Comparison of Hyperspectral Imagery and Physiological Characteristics of Bentazone-Tolerant and -Susceptible Soybean Cultivars
Abstract
1. Introduction
2. Materials and Methods
2.1. Experiment 1: Evaluation of the Reaction to Bentazone with Visual Scores
2.1.1. Soybean Cultivars and Herbicide Treatments
2.1.2. Visual Determination of Injury
2.2. Experiment 2: Physiological Assessments and Biomass of Bentazone-Treated Soybeans
Soybean Cultivars, Treatment, and Plot Design
2.3. Hyperspectral Imagery Collection
2.4. Calculation of Normalized Difference Vegetation Index in Hyperspectral Images
2.5. Determination of Photosynthetic Parameters
2.6. Fresh Weight and Plant Height
2.7. Statistical Analysis
3. Results
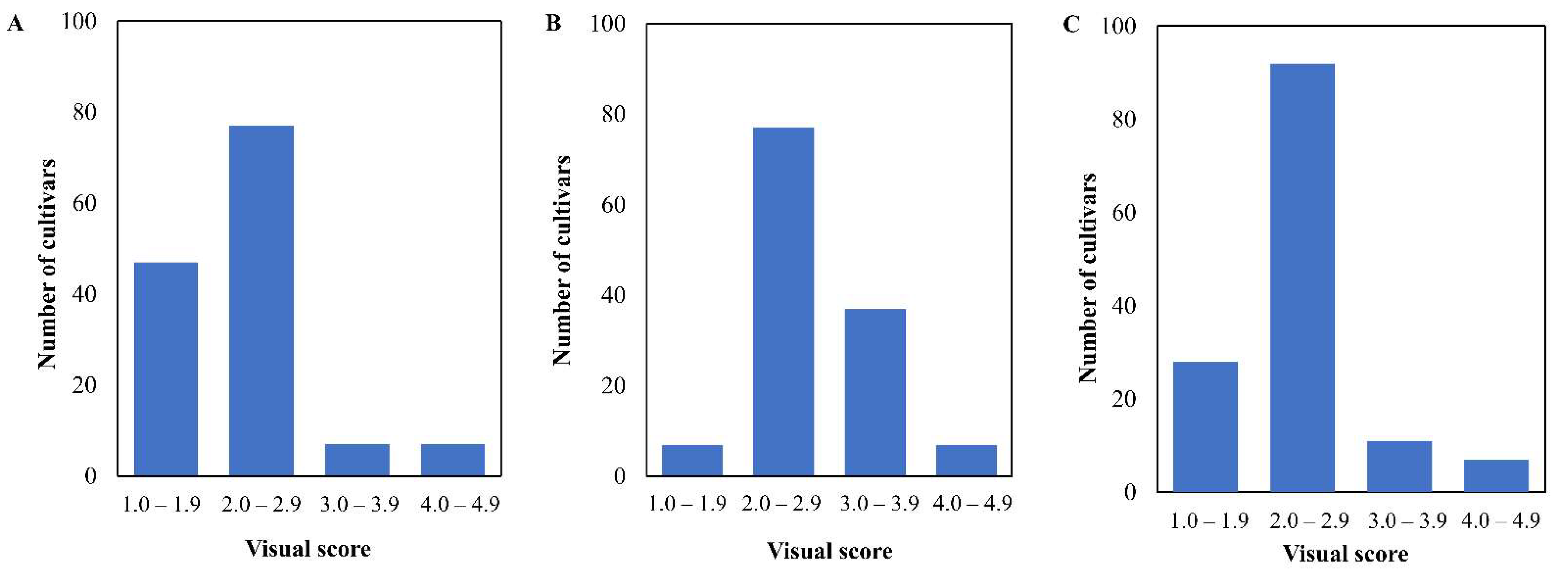
3.1. Phenotypic Distribution of Bentazone Tolerance in Korean Soybean Cultivars
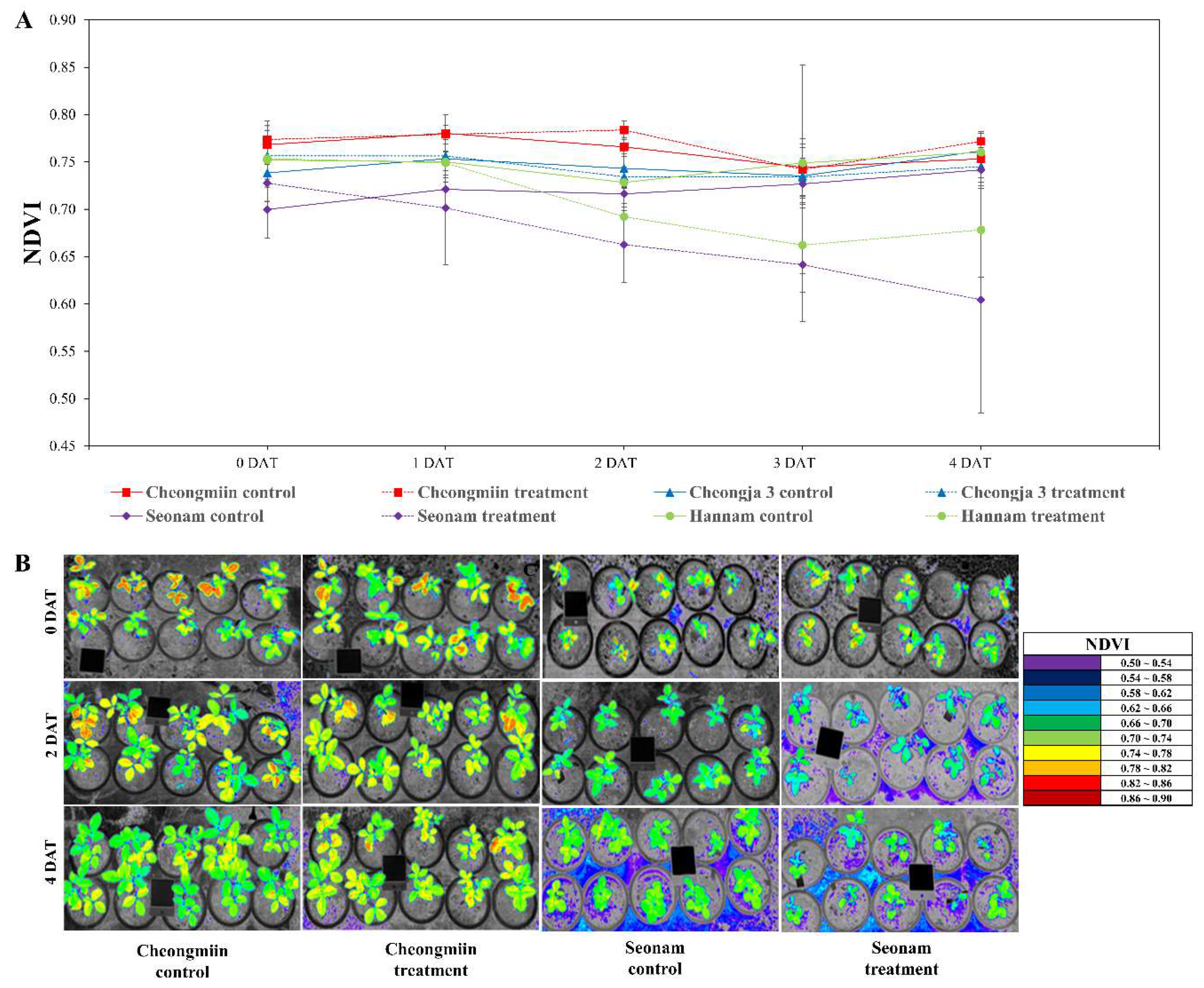
3.2. Determination of Leaf Greenness Using Hyperspectral Images of NDVI
3.3. Investigation of Photosynthesis-Related Parameters
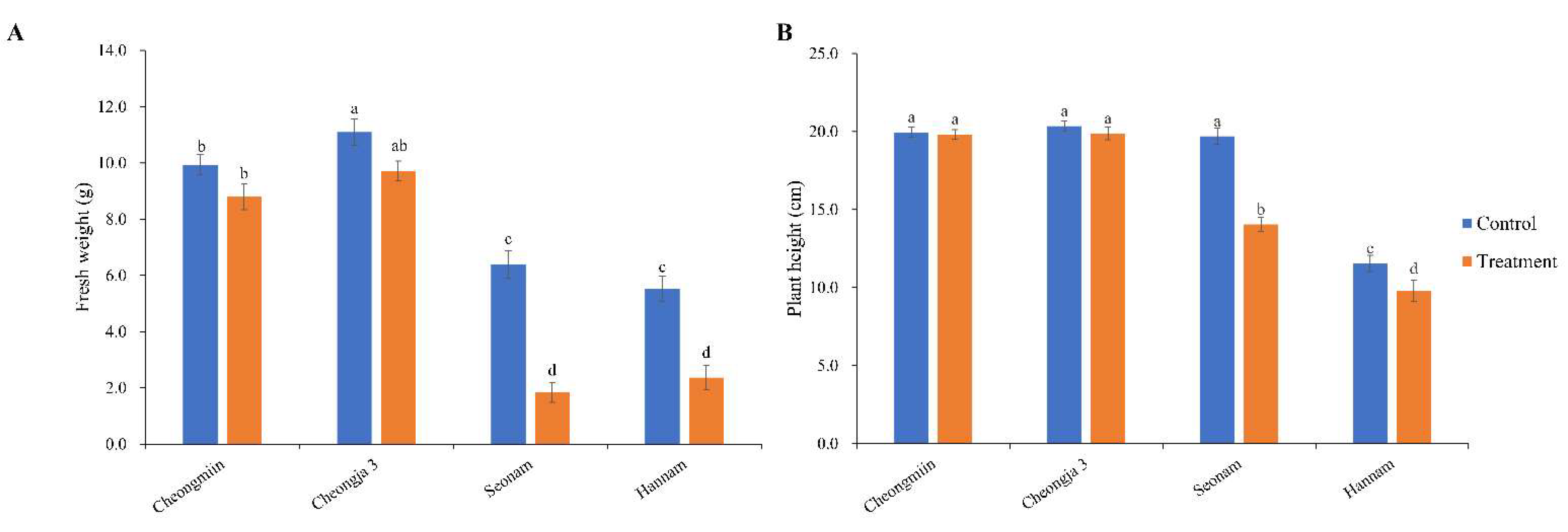
3.4. Effects on Plant Growth
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Czékus, Z.; Farkas, M.; Bakacsy, L.; Ördög, A.; Gallé, Á.; Poór, P. Time-Dependent Effects of Bentazon Application on the Key Antioxidant Enzymes of Soybean and Common Ragweed. Sustainability 2020, 12, 3872. [Google Scholar] [CrossRef]
- Hartman, G.L.; West, E.D.; Herman, T.K. Crops that feed the World 2. Soybean—worldwide production, use, and constraints caused by pathogens and pests. Food Secur. 2011, 3, 5–17. [Google Scholar] [CrossRef]
- Shin, D.; Jeong, D. Korean traditional fermented soybean products: Jang. J. Ethn. Foods 2015, 2, 2–7. [Google Scholar] [CrossRef]
- Jańczak-Pieniążek, M.; Buczek, J.; Bobrecka-Jamro, D.; Szpunar-Krok, E.; Tobiasz-Salach, R.; Jarecki, W. Morphophysiology, Productivity and Quality of Soybean (Glycine max (L.) Merr.) cv. Merlin in Response to Row Spacing and Seeding Systems. Agronomy 2021, 11, 403. [Google Scholar] [CrossRef]
- Lamichhane, J.R.; Devos, Y.; Beckie, H.; Owen, M.D.K.; Tillie, P.; Messéan, A.; Kudsk, P. Integrated weed management systems with herbicide-tolerant crops in the European Union: Lessons learnt from home and abroad. Crit. Rev. Biotechnol. 2017, 37, 459–475. [Google Scholar] [CrossRef]
- Meseldžija, M.; Rajković, M.; Dudić, M.; Vranešević, M.; Bezdan, A.; Jurišić, A.; Ljevnaić-Mašić, B. Economic Feasibility of Chemical Weed Control in Soybean Production in Serbia. Agronomy 2020, 10, 291. [Google Scholar] [CrossRef]
- Kato, S.; Yokota, Y.; Suzuki, R.; Fujisawa, Y.; Sayama, T.; Kaga, A.; Anai, T.; Komatsu, K.; Oki, N.; Kikuchi, A.; et al. Identification of a cytochrome P450 hydroxylase, CYP81E22, as a causative gene for the high sensitivity of soybean to herbicide bentazon. Theor. Appl. Genet. 2020, 133, 2105–2115. [Google Scholar] [CrossRef]
- Jhala, A.J.; Sandell, L.D.; Kruger, G.R. Control of Glyphosate-Resistant Giant Ragweed (Ambrosia trifida L.) with 2,4-D Followed by Pre-Emergence or Post-Emergence Herbicides in Glyphosate-Resistant Soybean (Glycine max L.). Am. J. Plant Sci. 2014, 5, 2289–2297. [Google Scholar] [CrossRef]
- Oliveira, M.C.; Feist, D.; Eskelsen, S.; Scott, J.E.; Knezevic, S.Z. Weed Control in Soybean with Preemergence- and Postemergence-applied Herbicides. Crop. Forage Turfgrass Manag. 2017, 3, 1–7. [Google Scholar] [CrossRef]
- Ali, L.; Jo, H.; Song, J.T.; Lee, J.-D. The Prospect of Bentazone-Tolerant Soybean for Conventional Cultivation. Agronomy 2020, 10, 1650. [Google Scholar] [CrossRef]
- De Sousa, C.P.; De Farias, M.E.; Schock, A.A.; Bacarin, M.A. Photosynthesis of soybean under the action of a photosystem II-inhibiting herbicide. Acta Physiol. Plant. 2014, 36, 3051–3062. [Google Scholar] [CrossRef]
- Fang, Y.; Lu, H.; Chen, S.; Zhu, K.; Song, H.; Qian, H. Leaf proteome analysis provides insights into the molecular mechanisms of bentazon detoxification in rice. Pestic. Biochem. Physiol. 2018, 125, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Radwan, D.E.; Mohamed, A.K.; Fayez, K.A.; Abdelrahman, A.M. Oxidative stress caused by Basagran® herbicide is altered by salicylic acid treatments in peanut plants. Heliyon 2019, 5, e01791. [Google Scholar] [CrossRef] [PubMed]
- Agostinetto, D.; Perboni, L.; Langaro, A.; Gomes, J.; Fraga, D.; Franco, J. Changes in Photosynthesis and Oxidative Stress in Wheat Plants Submmited to Herbicides Application. Planta Daninha 2016, 34, 1–9. [Google Scholar] [CrossRef]
- Hartwing, I.; Bertan, I.; Galon, L.; Noldin, J.; Concenço, G.; Silva, A.; Aspiazú, I.; Ferreira, E. Tolerância de trigo (Triticum aestivum) e aveia (Avena sp.) a herbicidas inibidores da enzima acetolactato sintase (ALS). Planta Daninha 2008, 26, 361–368. [Google Scholar] [CrossRef]
- Zhu, J.; Patzoldt, W.L.; Radwan, O.; Tranel, P.J.; Clough, S.J. Effects of Photosystem-II-Interfering Herbicides Atrazine and Bentazon on the Soybean Transcriptome. Plant Genome 2019, 2, 191–205. [Google Scholar] [CrossRef]
- Ranjan, P.N.; Ram, C.J.; Anurag, T.; Nilesh, J.; Kumar, P.B.; Suresh, Y.; Rahul, K. Breeding for herbicide tolerance in crops: A review. Res. J. Biotechnol. 2020, 15, 154–162. [Google Scholar]
- Connelly, J.A.; Johnson, M.D.; Gronwald, J.W.; Wyse, D.L. Bentazon Metabolism in Tolerant and Susceptible Soybean (Glycine max) Genotypes. Weed Sci. 1988, 36, 417–423. [Google Scholar] [CrossRef]
- Hayes, R.M.; Wax, L.M. Differential Intraspecific Responses of Soybean Cultivars to Bentazon. Weed Sci. 1975, 23, 516–521. [Google Scholar] [CrossRef]
- Darmency, H.; Pernès, J. Agronomic performance of a triazine resistant foxtail millet (Setaria italica (L.) Beauv.). Weed Res. 1989, 29, 147–150. [Google Scholar] [CrossRef]
- Samtani, J.B.; Rachuy, J.S.; Mou, B.; Fennimore, S.A. Evaluation of Tribenuron-Methyl on Sulfonylurea-Resistant Lettuce Germplasm. Weed Technol. 2014, 28, 510–517. [Google Scholar] [CrossRef]
- Durigon, M.; Camera, A.; Cechin, J.; Vargas, L.; Chavarria, G. Does Spraying of Atrazine on Triazine-Resistant Canola Hybrid Impair Photosynthetic Processes? Planta Daninha 2019, 37, 1–11. [Google Scholar] [CrossRef]
- Shi, C.; Zheng, Y.; Geng, J.; Liu, C.; Pei, H.; Ren, Y.; Dong, Z.; Zhao, L.; Zhang, N.; Chen, F. Identification of herbicide resistance loci using a genome-wide association study and linkage mapping in Chinese common wheat. Crop J. 2020, 8, 666–675. [Google Scholar] [CrossRef]
- Gaur, P.M.; Jukanti, A.K.; Samineni, S.; Chaturvedi, S.K.; Singh, S.; Tripathi, S.; Singh, I.; Singh, G.; Das, T.K.; Aski, M.; et al. Large Genetic Variability in Chickpea for Tolerance to Herbicides Imazethapyr and Metribuzin. Agronomy 2013, 3, 524–536. [Google Scholar] [CrossRef]
- Suarez, L.A.; Apan, A.; Werth, J. Detection of phenoxy herbicide dosage in cotton crops through the analysis of hyperspectral data. Plant Life Under Chang. Environ. 2017, 38, 237–250. [Google Scholar] [CrossRef]
- Mahlein, A.-K.; Rumpf, T.; Welke, P.; Dehne, H.-W.; Plümer, L.; Steiner, U.; Oerke, E.-C. Development of spectral indices for detecting and identifying plant diseases. Remote Sens. Environ. 2013, 128, 21–30. [Google Scholar] [CrossRef]
- Moghadam, P.; Ward, D.; Goan, E.; Jayawardena, S.; Sikka, P.; Hernandez, E. Plant Disease Detection Using Hyperspectral Imaging. In Proceedings of the 2017 International Conference on Digital Image Computing: Techniques and Applications (DICTA), Sydney, NSW, Australia, 29 November–1 December 2017. [Google Scholar] [CrossRef]
- Yang, G.; Liu, J.; Zhao, C.; Li, Z.; Huang, Y.; Yu, H.; Xu, B.; Yang, X.; Zhu, D.; Zhang, X.; et al. Unmanned Aerial Vehicle Remote Sensing for Field-Based Crop Phenotyping: Current Status and Perspectives. Front. Plant Sci. 2017, 8, 1111. [Google Scholar] [CrossRef]
- Li, X.; Li, R.; Wang, M.; Liu, Y.; Zhang, B.; Zhou, B.Z.A.J. Hyperspectral Imaging and Their Applications in the Nondestructive Quality Assessment of Fruits and Vegetables. In Hyperspectral Imaging in Agriculture, Food and Environment; IntechOpen: London, UK, 2018; pp. 27–63. [Google Scholar] [CrossRef]
- Sytar, O.; Brestic, M.; Zivcak, M.; Olsovska, K.; Kovar, M.; Shao, H.; He, X. Applying hyperspectral imaging to explore natural plant diversity towards improving salt stress tolerance. Sci. Total Environ. 2017, 578, 90–99. [Google Scholar] [CrossRef]
- Thomas, S.; Kuska, M.T.; Bohnenkamp, D.; Brugger, A.; Alisaac, E.; Wahabzada, M.; Behmann, J.; Mahlein, A.-K. Benefits of hyperspectral imaging for plant disease detection and plant protection: A technical perspective. J. Plant Dis. Prot. 2018, 125, 5–20. [Google Scholar] [CrossRef]
- Kuska, M.T.; Mahlein, A.-K. Aiming at decision making in plant disease protection and phenotyping by the use of optical sensors. Eur. J. Plant Pathol. 2018, 152, 987–992. [Google Scholar] [CrossRef]
- Lowe, A.; Harrison, N.; French, A.P. Hyperspectral image analysis techniques for the detection and classification of the early onset of plant disease and stress. Plant Methods 2017, 13, 80. [Google Scholar] [CrossRef] [PubMed]
- Tayade, R.; Yoon, J.; Lay, L.; Khan, A.L.; Yoon, Y.; Kim, Y. Utilization of Spectral Indices for High-Throughput Phenotyping. Plants 2022, 11, 1712. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Tiwari, S. Responses of plants to herbicides: Recent advances and future prospectives. In Plant Life Under Changing Environment; Academic Press: Cambridge, MA, USA, 2020; pp. 237–250. [Google Scholar] [CrossRef]
- Ryu, J.-H.; Jeong, H.; Cho, J. Performances of Vegetation Indices on Paddy Rice at Elevated Air Temperature, Heat Stress, and Herbicide Damage. Remote Sens. 2020, 12, 2654. [Google Scholar] [CrossRef]
- Cao, Z.; Yao, X.; Liu, H.; Liu, B.; Cheng, T.; Tian, Y.; Cao, W.; Zhu, Y. Comparison of the abilities of vegetation indices and photosynthetic parameters to detect heat stress in wheat. Agric. For. Meteorol. 2019, 265, 121–136. [Google Scholar] [CrossRef]
- Polder, G.; van der Heijden, G.W.; van Doorn, J.; Baltissen, T.A. Automatic detection of tulip breaking virus (TBV) in tulip fields using machine vision. Biosyst. Eng. 2014, 117, 35–42. [Google Scholar] [CrossRef]
- Wijekoon, C.; Goodwin, P.; Hsiang, T. Quantifying fungal infection of plant leaves by digital image analysis using Scion Image software. J. Microbiol. Methods 2008, 74, 94–101. [Google Scholar] [CrossRef]
- Neto, A.J.S.; Lopes, D.C.; Pinto, F.A.C.; Zolnier, S. Vis/NIR spectroscopy and chemometrics for non-destructive estimation of water and chlorophyll status in sunflower leaves. Biosyst. Eng. 2017, 155, 124–133. [Google Scholar] [CrossRef]
- Behmann, J.; Acebron, K.; Emin, D.; Bennertz, S.; Matsubara, S.; Thomas, S.; Bohnenkamp, D.; Kuska, M.T.; Jussila, J.; Salo, H.; et al. Specim IQ: Evaluation of a New, Miniaturized Handheld Hyperspectral Camera and Its Application for Plant Phenotyping and Disease Detection. Sensors 2018, 18, 441. [Google Scholar] [CrossRef]
- Teena, M.; Manickavasagan, A.; Mothershaw, A.; El Hadi, S.; Jayas, D.S. Potential of Machine Vision Techniques for Detecting Fecal and Microbial Contamination of Food Products: A Review. Food Bioprocess Technol. 2013, 6, 1621–1634. [Google Scholar] [CrossRef]
- Behmann, J.; Steinrücken, J.; Plümer, L. Detection of early plant stress responses in hyperspectral images. ISPRS J. Photogramm. Remote Sens. 2014, 93, 98–111. [Google Scholar] [CrossRef]
- Terentev, A.; Dolzhenko, V.; Fedotov, A.; Eremenko, D. Current State of Hyperspectral Remote Sensing for Early Plant Disease Detection: A Review. Sensors 2022, 22, 757. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, L.; Scherner, A.; Cuchiara, C.C.; Moraes, L.; Avila, L.A.; Deuner, S. Soybean plant osmotic and oxidative stress as affected by herbicide and salinity levels in soil. Planta Daninha 2020, 38, 1–7. [Google Scholar] [CrossRef]
- Hassannejad, S.; Ghafarbi, S.P.; Lotfi, R. The effect of nicosulfuron and bentazon on photosynthetic performance of common cocklebur (Xanthium strumarium L.). Environ. Sustain. Indic. 2020, 6, 100026. [Google Scholar] [CrossRef]
- Bigot, A.; Fontaine, F.; Clément, C.; Vaillant-Gaveau, N. Effect of the herbicide flumioxazin on photosynthetic performance of grapevine (Vitis vinifera L.). Chemosphere 2007, 67, 1243–1251. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, P.; Weber, J.F.; Gerhards, R. Early Identification of Herbicide Stress in Soybean (Glycine max (L.) Merr.) Using Chlorophyll Fluorescence Imaging Technology. Sensors 2018, 18, 21. [Google Scholar] [CrossRef]
- Radwan, D.E.M.; Fayez, K.A. Photosynthesis, antioxidant status and gas-exchange are altered by glyphosate application in peanut leaves. Photosynthetica 2016, 54, 307–316. [Google Scholar] [CrossRef]
- Sun, L.; Xu, H.; Hao, H.; An, S.; Lu, C.; Wu, R.; Su, W. Effects of bensulfuron-methyl residue on photosynthesis and chlorophyll fluorescence in leaves of cucumber seedlings. PLoS ONE 2019, 14, e0215486. [Google Scholar] [CrossRef]
- Reiling, K.L.; Simmons, F.W.; Riechers, D.E.; Steckel, L.E. Application timing and soil factors affect sulfentrazone phytotoxicity to two soybean (Glycine max (L.) Merr.) cultivars. Crop Prot. 2006, 25, 230–234. [Google Scholar] [CrossRef]
- Zobiole, L.H.S.; Bonini, E.A.; de Oliveira, R.S.; Kremer, R.J.; Ferrarese-Filho, O. Glyphosate affects lignin content and amino acid production in glyphosate-resistant soybean. Acta Physiol. Plant. 2010, 32, 831–837. [Google Scholar] [CrossRef]
- Matzrafi, M.; Herrmann, I.; Nansen, C.; Kliper, T.; Zait, Y.; Ignat, T.; Siso, D.; Rubin, B.; Karnieli, A.; Eizenberg, H. Hyperspectral Technologies for Assessing Seed Germination and Trifloxysulfuron-methyl Response in Amaranthus palmeri (Palmer Amaranth). Front. Plant Sci. 2017, 8, 474. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, C.; Shi, Y.; Long, M.; Islam, F.; Yang, C.; Yang, S.; He, Y.; Zhou, W. Evaluation of quinclorac toxicity and alleviation by salicylic acid in rice seedlings using ground-based visible/near-infrared hyperspectral imaging. Plant Methods 2020, 16, 30. [Google Scholar] [CrossRef] [PubMed]
- Cai, K.; Chen, X.; Han, Z.; Wu, X.; Zhang, S.; Li, Q.; Nazir, M.M.; Zhang, G.; Zeng, F. Screening of Worldwide Barley Collection for Drought Tolerance: The Assessment of Various Physiological Measures as the Selection Criteria. Front. Plant Sci. 2020, 11, 1159. [Google Scholar] [CrossRef] [PubMed]
- Wijewardana, C.; Alsajri, F.A.; Irby, J.T.; Krutz, L.J.; Golden, B.; Henry, W.B.; Gao, W.; Reddy, K.R. Physiological assessment of water deficit in soybean using midday leaf water potential and spectral features. J. Plant Interact. 2019, 14, 533–543. [Google Scholar] [CrossRef]
- Fehr, W.R.; Caviness, C.E.; Burmood, D.T.; Pennington, J.S. Stage of Development Descriptions for Soybeans, Glycine Max (L.) Merrill. Crop Sci. 1971, 11, 929–931. [Google Scholar] [CrossRef]
- Ali, L.; Jo, H.; Song, J.-T.; Lee, J.-D. Evaluation of Bentazone Reaction by Growth Stage and Bentazone Dose Across Soybean Genotypes. Plant Breed. Biotechnol. 2021, 9, 331–344. [Google Scholar] [CrossRef]
- Liu, M.; Liu, X.; Li, M.; Fang, M.; Chi, W. Neural-network model for estimating leaf chlorophyll concentration in rice under stress from heavy metals using four spectral indices. Biosyst. Eng. 2010, 106, 223–233. [Google Scholar] [CrossRef]
- Oerke, E.-C.; Dehne, H.-W. Safeguarding production—losses in major crops and the role of crop protection. Crop Prot. 2004, 23, 275–285. [Google Scholar] [CrossRef]
- Wax, L.M.; Bernard, R.L.; Hayes, R.M. Response of Soybean Cultivars to Bentazon, Bromoxynil, Chloroxuron, and 2,4-DB. Weed Sci. 1974, 22, 35–41. [Google Scholar] [CrossRef]
- Xiao, Q.; Bai, X.; Zhang, C.; He, Y. Advanced high-throughput plant phenotyping techniques for genome-wide association studies: A review. J. Adv. Res. 2021, 35, 215–230. [Google Scholar] [CrossRef]
- Agudelo, C.A.B. Effects of Fungicides on Physiological Parameters and Yield Formation of Wheat Assessed by Non-Invasive Sensors. Doctoral Dissertation, Universitäts-und Landesbibliothek Bonn, Bonn, Germany, 2014. [Google Scholar]
- Bloem, E.; Gerighausen, H.; Chen, X.; Schnug, E. The Potential of Spectral Measurements for Identifying Glyphosate Application to Agricultural Fields. Agronomy 2020, 10, 1409. [Google Scholar] [CrossRef]
- Henry, W.B.; Shaw, D.R.; Reddy, K.R.; Bruce, L.M.; Tamhankar, H.D. Remote Sensing to Detect Herbicide Drift on Crops. Weed Technol. 2004, 18, 358–368. [Google Scholar] [CrossRef]
- Huang, Y.; Yuan, L.; Reddy, K.N.; Zhang, J. In-situ plant hyperspectral sensing for early detection of soybean injury from dicamba. Biosyst. Eng. 2016, 149, 51–59. [Google Scholar] [CrossRef]
- Hinojosa, L.; Kumar, N.; Gill, K.S.; Murphy, K.M. Spectral Reflectance Indices and Physiological Parameters in Quinoa under Contrasting Irrigation Regimes. Crop Sci. 2019, 59, 1927–1944. [Google Scholar] [CrossRef]
- Sharma, A.; Kumar, V.; Shahzad, B.; Ramakrishnan, M.; Sidhu, G.P.S.; Bali, A.S.; Handa, N.; Kapoor, D.; Yadav, P.; Khanna, K.; et al. Photosynthetic Response of Plants Under Different Abiotic Stresses: A Review. J. Plant Growth Regul. 2020, 39, 509–531. [Google Scholar] [CrossRef]
- Hamim, H.; Violita, V.; Triadiati, T.; Miftahudin, M. Oxidative Stress and Photosynthesis Reduction of Cultivated (Glycine max L.) and Wild Soybean (G. tomentella L.) Exposed to Drought and Paraquat. Asian J. Plant Sci. 2017, 16, 65–77. [Google Scholar] [CrossRef][Green Version]
- Tao, B.; Sun, S.; Zhang, L.; Guo, J.; Shao, B. Production and Assessment of Potato Material Resistant to the Broadleaf Herbicide Bentazone. Potato Res. 2021, 64, 241–256. [Google Scholar] [CrossRef]
- Vivancos, P.D.; Driscoll, S.P.; Bulman, C.A.; Ying, L.; Emami, K.; Treumann, A.; Mauve, C.; Noctor, G.; Foyer, C.H. Perturbations of Amino Acid Metabolism Associated with Glyphosate-Dependent Inhibition of Shikimic Acid Metabolism Affect Cellular Redox Homeostasis and Alter the Abundance of Proteins Involved in Photosynthesis and Photorespiration. Plant Physiol. 2011, 157, 256–268. [Google Scholar] [CrossRef]
- Silva, D.; Vargas, L.; Agostinetto, D.; Santos, F. Photosynthetic Performance of Glyphosate Resistant and Glyphosate Susceptible Hairy Fleabane under Light Intensity. Planta Daninha 2018, 35, 1–8. [Google Scholar] [CrossRef]
- Alla, M.M.N.; Hassan, N.M.; El-Bastawisy, Z.M. Changes in antioxidants and kinetics of glutathione-S-transferase of maize in response to isoproturon treatment. Plant Biosyst. 2008, 142, 5–16. [Google Scholar] [CrossRef]
- Zobiole, L.H.S.; Kremer, R.J.; de Oliveira, R.S., Jr.; Constantin, J. Glyphosate effects on photosynthesis, nutrient accumulation, and nodulation in glyphosate-resistant soybean. J. Plant. Nutr. Soil Sci. 2012, 175, 319–330. [Google Scholar] [CrossRef]

| Source of Variation | Degree of Freedom | Sum Square | Mean Square | F-Value | p-Value |
|---|---|---|---|---|---|
| Test | 1 | 17.8 | 17.8 | 87.2 | <0.0001 |
| Replication in test | 2 | 3.9 | 1.9 | 9.5 | 0.0001 |
| Cultivar | 137 | 138.7 | 1.0 | 5.0 | <0.0001 |
| Test × Cultivar | 127 | 118.7 | 0.9 | 4.6 | <0.0001 |
| Error | 264 | 53.8 | 0.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, L.; Jo, H.; Choi, S.M.; Kim, Y.; Song, J.T.; Lee, J.-D. Comparison of Hyperspectral Imagery and Physiological Characteristics of Bentazone-Tolerant and -Susceptible Soybean Cultivars. Agronomy 2022, 12, 2241. https://doi.org/10.3390/agronomy12102241
Ali L, Jo H, Choi SM, Kim Y, Song JT, Lee J-D. Comparison of Hyperspectral Imagery and Physiological Characteristics of Bentazone-Tolerant and -Susceptible Soybean Cultivars. Agronomy. 2022; 12(10):2241. https://doi.org/10.3390/agronomy12102241
Chicago/Turabian StyleAli, Liakat, Hyun Jo, Seung Min Choi, Yoonha Kim, Jong Tae Song, and Jeong-Dong Lee. 2022. "Comparison of Hyperspectral Imagery and Physiological Characteristics of Bentazone-Tolerant and -Susceptible Soybean Cultivars" Agronomy 12, no. 10: 2241. https://doi.org/10.3390/agronomy12102241
APA StyleAli, L., Jo, H., Choi, S. M., Kim, Y., Song, J. T., & Lee, J.-D. (2022). Comparison of Hyperspectral Imagery and Physiological Characteristics of Bentazone-Tolerant and -Susceptible Soybean Cultivars. Agronomy, 12(10), 2241. https://doi.org/10.3390/agronomy12102241








