Organic Phosphorus Substantially Contributes to Crop Plant Nutrition in Soils with Low Phosphorus Availability
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil and Rhizobox System
2.2. Soil Chemical Analyses
2.3. Soil Microbial Analyses
3. Results and Discussion
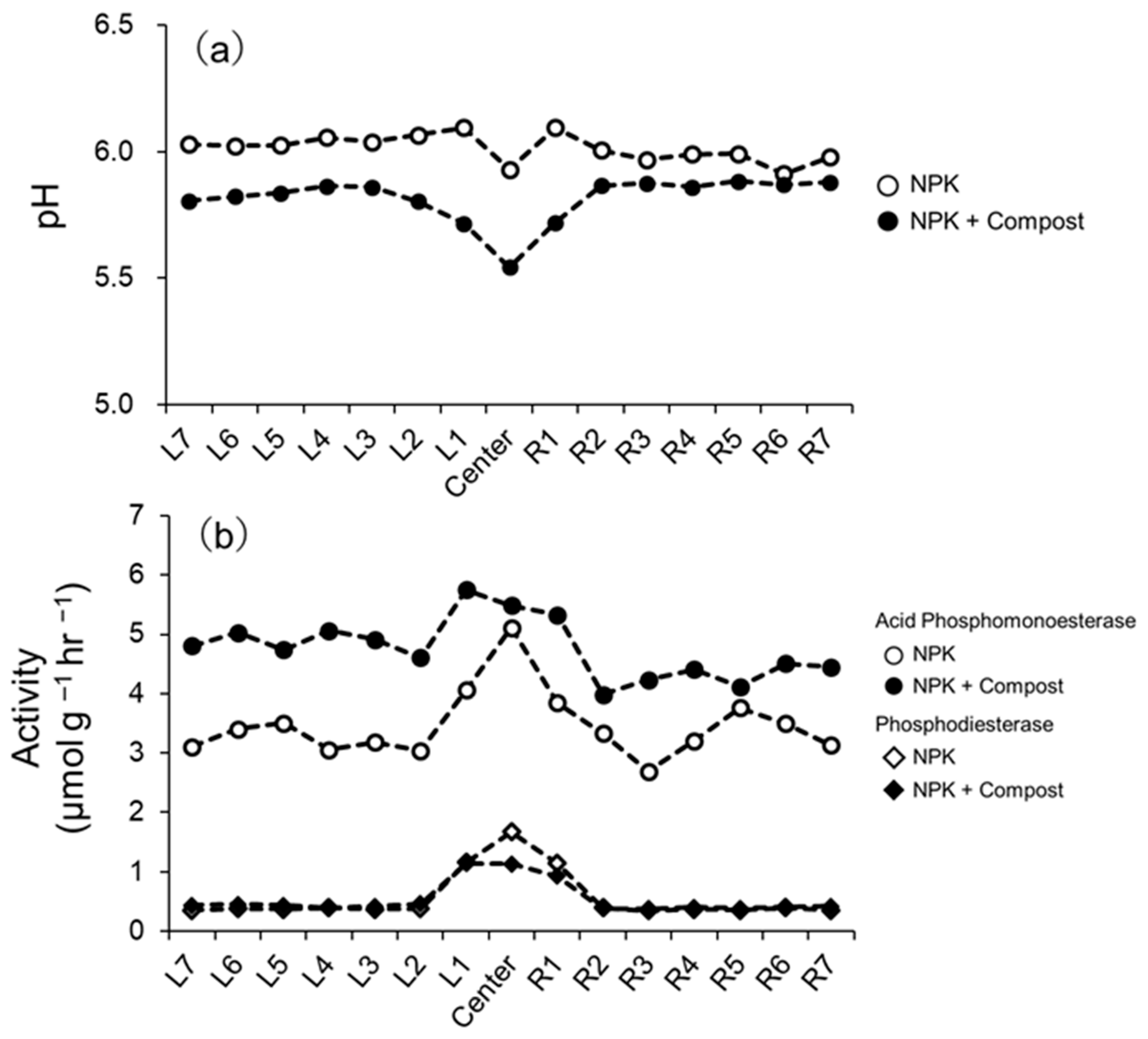
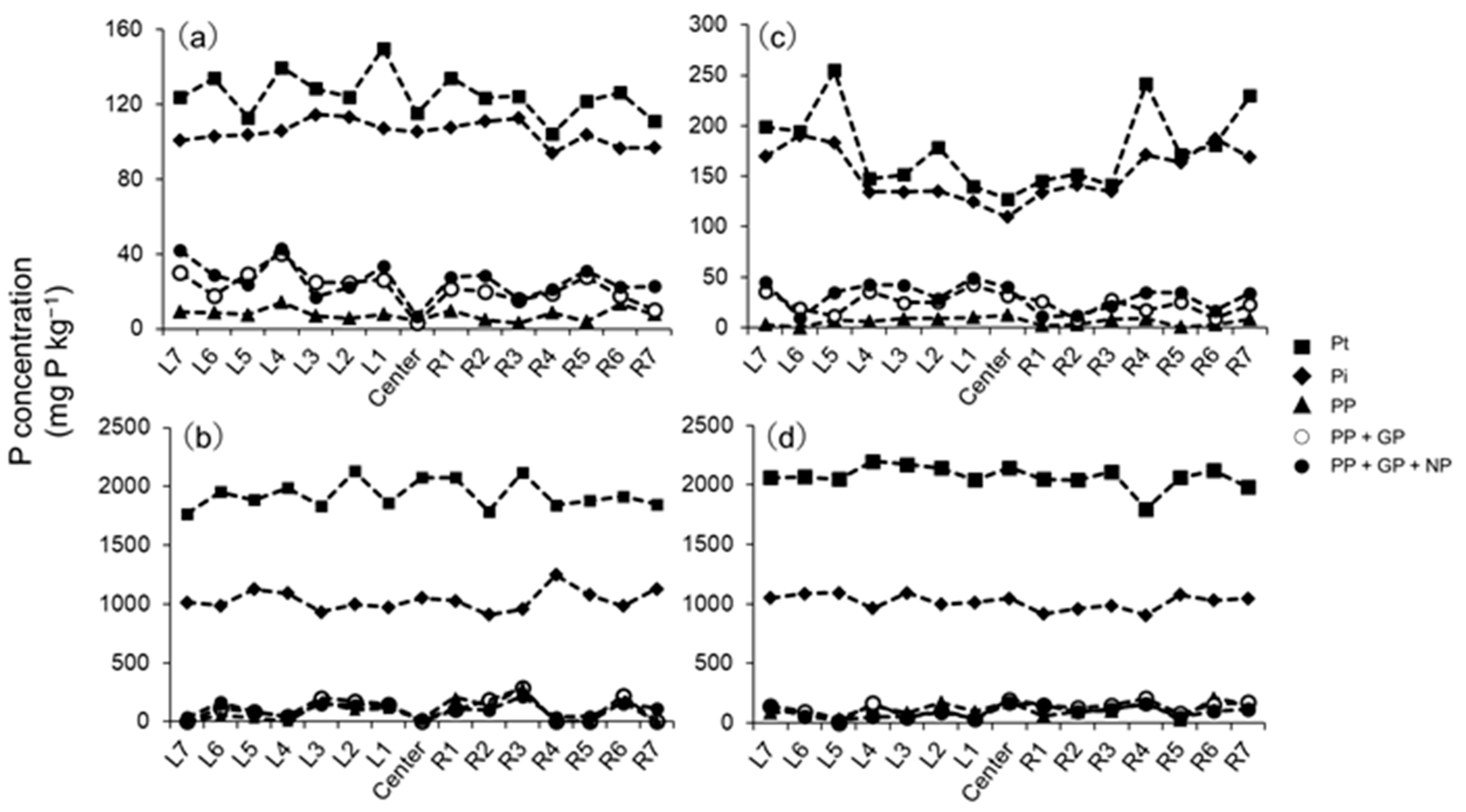
3.1. Phosphorus Fractions and Phosphatase-Labile Pool of Po
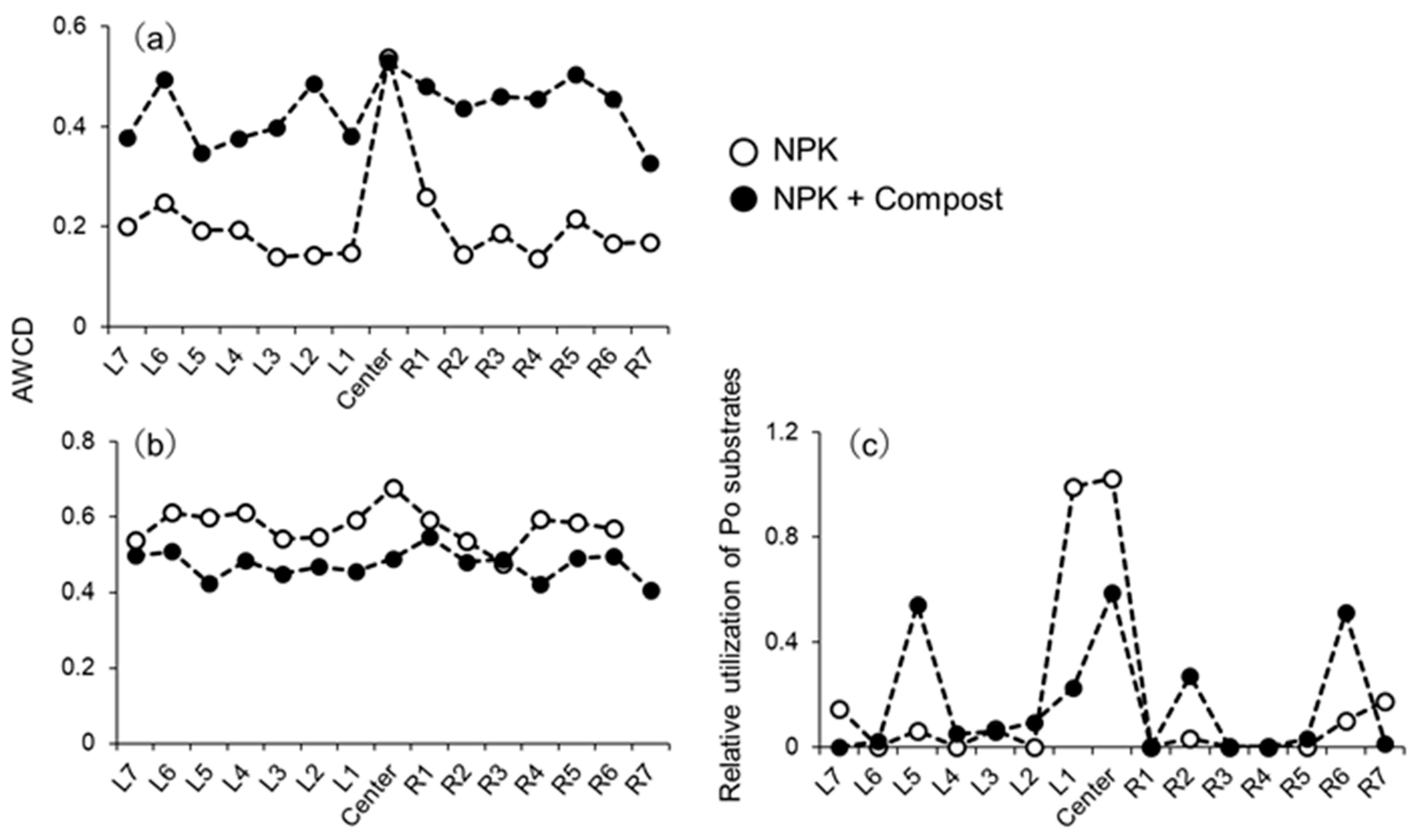
3.2. Phosphatase Activities and Substrate Utilization Profiles of Microbial Communities in the Rhizosphere
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Borie, F.; Zunino, H. Organic matter-phosphorus association as a sink in P-fixation processes in allophanic soils of Chile. Soil Biol. Biochem. 1983, 15, 599–603. [Google Scholar] [CrossRef]
- Moro, H.; Kunito, T.; Sato, T. Assessment of phosphorus bioavailability in cultivated Andisols from a long-term fertilization field experiment using chemical extractions and soil enzyme activities. Arch. Agron. Soil Sci. 2015, 61, 1107–1123. [Google Scholar] [CrossRef]
- Zubillaga, M.S.; Giuffré, L. Soil phosphorus mobilization in different taxonomic orders. J. Plant Nutr. Soil Sci. 1999, 162, 201–205. [Google Scholar] [CrossRef]
- Kunito, T.; Haraguchi, S.; Hanada, K.; Fujita, K.; Moro, H.; Nagaoka, K.; Otsuka, S. pH is the dominant factor controlling the levels of phytate-like and DNA-like phosphorus in 0.5M NaHCO3-extracts of soils: Evaluation with phosphatase-addition approach. Geoderma 2021, 398, 115113. [Google Scholar] [CrossRef]
- Tarafdar, J.C.; Claassen, N. Organic phosphorus compounds as a phosphorus source for higher plants through the activity of phosphatases produced by plant roots and microorganisms. Biol. Fertil. Soils 1988, 5, 308–312. [Google Scholar] [CrossRef]
- Nannipieri, P.; Giagnoni, L.; Landi, L.; Renella, G. Role of phosphatase enzymes in soil. In Phosphorus in Action: Biological Processes in Soil Phosphorus Cycling (Soil Biology 26); Bünemann, E.K., Oberson, A., Frossard, E., Eds.; Springer: Berlin, Germany, 2011; pp. 215–243. [Google Scholar]
- Spohn, M.; Treichel, N.S.; Cormann, M.; Schloter, M.; Fischer, D. Distribution of phosphatase activity and various bacterial phyla in the rhizosphere of Hordeum vulgare L. depending on P availability. Soil Biol. Biochem. 2015, 89, 44–51. [Google Scholar] [CrossRef]
- Tarafdar, J.C.; Jungk, A. Phosphatase activity in the rhizosphere and its relation to the depletion of soil organic phosphorus. Biol. Fertil. Soils 1987, 3, 199–204. [Google Scholar] [CrossRef]
- George, T.S.; Turner, B.L.; Gregory, P.J.; Cade-Menun, B.J.; Richardson, A.E. Depletion of organic phosphorus from Oxisols in relation to phosphatase activities in the rhizosphere. Eur. J. Soil Sci. 2006, 57, 47–57. [Google Scholar] [CrossRef]
- Waldrip, H.M.; He, Z.; Erich, M.S. Effects of poultry manure amendment on phosphorus uptake by ryegrass, soil phosphorus fractions and phosphatase activity. Biol. Fertil. Soils 2011, 47, 407–418. [Google Scholar] [CrossRef]
- Youssef, R.A.; Chino, M. Development of a new rhizobox system to study the nutrient status in the rhizosphere. Soil Sci. Plant Nutr. 1988, 34, 461–465. [Google Scholar] [CrossRef]
- Sumi, H.; Kunito, T.; Ishikawa, Y.; Sato, T.; Park, H.D.; Nagaoka, K.; Aikawa, Y. Plant roots influence microbial activities as well as cadmium and zinc fractions in metal-contaminated soil. Chem. Ecol. 2015, 31, 105–110. [Google Scholar] [CrossRef]
- Kitson, R.E.; Mellon, M.G. Colorimetric determination of phosphorus as molybdivanadophosphoric acid. Ind. Eng. Chem. Anal. Ed. 1944, 16, 379–383. [Google Scholar]
- He, Z.; Griffin, T.S.; Honeycutt, C.W. Enzymatic hydrolysis of organic phosphorus in swine manure and soil. J. Environ. Qual. 2004, 33, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar]
- Tabatabai, M.A. Soil enzymes. In Methods of Soil Analysis, Part 2, Microbiological and Biochemical Properties; Weaver, R.W., Angle, S., Bottomley, P., Bezdicek, D., Smith, S., Tabatabai, A., Wollum, A., Eds.; Soil Science Society of America: Madison, WI, USA, 1994; pp. 775–833. [Google Scholar]
- Classen, A.T.; Boyle, S.I.; Haskins, K.E.; Overby, S.T.; Hart, S.C. Community-level physiological profiles of bacteria and fungi: Plate type and incubation temperature influences on contrasting soils. FEMS Microbiol. Ecol. 2003, 44, 319–328. [Google Scholar] [CrossRef]
- Kunito, T.; Nagaoka, K. Effects of plant litter type and additions of nitrogen and phosphorus on bacterial community-level physiological profiles in a brown forest soil. Microbes Environ. 2009, 24, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Turner, B.L.; Papházy, M.J.; Haygarth, P.M.; McKelvie, I.D. Inositol phosphates in the environment. Phil. Trans. R. Soc. Lond. B 2002, 357, 449–469. [Google Scholar]
- Menezes-Blackburn, D.; Jorquera, M.A.; Greiner, R.; Gianfreda, L.; Mora, M.L. Phytases and phytase-labile organic phosphorus in manures and soils. Crit. Rev. Environ. Sci. Technol. 2013, 43, 916–954. [Google Scholar] [CrossRef]
- Richardson, A.E.; Hadobas, P.A.; Hayes, J.E.; O’Hara, C.P.; Simpson, R.J. Utilization of phosphorus by pasture plants supplied with myo-inositol hexaphosphate is enhanced by the presence of soil micro-organisms. Plant Soil 2001, 229, 47–56. [Google Scholar] [CrossRef]
- Greiner, R. Phytate-degrading enzymes: Regulation of synthesis in microorganisms and plants. In Inositol Phosphates: Linking Agriculture and the Environment; Turner, B.L., Richardson, A.E., Mullaney, E.J., Eds.; CAB International: Wallingford, UK, 2007; pp. 78–96. [Google Scholar]
- Ye, D.; Zhang, X.; Li, T.; Xu, J.; Chen, G. Phosphorus-acquisition characteristics and rhizosphere properties of wild barley in relation to genotypic differences as dependent on soil phosphorus availability. Plant Soil 2018, 423, 503–516. [Google Scholar]
- Sharpley, A.N. Phosphorus cycling in unfertilized and fertilized agricultural soils. Soil Sci. Soc. Am. J. 1985, 49, 905–911. [Google Scholar] [CrossRef]
- Kunito, T.; Tobitani, T.; Moro, H.; Toda, H. Phosphorus limitation in microorganisms leads to high phosphomonoesterase activity in acid forest soils. Pedobiologia 2012, 55, 263–270. [Google Scholar] [CrossRef]
- Fujita, K.; Kunito, T.; Moro, H.; Toda, H.; Otsuka, S.; Nagaoka, K. Microbial resource allocation for phosphatase synthesis reflects the availability of inorganic phosphorus across various soils. Biogeochemistry 2017, 136, 325–339. [Google Scholar] [CrossRef]
- Mise, K.; Maruyama, R.; Miyabara, Y.; Kunito, T.; Senoo, K.; Otsuka, S. Time-series analysis of phosphorus-depleted microbial communities in carbon/nitrogen-amended soils. Appl. Soil Ecol. 2020, 145, 103346. [Google Scholar] [CrossRef]
| NPK Soil | NPK + Compost Soil | |
|---|---|---|
| pH | 6.2 | 6.3 |
| Total C (g kg−1) | 34.8 | 43.4 |
| Total N (g kg−1) | 2.6 | 3.1 |
| Total P (mg kg−1) | 3390 | 3160 |
| Shoot | Root | |||||
|---|---|---|---|---|---|---|
| Dry weight | P concentration | P uptake | Dry weight | P concentration | P uptake | |
| (g) | (µg g−1) | (µg) | (g) | (µg g−1) | (µg) | |
| NPK soil | 0.60 | 652 | 391 | 1.12 | 837 | 937 |
| NPK + compost soil | 1.14 | 566 | 645 | 1.69 | 824 | 1393 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moro, H.; Park, H.-D.; Kunito, T. Organic Phosphorus Substantially Contributes to Crop Plant Nutrition in Soils with Low Phosphorus Availability. Agronomy 2021, 11, 903. https://doi.org/10.3390/agronomy11050903
Moro H, Park H-D, Kunito T. Organic Phosphorus Substantially Contributes to Crop Plant Nutrition in Soils with Low Phosphorus Availability. Agronomy. 2021; 11(5):903. https://doi.org/10.3390/agronomy11050903
Chicago/Turabian StyleMoro, Hitoshi, Ho-Dong Park, and Takashi Kunito. 2021. "Organic Phosphorus Substantially Contributes to Crop Plant Nutrition in Soils with Low Phosphorus Availability" Agronomy 11, no. 5: 903. https://doi.org/10.3390/agronomy11050903
APA StyleMoro, H., Park, H.-D., & Kunito, T. (2021). Organic Phosphorus Substantially Contributes to Crop Plant Nutrition in Soils with Low Phosphorus Availability. Agronomy, 11(5), 903. https://doi.org/10.3390/agronomy11050903






