Cattle Horn Shavings: A Possible Nitrogen Source for Apple Trees
Abstract
1. Introduction
2. Materials and Methods
- No fertilisers
- 50 kg/ha N equivalent applied in spring as NH4NO3
- 100 kg/ha N equivalent applied in spring as NH4NO3
- 50 kg/ha N equivalent applied in spring as horn shavings
- 100 kg/ha N equivalent applied in spring as horn shavings
- 50 + 50 N equivalent applied at equal parts in autumn and spring as horn shavings
- 50 kg/ha N equivalent applied in autumn as horn shavings
- 100 kg/ha N equivalent applied in autumn as horn shavings
3. Results
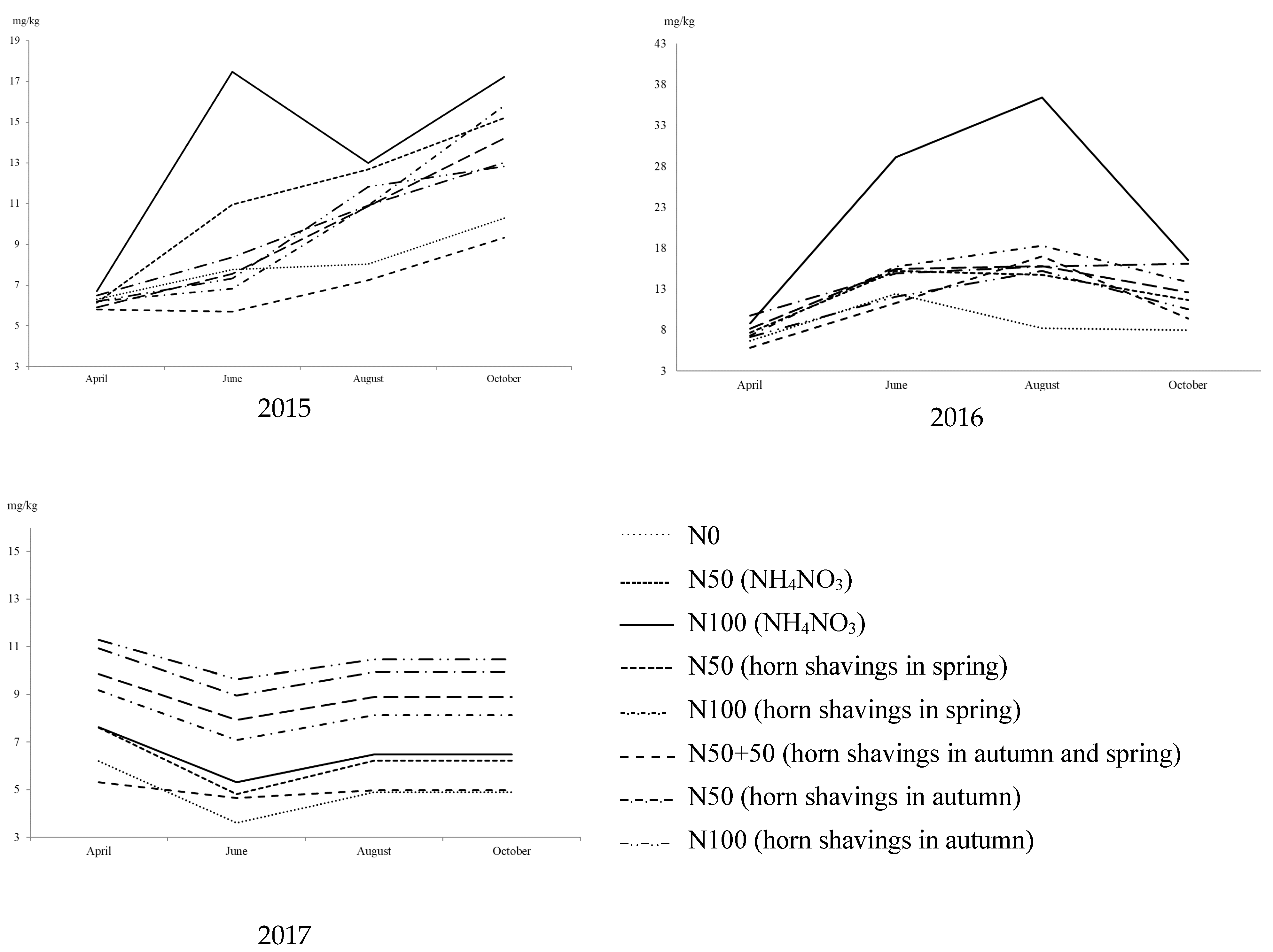
3.1. Soil Mineral Nitrogen
3.2. Leaf Nitrogen Content
3.3. Apple Tree Productivity
3.4. Fruit Quality
4. Discussion
4.1. Soil Mineral Nitrogen
4.2. Leaf Nitrogen Content
4.3. Apple Tree Productivity
4.4. Fruit Quality
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Xia, G.; Cheng, L.; Lakso, A.; Goffinet, M. Effects of nitrogen supply on source-sink balance and fruit size of ‘Gala Apple’ trees. J. Am. Soc. Hortic. Sci. 2009, 134, 126–133. [Google Scholar] [CrossRef]
- Komamura, K.; Suzuki, A.; Fukumoto, M.; Kato, K.; Sato, Y. Effects of Long-Term Nitrogen Application on Tree Growth, Yield, and Fruit Qualities in a ‘Jonathan’ Apple Orchard. J. Jpn. Soc. Hortic. Sci. 2000, 69, 617–623. [Google Scholar] [CrossRef]
- Ernani, P.R.; Rogeri, D.A.; Proença, M.M.; Dias, J. Addition of nitrogen had no effect on yield and quality of apples in a high density orchard carrying a dwarf rootstock. Rev. Bras. Frutic. 2008, 30, 1113–1118. [Google Scholar] [CrossRef]
- Wrona, D. The influence of nitrogen fertilization on growth, yield and fruit size of ‘Jonagored’ apple trees. Acta Sci. Pol. Hortorum Cultus. 2011, 10, 3–10. [Google Scholar]
- Kowalczyk, W.; Wrona, D.; Przybyłko, S. Content of minerals in soil, apple tree leaves and fruits depending on nitrogen fertilization. J. Elem. 2017, 22, 67–77. [Google Scholar] [CrossRef]
- Rütting, T.; Aronsson, H.; Delin, S. Efficient use of nitrogen in agriculture. Nutr. Cycl. Agroecosyst. 2018, 110, 1–5. [Google Scholar] [CrossRef]
- Palmer, J.W.; Wünsche, N. Organic Systems of Apple Production Bring New Horizons to Traditional Crop Physiology. Acta Hort. 2004, 638, 489–496. [Google Scholar] [CrossRef]
- Mays, N.; Brye, K.R.; Rom, C.R.; Savin, M.; Garcia, M.E. Groundcover Management and Nutrient Source Effects on Soil Carbon and Nitrogen Sequestration in an Organically Managed Apple Orchard in the Ozark Highlands. HortScience 2014, 49, 637–644. [Google Scholar] [CrossRef]
- Bhattacharya, A. Nitrogen-Use Efficiency under Changing Climatic Conditions. In Changing Climate and Resource Use Efficiency in Plants; Bhattacharya, A., Ed.; Academic Press: London, UK, 2019; pp. 181–240. [Google Scholar]
- Pekarskas, J.; Žibutis, S.; Gražulevičienė, V.; Grigalavičienė, I.; Mažeika, R. Cattle Horn Shavings as Slow Release Nitrogen Fertilizer. Pol. J. Environ. Stud. 2015, 24, 645–650. [Google Scholar]
- Veselá, M.; Friedrich, J. Amino Acid and Soluble Protein Cocktail from Waste Keratin Hydrolysed by a Fungal Keratinase of Paecilomyces marquandii. Biotechnol. Bioprocess Eng. 2009, 14, 84–90. [Google Scholar] [CrossRef]
- European Commission. COMMISSION REGULATION (EC) No 889/2008 of 5 September 2008 Laying Down Detailed Rules for the Implementation of Council Regulation (EC) No 834/2007 on Organic Production and Labelling of Organic Products with Regard to Organic Production, Labelling and Control; European Commission: Brussels, Belgium, 2008. [Google Scholar]
- Juroszek, P.; Drews, S.; Neuhoff, D.; Köpke, U. Effects of organic fertilisers on the development of weeds and winter wheat. J. Plant Dis. Prot. 2004, 19, 611–618. [Google Scholar]
- Möller, K.; Schultheiß, U. Organische Handelsdüngemittel Tierischer und Pflanzlicher Herkunft für den Ökologischen Landbau-Charakterisierung und Empfehlungen für die Praxis; Kuratorium für Technik und Bauwesen in der Landwirtschaft: Darmstadt, Germany, 2013; pp. 1–63. [Google Scholar]
- Jayathilakan, K.; Sultana, K.; Radhakrishna, K.; Bawa, A.S. Utilization of byproducts and waste materials from meat, poultry and fish processing industries: A review. J. Food Sci. Technol. 2012, 49, 278–293. [Google Scholar] [CrossRef]
- Meier, U. Growth Stages of Mono-and Dicotyledonous Plants. In BBCH Monograph; Open Agrar Repositorium: Quedlinburg, German, 2018; p. 204. [Google Scholar]
- Sadar, N.; Zanella, A.A. Study on the Potential of IAD as a Surrogate Index of Quality and Storability in cv. ‘Gala’ Apple Fruit. Agronomy 2019, 9, 642. [Google Scholar] [CrossRef]
- Žibutis, S.; Pekarskas, J.; Česonienė, L. Effect of horn shaving and horn core powder fertilizers on the dynamics of mineral nitrogen in the soil of organic farm. Ekologija 2012, 58, 375–390. [Google Scholar] [CrossRef]
- Agehara, S.; Warncke, D.D. Soil Moisture and Temperature Effects on Nitrogen Release from Organic Nitrogen Sources. Soil Sci. Soc. Am. J. 2005, 69, 1844–1855. [Google Scholar] [CrossRef]
- Gan, L. Simulation of Soil Nitrogen Mineralization by Temperature and Water in a Typical Period of Forest Succession in the Central Subtropical Zone. In Proceedings of the 5th International Conference on Mechatronics, Materials, Chemistry and Computer Engineering (ICMMCCE 2017), Advances in Engineering Research, Chongqing, China, 24–25 July 2017; Wang, M., Zhou, X., Eds.; Atlantis Press: Paris, France, 2017; Volume 141, pp. 184–189. [Google Scholar]
- Mažuolytė-Miškinė, E.; Grigalavičienė, I.; Gražulevičienė, V. Biodegradation of cattle horn shavings in soil and its effect on the agrochemical properties of soil. Environ. Prot. Eng. 2014, 6, 368–372. [Google Scholar]
- Cheng, L.; Raba, R. Accumulation of Macro- and Micronutrients and Nitrogen Demand-supply Relationship of ‘Gala’/‘Malling 26′ Apple Trees Grown in Sand Culture. J. Am. Soc. Hort. Sci. 2009, 134, 3–13. [Google Scholar] [CrossRef]
- Carranca, C.; Brunetto, G.; Tagliavini, M. Nitrogen Nutrition of Fruit Trees to Reconcile Productivity and Environmental Concerns. Plants 2018, 7, 4. [Google Scholar] [CrossRef]
- Deckers, T.; Schoofs, H.; Daemen, E.; Missotten, C.; Hähndel, R. Effect of long term soil and leaf nitrogen applications to apple cv. Jonagold and Boskoop on Nmin in the soil and on leaf and fruit quality. Acta Hortic. 2001, 564, 269–278. [Google Scholar] [CrossRef]
- Stiles, W.C. Nitrogen Management in the Orchard. In Tree Fruit Nutrition; Peterson, A.B., Stevens, R.G., Eds.; Good Fruit Grower: Yakima, WA, USA, 1994; pp. 41–50. [Google Scholar]
- Wargo, M.J.; Merwin, I.; Watkins, C. Fruit Size, Yield, and Market Value of ‘GoldRush’ Apple are Affected by Amount, Timing and Method of Nitrogen Fertilization. HortTechnology 2003, 13, 153–161. [Google Scholar] [CrossRef]
- Treder, W. Crop loading studies with ‘Jonagold’ apple tree. J. Fruit Ornam. Plant Res. 2010, 18, 59–69. [Google Scholar]
- Racsko, J.; Szabó, Z.; Nyéki, J. Effect of nutrient supply on fruit quality of apple (Malus domestica Borkh.). J. Cent. Eur. Agric. 2005, 6, 35–42. [Google Scholar] [CrossRef]
- Nava, G.; Dechen, A.R.; Nachtigall, G.R. Nitrogen and Potassium Fertilization Affect Apple Fruit Quality in Southern Brazil. Commun. Soil Sci. Plant Anal. 2007, 39, 96–107. [Google Scholar] [CrossRef]
- Jivan, C.; Sala, F. Relationship between tree nutritional status and apple quality. Hort. Sci. 2014, 41, 1–9. [Google Scholar] [CrossRef]
- Nyasordzi, J.; Friedman, H.; Schmilovitch, Z.; Ignat, T.; Weksler, A.; Rot, I.; Lurie, S. Utilizing the IAD index to determine internal quality attributes of apples at harvest and after storage. Postharvest Biol. Technol. 2013, 77, 80–86. [Google Scholar] [CrossRef]
- Farneti, B.; Gutierrez, M.S.; Novak, B.; Busatto, N.; Ravaglia, D.; Spinelli, F.; Costa, G. Use of the index of absorbance difference (IAD) as a tool for tailoring post-harvest 1-MCP application to control apple superficial scald. Sci. Hortic. 2015, 190, 110–116. [Google Scholar] [CrossRef]
- Cocetta, G.; Beghi, R.; Mignani, I.; Spinardi, A.M. Nondestructive Apple Ripening Stage Determination Using the Delta Absorbance Meter at Harvest and after Storage. HortTechnology 2017, 27, 54–64. [Google Scholar] [CrossRef]
- Wang, H.; Cheng, L. Differential Effects of Nitrogen Supply on Skin Pigmentation and Flesh Starch Breakdown of ‘Gala’ Apple. HortScience 2011, 46, 1116–1120. [Google Scholar] [CrossRef]
| Composition | Content, % |
|---|---|
| Dry matter | 91.20 ± 1.60 |
| Organic matter | 98.30 ± 0.90 |
| Organic carbon | 38.65 ± 1.30 |
| Total nitrogen (N): N in organic form, N in inorganic form | 14.10 ± 1.20 99.2 0.6 |
| C/N rate | 2.74 |
| Total phosphor (P), % | 0.25 ± 0.06 |
| Total potassium (K), % | 0.11 ± 0.02 |
| Total sulphur (S), % | 1.10 ± 0.20 |
| Calcium (Ca), % | 0.60 ± 0.10 |
| Magnesium (Mg), % | 0.02 ± 0.003 |
| Month | Precipitation, mm | Average Air Temperature, °C | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Multiannual * Average | Deviation from Multiannual Average, ± | Multiannual Average | Deviation from Multiannual Average, ± | |||||||
| 2015 | 2016 | 2017 | 2018 | 2015 | 2016 | 2017 | 2018 | |||
| April | 38.4 | +33.6 | +11.8 | −8.2 | +9.8 | 6.1 | +1.6 | −0.5 | +2.9 | +1.4 |
| May | 53.8 | +6.4 | +58.6 | +49.0 | +9.4 | 12.3 | +1.4 | +3.9 | +1.3 | −0.3 |
| June | 62.6 | +28.0 | −23.6 | +22.2 | −35.8 | 15.6 | −0.1 | +3.4 | −0.8 | +0.2 |
| July | 81.2 | +19.6 | −15.2 | −30.0 | +10 | 17.6 | +2.1 | +1.4 | +2.8 | +0.8 |
| August | 80.3 | −9.9 | −2.9 | −57.5 | −75.7 | 16.6 | +0.4 | +1.6 | +6.6 | +3.1 |
| September | 52.6 | −1.8 | +31.4 | −52.6 | −9.2 | 12.2 | +1.3 | +0.1 | −0.1 | +1.8 |
| October | 49.6 | +4.4 | −5.8 | −2.6 | −38.6 | 6.8 | +0.4 | +1.9 | +0.5 | −1.2 |
| November | 46.1 | +4.9 | −1.3 | −20.1 | +26.5 | 1.5 | +3.5 | +3.6 | +1.5 | +3.5 |
| Total period | 464.6 | +85.2 | +53.0 | −99.8 | −103.6 | 11.1 | +1.3 | +1.9 | +1.8 | +1.2 |
| Treatment | Year | |||
|---|---|---|---|---|
| 2015 | 2016 | 2017 | 2018 | |
| N0 | 2.13 ab * | 1.90 a | 2.06 a | 1.58 a |
| N50 (NH4NO3) | 2.14 ab | 2.11 d | 2.12 ab | 1.89 b |
| N100 (NH4NO3) | 2.17 b | 2.07 bcd | 2.17 b | 1.86 b |
| N50 (horn shavings in spring) | 2.13 ab | 1.95 ab | 2.11 ab | 1.61 a |
| N100 (horn shavings in spring) | 2.16 b | 2.05 bcd | 2.16 b | 1.79 b |
| N50+50 (horn shavings in autumn and spring) | 2.16 b | 2.06 bcd | 2.08 a | 1.77 b |
| N50 (horn shavings in autumn) | 2.14 ab | 2.07 bcd | 2.10 a | 1.80 b |
| N100 (horn shavings in autumn) | 2.08 a | 2.08 bcd | 2.13 b | 1.82 b |
| p | 0.2 | 0.2 | 0.2 | 0.05 |
| Treatment | Year | Average | |||
|---|---|---|---|---|---|
| 2015 | 2016 | 2017 | 2018 | ||
| N0 | 33.8 a * | 26.3 a | 60.9 a | 16.9 a | 34.5 a |
| N50 (NH4NO3) | 34.8 a | 33.6 ab | 54.6 a | 46.6 c | 42.4 c |
| N100 (NH4NO3) | 33.3 a | 36.4 ab | 56.9 a | 36.7 bc | 40.8 ab |
| N50 (horn shavings in spring) | 38.1 a | 24.7 a | 62.8 a | 20.9 a | 36.6 ab |
| N100 (horn shavings in spring) | 32.9 a | 32.5 ab | 59.2 a | 35.4 abc | 40.0 abc |
| N50+50 (horn shavings in autumn and spring) | 32.7 a | 34.9 ab | 60.9 a | 31.5 abc | 40.0 abc |
| N50 (horn shavings in autumn) | 31.9 a | 38.0 b | 57.2 a | 38.4 bc | 41.4 bc |
| N100 (horn shavings in autumn) | 31.6 a | 39.1 b | 56.0 a | 31.9 abc | 39.7 bc |
| p | ns ** | 0.1 | ns | 0.05 | 0.2 |
| Treatment | Year | Average | |||
|---|---|---|---|---|---|
| 2015 | 2016 | 2017 | 2018 | ||
| N0 | 1.44 ab * | 0.80 a | 1.37 ab | 0.34 a | 0.99 a |
| N50 (NH4NO3) | 1.44 ab | 1.12 ab | 1.28 ab | 0.93 b | 1.19 b |
| N100 (NH4NO3) | 1.41 ab | 1.23 b | 1.32 ab | 0.74 b | 1.18 b |
| N50 (horn shavings in spring) | 1.64 b | 0.97 ab | 1.43 b | 0.40 a | 1.11 ab |
| N100 (horn shavings in spring) | 1.38 ab | 1.11 ab | 1.37 ab | 0.71 ab | 1.14 ab |
| N50+50 (horn shavings in autumn and spring) | 1.28 ab | 1.02 ab | 1.27 ab | 0.57 ab | 1.04 ab |
| N50 (horn shavings in autumn) | 1.30 ab | 1.17 b | 1.27 ab | 0.73 b | 1.12 ab |
| N100 (horn shavings in autumn) | 1.19 a | 1.16 b | 1.18 a | 0.59 ab | 1.03 ab |
| p | 0.1 | 0.2 | 0.2 | 0.05 | 0.05 |
| Treatment | Year | Average | |||
|---|---|---|---|---|---|
| 2015 | 2016 | 2017 | 2018 | ||
| N0 | 257 | 265 ab * | 235 ab | 234 bc | 248 ab |
| N50 (NH4NO3) | 234 | 244 a | 239 ab | 210 ab | 232 a |
| N100 (NH4NO3) | 260 | 275 b | 222 a | 208 ab | 241 ab |
| N50 (horn shavings in spring) | 242 | 261 ab | 221 a | 210 ab | 234 a |
| N100 (horn shavings in spring) | 260 | 240 a | 247 b | 198 a | 236 ab |
| N50+50 (horn shavings in autumn and spring) | 260 | 276 b | 228 ab | 187 a | 238 ab |
| N50 (horn shavings in autumn) | 257 | 265 ab | 233 ab | 244 c | 250 b |
| N100 (horn shavings in autumn) | 270 | 258 ab | 236 ab | 219 bc | 246 ab |
| p | ns ** | 0.05 | 0.05 | 0.05 | 0.05 |
| Treatment | Year | Average | |||
|---|---|---|---|---|---|
| 2015 | 2016 | 2017 | 2018 | ||
| N0 | 7.6 | 7.8 ab * | 6.1 ab | 6.9 b | 7.1 ab |
| N50 (NH4NO3) | 7.8 | 8.1 ab | 6.1 ab | 6.3 ab | 7.1 ab |
| N100 (NH4NO3) | 7.6 | 7.7 ab | 5.9 a | 6.5 ab | 6.9 a |
| N50 (horn shavings in spring) | 7.6 | 8.3 b | 6.2 ab | 6.7 ab | 7.2 b |
| N100 (horn shavings in spring) | 7.9 | 7.6 a | 6.3 b | 6.0 a | 6.9 a |
| N50+50 (horn shavings in autumn and spring) | 7.6 | 7.6 ab | 6.1 ab | 6.2 ab | 6.9 a |
| N50 (horn shavings in autumn) | 7.7 | 7.7 ab | 6.3 b | 6.5 ab | 7.1 ab |
| N100 (horn shavings in autumn) | 7.8 | 7.6 ab | 6.3 b | 6.4 ab | 7.0 a |
| p | ns ** | 0.05 | 0.05 | 0.1 | 0.05 |
| Treatment | Year | Average | |||
|---|---|---|---|---|---|
| 2015 | 2016 | 2017 | 2018 | ||
| N0 | 12.0 a * | 13.6 | 12.8 | 12.1 a | 12.6 a |
| N50 (NH4NO3) | 12.5 ab | 14.2 | 13.5 | 12.9 ab | 13.3 bc |
| N100 (NH4NO3) | 12.5 ab | 13.8 | 12.7 | 13.4 bc | 13.1 b |
| N50 (horn shavings in spring) | 12.2 a | 14.3 | 12.8 | 14.3 c | 13.4 c |
| N100 (horn shavings in spring) | 12.7 b | 13.7 | 12.7 | 13.9 bc | 13.2 bc |
| N50+50 (horn shavings in autumn and spring) | 12.8 b | 13.3 | 12.9 | 12.5 ab | 12.9 ab |
| N50 (horn shavings in autumn) | 12.9 b | 13.6 | 12.6 | 12.5 ab | 12.9 ab |
| N100 (horn shavings in autumn) | 12.9 b | 13.4 | 13.0 | 13.2 b | 13.1 b |
| p | 0.05 | ns ** | ns | 0.05 | 0.1 |
| Treatment | Year | Average | |
|---|---|---|---|
| 2017 | 2018 | ||
| N0 | 0.83 a * | 0.31 a | 0.57 a |
| N50 (NH4NO3) | 1.11 b | 0.80 bc | 0.96 b |
| N100 (NH4NO3) | 1.14 b | 0.94 c | 1.04 b |
| N50 (horn shavings in spring) | 0.98 ab | 0.53 ab | 0.76 ab |
| N100 (horn shavings in spring) | 1.12 b | 0.80 bc | 0.96 b |
| N50+50 (horn shavings in autumn and spring) | 1.03 ab | 0.94 c | 0.98 b |
| N50 (horn shavings in autumn) | 1.12 b | 0.73 bc | 0.92 b |
| N100 (horn shavings in autumn) | 0.98 ab | 0.77 bc | 0.88 b |
| p | 0.05 | 0.05 | 0.05 |
| Treatment | Year | |||
|---|---|---|---|---|
| 2015 | 2016 | 2017 | 2018 | |
| N0 | 58 a * | 68 a | 72 b | 59 b |
| N50 (NH4NO3) | 59 ab | 66 a | 65 a | 51 a |
| N100 (NH4NO3) | 63 bc | 69 a | 65 a | 47 a |
| N50 (horn shavings in spring) | 61 ab | 69 a | 66 a | 56 ab |
| N100 (horn shavings in spring) | 61 ab | 65 a | 65 a | 45 a |
| N50+50 (horn shavings in autumn and spring) | 66 c | 68 a | 67 ab | 48 a |
| N50 (horn shavings in autumn) | 58 a | 64 a | 67 ab | 41 a |
| N100 (horn shavings in autumn) | 62 b | 63 a | 67 ab | 44 a |
| p | 0.1 | ns ** | 0.1 | 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lanauskas, J.; Uselis, N.; Buskienė, L.; Mažeika, R.; Staugaitis, G.; Kviklys, D. Cattle Horn Shavings: A Possible Nitrogen Source for Apple Trees. Agronomy 2021, 11, 540. https://doi.org/10.3390/agronomy11030540
Lanauskas J, Uselis N, Buskienė L, Mažeika R, Staugaitis G, Kviklys D. Cattle Horn Shavings: A Possible Nitrogen Source for Apple Trees. Agronomy. 2021; 11(3):540. https://doi.org/10.3390/agronomy11030540
Chicago/Turabian StyleLanauskas, Juozas, Nobertas Uselis, Loreta Buskienė, Romas Mažeika, Gediminas Staugaitis, and Darius Kviklys. 2021. "Cattle Horn Shavings: A Possible Nitrogen Source for Apple Trees" Agronomy 11, no. 3: 540. https://doi.org/10.3390/agronomy11030540
APA StyleLanauskas, J., Uselis, N., Buskienė, L., Mažeika, R., Staugaitis, G., & Kviklys, D. (2021). Cattle Horn Shavings: A Possible Nitrogen Source for Apple Trees. Agronomy, 11(3), 540. https://doi.org/10.3390/agronomy11030540






