Prospects for Increasing the Efficacy of Plant Resistance Inducers Stimulating Salicylic Acid
Abstract
1. Introduction
2. The Role of the SA Signaling Pathway in Plant Immunity
3. High Concentrations in SA Are also Associated with Increased Tolerance of Plants towards Abiotic Stresses
3.1. High SA Exerts Positive Effects on Photosynthesis of Stressed Plants
3.2. High SA Exerts Positive Effects on Growth and Yield of Stressed Plants
3.3. High SA May Exert Positive Effects on Quality of Production
3.4. High SA Increases Plant Tolerance to Exceedingly High Temperatures
3.5. High SA increases Plant Tolerance to Exceedingly Low Temperatures
3.6. High SA Can Help Crops to Overcome the Consequences of Frost
3.7. SA Acts as a Critical Hormone in Improving Uptake and Status of Mineral Nutrients under Stress Conditions
3.8. High SA Stimulates Flowering
4. The Dark Side of High SA
5. A Review of the Existing Means of Increasing SA in Plants and Crops
5.1. Analogs of SA and Chemical Inducers/Stimulators of the SA Signaling Pathway
5.2. Chitin and Chitosan
5.3. Phosphites
5.4. Laminarin and Sulfated Laminarin
5.5. COS-OGA
5.6. Plant and Yeast Extracts
5.7. Essential Oils
5.8. Biological PRIs
5.9. Physical PRIs
5.10. Flashes of UV-C Light, a Newcomer among PRIs with Unique Features
6. Combining PRIs: Playing Complementarity or Reinforcement Effects?
7. Reducing Disease Pressure
7.1. Chemical Substances with Antifungal Properties
7.2. Physical Methods of Surface Disinfection
8. Ensuring That Plants Have the Resources Required for Synthesis of Defense Compounds and Structures
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ma, C.-S.; Zhang, W.; Peng, Y.; Zhao, F.; Chang, X.-Q.; Xing, K.; Zhu, L.; Ma, G.; Yang, H.-P.; Rudolf, V.H.W. Climate warming promotes pesticide resistance through expanding overwintering range of a global pest. Nat. Commun. 2021, 12, 5351. [Google Scholar] [CrossRef] [PubMed]
- Rani, L.; Thapa, K.; Kanojia, N.; Sharma, N.; Singh, S.; Grewal, A.S.; Srivastav, A.L.; Kaushal, J. An extensive review on the consequences of chemical pesticides on human health and environment. J. Clean. Prod. 2020, 283, 124657. [Google Scholar] [CrossRef]
- Marolleau, B.; Gaucher, M.; Heintz, C.; Degrave, A.; Warneys, R.; Orain, G.; Lemarquand, A.; Brisset, M.-N. When a Plant Resistance Inducer Leaves the Lab for the Field: Integrating ASM into Routine Apple Protection Practices. Front. Plant Sci. 2017, 8, 1938. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, D.; Raikhy, G.; Kumar, D. Chemical elicitors of systemic acquired resistance—Salicylic acid and its functional analogs. Curr. Plant Biol. 2019, 17, 48–59. [Google Scholar] [CrossRef]
- White, R. Acetylsalicylic acid (aspirin) induces resistance to tobacco mosaic virus in tobacco. Virology 1979, 99, 410–412. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Zamioudis, C.; Berendsen, R.L.; Weller, D.M.; Van Wees, S.C.M.; Bakker, P.A.H.M. Induced Systemic Resistance by Beneficial Microbes. Annu. Rev. Phytopathol. 2014, 52, 347–375. [Google Scholar] [CrossRef]
- Van Wees, S.C.; Van der Ent, S.; Pieterse, C.M. Plant immune responses triggered by beneficial microbes. Curr. Opin. Plant Biol. 2008, 11, 443–448. [Google Scholar] [CrossRef]
- Yassin, M.; Ton, J.; Rolfe, S.A.; Valentine, T.A.; Cromey, M.; Holden, N.; Newton, A.C. The rise, fall and resurrection of chemical-induced resistance agents. Pest Manag. Sci. 2021, 77, 3900–3909. [Google Scholar] [CrossRef]
- Launay, M.; Caubel, J.; Bourgeois, G.; Huard, F.; de Cortazar-Atauri, I.G.; Bancal, M.-O.; Brisson, N. Climatic indicators for crop infection risk: Application to climate change impacts on five major foliar fungal diseases in Northern France. Agric. Ecosyst. Environ. 2014, 197, 147–158. [Google Scholar] [CrossRef]
- ACTA. Stimulateurs de Défense des Plantes (SDP), Technique à l’étude. Guide Pour la Conception de Systèmes de Production Fruitière Economes en Produits Phytosanitaires; GIS et Ministère de l’Agriculture: Paris, France, 2014; pp. 104–106. [Google Scholar]
- Sandroni, M.; Liljeroth, E.; Mulugeta, T.; Alexandersson, E. Plant resistance inducers (PRIs): Perspectives for future disease management in the field. CAB Rev. Perspect. Agric. Vet.-Sci. Nutr. Nat. Resour. 2020, 15, 1–10. [Google Scholar] [CrossRef]
- Ripoll, J.; Urban, L.; Staudt, M.; Lopez-Lauri, F.; Bidel, L.P.; Bertin, N. Water shortage and quality of fleshy fruits—Making the most of the unavoidable. J. Exp. Bot. 2014, 65, 4097–4117. [Google Scholar] [CrossRef]
- Walters, D.; Heil, M. Costs and trade-offs associated with induced resistance. Physiol. Mol. Plant Pathol. 2007, 71, 3–17. [Google Scholar] [CrossRef]
- Walters, D.R.; Fountaine, J.M. Practical application of induced resistance to plant diseases: An appraisal of effectiveness under field conditions. J. Agric. Sci. 2009, 147, 523–535. [Google Scholar] [CrossRef]
- Austin, C. Sunlight’s Influence on Grapevine Powdery Mildew: Direct Effects on Pathogen Development and Attendant Consequences of Canopy Management and Vineyard Variability. Ph.D. Dissertation, Cornell University, Ithaca, NY, USA, 2010. [Google Scholar]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef]
- Krantev, A.; Yordanova, R.; Janda, T.; Szalai, G.; Popova, L. Treatment with salicylic acid decreases the effect of cadmium on photosynthesis in maize plants. J. Plant Physiol. 2008, 165, 920–931. [Google Scholar] [CrossRef] [PubMed]
- Arfan, M.; Athar, H.R.; Ashraf, M. Does exogenous application of salicylic acid through the rooting medium modulate growth and photosynthetic capacity in two differently adapted spring wheat cultivars under salt stress? J. Plant Physiol. 2007, 164, 685–694. [Google Scholar] [CrossRef]
- Miao, Y.; Luo, X.; Gao, X.; Wang, W.; Li, B.; Hou, L. Exogenous salicylic acid alleviates salt stress by improving leaf photosynthesis and root system architecture in cucumber seedlings. Sci. Hortic. 2020, 272, 109577. [Google Scholar] [CrossRef]
- Wang, L.-J.; Fan, L.; Loescher, W.; Duan, W.; Liu, G.-J.; Cheng, J.-S.; Luo, H.-B.; Li, S.-H. Salicylic acid alleviates decreases in photosynthesis under heat stress and accelerates recovery in grapevine leaves. BMC Plant Biol. 2010, 10, 34. [Google Scholar] [CrossRef]
- Janda, T.; Gondor, O.K.; Yordanova, R.; Szalai, G.; Pál, M. Salicylic acid and photosynthesis: Signalling and effects. Acta Physiol. Plant. 2014, 36, 2537–2546. [Google Scholar] [CrossRef]
- Mateo, A.; Funck, D.; Mühlenbock, P.; Kular, B.; Mullineaux, P.M.; Karpinski, S. Controlled levels of salicylic acid are required for optimal photosynthesis and redox homeostasis. J. Exp. Bot. 2006, 57, 1795–1807. [Google Scholar] [CrossRef]
- Liu, T.; Li, T.; Zhang, L.; Li, H.; Liu, S.; Yang, S.; An, Q.; Pan, C.; Zou, N. Exogenous salicylic acid alleviates the accumulation of pesticides and mitigates pesticide-induced oxidative stress in cucumber plants (Cucumis sativus L.). Ecotoxicol. Environ. Saf. 2020, 208, 111654. [Google Scholar] [CrossRef]
- Dong, C.-J.; Li, L.; Shang, Q.-M.; Liu, X.-Y.; Zhang, Z.-G. Endogenous salicylic acid accumulation is required for chilling tolerance in cucumber (Cucumis sativus L.) seedlings. Planta 2014, 240, 687–700. [Google Scholar] [CrossRef]
- Metwally, A.; Finkemeier, I.; Georgi, M.; Dietz, K.-J. Salicylic Acid Alleviates the Cadmium Toxicity in Barley Seedlings. Plant Physiol. 2003, 132, 272–281. [Google Scholar] [CrossRef]
- Wael, M.S.; Mostafa, M.R.; Taia, A.A.E.-M.; Saad, M.H.; Magdi, T.A. Alleviation of cadmium toxicity in common bean (Phaseolus vulgaris L.) plants by the exogenous application of salicylic acid. J. Hortic. Sci. Biotechnol. 2015, 90, 83–91. [Google Scholar] [CrossRef]
- Roustakhiz, J.; Saboki, E. Effect of salicylic acid on yield and yield component of grapevine (Vitis vinifera) under salinity stress condition. Int. J. Farming Allied Sci. 2017, 6, 39–42. [Google Scholar]
- Zamaninejad, M.; Khorasani, S.K.; Moeini, M.J.; Heidarian, A.R. Effect of salicylic acid on morphological characteristics, yield and yield components of corn (Zea mays L.) under drought condition. Eur. J. Exp. Biol. 2013, 3, 153–161. [Google Scholar]
- Tahjib-Ul-Arif, M.; Siddiqui, M.N.; Sohag, A.A.M.; Sakil, M.A.; Rahman, M.M.; Polash, M.A.S.; Mostofa, M.G.; Tran, L.-S.P. Salicylic Acid-Mediated Enhancement of Photosynthesis Attributes and Antioxidant Capacity Contributes to Yield Improvement of Maize Plants Under Salt Stress. J. Plant Growth Regul. 2018, 37, 1318–1330. [Google Scholar] [CrossRef]
- Qados, M.M. Effects of salicylic acid on growth, yield and chemical contents of pepper (Capsicum annuum L.) plants grown under salt stress conditions. Int. J. Agric. Crop. Sci. 2015, 8, 107–113. [Google Scholar]
- Karlidag, H.; Yildirim, E.; Turan, M. Exogenous applications of salicylic acid affect quality and yield of strawberry grown under antifrost heated greenhouse conditions. J. Plant Nutr. Soil Sci. 2009, 172, 270–276. [Google Scholar] [CrossRef]
- Azimi, M.S.; Daneshian, J.; Sayfzadeh, S.; Zare, S. Evaluation of amino acid and salicylic acid application on yield and growth of wheat under water deficit. Int. J. Agric. Crop. Sci. 2013, 5, 816. [Google Scholar]
- Munir, M.; Shabbir, G. Salicylic acid mediated heat stress tolerance in selected bread wheat genotypes of Pakistan. Pak. J. Bot. 2018, 50, 2141–2146. [Google Scholar]
- Elwan, M.; El-Hamahmy, M. Improved productivity and quality associated with salicylic acid application in greenhouse pepper. Sci. Hortic. 2009, 122, 521–526. [Google Scholar] [CrossRef]
- Urban, L.; Léchaudel, M.; Lu, P. Effect of fruit load and girdling on leaf photosynthesis in Mangifera indica L. J. Exp. Bot. 2004, 55, 2075–2085. [Google Scholar] [CrossRef] [PubMed]
- Larkindale, J.; Hall, J.D.; Knight, M.; Vierling, E. Heat Stress Phenotypes of Arabidopsis Mutants Implicate Multiple Signaling Pathways in the Acquisition of Thermotolerance. Plant Physiol. 2005, 138, 882–897. [Google Scholar] [CrossRef]
- Clarke, S.M.; Mur, L.A.J.; Wood, J.E.; Scott, I.M. Salicylic acid dependent signaling promotes basal thermotolerance but is not essential for acquired thermotolerance in Arabidopsis thaliana. Plant J. 2004, 38, 432–447. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.-J.; Li, S.-H. Salicylic acid-induced heat or cold tolerance in relation to Ca2+ homeostasis and antioxidant systems in young grape plants. Plant Sci. 2006, 170, 685–694. [Google Scholar] [CrossRef]
- Saleem, M.; Fariduddin, Q.; Janda, T. Multifaceted Role of Salicylic Acid in Combating Cold Stress in Plants: A Review. J. Plant Growth Regul. 2020, 40, 464–485. [Google Scholar] [CrossRef]
- Suzuki, N.; Mittler, R. Reactive oxygen species and temperature stresses: A delicate balance between signaling and destruction. Physiol. Plant. 2005, 126, 45–51. [Google Scholar] [CrossRef]
- Bredow, M.; Walker, V.K. Ice-Binding Proteins in Plants. Front. Plant Sci. 2017, 8, 2153. [Google Scholar] [CrossRef]
- Janda, T.; Szalai, G.; Antunocis, Z.; Horvath, E.; Paldi, E. Effect of benzoic acid and aspirin on chilling tolerance and photosynthesis in young maize plants. Maydica 2000, 45, 29–33. [Google Scholar]
- Griffith, M.; Yaish, M.W. Antifreeze proteins in overwintering plants: A tale of two activities. Trends Plant Sci. 2004, 9, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Tasgin, E.; Atici, O.; Nalbantoglu, B.; Popova, L. Effects of salicylic acid and cold treatments on protein levels and on the activities of antioxidant enzymes in the apoplast of winter wheat leaves. Phytochemistry 2006, 67, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, X.; Zhang, J.; Huang, M.; Cai, J.; Zhou, Q.; Dai, T.; Jiang, D. Salicylic acid and cold priming induce late-spring freezing tolerance by maintaining cellular redox homeostasis and protecting photosynthetic apparatus in wheat. Plant Growth Regul. 2019, 90, 109–121. [Google Scholar] [CrossRef]
- Clarke, S.M.; Cristescu, S.M.; Miersch, O.; Harren, F.J.M.; Wasternack, C.; Mur, L.A.J. Jasmonates act with salicylic acid to confer basal thermotolerance in Arabidopsis thaliana. New Phytol. 2009, 182, 175–187. [Google Scholar] [CrossRef]
- López-Delgado, H.A.; Martínez-Gutiérrez, R.; Mora-Herrera, M.E.; Torres-Valdés, Y. Induction of Freezing Tolerance by the Application of Hydrogen Peroxide and Salicylic Acid as Tuber-Dip or Canopy Spraying in Solanum tuberosum L. Plants. Potato Res. 2018, 61, 195–206. [Google Scholar] [CrossRef]
- Zhang, B.B.; Guo, L.; Song, Z.Z.; Yu, M.L.; Ma, R.J. Effect of salicylic acid on freezing injury in peach floral organs and the expressions of CBF genes. Biol. Plant. 2017, 61, 622–630. [Google Scholar] [CrossRef]
- Matton, Y.; Sement, F.; Rouesné, B.; Ledermann, L.; Pascouau, N.; Charel, J.; Goncalves, H.; Urban, L.; Aaarrouf, J. Utilisation D’un Dispositif Mobile Délivrant Des Impulsions Lumineuses Sur Une Matière Végétale Pour La Prévention Contre Les Impacts Liés à Des Stress Abiotiques. European Patent PCT/EP2022/066721, 2021. [Google Scholar]
- Orrantia-Araujo, M.A.; Martínez-Téllez, M.; Rivera-Domínguez, M.; Hernández-Oñate, M.; Vargas-Arispuro, I. Changes in the Endogenous Content and Gene Expression of Salicylic Acid Correlate with Grapevine Bud Dormancy Release. J. Plant Growth Regul. 2020, 40, 254–262. [Google Scholar] [CrossRef]
- Sheteiwy, M.S.; An, J.; Yin, M.; Jia, X.; Guan, Y.; He, F.; Hu, J. Cold plasma treatment and exogenous salicylic acid priming enhances salinity tolerance of Oryza sativa seedlings. Protoplasma 2018, 256, 79–99. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, S.; Wang, P.; Hou, J.; Qian, J.; Ao, Y.; Lu, J.; Li, L. Salicylic acid involved in the regulation of nutrient elements uptake and oxidative stress in Vallisneria natans (Lour.) Hara under Pb stress. Chemosphere 2011, 84, 136–142. [Google Scholar] [CrossRef]
- Lee, T.T.; Skoog, F. Effect of substituted phenols on bud formation and growth of tobacco tissue and vineyard conditions. Physiol. Plant. 1965, 18, 386–402. [Google Scholar] [CrossRef]
- Hayat, Q.; Hayat, S.; Irfan, M.; Ahmad, A. Effect of exogenous salicylic acid under changing environment: A review. Environ. Exp. Bot. 2010, 68, 14–25. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, X. Salicylic acid: Biosynthesis, perception, and contributions to plant immunity. Curr. Opin. Plant Biol. 2019, 50, 29–36. [Google Scholar] [CrossRef]
- Yalpani, N.; Enyedi, A.J.; Raskin, I. Ultraviolet light and ozone stimulate accumulation of salicylic acid, pathogenesis-related proteins and virus resistance in tobacco. Planta 1994, 193, 372–376. [Google Scholar] [CrossRef]
- Martínez, C.; Pons, E.; Prats, G.; León, J. Salicylic acid regulates flowering time and links defence responses and reproductive development. Plant J. 2003, 37, 209–217. [Google Scholar] [CrossRef]
- Pancheva, T.; Popova, L.; Uzunova, A. Effects of salicylic acid on growth and photosynthesis in barley plants. J. Plant Physiol. 1996, 149, 57–63. [Google Scholar] [CrossRef]
- Janda, T.; Szalai, G.; Tari, I.; Páldi, E. Hydroponic treatment with salicylic acid decreases the effects of chilling injury in maize (Zea mays L.) plants. Planta 1999, 208, 175–180. [Google Scholar] [CrossRef]
- Mateo, A.; Mühlenbock, P.; Rustérucci, C.; Chang, C.C.-C.; Miszalski, Z.; Karpinska, B.; Parker, J.E.; Mullineaux, P.M.; Karpinski, S. LESION SIMULATING DISEASE 1 Is Required for Acclimation to Conditions That Promote Excess Excitation Energy. Plant Physiol. 2004, 136, 2818–2830. [Google Scholar] [CrossRef] [PubMed]
- Janda, K.; Hideg, É.; Szalai, G.; Kovács, L.; Janda, T. Salicylic acid may indirectly influence the photosynthetic electron transport. J. Plant Physiol. 2012, 169, 971–978. [Google Scholar] [CrossRef]
- Poór, P.; Tari, I. Regulation of stomatal movement and photosynthetic activity in guard cells of tomato abaxial epidermal peels by salicylic acid. Funct. Plant Biol. 2012, 39, 1028–1037. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zu, X.; Wang, X.; Sun, A.; Zhang, J.; Wang, S.; Chen, Y. Comparative Proteomic Analysis of the Effects of Salicylic Acid and Abscisic Acid on Maize (Zea mays L.) Leaves. Plant Mol. Biol. Rep. 2012, 31, 507–516. [Google Scholar] [CrossRef]
- Csinos, A.S.; Pappu, H.R.; McPherson, R.M.; Stephenson, M.G. Management of Tomato spotted wilt virus in Flue-Cured Tobacco with Acibenzolar-S-Methyl and Imidacloprid. Plant Dis. 2001, 85, 292–296. [Google Scholar] [CrossRef]
- Louws, F.J.; Wilson, M.; Campbell, H.L.; Cuppels, D.A.; Jones, J.B.; Shoemaker, P.B.; Sahin, F.; Miller, S.A. Field Control of Bacterial Spot and Bacterial Speck of Tomato Using a Plant Activator. Plant Dis. 2001, 85, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Romero, A.M.; Kousik, C.S.; Ritchie, D.F. Resistance to Bacterial Spot in Bell Pepper Induced by Acibenzolar-S-Methyl. Plant Dis. 2001, 85, 189–194. [Google Scholar] [CrossRef]
- Gent, D.H.; Schwartz, H.F. Management of Xanthomonas Leaf Blight of Onion with a Plant Activator, Biological Control Agents, and Copper Bactericides. Plant Dis. 2005, 89, 631–639. [Google Scholar] [CrossRef]
- Nair, C.B.; Anith, K.N.; Sreekumar, J. Mitigation of growth retardation effect of plant defense activator, acibenzolar-S-methyl, in amaranthus plants by plant growth-promoting rhizobacteria. World J. Microbiol. Biotechnol. 2006, 23, 1183–1187. [Google Scholar] [CrossRef]
- Heil, M.; Hilpert, A.; Kaiser, W.; Linsenmair, K.E. Reduced growth and seed set following chemical induction of pathogen defence: Does systemic acquired resistance (SAR) incur allocation costs? J. Ecol. 2000, 88, 645–654. [Google Scholar] [CrossRef]
- Babu, B.; Paret, M.L.; Martini, X.; Knox, G.W.; Riddle, B.; Ritchie, L.; Aldrich, J.; Kalischuk, M.; Da Silva, S. Impact of Foliar Application of Acibenzolar-S-Methyl on Rose Rosette Disease and Rose Plant Quality. Plant Dis. 2022, 106, 818–827. [Google Scholar] [CrossRef]
- Xue, L.J.; Guo, W.; Yuan, Y.; Anino, E.O.; Nyamdari, B.; Wilson, M.C.; Frost, C.J.; Chen, H.Y.; Babst, B.A.; Harding, S.A.; et al. Constitutively Elevated Salicylic Acid Levels Alter Photosynthesis and Oxidative State but Not Growth in Transgenic Populus. Plant Cell 2013, 25, 2714–2730. [Google Scholar] [CrossRef]
- Szalai, G.; Horgosi, S.; Soós, V.; Majláth, I.; Balázs, E.; Janda, T. Salicylic acid treatment of pea seeds induces its de novo synthesis. J. Plant Physiol. 2011, 168, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Gozzo, F.; Faoro, F. Systemic Acquired Resistance (50 Years after Discovery): Moving from the Lab to the Field. J. Agric. Food Chem. 2013, 61, 12473–12491. [Google Scholar] [CrossRef]
- Cole, D.L. The efficacy of acibenzolar-S-methyl, an inducer of systemic acquired resistance, against bacterial and fungal diseases of tobacco. Crop Prot. 1999, 18, 267–273. [Google Scholar] [CrossRef]
- Babu, R.M.; Sajeena, A.; Samundeeswari, A.V.; Sreedhar, A.; Vidhyasekeran, P.; Reddy, M.S. Induction of bacterial blight (Xanthomonas oryzae pv. oryzae) resistance in rice by treatment with acibenzolar-S-methyl. Ann. Appl. Biol. 2003, 143, 333–340. [Google Scholar] [CrossRef]
- Ishiga, T.; Iida, Y.; Sakata, N.; Ugajin, T.; Hirata, T.; Taniguchi, S.; Hayashi, K.; Ishiga, Y. Acibenzolar-S-methyl activates stomatal-based defense against Pseudomonas cannabina pv. alisalensis in cabbage. J. Gen. Plant Pathol. 2019, 86, 48–54. [Google Scholar] [CrossRef]
- Lopez-Moya, F.; Suarez-Fernandez, M.; Lopez-Llorca, L.V. Molecular Mechanisms of Chitosan Interactions with Fungi and Plants. Int. J. Mol. Sci. 2019, 20, 332. [Google Scholar] [CrossRef]
- Malerba, M.; Cerana, R. Recent Applications of Chitin- and Chitosan-Based Polymers in Plants. Polymers 2019, 11, 839. [Google Scholar] [CrossRef] [PubMed]
- De Bona, G.S.; Vincenzi, S.; De Marchi, F.; Angelini, E.; Bertazzon, N. Chitosan induces delayed grapevine defense mechanisms and protects grapevine against Botrytis cinerea. J. Plant Dis. Prot. 2021, 128, 715–724. [Google Scholar] [CrossRef]
- Chakraborty, M.; Hasanuzzaman, M.; Rahman, M.; Khan, A.R.; Bhowmik, P.; Mahmud, N.U.; Tanveer, M.; Islam, T. Mechanism of Plant Growth Promotion and Disease Suppression by Chitosan Biopolymer. Agriculture 2020, 10, 624. [Google Scholar] [CrossRef]
- Lobato, M.C.; Machinandiarena, M.F.; Tambascio, C.; Dosio, G.A.A.; Caldiz, D.O.; Daleo, G.R.; Andreu, A.B.; Olivieri, F.P. Effect of foliar applications of phosphite on post-harvest potato tubers. Eur. J. Plant Pathol. 2011, 130, 155–163. [Google Scholar] [CrossRef]
- Olivieri, F.P.; Feldman, M.L.; Machinandiarena, M.F.; Lobato, M.C.; Caldiz, D.O.; Daleo, G.R.; Andreu, A.B. Phosphite applications induce molecular modifications in potato tuber periderm and cortex that enhance resistance to pathogens. Crop Prot. 2012, 32, 1–6. [Google Scholar] [CrossRef]
- Eshraghi, L.; Anderson, J.; Aryamanesh, N.; McComb, J.A.; Shearer, B.; Hardy, G. Defence Signalling Pathways Involved in Plant Resistance and Phosphite-Mediated Control of Phytophthora cinnamomi. Plant Mol. Biol. Rep. 2013, 32, 342–356. [Google Scholar] [CrossRef]
- Gill, U.S.; Sun, L.; Rustgi, S.; Tang, Y.; Wettstein, D.; Mysore, K.S. Transcriptome-based analyses of phosphite-mediated suppression of rust pathogens Puccinia emaculata and Phakopsora pachyrhizi and functional characterization of selected fungal target genes. Plant J. 2018, 93, 894–904. [Google Scholar] [CrossRef]
- Massoud, K.; Barchietto, T.; Le Rudulier, T.; Pallandre, L.; Didierlaurent, L.; Garmier, M.; Ambard-Bretteville, F.; Seng, J.-M.; Saindrenan, P. Dissecting Phosphite-Induced Priming in Arabidopsis Infected with Hyaloperonospora arabidopsidis. Plant Physiol. 2012, 159, 286–298. [Google Scholar] [CrossRef]
- Thao, H.T.B.; Yamakawa, T. Phosphite (phosphorous acid): Fungicide, fertilizer or bio-stimulator? Soil Sci. Plant Nutr. 2009, 55, 228–234. [Google Scholar] [CrossRef]
- Aziz, A.; Poinssot, B.; Daire, X.; Adrian, M.; Bézier, A.; Lambert, B.; Joubert, J.-M.; Pugin, A. Laminarin Elicits Defense Responses in Grapevine and Induces Protection against Botrytis cinerea and Plasmopara viticola. Mol. Plant-Microbe Interact. 2003, 16, 1118–1128. [Google Scholar] [CrossRef]
- Renard-Merlier, D.; Randoux, B.; Nowak, E.; Farcy, F.; Durand, R.; Reignault, P. Iodus 40, salicylic acid, heptanoyl salicylic acid and trehalose exhibit different efficacies and defence targets during a wheat/powdery mildew interaction. Phytochemistry 2007, 68, 1156–1164. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, A.; Trouvelot, S.; Kelloniemi, J.; Frettinger, P.; Wendehenne, D.; Daire, X.; Joubert, J.-M.; Ferrarini, A.; Delledonne, M.; Flors, V.; et al. The Sulfated Laminarin Triggers a Stress Transcriptome before Priming the SA- and ROS-Dependent Defenses during Grapevine’s Induced Resistance against Plasmopara viticola. PLoS ONE 2014, 9, e88145. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-R.; Lin, Y.-C.; Chuang, H.-W. Laminarin modulates the chloroplast antioxidant system to enhance abiotic stress tolerance partially through the regulation of the defensin-like gene expression. Plant Sci. 2016, 247, 83–92. [Google Scholar] [CrossRef]
- Cabrera, J.-C.; Boland, A.; Cambier, P.; Frettinger, P.; Van Cutsem, P. Chitosan oligosaccharides modulate the supramolecular conformation and the biological activity of oligogalacturonides in Arabidopsis. Glycobiology 2010, 20, 775–786. [Google Scholar] [CrossRef]
- Van Cutsem, P.; Messiaen, J. Biological effects of pectin fragments in plant cells. Acta Bot. Neerl. 1994, 43, 231–245. [Google Scholar] [CrossRef]
- Moreau, S.; Van Aubel, G.; Janky, R.; Van Cutsem, P. Chloroplast Electron Chain, ROS Production, and Redox Homeostasis Are Modulated by COS-OGA Elicitation in Tomato (Solanum lycopersicum) Leaves. Front. Plant Sci. 2020, 11, 597589. [Google Scholar] [CrossRef] [PubMed]
- Fujita, M.; Fujita, Y.; Noutoshi, Y.; Takahashi, F.; Narusaka, Y.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Crosstalk between abiotic and biotic stress responses: A current view from the points of convergence in the stress signaling networks. Curr. Opin. Plant Biol. 2006, 9, 436–442. [Google Scholar] [CrossRef]
- Javaid, A.; Munir, R.; Khan, I.H.; Shoaib, A. Control of the chickpea blight, Ascochyta rabiei, with the weed plant, Withania somnifera. Egypt. J. Biol. Pest Control. 2020, 30, 114. [Google Scholar] [CrossRef]
- Javaid, A.; Khan, I.; Shoaib, A. Management of Charcoal Rot of Mungbean by Two Trichoderma Species and Dry Biomass of Coronopus didymus. Planta Daninha 2018, 36, e018182795. [Google Scholar] [CrossRef]
- Jabeen, N.; Javaid, A.; Shoaib, A.; Khan, I.H. Management of southern blight of bell pepper by soil amendment with dry biomass of Datura metel. J. Plant Pathol. 2021, 103, 901–913. [Google Scholar] [CrossRef]
- Sharf, W.; Javaid, A.; Shoaib, A.; Khan, I.H. Induction of resistance in chili against Sclerotium rolfsii by plant growth promoting rhizobacteria and Anagallis arvensis. Egypt. J. Biol. Pest Control. 2021, 31, 16. [Google Scholar] [CrossRef]
- Margaritopoulou, T.; Toufexi, E.; Kizis, D.; Balayiannis, G.; Anagnostopoulos, C.; Theocharis, A.; Rempelos, L.; Troyanos, Y.; Leifert, C.; Markellou, E. Reynoutria sachalinensis extract elicits SA-dependent defense responses in courgette genotypes against powdery mildew caused by Podosphaera xanthii. Sci. Rep. 2020, 10, 3354. [Google Scholar] [CrossRef]
- Teysssier, L.; Colussi, J.; Delemasure, S.; Chluba, J.; Wendehenne, D.; LaMotte, O.; Connat, J.-L. Inflammatory Effects of the Plant Protection Product Stifenia (FEN560) on Vertebrates. Front. Public Health 2017, 5, 74. [Google Scholar] [CrossRef] [PubMed]
- Angelini, R.M.D.M.; Rotolo, C.; Gerin, D.; Abate, D.; Pollastro, S.; Faretra, F. Global transcriptome analysis and differentially expressed genes in grapevine after application of the yeast-derived defense inducer cerevisane. Pest Manag. Sci. 2019, 75, 2020–2033. [Google Scholar] [CrossRef]
- Pavela, R.; Benelli, G. Essential Oils as Ecofriendly Biopesticides? Challenges and Constraints. Trends Plant Sci. 2016, 21, 1000–1007. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, P.; Dubey, N.; Shukla, A.K. Use of some essential oils as post-harvest botanical fungicides in the management of grey mould of grapes caused by Botrytis cinerea. World J. Microbiol. Biotechnol. 2007, 24, 39–46. [Google Scholar] [CrossRef]
- Pandey, A.K.; Kumar, P.; Singh, P.; Tripathi, N.N.; Bajpai, V.K. Essential Oils: Sources of Antimicrobials and Food Preservatives. Front. Microbiol. 2017, 7, 2161. [Google Scholar] [CrossRef] [PubMed]
- Viacava, G.E.; Goyeneche, R.; Goñi, M.G.; Roura, S.I.; Agüero, M.V. Natural elicitors as preharvest treatments to improve postharvest quality of Butterhead lettuce. Sci. Hortic. 2018, 228, 145–152. [Google Scholar] [CrossRef]
- Verdi, C.M.; Machado, V.S.; Machado, A.K.; Klein, B.; Bonez, P.C.; de Andrade, E.N.C.; Rossi, G.; Campos, M.M.; Wagner, R.; Sagrillo, M.R.; et al. Phytochemical characterization, genotoxicity, cytotoxicity, and antimicrobial activity of Gautheria procumbens essential oil. Nat. Prod. Res. 2020, 36, 1327–1331. [Google Scholar] [CrossRef] [PubMed]
- Vergnes, S.; Ladouce, N.; Fournier, S.; Ferhout, H.; Attia, F.; Dumas, B. Foliar treatments with Gaultheria procumbens essential oil induce defense responses and resistance against a fungal pathogen in Arabidopsis. Front. Plant Sci. 2014, 5, 477. [Google Scholar] [CrossRef]
- Jamiołkowska, A. Natural Compounds as Elicitors of Plant Resistance against Diseases and New Biocontrol Strategies. Agronomy 2020, 10, 173. [Google Scholar] [CrossRef]
- Kloepper, J.W.; Ryu, C.-M.; Zhang, S. Induced Systemic Resistance and Promotion of Plant Growth by Bacillus spp. Phytopathology 2004, 94, 1259–1266. [Google Scholar] [CrossRef] [PubMed]
- Narayanasamy, P. Biological Management of Diseases of Crops; Springer: Dordrecht, The Netherlands, 2013; 673p. [Google Scholar]
- Lastochkina, O.; Pusenkova, L.; Yuldashev, R.; Babaev, M.; Garipova, S.; Blagova, D.; Khairullin, R.; Aliniaeifard, S. Effects of Bacillus subtilis on some physiological and biochemical parameters of Triticum aestivum L. (wheat) under salinity. Plant Physiol. Biochem. 2017, 121, 80–88. [Google Scholar] [CrossRef]
- Giraud, R. Bacillus: Une Bactérie Auxilliaire Pour la Santé du Gazon; Clinique du Gazon: Saint-Verand, France, 2019. [Google Scholar]
- Stewart, A.; Hill, R. Applications of Trichoderma in Plant Growth Promotion. In Biotechnology and Biology of Trichoderma; Elsevier: Amsterdam, The Netherlands, 2014; pp. 415–428. [Google Scholar] [CrossRef]
- Mastouri, F.; Björkman, T.; Harman, G.E. Trichoderma harzianum Enhances Antioxidant Defense of Tomato Seedlings and Resistance to Water Deficit. Mol. Plant-Microbe Interact. 2012, 25, 1264–1271. [Google Scholar] [CrossRef]
- Morán-Diez, M.E.; Martínez de Alba, Á.E.; Rubio, M.B.; Hermosa, R.; Monte, E. Trichoderma and the Plant Heritable Priming Responses. J. Fungi 2021, 7, 318. [Google Scholar] [CrossRef]
- Karuppiah, V.; Sun, J.; Li, T.; Vallikkannu, M.; Chen, J. Co-cultivation of Trichoderma asperellum GDFS1009 and Bacillus amyloliquefaciens 1841 Causes Differential Gene Expression and Improvement in the Wheat Growth and Biocontrol Activity. Front Microbiol. 2019, 10, 1068. [Google Scholar] [CrossRef]
- Coutand, C. The Effect of Mechanical Stress on Plant Susceptibility to Pests: A Mini Opinion Review. Plants 2020, 9, 632. [Google Scholar] [CrossRef] [PubMed]
- L’Haridon, F.; Besson-Bard, A.; Binda, M.; Serrano, M.; Mansour, E.A.; Balet, F.; Schoonbeek, H.-J.; Hess, S.; Mir, R.; León, J.; et al. A Permeable Cuticle Is Associated with the Release of Reactive Oxygen Species and Induction of Innate Immunity. PLoS Pathog. 2011, 7, e1002148. [Google Scholar] [CrossRef] [PubMed]
- Orozco-Cardenas, M.; Ryan, C.A. Hydrogen peroxide is generated systemically in plant leaves by wounding and systemin via the octadecanoid pathway. Proc. Natl. Acad. Sci. USA 1999, 96, 6553–6557. [Google Scholar] [CrossRef]
- Mauch, F.; Kmecl, A.; Schaffrath, U.; Volrath, S.; Gorlach, J.; Ward, E.; Ryals, J.; Dudler, R. Mechanosensitive Expression of a Lipoxygenase Gene in Wheat. Plant Physiol. 1997, 114, 1561–1566. [Google Scholar] [CrossRef] [PubMed]
- Tretner, C.; Huth, U.; Hause, B. Mechanostimulation of Medicago truncatula leads to enhanced levels of jasmonic acid. J. Exp. Bot. 2008, 59, 2847–2856. [Google Scholar] [CrossRef]
- Mitchell, C.A.; Myers, P.N. Mechanical Stress Regulation of Plant Growth and Development. In Horticultural Reviews; Wiley: Hoboken, NJ, USA, 2010; Volume 17, pp. 1–42. [Google Scholar] [CrossRef]
- Turgeon, R.; Webb, J.A. Growth Inhibition by Mechanical Stress. Science 1971, 174, 961–962. [Google Scholar] [CrossRef]
- Börnke, F.; Rocksch, T. Thigmomorphogenesis—Control of plant growth by mechanical stimulation. Sci. Hortic. 2018, 234, 344–353. [Google Scholar] [CrossRef]
- Huché-Thélier, L.; Crespel, L.; Le Gourrierec, J.; Morel, P.; Sakr, S.; Leduc, N. Light signaling and plant responses to blue and UV radiations—Perspectives for applications in horticulture. Environ. Exp. Bot. 2016, 121, 22–38. [Google Scholar] [CrossRef]
- Ballaré, C.L. Light Regulation of Plant Defense. Annu. Rev. Plant Biol. 2014, 65, 335–363. [Google Scholar] [CrossRef]
- Urban, L.; Sari, D.C.; Orsal, B.; Lopes, M.; Miranda, R.; Aarrouf, J. UV-C light and pulsed light as alternatives to chemical and biological elicitors for stimulating plant natural defenses against fungal diseases. Sci. Hortic. 2018, 235, 452–459. [Google Scholar] [CrossRef]
- Lopes, M.M.; Silva, E.O.; Canuto, K.M.; Silva, L.M.; Gallão, M.I.; Urban, L.; Ayala-Zavala, J.F.; Miranda, M.R.A. Low fluence pulsed light enhanced phytochemical content and antioxidant potential of ‘Tommy Atkins’ mango peel and pulp. Innov. Food Sci. Emerg. Technol. 2016, 33, 216–224. [Google Scholar] [CrossRef]
- Filho, F.O.; Silva, E.D.O.; Lopes, M.M.D.A.; Ribeiro, P.R.V.; Oster, A.H.; Guedes, J.A.C.; Zampieri, D.D.S.; Bordallo, P.D.N.; Zocolo, G.J. Effect of pulsed light on postharvest disease control-related metabolomic variation in melon (Cucumis melo) artificially inoculated with Fusarium pallidoroseum. PLoS ONE 2020, 15, e0220097. [Google Scholar] [CrossRef]
- Wu, L.; Yang, H.-Q. CRYPTOCHROME 1 Is Implicated in Promoting R Protein-Mediated Plant Resistance to Pseudomonas syringae in Arabidopsis. Mol. Plant 2010, 3, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Jeong, R.-D.; Chandra-Shekara, A.C.; Barman, S.R.; Navarre, D.; Klessig, D.F.; Kachroo, A.; Kachroo, P. Cryptochrome 2 and phototropin 2 regulate resistance protein-mediated viral defense by negatively regulating an E3 ubiquitin ligase. Proc. Natl. Acad. Sci. USA 2010, 107, 13538–13543. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.; Zhang, M.; Lu, C.; Hettenhausen, C.; Tan, Q.; Cao, G.; Zhu, X.; Wu, G.; Wu, J. Ultraviolet-B enhances the resistance of multiple plant species to lepidopteran insect herbivory through the jasmonic acid pathway. Sci. Rep. 2018, 8, 277. [Google Scholar] [CrossRef]
- Escobar-Bravo, R.; Cheng, G.; Kim, H.K.; Grosser, K.; Van Dam, N.M.; Leiss, K.A.; Klinkhamer, P.G.L. Ultraviolet radiation exposure time and intensity modulate tomato resistance to herbivory through activation of jasmonic acid signaling. J. Exp. Bot. 2018, 70, 315–327. [Google Scholar] [CrossRef]
- Demkura, P.V.; Ballaré, C.L. UVR8 Mediates UV-B-Induced Arabidopsis Defense Responses against Botrytis cinerea by Controlling Sinapate Accumulation. Mol. Plant 2012, 5, 642–652. [Google Scholar] [CrossRef]
- Teramura, A.H.; Sullivan, J. Effects of UV-B radiation on photosynthesis and growth of terrestrial plants. Photosynth. Res. 1994, 39, 463–473. [Google Scholar] [CrossRef]
- Liu, J.; Stevens, C.; Khan, V.A.; Lu, J.Y.; Wilson, C.L.; Adeyeye, O.; Kabwe, M.K.; Pusey, P.L.; Chalutz, E.; Sultana, T.; et al. Application of Ultraviolet-C Light on Storage Rots and Ripening of Tomatoes. J. Food Prot. 1993, 56, 868–873. [Google Scholar] [CrossRef]
- Charles, M.T.; Goulet, A.; Arul, J. Physiological basis of UV-C induced resistance to Botrytis cinerea in tomato fruit. Postharvest Biol. Technol. 2008, 47, 41–53. [Google Scholar] [CrossRef]
- Charles, M.T.; Tano, K.; Asselin, A.; Arul, J. Physiological basis of UV-C induced resistance to Botrytis cinerea in tomato fruit. V. Constitutive defence enzymes and inducible pathogenesis-related proteins. Postharvest Biol. Technol. 2009, 51, 414–424. [Google Scholar] [CrossRef]
- Mercier, J.; Arul, J.; Julien, C. Effect of UV-C on Phytoalexin Accumulation and Resistance to Botrytis cinerea in Stored Carrots. J. Phytopathol. 1993, 139, 17–25. [Google Scholar] [CrossRef]
- Mercier, J.; Baka, M.; Reddy, B.; Corcuff, R.; Arul, J. Shortwave Ultraviolet Irradiation for Control of Decay Caused by Botrytis cinerea in Bell Pepper: Induced Resistance and Germicidal Effects. J. Am. Soc. Hortic. Sci. 2001, 126, 128–133. [Google Scholar] [CrossRef]
- Ouhibi, C.; Attia, H.; Rebah, F.; Msilini, N.; Chebbi, M.; Aarrouf, J.; Urban, L.; Lachaal, M. Salt stress mitigation by seed priming with UV-C in lettuce plants: Growth, antioxidant activity and phenolic compounds. Plant Physiol. Biochem. 2014, 83, 126–133. [Google Scholar] [CrossRef]
- Urban, L.; Charles, F.; de Miranda, M.R.A.; Aarrouf, J. Understanding the physiological effects of UV-C light and exploiting its agronomic potential before and after harvest. Plant Physiol. Biochem. 2016, 105, 1–11. [Google Scholar] [CrossRef]
- Aarrouf, J.; Urban, L. Flashes of UV-C light: An innovative method for stimulating plant defences. PLoS ONE 2020, 15, e0235918. [Google Scholar] [CrossRef]
- Aarrouf, J.; Goncalves, H.; Ledermann, L.; Matton, Y.; Lizzi, Y.; Junglee, S.; Suchail, S.; Urban, L. Simulation des défenses du fraisier par les flashs d’UV-C. Phytoma 2020, 732, 48–51. [Google Scholar]
- Ledermann, L.; Daouda, S.; Gouttesoulard, C.; Aarrouf, J.; Urban, L. Flashes of UV-C Light Stimulate Defenses of Vitis vinifera L. ‘Chardonnay’ Against Erysiphe necator in Greenhouse and Vineyard Conditions. Plant Dis. 2021, 105, 2106–2113. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Van der Does, D.; Zamioudis, C.; Leon-Reyes, A.; Van Wees, S.C.M. Hormonal Modulation of Plant Immunity. Annu. Rev. Cell Dev. Biol. 2012, 28, 489–521. [Google Scholar] [CrossRef]
- Dufour, M.C.; Lambert, C.; Bouscaut, J.; Mérillon, J.M.; Corio-Costet, M.F. Benzothiadiazole-primed defence responses and enhanced differential expression of defence genes in Vitis vinifera infected with biotrophic pathogens Erysiphe necator and Plasmopara viticola. Plant Pathol. 2012, 62, 370–382. [Google Scholar] [CrossRef]
- Banani, H.; Roatti, B.; Ezzahi, B.; Giovannini, O.; Gessler, G.; Pertot, I.; Perazzolli, M. Characterization of resistance mechanisms activated by Trichoderma harzianum T39 and benzothiadiazole to downy mildew in different grapevine cultivars. Plant Pathol. 2013, 63, 334–343. [Google Scholar] [CrossRef]
- Bellée, A.; Cluzet, S.; Dufour, M.-C.; Mérillon, J.-M.; Corio-Costet, M.-F. Comparison of the Impact of Two Molecules on Plant Defense and on Efficacy against Botrytis cinerea in the Vineyard: A Plant Defense Inducer (Benzothiadiazole) and a Fungicide (Pyrimethanil). J. Agric. Food Chem. 2018, 66, 3338–3350. [Google Scholar] [CrossRef] [PubMed]
- Guerreiro, A.; Figueiredo, J.; Silva, M.S.; Figueiredo, A. Linking Jasmonic Acid to Grapevine Resistance against the Biotrophic Oomycete Plasmopara viticola. Front. Plant Sci. 2016, 7, 565. [Google Scholar] [CrossRef]
- Belhadj, A.; Telef, N.; Cluzet, S.; Bouscaut, J.; Corio-Costet, M.-F.; Mérillon, J.-M. Ethephon Elicits Protection against Erysiphe necator in Grapevine. J. Agric. Food Chem. 2008, 56, 5781–5787. [Google Scholar] [CrossRef] [PubMed]
- Park, K.; Park, J.-W.; Lee, S.-W.; Balaraju, K. Disease suppression and growth promotion in cucumbers induced by integrating PGPR agent Bacillus subtilis strain B4 and chemical elicitor ASM. Crop. Prot. 2013, 54, 199–205. [Google Scholar] [CrossRef]
- El-Rahman, S.S.A.; Mohamed, H.I. Application of benzothiadiazole and Trichoderma harzianum to control faba bean chocolate spot disease and their effect on some physiological and biochemical traits. Acta Physiol. Plant. 2013, 36, 343–354. [Google Scholar] [CrossRef]
- De Jong, H.; Reglinski, T.; Elmer, P.A.G.; Wurms, K.; Vanneste, J.L.; Guo, L.F.; Alavi, M. Integrated Use of Aureobasidium pullulans Strain CG163 and Acibenzolar-S-Methyl for Management of Bacterial Canker in Kiwifruit. Plants 2019, 8, 287. [Google Scholar] [CrossRef]
- Reuveni, M.; Sheglov, D.; Cohen, Y. Control of Moldy-Core Decay in Apple Fruits by β-Aminobutyric Acids and Potassium Phosphites. Plant Dis. 2003, 87, 933–936. [Google Scholar] [CrossRef]
- Walters, D.R.; Havis, N.D.; Sablou, C.; Walsh, D.J. Possible trade-off associated with the use of a combination of resistance elicitors. Physiol. Mol. Plant Pathol. 2011, 75, 188–192. [Google Scholar] [CrossRef]
- Singh, U.B.; Malviya, D.; Singh, S.; Kumar, M.; Sahu, P.K.; Singh, H.V.; Kumar, S.; Roy, M.; Imran, M.; Rai, J.P.; et al. Trichoderma harzianum- and Methyl Jasmonate-Induced Resistance to Bipolaris sorokiniana through Enhanced Phenylpropanoid Activities in Bread Wheat (Triticum aestivum L.). Front. Microbiol. 2019, 10, 1697. [Google Scholar] [CrossRef]
- Zehra, A.; Meena, M.; Dubey, M.K.; Aamir, M.; Upadhyay, R.S. Synergistic effects of plant defense elicitors and Trichoderma harzianum on enhanced induction of antioxidant defense system in tomato against Fusarium wilt disease. Bot. Stud. 2017, 58, 44. [Google Scholar] [CrossRef] [PubMed]
- Langa-Lomba, N.; Buzón-Durán, L.; Martín-Ramos, P.; Casanova-Gascón, J.; Martín-Gil, J.; Sánchez-Hernández, E.; González-García, V. Assessment of Conjugate Complexes of Chitosan and Urtica dioica or Equisetum arvense Extracts for the Control of Grapevine Trunk Pathogens. Agronomy 2021, 11, 976. [Google Scholar] [CrossRef]
- Gómez-Merino, F.C.; Trejo-Téllez, L.I. Biostimulant activity of phosphite in horticulture. Sci. Hortic. 2015, 196, 82–90. [Google Scholar] [CrossRef]
- Nojosa, G.B.A.; Resende, M.L.V.; Barguil, B.M.; Moraes, S.A.G.; Vilas Boas, C.H. Effect of resistance inducers on coffee against Phoma leaf spot. Summa Phytopathol. 2009, 35, 60–62. [Google Scholar] [CrossRef]
- Ogoshi, C.; de Abreu, M.S.; da Silva, B.M.; Neto, H.S.; Junior, P.M.R.; de Resende, M.L.V. Potassium phosphite: A promising product in the management of diseases caused by Colletotrichum gloeosporioides in coffee plants. Biosci. J. 2013, 29, 1558–1565. [Google Scholar]
- Monteiro, A.C.A.; de Resende, M.L.V.; Valente, T.C.T.; Junior, P.M.R.; Pereira, V.F.; da Costa, J.R.; da Silva, J.A.G. Manganese Phosphite in Coffee Defence against Hemileia vastatrix, the Coffee Rust Fungus: Biochemical and Molecular Analyses. J. Phytopathol. 2016, 164, 1043–1053. [Google Scholar] [CrossRef]
- Lobato, M.C.; Olivieri, F.P.; Altamiranda, E.A.G.; Wolski, E.A.; Daleo, G.R.; Caldiz, D.O.; Andreu, A.B. Phosphite compounds reduce disease severity in potato seed tubers and foliage. Eur. J. Plant Pathol. 2008, 122, 349–358. [Google Scholar] [CrossRef]
- Achary, V.M.M.; Ram, B.; Manna, M.; Datta, D.; Bhatt, A.; Reddy, M.K.; Agrawal, P.K. Phosphite: A novel P fertilizer for weed management and pathogen control. Plant Biotechnol. J. 2017, 15, 1493–1508. [Google Scholar] [CrossRef]
- Diánez, F.; Santos, M.; Parra, C.; Navarro, M.; Blanco, R.; Gea, F. Screening of antifungal activity of 12 essential oils against eight pathogenic fungi of vegetables and mushroom. Lett. Appl. Microbiol. 2018, 67, 400–410. [Google Scholar] [CrossRef]
- Trebbi, G.; Negri, L.; Bosi, S.; Dinelli, G.; Cozzo, R.; Marotti, I. Evaluation of Equisetum arvense (Horsetail Macerate) as a Copper Substitute for Pathogen Management in Field-Grown Organic Tomato and Durum Wheat Cultivations. Agriculture 2020, 11, 5. [Google Scholar] [CrossRef]
- Misato, T.; Homma, Y.; Kō, K. The development of a natural fungicide, soybean lecithin. Eur. J. Plant Pathol. 1977, 83, 395–402. [Google Scholar] [CrossRef]
- Homma, Y.; Takahashi, H.; Arimoto, Y. Studies on the Mode of Action of Soybean Lecithin. Part 3. Effects on the Infection Process of Rice Blast Fungus, Pyricularia oryzae. Jpn. J. Phytopathol. 1992, 58, 514–521. [Google Scholar] [CrossRef]
- Kang, B.R.; Park, J.S.; Jung, W.-J. Antiviral activity by lecithin-induced fengycin lipopeptides as a potent key substrate against Cucumber mosaic virus. Microb. Pathog. 2021, 155, 104910. [Google Scholar] [CrossRef] [PubMed]
- Salgado, M.; Rodríguez-Rojo, S.; Alves-Santos, F.M.; Cocero, M.J. Encapsulation of resveratrol on lecithin and β-glucans to enhance its action against Botrytis cinerea. J. Food Eng. 2015, 165, 13–21. [Google Scholar] [CrossRef]
- Ma, Z.; Garrido-Maestu, A.; Jeong, K.C. Application, mode of action, and in vivo activity of chitosan and its micro-and nanoparticles as antimicrobial agents: A review. Carbohydr. Polym. 2017, 176, 257–265. [Google Scholar] [CrossRef]
- Mahendran, R.; Ramanan, K.R.; Barba, F.J.; Lorenzo, J.M.; López-Fernández, O.; Munekata, P.E.S.; Roohinejad, S.; Sant’Ana, A.S.; Tiwari, B.K. Recent advances in the application of pulsed light processing for improving food safety and increasing shelf life. Trends Food Sci. Technol. 2019, 88, 67–79. [Google Scholar] [CrossRef]
- Bintsis, T.; Litopoulou-Tzanetaki, E.; Robinson, R.K. Existing and potential applications of ultraviolet light in the food industry—A critical review. J. Sci. Food Agric. 2000, 80, 637–645. [Google Scholar] [CrossRef]
- Jay, J.M.; Loessner, M.A.J.; Golden, D.A. Modern Food Microbiology; Springer Science and Business Media: New York, NY, USA, 2008. [Google Scholar]
- Wuytack, E.Y.; Phuong, L.D.T.; Aertsen, A.; Reyns, K.M.F.; Marquenie, D.; De Ketelaere, B.; Masschalck, B.; Van Opstal, I.; Diels, A.M.J.; Michiels, C.W. Comparison of Sublethal Injury Induced in Salmonella enterica Serovar Typhimurium by Heat and by Different Nonthermal Treatments. J. Food Prot. 2003, 66, 31–37. [Google Scholar] [CrossRef]
- Santos, A.L.; Oliveira, V.; Baptista, I.; Henriques, I.; Gomes, N.C.M.; Almeida, A.; Correia, A.; Cunha, Â. Wavelength dependence of biological damage induced by UV radiation on bacteria. Arch. Microbiol. 2012, 195, 63–74. [Google Scholar] [CrossRef]
- Vanhaelewyn, L.; Van Der Straeten, D.; De Coninck, B.; Vandenbussche, F. Ultraviolet Radiation From a Plant Perspective: The Plant-Microorganism Context. Front. Plant Sci. 2020, 11, 597642. [Google Scholar] [CrossRef]
- Patel, J.S.; Radetsky, L.C.; Nagare, R.; Rea, M.S. Nighttime Application of UV-C to Control Cucumber Powdery Mildew. Plant Health Prog. 2020, 21, 40–46. [Google Scholar] [CrossRef]
- Onofre, R.B.; Gadoury, D.M.; Stensvand, A.; Bierman, A.; Rea, M.; Peres, N.A. Use of Ultraviolet Light to Suppress Powdery Mildew in Strawberry Fruit Production Fields. Plant Dis. 2021, 105, 2402–2409. [Google Scholar] [CrossRef] [PubMed]
- Kanwar, P.; Jha, G. Alterations in plant sugar metabolism: Signatory of pathogen attack. Planta 2018, 249, 305–318. [Google Scholar] [CrossRef] [PubMed]
- Calonnec, A.; Jolivet, J.; Vivin, P.; Schnee, S. Pathogenicity Traits Correlate with the Susceptible Vitis vinifera Leaf Physiology Transition in the Biotroph Fungus Erysiphe necator: An Adaptation to Plant Ontogenic Resistance. Front. Plant Sci. 2018, 9, 1808. [Google Scholar] [CrossRef]
- Lecompte, F.; Nicot, P.C.; Ripoll, J.; Abro, M.A.; Raimbault, A.K.; Lopez-Lauri, F.; Bertin, N. Reduced susceptibility of tomato stem to the necrotrophic fungus Botrytis cinerea is associated with a specific adjustment of fructose content in the host sugar pool. Ann. Bot. 2017, 119, 931–943. [Google Scholar] [CrossRef]
- Herms, D.A.; Mattson, W.J. The Dilemma of Plants: To Grow or Defend. Q. Rev. Biol. 1992, 67, 283–335. [Google Scholar] [CrossRef]
- Walters, D.; Walsh, D.; Newton, A.; Lyon, G. Induced Resistance for Plant Disease Control: Maximizing the Efficacy of Resistance Elicitors. Phytopathology 2005, 95, 1368–1373. [Google Scholar] [CrossRef]
- Walters, D.R.; Ratsep, J.; Havis, N. Controlling crop diseases using induced resistance: Challenges for the future. J. Exp. Bot. 2013, 64, 1263–1280. [Google Scholar] [CrossRef]
- Van Hulten, M.; Pelser, M.; van Loon, L.C.; Pieterse, C.M.J.; Ton, J. Costs and benefits of priming for defense in Arabidopsis. Proc. Natl. Acad. Sci. USA 2006, 103, 5602–5607. [Google Scholar] [CrossRef]
- Poiroux-Gonord, F.; Santini, J.; Fanciullino, A.-L.; Lopez-Lauri, F.; Giannettini, J.; Sallanon, H.; Berti, L.; Urban, L. Metabolism in orange fruits is driven by photooxidative stress in the leaves. Physiol. Plant. 2013, 149, 175–187. [Google Scholar] [CrossRef]
- Wang, F.; Sanz, A.; Brenner, M.L.; Smith, A. Sucrose Synthase, Starch Accumulation, and Tomato Fruit Sink Strength. Plant Physiol. 1993, 101, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Aslani, L.; Gholami, M.; Mobli, M.; Sabzalian, M.R. The influence of altered sink-source balance on the plant growth and yield of greenhouse tomato. Physiol. Mol. Biol. Plants 2020, 26, 2109–2123. [Google Scholar] [CrossRef] [PubMed]
- Pawar, R.; Rana, V.S. Manipulation of Source-Sink Relationship in Pertinence to Better Fruit Quality and Yield in Fruit Crops: A Review. Agric. Rev. 2019, 40, 200–207. [Google Scholar] [CrossRef]
- Lasa, B.; Menendez, S.; Sagastizabal, K.; Cervantes, M.E.C.; Irigoyen, I.; Muro, J.; Aparicio-Tejo, P.M.; Ariz, I. Foliar application of urea to “Sauvignon Blanc” and “Merlot” vines: Doses and time of application. Plant Growth Regul. 2012, 67, 73–81. [Google Scholar] [CrossRef]
- Gutiérrez-Gamboa, G.; Garde-Cerdán, T.; Gonzalo-Diago, A.; Moreno-Simunovic, Y.; Martínez-Gil, A.M. Effect of different foliar nitrogen applications on the must amino acids and glutathione composition in Cabernet Sauvignon vineyard. LWT Food Sci. Technol. 2017, 75, 147–154. [Google Scholar] [CrossRef]
- Gutiérrez-Gamboa, G.; Romanazzi, G.; Garde-Cerdán, T.; Pérez-Álvarez, E.P. A review of the use of biostimulants in the vineyard for improved grape and wine quality: Effects on prevention of grapevine diseases. J. Sci. Food Agric. 2018, 99, 1001–1009. [Google Scholar] [CrossRef]
- Massi, F.; Torriani, S.F.F.; Borghi, L.; Toffolatti, S.L. Fungicide Resistance Evolution and Detection in Plant Pathogens: Plasmopara viticola as a Case Study. Microorganisms 2021, 9, 119. [Google Scholar] [CrossRef]
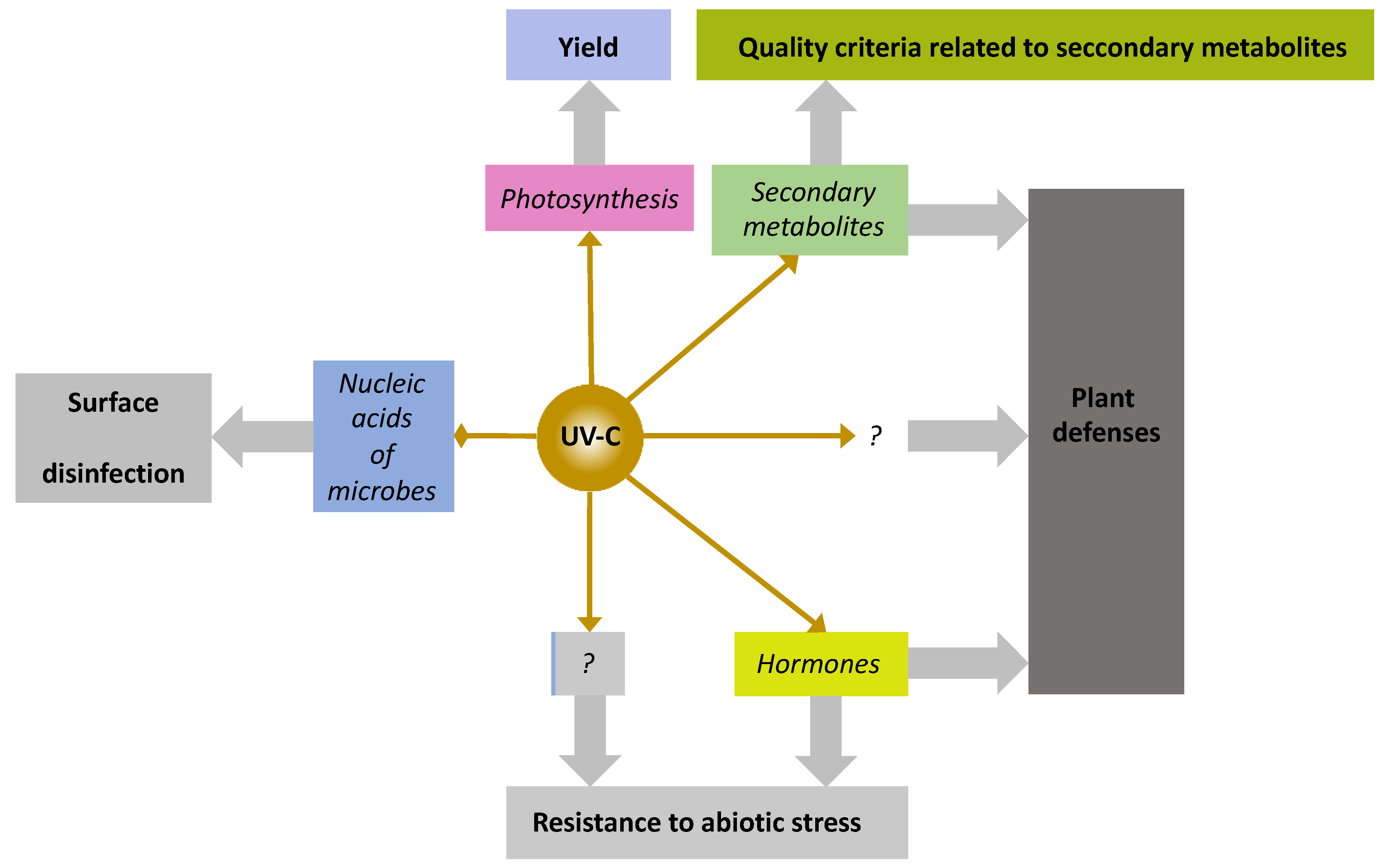
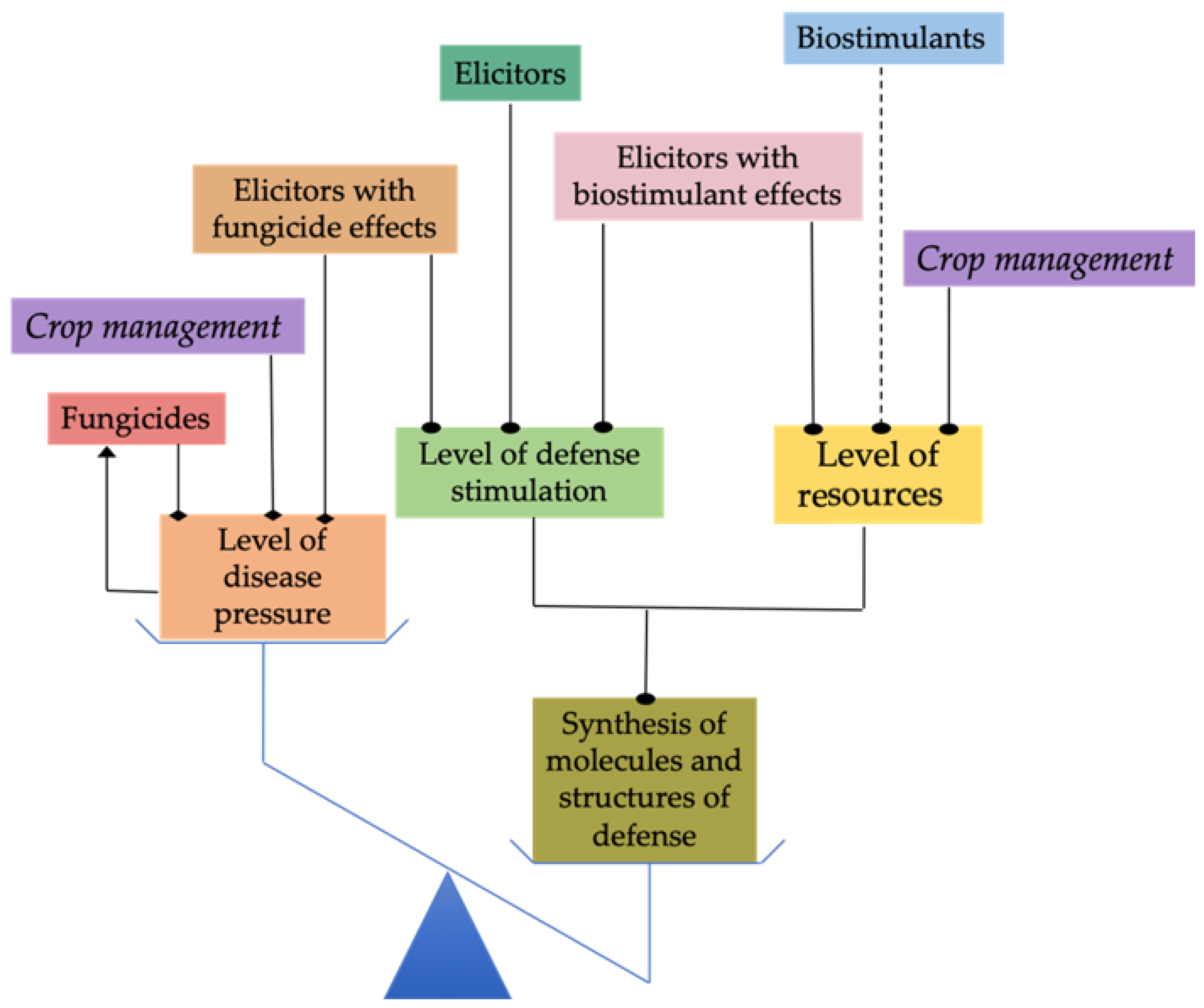
| Crop | Stress | Reference |
|---|---|---|
| Barley | High cadmium | [25] |
| Common bean | High cadmium | [26] |
| Cucumber | Cold | [24] |
| Grapevine | High salinity | [27] |
| Maize | Drought | [28] |
| Maize | High cadmium | [17] |
| Maize | High salinity | [29] |
| Pepper | High salinity | [30] |
| Strawberry | Cold | [31] |
| Wheat | Drought | [32] |
| Wheat | High temperature | [33] |
| Name | Category | SA Pathway | JA/ET Pathway | Fungicide Effect | Effect on Growth |
|---|---|---|---|---|---|
| ASM | Chemical elicitor | yes | yes | not documented | generally negative |
| Cerevisane | Chemical elicitor of natural origin | yes | yes | not documented | not documented |
| Chitin and chitosan | Chemical elicitor of natural origin | yes | yes | yes | positive |
| COS-OGA | Chemical elicitor of natural origin | yes | no | not documented | no negative effects reported |
| Essential oils | Chemical elicitor of natural origin | probable | not documented | some EOs are effective | not documented |
| Flashes of UV-C light | Physical elicitor | yes | (yes) | yes | no negative effect in the absence of stress |
| Laminarin/ PS3 | Chemical elicitor of natural origin | after inoculation | yes | not documented | positive in conditions of stress |
| Mechanical stress | Physical elicitor | unknown | yes | no | potentially negative |
| Phosphites | Chemical elicitor | indirectly | yes | yes | potentially negative |
| Trichoderma spp. | Biological elicitor | yes | yes | yes | positive, plays a role in the defense/growth balance |
| UV-B light | Physical elicitor | not documented | yes | yes | negative |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urban, L.; Lauri, F.; Ben Hdech, D.; Aarrouf, J. Prospects for Increasing the Efficacy of Plant Resistance Inducers Stimulating Salicylic Acid. Agronomy 2022, 12, 3151. https://doi.org/10.3390/agronomy12123151
Urban L, Lauri F, Ben Hdech D, Aarrouf J. Prospects for Increasing the Efficacy of Plant Resistance Inducers Stimulating Salicylic Acid. Agronomy. 2022; 12(12):3151. https://doi.org/10.3390/agronomy12123151
Chicago/Turabian StyleUrban, Laurent, Félicie Lauri, Douae Ben Hdech, and Jawad Aarrouf. 2022. "Prospects for Increasing the Efficacy of Plant Resistance Inducers Stimulating Salicylic Acid" Agronomy 12, no. 12: 3151. https://doi.org/10.3390/agronomy12123151
APA StyleUrban, L., Lauri, F., Ben Hdech, D., & Aarrouf, J. (2022). Prospects for Increasing the Efficacy of Plant Resistance Inducers Stimulating Salicylic Acid. Agronomy, 12(12), 3151. https://doi.org/10.3390/agronomy12123151






