Effect of Thiosemicarbazone Derivatives and Fusarium culmorum (Wm.G. Sm.) Sacc. Infection of Winter Wheat Seedlings on Their Health Status and Soil Biological Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Pant Material
2.2. Fusarium Culmorum Inoculum
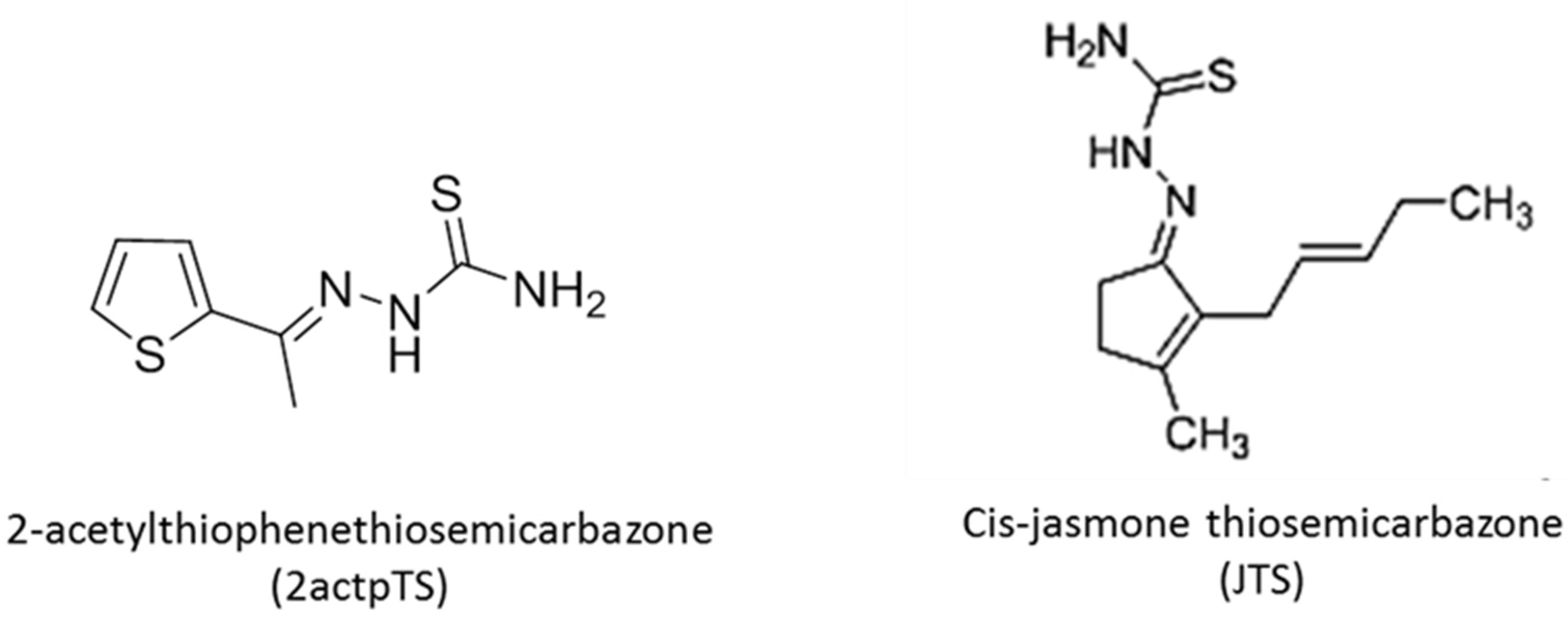
2.3. Chemical Synthesis of Thiosemicarbazone Derivatives
2.4. Thiosemicarbazones Treatments
2.5. Molecule Phytotoxicity (PI) and Plant Disease Index (DI) Determination
2.6. Assessment of Seedling Fresh and Dry Weight
2.7. Determination of Catalase and Dehydrogenase Activity in Soil
2.8. Analysis of Microorganism Abundance in Soil
2.9. Analysis of Soil Functional Biodiversity Using Biolog EcoPlates
2.10. Statistical Analysis
3. Results
3.1. Phytotoxicity of Compounds and Plant Infestation with F. culmorum
3.2. Plant Biomass Production
3.3. Biological Activity of the Soil
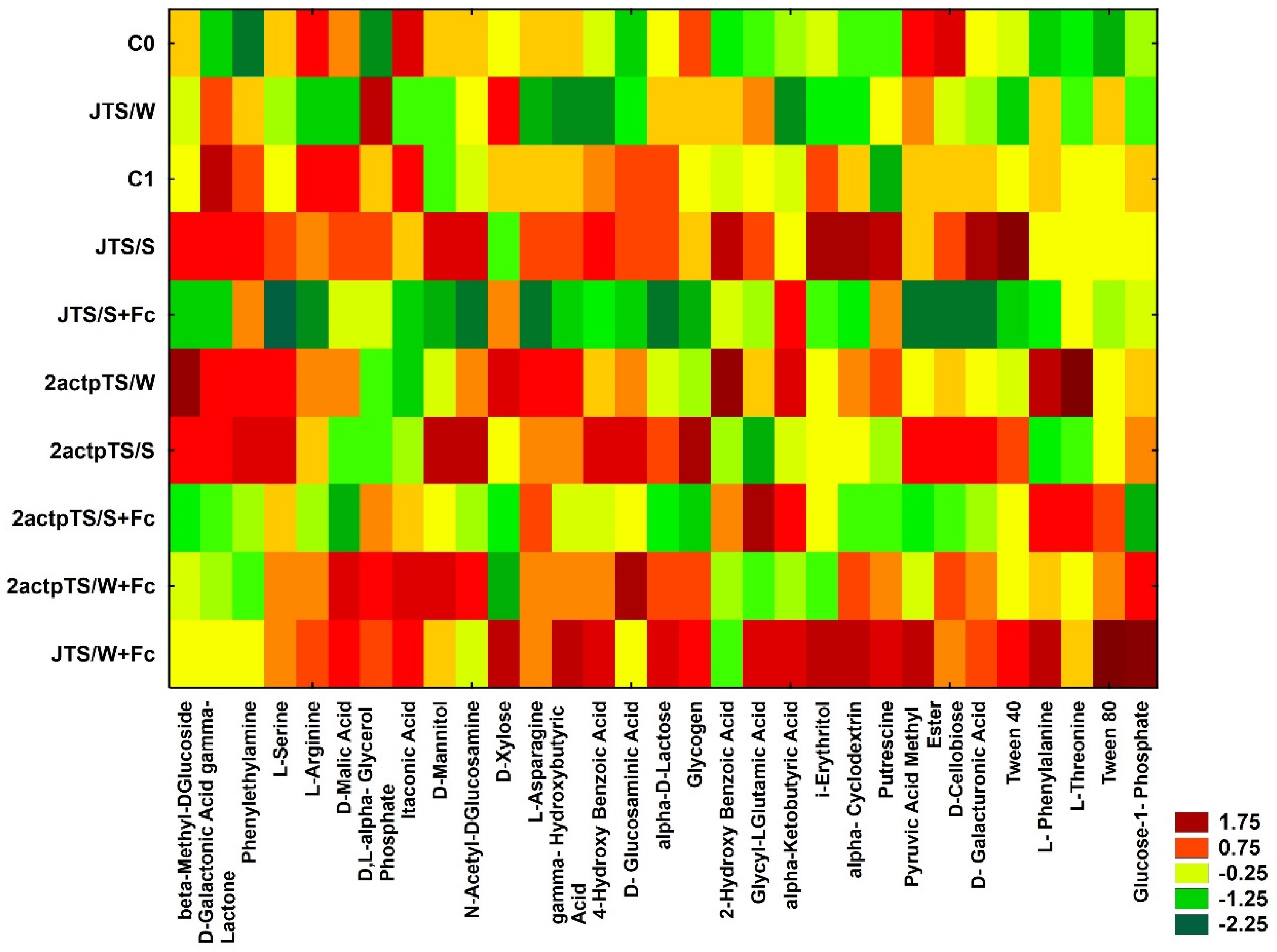
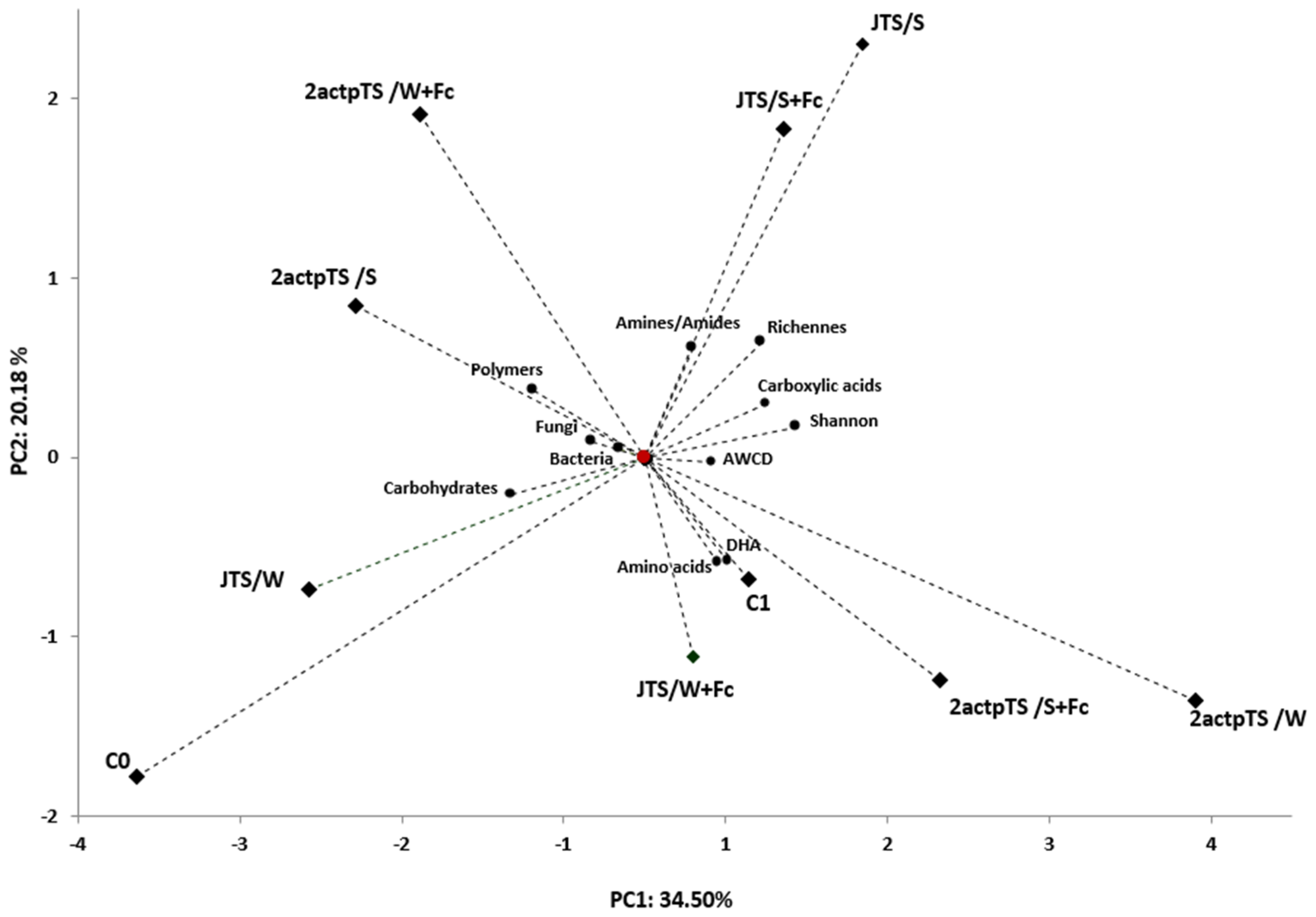
3.4. Biolog EcoPlates Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gruber-Dorninger, C.; Jenkins, T.; Schatzmayr, G. Global Mycotoxin Occurrence in Feed: A Ten-Year Survey. Toxins 2019, 11, 375. [Google Scholar] [CrossRef] [Green Version]
- Mielniczuk, E.; Skwaryło-Bednarz, B. Fusarium Head Blight, Mycotoxins and Strategies for Their Reduction. Agronomy 2020, 10, 509. [Google Scholar] [CrossRef] [Green Version]
- Beccari, G.; Covarelli, L.; Nicholson, P. Infection processes and soft wheat response to root rot and crown rot caused by Fusarium culmorum. Plant Pathol. 2011, 60, 671–684. [Google Scholar] [CrossRef]
- Hofgaard, I.S.; Eseehusen, T.; Aamot, H.U.; Eriley, H.; Erazzaghian, J.; Le, V.H.; Hjelkrem, A.-G.R.; Edill-Macky, R.; Ebrodal, G. Inoculum Potential of Fusarium spp. Relates to Tillage and Straw Management in Norwegian Fields of Spring Oats. Front. Microbiol. 2016, 7, 556. [Google Scholar] [CrossRef] [Green Version]
- Hofgaard, I.S.; Aamot, H.U.; Torp, T.; Jestoi, M.; Lattanzio, V.M.T.; Klemsdal, S.S.; Waalwijk, C.; van der Lee, T.; Brodal, G. Associations between Fusarium species and mycotoxins in oats and spring wheat from farmers’ fields in Norway over a six-year period. World Mycotoxin J. 2016, 9, 365–378. [Google Scholar] [CrossRef]
- Kraska, P.; Mielniczuk, E. The occurrence of fungi on the stem base and rootsof spring wheat (Triticum aestivum L.) grown in monoculture depending on tillage systems and catch crops. Acta Agrobot. 2012, 65, 79–90. [Google Scholar] [CrossRef] [Green Version]
- Popovski, S.; Celar, F.A. The impact of environmental factors on the infection of cereals with Fusarium species and mycotoxin production—A review. Acta Agric. Slov. 2013, 101, 105–116. [Google Scholar] [CrossRef]
- Bankina, B.; Bimšteine, G.; Ruza, A.; Priekule, I.; Paura, L.; Vaivade, I.; Fridmanis, D. Winter wheat crown and root rot are affected by soil tillage and crop rotation in Latvia. Acta Agric. Scand. Sect. B—Plant Soil Sci. 2013, 63, 723–730. [Google Scholar] [CrossRef]
- Parry, D.W.; Jenkinson, P.; McLeod, L. Fusarium ear blight (scab) in small grain cereals—A review. Plant Pathol. 1995, 44, 207–238. [Google Scholar] [CrossRef]
- Medina, A.; Akbar, A.; Baazeem, A.; Rodriguez, A.; Magan, N. Climate change, food security and mycotoxins: Do we know enough? Fungal Biol. Rev. 2017, 31, 143–154. [Google Scholar] [CrossRef] [Green Version]
- European Parliament. Regulation (EC) No 1107/2009 of the European Parliament and of the Council of 21 October 2009 Concerning the Placing of Plant Protection Products on the Market and Repealing Council Directives 79/117/EEC and 91/414/EEC; European Parliament: Brussels, Belgium, 2009.
- Benns, B.G.; Gingras, B.A.; Bayley, C.H. Antifungal activity of some thiosemicarbazones and their copper complexes. Appl. Microbiol. 1960, 8, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Paiva, R.O.; Kneipp, L.F.; Goular, C.M.; Albuquerque, M.A.; Echevarria, A. Antifungal activities of thiosemicarbazones and semicarbazones against mycotoxigenic fungi. Ciência Agrotecnologia 2014, 38, 531–537. [Google Scholar] [CrossRef] [Green Version]
- Degola, F.; Morcia, C.; Bisceglie, F.; Mussi, F.; Tumino, G.; Ghizzoni, R.; Pelosi, G.; Terzi, V.; Buschini, A.; Restivo, F.M.; et al. In vitro evaluation of the activity of thiosemicarbazone derivatives against mycotoxigenic fungi affecting cereals. Int. J. Food Microbiol. 2015, 200, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Neto, L.N.D.A.; de Lima, M.D.C.A.; de Oliveira, J.F.; de Souza, E.R.; Buonafina, M.D.S.; Anjos, M.N.V.; Brayner, F.A.; Alves, L.C.; Neves, R.P.; Mendonça-Junior, F.J.B. Synthesis, cytotoxicity and antifungal activity of 5-nitro-thiophene-thiosemicarbazones derivatives. Chem. Interact. 2017, 272, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Rogolino, D.; Gatti, A.; Carcelli, M.; Pelosi, G.; Bisceglie, F.; Restivo, F.M.; Degola, F.; Buschini, A.; Montalbano, S.; Feretti, D.; et al. Thiosemicarbazone scaffold for the design of antifungal and antiaflatoxigenic agents: Evaluation of ligands and related copper complexes. Sci. Rep. 2017, 7, 111214. [Google Scholar] [CrossRef]
- Bartoli, J.; Montalbano, S.; Spadola, G.; Rogolino, D.; Pelosi, G.; Bisceglie, F.; Restivo, F.M.; Degola, F.; Serra, O.; Buschini, A.; et al. Antiaflatoxigenic Thiosemicarbazones as Crop-Protective Agents: A Cytotoxic and Genotoxic Study. J. Agric. Food Chem. 2019, 67, 10947–10953. [Google Scholar] [CrossRef]
- Song, S.; You, A.; Chen, Z.; Zhu, G.; Wen, H.; Song, H.; Yi, W. Study on the design, synthesis and structure-activity relationships of new thiosemicarbazone compounds as tyrosinase inhibitors. Eur. J. Med. Chem. 2017, 139, 815–825. [Google Scholar] [CrossRef]
- Zhai, X.; Bao, G.; Wang, L.; Cheng, M.; Zhao, M.; Zhao, S.; Zhou, H.; Gong, P. Design, synthesis and biological evaluation of novel 4-phenoxy-6,7-disubstuìitted quinoline possessing (thio)semicarbazone as c-Met kinase inhibitors. Bioorg. Med. Chem. 2016, 14, 1331–1345. [Google Scholar] [CrossRef]
- Yu, Y.; Kalinowski, D.S.; Kovacevic, Z.; Siafakas, A.R.; Jansson, P.J.; Stefani, C.; Lovejoy, D.B.; Sharpe, P.; Bernhardt, P.; Richardson, D.R. Thiosemicarbazones from the Old to New: Iron Chelators That Are More than Just Ribonucleotide Reductase Inhibitors. J. Med. Chem. 2009, 52, 5271–5294. [Google Scholar] [CrossRef]
- Marquezin, C.; de Oliveira, C.; Vandresen, F.; Duarte, E.; Lamy, M.; Vequi-Suplicy, C. The interaction of a thiosemicarbazone derived from R-(+)-limonene with lipid membranes. Chem. Phys. Lipids 2021, 234, 105018. [Google Scholar] [CrossRef]
- Orsoni, N.; Degola, F.; Nerva, L.; Bisceglie, F.; Spadola, G.; Chitarra, W.; Terzi, V.; Delbono, S.; Ghizzoni, R.; Morcia, C.; et al. Double Gamers—Can Modified Natural Regulators of Higher Plants Act as Antagonists against Phytopathogens? The Case of Jasmonic Acid Derivatives. Int. J. Mol. Sci. 2020, 21, 8681. [Google Scholar] [CrossRef] [PubMed]
- Montalbano, S.; Degola, F.; Bartoli, J.; Bisceglie, F.; Buschini, A.; Carcelli, M.; Feretti, D.; Galati, S.; Marchi, L.; Orsoni, N.; et al. The AFLATOX® Project: Approaching the Development of New Generation, Natural-Based Compounds for the Containment of the Mycotoxigenic Phytopathogen Aspergillus flavus and Aflatoxin Contamination. Int. J. Mol. Sci. 2021, 22, 4520. [Google Scholar] [CrossRef]
- Ossowicki, A.; Tracanna, V.; Petrus, M.L.C.; van Wezel, G.; Raaijmakers, J.; Medema, M.H.; Garbeva, P. Microbial and volatile profiling of soils suppressive to Fusarium culmorum of wheat. Proc. R. Soc. B Boil. Sci. 2020, 287, 20192527. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jamiołkowska, A. Effect of Field Pea (Pisum arvense L.) as a Cover Plant on Health of Under-Ground Part of Field Tomato. Veg. Crop. Res. Bull. 2007, 67, 71–79. [Google Scholar] [CrossRef]
- Mielniczuk, E. Występowanie Grzybów Rodzaju Fusarium NA Owsie (Avena sativa L.) Ze Szczególnym Uwzględnieniem Gatunku Fusarium Avenaceum (FR.) Sacc., Jego Biologii Oraz Szkodliwości Dla Wybranych Odmian, 1st ed.; Wyd. Uniwersytetu Przyrodniczego w Lublinie: Lublin, Poland, 2018; pp. 1–156. [Google Scholar]
- Wenzel, H. Zur Erfassung des Schadenausmasses in Pflanzenschutzversuchen. Pflanzenschutz-Berichte 1948, 15, 81–84. [Google Scholar]
- Johnson, J.L.; Temple, K.L. Some Variables Affecting the Measurement of “Catalase Activity” in Soil. Soil Sci. Soc. Am. J. 1964, 28, 207–209. [Google Scholar] [CrossRef]
- Casida, L.E.; Klein, D.A.; Santoro, T. Soil Dehydrogenase Activity. Soil Sci. 1964, 98, 371–376. [Google Scholar] [CrossRef]
- Patkowska, E.; Błażewicz-Woźniak, M. The microorganisms communities in the soil under the cultivation of carrot (Daucus carota L.). Acta Sci. Pol. Hortorum Cultus 2014, 13, 103–115. [Google Scholar]
- Jamiołkowska, A.; Skwaryło-Bednarz, B.; Patkowska, E.; Buczkowska, H.; Gałązka, A.; Grządziel, J.; Kopacki, M. Effect of Mycorrhizal Inoculation and Irrigation on Biological Properties of Sweet Pepper Rhizosphere in Organic Field Cultivation. Agronomy 2020, 10, 1693. [Google Scholar] [CrossRef]
- Weber, K.P.; Legge, R.L. One-dimensional metric for tracking bacterial community divergence using sole carbon source utilization patterns. J. Microbiol. Methods 2009, 79, 55–61. [Google Scholar] [CrossRef]
- Hammer, O.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]
- Degola, F.; Bisceglie, F.; Pioli, M.; Palmano, S.; Elviri, L.; Pelosi, G.; Lodi, T.; Restivo, F.M. Structural modification of cuminaldehyde thiosemicarbazone increases inhibition specificity toward aflatoxin biosynthesis and sclerotia development in Aspergillus flavus. Appl. Microbiol. Biotechnol. 2017, 101, 6683–6696. [Google Scholar] [CrossRef] [PubMed]
- Bisceglie, F.; Degola, F.; Rogolino, D.; Giannelli, G.; Orsoni, N.; Spadola, G.; Pioli, M.; Restivo, F.M.; Carcelli, M.; Pelosi, G. Sisters in structure but different in character, some benzaldehyde and cinnamaldehyde derivatives differentially tune Aspergillus flavus secondary metabolism. Sci. Rep. 2020, 10, 17686. [Google Scholar] [CrossRef]
- Huang, Y.; Wang, S.; Wang, C.; Ding, G.; Cai, H.; Shi, L.; Xu, F. Induction of jasmonic acid biosynthetic genes inhibits Arabidopsis growth in response to low boron. J. Integr. Plant Biol. 2021, 63, 937–948. [Google Scholar] [CrossRef]
- Ali, S.; Baek, K.-H. Jasmonic Acid Signaling Pathway in Response to Abiotic Stresses in Plants. Int. J. Mol. Sci. 2020, 21, 621. [Google Scholar] [CrossRef] [Green Version]
- Chanclud, E.; Morel, J.-B. Plant hormones: A fungal point of view. Mol. Plant Pathol. 2016, 17, 1289–1297. [Google Scholar] [CrossRef]
- Baćmaga, M.; Kucharski, J.; Wyszkowska, J. Wpływ środków ochrony roślin na aktywność mikrobiologiczną gleby. J. Elementol. 2007, 12, 225–239. [Google Scholar]
- Anna, G.; Karolina, G.; Jarosław, G.; Magdalena, F.; Jerzy, K. Microbial community diversity and the interaction of soil under maize growth in different cultivation techniques. Plant Soil Environ. 2017, 63, 264–270. [Google Scholar] [CrossRef] [Green Version]
- Gałązka, A.; Gawyjołek, K.; Perzyński, A.; Gałązka, R.; Jerzy, K. Changes in Enzymatic Activities and Microbial Communities in Soil under Long-Term Maize Monoculture and Crop Rotation. Pol. J. Environ. Stud. 2017, 26, 39–46. [Google Scholar] [CrossRef]
- Gałązka, A.; Grzęda, E.; Jończyk, K. Changes of Microbial Diversity in Rhizosphere Soils of New Quality Varieties of Winter Wheat Cultivation in Organic Farming. Sustainability 2019, 11, 4057. [Google Scholar] [CrossRef] [Green Version]
- Furtak, K.; Gajda, A.M. Activity of Dehydrogenases as an Indicator of Soil Environment Quality. Pol. J. Soil Sci. 2017, 50, 33. [Google Scholar] [CrossRef] [Green Version]
- Furtak, K.; Gajda, A.M. Biochemical Methods for the Evaluation of the Functional and Structural Diversity of Micro-Organisms in the Soil Environment. Adv. Microbiol. 2019, 57, 194–202. [Google Scholar] [CrossRef]
- Wolińska, A.; Stępniewska, Z. Dehydrogenase activity in the soil environment. In Dehydrogenases, 1st ed.; Canuto, R.A., Ed.; InTech: Rijeka, Croatia, 2012; Available online: http://www.ebook3000.com/ (accessed on 18 November 2021).
- Calamai, L.; Laze, I.; Stotzky, G.; Fusi, P.; Ristori, G.G. Interaction of catalase with montmorillonite homoionic to cations with different hydrophobicity: Effect on enzyme activity and microbial utilization. Soil Biol. Biochem. 2000, 32, 815–823. [Google Scholar] [CrossRef]
- Masciandaro, G.; Ceccanti, B.; Ronchi, V.; Bauer, C. Kinetic parameters of dehydrogenase in the assessment of the response of soil to vermicom-post and inorganic fertilizers. Biol. Fertil. Soils 2000, 32, 479–483. [Google Scholar] [CrossRef]
- Siwik-Ziomek, A.; Szczepanek, M. Soil Extracellular Enzyme Activities and Uptake of N by Oilseed Rape Depending on Fertilization and Seaweed Biostimulant Application. Agronomy 2019, 9, 480. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.L.; Wu, Z.J.; Chen, L.J.; Jiang, Y.; Li, D.P. Kinetics of catalase and dehydrogenase in main soils of Northeast China under different soil moisture conditions. Agric. J. 2009, 4, 113–120. Available online: https://medwelljournals.com/abstract/?doi=aj.2009.113.120 (accessed on 7 November 2021).
- Dinesh, R.; Chaudhuri, S.G.; Sheeja, T.E. Soil biochemical and microbial indices in wet tropical forests: Effects of deforestation and cultivation. J. Plant Nutr. Soil Sci. 2004, 167, 24–32. [Google Scholar] [CrossRef]
- Brzezińska, M. Aktywność biologiczna i procesy jej towarzyszące w glebach organicznych nawadnianych oczyszczonymi ściekami miejskimi. Acta Agrophys. 2006, 131, 1–164. [Google Scholar]
- Kaczyńska, G.; Borowik, A.; Wyszkowska, J. Soil Dehydrogenases as an Indicator of Contamination of the Environment with Petroleum Products. Water Air Soil Pollut. 2015, 226, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tiwari, R.K.; Singh, S.; Pandey, R.S.; Sharma, B. Enzymes of Earthworm as Indicators of Pesticide Pollution in Soil. Adv. Enzym. Res. 2016, 4, 113–124. [Google Scholar] [CrossRef] [Green Version]
- Karaca, A.; Cetin, S.C.; Turgay, O.C.; Kizilkaya, R. Soil Enzymes as Indication of Soil Quality. In Soil Biology; Springer Science and Business Media LLC: Cham, Switzerland, 2010; pp. 119–148. [Google Scholar]
- Medo, J.; Maková, J.; Medová, J.; Lipková, N.; Cinkocki, R.; Omelka, R.; Javoreková, S. Changes in soil microbial community and activity caused by application of dimethachlor and linuron. Sci. Rep. 2021, 11, 12786. [Google Scholar] [CrossRef] [PubMed]
- Brodowska, M.S.; Onuch, J.; Krzepiłko, A.; Skwaryło-Bednarz, B.; Brodowski, R.; Ziemińska-Smyk, M.; Gradziuk, B. The impact of copper on catalase activity and antioxidant properties of soil under amaranth cultivation. J. Elem. 2018, 23, 825–836. [Google Scholar] [CrossRef]
- Kalam, A.; Tah, J.; Mukherjee, A.K. Pesticide effects on microbial population and soil enzyme activities during vermicom-posting of agricultural waste. J. Environ. Biol. 2004, 25, 201–208. [Google Scholar]
- Riah, W.; Laval, K.; Laroche-Ajzenberd, E.; Mougin, C.; Latour, X.; Trinsoutrot-Gattin, I. Effects of Pesticides on Soil Enzymatic Activities: General Trends. Environ. Chem. Lett. 2014, 12, 257–273. [Google Scholar] [CrossRef]
- Stępniewska, Z.; Wolińska, A.; Lipińska, R. Effect of fonofos on soil dehydrogenase activity. Int. Agrophysics 2006, 21, 101–105. [Google Scholar]
- Jastrzębska, E. The effect of chlorpyrifos and teflubenzuron on the enzymatic activity of soil. Pol. J. Environ. Stud. 2011, 20, 903–910. [Google Scholar]
- Caceres, T.; He, W.; Megharaj, M.; Naidu, R. Effect of insecticide fenamiphos on soil microbial activities in Australian and Ecuadorean soils. J. Environ. Sci. Health Part B 2008, 44, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Kalyani, S.S.; Sharma, J.; Dureja, P.; Singh, S.; Lata, N. Influence ofendosulfan on microbial biomass and soil enzymatic ac-tivities of a tropical. Alfisol. Bull. Environ. Contam. Toxicol. 2010, 84, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Jastrzebska, E.; Kucharski, J. Dehydrogenases, urease and phosphatases activities of soil contaminated with fungicides. Plant Soil Environ. 2008, 53, 51–57. [Google Scholar] [CrossRef] [Green Version]
- Małachowska-Jutsz, A.; Mrozowska, J.; Kozielska, M.; Miksch, K. Aktywność enzymatyczna w glebie skażonej związkami ropopochodnymi w procesie jej detoksykacji. Biotechnologia 1997, 1, 79–91. [Google Scholar]
- Skwaryło-Bednarz, B. Influence of contamination of soil with copper on the activity of dehydrogenases in areas where amaranthus is cultivated. Ecol. Chem. Eng. A 2012, 19, 155–160. [Google Scholar]
- Verma, J.P.; Jaiswal, D.K.; Sagar, R. Pesticide relevance and their microbial degradation: A-state-of-art. Rev. Environ. Sci. Bio/Technol. 2014, 13, 429–466. [Google Scholar] [CrossRef]
- Sud, D.; Kumar, J.; Kaur, P.; Bansal, P. Toxicity, Natural and Induced Degradation of Chlorpyrifos. J. Chil. Chem. Soc. 2020, 65, 4807–4816. [Google Scholar] [CrossRef]
- Kızılkaya, R.; Akça, İ.; Aşkın, T.; Yılmaz, R.; Olekhov, V.; Samofalova, I.; Mudrykh, N. Effect of soil contamination with azadirachtin on dehydrogenase and catalase activity of soil. Eurasian J. Soil Sci. 2012, 2, 98–103. [Google Scholar]
- Frąc, M.; Hannula, S.E.; Bełka, M.; Jędryczka, M. Fungal Biodiversity and Their Role in Soil Health. Front. Microbiol. 2018, 9, 707. [Google Scholar] [CrossRef] [Green Version]
- Verma, A. Bioremediation Techniques for Soil Pollution: An Introduction. In Biodegradation; IntechOpen Limited: London, UK, 2021. [Google Scholar] [CrossRef]
- Waraczewska, Z.; Niewiadomska, A.; Grzyb, A. Wybrane metody bioremediacji in situ z wykorzystaniem mikroorganizmów. Woda-Środowisko-Obszary Wiej. 2018, 18, 65–78. [Google Scholar]


| Experimental Combination | Mean Phytotoxicity Index (PI) | Fresh Weight (g fw−1) | Dry Weight (g dw−1) | Mean Disease Index (DI) |
|---|---|---|---|---|
| 2actpTS/W | 46.00 a | 3.192 abcd | 0.471 b | 11.00 a |
| 2actpTS/S | 5.00 c | 4.498 d | 0.638 ab | 13.30 ab |
| 2actpTS/W+Fc | 44.00 a | 4.147 bcd | 0.635 ab | 15.70 bc |
| 2actpTS/S+Fc | 12.00 bc | 5.903 a | 0.705 a | 16.70 c |
| JTS/W | 53.00 a | 4.046 cd | 0.746 a | 15.00 bc |
| JTS/S | 13.00 bc | 4.407 abcd | 0.592 ab | 12.00 ab |
| JTS/W+Fc | 5.00 c | 3.732 cd | 0.572 ab | 14.30 ab |
| JTS/S+Fc | 19.00 b | 5.02 abc | 0.667 a | 23.70 d |
| C1 | 0.00 c | 5.644 ab | 0.654 ab | 17.70 c |
| C0 | 0.00 c | 4.934 abc | 0.614 ab | 14.30 ab |
| Experimental Combination | Total Number of Bacteria (107 CFU g−1 dw Soil) | Total Number of Fungi (104 CFU G−1 Dw Soil) | Catalase Activity (mg H2O2 g−1 dw min−1) | DEHYDROGENASE Activity (µg TPF g−1 dw d−1) |
|---|---|---|---|---|
| 2actpTS/W | 114.868 b | 53.749 b | 0.081 b | 54.55 b |
| 2actpTS/S | 64.542 c | 64.542 b | 0.050 e | 46.34 c |
| 2actpTS/W+Fc | 151.254 b | 76.449 a | 0.088 a | 39.58 d |
| 2actpTS/S+Fc | 81.917 c | 41.830 b | 0.053 de | 50.28 bc |
| JTS/W | 289.116 a | 42.517 b | 0.065 c | 44.21 cd |
| JTS/S | 233.706 a | 49.348 b | 0.040 f | 42.26 d |
| JTS/W+Fc | 45.947 c | 55.483 b | 0.078 b | 54.37 b |
| JTS/S+Fc | 75.615 c | 49.217 b | 0.048 e | 47.93 bc |
| C1 | 285.277 a | 79.948 a | 0.092 a | 61.67 a |
| C0 | 141.304 b | 65.620 b | 0.058 d | 47.17 c |
| Experimental Combination | Amines and Amides | Amino Acids | Carbohydrates | Carboxylic Acids | Polymers |
|---|---|---|---|---|---|
| 2actpTS/W | 6.888 b | 19.752 a | 29.556 a | 29.909 b | 13.894 a |
| 2actpTS/S | 6.824 b | 18.112 b | 29.228 a | 31.171 a | 14.665 a |
| 2actpTS/W+Fc | 6.150 c | 19.848 a | 28.621 b | 30.543 b | 14.838 a |
| 2actpTS/S+Fc | 6.528 c | 20.443 a | 29.372 a | 29.082 b | 14.576 a |
| JTS/W | 7.461 a | 19.232 b | 27.344 b | 31.570 a | 14.392 a |
| JTS/S | 7.238 a | 18.781 b | 29.635 a | 29.874 b | 14.472 a |
| JTS/W+Fc | 7.025 b | 19.287 b | 28.481 b | 30.650 b | 14.557 a |
| JTS/S+Fc | 8.101 a | 19.556 a | 29.292 a | 29.132 b | 13.920 a |
| C1 | 6.444 c | 19.294 b | 30.226 a | 29.840 b | 14.196 a |
| C0 | 6.043 c | 19.093 b | 28.485 b | 31.858 a | 14.521 a |
| Experimental Combination | H’ | R | E | AWCD590 |
|---|---|---|---|---|
| 2actpTS/W | 3.405 a | 30.333 a | 0.992 a | 2.101 a |
| 2actpTS/S | 3.393 a | 29.667 a | 0.994 a | 2.016 a |
| 2actpTS/W+Fc | 3.395 a | 30.333 a | 0.992 a | 2.096 a |
| 2actpTS/S+Fc | 3.395 a | 30.000 a | 0.995 a | 1.896 b |
| JTS/W | 3.390 a | 29.333 a | 0.994 a | 1.918 a |
| JTS/S | 3.412 a | 31.000 a | 0.993 a | 2.201 a |
| JTS/W+Fc | 3.396 a | 30.000 a | 0.995 a | 2.122 a |
| JTS/S+Fc | 3.399 a | 30.333 a | 0.990 a | 1.795 b |
| C1 | 3.398 a | 30.000 a | 0.996 a | 2.010 a |
| C0 | 3.362 a | 28.667 b | 0.998 a | 1.916 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jamiołkowska, A.; Skwaryło-Bednarz, B.; Mielniczuk, E.; Bisceglie, F.; Pelosi, G.; Degola, F.; Gałązka, A.; Grzęda, E. Effect of Thiosemicarbazone Derivatives and Fusarium culmorum (Wm.G. Sm.) Sacc. Infection of Winter Wheat Seedlings on Their Health Status and Soil Biological Activity. Agronomy 2022, 12, 116. https://doi.org/10.3390/agronomy12010116
Jamiołkowska A, Skwaryło-Bednarz B, Mielniczuk E, Bisceglie F, Pelosi G, Degola F, Gałązka A, Grzęda E. Effect of Thiosemicarbazone Derivatives and Fusarium culmorum (Wm.G. Sm.) Sacc. Infection of Winter Wheat Seedlings on Their Health Status and Soil Biological Activity. Agronomy. 2022; 12(1):116. https://doi.org/10.3390/agronomy12010116
Chicago/Turabian StyleJamiołkowska, Agnieszka, Barbara Skwaryło-Bednarz, Elżbieta Mielniczuk, Franco Bisceglie, Giorgio Pelosi, Francesca Degola, Anna Gałązka, and Emilia Grzęda. 2022. "Effect of Thiosemicarbazone Derivatives and Fusarium culmorum (Wm.G. Sm.) Sacc. Infection of Winter Wheat Seedlings on Their Health Status and Soil Biological Activity" Agronomy 12, no. 1: 116. https://doi.org/10.3390/agronomy12010116
APA StyleJamiołkowska, A., Skwaryło-Bednarz, B., Mielniczuk, E., Bisceglie, F., Pelosi, G., Degola, F., Gałązka, A., & Grzęda, E. (2022). Effect of Thiosemicarbazone Derivatives and Fusarium culmorum (Wm.G. Sm.) Sacc. Infection of Winter Wheat Seedlings on Their Health Status and Soil Biological Activity. Agronomy, 12(1), 116. https://doi.org/10.3390/agronomy12010116