Diversity of Chemical Composition and Morphological Traits of Eight Iranian Wild Salvia Species during the First Step of Domestication
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Study Site
2.3. Seedling Production
2.4. Evaluation of Growth Parameters
2.5. Evaluation of Biochemical Parameters
2.5.1. Total Phenolic Compounds
2.5.2. Total Flavonoids
2.5.3. DPPH Radical Scavenging Activity
2.5.4. Rosmarinic Acid Content
2.5.5. Essential Oil Extraction
2.5.6. Gas Chromatography-Flame Ionization Detector and Gas Chromatography-Mass Spectrometry Analysis
2.6. Statistical Analysis
3. Results and Discussion
3.1. Phytochemical Evaluation
3.1.1. Total Phenolic Compounds (TP)
3.1.2. Total Flavonoids
3.1.3. DPPH Radical Scavenging Activity
3.1.4. Rosmarinic Acid Content
3.1.5. Essential Oil Content and Composition
3.2. Correlations among Traits
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Faezeh Taghizadeh, S.; Panahi, A.; Esmaeilzadeh Kashi, M.; Kretschmer, N.; Asili, J.; Ahmad Emami, S.; Azizi, M.; Shakeri, A. Structural Diversity of Complex Phloroglucinol Derivatives from Eucalyptus Species. Chem. Biodivers. 2022, 19, e202200025. [Google Scholar] [CrossRef] [PubMed]
- Azizi, M.; Davareenejad, G.; Bos, R.; Woerdenbag, H.J.; Kayser, O. Essential oil content and constituents of black zira (Bunium persicum [Boiss.] B. Fedtsch.) from Iran during field cultivation (domestication). J. Essent. Oil Res. 2009, 21, 78–82. [Google Scholar] [CrossRef]
- Azizi, M.; Tavana, M.; Farsi, M.; Oroojalian, F. Yield performance of Lingzhi or Reishi medicinal mushroom, Ganoderma lucidum (W. Curt.: Fr.) P. Karst.(higher Basidiomycetes), using different waste materials as substrates. Int. J. Med. Mushrooms 2012, 14, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Salma, U.; Kundu, S.; Mandal, N. Artificial polyploidy in medicinal plants: Advancement in the last two decades and impending prospects. J. Crop Sci. Biotechnol. 2017, 20, 9–19. [Google Scholar] [CrossRef]
- Tarakemeh, A.; Azizi, M.; Rowshan, V.; Salehi, H.; Spina, R.; Dupire, F.; Arouie, H.; Laurain-Mattar, D. Screening of Amaryllidaceae alkaloids in bulbs and tissue cultures of Narcissus papyraceus and four varieties of N. tazetta. J. Pharm. Biomed. Anal. 2019, 172, 230–237. [Google Scholar] [CrossRef]
- Mahdieh, M.; Talebi, S.M.; Akhani, M. Infraspecific essential oil and anatomical variations of Salvia nemorosa L.(Labiatae) populations in Iran. Ind. Crops Prod. 2018, 123, 35–45. [Google Scholar] [CrossRef]
- Chizzola, R.; Saeidnejad, A.H.; Azizi, M.; Oroojalian, F.; Mardani, H. Bunium persicum: Variability in essential oil and antioxidants activity of fruits from different Iranian wild populations. Genet. Resour. Crop Evol. 2014, 61, 1621–1631. [Google Scholar] [CrossRef]
- Kaur, N.; Kaur, N.; Saggoo, M. Conservation Strategies for Medicinal Plants in the Face of Environmental Challenges. In Environmental Challenges and Medicinal Plants; Springer: Berlin/Heidelberg, Germany, 2022; pp. 461–485. [Google Scholar]
- Heidari, S.; Azizi, M.; Soltani, F.; Hadian, J. Foliar application of Ca (NO3) 2 and KNO3 affects growth, essential oil content, and oil composition of French tarragon. Ind. Crops Prod. 2014, 62, 526–532. [Google Scholar] [CrossRef]
- Giannoulis, K.D.; Skoufogianni, E.; Bartzialis, D.; Solomou, A.D.; Danalatos, N.G. Growth and productivity of Salvia officinalis L. under Mediterranean climatic conditions depends on biofertilizer, nitrogen fertilization, and sowing density. Ind. Crops Prod. 2021, 160, 113136. [Google Scholar] [CrossRef]
- Matkowski, A.; Zielińska, S.; Oszmiański, J.; Lamer-Zarawska, E. Antioxidant activity of extracts from leaves and roots of Salvia miltiorrhiza Bunge, S. przewalskii Maxim., and S. verticillata L. Bioresour. Technol. 2008, 99, 7892–7896. [Google Scholar] [CrossRef]
- Hafez Ghoran, S.; Taktaz, F.; Mozafari, A.A.; Tunçtürk, M.; Sekeroglu, N.; Kijjoa, A. Uncommon Terpenoids from Salvia Species: Chemistry, Biosynthesis and Biological Activities. Molecules 2022, 27, 1128. [Google Scholar] [CrossRef]
- Tohma, H.; Köksal, E.; Kılıç, Ö.; Alan, Y.; Yılmaz, M.A.; Gülçin, İ.; Bursal, E.; Alwasel, S.H. RP-HPLC/MS/MS analysis of the phenolic compounds, antioxidant and antimicrobial activities of Salvia L. species. Antioxidants 2016, 5, 38. [Google Scholar] [CrossRef]
- Bahadori, M.B.; Dinparast, L.; Valizadeh, H.; Farimani, M.M.; Ebrahimi, S.N. Bioactive constituents from roots of Salvia syriaca L.: Acetylcholinesterase inhibitory activity and molecular docking studies. South Afr. J. Bot. 2016, 106, 1–4. [Google Scholar] [CrossRef]
- Boukhary, R.; Raafat, K.; Ghoneim, A.I.; Aboul-Ela, M.; El-Lakany, A. Anti-inflammatory and antioxidant activities of Salvia fruticosa: An HPLC determination of phenolic contents. Evid. -Based Complementary Altern. Med. 2016, 2016, 7178105. [Google Scholar] [CrossRef] [PubMed]
- Cardile, V.; Russo, A.; Formisano, C.; Rigano, D.; Senatore, F.; Arnold, N.A.; Piozzi, F. Essential oils of Salvia bracteata and Salvia rubifolia from Lebanon: Chemical composition, antimicrobial activity and inhibitory effect on human melanoma cells. J. Ethnopharmacol. 2009, 126, 265–272. [Google Scholar] [CrossRef]
- Mirmostafaee, S.; Azizi, M.; Fujii, Y. Study of allelopathic interaction of essential oils from medicinal and aromatic plants on seed germination and seedling growth of lettuce. Agronomy 2020, 10, 163. [Google Scholar] [CrossRef]
- Sekine, T.; Appiah, K.S.; Azizi, M.; Fujii, Y. Plant growth inhibitory activities and volatile active compounds of 53 spices and herbs. Plants 2020, 9, 264. [Google Scholar] [CrossRef]
- Liu, J.-J.; Wu, H.-H.; Chen, T.-H.; Leung, W.; Liang, Y.-C. 15, 16-Dihydrotanshinone I from the functional food Salvia miltiorrhiza exhibits anticancer activity in human HL-60 leukemia cells: In vitro and in vivo studies. Int. J. Mol. Sci. 2015, 16, 19387–19400. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, C.; Peng, L.; Liang, Z.; Yan, X.; Zhu, Y.; Liu, Y. Comparison of essential oil composition and phenolic acid content of selected Salvia species measured by GC–MS and HPLC methods. Ind. Crops Prod. 2015, 69, 329–334. [Google Scholar] [CrossRef]
- Velamuri, R.; Sharma, Y.; Fagan, J.; Schaefer, J. Application of UHPLC-ESI-QTOF-MS in phytochemical profiling of sage (Salvia officinalis) and rosemary (Rosmarinus officinalis). Planta Med. Int. Open 2020, 7, e133–e144. [Google Scholar] [CrossRef]
- Bahadori, M.B.; Dinparast, L.; Zengin, G.; Sarikurkcu, C.; Bahadori, S.; Asghari, B.; Movahhedin, N. Functional components, antidiabetic, anti-Alzheimer’s disease, and antioxidant activities of Salvia syriaca L. Int. J. Food Prop. 2017, 20, 1761–1772. [Google Scholar] [CrossRef]
- Jassbi, A.; Miri, R.; Alizadeh, M.; Asadollahi, M.; Massrorbabanari, M.; Baldwin, I. Quantification of Phenolic Diterpenoids and Rosmarinic Acid in Salvia eremophila and Salvia santolinifolia by LC-DAD-MS. Austin Chromatogr. 2014, 1, 1012–1017. [Google Scholar]
- Hassanzadeh, M.; Emami, S.; Asili, J.; Najaran, Z.T. Review of the essential oil composition of Iranian Lamiaceae. J. Essent. Oil Res. 2011, 23, 35–74. [Google Scholar] [CrossRef]
- Jamzad, Z. A survey of Lamiaceae in the flora of Iran. Rostaniha 2013, 14, 59–67. [Google Scholar]
- Ebrahimi, M.; Ranjbar, S. Some Autecological Properties of Medicinal Plant of Salvia hydrangea L. in Mazandaran, Iran. J. Rangel. Sci. 2016, 6, 253–263. [Google Scholar]
- Zarei, G.; Morovvati Sharifabad, A. Essential oil composition of Salvia eremophila Boiss. in different stages of plant growth in Yazd province. Eco-Phytochem. J. Med. Plants 2017, 4, 74–84. [Google Scholar]
- Cabrera-Santos, D.; Ordoñez-Salanueva, C.A.; Sampayo-Maldonado, S.; Campos, J.E.; Orozco-Segovia, A.; Flores-Ortiz, C.M. Quantifying Cardinal Temperatures of Chia (Salvia hispanica L.) Using Non-Linear Regression Models. Plants 2022, 11, 1142. [Google Scholar] [CrossRef]
- Feng, J.; Liao, F.; Kong, D.; Ren, R.; Sun, T.; Liu, W.; Yin, Y.; Ma, H.; Tang, J.; Li, G. Genetic diversity of the cultivated Salvia miltiorrhiza populations revealed by four intergenic spacers. PLoS ONE 2022, 17, e0266536. [Google Scholar] [CrossRef]
- Samadi, S.; Saharkhiz, M.J.; Azizi, M.; Samiei, L.; Ghorbanpour, M. Multi-walled carbon nanotubes stimulate growth, redox reactions and biosynthesis of antioxidant metabolites in Thymus daenensis celak. in vitro. Chemosphere 2020, 249, 126069. [Google Scholar] [CrossRef]
- Koşar, M.; Göger, F.; Can Başer, K.H. In vitro antioxidant properties and phenolic composition of Salvia virgata Jacq. from Turkey. J. Agric. Food Chem. 2008, 56, 2369–2374. [Google Scholar] [CrossRef]
- Menichini, F.; Tundis, R.; Bonesi, M.; Loizzo, M.R.; Conforti, F.; Statti, G.; De Cindio, B.; Houghton, P.J.; Menichini, F. The influence of fruit ripening on the phytochemical content and biological activity of Capsicum chinense Jacq. cv Habanero. Food Chem. 2009, 114, 553–560. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Komali, A.S.; Shetty, K. Comparison of the growth pattern and Rosharinic acid production in rosemary (Rosmarinus officinalis) shoots and genetically transformed callus cultures. Food Biotechnol. 1998, 12, 27–41. [Google Scholar] [CrossRef]
- Lopez-Arnaldos, T.; Lopez-Serrano, M.; Barcelo, A.R.; Calderon, A.; Zapata, J. Spectrophotometric determination of rosmarinic acid in plant cell cultures by complexation with Fe2+ ions. Fresenius J. Anal. Chem. 1995, 351, 311–314. [Google Scholar] [CrossRef]
- National Institute of Standards and Technology (NIST). Mass Spectral Library (NIST/EPA/NIH); National Institute of Standards and Technology: Gaithersburg, MD, USA, 2008.
- Duncan, D.B. Multiple range and multiple F tests. biometrics 1955, 11, 1–42. [Google Scholar] [CrossRef]
- Jonckheere, I.; Fleck, S.; Nackaerts, K.; Muys, B.; Coppin, P.; Weiss, M.; Baret, F. Methods for leaf area index determination. Part I: Theories, techniques and instruments. Agric. For. Meteorol. 2004, 121, 19–35. [Google Scholar] [CrossRef]
- Sánchez-Calderón, L.; Ibarra-Cortés, M.; Zepeda-Jazo, I. Root development and abiotic stress adaptation. In Abiotic Stress—Plant Responses and Applications in Agriculture; IntechOpen: London, UK, 2013; pp. 135–168. [Google Scholar]
- Azizi, M. Change in content and chemical composition of Hypericum perforatum L. oil at three harvest time. J. Herbs Spices Med. Plants 2008, 13, 79–85. [Google Scholar] [CrossRef]
- Shahhoseini, R.; Azizi, M.; Asili, J.; Moshtaghi, N.; Samiei, L. Effects of zinc oxide nanoelicitors on yield, secondary metabolites, zinc and iron absorption of Feverfew (Tanacetum parthenium (L.) Schultz Bip.). Acta Physiol. Plant. 2020, 42, 52. [Google Scholar] [CrossRef]
- Yahia, Y.; Benabderrahim, M.A.; Tlili, N.; Bagues, M.; Nagaz, K. Bioactive compounds, antioxidant and antimicrobial activities of extracts from different plant parts of two Ziziphus Mill. species. PLoS ONE 2020, 15, e0232599. [Google Scholar] [CrossRef]
- Pank, F. Breeding of medicinal plants. In Medicinal Plant Biotechnology: From Basic Research to Industrial Applications; Wiley-VCH: Weinheim, Germany, 2006; pp. 417–449. [Google Scholar]
- Jiang, R.-W.; Lau, K.-M.; Hon, P.-M.; Mak, T.C.; Woo, K.-S.; Fung, K.-P. Chemistry and biological activities of caffeic acid derivatives from Salvia miltiorrhiza. Curr. Med. Chem. 2005, 12, 237–246. [Google Scholar] [CrossRef]
- Shaerzadeh, F.; Ahmadiani, A.; Esmaeili, M.A.; Ansari, N.; Asadi, S.; Tusi, S.K.; Sonboli, A.; Ghahremanzamaneh, M.; Khodagholi, F. Antioxidant and antiglycating activities of Salvia sahendica and its protective effect against oxidative stress in neuron-like PC12 cells. J. Nat. Med. 2011, 65, 455–465. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, G.; Azizi, M.; Arouiee, H.; Vaezi, J. Anatomical and Morphological Properties of Trichomes in Four Iranian Native Salvia Species under Cultivated Conditions. Int. J. Hortic. Sci. Technol. 2019, 6, 189–200. [Google Scholar]
- Kołton, A.; Długosz-Grochowska, O.; Wojciechowska, R.; Czaja, M. Biosynthesis regulation of folates and phenols in plants. Sci. Hortic. 2022, 291, 110561. [Google Scholar] [CrossRef]
- Mander, L.; Liu, H.-W. Comprehensive Natural Products II; Elsevier Science: Oxford, UK, 2010. [Google Scholar]
- Mo, F.; Li, L.; Zhang, C.; Yang, C.; Chen, G.; Niu, Y.; Si, J.; Liu, T.; Sun, X.; Wang, S. Genome-wide analysis and expression profiling of the phenylalanine ammonia-lyase gene family in Solanum tuberosum. Int. J. Mol. Sci. 2022, 23, 6833. [Google Scholar] [CrossRef] [PubMed]
- Loizzo, M.R.; Abouali, M.; Salehi, P.; Sonboli, A.; Kanani, M.; Menichini, F.; Tundis, R. In vitro antioxidant and antiproliferative activities of nine Salvia species. Nat. Prod. Res. 2014, 28, 2278–2285. [Google Scholar] [CrossRef] [PubMed]
- Parvin, K.; Nahar, K.; Mohsin, S.M.; Mahmud, J.A.; Fujita, M.; Hasanuzzaman, M. Plant Phenolic Compounds for Abiotic Stress Tolerance. In Managing Plant Production Under Changing Environment; Springer: Singapore, 2022; pp. 193–237. [Google Scholar]
- Hamrouni-Sellami, I.; Rahali, F.Z.; Rebey, I.B.; Bourgou, S.; Limam, F.; Marzouk, B. Total phenolics, flavonoids, and antioxidant activity of sage (Salvia officinalis L.) plants as affected by different drying methods. Food Bioprocess Technol. 2013, 6, 806–817. [Google Scholar] [CrossRef]
- Deshmukh, V.P. Phytochemistry and Pharmacology of Salvia officinalis L.: A Review. In Bioactives and Pharmacology of Medicinal Plants; Apple Academic Press: Palm Bay, FL, USA, 2022; pp. 339–358. [Google Scholar]
- Ghorbani, A.; Esmaeilizadeh, M. Pharmacological properties of Salvia officinalis and its components. J. Tradit. Complementary Med. 2017, 7, 433–440. [Google Scholar] [CrossRef]
- Falcone Ferreyra, M.L.; Rius, S.P.; Casati, P. Flavonoids: Biosynthesis, biological functions, and biotechnological applications. Front. Plant Sci. 2012, 3, 222. [Google Scholar] [CrossRef]
- Lee, K.W.; Kim, Y.J.; Lee, H.J.; Lee, C.Y. Cocoa has more phenolic phytochemicals and a higher antioxidant capacity than teas and red wine. J. Agric. Food Chem. 2003, 51, 7292–7295. [Google Scholar] [CrossRef]
- Dai, J.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, X.; Wang, Z. Antioxidant activities of leaf extract of Salvia miltiorrhiza Bunge and related phenolic constituents. Food Chem. Toxicol. 2010, 48, 2656–2662. [Google Scholar] [CrossRef] [PubMed]
- Azizi, M.; Chizzola, R.; Ghani, A.; Oroojalian, F. Composition at different development stages of the essential oil of four Achillea species grown in Iran. Nat. Prod. Commun. 2010, 5, 283–290. [Google Scholar] [CrossRef]
- Said-Al Ahl, H.; Hussein, M.S.; Gendy, A.S.; Tkachenko, K.G. Quality of sage (Salvia officinalis L.) essential oil grown in Egypt. Int. J. Plant Sci. Ecol. 2015, 1, 119–123. [Google Scholar]
- Sefidkon, F.; Mirza, M. Chemical composition of the essential oils of two Salvia species from Iran, Salvia virgata Jacq. and Salvia syriaca L. Flavour Fragr. J. 1999, 14, 45–46. [Google Scholar] [CrossRef]
- Šulniūtė, V.; Baranauskienė, R.; Ragažinskienė, O.; Venskutonis, P.R. Comparison of composition of volatile compounds in ten Salvia species isolated by different methods. Flavour Fragr. J. 2017, 32, 254–264. [Google Scholar] [CrossRef]


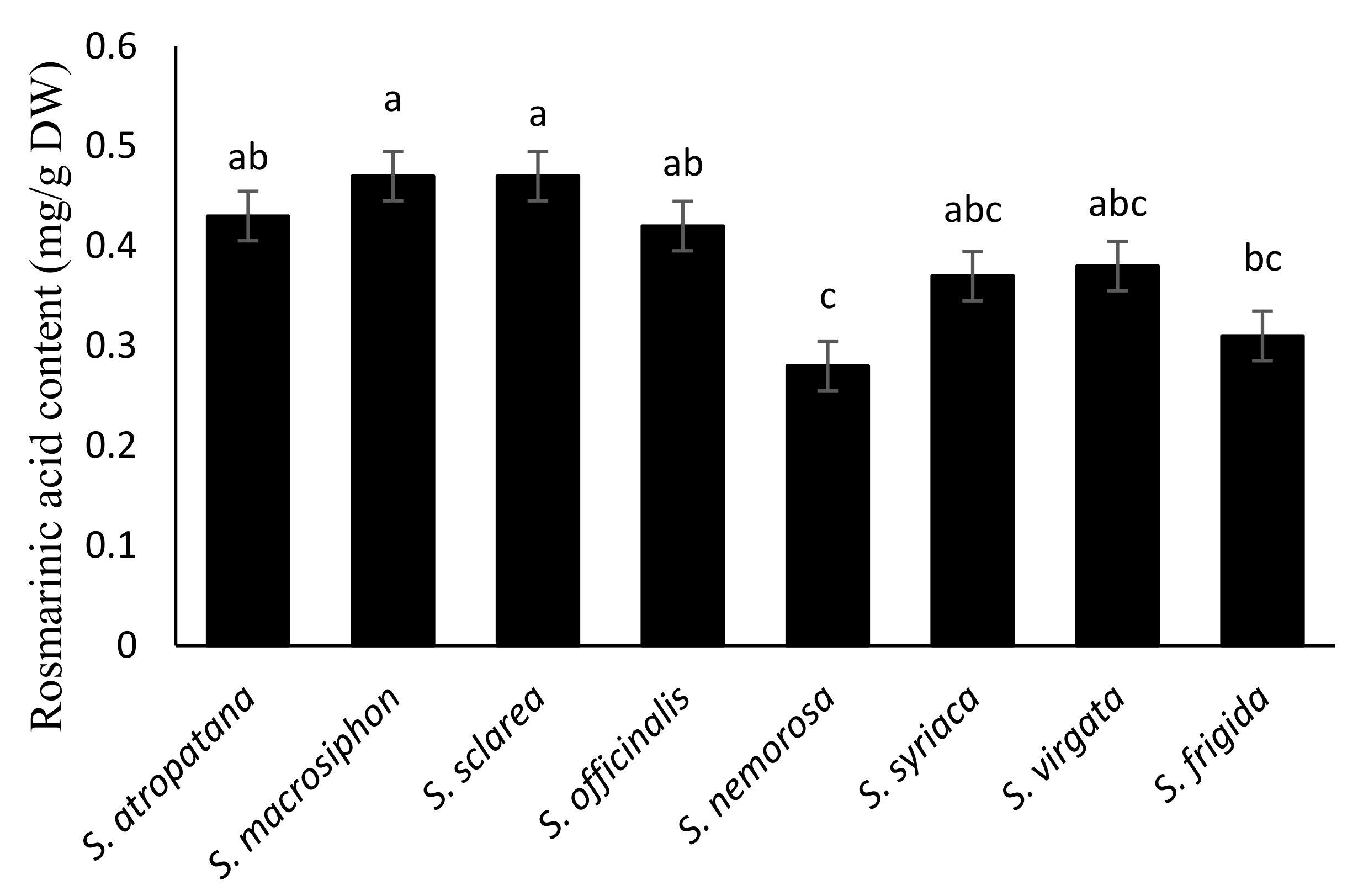

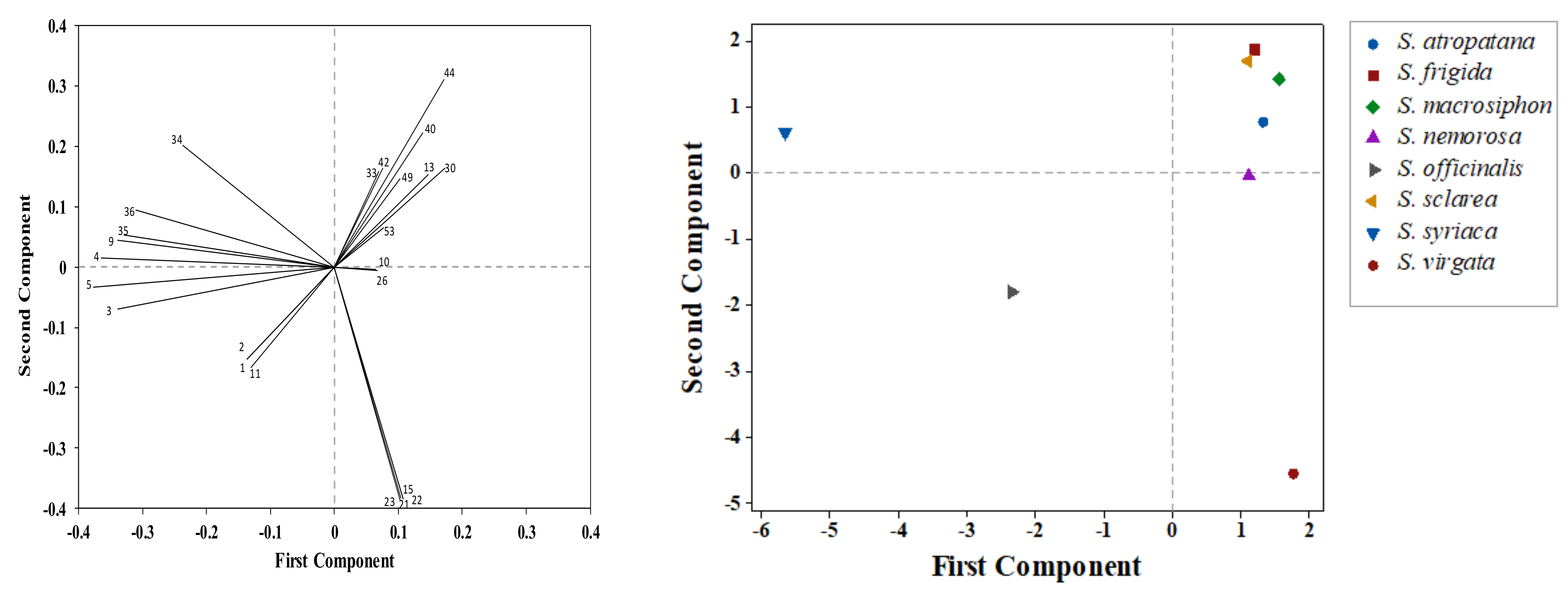
| Species | LA (1) | TFW | TDW | RFW | RDW | SFW | SDW | LFW | LDW | FFW | FDW | SW | TDB |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| S. atropatana | 547.7 cd | 91.7 d | 22.4 ef | 25.0 efg | 5.3 ef | 33.3 d | 6.9 d | 51.7 bcd | 8.9 cd | 6.2 d | 1.3 f | 4.20 b | 1182 cd |
| S. macrosiphon | 604.0 cd | 144.2 c | 52.7 b | 107.7 a | 23.2 a | 60.7 bc | 13.0 bc | 54.0 bcd | 10.7 bc | 29.0 b | 5.8 c | 2.65 d | 1764 c |
| S. sclarea | 9117.3 a | 570.2 a | 120.5 a | 25.7 efg | 5.3 f | 96.0 a | 20.7 a | 409.7 a | 79.1 a | 64.0 a | 15.4 a | 2.24 f | 6235 a |
| S. officinalis | 1128.3 c | 116.8 cd | 25.8 ef | 13.3 g | 3.2 f | 31.3 d | 6.3 d | 73.0 b | 14.4 b | 12.0 cd | 1.9 ef | 5.51 a | 1452 cd |
| S. nemorosa | 1936.3 b | 186.4 b | 43.6 bc | 32.5 def | 6.7 def | 74.9 b | 15.8 b | 55.5 bc | 10.5 bc | 55.5 a | 10.6 b | 0.77 j | 3288 b |
| S. syriaca | 348.0 d | 88.5 d | 19.3 f | 21.fg | 3.4 f | 42.7 cd | 7.4 d | 28.3 cd | 5.5 cd | 18.9 bcd | 3.0 def | 3.65 c | 1014 d |
| S. virgata (Afoos) | 839.7 cd | 101.7 d | 30.6 de | 46.8 de | 10.1 cd | 41.0 d | 9.1 cd | 36.5 cd | 8.0 cd | 23.8 bc | 3.4 c.f | 1.15 h | 1344 cd |
| S. virgata (Darkesh) | 760.0 cd | 89.5 d | 27.4 ef | 42.8 cde | 10.0 cde | 37.3 d | 7.9 d | 25.7 d | 4.9 d | 26.0 b | 4.5 cd | 0.95 i | 1110 cd |
| S. virgata (Eghled) | 926.8 cd | 119.4 cd | 38.9 cd | 78.2 b | 17.9 b | 45.0 cd | 8.6 cd | 50.5 bcd | 8.6 cd | 23.5 bc | 3.8 cde | 1.59 g | 1368 cd |
| S. frigida (Jahrom) | 1025.0 cd | 182.2 b | 28.7 ef | 58.4 c | 11.7 c | 27.3 d | 6.0 d | 56.3 bcd | 9.5 bcd | 9.0 d | 1.5 ef | 2.42 e | 924 d |
| S. frigida (Targavar) | 1073.3 c | 81.7 d | 22.8 ef | 40.8 cde | 7.0 def | 26.7 d | 5.4 d | 43.2 cd | 8.2 cd | 11.3 cd | 2.2 def | 2.48 e | 1230 cd |
| Species | Plant Parts | Total Phenolic Compounds (mg GAE/g Extract) | Flavonoids Content (mg QE/g Extract) | IC50 (1) |
|---|---|---|---|---|
| S. atropatana | Leaf | 22.7 ± 3.9 d–g | 23.9 ± 2.9 lm | 14.0 ± 1.1 ij |
| Root | - | - | - | |
| Stem | - | - | - | |
| Flower | - | - | - | |
| S. macrosiphon | Leaf | 14.7 ± 2.0 g–k | 51.9 ± 5.5 hij | 36.7 ± 1.9 e |
| Root | 5.8 ± 0.8 j–m | 15.9 ± 1.9 ms | 10.9 ± 1.7 j | |
| Stem | - | - | - | |
| Flower | - | - | - | |
| S. sclarea | Leaf | 19.3 ± 2.3 fi | 69.9 ± 3.2 fgh | 24.2 ± 1.8 fg |
| Root | 1.5 ± 0.2 m | 14.9 ± 2.1 m | 14.9 ± 2.0 ij | |
| Stem | - | - | - | |
| Flower | - | - | - | |
| S. officinalis | Leaf | 40.6 ± 2.9 a | 129.1 ± 7.9 b | 52.3 ± 3.1 c |
| Root | 4.9 ± 0.3 klm | 96.3 ± 8.4 cde | 42.9 ± 7.0 d | |
| Stem | - | - | - | |
| Flower | - | - | - | |
| S. nemorosa | Leaf | 29.0 ± 2.3 c–f | 141.9 ± 10.0 ab | 90.6 ± 3.3 a |
| Root | 7.3 ± 0.9 j–m | 71.9 ± 8.1 fh | 24.3 ± 0.8 fg | |
| Stem | 9.7 ± 2.1 i–m | 68.8 ± 3.0 ghi | 14.3 ± 1.2 ij | |
| Flower | 39.8 ± 6.9 ab | 121.7 ± 4.8 bc | 90.7 ± 7.2 a | |
| S. syriaca | Leaf | 13.7 ± 1.4 g–l | 43.6 ± 6.8 i–l | 52.0 ± 2.5 c |
| Root | - | - | - | |
| Stem | - | - | - | |
| Flower | - | - | - | |
| S. virgata (Afoos) | Leaf | 35.6 ± 7.6 abc | 83.9 ± 9.4 d–g | 89.1 ± 1.4 a |
| Root | 15.8 ± 3.3 g–j | 94.9 ± 4.7 def | 38.2 ± 5.3 de | |
| Stem | 18.7 ± 2.6 fi | 82.1 ± 7.3 d–g | 42.2 ± 3.6 de | |
| Flower | 36.7 ± 5.7 abc | 100.4 ± 8.7 cd | 89.6 ± 3.0 a | |
| S. virgata (Darkesh) | Leaf | 14.2 ± 2.3 g–l | 54.3 ± 7.7 hij | 90.3± 2.2 a |
| Root | 18.5 ± 0.3 ghi | 63.5 ± 4.3 ghi | 25.3 ± 1.7 fg | |
| Stem | - | - | - | |
| Flower | 32.5 ± 2.7 ad | 156.7 ± 6–5 a | 53.8 ± 1.6 c | |
| S. virgata (Eghled) | Leaf | 18.3 ± 3.4 ghi | 61.5 ± 8.8 ghi | 22.4 ± 4.1 gh |
| Root | 10.2 ± 1.3 i–m | 71.7 ± 2.7 e–h | 28.3 ± 1.4 f | |
| - | - | - | - | |
| - | - | - | - | |
| S. frigida (Jahrom) | Leaf | 10.4 ± 0.5 i–m | 50.9 ± 7.0 hk | 29.1 ± 2.3 f |
| Root | 11.1 ± 0.7 hm | 34.0 ± 4.7 j–m | 18.0 ± 2.6 hi | |
| Stem | - | - | - | |
| Flower | - | - | - | |
| S. frigida (Targavar) | Leaf | 30.1 ± 1.5 b–e | 95.1 ± 5.3 def | 88.5 ± 2.3 a |
| Root | 10.1 ± 24 i–m | 62.5 ± 4.7 ghi | 26.1 ± 2.6 fg | |
| Stem | 4.2 ± 0.8 lm | 26.3 ± 3.0 klm | 40.2 ± 3.6 de | |
| Flower | 21.1 ± 2.6 e–h | 58.8 ± 8.7 g–j | 81.0 ± 4.3 b |
| No. | Formula | Compounds | RI (1) | SO (2) | SV (3) | SN (4) | SS (5) | SC (6) | SM (7) | SA [36] | SF (9) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | C10H16O | α-Thujone | 1105 | 39.34 | - | - | - | - | - | - | - |
| 2 | C10H16O | β-Thujone | 1112 | 15.51 | - | - | - | - | - | - | - |
| 3 | C10H16O | (-)-Camphor | 1139 | 17.18 | - | - | 15.12 | - | 3.10 | - | - |
| 4 | C11H18O2 | Isobornyl formate | 1217 | 4.60 | - | - | 18.40 | - | - | - | - |
| 5 | C10H14 | 1,3,8-p-menthatriene | 1100 | 2.98 | - | - | 5.07 | - | - | - | - |
| 6 | C10H16O | trans-3-Caren-2-ol | 1655 | 0.67 | - | - | 4.17 | - | - | - | - |
| 7 | C10H14 | 2,8-Decadiyne | 1102 | 0.47 | - | - | - | - | - | - | - |
| 8 | C10H16O | Pulegone | 1234 | 1.34 | - | - | - | - | - | - | - |
| 9 | C10H16 | Camphene | 946 | 0.31 | - | - | 5.39 | - | - | - | - |
| 10 | C15H24 | Aromadendrene | 1442 | 1.33 | 0.48 | 47.50 | - | - | 2.88 | - | - |
| 11 | C15H24 | α-Caryophyllene | 1438 | 13.03 | 0.50 | - | - | - | - | 0.50 | - |
| 12 | C10H14O | Thymol | 1291 | - | 0.34 | - | - | 0.74 | - | - | 3.84 |
| 13 | C15H24 | β-Caryophyllene | 1441 | - | 7.08 | - | - | 6.18 | 13.50 | - | 31.05 |
| 14 | C15H24 | cis-β-Famesene | 1458 | - | 1.46 | - | - | - | - | - | - |
| 15 | C15H24 | δ-Cadinene | 1525 | - | 23.32 | - | - | - | - | 1.55 | - |
| 16 | C15H24 | Seychellene | 1449 | - | 1.20 | - | - | - | - | - | - |
| 17 | C15H24 | β-Elemen | 1391 | - | 0.96 | - | - | - | - | - | - |
| 18 | C15H24 | cis-Z-α-Bisabolene epoxide | 1816 | - | 2.73 | - | - | - | - | - | - |
| 19 | C22H32O2 | Doconexent | 2522 | - | 3.97 | - | - | - | - | - | - |
| 20 | C17H24O | Falcarinol | 2040 | - | 2.07 | - | - | - | - | - | - |
| 21 | C15H24 | Υ-Gurjunene | 1470 | - | 5.62 | - | - | - | - | 0.41 | - |
| 22 | C15H26O | Valeranone | 1672 | - | 26.09 | - | - | - | - | - | 0.60 |
| 23 | C21H34O2 | 3-Ethyl-3-hydroxyandrostan-17-one | 2651 | - | 10.22 | - | - | - | - | - | - |
| 24 | C7H16 | 2,3-Dimethylpentane | 675 | - | - | 4.76 | - | - | - | - | - |
| 25 | C7H16 | 3-Methylhexane | 671 | - | - | 0.34 | - | - | - | - | - |
| 26 | C8H18 | Tetramethylbutane | 716 | - | - | 18.12 | - | - | - | - | - |
| 27 | C18H31N | 2-Tridecylpyridine | 1920 | - | - | 4.21 | - | - | - | - | - |
| 28 | C10H17Br | Geranyl bromide | 1284 | - | - | 1.28 | - | - | - | - | - |
| 29 | C10H16 | Santolina triene | 911 | - | - | 3.72 | - | - | - | - | - |
| 30 | C15H24O | Caryophyllene oxide | 1581 | - | - | 13.48 | 0.55 | 14.63 | 24.30 | 5.5 | |
| 31 | C20H24N2O | Eseroline, benzyl ether | 2635 | - | - | 2.08 | - | - | - | - | - |
| 32 | C10H16 | α-Pinene | 937 | - | - | - | - | - | - | - | 6.15 |
| 33 | C10H18O | α-Terpineol | 1192 | - | - | - | 5.99 | 3.21 | 0.21 | - | 3.16 |
| 34 | C10H16O | cis-Verbenol | 1115 | - | - | - | 13.55 | - | - | - | - |
| 35 | C12H20O2 | Bornyl acetate | 1288 | - | - | - | 30.83 | - | - | 5.55 | 5.12 |
| 36 | C10H16 | Myrcene | 991 | - | - | - | - | 1.85 | - | - | - |
| 37 | C10H16 | transe-β-Ocimene | 1039 | - | - | - | - | 1.82 | - | - | - |
| 38 | C10H16 | Terpinolene | 1089 | - | - | - | - | 0.68 | 0.85 | - | 0.55 |
| 39 | C10H18O | Linalool | 1099 | - | - | - | - | 26.20 | 27.20 | 7.60 | - |
| 40 | C10H18O | Nerol | 1229 | - | - | - | - | 2 | - | - | - |
| 41 | C10H20O2 | Linalyl acetate | 1248 | - | - | - | - | 20.50 | 1.55 | - | 1.05 |
| 42 | C15H24 | β-Cubebene | 1387 | - | - | - | - | 0.65 | - | - | - |
| 43 | C15H24 | Germacrene D | 1583 | - | - | - | - | 16.40 | 7.59 | 10.50 | 19.5 |
| 44 | C15H26O | β-Eudesmol | 1651 | - | - | - | - | 0.71 | - | - | - |
| 45 | C10H18O | 1,8-cineole | 1035 | - | - | - | - | - | - | - | 2.70 |
| 46 | C10H16 | Limonene | 1029 | - | - | - | - | - | 0.22 | - | 1.01 |
| 47 | C10H18O | Borneol | 1169 | - | - | - | - | - | 0.77 | - | - |
| 48 | C15H24O | Spathulenol | 1578 | - | - | - | - | - | 5.80 | - | 0.87 |
| 49 | C15H24 | Germacrene B | 1552 | - | - | - | - | - | 1.32 | - | - |
| 50 | C15H26O | α-Eudesmol | 1653 | - | - | - | - | - | 1.25 | - | - |
| 51 | C27H56 | Heptacosane | 525 | - | - | - | - | - | 0.30 | - | - |
| 52 | C15H24 | α-Cubebene | 1353 | - | - | - | - | - | - | 16.4 | - |
| 53 | C20H36O2 | Sclareol | 2223 | - | - | - | - | - | - | 0.50 | - |
| 54 | C17H28O2 | Caryophyllene acetate | 1692 | - | - | - | - | - | - | 3.21 | - |
| 55 | C15H24O | Alloaromadendrene oxide | 1641 | - | - | - | - | - | - | 0.74 | - |
| 56 | C12H20O7 | Triethyl citrate | 1657 | - | - | - | - | - | - | 0.60 | - |
| 57 | C15H24 | Diepicedrene | 1402 | - | - | - | - | - | - | 0.81 | - |
| 58 | C15H24O | 14-Hydroxy-α-humulene | 1715 | - | - | - | - | - | - | 2.61 | - |
| 59 | C14H12O2 | Benzyl benzoate | 1763 | - | - | - | - | - | - | 0.45 | - |
| 60 | C10H8 | Naphthalene | 1185 | - | - | - | - | - | - | - | 1.25 |
| Oxygenated monoterpenes | 78.64 | 0.34 | 0.0 | 88.06 | 52.65 | 32.83 | 12.34 | 15.87 | |||
| Monoterpene hydrocarbons | 3.76 | 0.0 | 5.0 | 10.46 | 4.35 | 1.07 | 0.81 | 8.96 | |||
| Sesquiterpene hydrocarbons | 14.36 | 40.62 | 47.5 | 0.0 | 23.23 | 25.29 | 29.36 | 50.55 | |||
| Oxygenated sesquiterpenes | 0.0 | 28.82 | 13.48 | 0.0 | 1.26 | 21.68 | 31.67 | 6.97 | |||
| Others | 0.0 | 16.26 | 29.51 | 0.0 | 0.0 | 0.3 | 1.55 | 0.0 | |||
| Detected compound | 96.76 | 86.04 | 95.49 | 98.52 | 81.49 | 81.17 | 75.73 | 82.35 | |||
| Essential oil content | 1.45 | 0.70 | 0.85 | 0.45 | 0.85 | 0.55 | 0.35 | 0.40 |
| Salvia Species | S. atropatana | S. macrosiphon | S. sclarea | S. officinalis | S. nemorosa | S. syriaca | S. virgata | S. frigida |
|---|---|---|---|---|---|---|---|---|
| S. atropatana | 0.0 | |||||||
| S. macrosiphon | 32.4 | 0.0 | ||||||
| S. sclarea | 41.8 | 28.8 | 0.0 | |||||
| S. officinalis | 57.9 | 58.5 | 61.0 | 0.0 | ||||
| S. nemorosa | 57.0 | 58.4 | 65.2 | 70.6 | 0.0 | |||
| S. syriaca | 50.5 | 54.5 | 56.6 | 58.3 | 68.1 | 0.0 | ||
| S. virgata | 49.5 | 50.0 | 52.8 | 61.0 | 65.0 | 57.0 | 0.0 | |
| S. frigida | 41.1 | 36.7 | 41.4 | 60.3 | 52.9 | 41.4 | 36.7 | 0.0 |
| LA | TFW | TDW | RFW | RDW | SFW | SDW | LFW | LDW | FFW | FDW | SW | IC50 | TP | TF | RAC | EOC | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LA | 1 | ||||||||||||||||
| TFW | 0.98 ** | 1 | |||||||||||||||
| TDW | 0.97 ** | 0.98 ** | 1 | ||||||||||||||
| RFW | −0.2 | −0.12 | −0.14 | 1 | |||||||||||||
| RDW | −0.18 | −0.10 | −0.11 | 0.97 ** | 1 | ||||||||||||
| SFW | 0.75 ** | 0.83 ** | 0.85 ** | 0.10 | 0.12 | 1 | |||||||||||
| SDW | 0.74 ** | 0.81 ** | 0.86 ** | 0.08 | 0.11 | 0.97 ** | 1 | ||||||||||
| LFW | 0.99 ** | 0.98 ** | 0.94 ** | −0.17 | −0.14 | 0.73 ** | 0.70 ** | 1 | |||||||||
| LDW | 0.98 ** | 0.96 ** | 0.93 ** | −0.18 | −0.14 | 0.71 ** | 0.67 ** | 0.98 ** | 1 | ||||||||
| FFW | 0.67 ** | 0.72 ** | 0.77 ** | 0.00 | 0.01 | 0.65 ** | 0.71 ** | 0.60 ** | 0.58 ** | 1 | |||||||
| FDW | 0.72 ** | 0.76 ** | 0.79 ** | −0.05 | −0.05 | 0.61 ** | 0.67 ** | 0.67 ** | 0.64 ** | 0.97 ** | 1 | ||||||
| SW | −0.11 | −0.10 | −0.17 | −0.31 | −0.20 | 0.17 | 0.26 | 0.01 | −0.10 | −0.53 ** | −0.47 ** | 1 | |||||
| IC50 | −0.25 | −0.30 | −0.23 | 0.00 | 0.00 | −0.30 | −0.16 | 0.35 * | −0.35 * | 0.14 | 0.10 | −0.52 ** | 1 | ||||
| TP | 0.02 | −0.01 | 0.02 | 0.10 | 0.05 | 0.03 | 0.13 | −0.03 | −0.03 | 0.06 | −0.01 | −0.44 ** | 0.42 * | 1 | |||
| TF | 0.12 | 0.08 | 0.12 | −0.07 | 0.00 | 0.12 | 0.24 | 0.03 | 0.05 | 0.31 | 0.20 | −0.28 | 0.56 ** | 0.37 * | 1 | ||
| RAC | 0. 30 | 0.37 * | 0.29 | 0.03 | 0.10 | 0.29 | 0.18 | 0.41 * | 0.35 * | 0.00 | 0.08 | 0.48 * | −0.47 * | −0.28 | −0.37 * | 1 | |
| EOC | 0.25 | 0.26 | 0.29 | −0.30 | −0.20 | 0.18 | 0.20 | 0.25 | 0.22 | 0.26 | 0.23 | 0.24 | 0.05 | −0.26 | 0.49 ** | 0.22 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esmaeili, G.; Fatemi, H.; Baghani avval, M.; Azizi, M.; Arouiee, H.; Vaezi, J.; Fujii, Y. Diversity of Chemical Composition and Morphological Traits of Eight Iranian Wild Salvia Species during the First Step of Domestication. Agronomy 2022, 12, 2455. https://doi.org/10.3390/agronomy12102455
Esmaeili G, Fatemi H, Baghani avval M, Azizi M, Arouiee H, Vaezi J, Fujii Y. Diversity of Chemical Composition and Morphological Traits of Eight Iranian Wild Salvia Species during the First Step of Domestication. Agronomy. 2022; 12(10):2455. https://doi.org/10.3390/agronomy12102455
Chicago/Turabian StyleEsmaeili, Ghasem, Hamideh Fatemi, Mahnaz Baghani avval, Majid Azizi, Hossein Arouiee, Jamil Vaezi, and Yoshiharu Fujii. 2022. "Diversity of Chemical Composition and Morphological Traits of Eight Iranian Wild Salvia Species during the First Step of Domestication" Agronomy 12, no. 10: 2455. https://doi.org/10.3390/agronomy12102455
APA StyleEsmaeili, G., Fatemi, H., Baghani avval, M., Azizi, M., Arouiee, H., Vaezi, J., & Fujii, Y. (2022). Diversity of Chemical Composition and Morphological Traits of Eight Iranian Wild Salvia Species during the First Step of Domestication. Agronomy, 12(10), 2455. https://doi.org/10.3390/agronomy12102455










