1. Introduction
According to Kristek et al. [
1], sugar beet is a profitable industrial crop grown commercially for sugar production. It is grown in Europe over approximately 2,000,000 ha, which is about 70% of the total arable land in the world. In the Republic of Croatia, sugar represents an important export product. Until 2012, it was grown on 23,215 ha with an average yield of 50.95 t/ha [
1]. In recent years, sugar beet production has decreased not only in Croatia, but in all countries of the EU. For example, in 2018, sugar beet yield per hectare decreased by 15% compared to 2017 [
2]. Croatian sugar beet production in 2018 was reduced by 40%, which means up to 524 thousand tons compared to the production of the previous year [
3].
Sugar beet has a long growing season of up to 200 days, during which it can be exposed to many diseases, insect pests and fungal diseases [
4]. According to Meier et al. [
5], phenological growth stages are defined as follows: Germination from 00-dry seed to 09-emergence: shoot emerges through the soil surface; leaf development or youth stage from 10-first leaf visible: cotyledons unfolded horizontally to 19-nine and more leaves unfolded; rosette growth (crop cover) from 31-beginning of crop cover: leaves cover 10% of the ground to 39-crop cover complete: Leaves cover 90% of the ground; development of harvestable vegetative plant parts is defined by code 49-Beet root has reached harvestable size. Other stages represent the appearance of inflorescences in the 2nd year of growth. BBCH identification codes are shown in
Figure 1. The decrease in production is due to economic reasons and changing climatic conditions, which led to major problems related to the inability to effectively control pests. In Croatia, pests are a limiting factor in sugar beet production [
6].
The most common pests that attack sugar beet in the early stages of leaf development or the youth stage (BBCH 10–19) and cause major damage are wireworms (Agriotes spp., Coleoptera: Elateridae), which live in the soil and feed on roots. The main damage occurs in spring and is manifested by thinning of the crop stand and reduction in yield [
7]. Flea beetles (
Chaetocnema tibialis Ill., Coleoptera: Chrysomelidae) cause damage by feeding on leaves and forming small round holes (1 mm in diameter) that enlarge as leaves grow. Sometimes they feed on the stem in addition to the leaves [
7]. When the plant is at the cotyledon stage, one flea can cause 33% damage per day, three fleas up to 62% and five fleas can cause as much as 90% damage to the plant. Their activity increases with higher temperatures, i.e., warmer climate-more damage [
8]. Sugar beet weevils (
Bothynoderes punctiventris Germ., Coleoptera: Curculionidae) emerge from the soil in early spring when the upper layer reaches a temperature of 6–10 °C [
9]. Normally, sugar beets are at the cotyledon stage at this time, so the damage can be extensive. In one day, an adult weevil can consume up to 50% of the emerged plants in m
2. Again, the insect’s feeding rate increases with temperature. At 20 °C, an adult weevil eats 34 mm
2 of leaf area, while at 32 °C the area increases up to 145 mm
2 [
8]. The caterpillars of noctuid moths (
Agrotis segetum Schiff.,
Agrotis ypsilon Hubn. and
Euxoa temera Hb., Lepidoptera: Noctuidae) can cause damage to more than 150 host plants. The first generation of caterpillars is the most damaging, feeding on underground and aboveground parts of newly emerged plants. Infestation can lead to thinning of the crop stand and reduced yields. Often a caterpillar can bite off the plant haze. According to Čamprag [
10], 5–10 caterpillars of the species
A. ipsilon can damage 90% of plants up to 8 cm high. Later in vegetation, sugar beet can be attacked by several species of aphids (Hemiptera: Aphididae) such as
Smynthurodes phaseoli West,
Pemphigus fuscicornis Koch and
Pemphigus betae Doane on roots and
Aphis fabae Scopoli and
Myzus persicae Sulzer as the most common species on aboveground organs [
11]. In addition to aphids, caterpillars of the rapeseed moth (
Scrobipalpa ocellatella Boyd, Lepidoptera: Gelechiidae), the cabbage moth and bright-line brown-eye moth (
Mamestra brassicae L. and
Lacanobia oleracea L.) as well as silver Y (
Autographa gamma L., Lepidoptera: Noctuidae) can also attack sugar beet during vegetation, but these pests are rarely controlled [
12].
According to Bažok et al. [
13], soil pests (mainly wireworms) were regularly controlled in Croatia by the application of lindane, terbufos, forat, chlormephos, chlorpyrifos, phoxim and carbofuran. For flea beetle control, a wide range of active ingredients such as diazinon, phosalone, monocrotophos, thiometon, carbaryl, alphamethrin, cypermethrin and deltamethrin were used. Aphids were mostly controlled with systemic active ingredients tiometon, dimethoate, methyl demeton, carbamil etc. or permethrin. The average amount of active ingredient/ha of sugar beet grown was 1.64 kg during 1981–1989. During the 1990s to 2018, the pests were controlled with 0.05 to 0.1 kg active ingredient of neonicotinoids as seed treatment/ha of grown sugar beet. Foliar application of insecticides was made only when necessary to control sugar beet weevil. Therefore, neonicotinoids contributed to a large reduction in the amount of insecticide used in sugar beet cultivation [
13]. It has been confirmed that treatment of sugar beet seeds with imidacloprid provides satisfactory protection of young plants against low to moderate infestation by wireworms, flea beetles and aphids [
14]. Hauer et al. [
15] analyzed the possible consequences of the ban of neonicotinoids on pest incidence on sugar beet under production conditions in north and central Europe. They concluded that seed treatment with neonicotinoids provides sufficient protection against aphids, the vectors of sugar beet virus. Since aphids do not occur annually in every field, they predicted that the ban would not have serious consequences for sugar beet production. In addition, they suggested developing monitoring systems and models to identify regions (and years) with high pest risk and allowing the use of insecticides for seed treatment only when aphid pressure is expected to be high. In 2018, the EU Commission completely banned the use of the active substances imidacloprid, clothianidin and thiamethoxam in the field [
16,
17,
18], and only their use in permanent greenhouses remains possible because of the risk to bees. Now, a large proportion of arable and industrial crops, including sugar beet, remain unprotected while pest pressure increases. In their work, Hauer et al. [
15] analyzed sugar beet production in the countries of northwestern Europe and did not consider the different climatic conditions and pest occurrence in eastern and southeastern Europe. Considering climate change, the global economic and health crisis and the FAO Sustainable Development Goals (SDGs) (e.g., zero hunger by 2030 [
19]) the question remains whether it will be possible to grow food, in this case sugar beet, without pesticides.
The aim of this study was to determine: (1) the efficacy of seed treatment with neonicotinoids on the main sugar beet pests during two growing seasons and different climatic conditions at two locations; (2) the actual vulnerability to individual pests under the specific agro climatic conditions and the extent to which neonicotinoid seed treatment is effective in preventing damage; and (3) the possible consequences of the ban of neonicotinoids on the future of sugar beet production in southeastern Europe.
3. Results
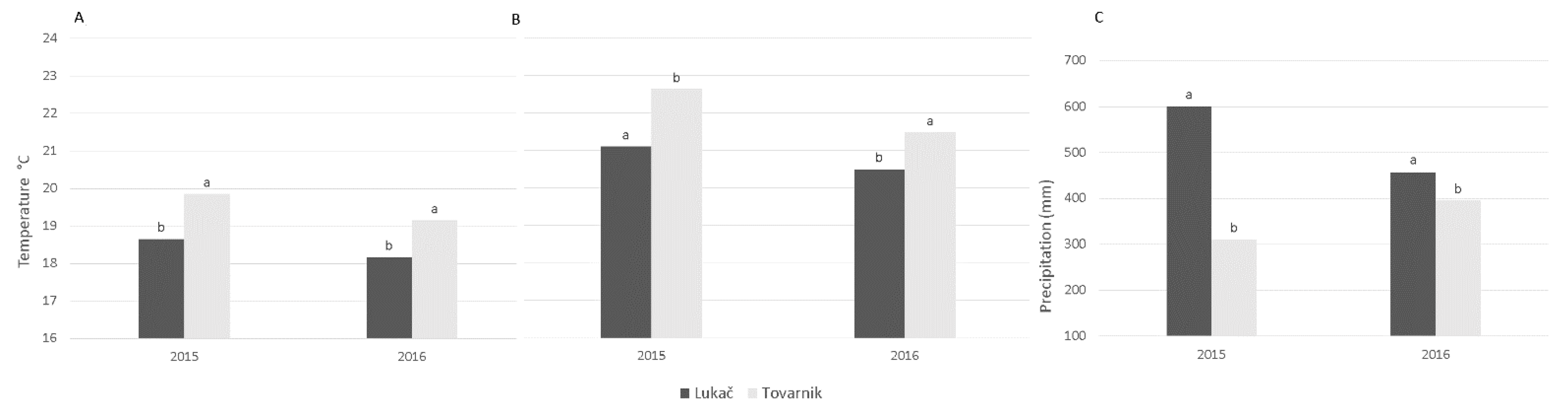
Figure 2A–C show the climatic analysis between Lukač and Tovarnik during two growing seasons. There were no significant differences between climatic conditions at both sites between the two years studied. Compared to Tovarnik, average air and soil temperatures were lower in Lukač in both years of study. The amount of precipitation was higher in Lukač in 2015, while in 2016 the differences were not significant.
During the two growing seasons, infestations of wireworms, flea beetles, sugar beet weevils, aphids and caterpillars were recorded depending on the location. No pests were detected before BBCH 12, which corresponds to youth stage of leaf development (first pair of leaves unfolded).
Table 1 shows the results on crop stand on the experimental plots and indicates wireworm damage on sugar beet plants. In 2015, a heavy wireworm infestation was detected at the Tovarnik site. Plant density on the untreated plots was significantly reduced during BBCH 19 and 31 compared to the treated plots. In Lukač, no significant difference was found between the variants. In 2016, the evaluation showed differences in the number of plants on treated and untreated plots at almost all stages of development, leading to the conclusion that both sites were infested with wireworms (
Table 1).
Crop stand as a result of wireworm damage was significantly affected by location, insecticide treatment and their combination at all stages of plant development (from BBCH 12 to BBCH 34), proving that wireworm infestation was significantly different at two locations and also that neonicotinoid seed treatments are able to protect plants from wireworm infestation under different environmental conditions (
Table 2). The third factor, year, significantly affected plant density in the first two observations (BBCH 12 and 16), while later in plant development plant density was not significantly affected by year. However, plant densities were significantly (
p > 0.05%) influenced by the combination of location and year and by the combination of insecticide treatment and year throughout the course of plant development from BBCH 12 to BBCH 34. The combination of all three factors significantly influenced plant densities over the period of plant development from BBCH 19 to BBCH 34.
Table 3 shows the percentage of flea beetle damage on sugar beet plants. In 2015, flea damage was higher at the very beginning of vegetation in Tovarnik than in Lukač and significantly higher on untreated varieties in both locations. In Lukač, the plants developed well until the end of BBCH 16 (about day 45), as the infestation stopped. All treated varieties in both trials and both years of the study significantly reduced pest infestation. Significant differences in flea beetle numbers on treated versus untreated variants were observed during sugar beet rosette growth and plant cover development. In 2016, a significant difference in flea damage between treated and untreated variants was observed in the Lukač trial throughout the development stage. At the same time, the damage in the Tovarnik trial was much lower and the differences between the treated and untreated variants were smaller (
Table 3).
Plant damage caused by flea beetles was significantly influenced by location and year at two plant development stages (BBCH 16 and 19) (
Table 4). The average percentage of damage at the Lukač site was significantly higher and amounted to 22.1 and 27.4% compared to the damage observed at the Tovarnik site, where it amounted to 17.4% and 18.6% in BBCH 16 and BBCH 19, respectively. At the same time, plant damage was significantly affected by insecticide treatments at all three stages of plant development (BBCH 12–19), proving that neonicotinoid seed treatments protect plants against flea beetle infestation. The significant (
p > 0.05%) interaction between all three factors (location × insecticide treatment × year) for flea beetle damage was present at all three plant developmental stages. A significant insecticide treatment × location interaction for flea beetle damage was observed at the first and last observed plant development stages (BBCH 12 and 19). Significant interactions between “location × year” and “insecticide application × year” for flea beetle damage existed at all three observed plant development stages (from BBCH 12–19). As no flea beetle damage was observed at the Lukač site in 2015 at BBCH 31 and BBCH 34, factorial analysis was not performed for these two samplings.
Table 5 shows the damage caused by the sugar beet weevil damages on sugar beet plants. The attack of sugar beet weevils was relatively weak in 2015 at both locations and treatments. As expected, some efficacy of insecticides in reducing the level of damage was observed in the Lukač trial. No more sugar beet weevils were observed during BBCH 31–34. In 2016, the infestation was significantly higher, especially in the trial in Tovarnik. Damage on untreated plots was significantly higher than on treated ones. Under these conditions, seed treatment achieved satisfactory results in protecting sugar beet at the most sensitive stages of development.
Plant damage caused by sugar beet weevil was significantly (
p > 0.05%) influenced by location and insecticide treatment at all three plant development stages (from BBCH 12 to BBCH 19) and significantly influenced by year at BBCH 16 and BBCH 19 (
Table 6). The average percentage of damage at the Lukač site was significantly lower. It was 0.63, 0.67 and 2.49% compared to the damage observed at the Tovarnik locality, where it was 3.42, 5.36 and 5.41% at BBCH 12, BBCH 16 and BBCH 19, respectively. At the same time, plant damage was significantly affected by the insecticide treatments at all three stages of plant development (BBCH 12–19), proving that the neonicotinoid seed treatments protect plants from sugar beet weevil attack at the early stages of development. The significant interaction (
p > 0.05%) between all the three factors (location × insecticide treatment × year) for sugar beet weevil damages was present only at BBCH 19. No significant insecticide “treatment × location” interaction for sugar beet weevil damage was observed at any stage of plant development. Significant interactions (
p > 0.05%) between “location × year” and “insecticide application × year” for sugar beet weevil damage existed at two of three observed plant developmental stages (BBCH 12 and BBCH 16). The factorial analysis was not performed for BBCH 31 and BBCH 34, as no sugar beet weevil damages were recorded at either location.
Table 7 shows the percentage of sugar beet plants infested with caterpillars at different stages of development. In 2015, no caterpillars were observed in the youth stage in the field trials in Tovarnik. In Lukač, caterpillars appeared 20 days earlier than expected, at the leaf development stage at BBCH 19. In 2016, no caterpillar damage was observed in Tovarnik, while in Lukač, caterpillars appeared from BBCH 31. The maximum caterpillar infestation was lower than in the previous year.
The percentage of plants damaged by caterpillars was significantly influenced by location on all sampling dates (
Table 8). The percentage of damaged plants was significantly higher in Lukač than in Tovarnik. At the same time, the percentage of damaged plants was significantly influenced by insecticide treatments at BBCH 19, 31, 34 and 36, proving that insecticide treatments significantly protected young sugar beet plants from caterpillars until BBCH 38. No significant differences were observed between two insecticide treatments in percentage of damaged plants except in Lukac at BBCH 19. The percentage of damaged plants was also significantly influenced by the year. Damage was higher in 2016 compared to 2015 at all observed plant development stages. The significant interaction (
p > 0.05%) between all three factors (location × insecticide treatment × year) for damage caused by caterpillars was present only at BBCH 36. Significant interaction between insecticide “treatment × location” and “location × year” for damage caused by caterpillars was observed at all stages of crop development. Significant interactions between “insecticide application × year” for damage caused by caterpillars were only present at one observed plant development stage (BBCH 36).
No significant aphid damage was observed in the 2015 trials (
Table 9). Some minor damage occurred at the later stage, during rosette growth (BBCH 31), but according to the Townsend-Heuberger formula the percentage of infested plants did not exceed 3.5%. In 2016, during the whole vegetation, the aphid infestation on the trial in Lukač was below 2%. In Tovarnik, damage occurred during BBCH 31 and ranged from 4% in the control to 12% in the imidacloprid treatment, while no significant damage was observed in the thiamtetoksam treatment.
Aphid infestation was very low in both study years and was significantly (
p > 0.05) influenced by location on four of five sampling dates (from BBCH 31 to 38) (
Table 10). Percentages of damage were significantly higher in Tovarnik (0.66, 1.19, 2.36 and 4.02% at BBCH 31, 34, 36 and 38, respectively) than in Lukač (0.03, 0.05, 0.77 and 0.15% at BBCH 31, 34, 36 and 38, respectively). The percentage of damage was significantly affected by insecticide treatments at the three observed plant development stages (BBCH 31–36). However, damage was significantly higher in imidacloprid treated plots compared to thiamethoxam and untreated plots. Percent damage was significantly influenced by year at two observed plant developmental stages. The significant interaction (
p > 0.05%) between all three factors (location × insecticide treatment × year) for aphid damage does not exist in any observed plant development stage. Significant (
p > 0.05%) interaction between insecticide “treatment × location” for aphid damage was observed in all observed plant developmental stages. A significant interaction between “location × year” and “insecticide application × year” for aphid damage exists in three out of three observed plant development stages (from BBCH 34 to BBCH 38).
4. Discussion
EFSA is requested to evaluate the justifications submitted by Member States that authorisations of neonicotinoids (imidacloprid, thiamethoxam and clothianidin) for seed coating of sugar beet are necessary due to a risk from certain pests that cannot be controlled by any reasonable means. EFSA is expected to report the results of its assessment by 2 October 2021. The results of our study provide important input and additional arguments for this assessment.
With the main objective of determining the efficacy of seed treatments with neonicotinoids on the main sugar beet pests, this study led to five main findings: (i) neonicotinoid treatments maintain crop stand and successfully suppress wireworms; (ii) neonicotinoid seed coating significantly reduces flea beetle damage; (iii) neonicotinoid seed coating can provide adequate control against weevils under low population pressure; (iv) neonicotinoid seed coating cannot reduce damage by noctuids at later growth stages of sugar beet; (v) at low population pressure of aphids, a solid conclusion on the effectiveness of neonicotinoid seed coating is not possible.
The experimental site in Tovarnik is located in the eastern part of Croatia, while Lukač is located in the northwestern part. No significant differences were found between years for all three observed climatic factors and at both sites. When comparing the sites, the Tovarnik site has higher average annual air and soil temperatures and lower precipitation, although the amount of precipitation in 2016 did not differ significantly between the sites. Therefore, we can conclude that our study was conducted in two regions with different climatic conditions. Similar results for these regions are reported by other authors [
23,
24,
25,
26].
4.1. Wireworms
Before the introduction of neonicotinoids in Croatia (between 1980 and 1990), wireworms were controlled on 50 to 95% of all sugar beet fields in the region of east Croatia. The average consumption of insecticides for wireworm control ranged from 0.8 to 1.4 kg active ingredient/ha of sown sugar beet, depending on the year. The most commonly used active ingredients were lindane (organochlorine), terbufos, forate, chlormephos, chlorpyrifos, phoxim (organophosphates) and carbofuran (carbamates) [
13]. After the introduction of neonicotinoids in the mid-1990s, all fields were sown with coated seed and 100% of the fields were treated. However, additional treatments against wireworms were not applied and the amount of insecticides used was much lower compared to the previous period. It was 0.073 kg active ingredient/ha of sown sugar beet [
13]. Routine prophylactic use of neonicotinoid seed coatings as comfort insecticides is no longer allowed in EU countries. Based on the present results as well as the reports of other authors [
27,
28], neonicotinoid seed coatings should only be applied when the wireworm population reaches a threshold level. For this purpose, different decision strategies are studied [
29] and proposed [
27,
30]. As outlined by Barcsics et al. [
31], rational IPM strategies exist and appropriate treatment options or monitoring tools are under development. However, it remains unclear whether the same tools are applicable to sugar beet. Based on the fact that other tools exist for wireworm management in other crops, further research will be conducted to determine if neonicotinoids can be fully substituted for wireworm management in sugar beet production. However, it would make sense to use neonicotinoids only as a very last resort for wireworm control when there is a real risk from infestation (based on forecasts), as also suggested by Hauer at al. [
15].
In Croatia, the economic thresholds for wireworms in sugar beet and maize fields are 1–3 larvae/m
2 in dry areas and 3–5 larvae/m
2 in areas with more rainfall, suggesting that these larval densities can cause the same economic damage in both maize and sugar beet. According to Furlan et al. [
27], no yield reduction is expected in maize when wireworm plant damage is less than 15% of the crop. In contrast, in France, an infestation of 10% of maize plants in a field corresponds to a loss of 500 to 1000 kg/ha [
32]. The occurrence of wireworms in the studied fields as well as the data presented by Čamprag et al. [
33] show that in Croatia and in the neighbouring countries the occurrence of wireworms could be significantly higher compared to north Europe, as presented by Hauer et al. [
15]. According to Hauer et al. [
15] and Furlan and Kreutzweisser [
34] there is less than 10% occurrence of wireworms in sugar beet fields in north Europe and very low occurrence in the Netherlands, Belgium, Germany, Sweden, Denmark and Italy. Furlan et al. [
27] reported that wireworm infestation was less than 15% in 70% of the fields observed over a period of 29 years. However, in more than 10% of the fields, the damage exceeded 40%. Poggi et al. [
28] reported damage above 15% in about half of the fields observed in northern France.
In our experiments, wireworm damage differed in terms of number of plant stands between sites and years, demonstrating that wireworms are serious pests at some sites and in some years. Plant stand on untreated plots was reduced by 43% at the Tovarnik site in 2015 and by 13% at the Lukač site in 2016. The application of insecticides in 2015 resulted in an increase in plant stand of about 11% in Lukač and 69% in Tovarnik. The increase in plant population in 2016 ranged from 22% to 32% in Lukač and from 37% to 55% in Tovarnik. Therefore, insecticide treatments significantly maintained plant stand at both locations and in both years. The obtained results are very similar to those of Kereši et al. [
35,
36] who showed that neonicotinoid seed treatment can ensure plant stand in sugar beet fields.
4.2. Beet Flea Beetle
Before the introduction of neonicotinoids, the beet flea beetle was controlled on 10 to 65% of all sugar beet fields in eastern Croatia. The average consumption of insecticides for beet flea beetle control ranged from 0.1 to 0.59 kg active ingredient/ha of sown sugar beet, depending on the year [
13]. After the introduction of neonicotinoids, all fields were sown with treated seeds, additional treatments against the beet flea beetle were not applied and the amount of insecticides used was significantly lower than in the period before neonicotinoids.
Kereši et al. [
36] reported very severe damage by flea beetles in the experiment under extremely hot and dry weather conditions in Vojvodina, where seed dressing with thiamethoxam resulted in a fourfold increase in seedling weight. However, due to the other factors affecting yield, the increase in yield in the plots treated with thiamethoxam was only 13%. Satisfactory protection of seedlings against beet flea beetle was achieved with thiamethoxam alone or in mixture with tefluthrin and a mixture of imidacloprid + tefluthrin [
35]. These treatments yielded significantly lower percentages of damaged plants than the untreated, while significantly increasing yield. Non-chemical alternatives for beet flea beetle control in sugar beet are not available and the only alternative is foliar spraying with pyrethroids. Therefore, the need to control the pests by spraying with pyrethroids has increased after the ban of neonicotinoids in 2018. In Croatia, we have already observed resistance of the sugar beet flea beetles to pyrethroids (Bažok, unpublished data). This could be one of the reasons why ten EU countries have requested an Emergency Authorisation of imidacloprid, thiamethoxam and clothianidin for seed treatment of sugar beet [
37].
Flea beetle damage observed in both years and locations averaged 44% on untreated plots in BBCH 16 and 52% in BBCH 19. The observed level of damage proves that flea beetles are a serious pest in Croatia, as in other neighbouring countries [
35]. At the same time, [
15] did not report beet flea beetle as a serious pest in north Europe. Although damage by beet flea beetle occurred regularly in our experiments, their intensity varied at different locations and in different years. Both location and year significantly influenced flea beetle infestation.
Seed coating with neonicotinoids resulted in significant damage reduction. In 2015, seed coating with imidacloprid reduced damage by 88 to 97% on Lukač and from 96 to 98% on Tovarnik compared to the untreated control. Slightly lower efficacy was observed on plots treated with thiamethoxam and tefluthrin (from 83 to 94% in Lukač and from 91 to 95% in Tovarnik, respectively). Insecticide efficacy was lower in 2016. Seed coating with imidacloprid reduced damage by 68 to 73% in Lukač and by 36 to 46% in Tovarnik. At the same time, the effectiveness of the combination of thiamethoxam and tefluthrin ranged from 74 to 77% in Lukač and from 43 to 46% in Tovarnik, respectively.
4.3. Sugar Beet Weevil
From 1965 until the early 2000s, the sugar beet weevil was not an important pest in Croatia. It was important in Serbia, in the region of Vojvodina, which borders eastern Croatia [
33]. As Čamprag [
9] stated, this species is the most important pest of sugar beet in Vojvodina. In the last 60 years, it has destroyed a total of more than 250,000 hectares of young sugar beet and caused reseeding of stands. Between 1975 and 2004, an average of 3.3 individuals per square meter was counted. In eastern Croatia (on the border with Vojvo-dina), the population of the pest was below the economic threshold until 2008 [
13]. After that, the population of the sugar beet weevil increased significantly and was regularly very high, causing severe damage [
6]. Increased occurrence of sugar beet weevil in Croatia, Ukraine and Vojvodina is associated with global climate change and increased temperatures [
13,
38,
39]. In Poland, Austria, Hungary and some eastern European countries, the sugar beet weevil is in a stage of downgrading and causes economically significant damage [
40]. The reason for the increase in abundance can also be found in the combination of favorable climatic conditions (hot and dry spring) with the prohibition of effective insecticides [
40].
In the eastern part of Croatia, the sugar beet weevil occurs regularly [
41]. At the same time, we did not expect its occurrence at the Lukač site. Weather conditions contributed to the low abundance of the pest in 2015 at both locations. However, the abundance of the pest in 2016 was high in Tovarnik, with plant damage on untreated plots of about 20% and significantly higher than in Lukač, where plant damage on untreated plots was up to 12%. Under these conditions, insecticide treatments significantly reduced plant damage. Seed treatments achieved satisfactory results in protecting sugar beet at the most sensitive stages of development under the condition of low weevil infestation.
4.4. Caterpillars
The surface-feeding species
Mamestra brassicae L,
Lacanobia oleracea L. and
Autographa gamma L. are among the most damaging Noctuidae pests in sugar beet in Croatia. They have the potential to remove much (or all) of the aboveground foliage from young plants and dramatically affect plant growth and development [
42]. The first appearance of the caterpillars is usually in June, two to three months after sowing. Due to the long period between sowing and the appearance of the pest, these pests are usually not controlled by seed dressing with neonicotinoids. In our experiments, significantly higher infestation was recorded in both years on Lukač, which is characterized by higher precipitation, confirming the results of Bažok et al. [
43] on the influence of weather conditions on moth occurrence and damage. They reported the decrease in caterpillar damage caused by a very warm and dry growing season. As expected and reported by other authors [
15], neonicotinoid seed coating did not significantly reduce damage. Due to their occurrence in the middle of the growing season, noctuid and moth caterpillars should be controlled by foliar application of insecticides.
4.5. Aphids
Aphids damage the crop mainly by sucking, resulting in reduced assimilate availability for plant growth and leaf area production [
15]. They can also transmit Virus Yellows [
44], which can cause significant damage in some countries of southern and eastern Europe [
10,
14], while in northern Europe, according to Kozlowska-Makulska et al. [
45], transmission of the virus does not play an important role in the spread of Virus Yellows in sugar beet.
Significant infestation of aphids was not detected in the experiment. Based on the results of other authors [
14,
46], we expected a high efficacy of seed coatings with neonicotinoids against aphids. Although the percentage of damage was very low, better efficacy of imidacloprid was observed in 2016 compared to thiamethoxam and untreated variants (
Table 9). However, our results do not provide a solid basis for conclusions on the efficacy of neonicotinoid seed coating against aphids.
5. Conclusions
In our trials, imidacloprid and thiamethoxam seed coatings provided satisfactory protection of young sugar beet plants against wireworms, sugar beet flea beetle, and, at low infestations, sugar beet weevil. These pests occur regularly in southern and eastern Europe and therefore require control measures.
Although there are many reports of high efficacy of neonicotinoid seed treatments against aphids, we could not draw any conclusions due to the low infestation of aphids in both trials. There are alternatives for the control of wireworms, sugar beet weevils, caterpillars and aphids. However, they should be further investigated as the application rate is not very high.
Further research program is needed to find alternative solutions and develop easily implementable strategies for all sugar beet pests. Based on the results obtained, we would propose an authorization of neonicotinoids for seed treatment of sugar beet in the regions with high infestation of the main sugar beet pests.