Selenium Biofortification of Lettuce Plants (Lactuca sativa L.) as Affected by Se Species, Se Rate, and a Biochar Co-Application in a Calcareous Soil
Abstract
:1. Introduction
2. Materials and Methods
2.1. Soil Selection and Characteristics
2.2. Experimental Design
2.3. Plant Tissue Analysis
2.4. Statistics
3. Results
3.1. Plant Growth Characteristics
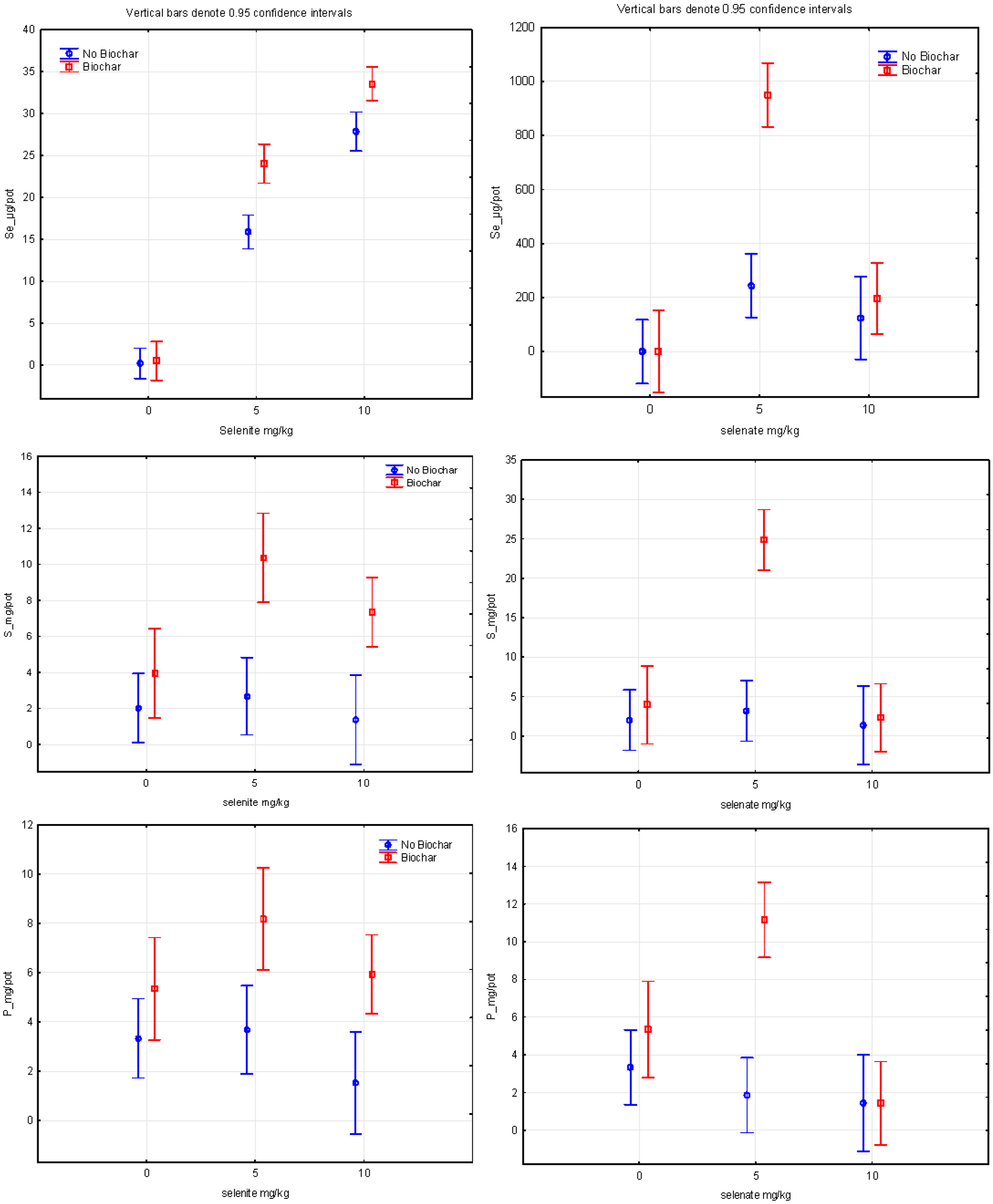
3.2. Selenium Concentration in Lettuce Plants
3.3. Sulfur Concentration in Lettuce Plants
3.4. Phosphorus Concentration in Lettuce Plants
4. Discussion
Total Uptake of Selenium, Sulfur and Phosphorous
5. Conclusions
- Biochar application reduced Se uptake with high selenite application rate and protected plants from Se toxicity leading also to the highest Se uptake (μg/pot). On the contrary, in the high selenate rate, biochar failed to secure plants from impaired growth probably due to the much higher mobility of selenate and availability in the alkaline soil environment compared to selenite.
- Visual and measurable toxic symptoms were observed in plants receiving the high selenate rate especially in the presence of biochar and the high selenite rate in the absence of biochar, as indicated by the decreased dry weight of the plants and the lower SPAD and NDVI values.
- In selenate treatments, Se concentration in lettuce shoots was up to 10 times higher than in the selenite treatments, verifying the greater mobility of selenates in the alkaline soil environment, and the higher translocation of selenate from roots to shoots.
- Most of the Se in selenite treatments remained in roots, regardless of the presence or absence of biochar. The opposite was observed in selenate treatments, i.e., most of the Se transferred to shoots, also irrespectively of biochar application.
- In general, biochar application increased S and P concentration in plants when selenium was applied in any form.
- The significant positive correlations between Se, S, and P total uptake by plants (mg/pot) of selenate treatments suggest mutually beneficial interactions between the three nutrients, whereas the significant negative respective correlations in selenite treatments imply antagonistic behavior.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Winkel, L.H.E.; Vriens, B.; Jones, G.D.; Schneider, L.S.; Pilon-Smits, E.; Bañuelos, G.S. Selenium Cycling Across Soil-Plant-Atmosphere Interfaces: A Critical Review. Nutrients 2015, 7, 4199–4239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rayman, M.P. The argument for increasing selenium intake. Proc. Nutr. Soc. 2002, 61, 203–215. [Google Scholar] [CrossRef]
- Holben, D.H.; Smith, A.M. The diverse role of selenium within selenoproteins: A review. J. Am. Diet. Assoc. 1999, 99, 836–843. [Google Scholar] [CrossRef]
- Murdolo, G.; Bartolini, D.; Tortoioli, C.; Piroddi, M.; Torquato, P.; Galli, F. Selenium and Cancer Stem Cells. Adv. Cancer Res. 2017, 136, 235–257. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Gupta, S. An Overview of Selenium Uptake, Metabolism, and Toxicity in Plants. Front. Plant Sci. 2017, 7, 2074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schiavon, M.; Pilon-Smits, E.A.H. The fascinating facets of plant selenium accumulation—Biochemistry, physiology, evolution and ecology. New Phytol. 2017, 213, 1582–1596. [Google Scholar] [CrossRef] [Green Version]
- Chauhan, R.; Awasthi, S.; Srivastava, S.; Dwivedi, S.; Pilon-Smits, E.A.; Dhankher, O.P.; Tripathi, R.D. Understanding selenium metabolism in plants and its role as a beneficial element. Crit. Rev. Environ. Sci. Technol. 2019, 49, 1937–1958. [Google Scholar] [CrossRef]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Frączek, A.; Pasternak, K. Selenium in medicine and treatment. J. Elem. 2013, 18, 145–163. [Google Scholar] [CrossRef]
- Sharma, S.; Kaur, N.; Kaur, S.; Nayyar, H. Selenium as a nutrient in biostimulation and biofortification of cereals. Indian J. Plant Physiol. 2016, 22, 1–15. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Global Health Risks: Mortality and Burden of Disease Attributable to Selected Major Risks; WHO: Geneva, Switzerland, 2009. [Google Scholar]
- Malagoli, M.; Schiavon, M.; Dall’Acqua, S.; Pilon-Smits, E.A.H. Effects of selenium biofortification on crop nutritional quality. Front. Plant Sci. 2015, 6, 280. [Google Scholar] [CrossRef] [Green Version]
- Kieliszek, M.; Błażejak, S. Selenium: Significance, and outlook for supplementation. Nutrition 2013, 29, 713–718. [Google Scholar] [CrossRef]
- Natasha Shahid, M.; Niazi, N.K.; Khalid, S.; Murtaza, B.; Bibi, I.; Rashid, M.I. A critical review of selenium biogeochemical behavior in soil-plant system with an inference to human health. Environ. Pollut. 2018, 234, 915–934. [Google Scholar] [CrossRef]
- Schiavon, M.; Nardi, S.; dalla Vecchia, F.; Ertani, A. Selenium biofortification in the 21st century: Status and challenges for healthy human nutrition. Plant Soil 2020, 453, 245–270. [Google Scholar] [CrossRef]
- Dinh, Q.T.; Cui, Z.; Huang, J.; Tran, T.A.T.; Wang, D.; Yang, W.; Zhou, F.; Wang, M.; Yu, D.; Liang, D. Selenium distribution in the Chinese environment and its relationship with human health: A review. Environ. Int. 2018, 112, 294–309. [Google Scholar] [CrossRef] [PubMed]
- Zafeiriou, I.; Gasparatos, D.; Massas, I. Adsorption/Desorption Patterns of Selenium for Acid and Alkaline Soils of Xerothermic Environments. Environments 2020, 7, 72. [Google Scholar] [CrossRef]
- Hossain, A.; Skalicky, M.; Brestic, M.; Maitra, S.; Sarkar, S.; Ahmad, Z.; Vemuri, H.; Garai, S.; Mondal, M.; Bhatt, R.; et al. Selenium Biofortification: Roles, Mechanisms, Responses and Prospects. Molecules 2021, 26, 881. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Ofner, J.; Kappler, A. Formation of binary and ternary colloids anddissolved complexes of organic matter, Fe and As. Environ. Sci. Technol. 2010, 44, 4479–4485. [Google Scholar] [CrossRef]
- Bassil, J.; Naveau, A.; Bueno, M.; Razack, M.; Kazpard, V. Leaching behavior of selenium from the karst infillings of the Hydrogeological Experimental Site of Poitiers. Chem. Geol. 2018, 483, 141–150. [Google Scholar] [CrossRef]
- De Temmerman, L.; Waegeneers, N.; Thiry, C.; Du Laing, G.; Tack, F.; Ruttens, A. Selenium content of Belgian cultivated soils and its uptake by field crops and vegetables. Sci. Total Environ. 2014, 468–469, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Alfthan, G.; Eurola, M.; Ekholm, P.; Venäläinen, E.; Root, T.; Korkalainen, K.; Hartikainen, H.; Salminen, P.; Hietaniemi, V.; Aspila, P.; et al. Effects of nationwide addition of selenium to fertilizers on foods, and animal and human health in Finland: From deficiency to optimal selenium status of the population. J. Trace Elem. Med. Biol. 2015, 31, 142–147. [Google Scholar] [PubMed]
- Lyons, G. Selenium in cereals: Improving the efficiency of agronomic biofortification in the UK. Plant Soil 2010, 332, 1–4. [Google Scholar] [CrossRef]
- Terry, N.; Zayed, A.M.; de Souza, M.P.; Tarun, A.S. Selenium in higher plants. Annu. Rev. Plant Biol. 2000, 51, 401–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- dos Santos, M.; da Silva Júnior, F.; Muccillo-Baisch, A. Selenium content of Brazilian foods: A review of the literature values. J. Food Compos. Anal. 2017, 58, 10–15. [Google Scholar] [CrossRef]
- Fordyce, F.M. Selenium deficiency and toxicity in the environment. In Essentials of Medical Geology; Selinus, O., Ed.; Elsevier: Amsterdam, The Netherlands, 2005; pp. 373–415. [Google Scholar]
- Paciolla, C.C.; De Leonardis, S.; Dipierro, S. Effects of selenite and selenate on the antioxidant systems in Senecio scandens L. Plant Biosyst. 2011, 145, 253–259. [Google Scholar] [CrossRef]
- Carvalho, K.M.; Gallardo-Williams, M.T.; Benson, R.F.; Martin, D.F. Effects of selenium supplementation on four agricultural crops. J. Agric. Food Chem. 2003, 51, 704–709. [Google Scholar] [CrossRef]
- White, P.J.; Broadley, M.R.; Bowen, H.C.; Johnson, S.E. Selenium and its relationship with sulfur. In Sulfur in Plants An Ecological Perspective; Hawkesford, M.J., De Kok, L.J., Eds.; Springer: Dordrecht, The Netherlands, 2007; Volume 6. [Google Scholar] [CrossRef]
- de Mello Prado, R.; Cruz, F.J.R.; da Cruz Ferreira, R.L. Selenium biofortification and the problem of its safety. In Superfood and Functional Food—An Overview of Their Processing and Utilization; IntechOpen: London, UK, 2017; pp. 229–238. [Google Scholar]
- Zayed, A.; Lytle, C.; Terry, N. Accumulation and volatilization of different chemical species of selenium by plants. Planta 1998, 206, 284–292. [Google Scholar] [CrossRef]
- Hu, T.; Li, H.; Li, J.; Zhao, G.; Wu, W.; Liu, L.; Wang, Q.; Guo, Y. Absorption and Bio-Transformation of Selenium Nanoparticles by Wheat Seedlings (Triticum aestivum L.). Front. Plant Sci. 2018, 9, 597. [Google Scholar] [CrossRef] [Green Version]
- Li, H.F.; McGrath, S.P.; Zhao, F.J. Selenium uptake, translocation and speciation in wheat supplied with selenate or selenite. New Phytol. 2008, 178, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Hawrylak-Nowak, B. Comparative effects of selenite and selenate on growth and selenium accumulation in lettuce plants under hydroponic conditions. Plant. Growth Regul. 2013, 70, 149–157. [Google Scholar] [CrossRef] [Green Version]
- Pezzarossa, B.; Petruzzelli, G.; Petacco, F.; Malorgio, F.; Ferri, T. Absorption of selenium by Lactuca sativa as affected by carboxymethylcellulose. Chemosphere 2007, 67, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Bilias, F.; Nikoli, T.; Kalderis, D.; Gasparatos, D. Towards a Soil Remediation Strategy Using Biochar: Effects on Soil Chemical Properties and Bioavailability of Potentially Toxic Elements. Toxics 2021, 9, 184. [Google Scholar] [CrossRef]
- Jeffery, S.; Verheijen, F.; van der Velde, M.; Bastos, A. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric. Ecosyst. Environ. 2011, 144, 175–187. [Google Scholar] [CrossRef]
- El-Naggar, A.; Lee, S.; Rinklebe, J.; Farooq, M.; Song, H.; Sarmah, A.; Zimmerman, A.; Ahmad, M.; Shaheen, S.; Ok, Y. Biochar application to low fertility soils: A review of current status, and future prospects. Geoderma 2019, 337, 536–554. [Google Scholar] [CrossRef]
- Kuppusamy, S.; Thavamani, P.; Megharaj, M.; Venkateswarlu, K.; Naidu, R. Agronomic and remedial benefits and risks of applying biochar to soil: Current knowledge and future research directions. Environ. Int. 2016, 87, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, T.; Chocano, C.; Moreno, H.L.; Garcia, C. Use of compost as an alternative to conventional inorganic fertilizers in intensive lettuce (Lactuca sativa L.) crops-effect on soil and Plant Soil. Soil Tillage Res. 2016, 160, 14–22. [Google Scholar] [CrossRef]
- Karapouloutidou, S.; Gasparatos, D. Effects of Biostimulant and Organic Amendment on Soil Properties and Nutrient Status of Lactuca Sativa in a Calcareous Saline-Sodic Soil. Agriculture 2019, 9, 164. [Google Scholar] [CrossRef] [Green Version]
- Ramos, S.J.; Faquin, V.; Guilherme, L.R.G.; Castro, E.M.; Ávila, F.W.; Carvalho, G.S.; Bastos, C.E.A.; Oliveira, C. Selenium biofortification and antioxidant activity in lettuce plants fed with selenate and selenite. Plant Soil Environ. 2010, 56, 584–588. [Google Scholar] [CrossRef] [Green Version]
- Page, A.L. (Ed.) Methods of Soil Analysis, Part. 2, 2nd ed.; American Society of Agronomy: Madison, WI, USA, 1982. [Google Scholar]
- Bouyoucos, G.J. A recalibration of the hydrometer method for making mechanical analysis of soils. Agron. J. 1951, 43, 434–438. [Google Scholar] [CrossRef] [Green Version]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon and organic matter. In Methods of Soil Analysis, Part. 2, Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA-SSSA: Madison, WI, USA, 1982. [Google Scholar]
- NF ISO 10693. Détermination de la Teneuren Carbonate—Méthode Volumétrique; Qualité des Sols AFNOR: Paris, France, 1995; pp. 177–186. [Google Scholar]
- Loeppert, R.H.; Suarez, D.L. Carbonate and gypsum. In Methods of Soil Analysis, Part. 3, Chemical Methods; Bigham, J.M., Bartels, J.M., Eds.; ASA-SSSA: Madison, WI, USA, 1982; pp. 437–474. [Google Scholar]
- Olsen, S.R.; Cole, C.V.; Watanabe, F.S.; Dean, L.A. Estimation of Available Phosphorus in Soils by Extraction with Sodium Bicarbonate; US Department of Agriculture: Washington, DC, USA, 1954; Volume 939, pp. 1–19. [Google Scholar]
- Gasparatos, D.; Haidouti, C.; Tarenidis, D. Characterization of iron oxides in Fe-rich concretions from an imperfectly-drained Greek soil: A study by selective-dissolution techniques and X-ray diffraction. Arch. Agron. Soil Sci. 2004, 50, 485–493. [Google Scholar] [CrossRef]
- Hagarová, I.; Žemberyová, M.; Bajčan, D. Sequential and single step extraction procedures used for fractionation of selenium in soil samples. Chem. Pap. 2005, 59, 93–98. [Google Scholar]
- Bachmann, H.; Bucheli, T.; Dieguez-Alonso, A.; Fabbri, D.; Knicker, H.; Schmidt, H.; Ulbricht, A.; Becker, R.; Buscaroli, A.; Buerge, D.; et al. Toward the Standardization of Biochar Analysis: The COST Action TD1107 Interlaboratory Comparison. J. Agric. Food Chem. 2016, 64, 513–527. [Google Scholar] [CrossRef]
- Bonfil, D.J.; Gitelson, A.A. RapidScan and CropCircle radio-meters: Opportunities and limitation in assessing wheat bio-mass and nitrogen. In Proceedings of the 12th International Conference on Precision Agriculture, Sacramento, CA, USA, 20–23 July 2014. CD-ROM #1369. [Google Scholar]
- Jones, J.B.; Case, V.W. Sampling, handling and analysing plant tissue samples, In Soil Testing and Plant Analysis, 3rd ed.; Westerman, R.L., Ed.; Book Series No. 3; Soil Science Society of America: Madison, WI, USA, 1990; pp. 389–427. [Google Scholar]
- Ros, G.; Rotterdam, A.; Bussink, D.; Bindraban, P. Selenium fertilization strategies for bio-fortification of food: An agro-ecosystem approach. Plant Soil 2016, 404, 99–112. [Google Scholar] [CrossRef]
- Zafeiriou, I.; Gasparatos Ioannou, D.; Massas, I. Selenium uptake by rocket plants (Eruca sativa) grown in a calcareous soil as affected by Se species, Se rate and a seaweed extract—Based biostimulant application. Crop Pasture Sci. 2021; in press. Available online: https://www.publish.csiro.au/CP/justaccepted/CP21529(accessed on 22 December 2021).
- Hayes, K.F.; Roe, L.A.; Brown, G.E.; Hodgson, K.O.; Leckie, J.O.; Parks, G.A. In situ X-ray absorption study of surface complexes: Selenium oxyanions on α-FeOOH. Science 1987, 238, 783–786. [Google Scholar] [CrossRef] [PubMed]
- Duc, M.; Lefevre, G.; Fedoroff, M. Sorption of selenite ions on hematite. J. Colloid Interface Sci. 2006, 298, 556–563. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, S.; Glaubig, R.A. Anion sorption on a calcareous, montmorillonitic soil-selenium. Soil Sci. Soc. Am. J. 1988, 52, 954e958. [Google Scholar] [CrossRef]
- Ramkissoon, C.; Degryse, F.; Young, S.; Bailey, E.; McLaughlin, M. Effect of soil properties on time-dependent fixation (ageing) of selenate. Geoderma 2021, 383, 114741. [Google Scholar] [CrossRef]
- Esringu, A.; Ekinci, M.; Usta, S.; Turan, M.; Dursun, A.; Ercisli, S.; Yildirim, E. Selenium supplementation affects the growth, yield and selenium accumulation in lettuce (Lactuca sativa L.). C. R. Acad. Bulg. Sci. 2015, 68, 801. [Google Scholar]
- Omondi, M.; Xia, X.; Nahayo, A.; Liu, X.; Korai, P.; Pan, G. Quantification of biochar effects on soil hydrological properties using meta-analysis of literature data. Geoderma 2016, 274, 28–34. [Google Scholar] [CrossRef]
- Akhtar, S.; Li, G.; Andersen, M.; Liu, F. Biochar enhances yield and quality of tomato under reduced irrigation. Agric. Water Manag. 2014, 138, 37–44. [Google Scholar] [CrossRef]
- Shaaban, M.; Van Zwieten, L.; Bashir, S.; Younas, A.; Núñez-Delgado, A.; Chhajro, M.; Kubar, K.; Ali, U.; Rana, M.; Mehmood, M.; et al. A concise review of biochar application to agricultural soils to improve soil conditions and fight pollution. J. Environ. Manag. 2018, 228, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Yang, J.; Kronzucker, H.; Shi, W. Selenium Biofortification and Interaction With Other Elements in Plants: A Review. Front. Plant Sci. 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Bannari, A.; Morin, D.; Bonn, F.; Huete, A.R. A review of vegetation indices. Remote Sens. Rev. 1995, 13, 95–120. [Google Scholar] [CrossRef]

| Clay g/kg | Sand g/kg | Silt g/kg | pH 1:1 | E.C. μS/cm | O.C. g/kg | CaCO3 g/kg | Act. CaCO3 g/kg | P Olsen mg/kg | C.E.C. cmolc/kg | Fed g/100 g | Feo g/100 g | Alo g/100 g | Mnd g/100 g | Se Total mg/kg |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 180 | 400 | 420 | 7.4 | 575 | 13 | 184 | 47.5 | 15.4 | 20.4 | 1.16 | 0.14 | 0.04 | 0.05 | 0.175 |
| Selenite | Selenate | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No Biochar | Biochar | No Biochar | Biochar | |||||||||
| Rate (mg/kg) | Rate (mg/kg) | Rate (mg/kg) | Rate (mg/kg) | |||||||||
| 0 | 5 | 10 | 0 | 5 | 10 | 0 | 5 | 10 | 0 | 5 | 10 | |
| Se (mg/kg) | 0.14 a (0.01) | 10.11 b (1.04) | 30.96 d (3.69) | 0.24 a (0.04) | 10.19 b (0.90) | 14.92 c (1.31) | 0.14 a (0.01) | 196.74 c (14.53) | 111.17 b (7.03) | 0.24 a (0.04) | 315.19 d (12.99) | 207.19 c (8.41) |
| S (mg/g) | 1.47 a (0.02) | 1.69 ab (0.13) | 1.53 ab (0.09) | 1.98 b (0.09) | 4.39 c (0.31) | 4.09 c (0.34) | 1.47 a (0.02) | 2.58 b (0.32) | 1.21 a (0.04) | 1.98 ab (0.09) | 8.13 c (0.81) | 2.48 b (0.18) |
| P (mg/g) | 2.42 b (0.15) | 2.33 ab (0.17) | 1.69 a (0.14) | 2.67 bc (0.07) | 3.47 c (0.53) | 3.30 c (0.36) | 2.42 b (0.15) | 1.50 a (0.16) | 1.28 a (0.09) | 2.67 b (0.07) | 3.60 c (0.61) | 1.51 b (0.10) |
| D.W. (g) | 1.38 b (0.08) | 1.59 b (0.23) | 0.90 a (0.09) | 2.01 c (0.10) | 2.37 c (0.23) | 2.25 c (0.11) | 1.38 ab (0.08) | 1.25 ab (0.27) | 1.12 ab (0.12) | 2.01 b (0.10) | 3.01 c (0.79) | 0.95 a (0.12) |
| SPAD | 24.36 b (1.17) | 32.80 c (3.06) | 11.37 a (0.96) | 27.27 bc (3.51) | 23.60 b (1.45) | 31.90 c (1.42) | 24.36 b (1.17) | 20.04 ab (5.23) | 15.27 a (0.65) | 27.27 bc (3.51) | 32.88 c (2.72) | 15.70 a (1.83) |
| NDVI | 0.916 b (0.011) | 0.908 b (0.008) | 0.683 a (0.013) | 0.917 b (0.015) | 0.946 c (0.004) | 0.939 bc (0.028) | 0.916 c (0.011) | 0.820 b (0.018) | 0.791 ab (0.078) | 0.917 c (0.015) | 0.928 c (0.015) | 0.725 a (0.026) |
| NDRE | 0.343 ab (0.091) | 0.368 b (0.048) | 0.230 a (0.027) | 0.333 ab (0.015) | 0.384 b (0.029) | 0.360 ab (0.030) | 0.343 ab (0.091) | 0.253 a (0.044) | 0.330 ab (0.020) | 0.333 ab (0.015) | 0.370 b (0.046) | 0.260 ab (0.029) |
| Selenite (n = 14) | ||||||
| Se (mg/kg) | S (mg/g) | P (mg/g) | D.W. (g) | SPAD | NDVI | |
| Se (mg/kg) | ||||||
| S (mg/g) | ||||||
| P (mg/g) | */− | ***/+ | ||||
| D.W. (g) | ***/− | ***/+ | ***/+ | |||
| SPAD | ***/− | */+ | ***/+ | ***/+ | ||
| NDVI | ***/− | **/+ | ***/+ | ***/+ | ***/+ | |
| Selenate (n = 17) | ||||||
| Se (mg/kg) | S (mg/g) | P (mg/g) | D.W. (g) | SPAD | NDVI | |
| Se (mg/kg) | ||||||
| S (mg/g) | ***/+ | |||||
| P (mg/g) | ***/+ | ***/+ | ||||
| D.W. (g) | ***/+ | ***/+ | ***/+ | |||
| SPAD | ***/+ | ***/+ | ***/+ | ***/+ | ||
| NDVI | **/+ | ***/+ | ***/+ | ***/+ | ***/+ | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zafeiriou, I.; Gasparatos, D.; Ioannou, D.; Kalderis, D.; Massas, I. Selenium Biofortification of Lettuce Plants (Lactuca sativa L.) as Affected by Se Species, Se Rate, and a Biochar Co-Application in a Calcareous Soil. Agronomy 2022, 12, 131. https://doi.org/10.3390/agronomy12010131
Zafeiriou I, Gasparatos D, Ioannou D, Kalderis D, Massas I. Selenium Biofortification of Lettuce Plants (Lactuca sativa L.) as Affected by Se Species, Se Rate, and a Biochar Co-Application in a Calcareous Soil. Agronomy. 2022; 12(1):131. https://doi.org/10.3390/agronomy12010131
Chicago/Turabian StyleZafeiriou, Ioannis, Dionisios Gasparatos, Dafni Ioannou, Dimitrios Kalderis, and Ioannis Massas. 2022. "Selenium Biofortification of Lettuce Plants (Lactuca sativa L.) as Affected by Se Species, Se Rate, and a Biochar Co-Application in a Calcareous Soil" Agronomy 12, no. 1: 131. https://doi.org/10.3390/agronomy12010131
APA StyleZafeiriou, I., Gasparatos, D., Ioannou, D., Kalderis, D., & Massas, I. (2022). Selenium Biofortification of Lettuce Plants (Lactuca sativa L.) as Affected by Se Species, Se Rate, and a Biochar Co-Application in a Calcareous Soil. Agronomy, 12(1), 131. https://doi.org/10.3390/agronomy12010131








