Effects of LED Light Quality on the Growth of Pepper (Capsicum spp.) Seedlings and the Development after Transplanting
Abstract
1. Introduction
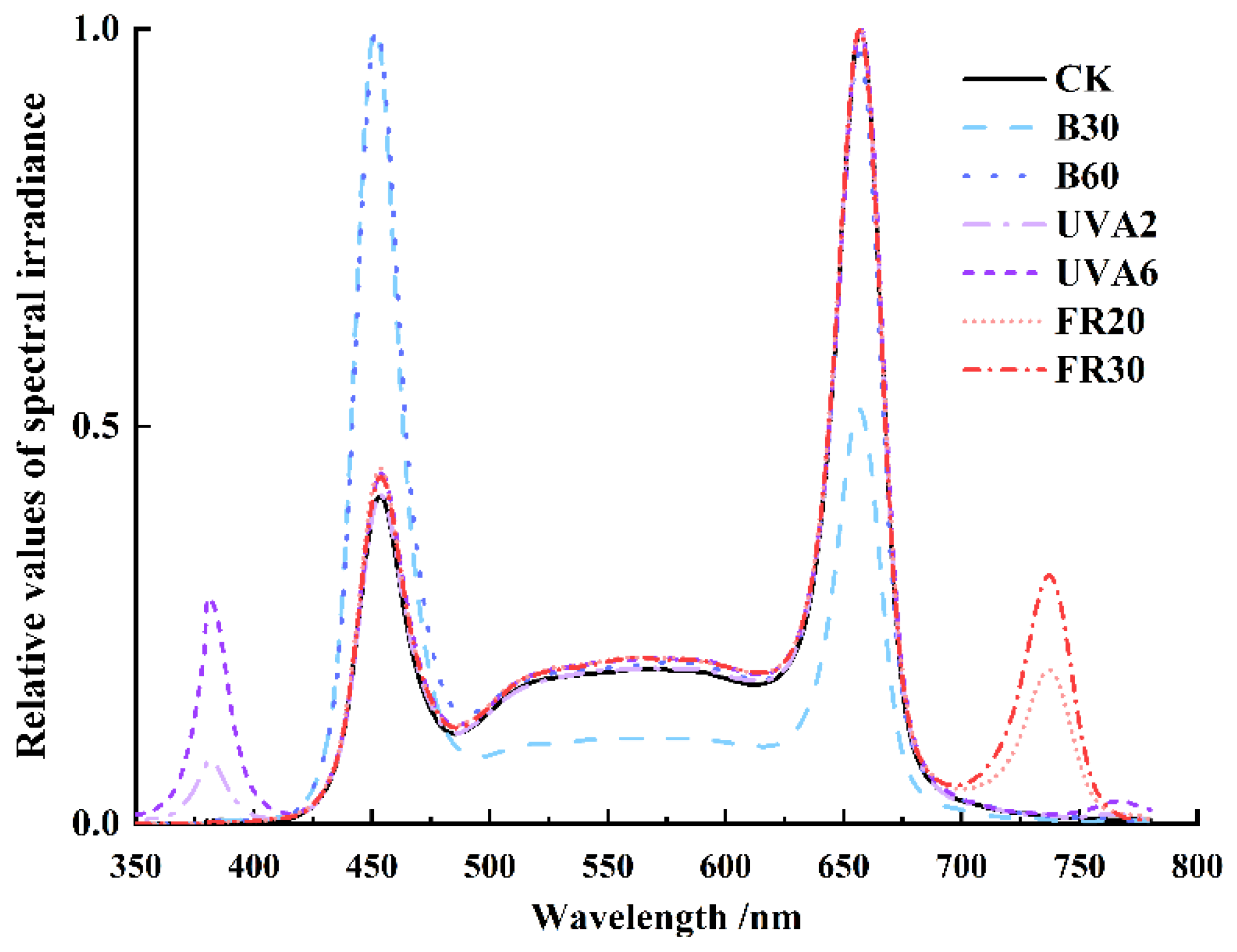
2. Materials and Methods
2.1. Plant Material and Growth Conditions
2.2. Determination of Morphological Traits and Fresh and Dry Weight
2.3. Calculation of Seedling Comprehensive Index
2.4. Measurement of Pigment Contents and Chlorophyll Fluorescence Parameters
2.5. Measurement of Phytochemical Substance and Enzyme Activities of Seedlings
2.6. Data of Flowering and Fruit Set after Transplanting
2.7. Statistical Analysis
3. Results
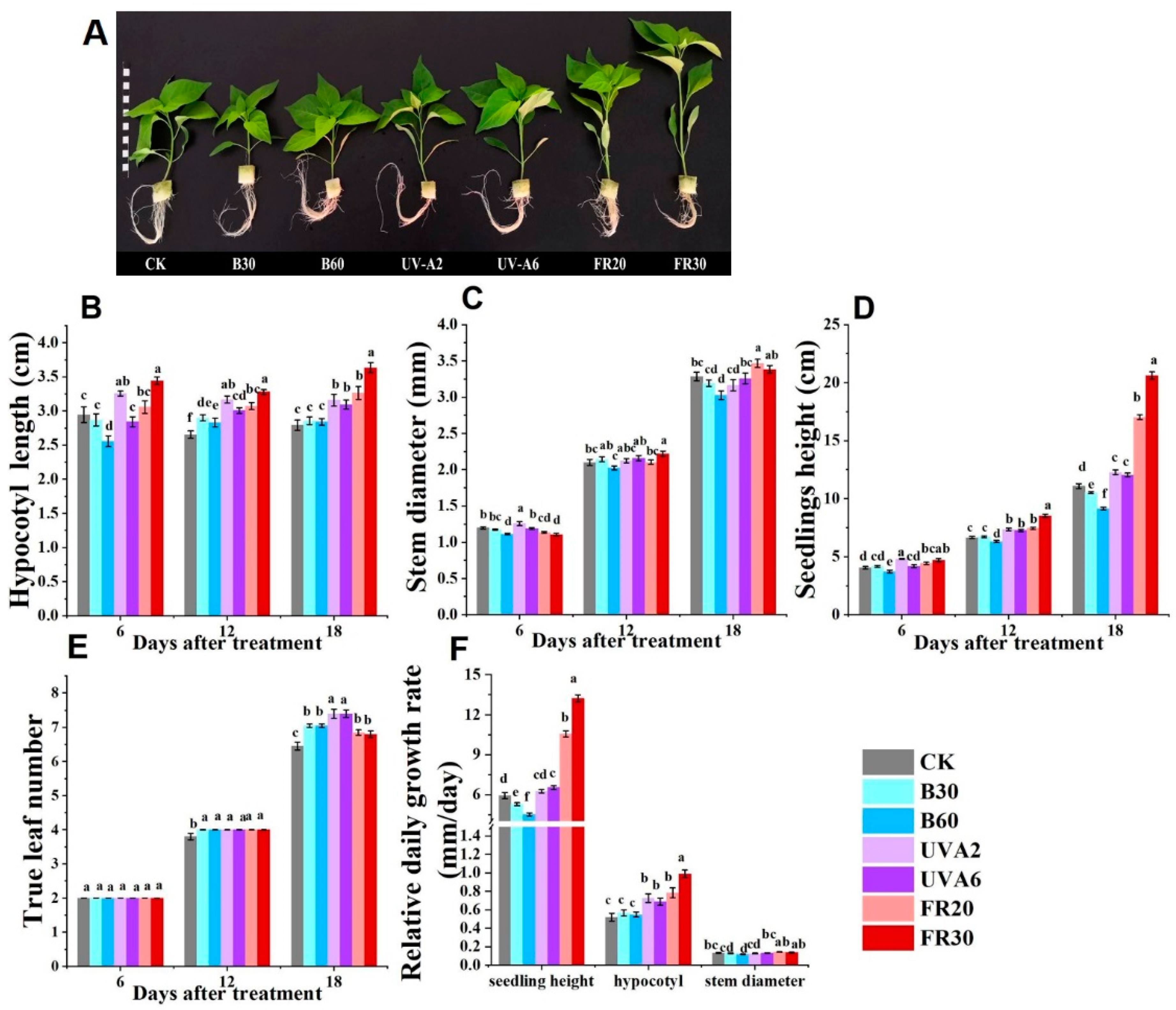
3.1. Impacts of Diverse LED Light Conditions on the Morphology and Growth of Pepper Seedlings
3.2. Influences of Diverse LED Light Conditions on the Comprehensive Growth Indexes of Pepper Seedlings
3.3. Effects of Different LED Light on Photosynthetic Pigment Content and Chlorophyll Fluorescence Parameters
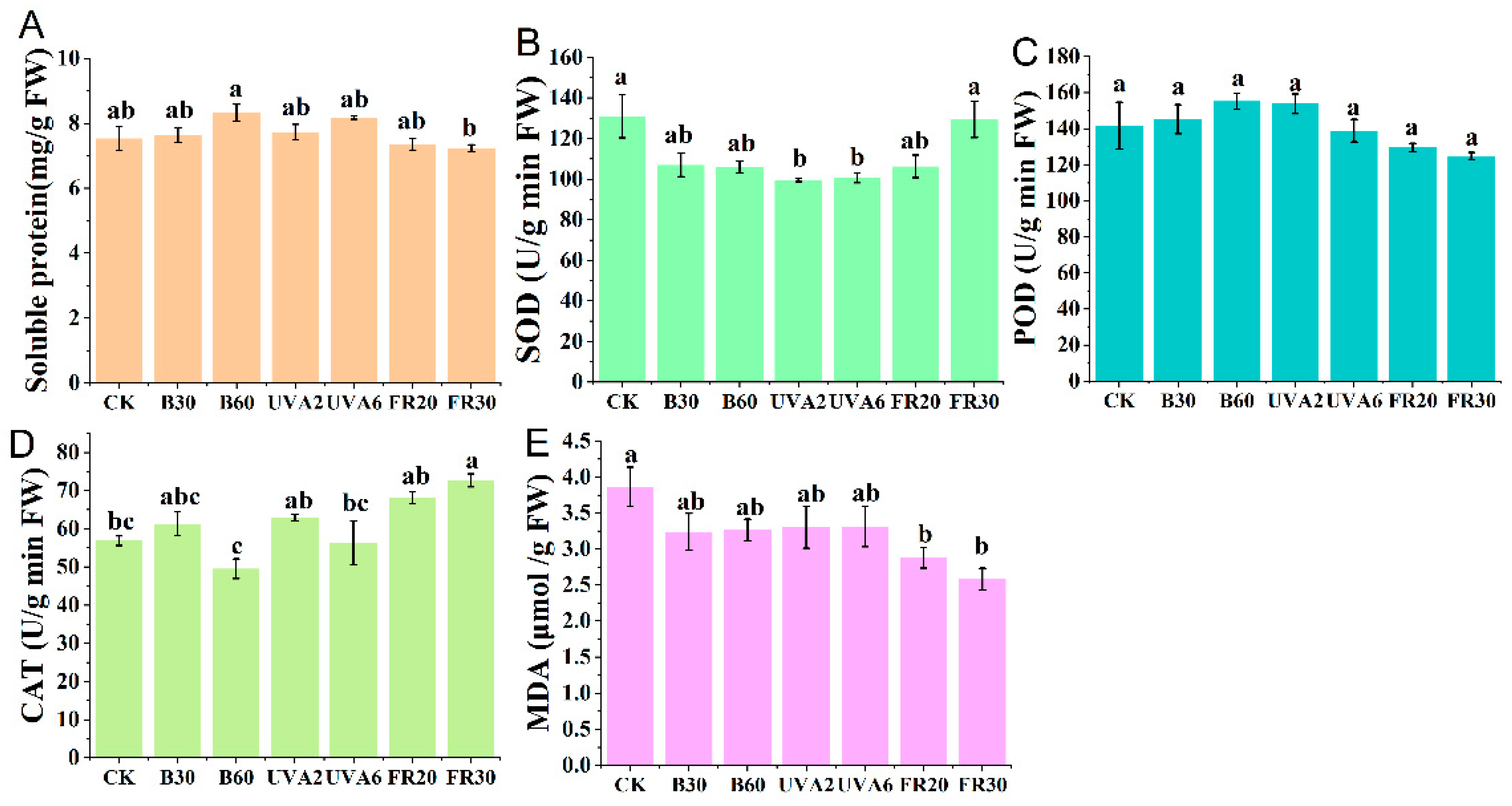
3.4. The Influence of Different LED Light Environment on Physiological Characteristics and Antioxidant Enzyme Activities of Pepper Seedlings
3.5. The Effect of Different LED Light on Flowering and Fruit Setting of Different Seedlings after Transplanting
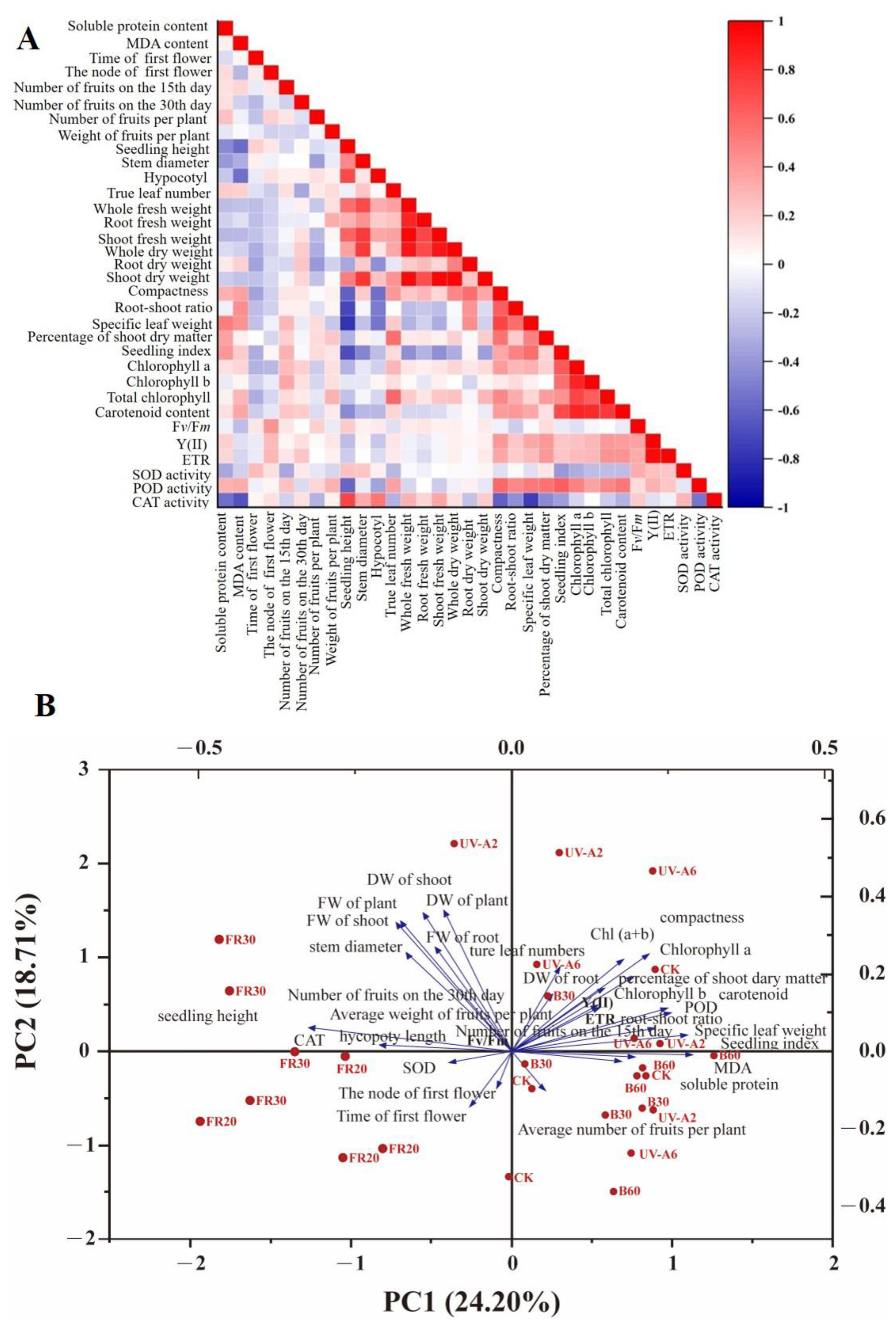
3.6. Correlation Analysis and Comprehensive Evaluation
4. Discussion
4.1. Impacts of Light Quality on the Growth Traits
4.2. Effects of Light Quality on Photosynthetic Pigment Content and Photosynthetic Characteristics
4.3. Effects of Light Quality on Physiological Characteristics of Pepper Seedlings and Transplant Development of Pepper Plants
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Franklin, K.A.; Quail, P.H. Phytochrome functions in Arabidopsis development. J. Exp. Bot. 2010, 61, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Nadimi, M.; Sun, D.-W.; Paliwal, J. Effect of laser biostimulation on germination of Wheat. Appl. Eng. Agric. 2022, 38, 77–84. [Google Scholar] [CrossRef]
- Sheerin, D.J.; Hiltbrunner, A. Molecular mechanisms and ecological function of far-red light signalling. Plant Cell Environ. 2017, 40, 2509–2529. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.Q.G.W. Effects of light quality on plant growth and development. Plant Physiol. J. 2015, 51, 1217–1234. [Google Scholar]
- Singh, D.; Basu, C.; Meinhardt-Wollweber, M.; Roth, B. LEDs for energy efficient greenhouse lighting. Renew. Sustain. Energy Rev. 2015, 49, 139–147. [Google Scholar] [CrossRef]
- Moradi, S.; Kafi, M.; Aliniaeifard, S.; Salami, S.A.; Shokrpour, M.; Pedersen, C.; Moosavi-Nezhad, M.; Wrobel, J.; Kalaji, H.M. Blue light improves photosynthetic performance and biomass partitioning toward harvestable organs in Saffron (Crocus sativus L.). Cells 2021, 10, 1994. [Google Scholar] [CrossRef]
- Zhang, T.; Folta, K.M. Green light signaling and adaptive response. Plant Signal. Behav. 2012, 7, 75–78. [Google Scholar] [CrossRef] [PubMed]
- He, R.; Zhang, Y.; Song, S.; Su, W.; Hao, Y.; Liu, H. UV-A and FR irradiation improves growth and nutritional properties of lettuce grown in an artificial light plant factory. Food Chem. 2021, 345, 128727. [Google Scholar] [CrossRef]
- Mao, P.; Duan, F.; Zheng, Y.; Yang, Q. Blue and UV-A light wavelengths positively affected accumulation profiles of healthy compounds in pak-choi. J. Sci. Food Agric. 2021, 101, 1676–1684. [Google Scholar] [CrossRef]
- Liu, Y.; Schouten, R.E.; Tikunov, Y.; Liu, X.; Visser, R.G.F.; Tan, F.; Bovy, A.; Marcelis, L.F.M. Blue light increases anthocyanin content and delays fruit ripening in purple pepper fruit. Postharvest Biol. Technol. 2022, 192, 112024. [Google Scholar] [CrossRef]
- Rodriguez-Calzada, T.; Qian, M.; Strid, A.; Neugart, S.; Schreiner, M.; Torres-Pacheco, I.; Guevara-Gonzalez, R.G. Effect of UV-B radiation on morphology, phenolic compound production, gene expression, and subsequent drought stress responses in chili pepper (Capsicum annuum L.). Plant Physiol. Biochem. 2019, 134, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Claypool, N.B.; Lieth, J.H. Physiological responses of pepper seedlings to various ratios of blue, green, and red light using LED lamps. Sci. Hortic. 2020, 268, 10931. [Google Scholar] [CrossRef]
- Hoffmann, A.M.; Noga, G.; Hunsche, M. Acclimations to light quality on plant and leaf level affect the vulnerability of pepper (Capsicum annuum L.) to water deficit. J. Plant Res. 2015, 128, 295–306. [Google Scholar] [CrossRef] [PubMed]
- McNellis, T.W.; Deng, X.W. Light control of seedling morphogenetic pattern. Plant Cell 1995, 11, 1749–1761. [Google Scholar]
- Kozai, T. Plant factories with artificial lighting (PFALs): Benefits, problems, and challenges. In Smart Plant Factory; Springer: Singapore, 2018; pp. 15–29. [Google Scholar]
- Hwang, H.; An, S.; Pham, M.D.; Cui, M.; Chun, C. The combined conditions of photoperiod, light Intensity, and air temperature control the growth and development of tomato and red pepper seedlings in a closed transplant production system. Sustainability 2020, 12, 9939. [Google Scholar] [CrossRef]
- Graamans, L.; Baeza, E.; van den Dobbelsteen, A.; Tsafaras, I.; Stanghellini, C. Plant factories versus greenhouses: Comparison of resource use efficiency. Agric. Syst. 2018, 160, 31–43. [Google Scholar] [CrossRef]
- Schuerger, A.C.; Brown, C.S.; Stryjewski, E.C. Anatomical features of pepper plants (Capsicum annuum L.) grown under red light-emitting diodes supplemented with blue or far-red light. Ann. Bot. 1997, 3, 273–282. [Google Scholar] [CrossRef]
- Hwang, H.; An, S.; Lee, B.; Chun, C. Improvement of growth and morphology of vegetable seedlings with supplemental Far-red enriched LED lights in a plant factory. Horticulturae 2020, 6, 109. [Google Scholar] [CrossRef]
- Bai Y, S.W.X.X. Study on tobacco vigorous seedling indexes model. Agric. Sci. China 2014, 6, 1086–1098. [Google Scholar]
- Li, J.; Shi, C.; Wang, X.; Liu, C.; Ding, X.; Ma, P.; Wang, X.; Jia, H. hydrogen sulfide regulates the activity of antioxidant enzymes through persulfidation and improves the resistance of tomato seedling to copper oxide nanoparticles (CuO NPs)-induced oxidative stress. Plant Physiol. Biochem. 2020, 156, 257–266. [Google Scholar] [CrossRef]
- Zhang, J.H.; Huang, W.D.; Liu, Y.P.; Pan, Q.H. Effects of Temperature acclimation pretreatment on the ultrastructure of mesophyll cells in young grape plants (Vitis vinifera L. cv. Jingxiu) under cross-temperature stresses. J. Integr. Plant Biol. 2005, 8, 959–970. [Google Scholar] [CrossRef]
- Huang, Y.; Li, Y.; Gao, X.; Zhang, Y.; Chen, C.; Wu, M.; Li, H.; Du, Y.; Kang, Y.; Yang, Y. Evaluation indexes of tomato seedling growth based on principal component analysis. China Cucurbits Veg. 2021, 34, 32–37. [Google Scholar]
- Sabzalian, M.R.; Heydarizadeh, P.; Zahedi, M.; Boroomand, A.; Agharokh, M.; Sahba, M.R.; Schoefs, B. High performance of vegetables, flowers, and medicinal plants in a red-blue LED incubator for indoor plant production. Agron. Sustain. Dev. 2014, 34, 879–886. [Google Scholar] [CrossRef]
- Kang, S.; Zhang, Y.; Zhang, Y.; Zou, J.; Yang, Q.; Li, T. Ultraviolet-A radiation stimulates growth of indoor cultivated tomato (Solanum lycopersicum) seedlings. HortScience 2018, 53, 1429–1433. [Google Scholar] [CrossRef]
- Hosseini, A.; Zare Mehrjerdi, M.; Aliniaeifard, S.; Seif, M. Photosynthetic and growth responses of green and purple basil plants under different spectral compositions. Physiol. Mol. Biol. Plants 2019, 25, 741–752. [Google Scholar] [CrossRef]
- Liscum, E.; Young, J.C.; Poff, K.L.; Hangarter, R.P. Genetic separation of phototropism and blue light inhibition of stem elongation. Plant Physiol. 1992, 100, 267–271. [Google Scholar] [CrossRef][Green Version]
- Shimizu, H.; Ma, Z.; Tazawa, S.; Douzono, M.; Runkle, E.S.; Heins, R.D. Blue light inhibits stem elongation of chrysanthemum. Int. Symp. Artif. Lighting Hortic. 2005, 711, 363–368. [Google Scholar] [CrossRef]
- Jeong, H.W.; Lee, H.R.; Kim, H.M.; Kim, H.M.; Hwang, H.S.; Hwang, S.J. Using light quality for growth control of cucumber seedlings in closed-type plant production system. Plants 2020, 9, 639. [Google Scholar] [CrossRef]
- Rodriguez-Concepcion, M.; Avalos, J.; Bonet, M.L.; Boronat, A.; Gomez-Gomez, L.; Hornero-Mendez, D.; Limon, M.C.; Melendez-Martinez, A.J.; Olmedilla-Alonso, B.; Palou, A.; et al. A global perspective on carotenoids: Metabolism, biotechnology, and benefits for nutrition and health. Prog. Lipid. Res. 2018, 70, 62–93. [Google Scholar] [CrossRef]
- Croft, H.; Chen, J.M.; Luo, X.; Bartlett, P.; Chen, B.; Staebler, R.M. Leaf chlorophyll content as a proxy for leaf photosynthetic capacity. Glob. Chang Biol. 2017, 23, 3513–3524. [Google Scholar] [CrossRef]
- Liu, N.; Ji, F.; Xu, L.; He, D. Effects of LED light quality on the growth of pepper seedling in plant factory. Int. J. Agric. Biol. Eng. 2019, 12, 44–50. [Google Scholar] [CrossRef]
- Tezuka, T.Y.F.; Ando, Y. Physiological activation in radish plants by UV-A radiation. J. Photochem. Photobiol. B Biol. 1994, 24, 33–40. [Google Scholar] [CrossRef]
- Li, Q.; Kubota, C. Effects of supplemental light quality on growth and phytochemicals of baby leaf lettuce. Environ. Exp. Bot. 2009, 67, 59–64. [Google Scholar]
- Tanaka, H.; Murai, K.; Nakanishi, T.; Tezuka, T.; Shiozaki, S.; Oda, M. Storage of plug seedlings of tomato under limited fertilisation, and growth, flowering and yield after planting. J. Hortic. Sci. Biotechnol. 2018, 93, 652–658. [Google Scholar]
- Liu, G.; Du, Q.; Li, J. Interactive effects of nitrate-ammonium ratios and temperatures on growth, photosynthesis, and nitrogen metabolism of tomato seedlings. Sci. Hortic. 2017, 214, 41–50. [Google Scholar]
- Ramalho, J.C.; Marques, N.C.; Semedo, J.N.; Matos, M.C.; Quartin, V.L. Photosynthetic performance and pigment composition of leaves from two tropical Species is determined by light quality. Plant Biol. 2002, 1, 112–120. [Google Scholar] [CrossRef]
- Rascher, U.; Liebig, M.; Lüttge, U. Evaluation of instant light-response curves of chlorophyll fluorescence parameters obtained with a portable chlorophyll fluorometer on site in the field. Plant Cell Environ. 2000, 12, 1397–1405. [Google Scholar]
- Yao, X.-y.; Liu, X.-y.; Xu, Z.-g.; Jiao, X.-l. Effects of light intensity on leaf microstructure and growth of rape seedlings cultivated under a combination of red and blue LEDs. J. Integr. Agric. 2017, 16, 97–105. [Google Scholar]
- Zheng J, H.M.; Guo, Y.P. Regulation of photosynthesis by light quality and its mechanism in plants. Ying Yong Sheng Tai Xue Bao Chin. 2018, 19, 1619–1624. [Google Scholar]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 179–194. [Google Scholar]
- Gao, S.; Liu, X.; Liu, Y.; Cao, B.; Chen, Z.; Xu, K. The spectral irradiance, growth, photosynthetic characteristics, antioxidant system, and nutritional status of green Onion (Allium fistulosum L.) grown under different photo-selective nets. Front. Plant Sci. 2021, 12, 650471. [Google Scholar] [CrossRef] [PubMed]
- Dehariya, P.; Kataria, S.; Pandey, G.P.; Guruprasad, K.N. Assessment of impact of solar UV components on growth and antioxidant enzyme activity in cotton plant. Physiol. Mol. Biol. Plants 2011, 17, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Cao, K.; Yu, J.; Xu, D.; Ai, K.; Bao, E.; Zou, Z. Exposure to lower red to far-red light ratios improve tomato tolerance to salt stress. BMC Plant Biol. 2018, 18, 92. [Google Scholar] [CrossRef] [PubMed]
- Mollá, S.; Villar-Salvador, P.; García-Fayos, P.; Peñuelas Rubira, J.L. Physiological and transplanting performance of Quercus ilex L. (holm oak) seedlings grown in nurseries with different winter conditions. For. Ecol. Manag. 2006, 237, 218–226. [Google Scholar] [CrossRef]
- Ros, C.; Bell, R.W.; White, P.F. Seedling vigour and the early growth of transplanted rice (Oryza sativa). Plant Soil 2003, 2, 325–337. [Google Scholar] [CrossRef]
- Cerdán PD, C.J. Regulation of flowering time by light quality. Nature 2003, 6942, 881–885. [Google Scholar] [CrossRef]
- Heo, J.W.; Lee, C.W.; Murthy, H.N.; Paek, K.Y. Influence of light quality and photoperiod on flowering of Cyclamen persicum Mill. cv. ‘Dixie White’. Plant Growth Regul. 2003, 1, 7–10. [Google Scholar] [CrossRef]
- Son, K.-H.; Kim, E.-Y.; Oh, M.-M. Growth and development of cherry tomato seedlings grown under various combined ratios of red to blue LED lights and fruit yield and quality after transplanting. Prot. Hortic. Plant Fact. 2018, 27, 54–63. [Google Scholar] [CrossRef]
| Treatment | Fresh Weight (g/per Plant) | Dry Weight (g/per Plant) | Percentage of Shoot Dry Matter (%) | ||||
|---|---|---|---|---|---|---|---|
| Shoot | Root | Whole | Shoot | Root | Whole | ||
| CK | 4.62 ± 0.18 c | 2.52 ± 0.12 ab | 7.13 ± 0.21 cd | 0.31 ± 0.01 cd | 0.45 ± 0.01 a | 0.75 ± 0.02 bc | 6.53 ± 0.13 bc |
| B30 | 4.49 ± 0.11 c | 2.28 ± 0.07 b | 6.77 ± 0.17 d | 0.29 ± 0.01 d | 0.44 ± 0.01 ab | 0.73 ± 0.01 c | 6.67 ± 0.05 ab |
| B60 | 4.34 ± 0.12 c | 2.27 ± 0.05 b | 6.61 ± 0.16 d | 0.29 ± 0.01 d | 0.43 ± 0.01 abc | 0.72 ± 0.01 c | 6.89 ± 0.09 a |
| UVA2 | 5.21 ± 0.21 b | 2.43 ± 0.08 ab | 7.64 ± 0.27 bc | 0.34 ± 0.017 bc | 0.44 ± 0.01 ab | 0.78 ± 0.02 ab | 6.58 ± 0.10 bc |
| UVA6 | 5.37 ± 0.19 ab | 2.46 ± 0.07 ab | 7.82 ± 0.25 ab | 0.36 ± 0.01 ab | 0.45 ± 0.01 a | 0.81 ± 0.02 a | 6.88 ± 0.06 a |
| FR20 | 5.50 ± 0.16 ab | 2.42 ± 0.06 ab | 7.92 ± 0.19 ab | 0.35 ± 0.01 ab | 0.43 ± 0.01 bc | 0.78 ± 0.01 ab | 6.35 ± 0.07 c |
| FR30 | 5.86 ± 0.21 a | 2.56 ± 0.11 a | 8.42 ± 0.28 a | 0.38 ± 0.02 a | 0.42 ± 0.01 c | 0.80 ± 0.02 a | 6.43 ± 0.07 bc |
| Treatment | Compactness | Root-Shoot Ratio | Specific Leaf Weight | The Seedling Index |
|---|---|---|---|---|
| CK | 0.03 ± 0.00 b | 0.27 ± 0.02 a | 1.53 ± 0.03 b | 0.210 ± 0.014 b |
| B30 | 0.03 ± 0.00 b | 0.30 ± 0.01 a | 1.33 ± 0.02 c | 0.242 ± 0.009 a |
| B60 | 0.03 ± 0.00 a | 0.32 ± 0.03 a | 1.65 ± 0.02 a | 0.248 ± 0.007 a |
| UVA2 | 0.03 ± 0.00 b | 0.28 ± 0.02 a | 1.48 ± 0.03 b | 0.236 ± 0.010 a |
| UVA6 | 0.03 ± 0.00 ab | 0.27 ± 0.01 a | 1.50 ± 0.03 b | 0.246 ± 0.006 a |
| FR20 | 0.02 ± 0.00 c | 0.21 ± 0.01 b | 1.22 ± 0.02 d | 0.182 ± 0.008 c |
| FR30 | 0.02 ± 0.00 c | 0.19 ± 0.02 b | 1.18 ± 0.02 d | 0.153 ± 0.007 d |
| Treatment | Photosynthetic Pigment Content (mg/g FW) | Fv/Fm | Y(II) | ETR | |||
|---|---|---|---|---|---|---|---|
| Chl a | Chl b | Total chl | Carotenoids | ||||
| CK | 1.14 ± 0.04 cd | 2.52 ± 0.12 ab | 1.51 ± 0.05 bc | 0.23 ± 0.00 ab | 0.75 ± 0.00 a | 0.59 ± 0.01 ab | 8.89 ± 0.26 ab |
| B30 | 1.20 ± 0.01 bc | 2.28 ± 0.07 b | 1.59 ± 0.01 ab | 0.23 ± 0.00 ab | 0.75 ± 0.00 a | 0.59 ± 0.01 ab | 8.86 ± 0.19 ab |
| B60 | 1.13 ± 0.02 cd | 2.27 ± 0.05 b | 1.50 ± 0.02 bc | 0.22 ± 0.00 ab | 0.76 ± 0.00 a | 0.60 ± 0.02 a | 9.13 ± 0.58 a |
| UVA2 | 1.31 ± 0.05 a | 2.43 ± 0.08 ab | 1.61 ± 0.09 ab | 0.24 ± 0.01 a | 0.75 ± 0.00 a | 0.59 ± 0.01 ab | 8.83 ± 0.19 ab |
| UVA6 | 1.23 ± 0.02 a | 2.46 ± 0.07 ab | 1.65 ± 0.026 a | 0.23 ± 0.00 ab | 0.75 ± 0.00 a | 0.60 ± 0.01 a | 9.05 ± 0.32 a |
| FR20 | 1.06 ± 0.04 d | 2.42 ± 0.06 ab | 1.42 ± 0.05 c | 0.21 ± 0.01 c | 0.75 ± 0.00 a | 0.57 ± 0.01 b | 8.52 ± 0.29 b |
| FR30 | 1.12 ± 0.02 cd | 2.56 ± 0.11 a | 1.51 ± 0.03 bc | 0.22 ± 0.00 bc | 0.76 ± 0.00 a | 0.58 ± 0.01 ab | 8.81 ± 0.27 ab |
| Treatment | Time of First Flower (days) | The Node of First Flower | Number of Fruits on the 15th Day | Number Of Fruits At The 30th Day | Average Number of Fruits per Plant | Average Weight of Fruits per Plant (g) |
|---|---|---|---|---|---|---|
| CK | 15.3 ± 0.3 a | 8.1 ± 0.1 bc | 0.2 ± 0.1 ab | 3.1 ± 0.5 ab | 5.0 ± 0.3 b | 13.8 ± 0.6 bc |
| B30 | 14.7 ± 0.3 a | 7.8 ± 0.1 c | 0.2 ± 0.1 ab | 2.5 ± 0.4 ab | 5.2 ± 0.3 b | 16.5 ± 1.9 ab |
| B60 | 14.8 ± 0.3 a | 8.0 ± 0.2 bc | 0.2 ± 0.1 ab | 3.2 ± 0.5 ab | 6.1 ± 0.2 a | 14.6 ± 0.5 abc |
| UVA2 | 14.8 ± 0.3 a | 7.9 ± 0.1 c | 0.3 ± 0.1 a | 3.8 ± 0.4 a | 5.4 ± 0.3 ab | 15.0 ± 0.7 abc |
| UVA6 | 14.9 ± 0.3 a | 8.1 ± 0.1 bc | 0.2 ± 0.1 ab | 2.7 ± 0.3 ab | 5.6 ± 0.3 ab | 17.3 ± 1.2 a |
| FR20 | 15.3 ± 0.3 a | 8.6 ± 0.1 a | 0.2 ± 0.1 ab | 2.5 ± 0.4 ab | 5.3 ± 0.3 ab | 11.7 ± 1.0 c |
| FR30 | 15.5 ± 0.3 a | 8.4 ± 0.2 ab | 0.0 ± 0.0 b | 2.1 ± 0.4 b | 5.1 ± 0.3 b | 11.9 ± 1.2 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, X.; Shi, R.; Gao, M.; He, R.; Li, Y.; Liu, H. Effects of LED Light Quality on the Growth of Pepper (Capsicum spp.) Seedlings and the Development after Transplanting. Agronomy 2022, 12, 2269. https://doi.org/10.3390/agronomy12102269
Liu X, Shi R, Gao M, He R, Li Y, Liu H. Effects of LED Light Quality on the Growth of Pepper (Capsicum spp.) Seedlings and the Development after Transplanting. Agronomy. 2022; 12(10):2269. https://doi.org/10.3390/agronomy12102269
Chicago/Turabian StyleLiu, Xiaojuan, Rui Shi, Meifang Gao, Rui He, Yamin Li, and Houcheng Liu. 2022. "Effects of LED Light Quality on the Growth of Pepper (Capsicum spp.) Seedlings and the Development after Transplanting" Agronomy 12, no. 10: 2269. https://doi.org/10.3390/agronomy12102269
APA StyleLiu, X., Shi, R., Gao, M., He, R., Li, Y., & Liu, H. (2022). Effects of LED Light Quality on the Growth of Pepper (Capsicum spp.) Seedlings and the Development after Transplanting. Agronomy, 12(10), 2269. https://doi.org/10.3390/agronomy12102269









