Agronomic Practices to Increase the Yield and Quality of Common Bean (Phaseolus vulgaris L.): A Systematic Review
Abstract
1. Introduction
2. Methods
2.1. Literature Research
2.2. Inclusion and Exclusion Criteria
2.3. Screening
2.4. Bibliometric and Concept Network Analysis
3. Results
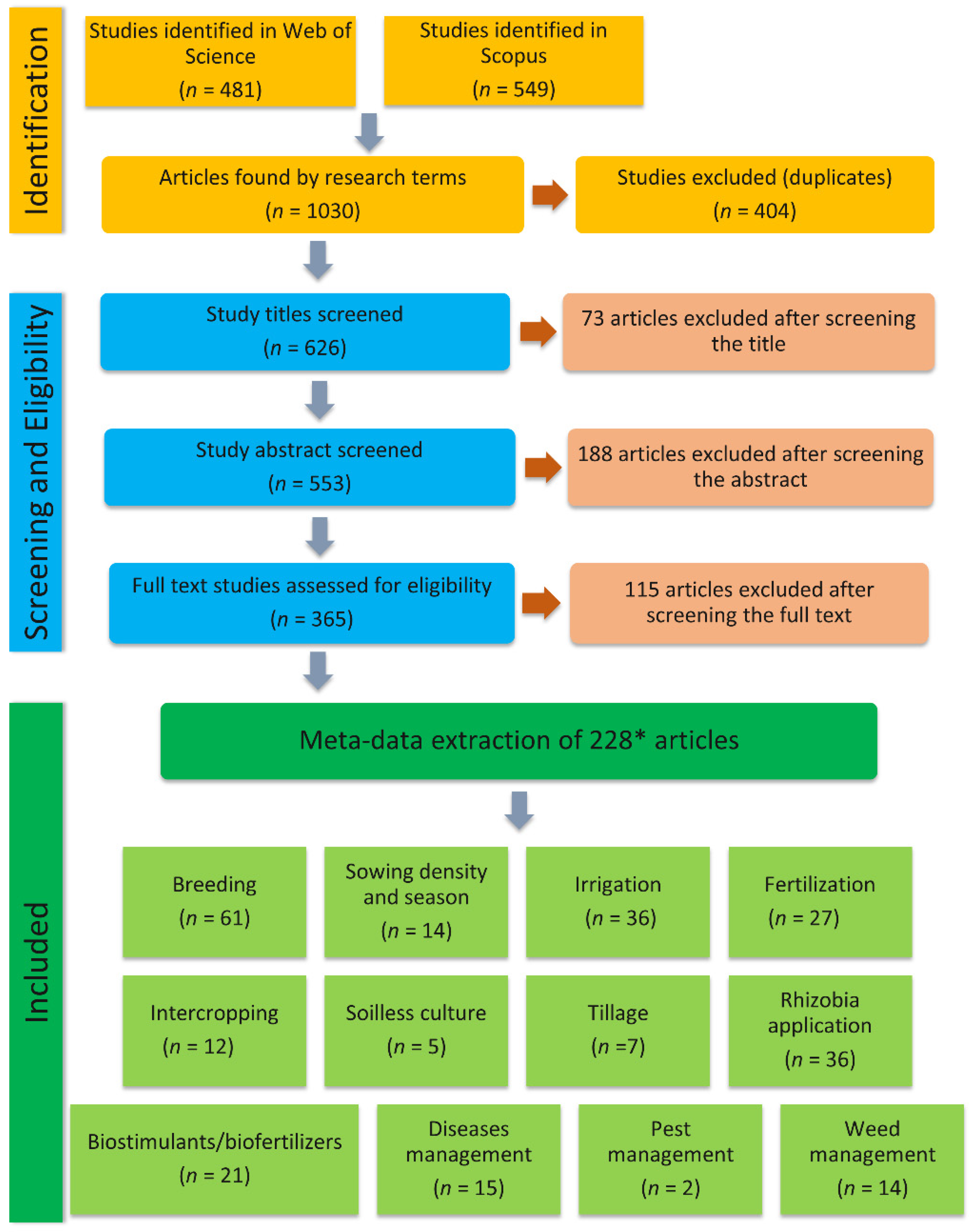
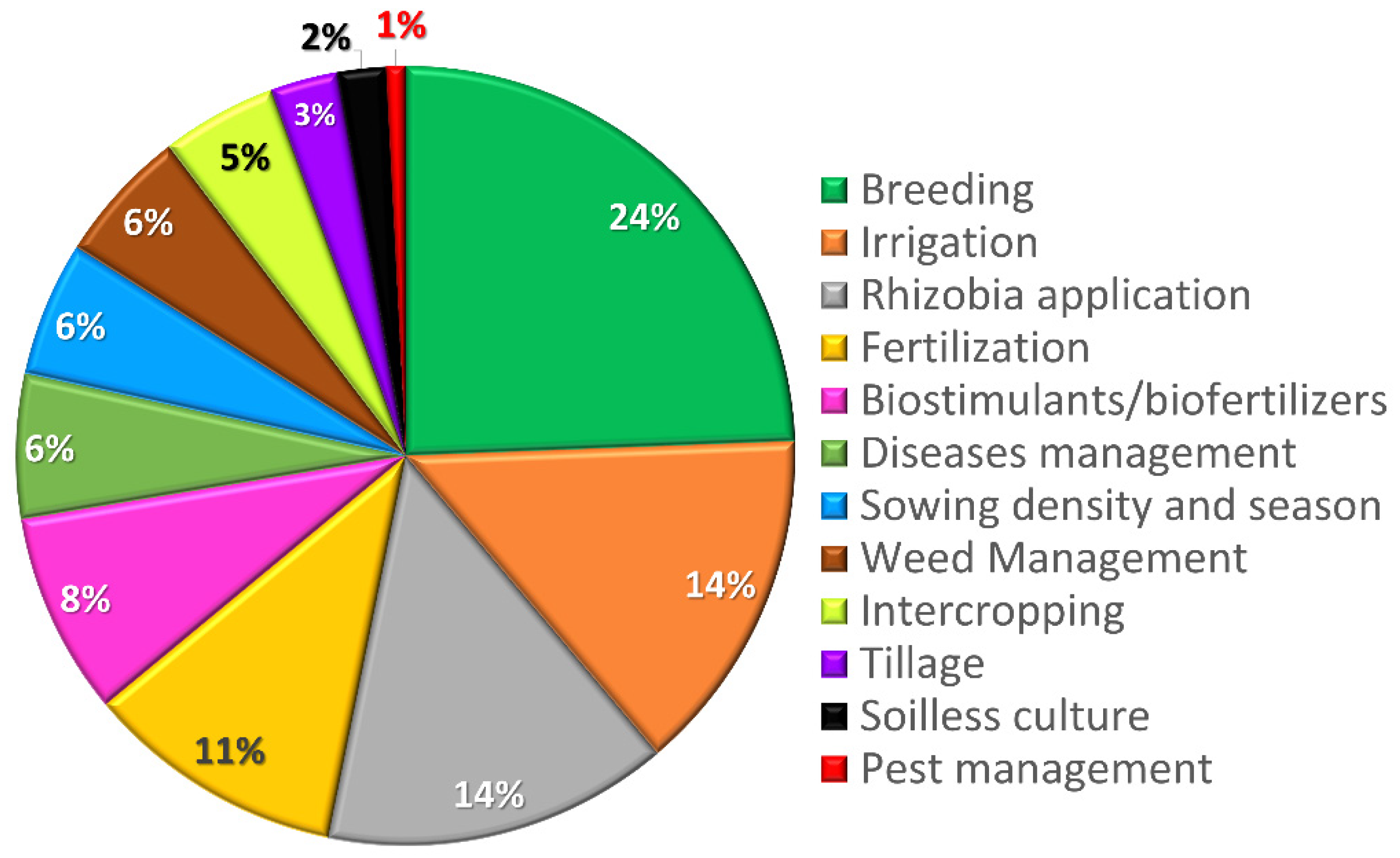
3.1. Screening Results
3.1.1. Breeding for Increased Yield and Quality
3.1.2. Sowing Density and Season
3.1.3. Irrigation
3.1.4. Fertilization
3.1.5. Intercropping
3.1.6. Soilless Culture
3.1.7. Tillage
3.1.8. Rhizobia Application
3.1.9. Biostimulant/Biofertilizer Application
3.1.10. Diseases Management
3.1.11. Pest Management
3.1.12. Weed Management
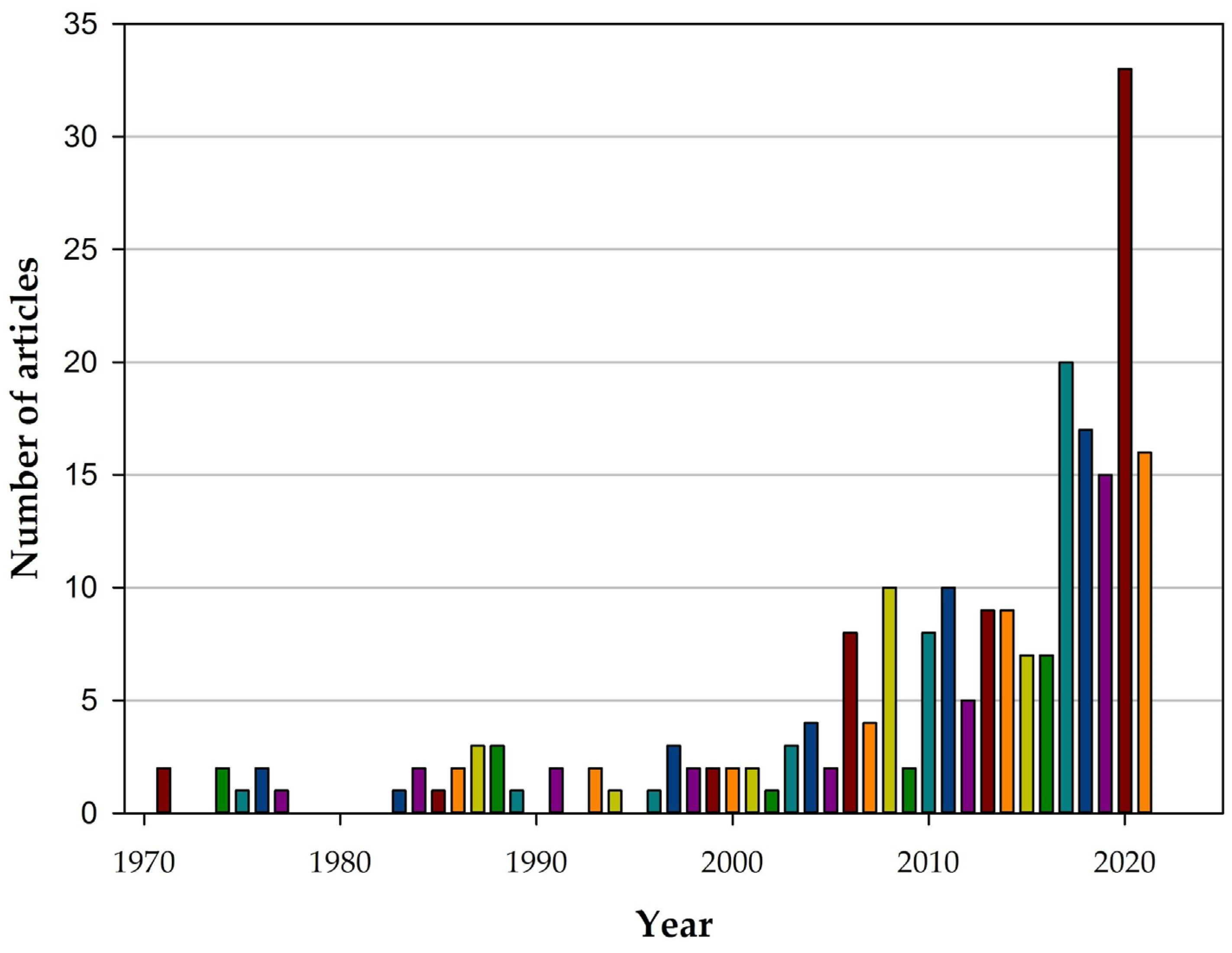
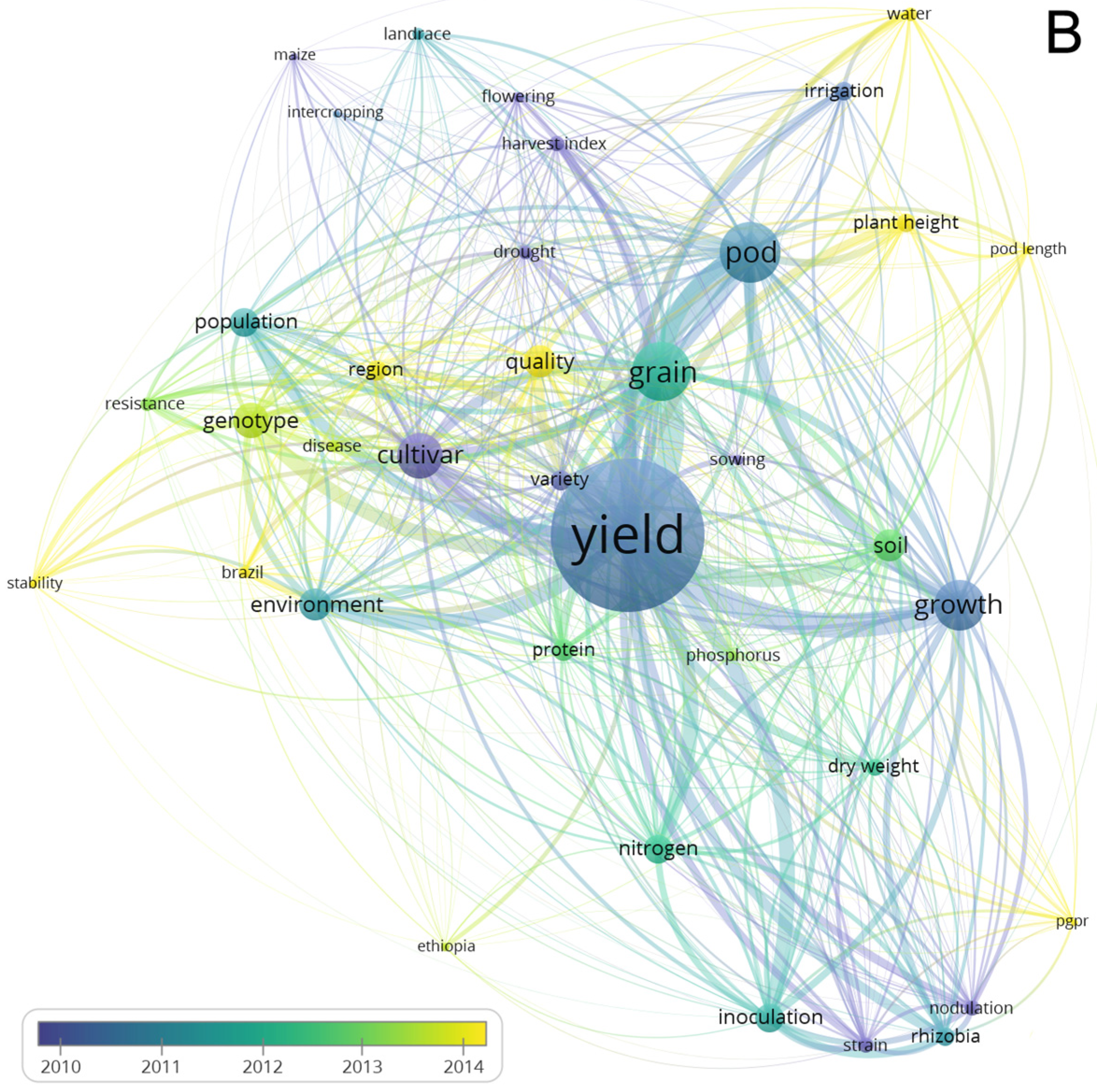
3.2. Evolution Articles over the Years
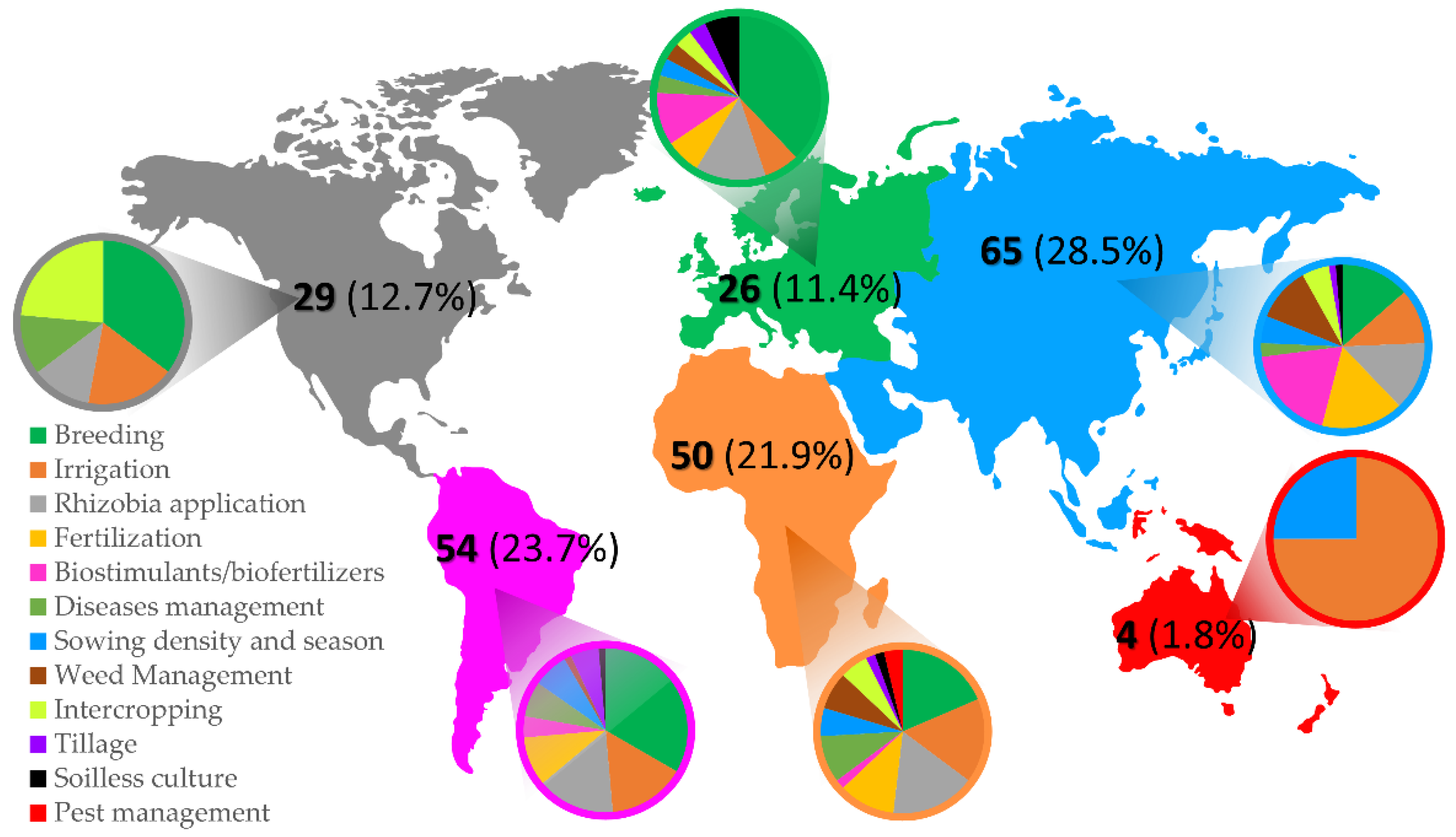
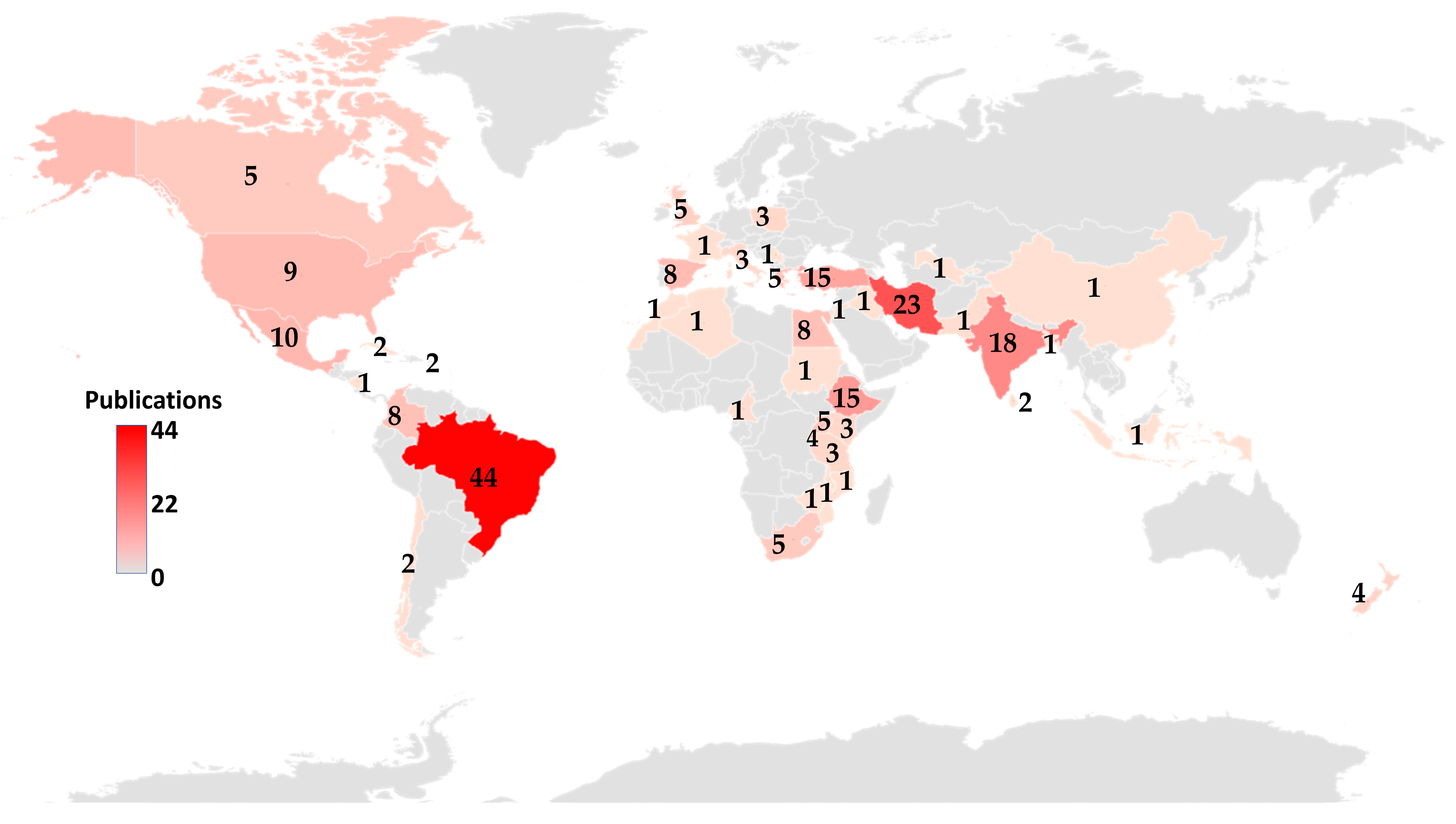
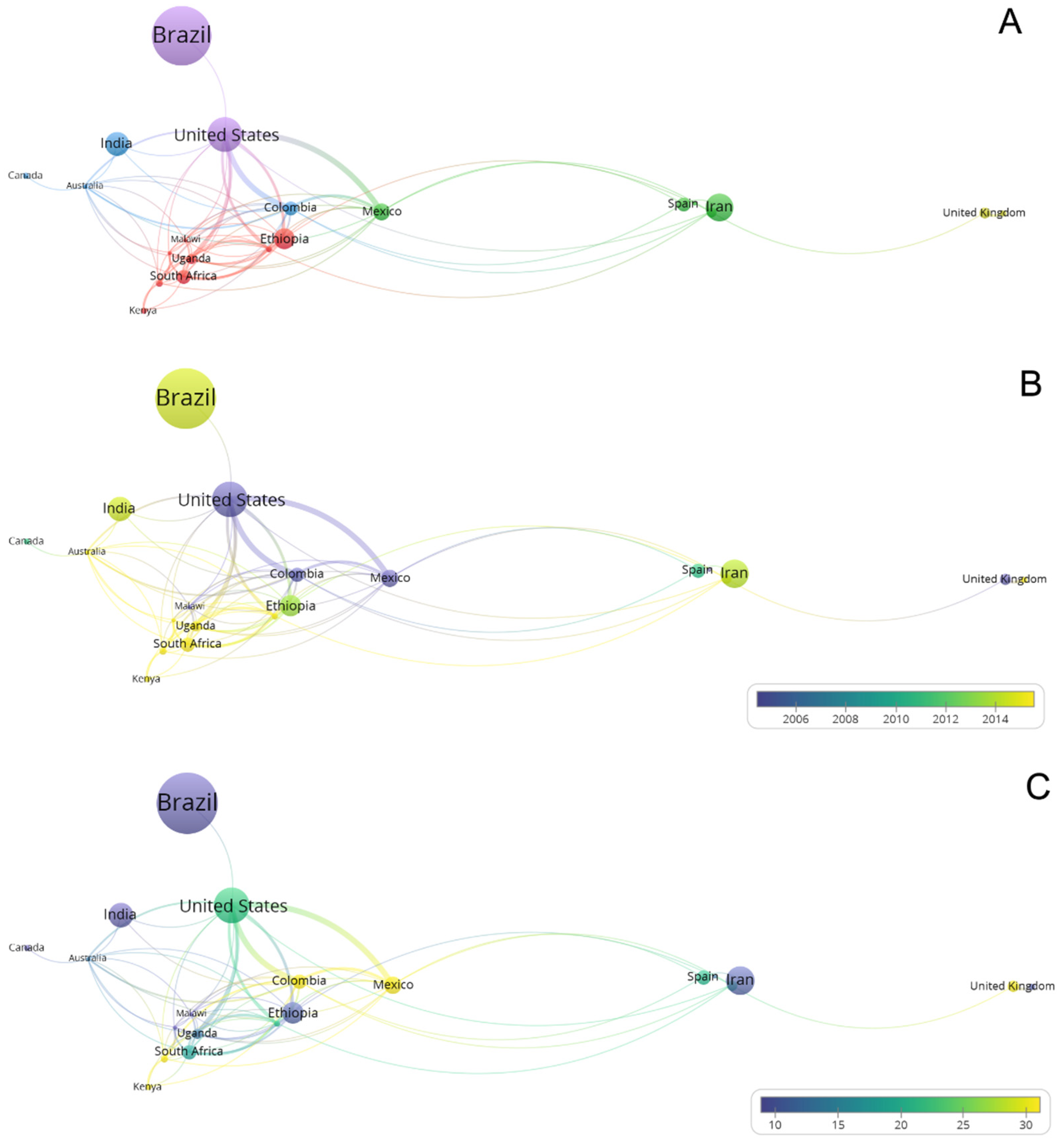
3.3. Geographical Distribution of Articles
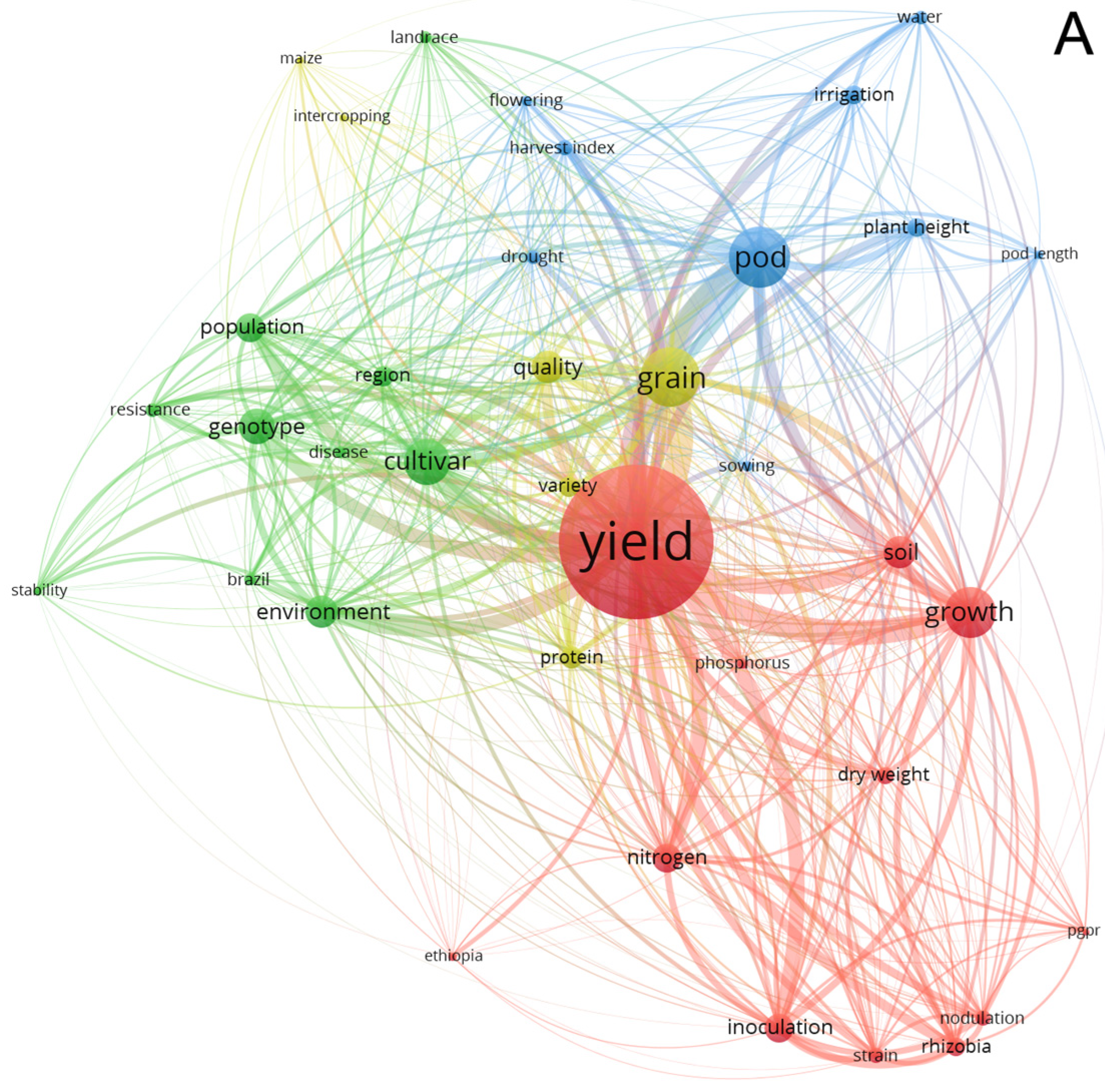
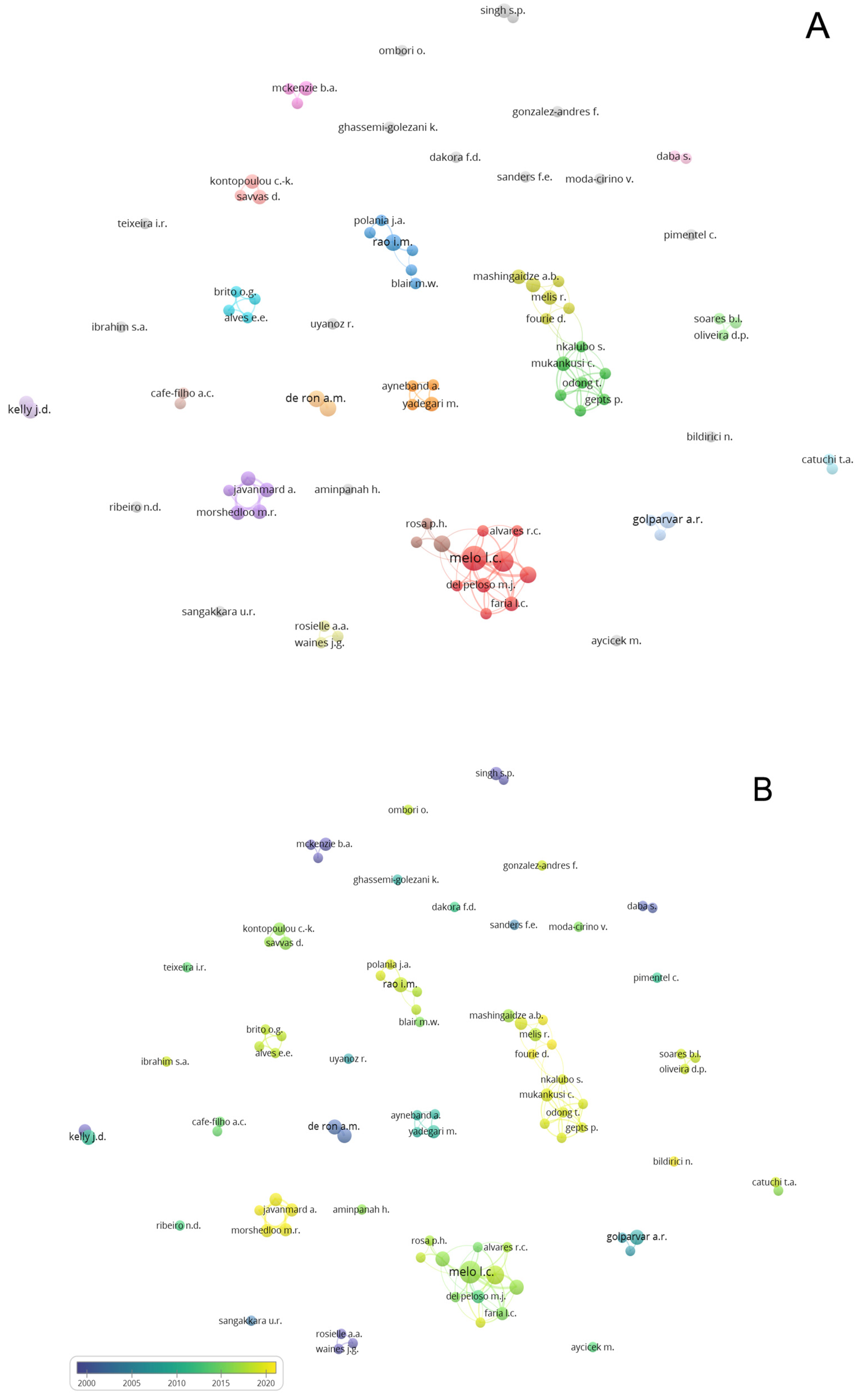
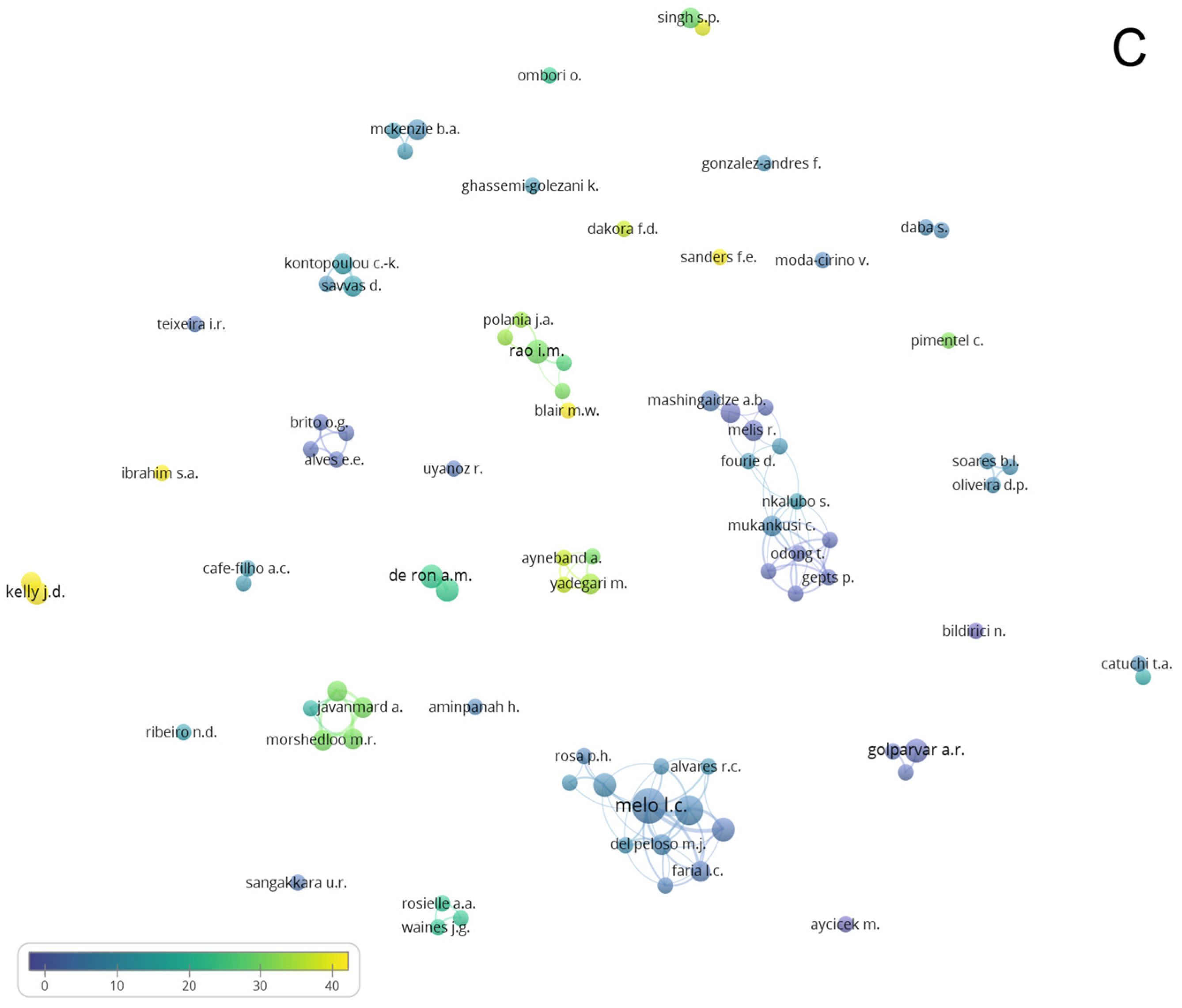
3.4. Network Analysis Subsection
3.4.1. Term Analysis
3.4.2. Authors and Countries Network Analysis
4. Discussion
4.1. Screening and Bibliometric Analysis
4.2. Breeding for Increased Yield and Quality
4.3. Sowing Density and Season
4.4. Irrigation
| Treatments | Yield Components | References | |||||
|---|---|---|---|---|---|---|---|
| Pod Yield | Seed Yield | Pod Nitrogen | Number Seeds/Pod | 100 Seed Weight | |||
| Irrigation regimes | rainfed | ND | ↓ | ND | ND | ND | [89] |
| ND | ↓ | ND | ND | ND | [90] | ||
| ND | ↓ | ↓ | ↓ | ↓ | [91] | ||
| ND | ↓ | ND | ND | ND | [92] | ||
| deficit irrigation | ND | ↓ | ND | ND | ↓ | [98] | |
| ND | ↓ | ND | ND | ↓ | [93] | ||
| ND | ↓ | ND | ND | ND | [94] | ||
| ND | ↓ | ↓ | ↓ | ↓ | [95] | ||
| ND | ↓ | ↓ | ↓ | ↓ | [82] | ||
| ND | ↓ | ND | ND | ND | [96] | ||
| ND | ↓ | ND | ND | ND | [97] | ||
| deficit evaporation | ND | ↓ | ↓ | ND | ND | [8] | |
| ND | ↓ | ↓ | ─ | ─ | [99] | ||
| ND | ─ | ─ | ↓ | ↓ | [10] | ||
| ↓ | ND | ND | ND | ND | [9] | ||
| deficit soil moisture | ↓ | ND | ↓ | ND | ND | [7] | |
| Irrigation impact on different growth stages | flowering | ND | ↓ | ↓ | ND | ND | [109] |
| flowering/ pod filling | ND | ↓/↓ | ↓/↓ | ─ | ─ | [111] | |
| ND | ↓/↓ | ↓/↓ | ↓/↓ | ↓/↓ | [112] | ||
| bud to pod filling | ND | ↓ | ↓ | ND | ND | [113] | |
| reproductive stage | ND | ↓ | ND | ND | ND | [110] | |
| vegetive/ reproductive | −/↓ | ND | −/↓ | −/↓ | ND | [115] | |
| ND | −/↓ | ND | ND | ND | [116] | ||
| vegetive/ flowering/reproductive | ND | ↓/↓/↓ | ↓/↓/↓ | ─/↓/↓ | ─/─/↓ | [114] | |
| Different irrigation intervals | 5, 7, 9 d | ND | ↓ (9d) | ↑ (7d) | ↓ (9d) | ↓ (9d) | [108] |
| 6, 12, 18 d | ND | ↓ (d > 6) | ↓ (d > 6) | ↓ (d > 6) | ↓ (d > 6) | [104] | |
| 4, 8, 12 d | ND | ↓ (12d) | ↓(12d) | ↓ (12d) | ─ | [107] | |
| 7, 14 d | ND | ↓ | ↓ | ↓ | ─ | [106] | |
4.5. Fertilization
4.6. Intercropping
4.7. Soilless Culture
4.8. Tillage
4.9. Rhizobia Application
| Bacterial Species | Strain | Yield Increase (%) | Protein Increase (%) | References |
|---|---|---|---|---|
| Rhizobium leguminosarum bv. phaseoli | LCS0306 | 26.56 | - | [16] |
| L-125 | 6.04–66.12 | - | [183] | |
| L-125, L-78 | 34.55–42.49 | - | [184] | |
| 6.35 | 20.32 | [185] | ||
| CO5 | no impact | - | [173] | |
| HB-429 or GT-9 | 30.56–33.59 | - | [186] | |
| Rhizobium leguminosarum | PhVyNOD3 | - | 9 | [18] |
| vicea | −10.90 (yield reduction) | 9.75 | [187] | |
| Rhizobium leguminosarum bv. phaseoli + Bacillus subtilis (OSU-142) + Bacillus megaterium (M-3) | OSU-142: B. subtilis M-3: B. megaterium | 6.18 | 23.13 | [185] |
| Rhizobium phaseoli | HAMBI3570 | 15.26–78.12 | - | [188] |
| 3644 and 3622 | 30.86–68.94 | [189] | ||
| - | 21.56 | - | [190] | |
| - | no impact | [176] | ||
| - | no impact | [175] | ||
| Rhizobium phaseoli + Pseudomonas fluorescens | Rb-133 + P-93 | 13.90–54.20 | - | [191,192] |
| Rhizobium etli | HAMBI3556 | 12.50–79.50 | - | [188] |
| Rhizobium phaseoli, Azotobacter vinelandii, Pseudomonas putida, Pantoea agglomerans, Pseudomonas koreensis, P. vancouverensis | - | 9.08 | 0.87 | [193] |
| Rhizobium tropici | CIAT 899 | no impact | - | [194] |
| 9.06 | - | [17] | ||
| 37.57–43.77 | - | [195] | ||
| SEMIA 4077, SEMIA 4080, and SEMIA 4088 | 11.05–16.62 | - | [182] | |
| SEMIA 4080 | 7.36–20.70 | - | [196] | |
| Rhizobium pisi Pseudomonas monteilii | R40982 | 41–59% (common bean genotype BAT-477) | - | [197] |
| Rhizobium sp. | CIAT isolates 384, 274, and 632 | 61.11–70.12 | - | [198] |
| B1 | 26.55 | - | [14] | |
| Rb-133 | 9.38–23.50 | 8.97–21.93 | [199] | |
| Rhizobium sp. | CIAT isolates 384, 274, and 632 | 19.94–70.18 (common bean intercropping with Sorghum bicolor) | [155] |
4.10. Biostimulant/Biofertilizer Application
4.11. Disease Management
4.12. Pest Management
4.13. Weed Management
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zandalinas, S.I.; Fritschi, F.B.; Mittler, R. Global Warming, Climate Change, and Environmental Pollution: Recipe for a Multifactorial Stress Combination Disaster. Trends Plant Sci. 2021, 26, 588–599. [Google Scholar] [CrossRef]
- Chojnacka, K.; Moustakas, K.; Witek-Krowiak, A. Bio-based fertilizers: A practical approach towards circular economy. Bioresour. Technol. 2020, 295, 122223. [Google Scholar] [CrossRef] [PubMed]
- van Dijk, M.; Morley, T.; Rau, M.L.; Saghai, Y. A meta-analysis of projected global food demand and population at risk of hunger for the period 2010–2050. Nat. Food 2021, 2, 494–501. [Google Scholar] [CrossRef]
- Young, M.D.; Ros, G.H.; de Vries, W. Impacts of agronomic measures on crop, soil, and environmental indicators: A review and synthesis of meta-analysis. Agric. Ecosyst. Environ. 2021, 319, 107551. [Google Scholar] [CrossRef]
- Morugán-Coronado, A.; Linares, C.; Gómez-López, M.D.; Faz, Á.; Zornoza, R. The impact of intercropping, tillage and fertilizer type on soil and crop yield in fruit orchards under Mediterranean conditions: A meta-analysis of field studies. Agric. Syst. 2020, 178, 102736. [Google Scholar] [CrossRef]
- Ghimire, R.; Norton, U.; Bista, P.; Obour, A.K.; Norton, J.B. Soil organic matter, greenhouse gases and net global warming potential of irrigated conventional, reduced-tillage and organic cropping systems. Nutr. Cycl. Agroecosyst. 2017, 107, 49–62. [Google Scholar] [CrossRef]
- El-Noemani, A.A.; Aboellil, A.A.A.; Dewedar, O.M. Influence of irrigation systems and water treatments on growth, yield, quality and water use efficiency of bean (Phaseolus vulgaris L.) plants. Int. J. ChemTech Res. 2015, 8, 248–258. [Google Scholar]
- da Conceição, C.G.; Robaina, A.D.; Peiter, M.X.; Parizi, A.R.C.; da Conceição, J.A.; Bruning, J. Economically optimal water depth and grain yield of common bean subjected to different irrigation depths. Rev. Bras. Eng. Agric. Ambient. 2018, 22, 482–487. [Google Scholar] [CrossRef]
- Abebe, A.; Tsige, A.; Work, M.; Enyew, A. Optimizing irrigation frequency and amount on yield and water productivity of snap bean (Phaseolus vulgaris L.) in NW Amhara, Ethiopia: A case study in Koga and Ribb irrigation scheme. Cogent Food Agric. 2020, 6, 1773690. [Google Scholar] [CrossRef]
- Campos, K.; Schwember, A.R.; Machado, D.; Ozores-Hampton, M.; Gil, P.M. Physiological and yield responses of green-shelled beans (Phaseolus vulgaris L.) grown under restricted irrigation. Agronomy 2021, 11, 562. [Google Scholar] [CrossRef]
- Alvares, R.C.; Silva, F.C.; Melo, L.C.; Melo, P.G.S.; Pereira, H.S. Estimation of genetic parameters and selection of high-yielding, upright common bean lines with slow seed-coat darkening. Genet. Mol. Res. 2016, 15, gmr15049081. [Google Scholar] [CrossRef]
- Rai, P.K.; Singh, M.; Anand, K.; Saurabh, S.; Kaur, T.; Kour, D.; Yadav, A.N.; Kumar, M. Role and Potential Applications of Plant Growth-Promoting Rhizobacteria for Sustainable Agriculture; Elsevier Inc.: Amsterdam, The Netherlands, 2020; ISBN 9780128205266. [Google Scholar]
- Niu, B.; Wang, W.; Yuan, Z.; Sederoff, R.R.; Sederoff, H.; Chiang, V.L.; Borriss, R. Microbial Interactions Within Multiple-Strain Biological Control Agents Impact Soil-Borne Plant Disease. Front. Microbiol. 2020, 11, 2452. [Google Scholar] [CrossRef]
- Negi, S.; Bharat, N.K.; Kumar, M. Effect of seed biopriming with indigenous pgpr, rhizobia and trichoderma sp. On growth, seed yield and incidence of diseases in french bean (Phaseolus vulgaris L.). Legum. Res. 2021, 44, 593–601. [Google Scholar] [CrossRef]
- Savvas, D.; Gruda, N. Application of soilless culture technologies in the modern greenhouse industry—A review. Eur. J. Hortic. Sci. 2018, 83, 280–293. [Google Scholar] [CrossRef]
- Pastor-Bueis, R.; Jiménez-Gómez, A.; Barquero, M.; Mateos, P.F.; González-Andrés, F. Yield response of common bean to co-inoculation with Rhizobium and Pseudomonas endophytes and microscopic evidence of different colonised spaces inside the nodule. Eur. J. Agron. 2021, 122, 126187. [Google Scholar] [CrossRef]
- Barros, R.L.N.; De Oliveira, L.B.; De Magalhães, W.B.; Pimentel, C. Growth and yield of common bean as affected by seed inoculation with rhizobium and nitrogen fertilization. Exp. Agric. 2018, 54, 16–30. [Google Scholar] [CrossRef]
- Massa, N.; Cesaro, P.; Todeschini, V.; Capraro, J.; Scarafoni, A.; Cantamessa, S.; Copetta, A.; Anastasia, F.; Gamalero, E.; Lingua, G.; et al. Selected autochthonous rhizobia, applied in combination with AM fungi, improve seed quality of common bean cultivated in reduced fertilization condition. Appl. Soil Ecol. 2020, 148, 103507. [Google Scholar] [CrossRef]
- Vasconcelos, M.W.; Grusak, M.A.; Pinto, E.; Gomes, A.; Ferreira, H.; Balázs, B.; Centofanti, T.; Ntatsi, G.; Savvas, D.; Karkanis; et al. The biology of legumes and their agronomic, economic, and social impact. In The Plant Family Fabaceae: Biology and Physiological Responses to Environmental Stresses; Hasanuzzaman, M., Araújo, S., Gill, S.S., Eds.; Springer Nature: Singapore, 2020; pp. 3–25. [Google Scholar] [CrossRef]
- Kocira, S.; Szparaga, A.; Findura, P.; Treder, K. Modification of yield and fiber fractions biosynthesis in phaseolus vulgaris by treatment with biostimulants containing amino acids and seaweed extract. Agronomy 2020, 10, 1338. [Google Scholar] [CrossRef]
- Guzmán-Maldonado, S.H.; Acosta-Gallegos, J.; Paredes-López, O. Protein and mineral content of a novel collection of wild and weedy common bean (Phaseolus vulgaris L.). J. Sci. Food Agric. 2000, 80, 1874–1881. [Google Scholar] [CrossRef]
- Celmeli, T.; Sari, H.; Canci, H.; Sari, D.; Adak, A.; Eker, T.; Toker, C. The nutritional content of common bean (Phaseolus vulgaris L.) landraces in comparison to modern varieties. Agronomy 2018, 8, 166. [Google Scholar] [CrossRef]
- Ntatsi, G.; Karkanis, A.; Tran, F.; Savvas, D.; Iannetta, P.P.M. Which agronomic practices increase the yield and quality of common bean (Phaseolus vulgaris L.)? A systematic review protocol. Agronomy 2020, 10, 1008. [Google Scholar] [CrossRef]
- Orduña-Malea, E.; Costas, R. Link-Based Approach to Study Scientific Software Usage: The Case of VOSviewer; Springer International Publishing: Cham, Switzerland, 2021; Volume 126, ISBN 0123456789. [Google Scholar]
- De Ron, A.M. Grain Legumes; Springer: Heidelberg, Germany, 2015; ISBN 9781493927975. [Google Scholar]
- Chávez-Servia, J.L.; Heredia-García, E.; Mayek-Pérez, N.; Aquino-Bolaños, E.N.; Hernández-Delgado, S.; Carrillo-Rodríguez, J.C.; Gill-Langarica, H.R.; Vera-Guzmán, A.M. Diversity of common Bean (Phaseolus vulgaris L.) landraces and the nutritional value of their grains. In Grain Legumes; InTech: Rijeka, Croatia, 2016. [Google Scholar] [CrossRef]
- Dawo, M.I.; Sanders, F.E.; Pilbeam, D.J. Yield, yield components and plant architecture in the F3 generation of common bean (Phaseolus vulgaris L.) derived from a cross between the determinate cultivar “Prelude” and an indeterminate landrace. Euphytica 2007, 156, 77–87. [Google Scholar] [CrossRef]
- Okii, D.; Mukankusi, C.; Sebuliba, S.; Tukamuhabwa, P.; Tusiime, G.; Talwana, H.; Odong, T.; Namayanja, A.; Paparu, P.; Nkalubo, S.; et al. Genetic variation, Heritability estimates and GXE effects on yield traits of Mesoamerican common bean (Phaseolus vulgaris L.) germplasm in Uganda. Plant Genet. Resour. Charact. Util. 2018, 16, 237–248. [Google Scholar] [CrossRef]
- López-Pedrouso, M.; Bernal, J.; Franco, D.; Zapata, C. Evaluating two-dimensional electrophoresis profiles of the protein phaseolin as markers of genetic differentiation and seed protein quality in common bean (Phaseolus vulgaris L.). J. Agric. Food Chem. 2014, 62, 7200–7208. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.P.; Teran, H.; Molina, A. Genetics of seed yield and its components in common beans (Phaseolus vulgaris L.) of andean origin. Orig. Plant Breed. 1991, 107, 254–257. [Google Scholar] [CrossRef]
- Corte, A.D.; Moda-Cirino, V.; Arias, C.A.A.; de Toledo, J.F.F.; Destro, D. Genetic analysis of seed morphological traits and its correlations with grain yield in common bean. Braz. Arch. Biol. Technol. 2010, 53, 27–34. [Google Scholar] [CrossRef][Green Version]
- Atuahene-Amankwa, G.; Michaels, T.E. Genetic variances, heritabilities and genetic correlations of grain yield, harvest index and yield components for common bean (Phaseolus vulgaris L.) in sole crop and in maize/bean intercrop. Can. J. Plant Sci. 1997, 77, 533–538. [Google Scholar] [CrossRef]
- Baldin, R.C.; Kavalco, S.A.F.; Woyann, L.G.; Junior, A.A.R.; Gobatto, D.R.; da Silva, G.R.; Beninand, G.; Finatto, T. Yield stability of common bean genotypes in the state of Santa Catarina, Brazil. Pesqui. Agropecu. Bras. 2021, 56, 1–9. [Google Scholar] [CrossRef]
- Trutmann, P.; Pyndji, M.M. Partial replacement of local common bean mixtures by high yielding angular leaf spot resistant varieties to conserve local genetic diversity while increasing yield. Ann. Appl. Biol. 1994, 125, 45–52. [Google Scholar] [CrossRef]
- Junior, C.F.D.S.; Correoso, C.C.; Copacheski, M.; Boff, P.; Boff, M.I.C. High dynamic dilutions and genetic variability to phytosanitary management and yield of beans (Phaseolus vulgaris L.). Aust. J. Crop Sci. 2021, 15, 821–826. [Google Scholar] [CrossRef]
- Bezaweletaw, K.; Belete, K.; Sripichitt, P. Genetic gain in grain yield potential and associated agronomic traits in haricot bean (Phaseolus vulgaris L.). Kasetsart J.-Nat. Sci. 2006, 40, 835–847. [Google Scholar]
- Fetahu, S.; Rusinovci, I.; Aliu, S.; Zeka, D.; Beluli, A. Genetic diversity of common bean landraces on seed imbibition and some quality traits. Acta Hortic. 2021, 1320, 71–78. [Google Scholar] [CrossRef]
- Kargiotidou, A.; Papathanasiou, F.; Baxevanos, D.; Vlachostergios, D.N.; Stefanou, S.; Papadopoulos, I. Yield and stability for agronomic and seed quality traits of common bean genotypes under Mediterranean conditions. Legum. Res. 2019, 42, 308–313. [Google Scholar] [CrossRef]
- Katuuramu, D.N.; Luyima, G.B.; Nkalubo, S.T.; Wiesinger, J.A.; Kelly, J.D.; Cichy, K.A. On-farm multi-location evaluation of genotype by environment interactions for seed yield and cooking time in common bean. Sci. Rep. 2020, 10, 3628. [Google Scholar] [CrossRef]
- Mekbib, F. Yield stability in common bean (Phaseolus vulgaris L.) genotypes. Euphytica 2003, 130, 147–153. [Google Scholar] [CrossRef]
- Poletine, J.P.; Gonçalves-Vidigal, M.C.; Coimbra, G.K.; Moiana, L.; Vidigal Filho, P.S.; Lacanallo, G.F.; de Lima Castro, S.A. Promising genotypes of common bean in relation to grain yield and resistance to anthracnose in Maringa and Umuarama counties. J. Food Agric. Environ. 2014, 12, 614–619. [Google Scholar]
- Salegua, V.; Melis, R.; Fourie, D.; Sibiya, J.; Musvosvi, C. Grain yield, stability and bacterial brown spot disease of dark red kidney dry bean (Phaseolus vulgaris L.) genotypes across six environments in South Africa. Aust. J. Crop Sci. 2020, 14, 1433–1442. [Google Scholar] [CrossRef]
- Silva, M.B.D.O.; De Carvalho, A.J.; Carneiro, J.E.D.S.; Aspiazú, I.; Alves, É.E.; David, A.M.S.D.S.; Brito, O.G.; Alves, P.F.S. Technological quality of grains of common beans selected genotypes from the carioca group. Semin. Agrar. 2016, 37, 1721–1732. [Google Scholar] [CrossRef]
- Lyngdoh, Y.A.; Thapa, U.; Shadap, A.; Singh, J.; Tomar, B.S. Studies on genetic variability and character association for yield and yield related traits in french bean Phaseolus vulgaris L.). Legum. Res. 2018, 41, 810–815. [Google Scholar] [CrossRef]
- Gómez, O.J.; Frankow-Lindberg, B.E. Yield formation in Nicaraguan landraces of common bean compared to bred cultivars. J. Agric. Sci. 2005, 143, 369–375. [Google Scholar] [CrossRef]
- Zilio, M.; Souza, C.A.; Medeiros Coelho, C.M.; Miquelluti, D.J.; Michels, A.F. Ciclo, arquitetura de parte aérea e produtividade de genótipos de feijão (Phaseolus vulgaris L.), no Estado de Santa Catarina. Acta Sci.-Agron. 2013, 35, 21–30. [Google Scholar] [CrossRef]
- Pereira, H.S.; Alvares, R.C.; De Cássia Silva, F.; De Faria, L.C.; Melo, L.C. Genetic, environmental and genotype x environment interaction effects on the common bean grain yield and commercial quality. Semin. Agrar. 2017, 38, 1241–1250. [Google Scholar] [CrossRef][Green Version]
- Nimbalkar, C.A.; Baviskar, A.P.; Navale, P.A. Genotype x environment interaction effect on seed yield of french bean (Phaseolus vulgaris L.). Indian J. Agric. Sci. 2004, 74, 366–369. [Google Scholar]
- Nicoletto, C.; Zanin, G.; Sambo, P.; Dalla Costa, L. Quality assessment of typical common bean genotypes cultivated in temperate climate conditions and different growth locations. Sci. Hortic. 2019, 256, 108599. [Google Scholar] [CrossRef]
- Hamblin, J. Effect of environment, seed size and competitive ability on yield and survival of Phaseolus vulgaris (L.) genotypes in mixtures. Euphytica 1975, 24, 435–445. [Google Scholar] [CrossRef]
- Escribano, M.R.; Santalla, M.; De Ron, A.M. Genetic diversity in pod and seed quality traits of common bean populations from northwestern Spain. Euphytica 1997, 93, 71–81. [Google Scholar] [CrossRef]
- Dias, P.A.S.; Almeida, D.V.; Melo, P.G.S.; Pereira, H.S.; Melo, L.C. Effectiveness of breeding selection for grain quality in common bean. Crop Sci. 2021, 61, 1127–1140. [Google Scholar] [CrossRef]
- Bulyaba, R.; Winham, D.M.; Lenssen, A.W.; Moore, K.J.; Kelly, J.D.; Brick, M.A.; Wright, E.M.; Ogg, J.B. Genotype by location effects on yield and seed nutrient composition of common bean. Agronomy 2020, 10, 347. [Google Scholar] [CrossRef]
- Meena, J.; Dhillon, T.S.; Meena, A.; Singh, K.K. Studies on performance of French bean (Phaseolus vulgaris L.) Genotypes for yield and quality traits under protected conditions. Plant Arch. 2017, 17, 615–619. [Google Scholar]
- Ribeiro, N.D.; Cargnelutti Filho, A.; Poersch, N.L.; Jost, E.; Rosa, S.S. Genetic progress in traits of yield, phenology and morphology of common bean. Crop. Breed. Appl. Biotechnol. 2008, 8, 232–238. [Google Scholar] [CrossRef]
- Zeffa, D.M.; Moda-Cirino, V.; Medeiros, I.A.; Freiria, G.H.; Neto, J.d.S.; Ivamoto-Suzuki, S.T.; Delfini, J.; Scapim, C.A.; Gonçalves, L.S.A. Genetic progress of seed yield and nitrogen use efficiency of brazilian carioca common bean cultivars using bayesian approaches. Front. Plant Sci. 2020, 11, 1168. [Google Scholar] [CrossRef] [PubMed]
- Nienhuis, J.; Singh, S.P. Genetics of Seed Yield and its Components in Common Bean (Phaseolus vulgaris L.) of Middle-American Origin: II. Genetic Variance, Heritability and expected response from selection. Plant Breed. 1988, 101, 155–163. [Google Scholar] [CrossRef]
- Kelly, J.D.; Kolkman, J.M.; Schneider, K. Breeding for yield in dry bean (Phaseolus vulgaris L.). Euphytica 1998, 102, 343–356. [Google Scholar] [CrossRef]
- Aycİcek, M. Yield and yield components of some common bean (Phaseolus vulgaris L.) local landraces and commercial varieties under Eastern Anatolia conditions. J. Food Agric. Environ. 2013, 11, 754–756. [Google Scholar]
- Aycİcek, M. Path analysis of yield and yield components of some common bean (Phaseolus vulgaris L.) genotypes under Bingol ecological conditions in Eastern Anatolia. J. Food Agric. Environ. 2013, 11, 750–753. [Google Scholar]
- Boylu, O.A.; Girgel, U. Molecular characterization and yield levels of local bean (Phaseolus vulgaris L.) genotypes growing in eastern mediterranean region. Fresenius Environ. Bull. 2021, 30, 4928–4934. [Google Scholar]
- Sozen, O.; Karadavuf, U.; Akcura, M. A study on the determination of the performance of some yield components in dry bean genotypes (Phaseolus vulgaris L.) in different environments. Fresenius Environ. Bull. 2018, 27, 8677–8686. [Google Scholar]
- Blair, M.W.; González, L.F.; Kimani, P.M.; Butare, L. Genetic diversity, inter-gene pool introgression and nutritional quality of common beans (Phaseolus vulgaris L.) from Central Africa. Theor. Appl. Genet. 2010, 121, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Ndlangamandla, V.V.; Ntuli, N.R. Variation on growth and yield traits among selected Phaseolus vulgaris landraces in kwazulu-natal, South Africa. Biodiversitas 2019, 20, 1597–1605. [Google Scholar] [CrossRef]
- Golparvar, A.R.; Ghasemi, P.A. Indirect selection for genetic improvement of seed yield and biological nitrogen fixation in Iranian common bean genotypes (Phaseolus vulgaris L.). Pakistan J. Biol. Sci. 2006, 9, 2097–2101. [Google Scholar]
- Golparvar, A.R. Multivariate analysis and determination of the best indirect selection criteria to genetic improvement the biological nitrogen fixation ability in common bean genotypes (Phaseolus vulgaris L.). Genetika 2008, 44, 279–284. [Google Scholar] [CrossRef]
- De Oliveira Silva, M.B.; De Carvalho, A.J.; De Souza David, A.M.S.; Aspiazú, I.; Alves, É.E.; De Souza Carneiro, J.E.; Brito, O.G.; De Souza, A.A. Technological quality of grain of common bean genotypes of the black commercial class. Rev. Bras. Ciencias Agrar. 2019, 14, 1–8. [Google Scholar] [CrossRef]
- de Steckling, S.M.; Ribeiro, N.D.; Arns, F.D.; Mezzomo, H.C.; Possobom, M.T.D.F. Genetic diversity and selection of common bean lines based on technological quality and biofortification. Genet. Mol. Res. 2017, 16. [Google Scholar] [CrossRef] [PubMed]
- Di Prado, P.R.C.; Faria, L.C.; Souza, T.L.P.O.; Melo, L.C.; Melo, P.G.S.; Pereira, H.S. Genetic control and selection of common bean parents and superior segregant populations based on high iron and zinc contents, seed yield and 100-seed weight. Genet. Mol. Res. 2019, 18, gmr18146. [Google Scholar] [CrossRef]
- Nkhata, W.; Shimelis, H.; Melis, R.; Chirwa, R.; Mzengeza, T.; Mathew, I.; Shayanowako, A. Combining ability analysis of common bean (Phaseolus vulgaris L.) genotypes for resistance to bean fly (Ophiomyia spp.), and grain yield and component traits. Euphytica 2021, 217, 93. [Google Scholar] [CrossRef]
- Ojwang, P.P.O.; Melis, R.; Githiri, M.S.; Songa, J.M. Genetic analysis for resistance to bean fly (Ophiomyia phaseoli) and seed yield among common bean genotypes in a semi-arid environment. Field Crop. Res. 2011, 120, 223–229. [Google Scholar] [CrossRef]
- Lioi, L.; Piergiovanni, A.R. Genetic diversity and seed quality of the Badda common Bean from sicily (Italy). Diversity 2013, 5, 843–855. [Google Scholar] [CrossRef]
- Wu, J.; Wang, L.; Fu, J.; Chen, J.; Wei, S.; Zhang, S.; Zhang, J.; Tang, Y.; Chen, M.; Zhu, J.; et al. Resequencing of 683 common bean genotypes identifies yield component trait associations across a north–south cline. Nat. Genet. 2020, 52, 118–125. [Google Scholar] [CrossRef]
- Plans, M.; Simó, J.; Casañas, F.; Sabaté, J. Near-infrared spectroscopy analysis of seed coats of common beans (Phaseolus vulgaris L.): A potential tool for breeding and quality evaluation. J. Agric. Food Chem. 2012, 60, 706–712. [Google Scholar] [CrossRef]
- Raatz, B.; Mukankusi, C.; Lobaton, J.D.; Male, A.; Chisale, V.; Amsalu, B.; Fourie, D.; Mukamuhirwa, F.; Muimui, K.; Mutari, B.; et al. Analyses of African common bean (Phaseolus vulgaris L.) germplasm using a SNP fingerprinting platform: Diversity, quality control and molecular breeding. Genet. Resour. Crop Evol. 2019, 66, 707–722. [Google Scholar] [CrossRef]
- Immer, A.M.; Fischer, R.A.; Joshue, K.S. Effects of plant density and thinning on high-yielding dry beans (Phaseolus vulgaris L.) in Mexico. Exp. Agric. 1977, 13, 325–335. [Google Scholar] [CrossRef]
- Chung, J.H.; Goulden, D.S. Yield components of haricot beans (Phaseolus vulgaris L.) grown at different plant densities. N Z. J. Agric. Res. 1971, 14, 227–234. [Google Scholar] [CrossRef]
- Musana, F.; Rucamumihigo, F.; Nirere, D.; Mbaraka, S. Growth and yield performance of common bean (Phaseolus vulgaris L.) as influenced by plant density at Nyagatare, East Rwanda. J. Ayurveda Integr. Med. 2020, 20, 16249–16261. [Google Scholar] [CrossRef]
- Babaeian, M. Effect of row spacing and sowing date on yield and yield components of common bean (Phaseolus vulgaris L.). Afr. J. Microbiol. Res. 2012, 6, 4340–4343. [Google Scholar] [CrossRef]
- Kouam, E.B.; Tsague-Zanfack, A.B. Effect of plant density on growth and yield attributes of common bean (Phaseolus vulgaris L.) genotypes. Not. Sci. Biol. 2020, 12, 399–408. [Google Scholar] [CrossRef]
- Baez-Gonzalez, A.D.; Fajardo-Diaz, R.; Padilla-Ramirez, J.S.; Osuna-Ceja, E.S.; Kiniry, J.R.; Meki, M.N.; Acosta-Díaz, E. Yield performance and response to high plant densities of dry bean (Phaseolus vulgaris L.) cultivars under semi-arid conditions. Agronomy 2020, 10, 1684. [Google Scholar] [CrossRef]
- Asemanrafat, M.; Honar, T. Effect of water stress and plant density on canopy temperature, yield components and protein concentration of red bean (Phaseolus vulgaris L. cv. akhtar). Int. J. Plant Prod. 2017, 11, 241–258. [Google Scholar] [CrossRef]
- Soratto, R.P.; Catuchi, T.A.; De Souza, E.D.F.C.; Garcia, J.L.N. Plant density and nitrogen fertilization on common bean nutrition and yield. Rev. Caatinga 2017, 30, 670–678. [Google Scholar] [CrossRef]
- Abubaker, S. Effect of Plant density on flowering date, yield and quality attribute of bush beans (Phaseolus vulgaris L.) under center pivot irrigation system. Am. J. Agric. Biol. Sci. 2008, 3, 666–668. [Google Scholar] [CrossRef]
- Balkaya, A.; Odabaş, M.S. The effects of sowing dates on seed yield and quality of red podded bean (Phaseolus vulgaris L.) cultivars. Acta Hortic. 2007, 729, 151–155. [Google Scholar] [CrossRef]
- Catuchi, T.A.; Guidorizzi, F.V.C.; Peres, V.J.S.; Dias, E.S.; Parmezan, G.C.; Galdi, L.V. Development and grain yield of common bean cultivars according to sowing season. Cientifica 2019, 47, 296–303. [Google Scholar] [CrossRef]
- Scarisbrick, D.H.; Carr, M.K.V.; Wilkes, J.M. The effect of sowing date and season on the development and yield of Navy beans (Phaseolus vulgaris L.) in south-east England. J. Agric. Sci. 1976, 86, 65–76. [Google Scholar] [CrossRef]
- Ishag, H.M.; Ayoub, A.T. Effect of sowing date and soil type on yield, yield components and survival of dry beans (Phaseolus vulgaris L.). J. Agric. Sci. 1974, 82, 343–347. [Google Scholar] [CrossRef]
- Smith, M.R.; Veneklaas, E.; Polania, J.; Rao, I.M.; Beebe, S.E.; Merchant, A. Field drought conditions impact yield but not nutritional quality of the seed in common bean (Phaseolus vulgaris L.). PLoS ONE 2019, 14, e0217099. [Google Scholar] [CrossRef] [PubMed]
- Polania, J.A.; Poschenrieder, C.; Beebe, S.; Rao, I.M. Effective use of water and increased dry matter partitioned to grain contribute to yield of common bean improved for drought resistance. Front. Plant Sci. 2016, 7, 660. [Google Scholar] [CrossRef] [PubMed]
- Assefa, T.; Wu, J.; Beebe, S.E.; Rao, I.M.; Marcomin, D.; Claude, R.J. Improving adaptation to drought stress in small red common bean: Phenotypic differences and predicted genotypic effects on grain yield, yield components and harvest index. Euphytica 2014, 203, 477–489. [Google Scholar] [CrossRef]
- Acosta-Gallegos, J.A.; Adams, M.W. Plant traits and yield stability of dry bean (Phaseolus vulgaris L.) cultivars under drought stress. J. Agric. Sci. 1991, 117, 213–219. [Google Scholar] [CrossRef]
- Nouralinezhad, A.; Babazadeh, H.; Amiri, E.; Sedghi, H. Effects of irrigation and nitrogen on yield and water productivity in common bean (Phaseolus vulgaris L.) and cowpea (Vigna unguiculata L.) in north of Iran. Appl. Ecol. Environ. Res. 2018, 16, 3113–3129. [Google Scholar] [CrossRef]
- Bourgault, M.; Madramootoo, C.A.; Webber, H.A.; Dutilleul, P.; Stulina, G.; Horst, M.G.; Smith, D.L. Legume production and irrigation strategies in the aral sea basin: Yield, yield components, water relations and crop development of common bean (Phaseolus vulgaris L.) and mungbean (Vigna radiata (L.) Wilczek). J. Agron. Crop Sci. 2013, 199, 241–252. [Google Scholar] [CrossRef]
- Rai, A.; Sharma, V.; Heitholt, J. Dry bean [Phaseolus vulgaris L.] growth and yield response to variable irrigation in the arid to semi-arid climate. Sustainability 2020, 12, 3851. [Google Scholar] [CrossRef]
- Parmar, S.K.; Patel, R.A.; Patel, H.K. Role of irrigation and nitrogen levels on yield, nutrient content, uptake and economics of French bean (Phaseolus vulgaris L.) under middle Gujarat condition. J. Pure Appl. Microbiol. 2016, 10, 657–662. [Google Scholar]
- Rosales-Serna, R.; Kohashi-Shibata, J.; Acosta-Gallegos, J.A.; Trejo-López, C.; Ortiz-Cereceres, J.; Kelly, J.D. Biomass distribution, maturity acceleration and yield in drought-stressed common bean cultivars. Field Crop. Res. 2004, 85, 203–211. [Google Scholar] [CrossRef]
- do Silva, A.N.; Ramos, M.L.G.; Ribeiro, W.Q.; de Alencar, E.R.; da Silva, P.C.; de Lima, C.A.; Vinson, C.C.; Silva, M.A.V. Water stress alters physical and chemical quality in grains of common bean, triticale and wheat. Agric. Water Manag. 2020, 231, 106023. [Google Scholar] [CrossRef]
- Ghassemi-Golezani, K.; Mardfar, R.A. Effects of limited irrigation on growth and grain yield of common bean. J. Plant Sci. 2008, 3, 230–235. [Google Scholar] [CrossRef]
- Dapaah, H.K.; McKenzie, B.A.; Hill, G.D. Influence of sowing date and irrigation on the growth and yield of pinto beans (Phaseolus vulgaris L.) in a sub-humid temperate environment. J. Agric. Sci. 2000, 134, 33–43. [Google Scholar] [CrossRef]
- Dapaah, H.K.; McKenzie, B.A.; Hill, G.D. Effects of irrigation and sowing date on phenology and yield of pinto beans (Phaseolus vulgaris L.) in Canterbury, New Zealand. N. Z. J. Crop Hortic. Sci. 1999, 27, 297–305. [Google Scholar] [CrossRef]
- Love, B.G.; Askin, D.C.; McKenzie, B.A. Effect of shelter, irrigation, and plant population on yield and yield components of navy beans (Phaseolus vulgaris L.). N. Z. J. Exp. Agric. 1988, 16, 231–237. [Google Scholar] [CrossRef]
- Efetha, A.; Harms, T.; Bandara, M. Irrigation management practices for maximizing seed yield and water use efficiency of Othello dry bean (Phaseolus vulgaris L.) in southern Alberta, Canada. Irrig. Sci. 2010, 29, 103–113. [Google Scholar] [CrossRef]
- Ibrahim, S.; Desoky, E.; Elrys, A. Influencing of water stress and micronutrients on physio-chemical attributes, yield and anatomical features of common bean plants (Phaseolus vulgaris L.). Egypt. J. Agron. 2017, 39, 251–264. [Google Scholar] [CrossRef]
- Kundu, M.; Chakraborty, P.K.; Mukherjee, A.; Sarkar, S. Influence of irrigation frequencies and phosphate fertilization on actual evapotranspiration rate, yield and water use pattern of rajmash (Phaseolus vulgaris L.). Agric. Water Manag. 2008, 95, 383–390. [Google Scholar] [CrossRef]
- Lizana, C.; Wentworth, M.; Martinez, J.P.; Villegas, D.; Meneses, R.; Murchie, E.H.; Pastenes, C.; Lercari, B.; Vernieri, P.; Horton, P.; et al. Differential adaptation of two varieties of common bean to abiotic stress I. Effects of drought on yield and photosynthesis. J. Exp. Bot. 2006, 57, 685–697. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, S.M.; Shahrokhnia, M.A. The effect of irrigation interval on yield, yield components and water productivity of common bean (Phaseolus vulgaris L.) cultivars in a semi-arid area. Ann. Biol. 2020, 36, 56–61. [Google Scholar]
- Okasha, E.M.; El-Metwally, I.M.; Taha, N.M.; Darwesh, R.K. Impact of drip and gated pipe irrigation systems, irrigation intervals on yield, productivity of irrigation water and quality of two common bean (Phaseolus vulgaris L.) cultivars in heavy clay soil. Egypt. J. Chem. 2020, 63, 5103–5116. [Google Scholar] [CrossRef]
- Guida Dos Santos, M.; Ribeiro, R.V.; Ferraz De Oliveira, R.; Pimentel, C. Gas exchange and yield response to foliar phosphorus application in Phaseolus vulgaris L. under drought. Braz. J. Plant Physiol. 2004, 16, 171–179. [Google Scholar] [CrossRef]
- González De Mejía, E.; Martínez-Resendiz, V.; Castaño-Tostado, E.; Loarca-Piña, G. Effect of drought on polyamine metabolism, yield, protein content and in vitro protein digestibility in tepary (Phaseolus acutifolius) and common (Phaseolus vulgaris) bean seeds. J. Sci. Food Agric. 2003, 83, 1022–1030. [Google Scholar] [CrossRef]
- Boutraa, T.; Sanders, F.E. Influence of water stress on grain yield and vegetative growth of two cultivars of bean (Phaseolus vulgaris L.). J. Agron. Crop Sci. 2001, 187, 251–257. [Google Scholar] [CrossRef]
- Mathobo, R.; Marais, D.; Steyn, J.M. The effect of drought stress on yield, leaf gaseous exchange and chlorophyll fluorescence of dry beans (Phaseolus vulgaris L.). Agric. Water Manag. 2017, 180, 118–125. [Google Scholar] [CrossRef]
- Mouhouche, B.; Ruget, F.; Delécolle, R. Effects of water stress applied at different phenological phases on yield components of dwarf bean (Phaseolus vulgaris L.). Agronomie 1998, 18, 197–205. [Google Scholar] [CrossRef]
- Acosta Gallegos, J.A.; Kohashi Shibata, J. Effect of water stress on growth and yield of indeterminate dry—bean (Phaseolus vulgaris L.). Field Crop. Res. 1989, 20, 81–93. [Google Scholar] [CrossRef]
- Simsek, M.; Comlekcioglu, N.; Ozturk, I. The effects of the regulated deficit irrigation on yield and some yield components of common bean (Phaseolus vulgaris L.) under semi-arid conditions. Afr. J. Biotechnol. 2011, 10, 4057–4064. [Google Scholar] [CrossRef]
- Peña-Cabriales, J.J.; Castellanos, J.Z. Effects of water stress on N2 fixation and grain yield of Phaseolus vulgaris L. Plant Soil 1993, 152, 151–155. [Google Scholar] [CrossRef]
- Desoky, E.S.M.; Ibrahim, S.A.; Merwad, A.R.M. Mitigation of salinity stress effects on growth, physio-chemical parameters and yield of snapbean (Phaseolus vulgaris l.) by exogenous application of glycine betaine. Int. Lett. Nat. Sci. 2019, 76, 60–71. [Google Scholar] [CrossRef]
- Kontopoulou, C.K.; Bilalis, D.; Pappa, V.A.; Rees, R.M.; Savvas, D. Effects of organic farming practices and salinity on yield and greenhouse gas emissions from a common bean crop. Sci. Hortic. 2015, 183, 48–57. [Google Scholar] [CrossRef]
- Rady, M.M. Effect of 24-epibrassinolide on growth, yield, antioxidant system and cadmium content of bean (Phaseolus vulgaris L.) plants under salinity and cadmium stress. Sci. Hortic. 2011, 129, 232–237. [Google Scholar] [CrossRef]
- Santosa, M.; Maghfoer, M.D.; Tarno, H. The influence of organic and inorganic fertilizers on the growth and yield of green bean, Phaseolus vulgaris L. Grown in dry and rainy season. Agrivita 2017, 39, 296–302. [Google Scholar] [CrossRef]
- Uyanoz, R. The Effects of different bio-organic, chemical fertilizers and their combination on yield, macro and micro nutrition content of dry bean (Phaseolus vulgaris L.). Int. J. Agric. Res. 2007, 2, 115–125. [Google Scholar] [CrossRef]
- Karungi, J.; Ekbom, B.; Kyamanywa, S. Effects of organic versus conventional fertilizers on insect pests, natural enemies and yield of Phaseolus vulgaris L. Agric. Ecosyst. Environ. 2006, 115, 51–55. [Google Scholar] [CrossRef]
- Magalhaes, A.C.; Montojos, J.C.; Miyasaka, S. Effect of dry organic matter on growth and yield of beans (Phaseolus vulgaris L.). Exp. Agric. 1971, 7, 137–143. [Google Scholar] [CrossRef]
- Prabhakar, M.; Hebbar, S.S.; Nair, A.K. Growth and yield of French bean (Phaseolus vulgaris L.) under organic farming. J. Appl. Hortic. 2011, 13, 71–73. [Google Scholar] [CrossRef]
- Fernández-Luqueño, F.; Reyes-Varela, V.; Martínez-Suárez, C.; Salomón-Hernández, G.; Yáñez-Meneses, J.; Ceballos-Ramírez, J.M.; Dendooven, L. Effect of different nitrogen sources on plant characteristics and yield of common bean (Phaseolus vulgaris L.). Bioresour. Technol. 2010, 101, 396–403. [Google Scholar] [CrossRef]
- Etminani, A.; Mohammadi, K.; Saberali, S.F. Effect of organic and inorganic amendments on growth indices and seed yield of red kidney bean (Phaseolus vulgaris L.) in competition with Amaranthus retroflexus. J. Plant Nutr. 2021, 44, 421–437. [Google Scholar] [CrossRef]
- Kawaka, F.; Dida, M.; Opala, P.; Ombori, O.; Maingi, J.; Amoding, A.; Muoma, J. Effect of nitrogen sources on the yield of common bean (Phaseolus vulgaris L.) in western Kenya. J. Plant Nutr. 2018, 41, 1652–1661. [Google Scholar] [CrossRef]
- Kumar, R.; Deka, B.C.; Kumawat, N.; Thirugnanavel, A. Effect of integrated nutrition on productivity, profitability and quality of French bean (Phaseolus vulgaris L.). Indian J. Agric. Sci. 2020, 90, 431–435. [Google Scholar]
- Sharma, A.; Sharma, R.P.; Katoch, V.; Sharma, G.D. Influence of vermicompost and split applied nitrogen on growth, yield, nutrient uptake and soil fertility in pole type french bean (Phaseolus vulgaris L.) in an acid alfisol. Legum. Res. 2018, 41, 126–131. [Google Scholar] [CrossRef]
- Singh, B.; Pathak, K.; Verma, A.; Verma, V.; Deka, B. Effects of vermicompost, fertilizer and mulch on plant growth, nodulation and pod yield of French bean (Phaseolus vulgaris L.). Veg. Crop. Res. Bull. 2011, 74, 153–165. [Google Scholar] [CrossRef]
- Zafar, M.; Abbasi, M.K.; Rahim, N.; Khaliq, A.; Shaheen, A.; Jamil, M.; Shahid, M. Influence of integrated phosphorus supply and plant growth promoting rhizobacteria on growth, nodulation, yield and nutrient uptake in Phaseolus vulgaris. Afr. J. Biotechnol. 2011, 10, 16793–16807. [Google Scholar] [CrossRef]
- Rurangwa, E.; Vanlauwe, B.; Giller, K.E. Benefits of inoculation, P fertilizer and manure on yields of common bean and soybean also increase yield of subsequent maize. Agric. Ecosyst. Environ. 2018, 261, 219–229. [Google Scholar] [CrossRef]
- Saikia, J.; Saikia, L.; Phookan, D.B.; Nath, D.J. Effect of biofertilizer consortium on yield, quality and soil health of french bean (Phaseolus vulgaris L.). Legum. Res. 2018, 41, 755–758. [Google Scholar] [CrossRef]
- da Silva, H.C.; de Lima, L.C.; de Camargo, R.; Lana, R.M.Q.; Lemes, E.M.; Cardoso, A.F. Effects of organomineral fertilizers formulated with biosolids and filter cake on common bean yield crop (Phaseolus vulgaris L.). Aust. J. Crop Sci. 2019, 13, 1566–1571. [Google Scholar] [CrossRef]
- Musse, Z.A.; Yoseph Samago, T.; Bisher, H.M. Effect of liquid bio-slurry and nitrogen rates on soil physico-chemical properties and quality of green bean (Phaseolus vulgaris L.) at Hawassa Southern Ethiopia. J. Plant Interact. 2020, 15, 207–212. [Google Scholar] [CrossRef]
- D’Amico-Damião, V.; Nunes, H.D.; Couto, P.A.; Lemos, L.B. Straw type and nitrogen fertilization influence winter common bean yield and quality. Int. J. Plant Prod. 2020, 14, 703–712. [Google Scholar] [CrossRef]
- Patel, A.G.; Patel, B.S.; Patel, P.H. Effect of irrigation levels based on IW: CPE ratios and time of nitrogen application on yield and monetary return of frenchbean (Phaseolus vulgaris L.). Legum. Res. 2010, 33, 42–45. [Google Scholar]
- Garcia, P.L.; Sermarini, R.A.; Trivelin, P.C.O. Nitrogen fertilization management with blends of controlled-release and conventional urea affects common bean growth and yield during mild winters in Brazil. Agronomy 2020, 10, 1935. [Google Scholar] [CrossRef]
- Suárez, J.C.; Polanía, J.A.; Anzola, J.A.; Contreras, A.T.; Méndez, D.L.; Vanegas, J.I.; Noriega, J.E.; Rodríguez, L.; Urban, M.O.; Beebe, S.; et al. Influence of nitrogen supply on gas exchange, chlorophyll fluorescence and grain yield of breeding lines of common bean evaluated in the Amazon region of Colombia. Acta Physiol. Plant. 2021, 43, 66. [Google Scholar] [CrossRef]
- Nascente, A.S.; Carvalho, M.d.C.S.; Melo, L.C.; Rosa, P.H. Nitrogen management effects on soil mineral nitrogen, plant nutrition and yield of super early cycle common bean genotype. Acta Sci.-Agron. 2017, 39, 369–378. [Google Scholar] [CrossRef][Green Version]
- Ovacikli, E.; Tolay, I. Morpho-agronomic and cooking quality of common bean (Phaseolus vulgaris L.) grown under different nitrogen sources and nitrogen levels. Appl. Ecol. Environ. Res. 2020, 18, 8343–8354. [Google Scholar] [CrossRef]
- Abebe, G. Effect of np fertilizer and moisture conservation on the yield and yield components of haricot bean (Phaseolus vulgaris L.) in the semi arid zones of the central rift valley in Ethiopia. Adv. Environ. Biol. 2009, 3, 302–307. [Google Scholar]
- da Carvalho, M.C.S.; Nascente, A.S.; Ferreira, G.B.; Mutadiua, C.A.P.; Denardin, J.E. Phosphorus and potassium fertilization increase common bean grain yield in Mozambique Maria. Rev. Bras. Eng. Agrícola Ambient. 2018, 22, 308–314. [Google Scholar] [CrossRef]
- Bildirici, N.; Oral, E. The effect of phosphorus and zinc doses on yield and yield components of beans (Phaseolus vulgaris L.) in Van-Gevaş, Turkey. Appl. Ecol. Environ. Res. 2020, 18, 2539–2553. [Google Scholar] [CrossRef]
- Da Silva, P.M.; Tsai, S.M.; Bonetti, R. Response to inoculation and N fertilization for increased yield and biological nitrogen fixation of common bean (Phaseolus vulgaris L.). Plant Soil 1993, 152, 123–130. [Google Scholar] [CrossRef]
- Aslani, M.; Souri, M.K. Growth and quality of green bean (Phaseolus vulgaris L.) under foliar application of organic-chelate fertilizers. Open Agric. 2018, 3, 146–154. [Google Scholar] [CrossRef]
- Khaber, M.S.; Aboohanah, M.A. Response of bean plant Phaseolus vulgaris L. to spray with hornwort extract and nano potassium on growth and yield parameters. Plant Arch. 2020, 20, 946–950. [Google Scholar]
- Balcha, A. Genetic variation for grain yield of common bean (Phaseolus vulgaris L.) in sole and maize/bean intercropping systems. Asian J. Crop Sci. 2014, 6, 158–164. [Google Scholar] [CrossRef]
- Zimmermann, M.J.O.; Rosielle, A.A.; Waines, J.G.; Foster, K.W. A heritability and correlation study of grain yield, yield components, and harvest index of common bean in sole crop and intercrop. Field Crop. Res. 1984, 9, 109–118. [Google Scholar] [CrossRef]
- Amani Machiani, M.; Rezaei-Chiyaneh, E.; Javanmard, A.; Maggi, F.; Morshedloo, M.R. Evaluation of common bean (Phaseolus vulgaris L.) seed yield and quali-quantitative production of the essential oils from fennel (Foeniculum vulgare Mill.) and dragonhead (Dracocephalum moldavica L.) in intercropping system under humic acid application. J. Clean. Prod. 2019, 235, 112–122. [Google Scholar] [CrossRef]
- Zimmermann, M.J.O.; Rosielle, A.A.; Waines, J.G. Heritabilities of grain yield of common bean in sole crop and in intercrop with maize 1. Crop Sci. 1984, 24, 641–644. [Google Scholar] [CrossRef]
- Santalla, M.; Fueyo, M.A.; Paula Rodino, A.; Montero, I.; de Ron, A.M. Breeding for culinary and nutritional quality of common bean (Phaseolus vulgaris L.) in intercropping systems with maize (Zea mays L.). Biotechnol. Agron. Soc. Environ. 1999, 3, 225–229. [Google Scholar]
- Dua, V.K.; Kumar, S.; Jatav, M.K. Effect of nitrogen application to intercrops on yield, competition, nutrient use efficiency and economics in potato (Solanum tuberosum L.) + french bean (Phaseolus vulgaris L.) system in north-western Hills of India. Legum. Res. 2017, 40, 698–703. [Google Scholar] [CrossRef]
- Barbeau, C.D.; Wilton, M.J.; Oelbermann, M.; Karagatzides, J.D.; Tsuji, L.J.S. Local food production in a subarctic Indigenous community: The use of willow (Salix spp.) windbreaks to increase the yield of intercropped potatoes (Solanum tuberosum) and bush beans (Phaseolus vulgaris L.). Int. J. Agric. Sustain. 2018, 16, 29–39. [Google Scholar] [CrossRef]
- Daba, S.; Haile, M. Effects of rhizobial inoculant and nitrogen fertilizer on yield and nodulation of common bean under intercropped conditions. J. Plant Nutr. 2002, 25, 1443–1455. [Google Scholar] [CrossRef]
- Abd El-Gai, M.A.; Al-Dokeshy, M.H.; Nasse, D.M.T. Effects of intercropping system of tomato and common bean on growth, yield components and land equivalent ratio in new valley governorate. Asian J. Crop Sci. 2014, 6, 254–261. [Google Scholar] [CrossRef][Green Version]
- Sadeghi, H.; Sasanfar, I. Effect of different safflower (Carthamus tinctorius L.)-bean (Phaseolus vulgaris L.) intercropping patterns on growth and yield under weedy and weed-free conditions. Arch. Agron. Soil Sci. 2013, 59, 765–777. [Google Scholar] [CrossRef]
- Raey, Y.; Ghassemi-Golezani, K. Yield-density relationship for potato (Solarium tuberosum) and common bean (Phaseolus vulgaris L.) in intercropping. N. Z. J. Crop Hortic. Sci. 2009, 37, 141–147. [Google Scholar] [CrossRef]
- Kontopoulou, C.K.; Giagkou, S.; Stathi, E.; Savvas, D.; Iannetta, P.P.M. Responses of hydroponically grown common bean fed with nitrogen-free nutrient solution to root inoculation with N2-fixing bacteria. HortScience 2015, 50, 597–602. [Google Scholar] [CrossRef]
- Kontopoulou, C.-K.; Liasis, E.; Iannetta, P.P.; Tampakaki, A.; Savvas, D. Impact of rhizobial inoculation and reduced N supply on biomass production and biological N2 fixation in common bean grown hydroponically. J. Sci. Food Agric. 2017, 97, 4353–4361. [Google Scholar] [CrossRef]
- Bildirici, N. The effects of copper-zinc interactions on yield and yield components in soilless grown beans (Phaseolus vulgaris L.). Appl. Ecol. Environ. Res. 2020, 18, 2581–2598. [Google Scholar] [CrossRef]
- da Silva, D.A.; Gonçalves, J.G.R.; Ribeiro, T.; Chiorato, A.F.; Carbonell, S.A.M. Morphophysiological and agronomic performance of 42 common bean genotypes grown hydroponically under phosphorus deficiency. Genet. Mol. Res. 2021, 20, gmr18753. [Google Scholar] [CrossRef]
- Azariz, L.; Elblidi, S.; Fekhaoui, M.; Yahyaoui, A. Uptake and accumulation of lead in Lycopersicon esculentum and Phaseolus vulgaris L. planted on organic hydroponics. Int. J. Environ. Anal. Chem. 2021, 101, 2242–2254. [Google Scholar] [CrossRef]
- Sangakkara, U.R. Effect of tillage and moisture levels on growth, yield and nodulation of common bean (Phaseolus vulgaris L.) and mungbean (Phaseolus radiatus) in the dry season. Indian J. Agron. 2004, 49, 60–63. [Google Scholar]
- Costa-Coelho, G.R.; de Toledo-Souza, E.D.; Café-Filho, A.C.; Lobo, M. Dynamics of common bean web blight epidemics and grain yields in different tillage systems. Trop. Plant Pathol. 2016, 41, 306–311. [Google Scholar] [CrossRef]
- de Toledo-Souza, E.D.; da Silveira, P.M.; Café-Filho, A.C.; Lobo Junior, M. Fusarium wilt incidence and common bean yield according to the preceding crop and the soil tillage system. Pesqui. Agropecu. Bras. 2012, 47, 1031–1037. [Google Scholar] [CrossRef]
- Mulas, D.; Seco, V.; Casquero, P.A.; Velázquez, E.; González-Andrés, F. Inoculation with indigenous rhizobium strains increases yields of common bean (Phaseolus vulgaris L.) in northern Spain, although its efficiency is affected by the tillage system. Symbiosis 2015, 67, 113–124. [Google Scholar] [CrossRef]
- Alguacil, M.d.M.; Roldán, A.; Salinas-García, J.R.; Querejeta, J.I. No tillage affects the phosphorus status, isotopic composition and crop yield of Phaseolus vulgaris in a rain-fed farming system. J. Sci. Food Agric. 2011, 91, 268–272. [Google Scholar] [CrossRef]
- Fatumah, N.; Tilahun, S.A.; Mohammed, S. Water use efficiency, grain yield, and economic benefits of common beans (Phaseolus vulgaris L.) under four soil tillage systems in Mukono District, Uganda. Heliyon 2021, 7, e06308. [Google Scholar] [CrossRef] [PubMed]
- Soratto, R.P.; Perez, A.A.G.; Fernandes, A.M. Age of no-till system and nitrogen management on common bean nutrition and yield. Agron. J. 2014, 106, 809–820. [Google Scholar] [CrossRef]
- Koskey, G.; Mburu, S.W.; Njeru, E.M.; Kimiti, J.M.; Ombori, O.; Maingi, J.M. Potential of native rhizobia in enhancing nitrogen fixation and yields of climbing beans (Phaseolus vulgaris L.) in contrasting environments of eastern Kenya. Front. Plant Sci. 2017, 8, 443. [Google Scholar] [CrossRef]
- Berton, J.F.; Santos, J.C.P.; Coelho, C.M.M.; Klauberg Filho, O. Effect of inoculation associated to leaf sprayed Co+Mo on the yield and grain nutrients in common bean (Phaseolus vulgaris L.). Brazilian Arch. Biol. Technol. 2008, 51, 1089–1096. [Google Scholar] [CrossRef]
- Lucrecia, M.; Ramos, G.; Boddey, R.M. Yield and nodulation of Phaseolus vulgaris and the competitivity of an introduced Rhizobium strain: Effects of lime, mulch and repeated cropping. Soil Biol. Biochem. 1987, 19, 171–177. [Google Scholar] [CrossRef]
- Buttery, B.R.; Park, S.J.; Findlay, W. Growth and yield of white bean (Phaseolus vulgaris L.) in response to nitrogen. phosphorus and potassium fertilizer and to inoculation with Rhizobium. Can. J. Plant Sci. 1987, 67, 425–432. [Google Scholar] [CrossRef]
- Crespo, G.M.; Kluson, R.; Schroder, E. Nitrogen levels and rhizobium inoculation and yields of native white bean (Phaseolus vulgaris L.). J. Agric. Univ. P. R. 1987, 71, 1–6. [Google Scholar]
- Karasu, A.; Oz, M.; Dogan, R. The effect of bacterial inoculation and different nitrogen doses on yield and yield components of some dwarf dry bean cultivars (Phaseolus vulgaris L.). Bulg. J. Agric. Sci. 2011, 17, 296–305. [Google Scholar]
- Efstathiadou, E.; Ntatsi, G.; Savvas, D.; Tampakaki, A.P. Genetic characterization at the species and symbiovar level of indigenous rhizobial isolates nodulating Phaseolus vulgaris in Greece. Sci. Rep. 2021, 11, 8674. [Google Scholar] [CrossRef]
- Rodiño, A.P.; De La Fuente, M.; De Ron, A.M.; Lema, M.J.; Drevon, J.J.; Santalla, M. Variation for nodulation and plant yield of common bean genotypes and environmental effects on the genotype expression. Plant Soil 2011, 346, 349–361. [Google Scholar] [CrossRef]
- Argaw, A.; Muleta, D. Inorganic nitrogen application improves the yield and yield traits of common bean (Phaseolus vulgaris L.) irrespective of the indigenous rhizobial population. S. Afr. J. Plant Soil 2017, 34, 97–104. [Google Scholar] [CrossRef]
- Remans, R.; Ramaekers, L.; Schelkens, S.; Hernandez, G.; Garcia, A.; Reyes, J.L.; Mendez, N.; Toscano, V.; Mulling, M.; Galvez, L.; et al. Effect of Rhizobium-Azospirillum coinoculation on nitrogen fixation and yield of two contrasting Phaseolus vulgaris L. genotypes cultivated across different environments in Cuba. Plant Soil 2008, 312, 25–37. [Google Scholar] [CrossRef]
- Filipini, L.D.; Pilatti, F.K.; Meyer, E.; Ventura, B.S.; Lourenzi, C.R.; Lovato, P.E. Application of Azospirillum on seeds and leaves, associated with Rhizobium inoculation, increases growth and yield of common bean. Arch. Microbiol. 2021, 203, 1033–1038. [Google Scholar] [CrossRef]
- Steiner, F.; Ferreira, H.C.P.; Zuffo, A.M. Can co-inoculation of Rhizobium tropici and Azospirillum brasilense increase common bean nodulation and grain yield? Semin. Agrar. 2019, 40, 81–98. [Google Scholar] [CrossRef]
- Pirbalouti, A.G.; Allahdadi, I.; Akbari, G.A.; Golparvar, A.R.; Rostampoor, S.A. Effects of different strains of Rhizobium legominosarum biovar phaseoli on yield and N2 fixation rate of common bean (Phaseolus vulgaris L.) Iranian cultivars. Pak. J. Biol. Sci. 2006, 9, 1738–1743. [Google Scholar] [CrossRef]
- Pirbalouti, A.G.; Golparvar, A.R.; Rostampoor, S.A. Evaluation of seed yield and yield components of common bean Iranian cultivars for inoculation with four strains of Rhizobium legominosarum biovar phaseoli. J. Agron. 2006, 5, 382–386. [Google Scholar]
- Elkoca, E.; Turan, M.; Donmez, M.F. Effects of single, dual and triple inoculations with Bacillus subtilis, Bacillus megaterium and Rhizobium leguminosarum bv. phaseoli on nodulation, nutrient uptake, yield and yield parameters of common bean (Phaseolus vulgaris L.cv. ’Elkoca-05′). J. Plant Nutr. 2010, 33, 2104–2119. [Google Scholar] [CrossRef]
- Samago, T.Y.; Anniye, E.W.; Dakora, F.D. Grain yield of common bean (Phaseolus vulgaris L.) varieties is markedly increased by rhizobial inoculation and phosphorus application in Ethiopia. Symbiosis 2018, 75, 245–255. [Google Scholar] [CrossRef]
- Uyanöz, R.; Akbulut, M.; Çetin, Ü.; Gültepe, N. Effects of microbial inoculation, organic and chemical fertilizer on yield and physicochemical and cookability properties of bean (Phaseolus vulgaris L.) seeds. Philipp. Agric. Sci. 2007, 90, 168–172. [Google Scholar]
- Aserse, A.A.; Markos, D.; Getachew, G.; Yli-Halla, M.; Lindström, K. Rhizobial inoculation improves drought tolerance, biomass and grain yields of common bean (Phaseolus vulgaris L.) and soybean (Glycine max L.) at Halaba and Boricha in Southern Ethiopia. Arch. Agron. Soil Sci. 2020, 66, 488–501. [Google Scholar] [CrossRef]
- Taylor, J.D.; Day, J.M.; Dudley, C.L. The effect of Rhizobium inoculation and nitrogen fertiliser on nitrogen fixation and seed yield of dry beans (Phaseolus vulgaris). Ann. Appl. Biol. 1983, 103, 419–429. [Google Scholar] [CrossRef]
- Ndlovu, T.J.; Mariga, I.K.; Mafeo, T.P. Influence of Rhizobium phaseoli inoculation and phosphorus application on nodulation and yield of two dry bean (Phaseolus vulgaris) cultivars. Int. J. Agric. Biol. 2017, 19, 1332–1338. [Google Scholar] [CrossRef]
- Yadegari, M.; Rahmani, H.A. Evaluation of bean (Phaseolus vulgaris) seeds’ inoculation with Rhizobium phaseoli and plant growth promoting rhizobacteria (PGPR) on yield and yield components. Afr. J. Agric. Res. 2010, 5, 792–799. [Google Scholar]
- Yadegari, M.; Rahmani, H.A.; Noormohammadi, G.; Ayneband, A. Evaluation of bean (Phaseolus vulgaris) seeds inoculation with Rhizobium phaseoli and plant growth promoting rhizobacteria on yield and yield components. Pak. J. Biol. Sci. 2008, 11, 1935–1939. [Google Scholar] [CrossRef] [PubMed]
- Rezaei-Chiyaneh, E.; Amirnia, R.; Amani Machiani, M.; Javanmard, A.; Maggi, F.; Morshedloo, M.R. Intercropping fennel (Foeniculum vulgare L.) with common bean (Phaseolus vulgaris L.) as affected by PGPR inoculation: A strategy for improving yield, essential oil and fatty acid composition. Sci. Hortic. 2020, 261, 108951. [Google Scholar] [CrossRef]
- da Silveira Cardillo, B.E.; Oliveira, D.P.; Soares, B.L.; Martins, F.A.D.; Rufini, M.; da Silva, J.S.; Neto, G.G.F.; de Andrade, M.J.B.; de Souza Moreira, F.M. Nodulation and yields of common bean are not affected either by fungicides or by the method of inoculation. Agron. J. 2019, 111, 694–701. [Google Scholar] [CrossRef]
- Ndakidemi, P.A.; Dakora, F.D.; Nkonya, E.M.; Ringo, D.; Mansoor, H. Yield and economic benefits of common bean (Phaseolus vulgaris) and soybean (Glycine max) inoculation in northern Tanzania. Aust. J. Exp. Agric. 2006, 46, 571–577. [Google Scholar] [CrossRef]
- Jalal, A.; Galindo, F.S.; Boleta, E.H.M.; da Silva Oliveira, C.E.; Dos Reis, A.R.; Nogueira, T.A.R.; Moretti Neto, M.J.; Mortinho, E.S.; Fernandes, G.C.; Teixeira Filho, M.C.M. Common bean yield and zinc use efficiency in association with diazotrophic bacteria co-inoculations. Agronomy 2021, 11, 959. [Google Scholar] [CrossRef]
- Sánchez, A.C.; Gutiérrez, R.T.; Santana, R.C.; Urrutia, A.R.; Fauvart, M.; Michiels, J.; Vanderleyden, J. Effects of co-inoculation of native Rhizobium and Pseudomonas strains on growth parameters and yield of two contrasting Phaseolus vulgaris L. genotypes under Cuban soil conditions. Eur. J. Soil Biol. 2014, 62, 105–112. [Google Scholar] [CrossRef]
- Daba, S.; Haile, M. Effects of rhizobial inoculant and nitrogen fertilizer on yield and nodulation of common bean. J. Plant Nutr. 2000, 23, 581–591. [Google Scholar] [CrossRef]
- Yadegari, M.; Asadi Rahmani, H.; Noormohammadi, G.; Ayneband, A. Plant growth promoting rhizobacteria increase growth, yield and nitrogen fixation in Phaseolus vulgaris. J. Plant Nutr. 2010, 33, 1733–1743. [Google Scholar] [CrossRef]
- EU. Regulation of the European parliament and of the Council laying down rules on the making available on the market of EU fertilising products and amending Regulations (EC) No 1069/2009 and (EC) No 1107/2009 and repealing Regulation (EC) No 2003/2003. Off. J. Eur. Union 2019, L170, 1–114. [Google Scholar]
- Ozaktan, H.; Ciftci, C.Y.; Uzun, S.; Uzrni, O.; Kaya, M. Effects of humic acid, microbiological fertilizer and phosphate rock on yield and yield components of field bean (Phaseolus vulgaris). Fresenius Environ. Bull. 2020, 29, 856–863. [Google Scholar] [CrossRef]
- Ibrahim, E.A.; Ramadan, W.A. Effect of zinc foliar spray alone and combined with humic acid or/and chitosan on growth, nutrient elements content and yield of dry bean (Phaseolus vulgaris L.) plants sown at different dates. Sci. Hortic. 2015, 184, 101–105. [Google Scholar] [CrossRef]
- Neeraj; Singh, K. Organic amendments to soil inoculated arbuscular mycorrhizal fungi and Pseudomonas fluorescens treatments reduce the development of root-rot disease and enhance the yield of Phaseolus vulgaris L. Eur. J. Soil Biol. 2011, 47, 288–295. [Google Scholar] [CrossRef]
- Bağdatli, M.C.; Erdoğan, O. Effects of Different irrigation levels and arbuscular mycorrhizal fungi (AMF), photosynthesis activator, traditional fertilizer on yield and growth parameters of dry bean (Phaseolus vulgaris L.) in arid climatic conditions. Commun. Soil Sci. Plant Anal. 2019, 50, 527–537. [Google Scholar] [CrossRef]
- Petropoulos, S.A.; Fernandes, Â.; Plexida, S.; Chrysargyris, A.; Tzortzakis, N.; Barreira, J.C.M.; Barros, L.; Ferreira, I.C.F.R. Biostimulants application alleviates water stress effects on yield and chemical composition of greenhouse green bean (Phaseolus vulgaris L.). Agronomy 2020, 10, 181. [Google Scholar] [CrossRef]
- Kocira, A.; Lamorska, J.; Kornas, R.; Nowosad, N.; Tomaszewska, M.; Leszczyńska, D.; Kozłowicz, K.; Tabor, S. Changes in Biochemistry and yield in response to biostimulants applied in bean (Phaseolus vulgaris L.). Agronomy 2020, 10, 189. [Google Scholar] [CrossRef]
- Moreira, A.; Moraes, L.A.C. Yield, nutritional status and soil fertility cultivated with common bean in response to amino-acids foliar application. J. Plant Nutr. 2017, 40, 344–351. [Google Scholar] [CrossRef]
- Tabesh, M.; Kiani, S.; Khoshgoftarmanesh, A.H. The effectiveness of seed priming and foliar application of zinc- amino acid chelates in comparison with zinc sulfate on yield and grain nutritional quality of common bean. J. Plant Nutr. 2020, 43, 2106–2116. [Google Scholar] [CrossRef]
- Kumar, V.; Kumar, P.; Khan, A. Optimization of PGPR and silicon fertilization using response surface methodology for enhanced growth, yield and biochemical parameters of French bean (Phaseolus vulgaris L.) under saline stress. Biocatal. Agric. Biotechnol. 2020, 23, 101463. [Google Scholar] [CrossRef]
- Bruno, A.; Clare, M.M.; Stanley, N.T.; Paul, G.; Maxwell, M.G.; Patrick, R.; Richard, E. Variety × environment × management interaction of diseases and yield in selected common bean varieties. Agron. J. 2017, 109, 2450–2462. [Google Scholar] [CrossRef]
- Mongi, R.; Tongoona, P.; Shimelis, H.; Sibiya, J. Agronomic performance and economics of yield loss associated with angular leaf spot disease of common bean in the southern highlands of Tanzania. Plant Dis. 2018, 102, 85–90. [Google Scholar] [CrossRef]
- Gutiérrez-Moreno, K.; Ruocco, M.; Monti, M.M.; de la Vega, O.M.; Heil, M. Context-dependent effects of trichoderma seed inoculation on anthracnose disease and seed yield of bean (Phaseolus vulgaris L.): Ambient conditions override cultivar-specific differences. Plants 2021, 10, 1739. [Google Scholar] [CrossRef]
- Pereira, H.S.; Wendland, A.; Melo, L.C.; Del Peloso, M.J.; de Faria, L.C.; da Costa, J.G.C.; Nascente, A.S.; Díaz, J.L.C.; de Carvalho, H.W.L.; de Almeida, V.M.; et al. BRS notável: A medium-early-maturing, disease-resistant carioca common bean cultivar with high yield potential. Crop Breed. Appl. Biotechnol. 2012, 12, 220–223. [Google Scholar] [CrossRef][Green Version]
- Naseri, B. Root rot of common bean in Zanjan, Iran: Major pathogens and yield loss estimates. Australas. Plant Pathol. 2008, 37, 546–551. [Google Scholar] [CrossRef]
- El-Mohamedy, R.S.R.; Shafeek, M.R.; El-Samad, E.E.D.H.A.; Salama, D.M.; Rizk, F.A. Field application of plant resistance inducers (PRIs) to control important root rot diseases and improvement growth and yield of green bean (Phaseolus vulgaris L.). Aust. J. Crop Sci. 2017, 11, 496–505. [Google Scholar] [CrossRef]
- Rodríguez, R.; Meléndez, P.L. Effect of fungicide on disease incidence and yield of bean (Phaseolus vulgaris L.) infected with Isariopsis griseola Sacc. and Ascochyta phaseolorum Sacc. J. Agric. Univ. Puerto Rico 1986, 2, 127–134. [Google Scholar] [CrossRef]
- Ellis, M.A.; Galvez, G.E.; Sinclair, J. Effect of foliar applications of systemic fungicides and late harvest on seed quality of dry bean (Phaseolus vulgaris). Plant Dis. Rep. 1976, 60, 1073–1076. [Google Scholar]
- Boersma, J.G.; Hou, A.; Gillard, C.L.; McRae, K.B.; Conner, R.L. Impact of common bacterial blight on the yield, seed weight and seed discoloration of different market classes of dry beans (Phaseolus vulgaris L.). J. Can. J. Plant Sci. 2015, 95, 703–710. [Google Scholar] [CrossRef]
- Tefera, T. Effect of common bacterial blight severity on common bean yield. Trop. Sci. 2006, 46, 41–44. [Google Scholar] [CrossRef]
- Sarrafi, A.; Ecochard, R. Modification of heterosis for protein and yield components by bean common mosaic virus in Phaseolus vulgaris. Plant Breed. 1986, 97, 279–282. [Google Scholar] [CrossRef]
- Souza, T.L.P.O.; Faria, J.C.; Aragão, F.J.L.; Del Peloso, M.J.; Faria, L.C.; Wendland, A.; Aguiar, M.S.; Quintela, E.D.; Melo, C.L.P.; Hungria, M.; et al. Agronomic performance and yield stability of the RNA interference-based Bean golden mosaic virus-resistant common bean. Crop Sci. 2018, 58, 579–591. [Google Scholar] [CrossRef]
- Karel, A.K.; Mghogho, R.M.K. Effects of Insecticide and Plant Populations on the Insect Pests and Yield of Common Bean Phaseolus vulgaris L.). J. Econ. Entomol. 1985, 78, 917–921. [Google Scholar] [CrossRef]
- Srivastava, A.K.; Kumar, A.; Yadav, D.D.; Singh, V. Influence of weed management practices on weeds, crop yield and economics of Rajmash (Phaseolus vulgaris L.). Plant Arch. 2013, 13, 235–238. [Google Scholar]
- da Costa, D.S.; Barbosa, R.M.; de Sá, M.E. Weed management and its relation to yield and seed physiological potential in common bean cultivars. Pesqui. Agropecuária Trop. 2013, 43, 147–154. [Google Scholar] [CrossRef]
- Esmaeilzadeh, S.; Aminpanah, H. Effects of planting date and spatial arrangement on common bean (Phaseolus vulgaris) yield under weed-free and weedy conditions. Planta Daninha 2015, 33, 425–432. [Google Scholar] [CrossRef]
- Mekonnen, G. Effect of pre emergence herbicides and their combinations on weeds infestation, yield components and yield of common bean (Phaseolus vulgaris L.) at Guraferda and Menitshashaworeda, South West Ethiopia. Plant Cell Biotechnol. Mol. Biol. 2020, 21, 12–23. [Google Scholar]
- Rashidi, S.; Yousefi, A.R.; Pouryousef, M.; Goicoechea, N. Mycorrhizal impact on competitive relationships and yield parameters in Phaseolus vulgaris L.—Weed mixtures. Mycorrhiza 2021, 31, 599–612. [Google Scholar] [CrossRef] [PubMed]
- Feizollah, J.; Aminpanah, H. Effects of planting distance and weeding regime on green bean (Phaseolus vulgaris L.) growth and yield. Rev. Fac. Agron. 2016, 33, 325–345. [Google Scholar]
- Ngadze, E.; Mashingaidze, A.B.; Sibiya, J. Weed density and biomass are reduced and plant growth and seed yield increased in common bean after solarisation with clear and black plastic. S. Afr. J. Plant Soil 2018, 35, 223–230. [Google Scholar] [CrossRef]
- Singh, S.; Singh, R.P.; Shukla, U.N.; Singh, J.K.; Singh, O.N. Efficacy of herbicides and nutrient management on weed dynamics and yield of French bean (Phaseolus vulgaris). Indian J. Agric. Sci. 2018, 88, 1794–1800. [Google Scholar]
- Ayonoadu, U.W.U.; Norrington-Davies, J.; Edje, O.T.; Mughogho, L.K. Weed control and its effects on yield of Phaseolus vulgaris beans in Malawi. J. Agric. Sci. 1974, 82, 283–286. [Google Scholar] [CrossRef]
- Głowacka, A. The effects of strip cropping and weed control methods on yield and yield components of dent maize, common bean and spring barley. Polish J. Nat. Sci. 2013, 28, 389–408. [Google Scholar]
- Dusabumuremyi, P.; Niyibigira, C.; Mashingaidze, A.B. Narrow row planting increases yield and suppresses weeds in common bean (Phaseolus vulgaris L.) in a semi-arid agro-ecology of Nyagatare, Rwanda. Crop Prot. 2014, 64, 13–18. [Google Scholar] [CrossRef]
- Byiringiro, B.; Birungi, S.; Musoni, A.; Mashingaidze, A.B. The effect of planting date on weed density, biomass and seed yield in common bean (Phaseolus vulgaris L.) in the semi-arid region of Nyagatare, Rwanda. Trop. Agric. 2017, 94, 335–345. [Google Scholar]
- Rahman, M.A.; Yahata, H.; Miah, M.G.; Ahamed, T.; Begum, M.N. Effectiveness of tree leaf mulch comparing with conventional mulches on common bean at different irrigation levels: Growth, yield, water use efficiency and weed infestation. Arch. Agron. Soil Sci. 2008, 54, 331–342. [Google Scholar] [CrossRef]
| Agronomic Practice | Topic Words |
|---|---|
| Breeding | genetic * or genotype * or landrace * or breed * |
| Sowing density and season | sowing date or plant density or sowing rate or sowing season |
| Irrigation | drought or water stress or deficit irrigation or irrigation or salinity or saline or salt stress or irrigation quality or water quality |
| Fertilization | organic or conventional or fertilizer or inorganic or nutrition or nitrogen or potassium or phosphorus |
| Intercropping | intercrop * |
| Soilless culture | hydroponic * or soilless or floating or nft or nutrient solution or vertical |
| Tillage | Till * |
| Rhizobia application | rhizob * or inocul * |
| Biostimulant/biofertilizer application | arbuscular mycorrhizal fungi or PGPR or azospirillum or plant growth-promot * or rhizobacteria or alga * or amino or biostimulant * or fulvi * or humi * or pgpb or biofertili * |
| Disease management | Fung * or biotic or virus or pathogen or bacter * or disease |
| Pest management | Insect *or pest * or acari * |
| Weed management | Weed * or herbicide * |
| Agronomic Practice | Breeding | Sowing Density and Season | Irrigation | Fertilization | Intercropping | Soilless Culture | Tillage | Rhizobia Application | Biostimulant/Biofertilizer Application | Disease Management | Pest Management | Weed Management |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Breeding | √ | √ | √ | |||||||||
| Sowing density and season | √ | √ | ||||||||||
| Irrigation | √ | √ | ||||||||||
| Fertilization | √ | √ | √ | √ | ||||||||
| Intercropping | √ | √ | √ | |||||||||
| Soilless culture | ||||||||||||
| Tillage | √ | |||||||||||
| Rhizobia application | √ | √ | √ | √ * | √ * | |||||||
| Biostimulant/biofertilizer application | √ | √ | √ * | * | √ | |||||||
| Disease management | √ | * | * | |||||||||
| Pest management | √ | |||||||||||
| Weed management | √ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karavidas, I.; Ntatsi, G.; Vougeleka, V.; Karkanis, A.; Ntanasi, T.; Saitanis, C.; Agathokleous, E.; Ropokis, A.; Sabatino, L.; Tran, F.; et al. Agronomic Practices to Increase the Yield and Quality of Common Bean (Phaseolus vulgaris L.): A Systematic Review. Agronomy 2022, 12, 271. https://doi.org/10.3390/agronomy12020271
Karavidas I, Ntatsi G, Vougeleka V, Karkanis A, Ntanasi T, Saitanis C, Agathokleous E, Ropokis A, Sabatino L, Tran F, et al. Agronomic Practices to Increase the Yield and Quality of Common Bean (Phaseolus vulgaris L.): A Systematic Review. Agronomy. 2022; 12(2):271. https://doi.org/10.3390/agronomy12020271
Chicago/Turabian StyleKaravidas, Ioannis, Georgia Ntatsi, Vasiliki Vougeleka, Anestis Karkanis, Theodora Ntanasi, Costas Saitanis, Evgenios Agathokleous, Andreas Ropokis, Leo Sabatino, Fanny Tran, and et al. 2022. "Agronomic Practices to Increase the Yield and Quality of Common Bean (Phaseolus vulgaris L.): A Systematic Review" Agronomy 12, no. 2: 271. https://doi.org/10.3390/agronomy12020271
APA StyleKaravidas, I., Ntatsi, G., Vougeleka, V., Karkanis, A., Ntanasi, T., Saitanis, C., Agathokleous, E., Ropokis, A., Sabatino, L., Tran, F., Iannetta, P. P. M., & Savvas, D. (2022). Agronomic Practices to Increase the Yield and Quality of Common Bean (Phaseolus vulgaris L.): A Systematic Review. Agronomy, 12(2), 271. https://doi.org/10.3390/agronomy12020271













