Effects of Growing Cycle and Genotype on the Morphometric Properties and Glucosinolates Amount and Profile of Sprouts, Microgreens and Baby Leaves of Broccoli (Brassica oleracea L. var. italica Plenck) and Kale (B. oleracea L. var. acephala DC.)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and Experimental Conditions
2.2. Morphometric Parameters
2.3. Glucosinolate Analyses
2.3.1. HPLC-DAD Analyses
2.3.2. HPLC-DAD-ESI-MSn Analysis of GLSs
2.4. Statistical Analysis
3. Results
3.1. Plant Characteristics
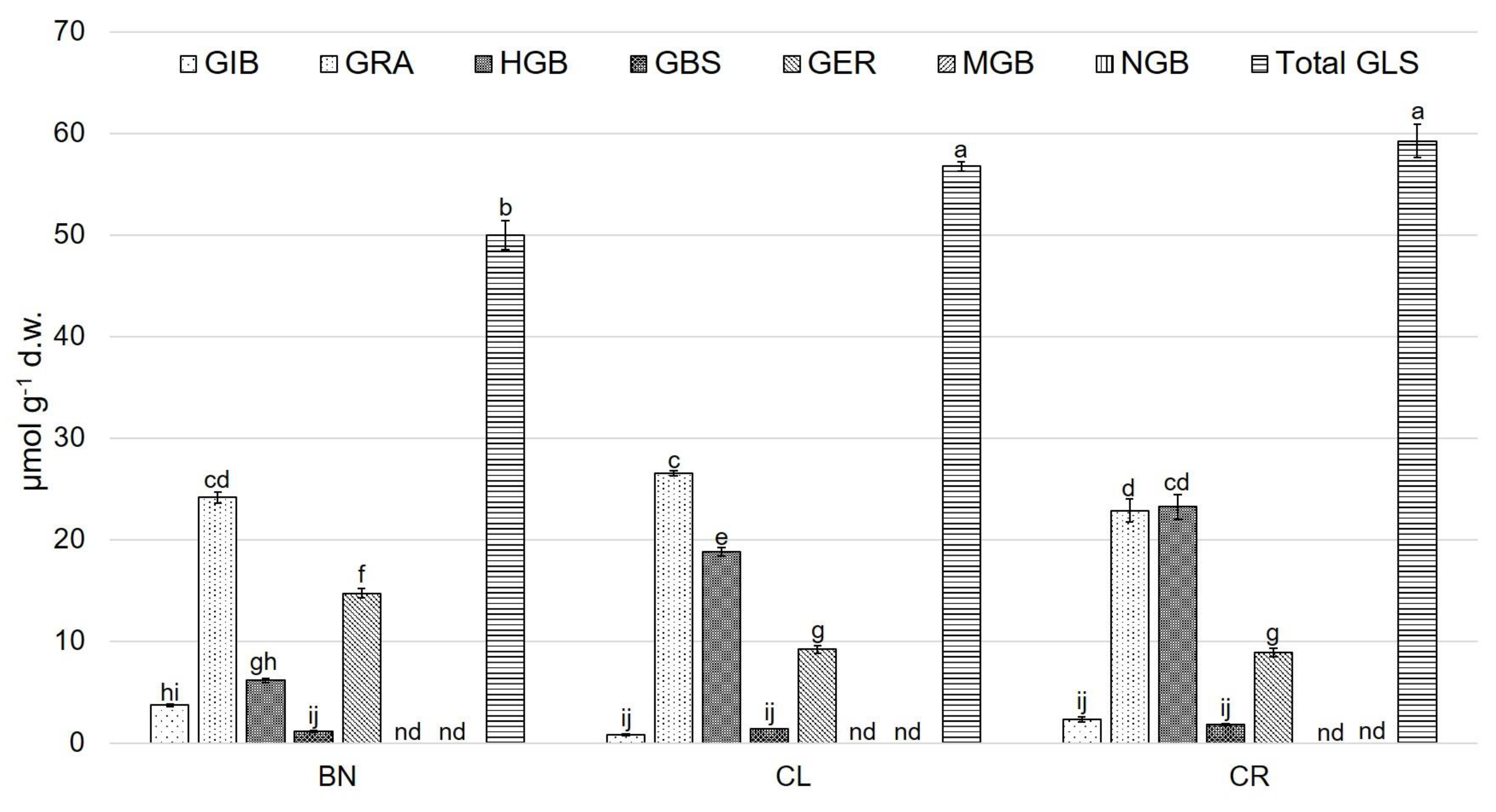
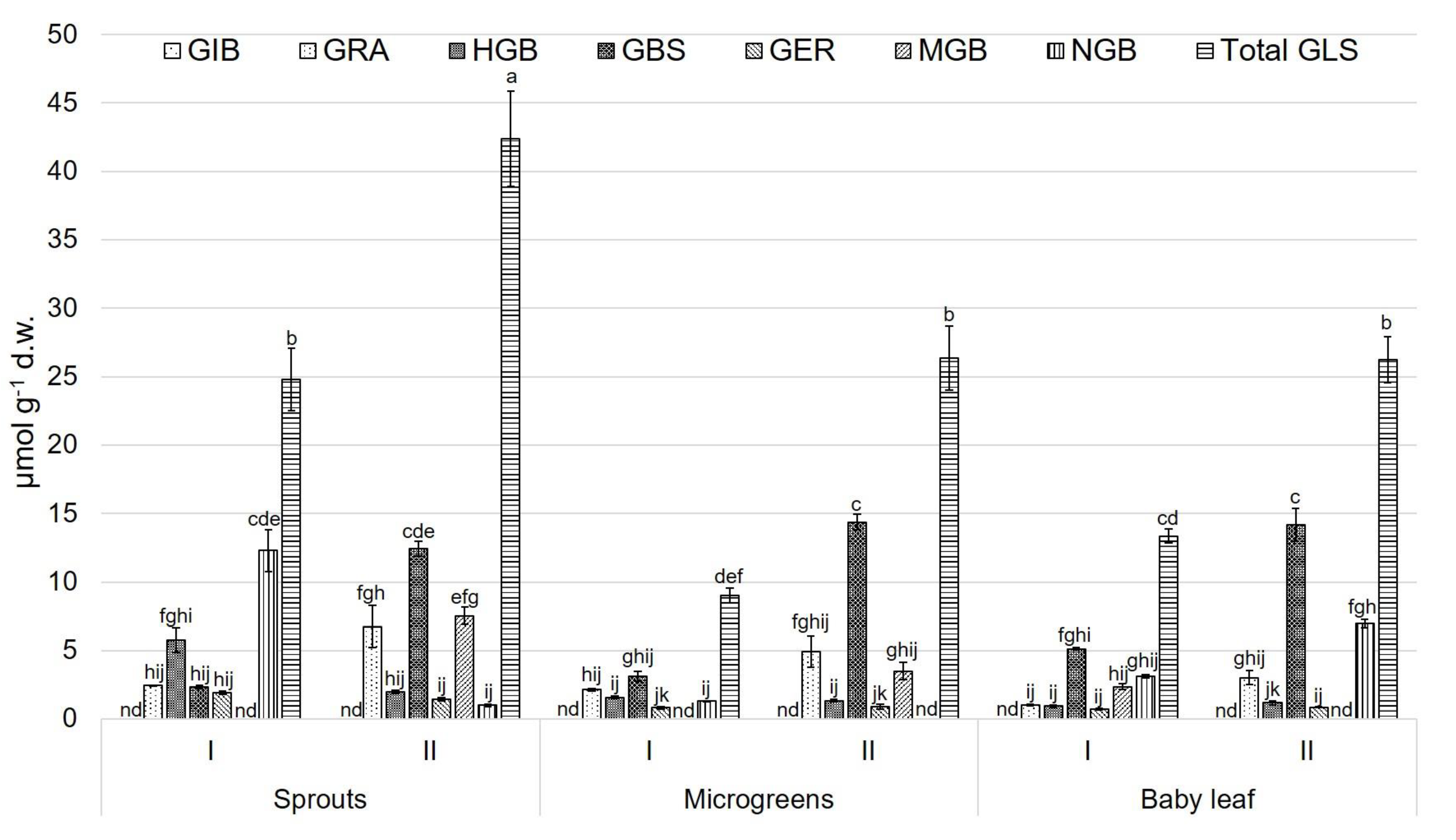
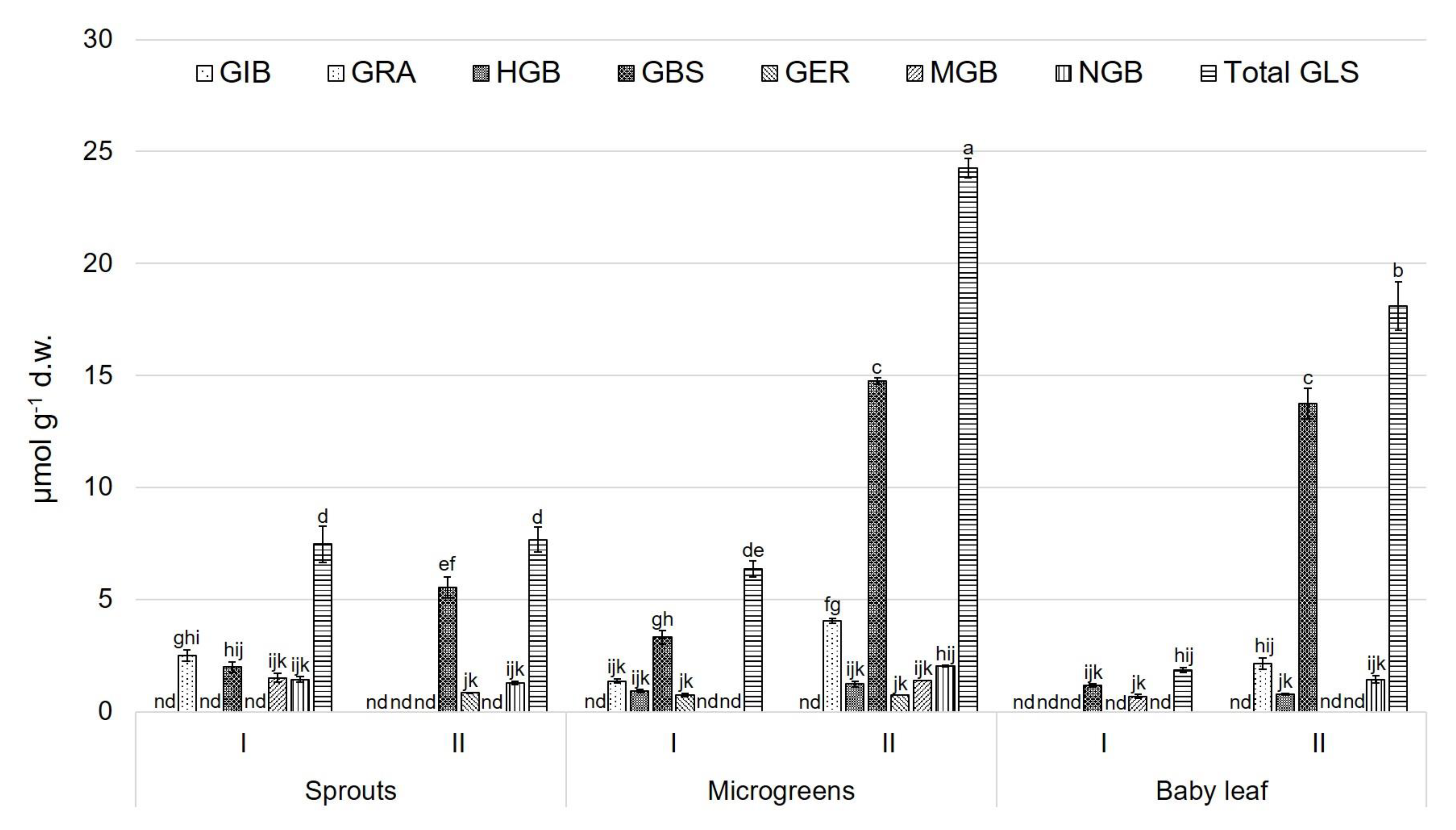
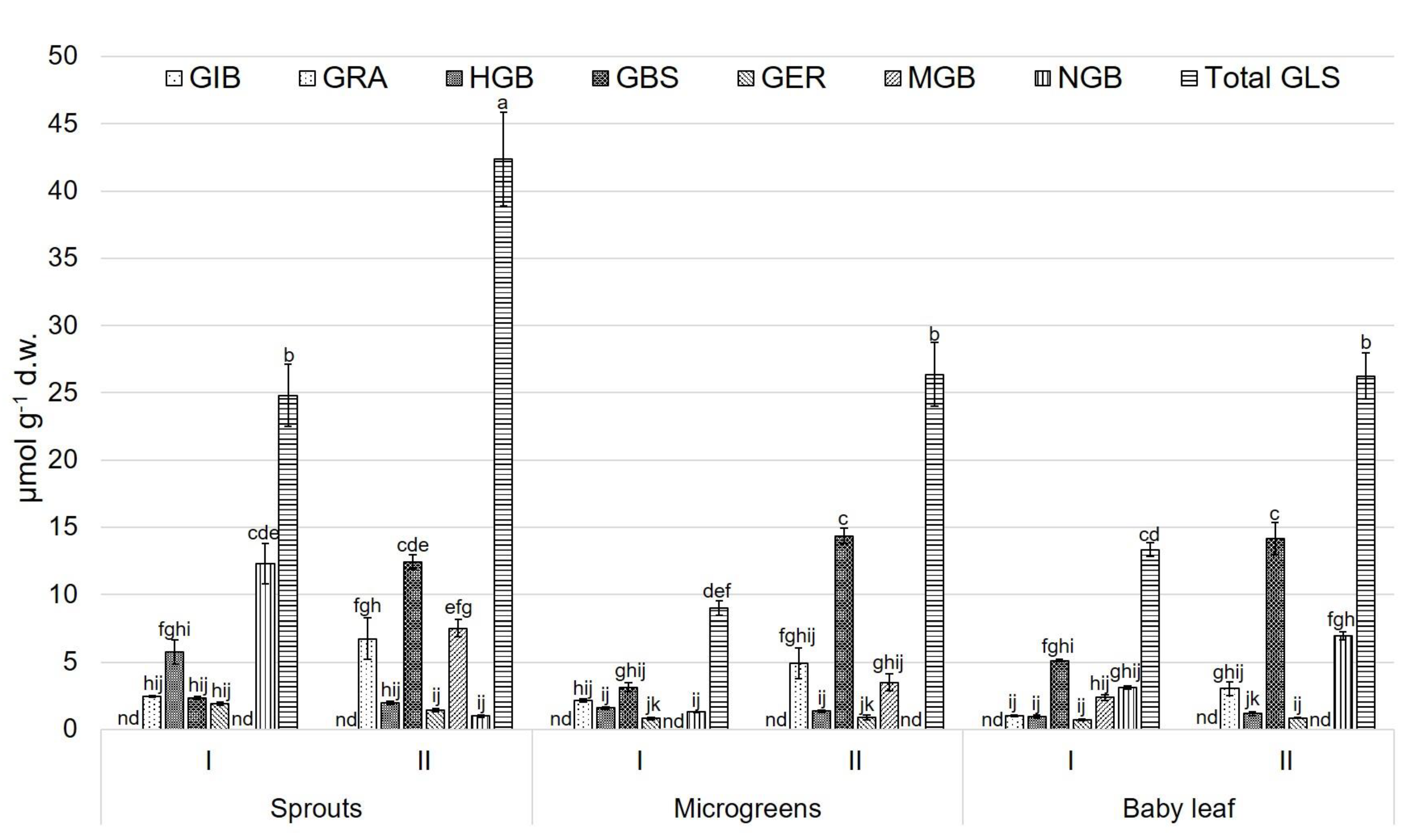
3.2. Glucosinolate (GLS) Profile
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Saini, P.; Kumar, N.; Kumar, S.; Mwaurah, P.W.; Panghal, A.; Attkan, A.K.; Singh, V.K.; Garg, M.K.; Singh, V. Bioactive compounds, nutritional benefits and food applications of colored wheat: A comprehensive review. Crit. Rev. Food Sci. Nutr. 2020, 1–14. [Google Scholar] [CrossRef]
- Di Gioia, F.; Renna, M.; Santamaria, P. Sprouts, Microgreens and Baby Leaf Vegetables. In Minimally Processed Refrigerated Fruits and Vegetables; Yildiz, F., Wiley, R., Eds.; Springer: Boston, MA, USA, 2017; pp. 403–432. [Google Scholar] [CrossRef]
- Renna, M.; Di Gioia, F.; Leoni, B.; Santamaria, P. Due Espressioni dell’agrobiodiversità in orticoltura: Germogli e micro-ortaggi. Italus Hortus 2016, 23, 31–44. [Google Scholar]
- Abellán, Á.; Domínguez-Perles, R.; Moreno, D.A.; García-Viguera, C. Sorting out the value of cruciferous sprouts as sources of bioactive compounds for nutrition and health. Nutrients 2019, 11, 429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le, T.N.; Chiu, C.-H.; Hsieh, P.-C. Bioactive Compounds and Bioactivities of Brassica oleracea L. var italica Sprouts and Microgreens: An Updated Overview from a Nutraceutical Perspective. Plants 2020, 9, 946. [Google Scholar] [CrossRef]
- Ragusa, L.; Picchi, V.; Tribulato, A.; Cavallaro, C.; Lo Scalzo, R.; Branca, F. The effect of the germination temperature on the phytochemical content of broccoli and rocket sprouts. Int. J. Food Sci. Nutr. 2017, 68, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Di Bella, M.C.; Niklas, A.; Toscano, S.; Picchi, V.; Romano, D.; Lo Scalzo, R.; Branca, F. Morphometric characteristics, polyphenols and ascorbic acid variation in Brassica oleracea L. novel foods: Sprouts, microgreens and baby leaves. Agronomy 2020, 10, 782. [Google Scholar] [CrossRef]
- Almuhayawi, M.S.; AbdElgawad, H.; Al Jaouni, S.K.; Selim, S.; Hassan, A.H.A.; Khamis, G. Elevated CO2 improves glucosinolate metabolism and stimulates anticancer and anti-inflammatory properties of broccoli sprouts. Food Chem. 2020, 328, 127102. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Hirami, A.H.; Li, Z.; Wu, C.; McVetty, P.B.E.; Daayf, F.; Li, G. QTL controlling glucosinolate content in seeds of Brassica napus L. Aust. J. Crop Sci. 2016, 10, 152–160. [Google Scholar]
- Martínez-Sánchez, A.; Luna, M.C.; Selma, M.V.; Tudela, J.A.; Abad, J.; Gil, M.I. Baby-leaf and multi-leaf of green and red lettuces are suitable raw materials for the fresh-cut industry. Postharvest Biol. Technol. 2012, 63, 1–10. [Google Scholar] [CrossRef]
- Warwick, S.I. Brassicaceae in Agriculture. In Genetics and Genomics of the Brassicaceae; Warwick, S.I., Ed.; Springer: New York, NY, USA, 2010; pp. 33–65. [Google Scholar]
- Mir, S.A.; Shah, M.A.; Mir, M.M. Microgreens: Production, shelf life, and bioactive components. Crit. Rev. Food Sci. Nutr. 2017, 57, 2730–2736. [Google Scholar] [CrossRef]
- Schmidt, R.; Bancroft, I. Genetics and Genomics of the Brassicaceae, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Weber, C.F. Broccoli microgreens: A mineral-rich crop that can diversify food systems. Front. Nutr. 2017, 4, 7. [Google Scholar] [CrossRef] [PubMed]
- Ares, A.M.; Nozal, M.J.; Bernal, J. Extraction, chemical characterization, and biological activity determination of broccoli health promoting compounds. J. Chromatogr. A 2013, 1313, 78–95. [Google Scholar] [CrossRef] [PubMed]
- Ying, Q.; Kong, Y.; Jones-Baumgardt, C.; Zheng, Y. Responses of yield and appearance quality of four Brassicaceae microgreens to varied blue light proportion in red and blue light-emitting diodes lighting. Sci. Hortic. 2020, 259, 108857. [Google Scholar] [CrossRef]
- Abdull Razis, A.F.; Iori, R.; Ioannides, C. The natural chemopreventive phytochemical R-sulforaphane is a far more potent inducer of the carcinogen-detoxifying enzyme systems in rat liver and lung than the S-isomer. Int. J. Cancer 2011, 128, 2775–2782. [Google Scholar] [CrossRef]
- De Oliveira, M.R.; de Bittencourt Brasil, F.; Furstenau, C.R. Sulforaphane Promotes Mitochondrial protection in SH-SY5Y cells exposed to hydrogen peroxide by an Nrf2-dependent mechanism. Mol. Neurobiol. 2018, 55, 4777–4787. [Google Scholar] [CrossRef]
- Halkier, B.A.; Gershenzon, J. Biology and biochemistry of glucosinolates. Annu. Rev. Plant Biol. 2006, 57, 303–333. [Google Scholar] [CrossRef] [Green Version]
- Petropoulos, S.; Di Gioia, F.; Ntatsi, G. Vegetable organosulfur compounds and their health promoting effects. Curr. Pharm. Des. 2017, 23, 2850–2875. [Google Scholar] [CrossRef] [PubMed]
- Tian, Q.; Rosselot, R.A.; Schwartz, S.J. Quantitative determination of intact glucosinolates in broccoli, broccoli sprouts, Brussels sprouts, and cauliflower by high-performance liquid chromatography–electrospray ionization–tandem mass spectrometry. Anal. Biochem. 2005, 343, 93–99. [Google Scholar] [CrossRef]
- Glawischnig, E.; Mikkelsen, M.D.; Halkier, B.A. Glucosinolates: Biosynthesis and Metabolism. In Sulphur in Plants; Abrol, Y.P., Ahmad, A., Eds.; Springer: Dordrecht, The Netherlands, 2003; pp. 145–162. [Google Scholar]
- Velasco, P.; Cartea, M.E.; González, C.; Vilar, M.; Ordás, A. Factors affecting the glucosinolate content of kale (Brassica oleracea acephala group). J. Agric. Food Chem. 2007, 55, 955–962. [Google Scholar] [CrossRef]
- Yuan, G.; Wang, X.; Guo, R.; Wang, Q. Effect of salt stress on phenolic compounds, glucosinolates, myrosinase and antioxidant activity in radish sprouts. Food Chem. 2010, 121, 1014–1019. [Google Scholar] [CrossRef]
- Stansell, Z.; Björkman, T. From landrace to modern hybrid broccoli: The genomic and morphological domestication syndrome within a diverse B. oleracea collection. Hortic. Res. 2020, 7, 159. [Google Scholar] [CrossRef] [PubMed]
- Cartea, M.E.; Di Bella, M.C.; Velasco, P.; Soengas, P.; Toscano, S.; Branca, F. Evaluation of Italian and Spanish Accessions of Brassica rapa L.: Effect of flowering earliness on fresh yield and biological value. Agronomy 2021, 11, 29. [Google Scholar] [CrossRef]
- Davis, D.R.; Epp, M.D.; Riordan, H.D. Changes in USDA food composition data for 43 garden crops, 1950 to 1999. J. Am. Coll. Nutr. 2004, 23, 669–682. [Google Scholar] [CrossRef]
- Ebert, A.W. High value specialty vegetable produce. In Handbook of Vegetables; Peter, K.V., Hazra, P., Eds.; Studium Press LLC: Houston, TX, USA, 2015; pp. 119–143. [Google Scholar]
- Ciancaleoni, S.; Negri, V. A method for obtaining flexible broccoli varieties for sustainable agriculture. BMC Genet. 2020, 21, 51. [Google Scholar] [CrossRef] [PubMed]
- Terzo, M.N.; Russo, A.; Ficili, B.; Pezzino, F.M.; Tribulato, A.; Russello, D.; Branca, F.; Travali, S. Neglected Sicilian landraces of black broccoli (Brassica oleracea var. italica Plenck) and health benefits: An in vivo study. Acta Hortic. 2020, 1267, 91–96. [Google Scholar] [CrossRef]
- Baenas, M.; Moreno, A.D.; Garcia-Viguera, C. Selecting Sprouts of Brassicaceae for Optimum Phytochemical Composition. Agric. Food Chem. 2012, 60, 45–11409. [Google Scholar] [CrossRef]
- Avato, P.; Argentieri, M.P. Brassicaceae: A rich source of health improving phytochemicals. Phytochem. Rev. 2015, 14, 1019–1033. [Google Scholar] [CrossRef]
- Cáceres, P.J.; Martínez-Villaluenga, C.; Amigo, L.; Frias, J. Maximising the phytochemical content and antioxidant activity of Ecuadorian brown rice sprouts through optimal germination conditions. Food Chem. 2014, 152, 407–414. [Google Scholar] [CrossRef] [Green Version]
- Lafarga, T.; Bobo, G.; Viñas, I.; Collazo, C.; Aguiló-Aguayo, I. Effects of thermal and non-thermal processing of cruciferous vegetables on glucosinolates and its derived forms. J. Food Sci. Technol. 2018, 55, 1973–1981. [Google Scholar] [CrossRef]
- Mewis, I.; Schreiner, M.; Nguyen, C.N.; Krumbein, A.; Ulrichs, C.; Lohse, M.; Zrenner, R. UV-B irradiation changes specifically the secondary metabolite profile in broccoli sprouts: Induced signaling overlaps with defense response to biotic stressors. Plant Cell Physiol. 2012, 53, 1546–1560. [Google Scholar] [CrossRef] [Green Version]
- Drozdowska, M.; Leszczyńska, T.; Koronowicz, A.; Piasna-Słupecka, E.; Domagała, D.; Kusznierewicz, B. Young shoots of red cabbage are a better source of selected nutrients and glucosinolates in comparison to the vegetable at full maturity. Eur. Food Res. Technol. 2020, 246, 2505–2515. [Google Scholar] [CrossRef]
- Guijarro-Real, C.; Rodríguez-Burruezo, A.; Fita, A. Volatile Profile of Wall Rocket Baby-Leaves (Diplotaxis erucoides) Grown under Greenhouse: Main Compounds and Genotype Diversity. Agronomy 2020, 10, 802. [Google Scholar] [CrossRef]
- Sasaki, K.; Neyazaki, M.; Shindo, K.; Ogawa, T.; Momose, M. Quantitative profiling of glucosinolates by LC–MS analysis reveals several cultivars of cabbage and kale as promising sources of sulforaphane. J. Chromatogr. B Biomed. Appl. 2012, 903, 171–176. [Google Scholar] [CrossRef]
- Li, H.; Qiang, Y.; Zhao, W.; Zhang, S. A green Brassica oleracea Lextract as a novel corrosion inhibitor for Q235 steel in two typical acid media. Colloids Surf. A Physicochem. Eng. Asp. 2021, 616, 126077. [Google Scholar] [CrossRef]
- Paulsen, E.; Moreno, D.A.; Baenas, N.; Heinzen, H. Effect of temperature on glucosinolate content and shelf life of ready-to-eat broccoli florets packaged in passive modified atmosphere. Postharvest Biol. Tec. 2018, 138, 125–133. [Google Scholar] [CrossRef]
- Bell, L.; Lignou, S.; Wagstaff, C. High Glucosinolate Content in Rocket Leaves (Diplotaxis tenuifolia and Eruca sativa) after Mul-tiple Harvests Is Associated with Increased Bitterness, Pungency, and Reduced Consumer. Foods 2020, 9, 1799. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.-Q.; Chen, X.-Q.; Wang, K.-H.; Zhu, Z.-J.; Yang, J.; Zhu, B. Effect of short-term high temperature on the accumulation of glucosinolates in Brassica rapa. Plant Physiol. Biochem. 2021, 161, 222–233. [Google Scholar] [CrossRef] [PubMed]
- Schonhof, I.; Kläring, H.P.; Krumbein, A.; Claußen, W.; Schreiner, M. Effect of temperature increase under low radiation conditions on phytochemicals and ascorbic acid in greenhouse grown broccoli. Agric. Ecosyst. Environ. 2007, 119, 103–111. [Google Scholar] [CrossRef]
- Johansen, T.J.; Hagen, S.F.; Bengtsson, G.B.; Mølmann, J.A. Growth temperature affects sensory quality and contents of glucosinolates, vitamin C and sugars in swede roots (Brassica napus L. ssp. rapifera Metzg.). Food Chem. 2016, 196, 228–235. [Google Scholar] [CrossRef]
| W (g) | HL (mm) | S (mm) | CW (mm) | ||
|---|---|---|---|---|---|
| Cycle (C) | |||||
| I | 1.20 ± 0.11 | 35.97 ± 2.25 b | 11.71 ± 0.76 b | 14.37 ± 0.73 b | |
| II | 1.38 ± 0.09 | 38.79 ± 2.03 a | 13.89 ± 0.64 a | 16.67 ± 0.51 a | |
| n.s. | * | *** | *** | ||
| Genotype (G) | |||||
| BN | 1.12 ± 0.12 | 40.91 ± 0.64 a | 11.35 ± 0.36 b | 14.81 ± 0.18 b | |
| CL | 1.28 ± 0.10 | 30.27 ± 1.69 b | 11.77 ± 0.75 b | 14.37 ± 1.09 b | |
| CR | 1.47 ± 0.13 | 40.96 ± 2.20 a | 15.28 ± 0.72 a | 17.37 ± 0.66 a | |
| n.s. | *** | *** | ** | ||
| C × G | |||||
| I | BN | 1.03 ± 0.11 | 42.1 ± 0.75 ab | 10.61 ± 0.24 de | 14.5 ± 0.23 bc |
| CL | 1.16 ± 0.17 | 29.2 ± 3.61 d | 10.30 ± 0.81 e | 12.2 ± 0.81 c | |
| CR | 1.41 ± 0.28 | 36.6 ± 2.31b cd | 14.22 ± 1.22 bc | 16.40 ± 1.10 ab | |
| II | BN | 1.22 ± 0.23 | 39.71 ± 0.17 abc | 12.09 ± 0.17 bc | 15.13 ± 0.11 bc |
| CL | 1.40 ± 0.12 | 31.34 ± 0.40 cd | 13.24 ± 0.12 bc | 16.53 ± 0.75 ab | |
| CR | 1.52 ± 0.07 | 45.32 ± 0.12 a | 16.34 ± 0.23 a | 18.34 ± 0.16 a | |
| n.s. | * | ** | *** |
| W (g) | HL (mm) | S (mm) | CW (mm) | LL (mm) | LW (mm) | ||
|---|---|---|---|---|---|---|---|
| Cycle (C) | |||||||
| I | 2.99 ± 0.22 | 33.91 ± 2.63 b | 27.19 ± 1.43 | 19.50 ± 1.14 | 25.51 ± 1.25 b | 17.68 ± 0.87 b | |
| II | 3.49 ± 0.22 | 42.97 ± 3.83 a | 28.61 ± 1.54 | 20.84 ± 1.43 | 31.29 ± 2.01 a | 20.75 ± 0.39 a | |
| n.s. | *** | n.s. | n.s. | ** | ** | ||
| Genotype (G) | |||||||
| BN | 3.48 ± 0.22 | 50.31 ± 3.15 a | 33.36 ± 0.49 a | 24.45 ± 0.63 a | 32.10 ± 3.15 a | 18.97 ± 1.32 | |
| CL | 2.81 ± 0.19 | 30.49 ± 1.32 b | 24.29 ± 0.91 b | 17.91 ± 0.99 b | 24.82 ± 1.04 b | 18.50 ± 0.64 | |
| CR | 3.43 ± 0.36 | 34.51 ± 2.94 b | 26.05 ± 0.77 b | 18.16 ± 1.18 b | 28.27 ± 1.44 ab | 20.17 ± 1.08 | |
| n.s. | *** | *** | *** | ** | n.s. | ||
| C × G | |||||||
| I | BN | 3.42 ± 0.46 | 43.30 ± 0.64 b | 32.40 ± 0.40 | 23.10 ± 0.35 | 25.62 ± 2.36 b | 16.43 ± 1.50 |
| CL | 2.72 ± 0.41 | 29.54 ± 2.62 d | 23.47 ± 0.20 | 17.60 ± 0.98 | 23.70 ± 1.73 b | 17.58 ± 0.95 | |
| CR | 2.82 ± 0.28 | 28.90 ± 3.18 d | 25.70 ± 1.62 | 17.80 ± 2.18 | 27.20 ± 2.66 b | 19.02 ± 2.07 | |
| II | BN | 3.53 ± 0.17 | 57.33 ± 0.23 a | 34.31 ± 0.35 | 25.81 ± 0.17 | 38.59 ± 1.45 a | 21.51 ± 0.17 |
| CL | 2.90 ± 0.12 | 31.44 ± 0.99 cd | 25.10 ± 1.85 | 18.21 ± 1.96 | 25.94 ± 1.09 b | 19.42 ± 0.52 | |
| CR | 4.03 ± 0.46 | 40.12 ± 1.33 bc | 26.41 ± 0.46 | 18.52 ± 1.45 | 29.34 ± 1.49 b | 21.32 ± 0.46 | |
| n.s. | * | n.s. | n.s. | * | n.s. |
| W (g) | SL (mm) | N (n) | LL (mm) | LW (mm) | ||
|---|---|---|---|---|---|---|
| Cycle (C) | ||||||
| I | 7.63 ± 0.40 | 118.59 ± 3.88 | 3.38 ± 0.14 b | 119.43 ± 1.86 | 36.82 ± 1.58 b | |
| II | 8.24 ± 0.39 | 122.26 ± 3.49 | 4.68 ± 0.10 a | 121.56 ± 1.59 | 39.33 ± 2.34 a | |
| n.s. | n.s. | *** | n.s. | * | ||
| Genotype (G) | ||||||
| BN | 8.97 ± 0.49 a | 108.76 ± 2.36 b | 4.01 ± 0.28 | 125.02 ± 1.03 a | 45.42 ± 1.49 a | |
| CL | 7.27 ± 0.40 b | 128.97 ± 3.46 a | 4.21 ± 0.30 | 114.45 ± 1.05 b | 32.92 ± 0.62 c | |
| CR | 7.57 ± 0.2 ab | 123.55 ± 2.47 a | 3.87 ± 0.38 | 122.02 ± 0.98 a | 35.88 ± 0.60 b | |
| * | ** | n.s. | *** | *** | ||
| C × G | ||||||
| I | BN | 8.42 ± 0.64 | 107.77 ± 3.84 | 3.51 ± 0.23 | 124.32 ± 2.08 | 42.53 ± 1.41 b |
| CL | 7.05 ± 0.83 | 125.53 ± 6.90 | 3.61 ± 0.29 | 113.28 ± 1.77 | 32.61 ± 0.87 c | |
| CR | 7.42 ± 0.52 | 122.47 ± 5.34 | 3.03 ± 0.04 | 120.71 ± 1.50 | 35.31 ± 0.86 c | |
| II | BN | 9.52 ± 0.69 | 109.74 ± 3.47 | 4.50 ± 0.29 | 125.72 ± 0.68 | 48.31 ± 0.87 a |
| CL | 7.50 ± 0.23 | 132.41 ± 0.75 | 4.82 ± 0.06 | 115.63 ± 1.04 | 33.23 ± 1.04 c | |
| CR | 7.71 ± 0.16 | 124.63 ± 0.87 | 4.71 ± 0.11 | 123.33 ± 0.92 | 36.44 ± 0.87 c | |
| n.s. | n.s. | n.s. | n.s. | * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Bella, M.C.; Toscano, S.; Arena, D.; Moreno, D.A.; Romano, D.; Branca, F. Effects of Growing Cycle and Genotype on the Morphometric Properties and Glucosinolates Amount and Profile of Sprouts, Microgreens and Baby Leaves of Broccoli (Brassica oleracea L. var. italica Plenck) and Kale (B. oleracea L. var. acephala DC.). Agronomy 2021, 11, 1685. https://doi.org/10.3390/agronomy11091685
Di Bella MC, Toscano S, Arena D, Moreno DA, Romano D, Branca F. Effects of Growing Cycle and Genotype on the Morphometric Properties and Glucosinolates Amount and Profile of Sprouts, Microgreens and Baby Leaves of Broccoli (Brassica oleracea L. var. italica Plenck) and Kale (B. oleracea L. var. acephala DC.). Agronomy. 2021; 11(9):1685. https://doi.org/10.3390/agronomy11091685
Chicago/Turabian StyleDi Bella, Maria Concetta, Stefania Toscano, Donata Arena, Diego A. Moreno, Daniela Romano, and Ferdinando Branca. 2021. "Effects of Growing Cycle and Genotype on the Morphometric Properties and Glucosinolates Amount and Profile of Sprouts, Microgreens and Baby Leaves of Broccoli (Brassica oleracea L. var. italica Plenck) and Kale (B. oleracea L. var. acephala DC.)" Agronomy 11, no. 9: 1685. https://doi.org/10.3390/agronomy11091685
APA StyleDi Bella, M. C., Toscano, S., Arena, D., Moreno, D. A., Romano, D., & Branca, F. (2021). Effects of Growing Cycle and Genotype on the Morphometric Properties and Glucosinolates Amount and Profile of Sprouts, Microgreens and Baby Leaves of Broccoli (Brassica oleracea L. var. italica Plenck) and Kale (B. oleracea L. var. acephala DC.). Agronomy, 11(9), 1685. https://doi.org/10.3390/agronomy11091685










