Hydroponic Lettuce Cultivation Using Organic Nutrient Solution from Aerobic Digested Aquacultural Sludge
Abstract
1. Introduction
2. Materials and Methods
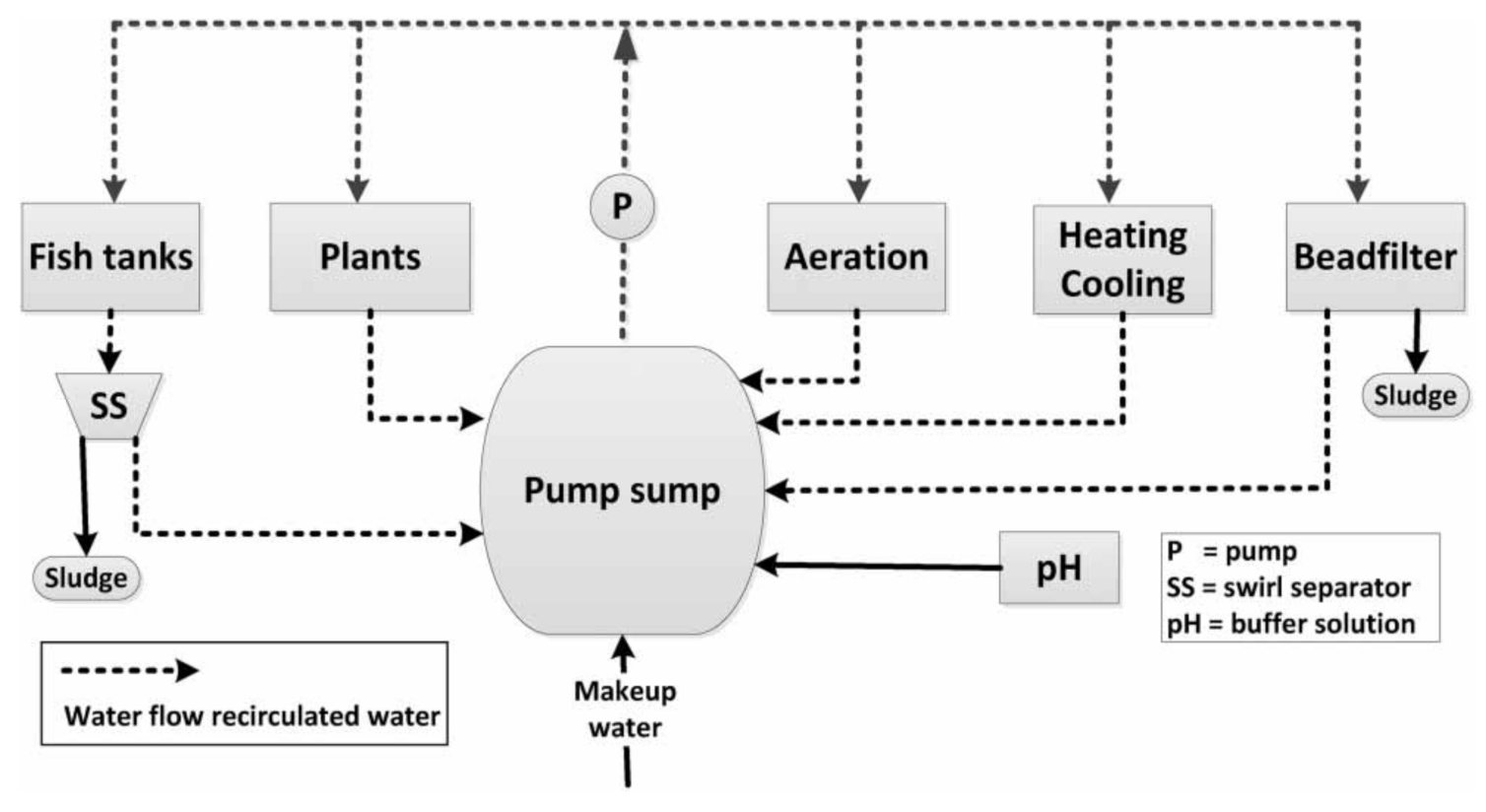
2.1. The Aquaponic Facility and Sludge Collection
2.2. Aerobic Digestion and Solid Separation
2.3. Lettuce Growth
2.4. Analytical Methods
2.5. Data Analysis and Statistics
3. Results and Discussion
3.1. Sludge Characterisation before and after AD
3.2. Nutrient Mobilization during Aerobic Digestion
3.3. Comparative Lettuce Growth Studies
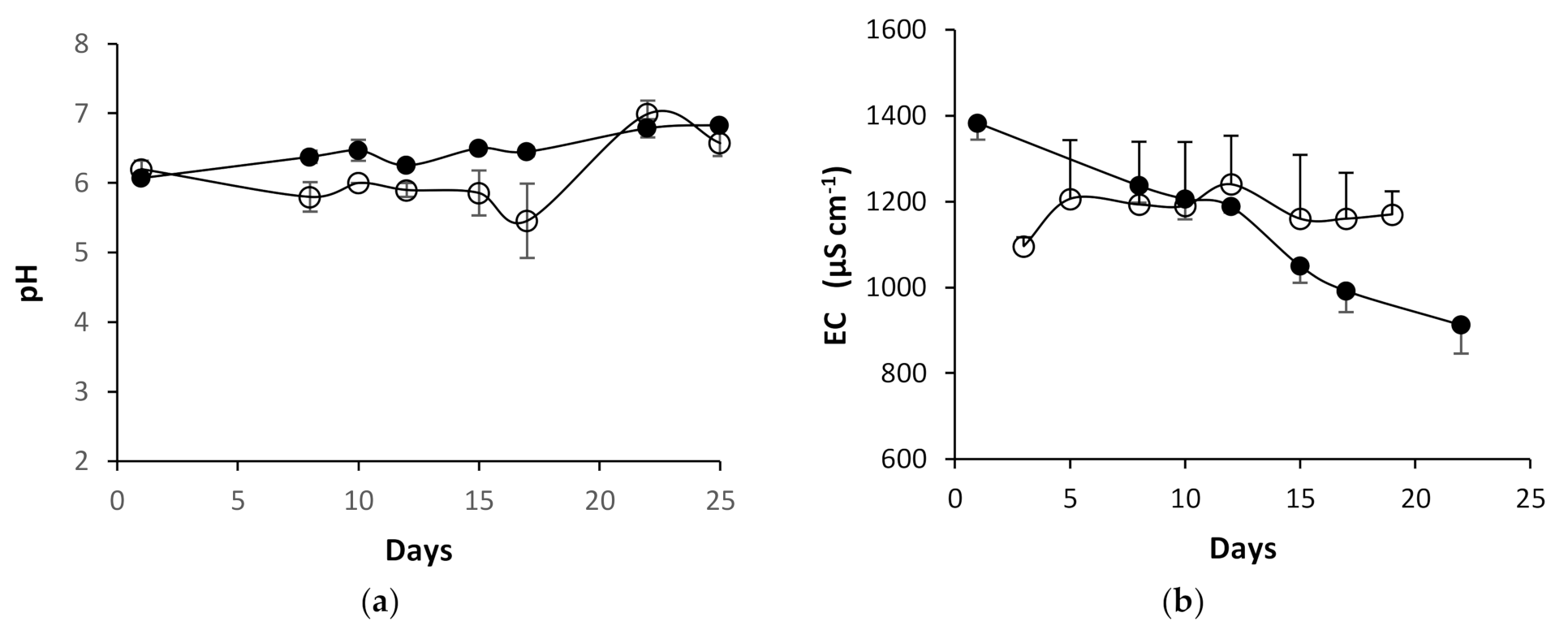
3.3.1. pH and EC Values during Growth in the NFT-System
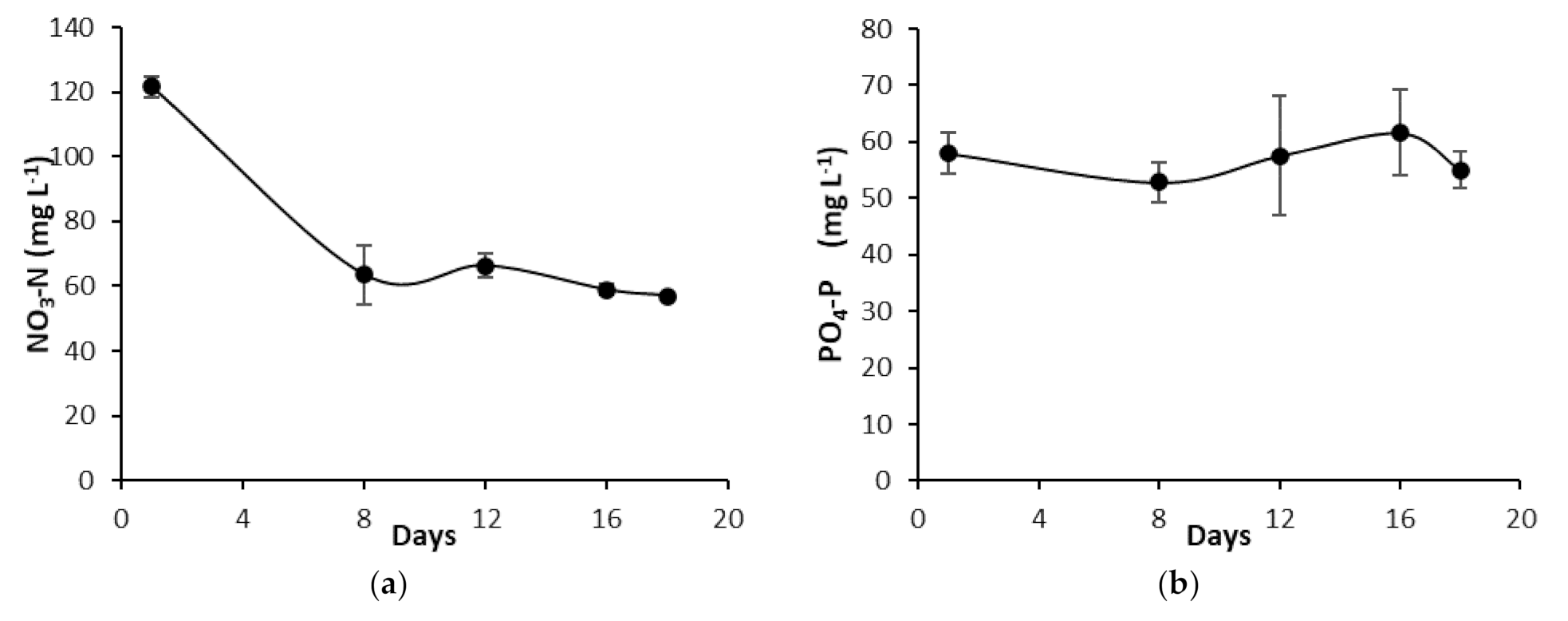
3.3.2. PO4-P and NO3-N Concentrations of the ONS during Growth in the NFT-System
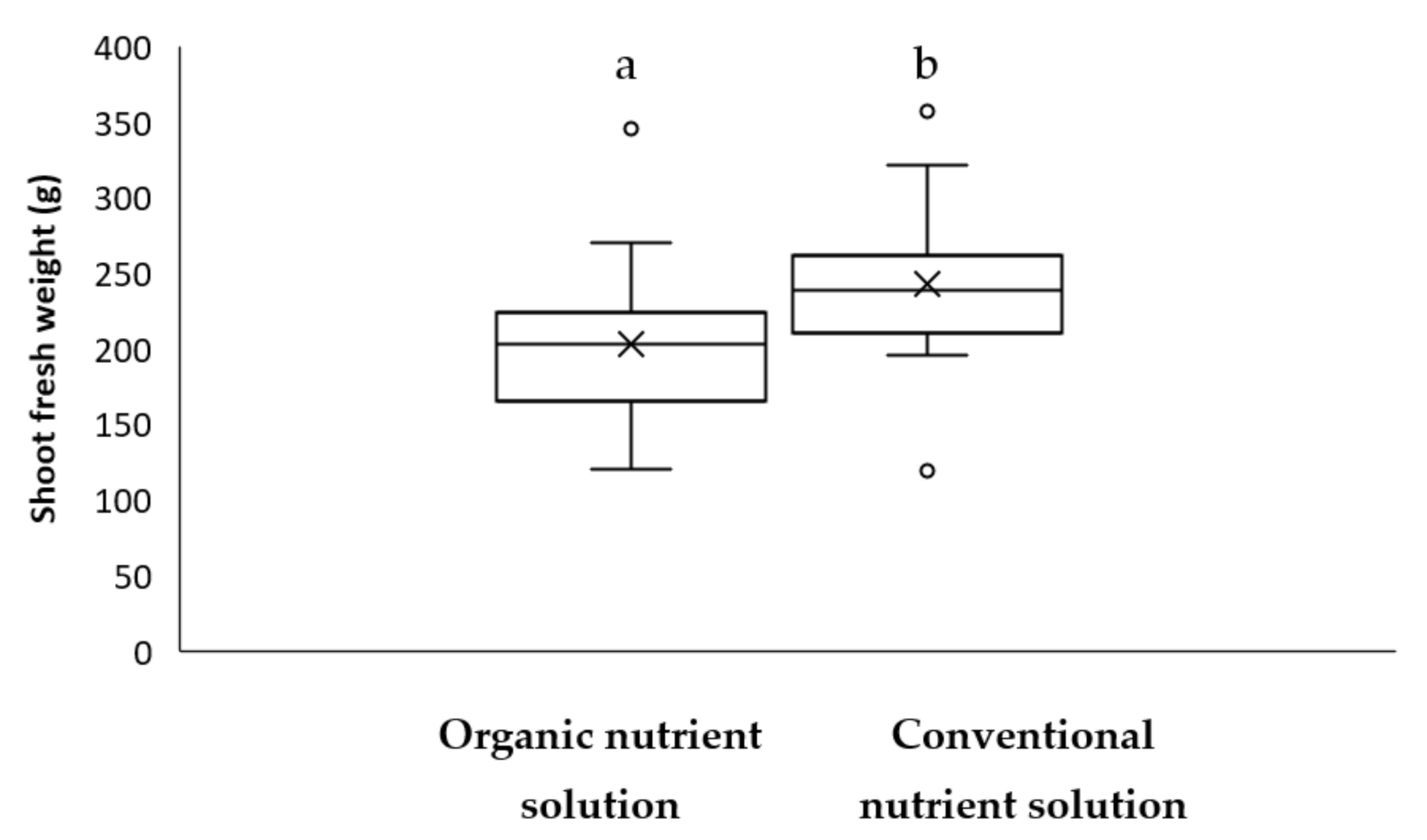
3.3.3. Effect of ONS versus CNS on Lettuce Growth and Yield
3.4. Heavy Metals Content in Lettuce Leaves
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Maucieri, C.; Nicoletto, C.; van Os, E.; Anseeuw, D.; Van Havermaet, R.J.R. Hydroponic Technologies. In Aquaponics Food Production Systems; Goddek, S., Joyce, A., Kotzen, B., Burnell, G.M., Eds.; Springer Nature: Cham, Switzerland, 2019. [Google Scholar]
- Jones, J.B. Hydroponics A Practical Guide for the Soilless Grower, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2005; Volume 53, ISBN 9788578110796. [Google Scholar]
- Somerville, C.; Cohen, M.; Pantanella, E.; Stankus, A.; Lovatelli, A. Small-Scale Aquaponic Food Production; FAO Fisheries and Aquaculture Technical Paper; FAO: Rome, Italy, 2014; ISBN 9789251085325. [Google Scholar]
- Shinohara, M.; Aoyama, C.; Fujiwara, K.; Watanabe, A.; Ohmori, H.; Uehara, Y.; Takano, M. Microbial mineralization of organic nitrogen into nitrate to allow the use of organic fertilizer in hydroponics. Soil Sci. Plant Nutr. 2011, 57, 190–203. [Google Scholar] [CrossRef]
- Atkin, K.; Nichols, M.A. Organic hydroponics. Acta Hortic. 2004, 648, 121–127. [Google Scholar] [CrossRef]
- Mehta, C.M.; Khunjar, W.O.; Nguyen, V.; Tait, S.; Batstone, D.J. Technologies to recover nutrients from waste streams: A critical review. Crit. Rev. Environ. Sci. Technol. 2015, 45, 385–427. [Google Scholar] [CrossRef]
- Ekpo, U.; Ross, A.B.; Camargo-Valero, M.A.; Williams, P.T. A comparison of product yields and inorganic content in process streams following thermal hydrolysis and hydrothermal processing of microalgae, manure and digestate. Bioresour. Technol. 2016, 200, 951–960. [Google Scholar] [CrossRef]
- Kawamura-Aoyama, C.; Fujiwara, K.; Shinohara, M.; Takano, M. Study on the hydroponic culture of lettuce with microbially degraded solid food waste as a nitrate source. Jpn. Agric. Res. Q. 2014, 48, 71–76. [Google Scholar] [CrossRef]
- Phibunwatthanawong, T.; Riddech, N. Liquid organic fertilizer production for growing vegetables under hydroponic condition. Int. J. Recycl. Org. Waste Agric. 2019, 8, 369–380. [Google Scholar] [CrossRef]
- Kano, K.; Kitazawa, H.; Suzuki, K.; Widiastuti, A.; Odani, H.; Zhou, S.; Chinta, Y.D.; Eguchi, Y.; Shinohara, M.; Sato, T. Effects of Organic Fertilizer on Bok Choy Growth and Quality in Hydroponic Cultures. Agronomy 2021, 11, 491. [Google Scholar] [CrossRef]
- Williams, K.A.; Nelson, J.S. Challenges of using organic fertilizers in hydroponic production systems. In Proceedings of the Symposium on Water, Eco-Efficiency and Transformation of Organic Waste in Horticultural Production, Brisbane, Australia, 17–22 August 2014; Volume 1112, pp. 365–370. [Google Scholar]
- Garland, J.L.; Mackowiak, C.L.; Strayer, R.F.; Finger, B.W. Integration of waste processing and biomass production systems as part of the KSC breadboard project. Adv. Space Res. 1997, 20, 1821–1826. [Google Scholar] [CrossRef]
- Del Campo, L.M.; Ibarra, P.; Gutiérrez, X.; Takle, H. Utilization of Sludge from Recirculation Aquaculture Systems; Nofima: Tromsø, Norway, 2010. [Google Scholar]
- Olsen, L.; Holmer, M.; Olsen, Y. Perspectives of Nutrient Emission from Fish Aquaculture in Coastal Waters; The Fishery and Aquaculture Industry Research Fund: Oslo, Norway, 2008. [Google Scholar]
- Delaide, B.P.L.; Goddek, S.; Keesman, K.J.; Jijakli, M.H. A methodology to quantify the aerobic and anaerobic sludge digestion performance for nutrient recycling in aquaponics. Biotechnol. Agron. Soc. Environ. 2018, 22, 106–112. [Google Scholar]
- Ezziddine, M.; Liltved, H.; Homme, J.M. A method for reclaiming nutrients from aquacultural waste for use in soilless growth systems. Water Sci. Technol. 2020, 81, 81–90. [Google Scholar] [CrossRef]
- Monsees, H.; Keitel, J.; Paul, M.; Kloas, W.; Wuertz, S. Potential of aquacultural sludge treatment for aquaponics: Evaluation of nutrient mobilization under aerobic and anaerobic conditions. Aquac. Environ. Interact. 2017, 9, 9–18. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, L. Removal of phosphate anions using the modified chitosan beads: Adsorption kinetic, isotherm and mechanism studies. Powder Technol. 2015, 277, 112–119. [Google Scholar] [CrossRef]
- Davidson, J.; Good, C.; Welsh, C.; Brazil, B.; Summerfelt, S. Heavy metal and waste metabolite accumulation and their potential effect on rainbow trout performance in a replicated water reuse system operated at low or high system flushing rates. Aquac. Eng. 2009, 41, 136–145. [Google Scholar] [CrossRef]
- Ferreira, L.M.H.; Lara, G.; Wasielesky, W., Jr.; Abreu, P.C. Biofilm versus biofloc: Are artificial substrates for biofilm production necessary in the BFT system? Aquac. Int. 2016, 24, 921–930. [Google Scholar] [CrossRef]
- Wagner, S.C. Biological Nitrogen Fixation. Nat. Educ. Knowl. 2011, 3, 1–15. [Google Scholar]
- Senbayram, M.; Gransee, A.; Wahle, V.; Thiel, H. Role of magnesium fertilisers in agriculture: Plant-soil continuum. Crop Pasture Sci. 2015, 66, 1219–1229. [Google Scholar] [CrossRef]
- Jordan, R.A.; Ribeiro, E.F.; de Oliveira, F.C.; Geisenhoff, L.O.; Martins, E.A.S. Yield of lettuce grown in hydroponic and aquaponic systems using different substrates. Rev. Bras. Eng. Agric. Ambient. 2018, 22, 525–529. [Google Scholar] [CrossRef]
- de Lira, R.M.; de França e Silva, Ê.F.; da Silva, G.F.; dos Santos, A.N.; Rolim, M.M. Production, water consumption and nutrient content of Chinese cabbage grown hydroponically in brackish water. Rev. Cienc. Agron. 2015, 46, 497–505. [Google Scholar] [CrossRef]
- Eissa, M.A.; Negim, O.E. Heavy metals uptake and translocation by lettuce and spinach grown on a metal-contaminated soil. J. Soil Sci. Plant Nutr. 2018, 18, 1097–1107. [Google Scholar] [CrossRef]
- Zubillaga, M.S.; Lavado, R.S. Heavy metal content in lettuce plants grown in biosolids compost. Compost Sci. Util. 2002, 10, 363–367. [Google Scholar] [CrossRef]
- Qasem, N.A.A.; Mohammed, R.H.; Lawal, D.U. Removal of heavy metal ions from wastewate: A comprehensive and critical review. Npj Clean Water 2021, 4, 1–15. [Google Scholar] [CrossRef]
- Zhang, G. Improving Productivity and Quality of Low-Potassium Lettuce in a Plant Factory with Artificial Lighting. Ph.D. Thesis, Graduate Schoold of Horticulture, Chiba University, Chiba, Japan, June 2016. [Google Scholar]
- Adamse, P.; Van der Fels-Klerx, H.J.; de Jong, J. Cadmium, lead, mercury and arsenic in animal feed and feed materials–trend analysis of monitoring results. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2017, 34, 1298–1311. [Google Scholar] [CrossRef] [PubMed]

| Parameter | Before AD | After AD | Remaining Concentrated Sludge after AD and Solid Separation |
|---|---|---|---|
| Volume of sludge (L) | 120 | 101 | 5 out of the 101 |
| pH | 7.1 | 7.0 | 7.0 |
| TSS (g L−1) | 1.10 (132.0) | 0.46 (46.5) | 10.00 (50.0) |
| BOD5 (mg L−1) | 145 (17.4) | 16 (1.6) | - |
| COD (mg L-1) | 1750 (210) | 390 (39.4) | 7900 (39.5) |
| Total P (TP) (mg L−1) | 92 (11.0) | 93 (9.4) | 560 (2.8) |
| Total N (TN) (mg L−1) | 77.5 (9.3) | 120 (12.1) | 170 (0.9) |
| Parameter | Before AD | After AD and Solids Separation | Standard Recommended Range [2] | ||
|---|---|---|---|---|---|
| Mean | STD | Mean | STD | ||
| Soluble macronutrients | |||||
| NH4-N (mg L−1) | <1.5 | <1.5 | 100 to 200 | ||
| NO3-N (mg L−1) | 40.5 (4.9) | 1.5 | 121.7 (12.3) | 3.09 | |
| P (mg L−1) | 22.5 (2.7) | 0.5 | 58.0 (5.9) | 3.74 | 15 to 90 |
| K (mg L−1) | 104.0 | 2 | 185.3 | 12.36 | 80 to 350 |
| Mg (mg L−1) | 9.7 | 0 | 17.0 | 0 | 26 to 96 |
| Ca (mg L−1) | 64.0 | 0 | 124.0 | 6.53 | 122 to 220 |
| S (mg L−1) | 13.0 | 0 | 18.0 | 1.41 | |
| Soluble micronutrients | |||||
| B (mg L−1) | 0.025 | 0.005 | 0.050 | 0.14 to 1.5 | |
| Cu (mg L−1) | 0.02 | 0 | 0.03 | 0 | 0.07 to 0.1 |
| Mn (mg L−1) | 0.085 | 0.005 | 0.020 | 0 | 0.5 to 1 |
| Mo (mg L−1) | <0.01 | <0.01 | 0.05 to 0.06 | ||
| Zn (mg L−1) | 0.56 | 0 | 0.83 | 0.03 | 0.5 to 2.5 |
| Fe (mg L−1) | 1.15 | 0.025 | 1.24 | 0.05 | 4 to 10 |
| Others | |||||
| Na (mg L−1) | 18 | 0 | 22.3 | 0.94 | |
| Cl (mg L−1) | 26.5 | 1.5 | 41.3 | 4.64 | |
| Parameter | Soluble Concentration (mg L−1) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NO3-N | NH4-N | P | K | Ca | Mg | S | Zn | B | Cu | Fe | Mn | Mo | |
| Applied ONS | 121.7 | <1.5 | 58 | 185 | 124 | 17 | 18 | 0.83 | 0.05 | 0.03 | 1.2 | 0.02 | <0.01 |
| Applied CNS | 107 | 4.0 | 23 | 140 | 94 | 23 | 23 | 0.26 | 0.19 | 0.07 | 1.7 | 0.42 | 0.04 |
| Organic Nutrient Solution | Conventional Nutrien Solution | Statistical Significance | |||
|---|---|---|---|---|---|
| Mean | STD | Mean | STD | ||
| Macronutrients (g/kg Dry Matter) | |||||
| N | 53 a | 0 | 38.6 a | 23.2 | 1 |
| P | 10.26 a | 0.5 | 6.4 b | 0.5 | 0.002 |
| K | 125 a | 1.6 | 50 b | 19.8 | 0.006 |
| Mg | 1.7 a | 0.08 | 3.36 b | 0.33 | 0.002 |
| Ca | 30 a | 2.16 | 15 b | 14.14 | 0.001 |
| S | 3.43 a | 0.1 | 2.7 b | 0.2 | 0.012 |
| Micronutrients (mg/kg Dry Matter) | |||||
| B | 34 a | 2.16 | 22.33 b | 1.25 | 0.003 |
| Cu | 5.4 a | 1.2 | 6.56 a | 0.6 | 0.28 |
| Mn | 119 a | 22.5 | 143.3 a | 26.25 | 0.37 |
| Mo | 0.6 a | 0.04 | 1.43 b | 0.4 | 0.046 |
| Zn | 52 a | 21.23 | 50.3 a | 14.38 | 0.93 |
| Fe | 102 a | 13.95 | 130 a | 8.16 | 0.07 |
| Heavy Metal Content in Leaves of Lettuce (mg kg−1 Dry Weights) | Maximum Permissible Limit (mg kg−1 Dry Weights) | ||
|---|---|---|---|
| Mean | STD | ||
| Lead (Pb) | 0.21 | 0.06 | 0.3 |
| Cadmium (Cd) | 0.23 | 0.05 | 0.2 |
| Nickel (Ni) | <0.01 | - | 1.5 |
| Zinc (Zn) | 52 | 26.0 | 60–80 |
| Copper (Cu) | 5.4 | 1.21 | 40 |
| Arsene (As) | <0,04 | - | - |
| Chromium (Cr) | <1.0 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ezziddine, M.; Liltved, H.; Seljåsen, R. Hydroponic Lettuce Cultivation Using Organic Nutrient Solution from Aerobic Digested Aquacultural Sludge. Agronomy 2021, 11, 1484. https://doi.org/10.3390/agronomy11081484
Ezziddine M, Liltved H, Seljåsen R. Hydroponic Lettuce Cultivation Using Organic Nutrient Solution from Aerobic Digested Aquacultural Sludge. Agronomy. 2021; 11(8):1484. https://doi.org/10.3390/agronomy11081484
Chicago/Turabian StyleEzziddine, Maha, Helge Liltved, and Randi Seljåsen. 2021. "Hydroponic Lettuce Cultivation Using Organic Nutrient Solution from Aerobic Digested Aquacultural Sludge" Agronomy 11, no. 8: 1484. https://doi.org/10.3390/agronomy11081484
APA StyleEzziddine, M., Liltved, H., & Seljåsen, R. (2021). Hydroponic Lettuce Cultivation Using Organic Nutrient Solution from Aerobic Digested Aquacultural Sludge. Agronomy, 11(8), 1484. https://doi.org/10.3390/agronomy11081484





