Postharvest Storage Techniques and Quality Evaluation of Fruits and Vegetables for Reducing Food Loss
Conflicts of Interest
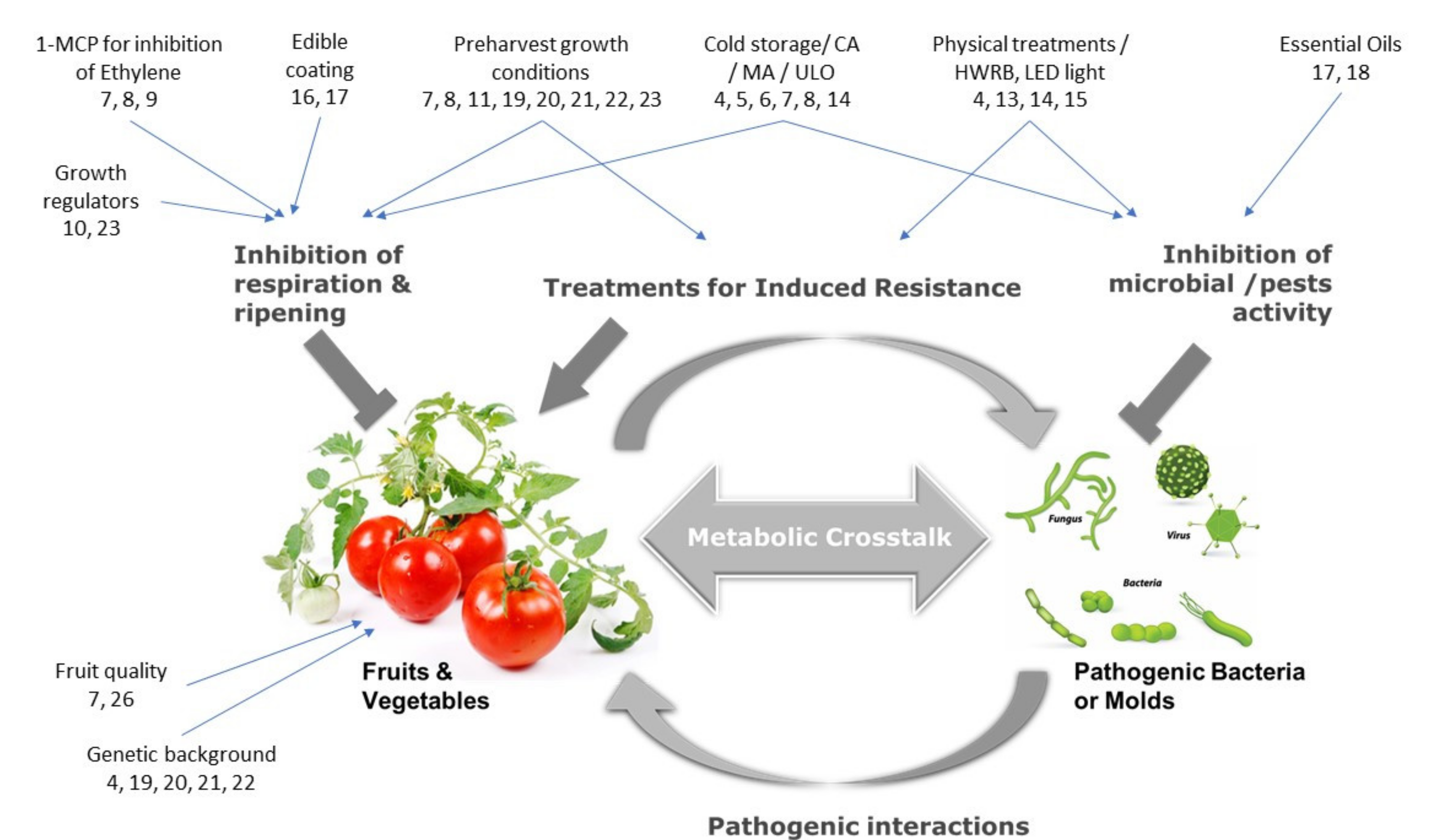
References
- Kyriacou, M.C.; Rouphael, Y. Towards a new definition of quality for fresh fruits and vegetables. Sci. Hortic. 2018, 234, 463–469. [Google Scholar] [CrossRef]
- FAO. Global Food Losses and Food Waste–Extent, Causes and Prevention; FAO: Rome, Italy, 2011. [Google Scholar]
- Buzby, J.C.; Wells, H.F.; Hyman, J. The Estimated Amount, Value, and Calories of Postharvest Food Losses at the Retail and Consumer Levels in the United States; EIB-121; U.S. Department of Agriculture, Economic Research Service: Washington, DC, USA, 2014.
- Adeeko, A.; Yudelevich, F.; Raphael, G.; Avraham, L.; Alon, H.; Presman, M.Z.; Alkalai-Tuvia, S.; Paris, H.S.; Fallik, E.; Ziv, C. Quality and Storability of Trellised Greenhouse-Grown, Winter-Harvested, New Sweet Acorn Squash Hybrids. Agronomy 2020, 10, 1443. [Google Scholar] [CrossRef]
- Paulauskienė, A.; Tarasevičienė, Ž.; Žebrauskienė, A.; Pranckietienė, I. Effect of Controlled Atmosphere Storage Conditions on the Chemical Composition of Super Hardy Kiwifruit. Agronomy 2020, 10, 822. [Google Scholar] [CrossRef]
- Korićanac, A.; Miletić, N.; Popović, B.; Mitrović, O.; Lukić, M.; Pešaković, M.; Tomić, J. The Effect of ULO and NA Storage on Changes in the Quality of Apple Fruit (Malus domestica Borkh.) during Shelf Life. Agronomy 2020, 10, 25. [Google Scholar] [CrossRef]
- Dällenbach, L.J.; Eppler, T.; Bühlmann-Schütz, S.; Kellerhals, M.; Bühlmann, A. Pre- and Postharvest Factors Control the Disease Incidence of Superficial Scald in the New Fire Blight Tolerant Apple Variety “Ladina”. Agronomy 2020, 10, 464. [Google Scholar] [CrossRef]
- Jung, S.-K.; Choi, H.-S. Browning of Early and Late-Harvested ‘Empire’ Apples Affected by Cold Storage and 1-MCP. Agronomy 2020, 10, 1050. [Google Scholar] [CrossRef]
- Gamrasni, D.; Feldmesser, E.; Ben-Arie, R.; Raz, A.; Tabatznik Asiag, A.; Glikman, M.; Aharoni, A.; Goldway, M. Gene Expression in 1-Methylcyclopropene (1-MCP) Treated Tomatoes during Pre-Climacteric Ripening Suggests Shared Regulation of Methionine Biosynthesis, Ethylene Production and Respiration. Agronomy 2020, 10, 1669. [Google Scholar] [CrossRef]
- Ennab, H.A.; El-Shemy, M.A.; Alam-Eldein, S.M. Salicylic Acid and Putrescine to Reduce Post-Harvest Storage Problems and Maintain Quality of Murcott Mandarin Fruit. Agronomy 2020, 10, 115. [Google Scholar] [CrossRef]
- Shimshoni, J.A.; Bommuraj, V.; Chena, Y.; Sperling, R.; Barel, S.; Kaye, Y.; Fallik, E. Residual Distribution Kinetics of Pesticides in Cherry Tomato Peel, Pulp, and Fruit as a Function of Irrigation Water Salinity, Household Rinsing, and Storage Regimen. Agronomy 2019, 9, 800. [Google Scholar] [CrossRef]
- Munhuweyi, K.; Mpai, S.; Sivakumar, D. Extension of Avocado Fruit Postharvest Quality Using Non-Chemical Treatments. Agronomy 2020, 10, 212. [Google Scholar] [CrossRef]
- Mpai, S.; Sivakumar, D. Stimulation of Light-Emitting Diode Treatment on Defence System and Changes in Mesocarp Metabolites of Avocados Cultivars (Hass and Fuerte) during Simulated Market Shelf Conditions. Agronomy 2020, 10, 1654. [Google Scholar] [CrossRef]
- Lama, K.; Alkalai-Tuvia, S.; Chalupowicz, D.; Fallik, E. Extended Storage of Yellow Pepper Fruits at Suboptimal Temperatures May Alter Their Physical and Nutritional Quality. Agronomy 2020, 10, 1109. [Google Scholar] [CrossRef]
- Wang, Y.; Naber, M.R.; Crosby, T.W. Effects of Wound-Healing Management on Potato Post-Harvest Storability. Agronomy 2020, 10, 512. [Google Scholar] [CrossRef]
- Fawole, O.A.; Riva, S.C.; Opara, U.L. Efficacy of Edible Coatings in Alleviating Shrivel and Maintaining Quality of Japanese Plum (Prunus salicina Lindl.) during Export and Shelf Life Conditions. Agronomy 2020, 10, 1023. [Google Scholar] [CrossRef]
- Tzortzakis, N.; Xylia, P.; Chrysargyris, A. Sage Essential Oil Improves the Effectiveness of Aloe vera Gel on Postharvest Quality of Tomato Fruit. Agronomy 2019, 9, 635. [Google Scholar] [CrossRef]
- Shehata, S.A.; Abdeldaym, E.A.; Ali, M.R.; Mohamed, R.M.; Bob, R.I.; Abdelgawad, K.F. Effect of Some Citrus Essential Oils on Post-Harvest Shelf Life and Physicochemical Quality of Strawberries during Cold Storage. Agronomy 2020, 10, 1466. [Google Scholar] [CrossRef]
- Conversa, G.; Lazzizera, C.; Bonasia, A.; Elia, A. Harvest Season and Genotype Affect Head Quality and Shelf-Life of Ready-to-Use Broccoli. Agronomy 2020, 10, 527. [Google Scholar] [CrossRef]
- Koukounaras, A.; Bantis, F.; Karatolos, N.; Melissas, C.; Vezyroglou, A. Influence of Pre-Harvest Factors on Postharvest Quality of Fresh-Cut and Baby Leafy Vegetables. Agronomy 2020, 10, 172. [Google Scholar] [CrossRef]
- Farina, V.; Gentile, C.; Sortino, G.; Gianguzzi, G.; Palazzolo, E.; Mazzaglia, A. Tree-Ripe Mango Fruit: Physicochemical Characterization, Antioxidant Properties and Sensory Profile of Six Mediterranean-Grown Cultivars. Agronomy 2020, 10, 884. [Google Scholar] [CrossRef]
- Fallik, E.; Alkalai-Tuvia, S.; Chalupowicz, D.; Popovsky-Sarid, S.; Zaaroor-Presman, M. Relationships between Rootstock-Scion Combinations and Growing Regions on Watermelon Fruit Quality. Agronomy 2019, 9, 536. [Google Scholar] [CrossRef]
- Kumar, S.P.; Maurer, D.; Feygenberg, O.; Love, C.; Alkan, N. Improving the Red Color and Fruit Quality of ‘Kent’ Mango Fruit by Pruning and Preharvest Spraying of Prohydrojasmon or Abscisic Acid. Agronomy 2020, 10, 944. [Google Scholar] [CrossRef]
- Fallik, E.; Ziv, C. How rootstock/scion combinations affect watermelon fruit quality after harvest? J. Sci. Food Agric. 2020, 100, 3275–3282. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Barman, K.; Siddiqui, M.W.; Nath, V. Chapter 10-Training and Pruning for Improved Postharvest Fruit Quality. In Preharvest Modulation of Postharvest Fruit and Vegetable Quality; Siddiqui, M.W., Ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 257–276. [Google Scholar] [CrossRef]
- Vázquez-Espinosa, M.; González-de-Peredo, A.V.; Espada-Bellido, E.; Ferreiro-González, M.; Toledo-Domínguez, J.J.; Carrera, C.; Palma, M.; Barbero, G.F. Ultrasound-Assisted Extraction of Two Types of Antioxidant Compounds (TPC and TA) from Black Chokeberry (Aronia melanocarpa L.): Optimization of the Individual and Simultaneous Extraction Methods. Agronomy 2019, 9, 456. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ziv, C.; Fallik, E. Postharvest Storage Techniques and Quality Evaluation of Fruits and Vegetables for Reducing Food Loss. Agronomy 2021, 11, 1133. https://doi.org/10.3390/agronomy11061133
Ziv C, Fallik E. Postharvest Storage Techniques and Quality Evaluation of Fruits and Vegetables for Reducing Food Loss. Agronomy. 2021; 11(6):1133. https://doi.org/10.3390/agronomy11061133
Chicago/Turabian StyleZiv, Carmit, and Elazar Fallik. 2021. "Postharvest Storage Techniques and Quality Evaluation of Fruits and Vegetables for Reducing Food Loss" Agronomy 11, no. 6: 1133. https://doi.org/10.3390/agronomy11061133
APA StyleZiv, C., & Fallik, E. (2021). Postharvest Storage Techniques and Quality Evaluation of Fruits and Vegetables for Reducing Food Loss. Agronomy, 11(6), 1133. https://doi.org/10.3390/agronomy11061133





