Ensifer aridi LMR001T Symbiosis and Tolerance to Stress Do Not Require the Alternative Sigma Factor RpoE2
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Growth
2.2. Phenotypic Characterization of the ΔrpoE2 Strain
2.2.1. Plant Nodulation Assay
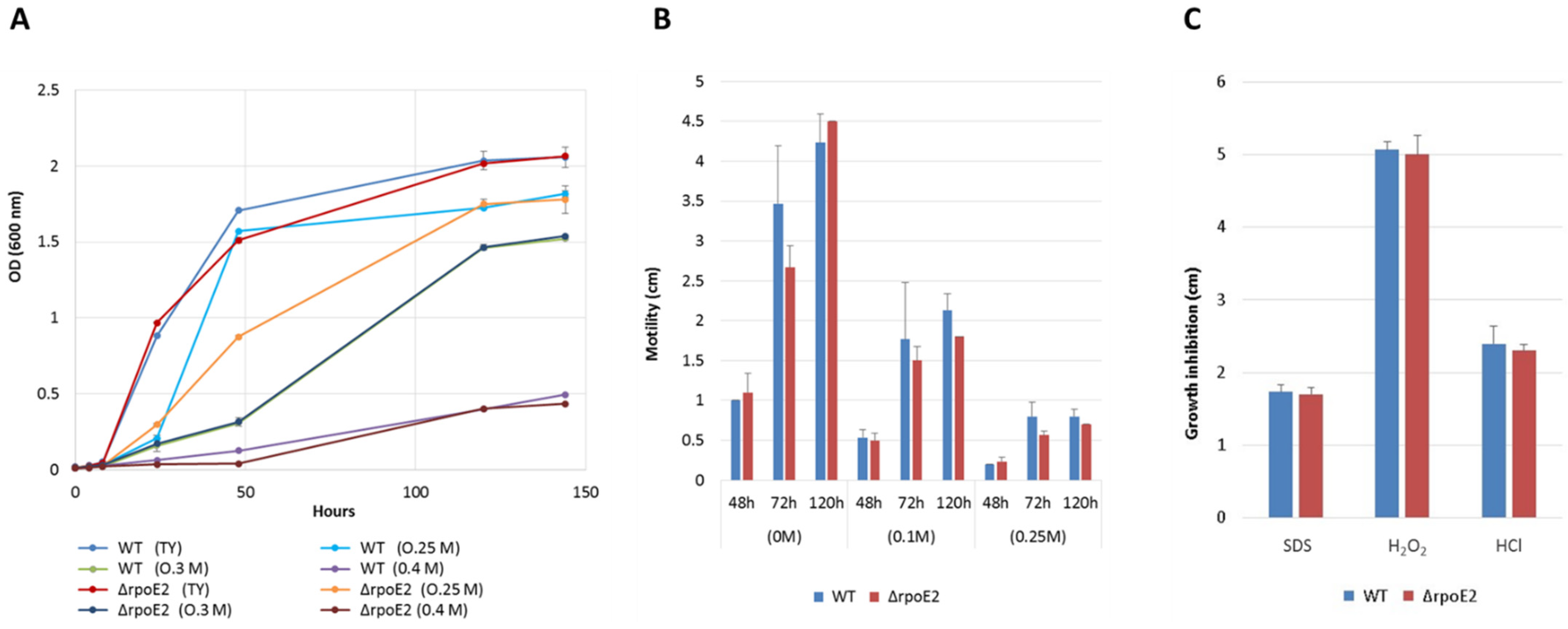
2.2.2. Motility Assay
2.2.3. Salinity Tolerance
2.2.4. Sensitivity to Detergent, Acid, and Oxidative Stresses
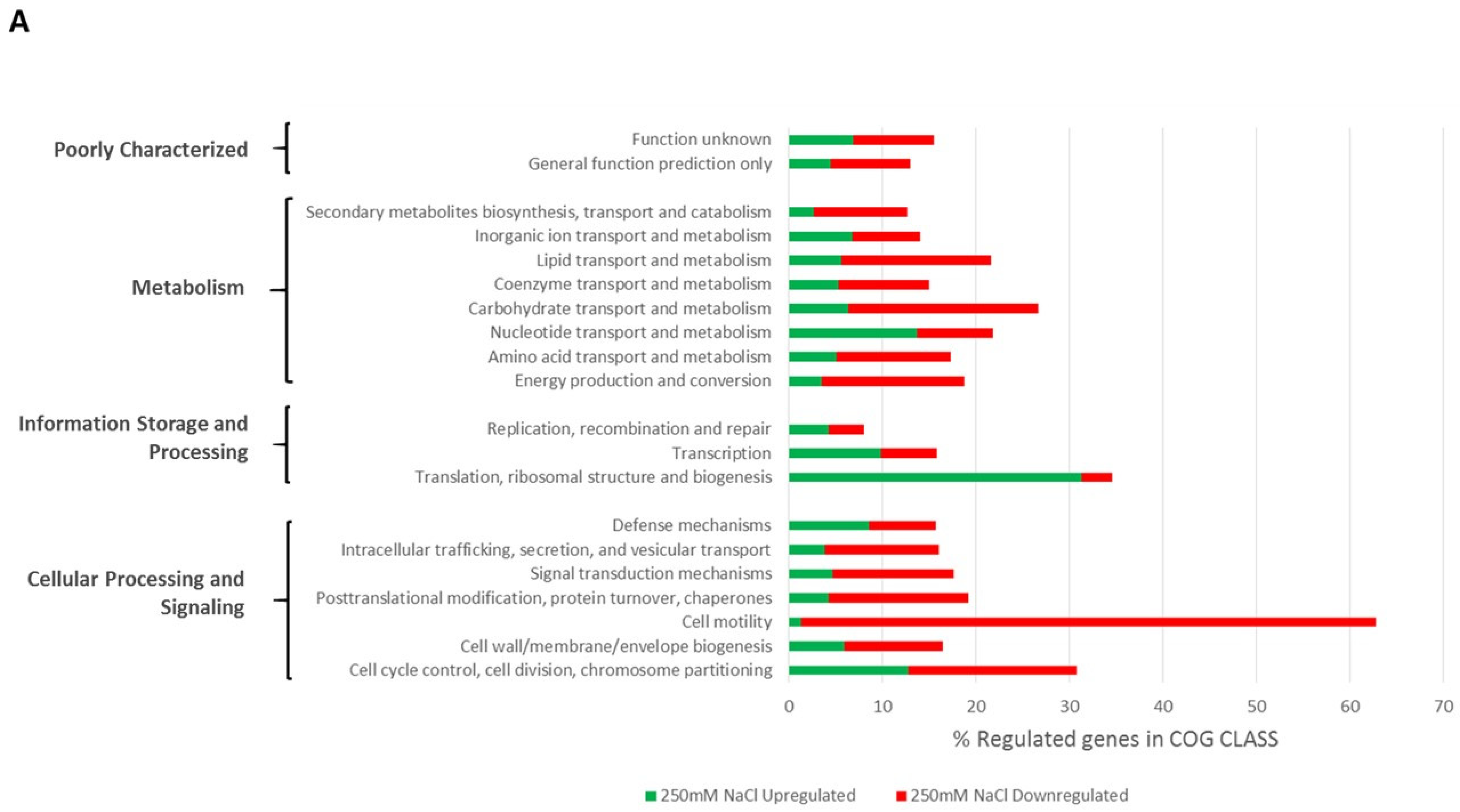
2.3. RNAseq Analysis and Characterization of Differentially Regulated Genes in LMR001 upon NaCl- or PEG8000-Induced Hyperosmotic Stresses
2.4. Real-Time PCR on Selected Genes
2.5. LMR001 ΔrpoE2 Mutant Construction
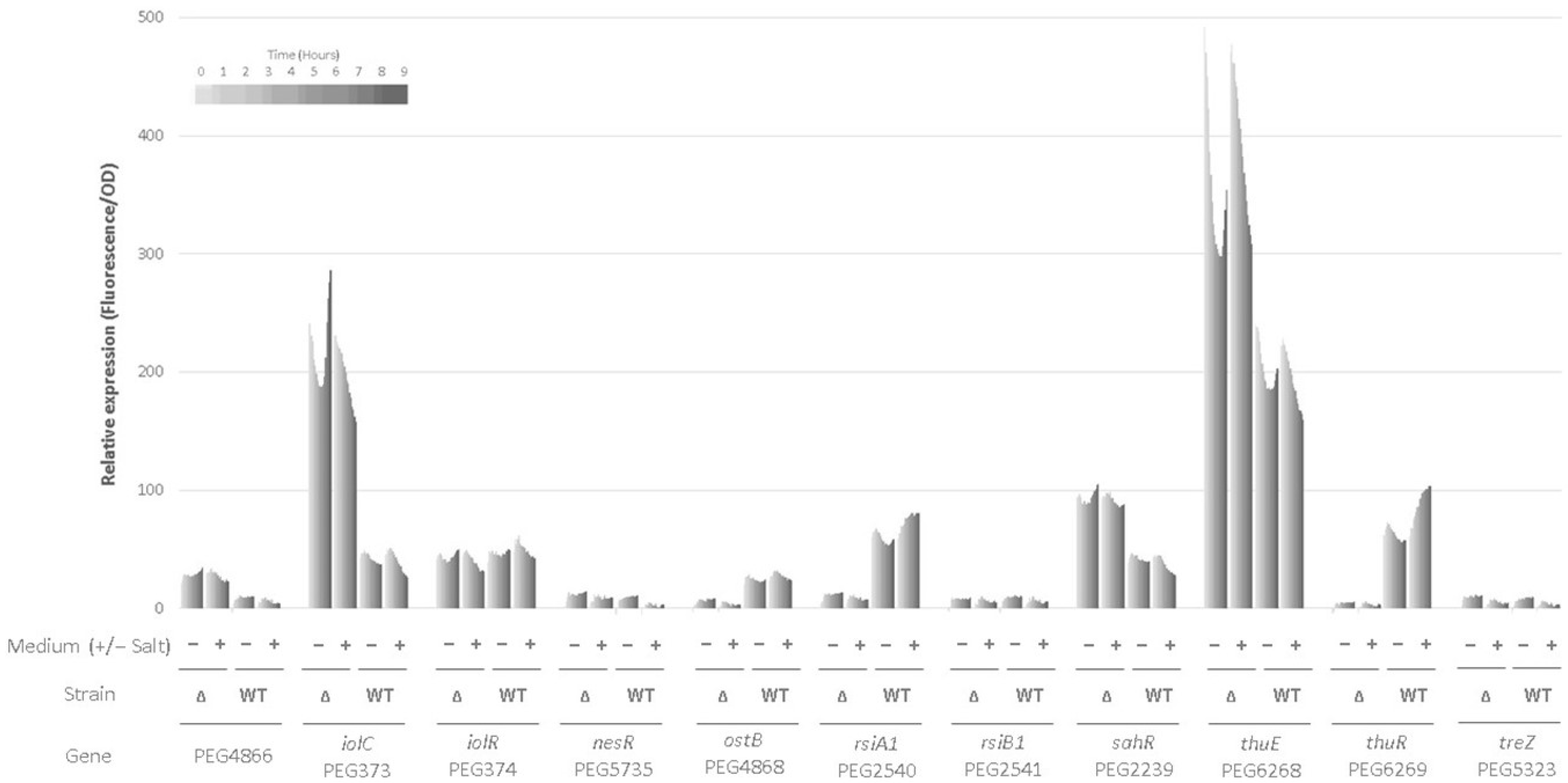
2.6. Use of Promoter-Gfp Fusions for Transcriptional Analyses in the Wild-Type and the ∆rpoE2 Strains
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sakrouhi, I.; Belfquih, M.; Sbabou, L.; Moulin, P.; Bena, G.; Filali-Maltouf, A.; Le Quéré, A. Recovery of Symbiotic Nitrogen Fixing Acacia Rhizobia from Merzouga Desert Sand Dunes in South East Morocco—Identification of a Probable New Species of Ensifer Adapted to Stressed Environments. Syst. Appl. Microbiol. 2016, 39, 122–131. [Google Scholar] [CrossRef]
- Rocha, G.; Le Queré, A.; Medina, A.; Cuéllar, A.; Contreras, J.-L.; Carreño, R.; Bustillos, R.; Muñoz-Rojas, J.; Villegas, M. del C.; Chaintreuil, C.; et al. Diversity and Phenotypic Analyses of Salt- and Heat-Tolerant Wild Bean Phaseolus filiformis Rhizobia Native of a Sand Beach in Baja California and Description of Ensifer aridi Sp. Nov. Arch. Microbiol. 2020, 202, 309–322. [Google Scholar] [CrossRef] [PubMed]
- Tak, N.; Awasthi, E.; Bissa, G.; Meghwal, R.R.; James, E.K.; Sprent, J.S.; Gehlot, H.S. Multi Locus Sequence Analysis and Symbiotic Characterization of Novel Ensifer Strains Nodulating Tephrosia Spp. in the Indian Thar Desert. Syst. Appl. Microbiol. 2016, 39, 534–545. [Google Scholar] [CrossRef] [PubMed]
- Hakim, S.; Imran, A.; Mirza, M.S. Phylogenetic Diversity Analysis Reveals Bradyrhizobium yuanmingense and Ensifer aridi as Major Symbionts of Mung Bean (Vigna radiata L.) in Pakistan. Braz. J. Microbiol. 2021, 52, 311–324. [Google Scholar] [CrossRef] [PubMed]
- Lamin, H.; Alami, S.; Bouhnik, O.; ElFaik, S.; Abdelmoumen, H.; Bedmar, E.J.; Missbah-El Idrissi, M. Nodulation of Retama monosperma by Ensifer aridi in an Abandonned Lead Mine Soils in Eastern Morocco. Front. Microbiol. 2019, 10, 1456. [Google Scholar] [CrossRef]
- Le Quéré, A.; Tak, N.; Gehlot, H.S.; Lavire, C.; Meyer, T.; Chapulliot, D.; Rathi, S.; Sakrouhi, I.; Rocha, G.; Rohmer, M.; et al. Genomic Characterization of Ensifer aridi, a Proposed New Species of Nitrogen-Fixing Rhizobium Recovered from Asian, African and American Deserts. BMC Genom. 2017, 18, 85. [Google Scholar] [CrossRef] [PubMed]
- Zahran, H.H. Rhizobium -Legume Symbiosis and Nitrogen Fixation under Severe Conditions and in an Arid Climate. Microbiol. Mol. Biol. Rev. 1999, 63, 968–989. [Google Scholar] [CrossRef]
- Denich, T.J.; Beaudette, L.A.; Lee, H.; Trevors, J.T. Effect of Selected Environmental and Physico-Chemical Factors on Bacterial Cytoplasmic Membranes. J. Microbiol. Methods 2003, 52, 149–182. [Google Scholar] [CrossRef]
- Chang, W.-S.; van de Mortel, M.; Nielsen, L.; Nino de Guzman, G.; Li, X.; Halverson, L.J. Alginate Production by Pseudomonas putida Creates a Hydrated Microenvironment and Contributes to Biofilm Architecture and Stress Tolerance under Water-Limiting Conditions. J. Bacteriol. 2007, 189, 8290–8299. [Google Scholar] [CrossRef]
- Finn, S.; Condell, O.; McClure, P.; Amézquita, A.; Fanning, S. Mechanisms of Survival, Responses and Sources of Salmonella in Low-Moisture Environments. Front. Microbiol. 2013, 4, 331. [Google Scholar] [CrossRef]
- Lebre, P.H.; De Maayer, P.; Cowan, D.A. Xerotolerant Bacteria: Surviving through a Dry Spell. Nat. Rev. Microbiol. 2017, 15, 285–296. [Google Scholar] [CrossRef]
- Vriezen, J.A.C.; de Bruijn, F.J.; Nüsslein, K. Responses of Rhizobia to Desiccation in Relation to Osmotic Stress, Oxygen, and Temperature. Appl. Environ. Microbiol. 2007, 73, 3451–3459. [Google Scholar] [CrossRef]
- Sharma, M.P.; Grover, M.; Chourasiya, D.; Bharti, A.; Agnihotri, R.; Maheshwari, H.S.; Pareek, A.; Buyer, J.S.; Sharma, S.K.; Schütz, L.; et al. Deciphering the Role of Trehalose in Tripartite Symbiosis Among Rhizobia, Arbuscular Mycorrhizal Fungi, and Legumes for Enhancing Abiotic Stress Tolerance in Crop Plants. Front. Microbiol. 2020, 11, 509919. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, C.Y. Generally Stressed Out Bacteria: Environmental Stress Response Mechanisms in Gram-Positive Bacteria. Integr. Comp. Biol. 2020, 60, 126–133. [Google Scholar] [CrossRef]
- Fiebig, A.; Herrou, J.; Willett, J.; Crosson, S. General Stress Signaling in the Alphaproteobacteria. Annu. Rev. Genet. 2015, 49, 603–625. [Google Scholar] [CrossRef]
- Lori, C.; Kaczmarczyk, A.; de Jong, I.; Jenal, U. A Single-Domain Response Regulator Functions as an Integrating Hub To Coordinate General Stress Response and Development in Alphaproteobacteria. mBio 2018, 9, e00809-18. [Google Scholar] [CrossRef]
- Beringer, J.E. R Factor Transfer in Rhizobium leguminosarum. Microbiology 1974, 84, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Sambrook, J.; Fritsch, E.; Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd ed.; Cold Spring Harbor: New York, NY, USA, 1989; Volume 2, ISBN 978-0-87969-309-1. [Google Scholar]
- Barra, L.; Fontenelle, C.; Ermel, G.; Trautwetter, A.; Walker, G.C.; Blanco, C. Interrelations between Glycine Betaine Catabolism and Methionine Biosynthesis in Sinorhizobium meliloti Strain 102F34. J. Bacteriol. 2006, 188, 7195–7204. [Google Scholar] [CrossRef]
- Broughton, W.J.; Dilworth, M.J. Control of Leghaemoglobin Synthesis in Snake Beans. Biochem. J. 1971, 125, 1075–1080. [Google Scholar] [CrossRef] [PubMed]
- Maynaud, G.; Brunel, B.; Mornico, D.; Durot, M.; Severac, D.; Dubois, E.; Navarro, E.; Cleyet-Marel, J.-C.; Le Quéré, A. Genome-Wide Transcriptional Responses of Two Metal-Tolerant Symbiotic Mesorhizobium Isolates to Zinc and Cadmium Exposure. BMC Genom. 2013, 14, 292. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S.; Sato, Y.; Kawashima, M.; Furumichi, M.; Tanabe, M. Data, Information, Knowledge and Principle: Back to Metabolism in KEGG. Nucl. Acids Res. 2014, 42, D199–D205. [Google Scholar] [CrossRef]
- Tatusov, R.L. The COG Database: New Developments in Phylogenetic Classification of Proteins from Complete Genomes. Nucl. Acids Res. 2001, 29, 22–28. [Google Scholar] [CrossRef]
- Pfaffl, M.W. A New Mathematical Model for Relative Quantification in Real-Time RT-PCR. Nucl. Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Kuo, T. A Simple and Rapid Method for the Preparation of Gram-Negative Bacterial Genomic DNA. Nucl. Acids Res. 1993, 21, 2260. [Google Scholar] [CrossRef]
- Quandt, J.; Hynes, M.F. Versatile Suicide Vectors Which Allow Direct Selection for Gene Replacement in Gram-Negative Bacteria. Gene 1993, 127, 15–21. [Google Scholar] [CrossRef]
- Simon, R.; Priefer, U.; Pühler, A. A Broad Host Range Mobilization System for In Vivo Genetic Engineering: Transposon Mutagenesis in Gram Negative Bacteria. Nat. Biotechnol. 1983, 1, 784–791. [Google Scholar] [CrossRef]
- Miller, W.G.; Leveau, J.H.J.; Lindow, S.E. Improved Gfp and InaZ Broad-Host-Range Promoter-Probe Vectors. Mol. Plant Microbe Interact. 2000, 13, 1243–1250. [Google Scholar] [CrossRef] [PubMed]
- An, Z.; Wang, Q.; Hu, Y.; Zhao, Y.; Li, Y.; Cheng, H.; Huang, H. Co-Extraction of High-Quality RNA and DNA from Rubber Tree (Hevea brasiliensis). Afr. J. Biotechnol. 2012, 11, 9308–9314. [Google Scholar] [CrossRef]
- Roose-Amsaleg, C.L.; Garnier-Sillam, E.; Harry, M. Extraction and Purification of Microbial DNA from Soil and Sediment Samples. Appl. Soil Ecol. 2001, 18, 47–60. [Google Scholar] [CrossRef]
- Anders, S.; Huber, W. Differential Expression Analysis for Sequence Count Data. Genome Biol. 2010, 11, R106. [Google Scholar] [CrossRef]
- Alavi, P.; Starcher, M.R.; Zachow, C.; Müller, H.; Berg, G. Root-Microbe Systems: The Effect and Mode of Interaction of Stress Protecting Agent (SPA) Stenotrophomonas rhizophila DSM14405T. Front. Plant Sci. 2013, 4, 141. [Google Scholar] [CrossRef] [PubMed]
- Dressaire, C.; Moreira, R.N.; Barahona, S.; Alves de Matos, A.P.; Arraiano, C.M. BolA Is a Transcriptional Switch That Turns Off Motility and Turns On Biofilm Development. mBio 2015, 6, e02352-14. [Google Scholar] [CrossRef]
- Li, S.; Liang, H.; Wei, Z.; Bai, H.; Li, M.; Li, Q.; Qu, M.; Shen, X.; Wang, Y.; Zhang, L. An Osmoregulatory Mechanism Operating through OmpR and LrhA Controls the Motile-Sessile Switch in the Plant Growth-Promoting Bacterium Pantoea alhagi. Appl. Environ. Microbiol. 2019, 85, e00077-19. [Google Scholar] [CrossRef] [PubMed]
- Steil, L.; Hoffmann, T.; Budde, I.; Völker, U.; Bremer, E. Genome-Wide Transcriptional Profiling Analysis of Adaptation of Bacillus subtilis to High Salinity. J. Bacteriol. 2003, 185, 6358–6370. [Google Scholar] [CrossRef]
- Zhou, A.; Baidoo, E.; He, Z.; Mukhopadhyay, A.; Baumohl, J.K.; Benke, P.; Joachimiak, M.P.; Xie, M.; Song, R.; Arkin, A.P.; et al. Characterization of NaCl Tolerance in Desulfovibrio vulgaris Hildenborough through Experimental Evolution. ISME J. 2013, 7, 1790–1802. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Ferreras, A.; Pérez-Arnedo, R.; Becker, A.; Olivares, J.; Soto, M.J.; Sanjuán, J. Transcriptome Profiling Reveals the Importance of Plasmid PSymB for Osmoadaptation of Sinorhizobium meliloti. J. Bacteriol. 2006, 188, 7617–7625. [Google Scholar] [CrossRef] [PubMed]
- Bahlawane, C.; McIntosh, M.; Krol, E.; Becker, A. Sinorhizobium meliloti Regulator MucR Couples Exopolysaccharide Synthesis and Motility. Mol. Plant Microbe Interact. 2008, 21, 1498–1509. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Gao, W.; Wang, Y.; Wu, L.; Liu, X.; Yan, T.; Alm, E.; Arkin, A.; Thompson, D.K.; Fields, M.W.; et al. Transcriptome Analysis of Shewanella oneidensis MR-1 in Response to Elevated Salt Conditions. J. Bacteriol. 2005, 187, 2501–2507. [Google Scholar] [CrossRef] [PubMed]
- Gibson, K.E.; Barnett, M.J.; Toman, C.J.; Long, S.R.; Walker, G.C. The Symbiosis Regulator CbrA Modulates a Complex Regulatory Network Affecting the Flagellar Apparatus and Cell Envelope Proteins. J. Bacteriol. 2007, 189, 3591–3602. [Google Scholar] [CrossRef]
- Sourjik, V.; Muschler, P.; Scharf, B.; Schmitt, R. VisN and VisR Are Global Regulators of Chemotaxis, Flagellar, and Motility Genes in Sinorhizobium (Rhizobium) meliloti. J. Bacteriol. 2000, 182, 782–788. [Google Scholar] [CrossRef] [PubMed]
- Hoang, H.H.; Gurich, N.; González, J.E. Regulation of Motility by the ExpR/Sin Quorum-Sensing System in Sinorhizobium meliloti. J. Bacteriol. 2008, 190, 861–871. [Google Scholar] [CrossRef]
- Janczarek, M. Environmental Signals and Regulatory Pathways That Influence Exopolysaccharide Production in Rhizobia. Int. J. Mol. Sci. 2011, 12, 7898–7933. [Google Scholar] [CrossRef]
- Domínguez-Ferreras, A.; Soto, M.J.; Pérez-Arnedo, R.; Olivares, J.; Sanjuán, J. Importance of Trehalose Biosynthesis for Sinorhizobium meliloti Osmotolerance and Nodulation of Alfalfa Roots. J. Bacteriol. 2009, 191, 7490–7499. [Google Scholar] [CrossRef]
- Flechard, M.; Fontenelle, C.; Blanco, C.; Goude, R.; Ermel, G.; Trautwetter, A. RpoE2 of Sinorhizobium meliloti Is Necessary for Trehalose Synthesis and Growth in Hyperosmotic Media. Microbiology 2010, 156, 1708–1718. [Google Scholar] [CrossRef]
- Sugawara, M.; Cytryn, E.J.; Sadowsky, M.J. Functional Role of Bradyrhizobium Japonicum Trehalose Biosynthesis and Metabolism Genes during Physiological Stress and Nodulation. Appl. Environ. Microbiol. 2010, 76, 1071–1081. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.B.; Peters, N.K.; Bhuvaneswari, T.V. Redundancy in Periplasmic Binding Protein-Dependent Transport Systems for Trehalose, Sucrose, and Maltose in Sinorhizobium meliloti. J. Bacteriol. 2002, 184, 2978–2986. [Google Scholar] [CrossRef] [PubMed]
- Jensen, J.B.; Ampomah, O.Y.; Darrah, R.; Peters, N.K.; Bhuvaneswari, T.V. Role of Trehalose Transport and Utilization in Sinorhizobium meliloti--Alfalfa Interactions. Mol. Plant Microbe Interact. 2005, 18, 694–702. [Google Scholar] [CrossRef] [PubMed]
- Ampomah, O.Y.; Jensen, J.B.; Bhuvaneswari, T.V. Lack of Trehalose Catabolism in Sinorhizobium Species Increases Their Nodulation Competitiveness on Certain Host Genotypes. New Phytol. 2008, 179, 495–504. [Google Scholar] [CrossRef]
- Geddes, B.A.; Oresnik, I.J. Genetic Characterization of a Complex Locus Necessary for the Transport and Catabolism of Erythritol, Adonitol and l-Arabitol in Sinorhizobium meliloti. Microbiology 2012, 158, 2180–2191. [Google Scholar] [CrossRef] [PubMed]
- Yost, C.K.; Rath, A.M.; Noel, T.C.; Hynes, M.F. Characterization of Genes Involved in Erythritol Catabolism in Rhizobium leguminosarum Bv. viciae. Microbiology 2006, 152, 2061–2074. [Google Scholar] [CrossRef] [PubMed]
- Poysti, N.J.; Oresnik, I.J. Characterization of Sinorhizobium meliloti Triose Phosphate Isomerase Genes. J. Bacteriol. 2007, 189, 3445–3451. [Google Scholar] [CrossRef]
- Geddes, B.A.; Pickering, B.S.; Poysti, N.J.; Collins, H.; Yudistira, H.; Oresnik, I.J. A Locus Necessary for the Transport and Catabolism of Erythritol in Sinorhizobium meliloti. Microbiology 2010, 156, 2970–2981. [Google Scholar] [CrossRef]
- Kohler, P.R.A.; Choong, E.-L.; Rossbach, S. The RpiR-Like Repressor IolR Regulates Inositol Catabolism in Sinorhizobium meliloti. J. Bacteriol. 2011, 193, 5155–5163. [Google Scholar] [CrossRef]
- Herrou, J.; Crosson, S. Myo -Inositol and d-Ribose Ligand Discrimination in an ABC Periplasmic Binding Protein. J. Bacteriol. 2013, 195, 2379–2388. [Google Scholar] [CrossRef] [PubMed]
- Kohler, P.R.A.; Zheng, J.Y.; Schoffers, E.; Rossbach, S. Inositol Catabolism, a Key Pathway in Sinorhizobium meliloti for Competitive Host Nodulation. Appl. Environ. Microbiol. 2010, 76, 7972–7980. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Krishnan, A.H.; Kim, Y.-W.; Wacek, T.J.; Krishnan, H.B. A Functional Myo -Inositol Dehydrogenase Gene Is Required for Efficient Nitrogen Fixation and Competitiveness of Sinorhizobium fredii USDA191 To Nodulate Soybean (Glycine max [L.] Merr.). J. Bacteriol. 2001, 183, 2595–2604. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fry, J.; Wood, M.; Poole, P.S. Investigation of Myo -Inositol Catabolism in Rhizobium leguminosarum Bv. viciae and Its Effect on Nodulation Competitiveness. Mol. Plant Microbe Interact. 2001, 14, 1016–1025. [Google Scholar] [CrossRef] [PubMed]
- Gourion, B.; Berrabah, F.; Ratet, P.; Stacey, G. Rhizobium–Legume Symbioses: The Crucial Role of Plant Immunity. Trends Plant Sci. 2015, 20, 186–194. [Google Scholar] [CrossRef]
- Rodrigo da-Silva, J.; Alexandre, A.; Brígido, C.; Oliveira, S. Can Stress Response Genes Be Used to Improve the Symbiotic Performance of Rhizobia? AIMS Microbiol. 2017, 3, 365–382. [Google Scholar] [CrossRef]
- Patankar, A.V.; González, J.E. An Orphan LuxR Homolog of Sinorhizobium meliloti Affects Stress Adaptation and Competition for Nodulation. Appl. Environ. Microbiol. 2009, 75, 946–955. [Google Scholar] [CrossRef]
- Novichkov, P.S.; Li, X.; Kuehl, J.V.; Deutschbauer, A.M.; Arkin, A.P.; Price, M.N.; Rodionov, D.A. Control of Methionine Metabolism by the SahR Transcriptional Regulator in Proteobacteria: Control of Methionine Metabolism in Proteobacteria. Environ. Microbiol. 2014, 16, 1–8. [Google Scholar] [CrossRef]
- Jiang, J.Q.; Wei, W.; Du, B.H.; Li, X.H.; Wang, L.; Yang, S.S. Salt-Tolerance Genes Involved in Cation Efflux and Osmoregulation of Sinorhizobium fredii RT19 Detected by Isolation and Characterization of Tn5 Mutants. FEMS Microbiol. Lett. 2004, 239, 139–146. [Google Scholar] [CrossRef][Green Version]
- Taté, R.; Riccio, A.; Caputo, E.; laccarino, M.; Patriarca, E.J. The Rhizobium etli metZ Gene Is Essential for Methionine Biosynthesis and Nodulation of Phaseolus vulgaris. Mol. Plant Microbe Interact. 1999, 12, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Francez-Charlot, A.; Kaczmarczyk, A.; Fischer, H.-M.; Vorholt, J.A. The General Stress Response in Alphaproteobacteria. Trends Microbiol. 2015, 23, 164–171. [Google Scholar] [CrossRef]
- Gourion, B.; Sulser, S.; Frunzke, J.; Francez-Charlot, A.; Stiefel, P.; Pessi, G.; Vorholt, J.A.; Fischer, H.-M. The PhyR-σ EcfG Signalling Cascade Is Involved in Stress Response and Symbiotic Efficiency in Bradyrhizobium japonicum. Mol. Microbiol. 2009, 73, 291–305. [Google Scholar] [CrossRef]
- Ledermann, R.; Bartsch, I.; Müller, B.; Wülser, J.; Fischer, H.-M. A Functional General Stress Response of Bradyrhizobium diazoefficiens Is Required for Early Stages of Host Plant Infection. Mol. Plant Microbe Interact. 2018, 31, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Bastiat, B.; Sauviac, L.; Bruand, C. Dual Control of Sinorhizobium meliloti RpoE2 Sigma Factor Activity by Two PhyR-Type Two-Component Response Regulators. J. Bacteriol. 2010, 192, 2255–2265. [Google Scholar] [CrossRef] [PubMed]
- Sauviac, L.; Philippe, H.; Phok, K.; Bruand, C. An Extracytoplasmic Function Sigma Factor Acts as a General Stress Response Regulator in Sinorhizobium meliloti. J. Bacteriol. 2007, 189, 4204–4216. [Google Scholar] [CrossRef]
- Jans, A.; Vercruysse, M.; Gao, S.; Engelen, K.; Lambrichts, I.; Fauvart, M.; Michiels, J. Canonical and Non-Canonical EcfG Sigma Factors Control the General Stress Response in Rhizobium etli. MicrobiologyOpen 2013, 2, 976–987. [Google Scholar] [CrossRef]
- Foreman, R.; Fiebig, A.; Crosson, S. The LovK-LovR Two-Component System Is a Regulator of the General Stress Pathway in Caulobacter crescentus. J. Bacteriol. 2012, 194, 3038–3049. [Google Scholar] [CrossRef]
- Lang, C.; Barnett, M.J.; Fisher, R.F.; Smith, L.S.; Diodati, M.E.; Long, S.R. Most Sinorhizobium meliloti Extracytoplasmic Function Sigma Factors Control Accessory Functions. mSphere 2018, 3, e00454-18. [Google Scholar] [CrossRef] [PubMed]
- Flechard, M.; Fontenelle, C.; Trautwetter, A.; Ermel, G.; Blanco, C. Sinorhizobium meliloti rpoE2 Is Necessary for H2O2 Stress Resistance during the Stationary Growth Phase. FEMS Microbiol. Lett. 2009, 290, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Ledermann, R.; Emmenegger, B.; Couzigou, J.-M.; Zamboni, N.; Kiefer, P.; Vorholt, J.A.; Fischer, H.-M. Bradyrhizobium diazoefficiens Requires Chemical Chaperones To Cope with Osmotic Stress during Soybean Infection. mBio 2021, 12, e00390-21. [Google Scholar] [CrossRef] [PubMed]
- Ravcheev, D.A.; Khoroshkin, M.S.; Laikova, O.N.; Tsoy, O.V.; Sernova, N.V.; Petrova, S.A.; Rakhmaninova, A.B.; Novichkov, P.S.; Gelfand, M.S.; Rodionov, D.A. Comparative Genomics and Evolution of Regulons of the LacI-Family Transcription Factors. Front. Microbiol. 2014, 5, 294. [Google Scholar] [CrossRef]

| Strains Plasmids Primers | Description/Primer Sequence | Source/Reference |
|---|---|---|
| Strains | ||
| Escherichia coli strains | ||
| XL2 Blue Ultra-competent Cells | endA1 supE44 thi-1 hsdR17 recA1 gyrA96 relA1 lac [F’ proAB lacIqZΔM15 Tn10 (Tetr) Amy Camr] | Stratagene |
| S17-1 | recA [SmR], thi, pro, RP4-2-Tc:Mu: aph::Tn7λpir. | [27] |
| Ensifer aridi strains | ||
| LMR001T | Ensifer aridi LMR001T (=LMG 31426T; =HAMBI 3707T) | [1,2] |
| LMR001 ∆rpoE2 | rpoE2 deletion mutant of Ensifer aridi LMR001T | This work |
| Plasmids | ||
| pGEM-T Easy | AmpR, pUC origin, Multi Cloning Sites), lacZ gene fusion, β-lactamase coding region, lac operon sequences. | Promega |
| TOPO vector | KmR, AmpR, pUC origin, lacZ α-ccdB gene fusion. Topoisomerase enzyme | Invitrogen |
| pPROBE NT (pNT) | KmR, pBBR1 replicon, Multi Cloning Sites upstream of gfp reporter gene | [28] |
| pJQ200SK+ | GmR, SacB; Ori origin replication, mob region, Multi Cloning Sites | [26] |
| pNT-sahRp | pPROBE NT containing sahR (PEG2239) promoter fused to gfp gene, KmR | This work |
| pNT-nesRp | pPROBE NT containing nesR (PEG5735) promoter fused to gfp gene, KmR | This work |
| pNT-rsiA1p | pPROBE NT containing rsiA1 (PEG2540) promoter fused to gfp gene, KmR | This work |
| pNT-rsiB1p | pPROBE NT containing rsiB1 (PEG2541) promoter fused to gfp gene, KmR | This work |
| pNT-thuEp | pPROBE NT containing thuE (PEG6268) promoter fused to gfp gene, KmR | This work |
| pNT-otsBp | pPROBE NT containing otsB (PEG4868) promoter fused to gfp gene, KmR | This work |
| pNT-treZp | pPROBE NT containing treZ (PEG5323) promoter fused to gfp gene, KmR | This work |
| pNT-PEG4866p | pPROBE NT containing a glycosyl transferase encoding gene (PEG4866) promoter fused to gfp gene, KmR | This work |
| pNT-thuRp | pPROBE NT containing thuR (PEG6269) promoter fused to gfp gene, KmR | This work |
| pNT-iolCp | pPROBE NT containing iolC (PEG373) promoter fused to gfp gene, KmR | This work |
| pNT-iolRp | pPROBE NT containing iolR (PEG374) promoter fused to gfp gene, KmR | This work |
| Primers | ||
| Targeted gene primers for qPCR | ||
| iolB (PEG370) Forward | 5′-CGGGCACACGTCCTCCTATC-3′ | This work |
| iolB (PEG370) Reverse | 5′-CCCTTGGGTACCAGCGTGAC-3′ | This work |
| flgB (PEG666) Forward | 5′-CCCAGGTGACCGAGGTGAGT-3′ | This work |
| flgB (PEG666) Reverse | 5′-CCCCGCATTGAGCTCGTAGT-3′ | This work |
| hrcA (PEG397) Forward | 5′-ACCAAGGGGATCGCATCGAC-3′ | This work |
| hrcA (PEG397) Reverse | 5′-GGCTCGCCGCTTTCCAGATA-3′ | This work |
| metK (PEG438) Forward | 5′-CCCAGCCGCTGTCGATCTAT-3′ | This work |
| metK (PEG438) Reverse | 5′-GGCGAGAGGTCCATCGTCTT-3′ | This work |
| cheA (PEG646) Forward | 5′-TCCGCTCCGTTTTCGAGTTC-3′ | This work |
| cheA (PEG646) Reverse | 5′-GCAACGGTCCGATCCTCTTC-3′ | This work |
| gltA (PEG1564) Forward | 5′-ATTGCGCTGACGGACGAGTAC-3′ | This work |
| gltA (PEG1564) Reverse | 5′-GCGTGATGCCGGAATAGAAG-3′ | This work |
| ahcY (PEG3556) Forward | 5′-AAGGGCAACCGCATCATTC-3′ | This work |
| ahcY (PEG3556) Reverse | 5′-GCTTCGGCAGCACGTAGAC-3′ | This work |
| metH (PEG3206) Forward | 5′-TGGGAGCTGAAGGGCGTCTA-3′ | This work |
| metH (PEG3206) Reverse | 5′-TCGGTGAAAAGGCGGATGTC-3′ | This work |
| bhMT (PEG1969) Forward | 5′-GCGGGTCCGAGATCCACTATT-3′ | This work |
| bhMT (PEG1969) Reverse | 5′-CGCAGCAGCCTCCGATGAT-3′ | This work |
| sahR (PEG2239) Forward | 5′-ATTTCCGCCTGCGTCAAGAG-3′ | This work |
| sahR (PEG2239) Reverse | 5′-CGGCGTTGCGACTGAAATAG-3′ | This work |
| L11MT (PEG2189) Forward | 5′-ACAAGGTGAAGGCGGGTGAG-3′ | This work |
| L11MT (PEG2189) Reverse | 5′-GAGGTCGAGGACGTTGCAGAA-3′ | This work |
| xylF (PEG2505) Forward | 5′-GCTCGGCACCCAGACTGTTT-3′ | This work |
| xylF (PEG2505) Reverse | 5′-GGTCTGCACGCCTTCGATCT-3′ | This work |
| thuE (PEG6268) Forward | 5′-GCTCCGGCGCTCTACTATCG-3′ | This work |
| thuE (PEG6268) Reverse | 5′-GGCGTTTGCCTGGAAGACA-3′ | This work |
| Primers for rpoE2 deletion mutant construction | ||
| RPOE2-P2539-A-XI | 5′-CCTCTAGACATCGCCTGAGGTCTGAGAT-3′ | This work |
| RPOE2-P2539-B-HIII | 5′-CGCCCTTCACTTGAAGCTTGCATCTCACGCTTGAACTCT-3′ | This work |
| RPOE2-P2539-C-HIII | 5′-AAGCTTCAAGTGAAGGGCGAGAAC-3′ | This work |
| RPOE2-P2539-D-XI | 5′-CCTCTAGATTGACCCATTGCCATTCACG-3′ | This work |
| Targeted gene promoter/Primers for promoter::gfp fusion cloning | ||
| iolRp Forward/iolR-1 | 5′-GACTTCCGCGAGGGCTAC-3′ | This work |
| iolRp Reverse/iolR-B | 5′-TCGGCCTCGACGGAGGGACATCGGTGGTCG-3′ | This work |
| iolCp Forward/iolC-1 | 5′-CTCGCTTTCGGCAAGC-3′ | This work |
| iolCp Reverse/iolC-B | 5′-GGCGACGGCTTGGCCGATCGTGATGATGTC-3′ | This work |
| sahRp Forward/metReg-1 | 5′-CAATCCGAAGCGGGTG-3′ | This work |
| sahRp Reverse/metReg-B | 5′-GTTTTCCTTCCTGGCGTTCCTCTGTCACCC-3′ | This work |
| nesRp Forward/nesR-1 | 5′-GGTTTCCGGGCCTGCAC-3′ | This work |
| nesRp Reverse/nesR-B | 5′-TTAAGCCGCGGCGGTGAACTGGTTCCTGATC-3′ | This work |
| rsiA1p Forward/rsiA1-1 | 5′-GAAGGTAAGGAAGGAATG-3′ | This work |
| rsiA1p Reverse/rsiA1-B | 5′-GAATCGATTGTCCCGCAATCTGTACATTCG-3′ | This work |
| risB1p Forward/rsiA1-B | 5′-GAATCGATTGTCCCGCAATCTGTACATTCG-3′ | This work |
| risB1p Reverse/rsiA1-1 | 5′-GAAGGTAAGGAAGGAATG-3′ | This work |
| otsBp Forward/peg4866-B | 5′-AGCTTATCGAGAGCAATTTGCGCGATCTTC-3′ | This work |
| otsBp Reverse/peg4866-1 | 5′-CTCGATCATGGAGAGG-3′ | This work |
| PEG4866p Forward/peg4866-1 | 5′-CTCGATCATGGAGAGG-3′ | This work |
| PEG4866p Reverse/peg4866-B | 5′-AGCTTATCGAGAGCAATTTGCGCGATCTTC-3′ | This work |
| treZp Forward/treZ-1 | 5′-GAGCTCGGGTTTGATG-3′ | This work |
| treZp Reverse/treZ-B | 5′-CGAAAGGTTGACCGTCGCTGTCTCTCGCAT-3′ | This work |
| thuRp Forward/thuR-1 | 5′-CTCGAAGGCTCCTCAGC-3′ | This work |
| thuRp Reverse/thuR-B | 5′-AGCAATTGCAGCGCGAACTCCTTGAGCTTC-3′ | This work |
| thuEp Forward/thuR-B | 5′-AGCAATTGCAGCGCGAACTCCTTGAGCTTC-3′ | This work |
| thuEp Reverse/thuR-1 | 5′-CTCGAAGGCTCCTCAGC-3′ | This work |
| gfp Reverse/gfp-Rev50 | 5′-ACATCACCATCTAATTCAAC-3′ | This work |
| Shoot Weight (g) | Root Weight (g) | Nodule Number | ||||
|---|---|---|---|---|---|---|
| Plant/Treatment | Mean | Standard Deviation | Mean | Standard Deviation | Mean | Standard Deviation |
| Vachellia gummifera | ||||||
| LMR001 WT | 2.27 | 0.21 | 0.90 | 0.09 | 33 | 3.22 |
| LMR001 ∆rpoE2 | 2.33 | 0.37 | 1.03 | 0.19 | 35.67 | 5.24 |
| Positive Control | 3.13 | 0.10 | 2.83 | 0.26 | 0 | |
| Negative Control | 1.37 | 0.14 | 1.07 | 0.10 | 0 | |
| Vachellia tortilis | ||||||
| LMR001 WT | 2.72 | 0.09 | 0.83 | 0.05 | 39 | 1.79 |
| LMR001 ∆rpoE2 | 2.16 | 0.21 | 1.08 | 0.15 | 38 | 3.94 |
| Positive Control | 3.73 | 0.58 | 2.33 | 0.26 | 0 | |
| Negative Control | 1.83 | 0.14 | 1.10 | 0.09 | 0 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belfquih, M.; Sakrouhi, I.; Ait-Benhassou, H.; Dubois, E.; Severac, D.; Filali-Maltouf, A.; Le Quere, A. Ensifer aridi LMR001T Symbiosis and Tolerance to Stress Do Not Require the Alternative Sigma Factor RpoE2. Agronomy 2021, 11, 1787. https://doi.org/10.3390/agronomy11091787
Belfquih M, Sakrouhi I, Ait-Benhassou H, Dubois E, Severac D, Filali-Maltouf A, Le Quere A. Ensifer aridi LMR001T Symbiosis and Tolerance to Stress Do Not Require the Alternative Sigma Factor RpoE2. Agronomy. 2021; 11(9):1787. https://doi.org/10.3390/agronomy11091787
Chicago/Turabian StyleBelfquih, Meryem, Ilham Sakrouhi, Hassan Ait-Benhassou, Emeric Dubois, Dany Severac, Abdelkarim Filali-Maltouf, and Antoine Le Quere. 2021. "Ensifer aridi LMR001T Symbiosis and Tolerance to Stress Do Not Require the Alternative Sigma Factor RpoE2" Agronomy 11, no. 9: 1787. https://doi.org/10.3390/agronomy11091787
APA StyleBelfquih, M., Sakrouhi, I., Ait-Benhassou, H., Dubois, E., Severac, D., Filali-Maltouf, A., & Le Quere, A. (2021). Ensifer aridi LMR001T Symbiosis and Tolerance to Stress Do Not Require the Alternative Sigma Factor RpoE2. Agronomy, 11(9), 1787. https://doi.org/10.3390/agronomy11091787







