Methane Mitigation Potential of Foliage of Fodder Trees Mixed at Two Levels with a Tropical Grass
Abstract
:1. Introduction
2. Materials and Methods
2.1. Description of the Study Area
2.2. Sampling
2.3. Treatments
2.4. Chemical Analysis
2.5. In Vitro Gas Production Technique
2.6. Methane Quantification
2.7. Statistical Analysis
3. Results
3.1. Chemical Composition, In Vitro Digestibility, and Phytochemical Screening
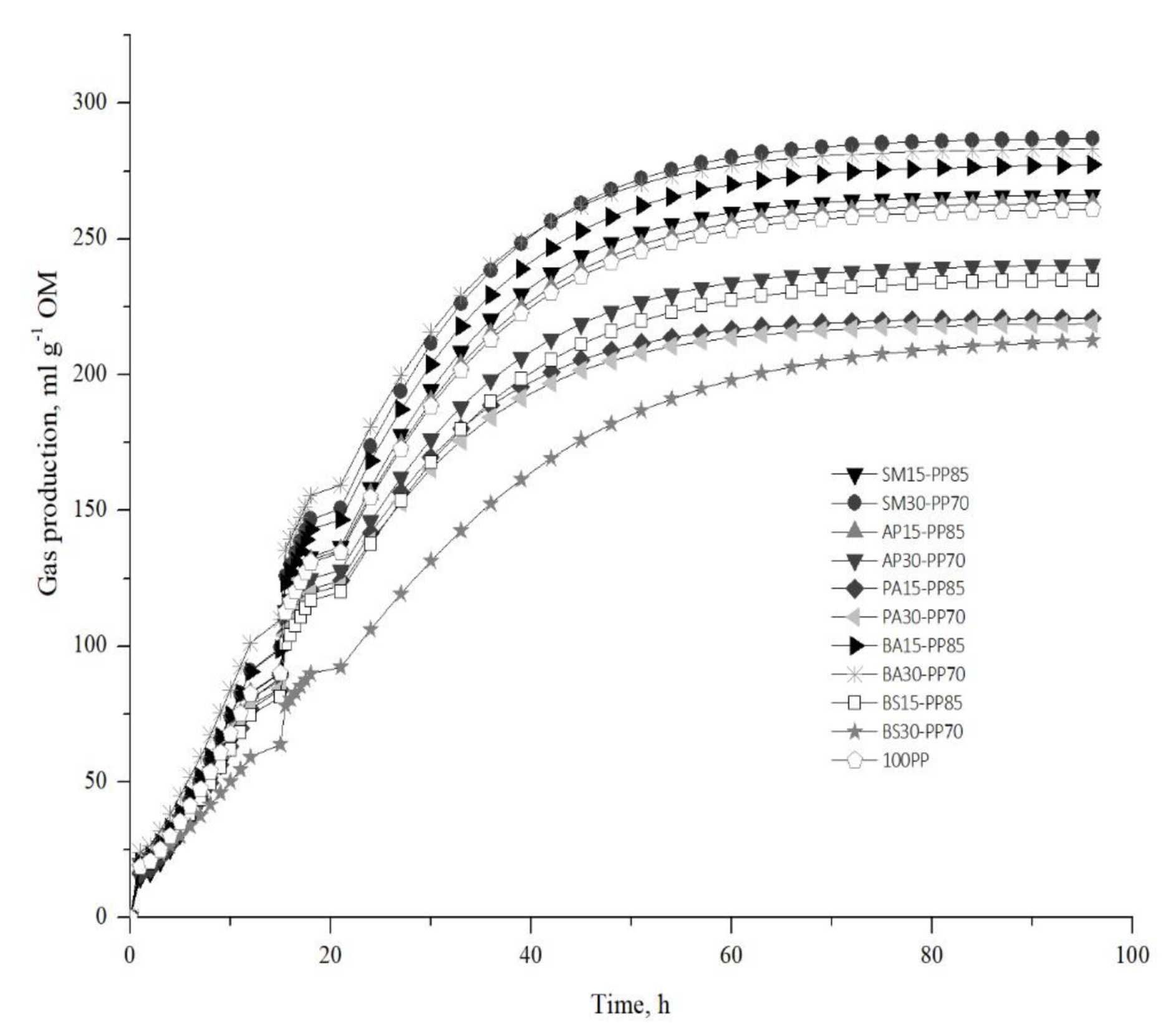
3.2. In Vitro Gas Production Parameters
3.3. Dry Matter Degradability, pH, Short Chain Fatty Acids and CH4 Production
4. Discussion
4.1. Chemical Composition and In Vitro Digestibility
4.2. Dry Matter Degradability, In Vitro Gas Production and pH
4.3. Methane Production
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Beauchemin, K.; Ungerfeld, E.; Eckard, R.; Wang, M. Fifty years of research on rumen methanogenesis: Lessons learned and future challenges for mitigation. Animal 2020, 14 (Suppl. S1), s2–s16. [Google Scholar] [CrossRef] [Green Version]
- Glatzle, A. Questioning key conclusions of FAO publications ‘Livestock’s Long Shadow’ (2006) appearing again in ‘Tackling Climate Change Through Livestock’. Pastoralism 2013, 4, 1. [Google Scholar] [CrossRef] [Green Version]
- Haque, N. Dietary manipulation: A sustainable way to mitigate methane emissions from ruminants. Anim. Feed Sci. Technol. 2018, 60, 15. [Google Scholar] [CrossRef] [Green Version]
- Kurihara, M.; Magner, T.; Hunter, R.; McCrabb, G. Methane production and energy partition of cattle in the tropics. Br. J. Nutr. 1999, 81, 227–234. [Google Scholar] [CrossRef]
- Hyland, J.J.; Styles, D.; Jones, D.J.; Williams, A.P. Improving livestock production efficiencies presents a major opportunity to reduce sectoral greenhouse gas emissions. Agric. Syst. 2016, 147, 123–131. [Google Scholar] [CrossRef] [Green Version]
- Cammack, K.M.; Austin, K.J.; Lamberson, W.R.; Conant, G.C.; Cunningham, H.C. Ruminnat nutrition symposium: Tiny but mighty: The role of the rumen microbes in livestock production. Anim. Sci. J. 2018, 96, 752–770. [Google Scholar] [CrossRef]
- Henderson, G.; Cox, F.; Ganesh, S.; Jonker, A.; Young, W.; Janssen, P.H. Rumen microbial community composition varies with diet and host, but a core microbiome is found across a wide geographical range. Sci. Rep. 2015, 5, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Lan, W.; Yang, C. Ruminal methane production: Associated microorganisms and the potential of applying hydrogen-utilizing bacteria for mitigation. Sci. Total Environ. 2019, 654, 1270–1283. [Google Scholar] [CrossRef] [PubMed]
- Huws, S.A.; Creevey, C.J.; Oyama, L.B.; Mizrahi, I.; Denman, S.E.; Popova, M.; Mayorga, O.L.; Morrisson, S.; Elliott, C.; Morgavi, D.P.; et al. Addressing global ruminant agricultural challenges through understanding the rumen microbiome: Past, present, and future. Front. Microb. 2018, 9, 2161. [Google Scholar] [CrossRef] [PubMed]
- Belanche, A.; De La Fuente, G.; Newbold, C.J. Study of methanogen communities associated with different rumen protozoal populations. FEMS Microbiol. Ecol. 2014, 90, 663–677. [Google Scholar] [CrossRef] [Green Version]
- Tapio, I.; Snelling, T.J.; Strozzi, F.; Wallace, R.J. The ruminal microbiome associated with methane emissions from ruminant livestock. J. Anim. Sci. Biotechnol. 2017, 8, 7. [Google Scholar] [CrossRef]
- Berghuis, B.A.; Yu, F.B.; Schulz, F.; Blainey, P.C.; Woyke, T.; Quake, S.R. Hydrogenotrophic methanogenesis in archaeal phylum Verstraetearchaeota reveals the shared ancestry of all methanogens. Proc. Natl. Acad. Sci. USA 2019, 116, 5037–5044. [Google Scholar] [CrossRef] [Green Version]
- Arndt, C.; Hristov, A.N.; Price, W.J.; McClelland, S.C.; Pelaez, A.M.; Cueva, S.F.; Martin, C.; Schwarm, A.; Veneman, J.B.; ZhongTang, Y.; et al. Strategies to mitigate enteric methane emissions by ruminants-a way to approach the 2.0 °C target. Preprint 2021, in press. [Google Scholar]
- Arango, J.; Ruden, A.; Martinez-Baron, D.; Loboguerrero, A.M.; Berndt, A.; Chacón, M.; Torres, C.F.; Oyhantcabal, W.; Gomez, C.A.; Ricci, P.; et al. Ambition Meets Reality: Achieving GHG Emission Reduction Targets in the Livestock Sector of Latin America. Front. Sustain. Food Syst. 2020, 4, 65. [Google Scholar] [CrossRef]
- Ku-Vera, J.C.; Jiménez-Ocampo, R.; Valencia-Salazar, S.; Flores-Santiago, E.J.; Montoya-Flores, M.D.; Molina-Botero, I.C.; Arango, J.; Gomez-Bravo, C.A.; Aguilar-Perez, C.F.; Solorio-Sanchez, F.J. Role of Secondary Plant Metabolites on Enteric Methane Mitigation in Ruminants. Front. Vet. Sci. 2020, 7, 584. [Google Scholar] [CrossRef]
- Patra, A.K.; Park, T.; Kim, M.; Yu, Z. Rumen methanogens and mitigation of methane emissions by anti-methanogenic compounds and substances. J. Anim. Sci. Biotechnol. 2017, 8, 13. [Google Scholar] [CrossRef] [Green Version]
- Rao, I.; Peters, M.; Castro, A.; Schultze-Kraft, R.; White, D.; Fisher, M.; Jenet, A.; Schmidt, A.; Vivas, N.; Rudel, T. LivestockPlus–The sustainable intensification of forage-based agricultural systems to improve livelihoods and ecosystem services in the tropics. Trop. Grassl. 2015, 3, 59. [Google Scholar] [CrossRef]
- Valencia-Salazar, S.; Piñeiro, A.T.; Molina, I.C.; Lazos, F.J.; Uuh, J.J.; Segura, M.; Ramírez, L.; Solorio, F.J.; Ku, J.C. Potential of Samanea saman pod meal for enteric methane mitigation in crossbred heifers fed low-quality tropical grass. Agric. Forest. Meteorol. 2018, 258, 108–116. [Google Scholar] [CrossRef]
- Valencia-Salazar, S.S.; Jiménez-Ferrer, G.; Arango, J.; Molina-Botero, I.C.; Chirinda, N.; Piñeiro-Vázquez, A.; Jiménez-Ocampo, R.; Nahed-Toral, J.; Ku-Vera, J. Enteric methane mitigation and fermentation kinetics of forage species from Southern Mexico: In vitro screening. Agroforest Syst. 2021, 95, 293–305. [Google Scholar] [CrossRef]
- García del Valle, Y.; Naranjo, E.; Caballero, J.; Martorell, C.; Ruan-Soto, F.; Enríquez, P. Cultural significance of wild mammals in mayan and mestizo communities of the Lacandon Rainforest, Chiapas, Mexico. J. Ethnobiol. Ethnomed. 2015, 11, 36. [Google Scholar] [CrossRef] [Green Version]
- ISO. ISO 6496. Animal Feeding Stuffs–Determination of Moisture and Other Volatile Matter Content; International Organization for Standardization: Geneva, Switzerland, 1999. [Google Scholar]
- AOAC. Official Methods of Analysis, 16th ed.; Association of Official Analytical Chemists (AOAC): Washington, DC, USA, 2005. [Google Scholar]
- AOAC. Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemists (AOAC): Washington, DC, USA, 1990. [Google Scholar]
- Van Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fibre, neutral detergent fibre and non-starch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1999, 74, 3583–3597. [Google Scholar] [CrossRef]
- ISO. ISO 9831. Animal Feeding Stuffs, Animal Products, and Faeces or Urine–Determination of Gross Calorific Value–Bomb Calorimeter method; International Organization for Standardization: Geneva, Switzerland, 1998. [Google Scholar]
- Tilley, J.M.A.; Terry, R.A. A two-stage technique for the in vitro digestion of forage crops. J. Br. Grassl. Soc. 1963, 18, 104–111. [Google Scholar] [CrossRef]
- Hagerman, A.E.; Butler, L.G. Protein precipitation method for the quantitative determination of tannins. J. Agric. Food Chem. 1978, 26, 809–812. [Google Scholar] [CrossRef]
- Domínguez, X.A. Métodos de Investigación Fitoquímica; Editorial Limusa: Mexico City, Mexico, 1979. [Google Scholar]
- ICONTEC. NTC 668. Alimentos y Materias Primas. Determinación de los Contenidos de Grasa y Fibra Cruda; Instituto Colombiano de Normas Técnicas y Certificación (ICONTEC): Bogotá, Colombia, 1973; p. 8. [Google Scholar]
- Menke, K.H.; Raab, L.; Salewski, A.; Steingass, H.; Fritz, D.; Schneider, W. The estimation of the digestibility and metabolisable energy content of ruminant feedstuffs from the gas production when they are incubated with rumen liquor. J. Agric. Sci. 1979, 93, 217. [Google Scholar] [CrossRef] [Green Version]
- Menke, K.H.; Steingass, H. Estimation of the energetic feed value obtained from chemical analysis and in vitro gas production using rumen fluid. Anim. Res. Dev. 1988, 28, 7–55. [Google Scholar]
- Getachew, G.; Makkar, H.P.S.; Becker, K. Tropical browses: Contents of phenolic compounds, in vitro gas production and stoichiometric relationship between short chain fatty acid and in vitro gas production. J. Agric. Sci. 2002, 139, 341–352. [Google Scholar] [CrossRef]
- Theodorou, M.K.; Williams, B.A.; Dhanoa, M.S.; McAllan, A.B.; France, J.A. Simple gas production method using a pressure transducer to determine the fermentation kinetics of ruminant feeds. Anim. Feed Sci. Technol. 1994, 48, 185–197. [Google Scholar] [CrossRef]
- Yáñez-Ruiz, D.R.; Bannink, A.; Dijkstra, J.; Kebreab, E.; Morgavi, D.P.; O’Kiely, P.; Reynolds, C.K.; Schwarm, A.; Shingfield, K.J.; Yu, Z.; et al. Design, implementation and interpre-tation of in vitro batch culture experiments to assess enteric methane mitigation in ruminants—A review. Anim. Feed Sci. Technol. 2016, 216, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Lavrenčič, A.; Mills, C.R.; Stefanon, B. Application of the Gompertz model to describe the fermentation characteristics of chemical components in forages. Anim. Sci. J. 1998, 66, 155–161. [Google Scholar] [CrossRef]
- SAS Institute. User’s Guide: Statistics Version 9.4; SAS Inst. Inc.: Cary, NC, USA, 2012. [Google Scholar]
- Gaviria-Uribe, X.; Bolivar, D.; Rosenstock, T.; Molina-Botero, I.C.; Chirinda, N.; Barahona, R.; Arango, J. Nutritional quality, voluntary intake and enteric methane emissions of diets based on novel Cayman grass and its associations with two Leucaena shrub legumes. Vet. Sci. Front. 2020, 7, 579189. [Google Scholar] [CrossRef] [PubMed]
- Albores-Moreno, S.; Alayón-Gamboa, J.A.; Miranda-Romero, L.A.; Alarcón-Zúñiga, B.; Jiménez-Ferrer, G.; Ku-Vera, J.C.; Piñeiro-Vázquez, A.T. 2018 Effect of tree foliage supplementation of tropical grass diet on in vitro digestibility and fermentation, microbial biomass synthesis and enteric methane production in ruminants. Trop. Anim. Health Prod. 2018, 58, 893–904. [Google Scholar] [CrossRef]
- Ortega-Gómez, R.; Castillo-Gallegos, E.; Jarillo-Rodríguez, J.; Escobar-Hernández, R.; Ocaña-Zavaleta, E.; Valles de la Mora, B. Nutritive Quality of Ten Grasses During the Rainy Season In A Hot-Humid Climate And Ultisol Soil. Trop. Subtrop. Agroecosyst. 2011, 13, 481–491. [Google Scholar]
- Yusuf, A.O.; Egbinola, O.O.; Ekunseitan, D.A.; Salem, A.Z.M. Chemical characterization and in vitro methane production of selected agroforestry plants as dry season feeding of ruminants livestock. Agroforest Syst. 2020, 94, 1481–1489. [Google Scholar] [CrossRef]
- Tagliapietra, F.; Cattani, M.; Hansen, H.H.; Hindrichsen, I.K.; Bailoni, L.; Schiavon, S. Metabolizable energy content of feeds based on 24 or 48 h in situ NDF digestibility and on in vitro 24 h gas production methods. Anim. Feed Sci. Technol. 2011, 170, 182–191. [Google Scholar] [CrossRef]
- Krizsan, S.J.; Pang, D.; Fatehi, F.; Rinne, M.; Huhtanen, P. Metabolisable energy of grass and red clover silages fed to sheep at maintenance level. Animal 2020, 14, 753–762. [Google Scholar] [CrossRef] [PubMed]
- Narváez, N.; Lascano, C.E. Caracterización química de especies arbóreas tropicales con potencial forrajero en Colombia. Pasturas Trop. 2004, 26, 1–8. [Google Scholar]
- Lee, M.R.F.; Jordana Rivero, M.; Cone, J.W. The role of pasture in the diet of ruminant livestock. In Improving Grassland and Pasture Management in Temperature Agriculture; Marshall, A., Collins, R., Eds.; Burleigh Dodds Science Publishing Limited: Cambridge, UK, 2018. [Google Scholar] [CrossRef]
- Goel, G.; Makkar, H.P. Methane mitigation from ruminants using tannins and saponins. Trop. Anim. Health Prod. 2012, 44, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Tedeschi, L.O.; Muir, J.P.; Naumann, H.D.; Norris, A.B.; Ramírez-Restrepo, C.A.; Mertens-Talcott, S.U. Nutritional Aspects of Ecologically Relevant Phytochemicals in Ruminant Production. Front. Vet. Sci. 2021, 8, 628445. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, D.E.; Arreaza, L.C.; Abadía, B. Estudio de la cinética de degradación in vitro de cuatro forrajes tropicales y una leguminosa de clima templado. Cienc. Tecnol. Agropecu. 2005, 6, 58–68. [Google Scholar] [CrossRef] [Green Version]
- Molina-Botero, I.C.; Mazabel, J.; Arceo-Castillo, J.; Urrea-Benitez, J.L.; Olivera-Castillo, L.; Barahona-Rosales, R.; Chirinda, N.; Ku-Vera, J.; Arango, J. Effect of the addition of Enterolobium cyclocarpum pods and Gliricidia sepium forage on dry matter degradation, volatile fatty acid concentration, and in vitro methane production. Trop. Anim. Health Prod. 2020, 52, 2787–2798. [Google Scholar] [CrossRef]
- Patra, A.K.; Yu, Z. Effect s of vanillin, quillaja saponin, and essential oils on in vitro fermentation and protein degrading microorganisms of the rumen. Appl. Microbiol. Biotechnol. 2014, 98, 897–905. [Google Scholar] [CrossRef]
- Aguiar, R.; Wink, M. Do naïve ruminants degrade alkaloids in the rumen? J. Chem. Ecol. 2005, 31, 761–787. [Google Scholar] [CrossRef] [PubMed]
- Li, M.M.; White, R.R.; Guan, L.L.; Harthan, L.; Hanigan, M.D. Metatranscriptomic analyses reveal ruminal pH regulates fiber degradation and fermentation by shifting the microbial community and gene expression of carbohydrate-active enzymes. Anim. Microbiome 2021, 3, 32. [Google Scholar] [CrossRef]
- Bodas, R.; Prieto, N.; García-González, R.; Andrés, S.; Giráldez, F.J.; López, S. Manipulation of rumen fermentation and methane production with plant secondary metabolites. Anim. Feed Sci. Technol. 2012, 176, 78–93. [Google Scholar] [CrossRef]
- Meale, S.J.; Chaves, A.V.; Baah, J.; McAllister, T.A. Methane production of different forages in in vitro ruminal fermentation. Asian-Aust. J. Anim. Sci. 2012, 25, 86. [Google Scholar] [CrossRef]
- Vongsamphanh, P.; Inthapanya, S.; Preston, T.; Van Dung, D.; Xuan Ba, N. Effect of leaves from sweet or bitter cassava and brewers’ grains on methane production in an in vitro rumen incubation of cassava root pulp-urea. Livest. Res. Rural Dev. 2018, 30, 167. [Google Scholar]
- Randami, D.; Chaudhry, A.S.; Seal, C.J. Alkaloid and polyphenol analysis by HPLC in green and black tea powders and their potential use as additives in ruminant diets. AIP Conf. Proc. 2018, 1927, 030008. [Google Scholar] [CrossRef] [Green Version]
- Khiaosa-Ard, R.; Mahmood, M.; Lerch, F.; Traintinger, F.P.; Petri, R.M.; Münnich, M.; Zebeli, Q. Physicochemical stressors and mixed alkaloid supplementation modulate ruminal microbiota and fermentation in vitro. Anaerobe 2020, 65, 102263. [Google Scholar] [CrossRef]
- Patra, A.K.; Saxena, J. A new perspective on the use of plant secondary metabolites to inhibit methanogenesis in the rumen. Phytochemistry 2010, 71, 1198–1222. [Google Scholar] [CrossRef] [PubMed]
- Puchala, R.; Animut, G.; Patra, A.K.; Detweiler, G.D.; Wells, J.E.; Varel, V.H.; Sahlu, T. Methane emissions by goats consuming Sericea lespedeza at different feeding frequencies. Anim. Feed Sci. Technol. 2012, 175, 76–84. [Google Scholar] [CrossRef]
- Min, B.R.; Solaiman, S.; Shange, R.; Eun, J.S. Gastrointestinal bacterial and methanogenic archaea diversity dynamics associated with condensed tannin-containing pine bark diet in goats using 16S rDNA amplicon pyrosequencing. Int. J. Food Microbiol. 2014, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- McSweeney, C.S.; Palmer, B.; Bunch, R.; Krause, D.O. Microbial interactions with tannins: Nutritional consequences for ruminants. Anim. Feed. Sci. Technol. 2001, 91, 83–93. [Google Scholar] [CrossRef]
- Vargas, J.; Cárdenas, E.; Pabón, M.; Carulla, J. Emisión de metano entérico en rumiantes en pastoreo. Arch. De Zootec. 2012, 61, 51–66. [Google Scholar] [CrossRef] [Green Version]
- Piñeiro-Vázquez, A.T.; Canul-Solis, J.R.; Casanova-Lugo, F.; Chay-Canul, A.J.; Ayala-Burgos, A.; Solorio-Sánchez, J.; Aguilar-Pérez, C.F.; Ku-Vera, J.C. Emisión de metano en ovinos alimentados con Pennisetum purpureum y árboles que contienen taninos condensados. Rev. Mex. Cienc. Pecu. 2017, 2, 111–119. [Google Scholar] [CrossRef] [Green Version]
- Bhatta, R.; Tajima, K.; Takusari, N.; Higuchi, K.; Enishi, O.; Kurihara, M. Comparison of in vivo and in vitro techniques for methane production from Ruminant diets. Asian-Aust. J. Anim. Sci. 2007, 20, 1049–1056. [Google Scholar] [CrossRef]

| g kg−1 DM | MJ kg−1 DM | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Species | DM | OM | NDF | ADF | CP | EE | IVDDM | CT | GE | ME | NE |
| Spondias mombin (SM) | 259.75 | 852.29 | 307.77 | 171.18 | 126.95 | 44.78 | 638.59 | 9.90 | 16.25 | ||
| Acacia pennatula (AP) | 505.42 | 924.85 | 492.56 | 210.34 | 192.69 | 39.25 | 447.44 | 31.1 | 20.92 | ||
| Parmentiera aculeata (PA) | 308.95 | 874.47 | 614.35 | 268.81 | 183.17 | 13.85 | 548.37 | 0.00 | 18.04 | ||
| Brosimum alicastrum (BA) | 489.18 | 821.28 | 298.17 | 269.22 | 116.21 | 29.92 | 686.38 | 0.00 | 15.65 | ||
| Bursera simaruba (BS) | 356.71 | 900.82 | 354.37 | 249.23 | 99.07 | 25.05 | 471.37 | 200.1 | 18.92 | ||
| Treatments | |||||||||||
| SM15-PP85 | 213.82 | 839.92 | 542.46 | 311.22 | 132.88 | 23.42 | 542.46 | 1.50 | 15.99 | 7.40 | 4.54 |
| SM30-PP70 | 221.93 | 842.10 | 501.04 | 286.50 | 131.83 | 27.19 | 501.04 | 3.00 | 16.04 | 7.75 | 4.80 |
| AP15-PP85 | 250.66 | 850.80 | 570.18 | 317.09 | 142.74 | 22.59 | 570.18 | 4.70 | 16.69 | 7.56 | 4.58 |
| AP30-PP70 | 295.61 | 863.87 | 556.48 | 298.25 | 151.56 | 25.53 | 556.48 | 9.30 | 17.44 | 7.46 | 4.51 |
| PA15-PP85 | 221.18 | 843.24 | 588.45 | 325.86 | 141.31 | 18.78 | 588.45 | 0.00 | 16.26 | 7.17 | 4.31 |
| PA30-PP70 | 236.66 | 848.75 | 593.02 | 315.79 | 148.70 | 17.91 | 593.02 | 0.00 | 16.58 | 7.18 | 4.27 |
| BA15-PP85 | 248.23 | 835.26 | 541.02 | 325.92 | 131.27 | 21.19 | 541.02 | 0.00 | 15.90 | 7.60 | 4.65 |
| BA30-PP70 | 290.75 | 832.80 | 498.17 | 315.92 | 128.61 | 22.73 | 498.17 | 0.00 | 15.86 | 7.82 | 4.82 |
| BS15-PP85 | 228.35 | 847.20 | 549.45 | 322.92 | 128.70 | 20.46 | 549.45 | 30.0 | 16.39 | 6.92 | 4.23 |
| BS30-PP70 | 251.00 | 856.66 | 515.03 | 309.92 | 123.47 | 21.27 | 515.03 | 60.0 | 16.84 | 6.05 | 3.72 |
| 100PP | 205.70 | 837.73 | 583.88 | 335.93 | 133.93 | 19.65 | 583.88 | 0.00 | 15.95 | 7.08 | 4.30 |
| Species | Alkaloids | Cyanogenic Glycosides | Saponins | ||
|---|---|---|---|---|---|
| Mayer | Draggendorff | Wagner | |||
| Spondias mombin (SM) | − | + | ++ | − | − |
| Acacia pennatula (AP) | − | ++ | ++ | ++++ | + |
| Parmentiera aculeata (PA) | ++++ | ++++ | ++++ | − | − |
| Brosimum alicastrum (BA) | +++ | + | − | − | − |
| Bursera simaruba (BS) | − | − | − | − | − |
| Parameters | |||||||
|---|---|---|---|---|---|---|---|
| Rations | a | b | c | TIP (h) | GIP (mL) | MGPR (mL h−1) | LP |
| 100PP | 261.304 | 1.009 | 0.079 | 12.786 | 96.110 | 7.580 | 0.120 |
| SM15-PP85 | 266.533 | 1.101 * | 0.084 | 13.170 | 98.033 | 8.200 * | 1.206 * |
| SM30-PP70 | 287.369 * | 1.048 | 0.083 | 12.690 | 105.697 * | 8.730 * | 0.583 |
| AP15-PP85 | 263.756 | 1.017 | 0.079 | 12.866 | 97.010 | 7.670 | 0.216 |
| AP30-PP70 | 240.909 * | 0.953 * | 0.078 | 12.160 | 88.607 * | 6.947 * | −0.593 * |
| PA15-PP85 | 220.971 * | 1.003 | 0.086 * | 11.616 * | 81.270 * | 6.613 * | 0.030 |
| PA30-PP70 | 219.015 * | 0.925 * | 0.081 | 11.380 * | 80.553 * | 5.546 * | −0.926 * |
| BA15-PP85 | 277.702 | 0.998 | 0.081 | 12.396 | 102.140 | 8.220 * | −0.020 |
| BA30-PP70 | 283.543 * | 0.944 * | 0.083 | 11.370 * | 104.287 * | 8.676 * | −0.676 * |
| BS15-PP85 | 235.527 * | 0.979 | 0.076 | 12.826 | 86.620 * | 6.610 * | −0.286 |
| BS30-PP70 | 214.480 * | 0.917 * | 0.060 * | 15.173 * | 78.887 * | 4.766 * | −1.386 * |
| MSE | 8.089 | 0.0201 | 0.003 | 0.456 | 2.975 | 0.177 | 0.267 |
| p-Value | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.00001 |
| DM DEG % | pH | SCFA mMol l−1 | CO2 mL g−1 IOM | CO2 mL g−1 DOM | CH4 mL g−1 IOM | CH4 mL g−1 DOM | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 24 | 48 | 24 | 48 | 24 | 48 | 24 | 48 | 24 | 48 | 24 | 48 | ||
| 100PP | 47.01 | 55.84 | 6.68 | 6.69 | 77.45 | 168.07 | 155.89 | 102.03 | 112.27 | 14.08 | 26.74 | 8.55 | 19.27 |
| SM15-PP85 | 47.13 | 60.69 * | 6.78 * | 6.79 | 84.86 * | 176.37 | 202.58 | 153.78 | 158.15 | 13.58 | 26.20 | 8.22 | 20.47 |
| SM30-PP70 | 56.65 * | 63.21 * | 6.67 | 6.63 | 92.80 * | 221.38 | 268.74 * | 185.08 * | 219.06 * | 16.75 | 37.51 * | 12.44 | 30.97 * |
| AP15-PP85 | 46.29 | 55.02 | 6.72 | 6.68 | 85.29 * | 148.72 | 166.34 | 87.30 | 116.35 | 1256 | 25.81 | 7.38 | 18.04 |
| AP30-PP70 | 46.04 | 51.43 * | 6.69 | 6.74 | 80.50 | 156.73 | 225.55 | 109.89 | 139.02 | 12.32 | 29.47 | 9.74 | 18.91 |
| PA15-PP85 | 43.77 | 51.73 * | 6.72 | 6.83 | 77.25 | 142.47 | 244.72 * | 79.88 | 149.69 | 11.37 | 29.87 | 6.39 | 19.86 |
| PA30-PP70 | 41.92* | 50.95 * | 6.70 | 6.94 | 75.13 | 321.66 * | 267.98 * | 171.45 | 174.07 * | 26.16 * | 35.68 * | 13.95 * | 23.17 * |
| BA15-PP85 | 51.37 | 60.11 * | 6.68 | 6.76 | 89.62 * | 259.01 | 272.44 * | 172.51 | 211.89 * | 23.61 * | 37.76 * | 14.14 * | 29.39 * |
| BA30-PP70 | 55.98 * | 62.24 * | 6.68 | 6.87 | 95.42 * | 269.39 | 285.77 * | 195.74 * | 230.51 * | 22.95 * | 38.06 * | 16.68 * | 30.69 * |
| BS15-PP85 | 45.01 | 54.52 | 6.70 | 6.77 | 75.49 | 305.59 * | 265.79 * | 175.28 * | 184.96 * | 20.68 | 31.81 | 12.95 | 22.16 |
| BS30-PP70 | 39.17 * | 54.47 | 6.70 | 6.72 | 57.88 * | 240.56 * | 208.22 | 118.20 | 143.09 | 16.35 | 21.02 | 6.65 | 14.43 |
| MSE | 2.06 | 1.48 | 0.02 | 0.11 | 1.67 | 38.30 | 30.51 | 29.03 | 24.74 | 2.30 | 2.98 | 1.94 | 2.11 |
| p-value | <0.0001 | <0.0001 | 0.0028 | 0.1467 | <0.0001 | <0.0001 | 0.0003 | 0.0003 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valencia-Salazar, S.S.; Jiménez-Ferrer, G.; Molina-Botero, I.C.; Ku-Vera, J.C.; Chirinda, N.; Arango, J. Methane Mitigation Potential of Foliage of Fodder Trees Mixed at Two Levels with a Tropical Grass. Agronomy 2022, 12, 100. https://doi.org/10.3390/agronomy12010100
Valencia-Salazar SS, Jiménez-Ferrer G, Molina-Botero IC, Ku-Vera JC, Chirinda N, Arango J. Methane Mitigation Potential of Foliage of Fodder Trees Mixed at Two Levels with a Tropical Grass. Agronomy. 2022; 12(1):100. https://doi.org/10.3390/agronomy12010100
Chicago/Turabian StyleValencia-Salazar, Sara Stephanie, Guillermo Jiménez-Ferrer, Isabel Cristina Molina-Botero, Juan Carlos Ku-Vera, Ngonidzashe Chirinda, and Jacobo Arango. 2022. "Methane Mitigation Potential of Foliage of Fodder Trees Mixed at Two Levels with a Tropical Grass" Agronomy 12, no. 1: 100. https://doi.org/10.3390/agronomy12010100
APA StyleValencia-Salazar, S. S., Jiménez-Ferrer, G., Molina-Botero, I. C., Ku-Vera, J. C., Chirinda, N., & Arango, J. (2022). Methane Mitigation Potential of Foliage of Fodder Trees Mixed at Two Levels with a Tropical Grass. Agronomy, 12(1), 100. https://doi.org/10.3390/agronomy12010100







