1. Introduction
Crown rot and common root rot are important soil-borne diseases of wheat (
Triticum aestivum L.) and barley (
Hordeum vulgare L.) worldwide, including countries such as Australia, Canada, China, New Zealand, North Africa, South Africa, and the USA [
1,
2]. In Australia, annual crown rot and common root rot losses in wheat and barley were estimated at 97 M AUD and 43 M AUD, respectively [
3,
4].
Crown rot has been associated with several
Fusarium species including
F. pseudograminearum, F. culmorum, F. graminearum, F. avenaceum, F. crookwellense, and
F. poae [
5,
6,
7,
8]. In Australia,
F. pseudograminearum and
F. culmorum are the predominant pathogens causing crown rot in barley and wheat [
6,
9,
10].
Fusarium pseudograminearum occurs across all wheat growing regions and can be severe under the hot, dry conditions found in Australia [
8,
9].
Fusarium culmorum is more widely distributed in high rainfall temperate regions of Victoria (VIC), South Australia, and in the summer rainfall regions of the northern region [eastern Darling Downs in southern Queensland (QLD)] [
6,
9]. In Australia,
F. graminearum (
F. graminearum sensu stricto) has been reported to cause localised Fusarium head blight epidemics in subtropical areas with high summer rainfall [
9]. Although
F. graminearum has been associated with crown rot under wet conditions in Australia, it has not been considered a significant pathogen to date [
10,
11].
Bipolaris sorokiniana is both a root and foliar pathogen causing common root rot, seedling blight, and spot blotch [
12,
13]. This fungus occurs in most regions where barley and wheat are grown [
13]. Common root rot is widespread and causes the most damage to plants grown in warmer areas with high rainfall at the beginning of the growing season and water-stress conditions at the end of the season [
14].
Both
B. sorokiniana and
Fusarium species can affect the sub-crown internode (SCI), crown, leaf base, coleoptile, and roots of plants [
1,
2,
8,
15]. The first symptoms of crown rot and common root rot are lesions on the coleoptile, SCI, and leaf sheaths, followed by brown discolouration along the basal stem tissues. These crown rot and common root rot symptoms are difficult to distinguish, however, discolouration caused by
B. sorokiniana does not extend as high up the stem as lesions caused by
F. pseudograminearum [
8,
16]. During severe crown rot infection, visual symptoms progress up the stem, sometimes as high as the fifth node [
8]. Lesions caused by
B. sorokiniana are usually dark brown to black [
17] compared to the honey brown coloured lesions typical of crown rot infections [
8,
18]. At flowering,
Fusarium infected stems may undergo premature senescence, resulting in whiteheads containing no or shrivelled grain [
1,
8,
19]. No reports are suggesting that
B. sorokiniana is associated with whiteheads [
20]. A positive correlation has been reported between the severity of infection, yield loss and the severity of discolouration of the SCI caused by
B. sorokiniana for wheat cultivars [
12,
15]. The SCI tissue discolouration has been adopted as a standard method to measure common root rot severity, while stem tissue browning has been used to measure crown rot disease [
15,
18,
21,
22].
Crown rot and common root rot pathogens have been associated with an extensive range of hosts including barley, bread wheat, durum wheat (
Triticum turgidum L. var.
durum), oat (
Avena sativa L.), rye (
Secale cereale L.), maize (
Zea mays L.), sorghum (
Sorghum bicolor L. Moench), and triticale (×
Triticosecale Wittmack) [
1,
2,
8,
23]. The incidence and severity of these diseases can vary significantly within the host, field, and season [
2,
8,
24]. The importance of barley and wheat associated with
F. pseudograminearum is clear. However, other host–pathogen associations in Australian cropping systems are unclear.
Crown rot research in Australia has focussed on
F. pseudograminearum as the dominant pathogen due to the wide distribution of the
F. pseudograminearum [
6,
8]. Most of the bread wheat cultivars and all of the durum wheat cultivars are very susceptible to susceptible to
F. pseudograminearum [
18,
24,
25,
26]. Barley cultivars exhibit a high level of disease severity to crown rot caused by
F. pseudograminearum, similar or greater than bread wheat cultivars [
18,
27,
28,
29]. However, estimated yield losses of barley are less than those in bread wheat and the formation of whiteheads is less common [
30].
Bipolaris sorokiniana can cause a range of common root rot reactions depending on the host [
12,
15,
25]. In Australia, bread and durum wheat cultivars range from susceptible to moderately resistant [
31,
32], and barley cultivars range from susceptible to moderately susceptible to common root rot. [
32,
33].
Oats have been shown to exhibit a low level of brown discolouration when they are inoculated with crown rot (
F. pseudograminearum and
F. culmorum) and common root rot pathogens [
18,
28,
29,
34]. Collins et al. [
35] reported that oats are more susceptible to
F. culmorum than
F. pseudograminearum. However, the role of oat cultivars as a rotation crop to crown rot and common root rot is not clear.
Long-term management and control of crown rot and common root rot pathogens can be difficult as the fungus can survive in stubble and soil for several years [
8,
36]. Stubble is managed in the field using incorporation by disc cultivators that invert the soil and surface residue and stubble retention on the soil surface with minimum tillage [
8,
36]. Good tillage and removal of stubble reduce crown rot and common root rot inoculum levels [
36,
37], however, this practice may have a negative effect on crop yield by removing moisture that would otherwise be available to the crop [
8]. Crop rotation is an effective method for crown rot management to reduce the levels of inoculum in the field [
1,
8]. Non-host cereal crops such as chickpea, canola, and sorghum can be efficient at reducing crown rot in subsequent plantings with at least two years of non-host crops [
8,
22].
Glasshouse studies examining seedling reactions of a range of cereals to crown rot and common root rot pathogens have reported variation in the visual discolouration and the host response among the hosts for both diseases [
28,
29]. Disease reactions in the field are an essential extension of this research and will assist in providing a robust understanding of winter cereal host responses to crown rot and common root rot pathogens. In Australia, only one study has compared the visual discolouration and the yield loss of wheat cultivars infected with a single strain each of
F. pseudograminearum and
F. culmorum in field trials [
38]. It was reported that
F. culmorum caused the same or less visual discolouration on wheat cultivars as
F. pseudograminearum. In Australia,
F. graminearum has not frequently been associated with crown rot [
11]. However, with the predicted climate changes [
39],
F. graminearum could become a significant pathogen causing crown rot on a range of winter cereals.
The primary aim of the current study was to determine the comparative disease-causing capabilities of the four crown rot and common root rot pathogen species across commercially grown cultivars of barley, bread wheat, durum wheat, oat, and triticale grown under field conditions. These crops were inoculated with strains of F. pseudograminearum, F. culmorum, F. graminearum, and B. sorokiniana in two field trials. Cultivars and pathogens were compared for visual disease severity on SCI and stem tissues of inoculated and non-inoculated treatments. The impact of each pathogen on host cultivars was further compared by reporting differences in plant height and plant dry weight at tillering, flowering, and maturity. This research is the first detailed comparative study of the pathogen impacts on different hosts, which may help to improve crown rot and common root rot resistance breeding. The comparison of cereal reactions to these pathogens in the field will provide valuable information for management strategies in crop rotation targeting crown rot and common root rot diseases.
2. Materials and Methods
2.1. Strains and Inoculum Preparation
Colonised wheat/barley grain inoculum was prepared for two strains each of
F. pseudograminearum [
Fp1, BRIP no. (Queensland Plant Pathology Herbarium number) 64949 and
Fp2, BRIP no. 64952],
F. graminearum (
Fg1, BRIP no. 64975 and
Fg2, BRIP no. 64976)
, F. culmorum (
Fc1, BRIB no. 64973 and
Fc2, BRIP no. 64974)
, and
B. sorokiniana (
Bs1, BRIP no. 64970 and
Bs2, BRIP no. 64972). Further details of inoculum production and isolates used in this study are presented in Saad et al. [
29]. Inoculum was stored at 4 ℃ and ground to pass through a 2 mm sieve prior to application in the field.
2.2. Field Site Characteristics
Field trials were conducted in 2016 and 2017 at the Wellcamp Field Station, Department of Agriculture and Fisheries, Queensland, Australia (coordinates: 27°33′54″ S 151°51′51″ E). The soil at the site was a self-mulching black Vertisol of the Irving clay soil association (Australian black earth) [
40]. In each year, urea (100 kg N/ha) was applied three weeks before sowing at a depth of 50 mm. In 2017, 40 mL of water was applied with overhead irrigation eight weeks after planting.
In both years, DNA levels of important soilborne pathogens, including
B. sorokiniana,
F. culmorum, F. graminearum, and
F. pseudograminearum, were determined with a PreDicta B full crop test provided by South Australian Research and Development Institute (SARDI) for soil and stubble borne disease DNA detection [
38]. Three soil samples were taken from the site pre-planting and twenty soil samples were taken from the non-inoculated controls at post-harvest for both years using an Accucore sampler at a depth of 30 cm (15 to 20 cores were included for each sample).
In both years, trials were planted in July and plants at the tillering (Zadoks growth stage GS20–29) and flowering stages (Zadoks growth stage GS60–69) were harvested in September and November, respectively. Mature plants (Zadoks growth stage GS90–99) were harvested in December of each year.
The minimum and maximum temperature and rainfall were recorded for both years from the Toowoomba Airport station, 8.1 km from the site of the trials (site number 041529, coordinates: Latitude: 27.54° S; Longitude: 151.91° E).
2.3. Experimental Design
Both trials were conducted as randomised completed block designs, where each treatment (combination of harvest time, pathogen, strain, and cultivar) was randomly allocated to a plot within each replicate block (three replicates for each harvest time). Five cultivars [barley (cv. Grimmett), bread wheat (cv. Livingston), durum wheat (cv. Hyperno), oat (cv. Genie), and triticale (cv. Endeavour)] were exposed to the four crown rot and common root rot pathogens. Two individual strains were used for each pathogen. In both years, twenty seeds were sown in 1 m rows, then covered with soil. In 2016, treatments consisted of individual plots inoculated with individual strains of each pathogen species. In 2017, the individual strains were combined to inoculate each plot. Control treatments consisted of non-inoculated plots for each trial. Two grams of ground colonised grain inoculum were applied on top of a layer of soil above the seed along the 1 m furrow at planting. In 2016 seed was hand planted into furrows. In 2017 the seed was machine planted. Inoculum was applied manually in both years to avoid cross contamination. Plants from three replicates of each treatment were harvested at the three harvest times, tillering, flowering, and maturity.
For both trials, harvest at tillering was conducted nine weeks after planting. The flowering harvest varied depending on each host. Bread and durum wheat cultivars were harvested at 13 weeks and barley, oat, and triticale cultivars at 15 weeks after planting. Mature plants were harvested at 17 weeks after planting for barley, bread and durum wheat cultivars, and 18 weeks after planting for oat and triticale cultivars.
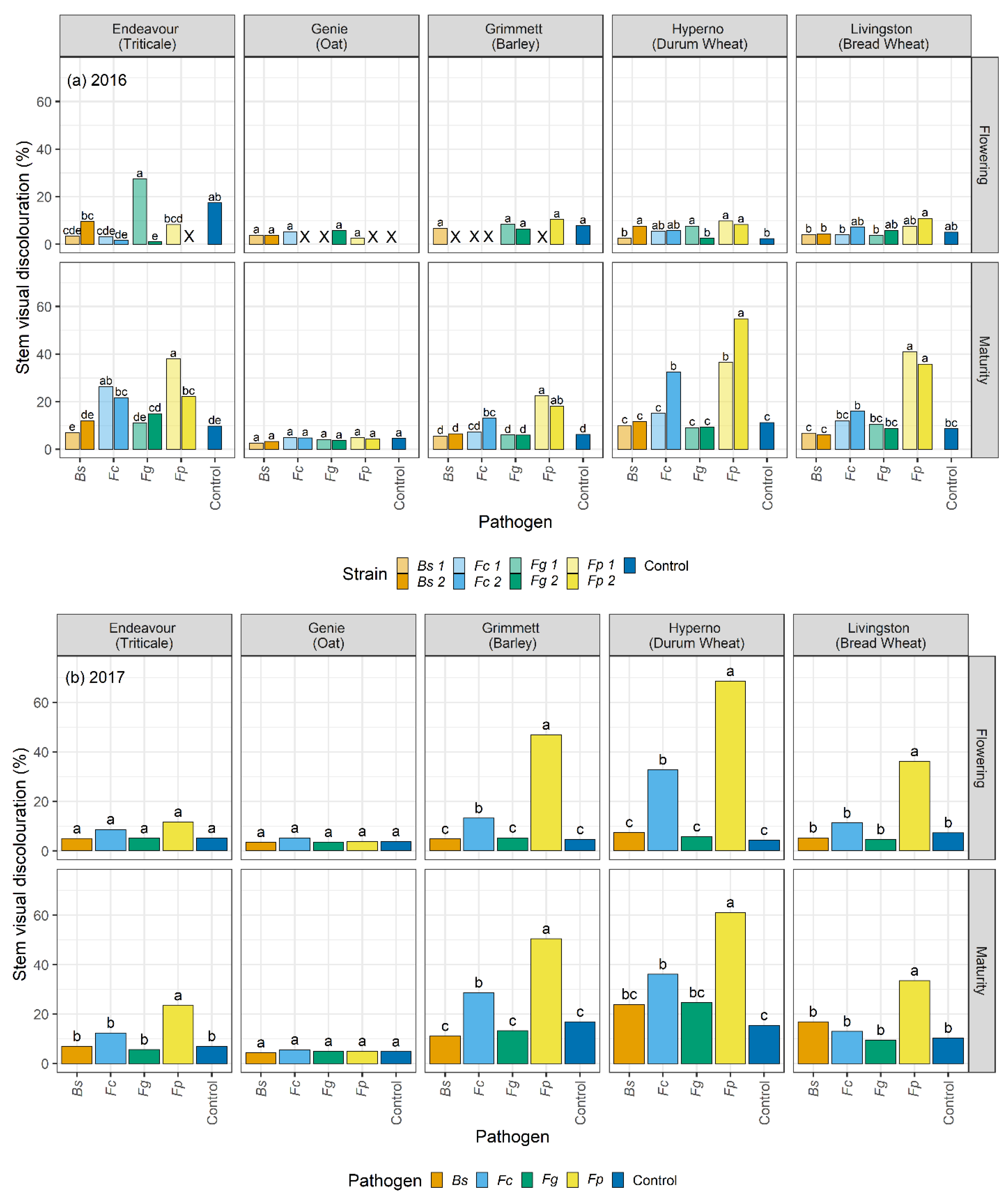
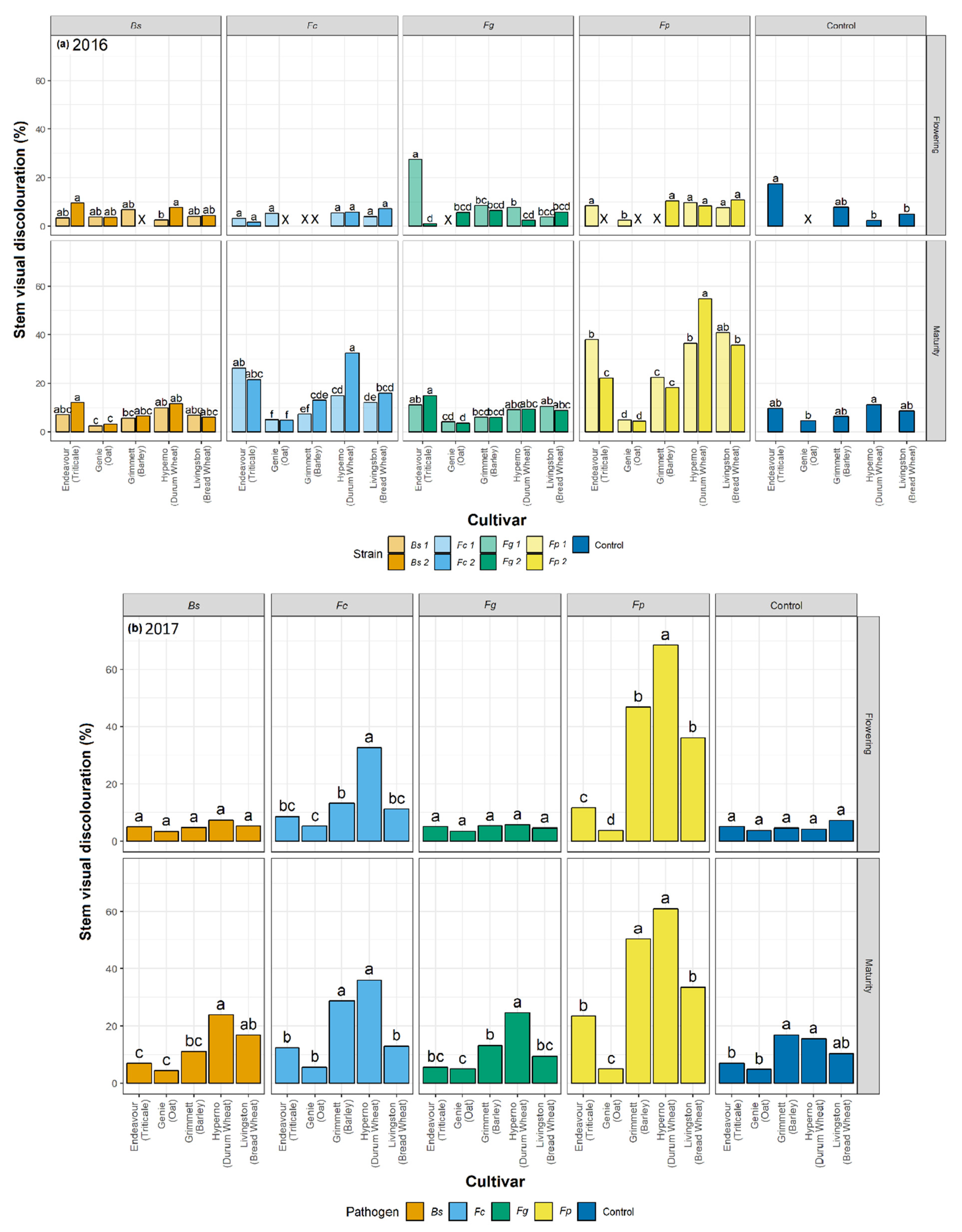
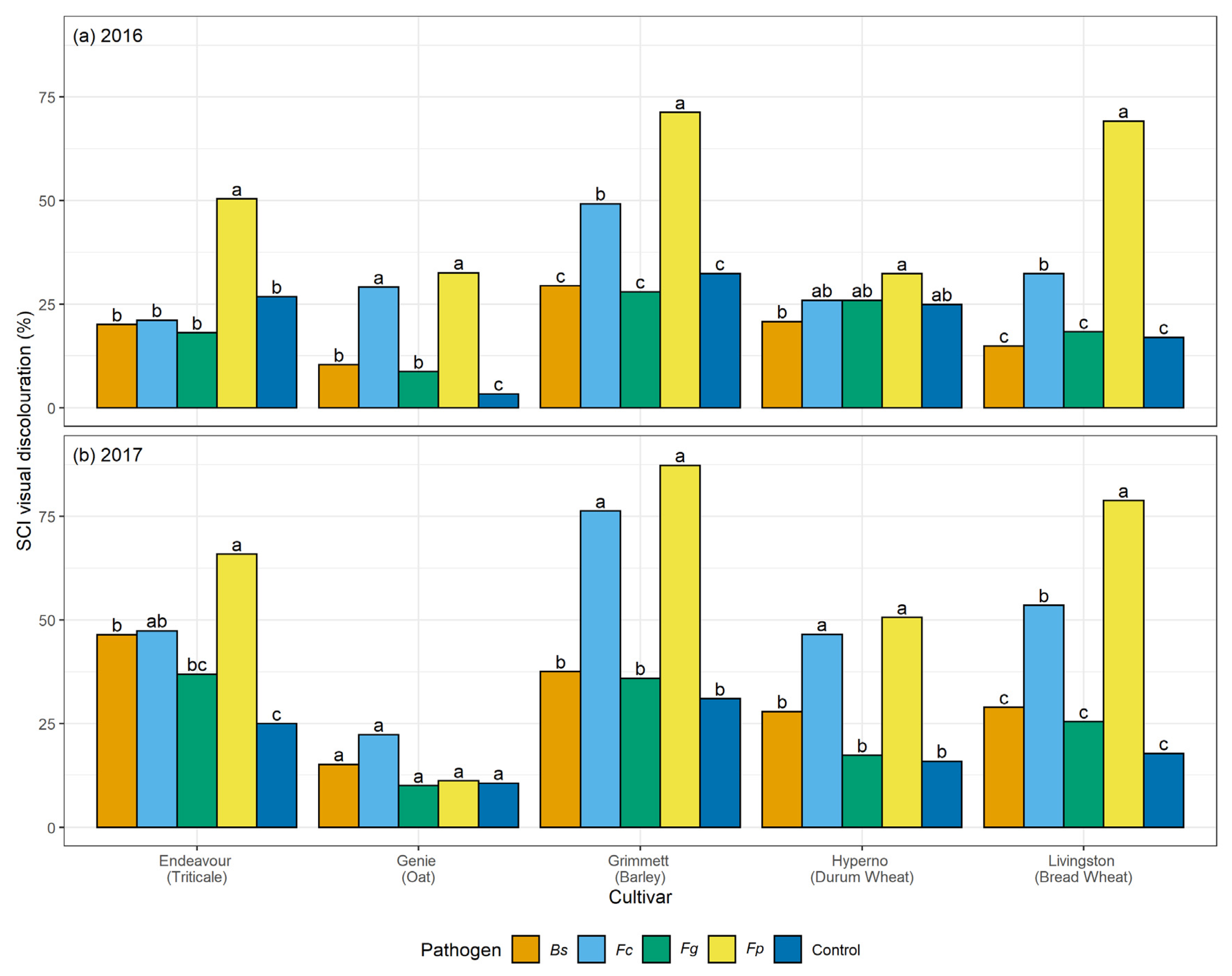
2.4. Visual Discolouration Rating and Physiological Response
At each harvest, ten plants were collected from each plot. At tillering, flowering, and maturity, the stem number and the number of stems with visual discolouration were counted for each plant. The SCI and basal 15 cm of each cleaned stem was rated for visual discolouration, measured as a honey brown to black discolouration of tissue, using a 0 to 100% rating scale where 0 = no discolouration and 100% = completely discoloured tissue (
Figure 1). Following visual discolouration rating, the roots and the SCI were removed, and plant height of each plant was measured from the base of the stem to the tip of the longest leaf at tillering stage and from the base of the stem to the tip of the longest stem with the longest head at flowering and maturity stages. All heads were removed, and the individual plants (leaves and stems) were dried at 60 °C in a drying oven (Wessberg Martin Engineering Pty Ltd., Sydney, NSW, Australia) for three days. Following drying, the individual plants were weighed.
2.5. Data Analysis
The incidence of discoloured stems (%) was calculated by dividing the number of discoloured stems by the total number of stems per plant. The visual discolouration (%) was calculated by using the average value of all stems in one plant. To ensure the assumptions of normality were not violated, the total stem number per plant, the incidence of discoloured stems, the visual discolouration assessed on each stem and the visual discolouration of the SCI were logit transformed, and the plant dry weight and stem height were log transformed. The analysis of each variable was performed using a linear mixed model. The model included fixed effects for pathogen, harvest time, cultivars, and their interactions. Terms to account for such as the replicate blocks, plots, and plants within plots were included as random effects. Estimates of variance parameters were generated using Residual maximum likelihood (REML) estimations [
41]. Predictions for each trait were generated from their respective models as empirical Best Linear Unbiased Estimators (eBLUEs). Where a transformation had been used, predicted means were back-transformed to the original scale, and approximate standard errors were calculated using the Taylor series approximation. All analyses were performed using ASReml-R [
42], in the R software environment [
43].
2.6. Tissue Processing and DNA Extraction
Fungal DNA from each strain was extracted from mycelium grown under 24 h darkness for 14 days on Potato Dextrose Agar (PDA) at 25 °C for Fusarium species and 20 °C for B. sorokiniana. Mycelium was scraped off plates and placed in a 2 mL centrifuge tube with two 2 mm metal beads (Winchester Australia limited, Moolap, VIC, Australia) and ground twice at 6.5 m/s for 15 s using a FastPrep-24 instrument (MP Biomedicals, Santa Ana, CA, USA). The extraction was conducted using the Wizard Genomic DNA Purification Kit for plant tissue (Promega, Sydney, NSW, Australia) according to the manufacturer’s recommended procedure.
In 2017, DNA was extracted from one random stem from each of five randomly selected plants from each plot, resulting in a total of 15 stems from each of the four pathogen treatments, along with the non-inoculated control treatments collected at flowering stage. The basal 3 cm of each stem was cleaned and stored separately in 5 cm tubes. Stems were dried at 60 °C in a drying oven (Wessberg Martin Engineering Pty Ltd., Sydney, NSW, Australia) for three days and stored in a −80 °C freezer. The basal 3 cm portion of the stem was cut into approximately 2 mm lengths and placed in 2 mL tubes. Prior to the extraction, each sample was again dried overnight in a dehydrator set at 65 °C (UF160-en, Memmert, Schwabach, Germany). Four 2 mm metal beads (Winchester Australia limited, Moolap, VIC, Australia) were placed into the 2 mL tube and each stem was ground twice at 6.5 m/s for 60 s using a FastPrep-24 instrument (MP Biomedicals, Valiant Co., Ltd., Santa Ana, CA, USA). Genomic DNA was extracted using the Wizard Genomic DNA Purification Kit for plant tissue (Promega, Sydney, Australia). The protocol provided by the manufacturer was used with the exception that incubation time was increased to 60 min at 65 °C after the addition of the Nuclei Lysis Solution. DNA was resuspended in 100 μL of autoclaved deionised water and stored at −20 °C until required.
2.7. Species-Specific PCR Assays
Species-specific PCR primers were used to amplify DNA of
F. pseudograminearum [Fp_TEF1α.2F (5′-ATCATTCGAATCGCTCGACG-3′) and Fp_TEF1α.2R (5′-AAAAATTACGACAAAGCCGTAAAAA-3′)] [
44],
F. culmorum [Fc_K13B-P_F (5′- ATGACCGAAGACTCGGAGAA-3′) and Fc_K13B-P_R (5′-CCTTGTGCTGAGCTCGTCT-3′)] [
28,
44],
F. graminearum [Fg16NF (5′-ACAGATGACAAGATTCAGGCACA-3′) and Fg16NR (5′-TTCTTTGACATCTGTTCAACCCA-3′)] [
45], and
B. sorokiniana [CosA_F_519 (5′-TCAAGCTGACCAAATCACCTTC-3′) & CosA_R_248 (5′-AATGTCGAGCTTGCCAAAGT-3′) [
46]. Pure genomic DNA standards of each pathogen strain were used as positive controls.
Each 10 uL PCR reaction consisted of 0.5 Units Immolase DNA polymerase (Bioline Pty Ltd., Eveleigh, NSW, Australia), 100 μM dNTPs, 1× Immolase buffer, 2.5 mM MgCl2, 0.25 mM forward and reverse primers, and 3 μL of DNA template. Thermal cycling conditions for primers Fc_K13B-P_F & Fc_K13B-P_R and Fg16NF & Fg16NR were 95 °C for 7 min followed by 40 cycles at 94 °C for 30 s, 60 °C for 30 s, and 72 °C for 30 s, with a final extension at 72 °C for 10 min. Thermal cycling conditions for primers Fp_TEF1α.2F & Fp_TEF1α.2R and CosA_F_519 & CosA_R_248 were 7 min at 95 °C, followed by 35 cycles of 94 °C for 30 s, 55 °C for 30 s, and 72 °C for 30 s, with a final extension at 72 °C for 10 min. DNA amplicons were separated by electrophoresis (100 V) in a 1% agarose gel with 5 μL GelRed and visualised using a Fusion FX (Vilber Lourmat, Fisher Biotec, Wembley, WA, Australia). For size assessment, a 100 bp DNA ladder was used (New England, Biolabs Inc., Dallas, TX, USA).
4. Discussion
This research is the most extensive study to date reporting a comparative analysis of four crown rot and common root rot pathogens in a single cultivar of five winter cereal species across different stages of growth in two field trials. This study has been employed along with a previously reported direct comparison of the four pathogens across the five-grain cereals in glasshouse tests [
29]. In both studies, significant variation among the visual discolouration ratings was caused by the four pathogens on stems and SCI tissues of the five winter cereals across harvest time. Climatic variables were also a major factor in our study, impacting the disease and the host response throughout the seasons. This study has identified significant differences in the visual discolouration caused by the three
Fusarium species and
B. sorokiniana in the five winter cereals in the field. In particular,
F. pseudograminearum caused the greatest visual discolouration on the stem and SCI followed by
F. culmorum,
B. sorokiniana, and
F. graminearum. The current study informed a significant variation in the extent of visual discolouration across the cultivars depending on which pathogen they were exposed to, with durum wheat cv. Hyperno, barley cv. Grimmett, bread wheat cv. Livingston and triticale cv. Endeavour recording the most disease symptoms, while oat cv. Genie developing the least visual discolouration when exposed to each pathogen.
Differences between the years in the levels of visual discolouration caused by
F. pseudograminearum and
F. culmorum strains were observed on cv. Hyperno and cv. Grimmett stems at flowering and maturity, with the levels of visual discolouration higher in 2017 than in 2016. This may be due to the wet conditions at the start of the 2016 season compared to the dry start of the 2017 season. This outcome is in contrast with other reports, which suggest that crown rot pathogens become more aggressive in the wet start and dry finish of the season [
6,
8,
19], which indicates that the increase in the infection due to crown rot pathogens was intensified by limited rainfall towards the end of the season [
8,
38]. Our study suggests that the escalation in the infection due to crown rot pathogens for some hosts could be increased by water stress early in the season, providing moisture is sufficient for plant establishment and initiation of infection by the pathogen in the host. While differences in pathogenicity have not been reported thus far in any of these host pathogen combinations, differences in aggressiveness do occur and interactions between cultivars and strains is possible. Inoculation with a mixture of strains from different geographic areas has been recommended to determine the potential value of bread wheat cultivars in local crown rot improvement breeding programs [
47,
48]. The higher levels of disease observed in 2017 could be due to combining the strains of each pathogen in this trial, compared to single isolates used in 2016, which could enhance the aggressiveness of each pathogen towards the host.
In this study, the durum wheat cv. Hyperno had a high level of visual discolouration on stems in both years compared to other cultivars when inoculated with either
F. pseudograminearum or
F. culmorum. This result agrees with other reports that have indicated that durum wheat cultivars are very susceptible to susceptible to
F. pseudograminearum [
18,
32,
49]. In contrast Saad et al. [
29] reported a low level of visual discolouration on both the SCI and the leaf sheaths of cv. Hyperno seedlings when inoculated with
F. pseudograminearum and
F. culmorum. In both field trials reported here, we observed no differences in the visual discolouration among inoculated treatments and the non-inoculated controls at the tillering stage in cv. Hyperno (data not shown). This suggests that different genes may be responsible for crown rot resistance at an early developmental stage in durum wheat cv. Hyperno and that resistance may not be expressed at the later stages of development [
50].
In contrast to differences in disease severity observed between years in cultivars Hyperno and Grimmett, a higher level of visual discolouration was observed in 2016 in the triticale cv. Endeavour on stems at maturity infected with one strain of
F. pseudograminearum and both strains of
F. culmorum compared to the 2017 trial. This further highlights the complex environment × cultivar × pathogen interactions that occur in crown rot infections where higher levels of rainfall in this instance may have resulted in higher disease severity in cv. Endeavour. The results agree with previous studies, which suggested that during the season, the increased growth of plants reduces the sub-soil moisture, making the plant more susceptible to water stress late in the season and more vulnerable to disease infection [
2,
6]. Previous reports have shown that triticale may be susceptible to the crown rot pathogen
F. pseudograminearum [
28,
51]. In our study,
F. pseudograminearum inoculation resulted in a higher level of visual discolouration compared to the other inoculated treatments and the non-inoculated control on cv. Endeavour at maturity for both years. Our study also demonstrated significant discolouration on the SCI of cv. Endeavour caused by
B. sorokiniana and
F.
culmorum in the 2017 field trial. A study by Klein et al. [
51] reported high levels of visual discolouration caused by
F. pseudograminearum infection on the triticale cv. Ningadhu (ranged from 85 to 91%). The findings of our study are in contrast with the results in Saad et al. [
29] where cv. Endeavour seedlings showed a low level of visual discolouration on both the SCI and the leaf sheaths when inoculated with
F. pseudograminearum.
Oat is considered to be a resistant or asymptomatic host to crown rot [
18,
28,
29,
34], and common root rot [
22] pathogens; however, other studies have recommended that oat should not be employed in crop rotation for crown rot [
34] nor common root rot management due to an increase in the levels of inoculum in the soil after planting oat [
22]. Low levels of visual discolouration were exhibited on the stems of the oat cv. Genie when inoculated with each pathogen in these field trials. However, these were not significantly different from the non-inoculated controls. In the 2016 field trial, the visual discolouration was significantly greater than the non-inoculated control on the SCI of Genie when inoculated with both
F. pseudograminearum and
F.
culmorum (
p < 0.05). These findings are consistent with the results of Saad et al. [
29], which reported
F. pseudograminearum or
F.
culmorum had higher visual discolouration than the control on the SCI of oat cv. Genie. Collins et al. [
35] tested the resistance and tolerance in six oat cultivars (Bannister, Carrolup, Durack, Kojonup, Mitika, Williams and Yallara), and two bread wheat cultivars (Mace and Emu Rock) to
F. pseudograminearum and
F.
culmorum through two seasons of field experimentation (total of four trials) between 2016 and 2017. All oat cultivars were more resistant (lower level of visual discolouration) to
F. pseudograminearum than wheat cultivars and had four-times lower yield loss. However, in 2016 at post-harvest, the inoculum levels of
F.
culmorum in the oats trials were higher than that in the wheat trials and similar to levels recorded for
F. pseudograminearum [
35].
Knight and Sutherland [
28] reported low or no discolouration and low levels of fungal biomass measured in
F. pseudograminearum infected oat seedlings (cv. Taipan and Quoll), suggesting that oat is a poor host and may be a potential crop for rotation. In the current report, the oat cv. Genie was observed to have little or no visual discolouration across all pathogen treatments in the 2017 field trial. However, the PCR assay detected the DNA of all pathogens in the base of cv. Genie stems. Furthermore,
F. culmorum had a higher incidence of DNA amplified on cv. Genie followed by
F. pseudograminearum compared to the other strains, suggesting that greater levels of
F. culmorum colonisation are occurring in cv. Genie then expressed disease symptoms. Thus, oat is not recommended for crop rotation as it may host crown rot and common root rot pathogens and escalate the pathogen population in the field. This requires further investigation using qPCR assays to identify the fungal biomass in oat cultivars.
The symptoms of crown rot pathogens might differ in the same field on the same host [
8,
13]. Initially, the brown discolouration that is associated with crown rot manifests as small necrotic lesions on the coleoptile tissue which is followed by brown discolouration on the SCI and the first leaf sheaths. The discolouration then develops on the base of the first internode of the stem tissue [
1,
8,
16]. In our study, across harvest time, the brown discolouration was observed mostly to start at the base of the stem, with lesions progressing into higher internodes. However, in some cases, at flowering, the discolouration had only developed on the second internode in both years. Knight and Sutherland [
52] indicated that the fungal invasion of internodes above ground level could occur via stomata, with penetration hyphae growing across from the infected subtending leaf sheaths in close contact with the stem. This may indicate that while the fungus colonised on the first leaf sheath tissue, penetration hyphae did not invade the second or the third leaf sheath tissue until late in the development of the plant more likely after or during stem extension.
An increase in the plant dry weight was observed from tillering to maturity for the cultivars tested when inoculated with
F. pseudograminearum and
F. culmorum strains in the 2016 trial compared to the non-inoculated control. This may be due to higher stem numbers observed at flowering and maturity in
F. pseudograminearum and
F. culmorum inoculated treatments compared to the non-inoculated controls. In the 2017 trial, the increase in plant dry weight was observed but rather
F. pseudograminearum had an overall negative effect on all cultivars tested compared to the non-inoculated control. This result is in line with Saad et al. [
29] who found that there was a decrease in the plant dry weight in seedlings across the cultivars tested when infected with
F. pseudograminearum,
F. culmorum, and
F. graminearum. Further research is needed to examine the impact of crown rot and common root rot pathogens on the physiological response of the host at different stages of growth.
A higher than expected incidence of visual discolouration was detected in the non-inoculated control treatments in both years. The pre-sowing Predicta-B results indicated that there was no evidence of any of the crown rot nor common root rot pathogens or any other soilborne pathogens such as take-all (Gaeumannomyces graminis) in the paddock for both years. Furthermore, Fusarium species and B. sorokiniana DNA were not detected on the sub-sample of control plots for both years. The low levels of visual discolouration that were detected on the non-inoculated plants (1 to 5%) could be due to physiological or other biological impacts, which cause brown discolouration similar to the disease.