Agronomic Traits in Oilseed Rape (Brassica napus) Can Predict Foraging Resources for Insect Pollinators
Abstract
1. Introduction
1.1. Pollinator Decline
1.2. Interdependence of Oilseed Rape and Insect Pollinators
1.3. Oilseed Rape Breeding Programmes
1.4. Agronomic Traits of Oilseed Rape
1.5. Aim of this Study
2. Materials and Methods
2.1. Study Site
2.2. Environmental Conditions
2.3. Nectar Collection and Quantification
2.4. Pollen Collection and Quantification
2.5. Statistical Analysis
3. Results
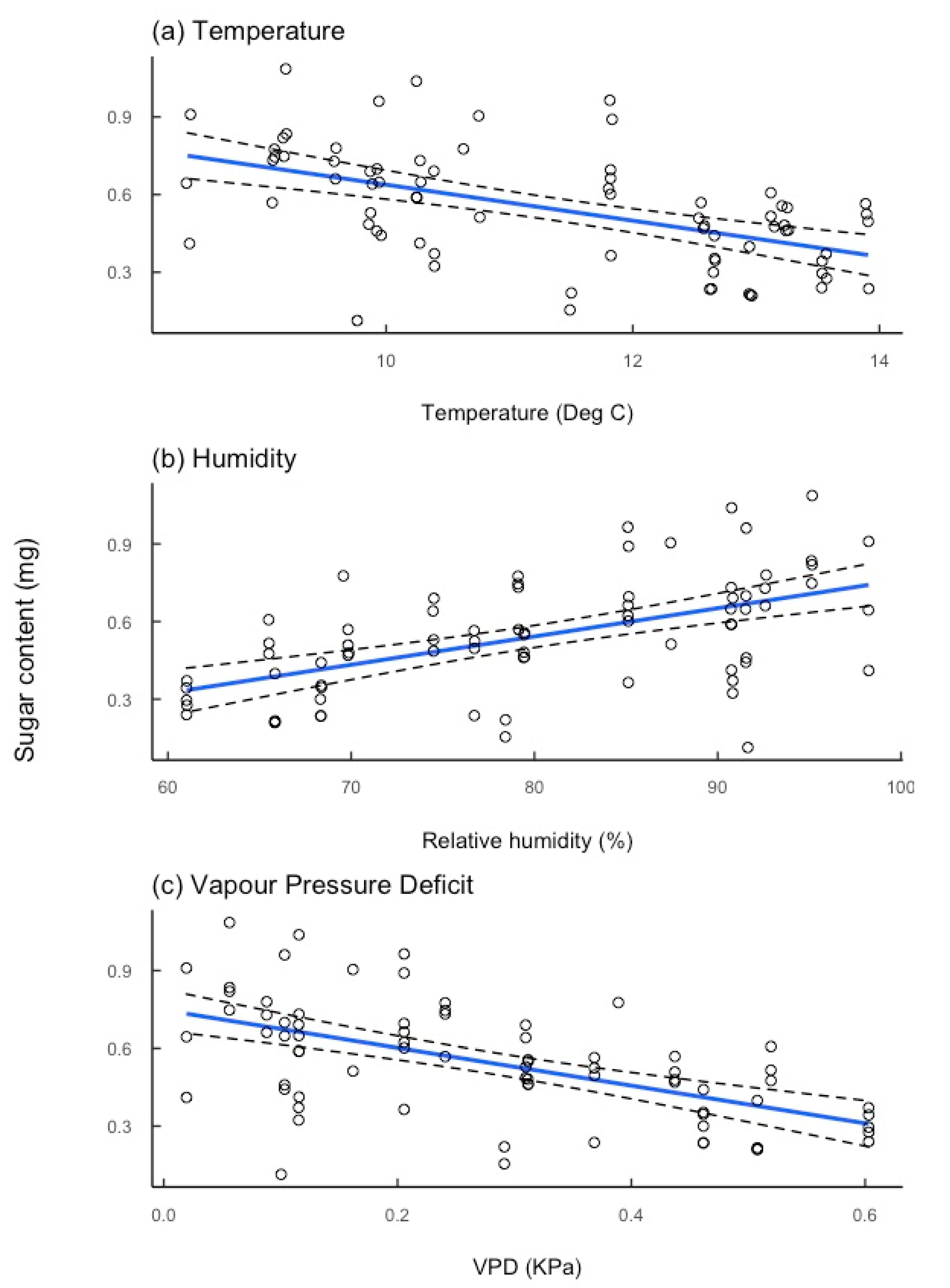
3.1. Environmental Effects on Sugar Quantity
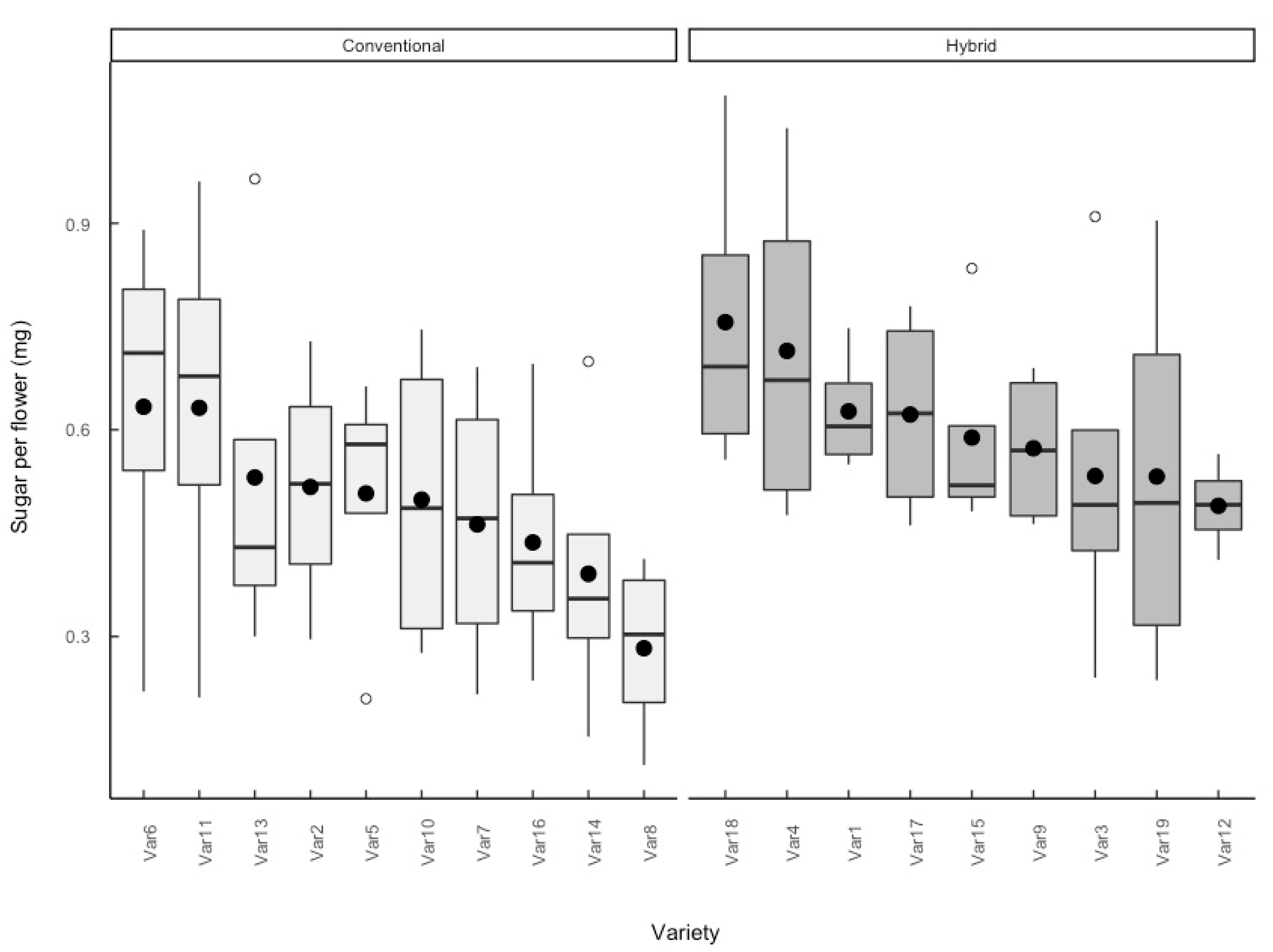
3.2. Sugar per Flower
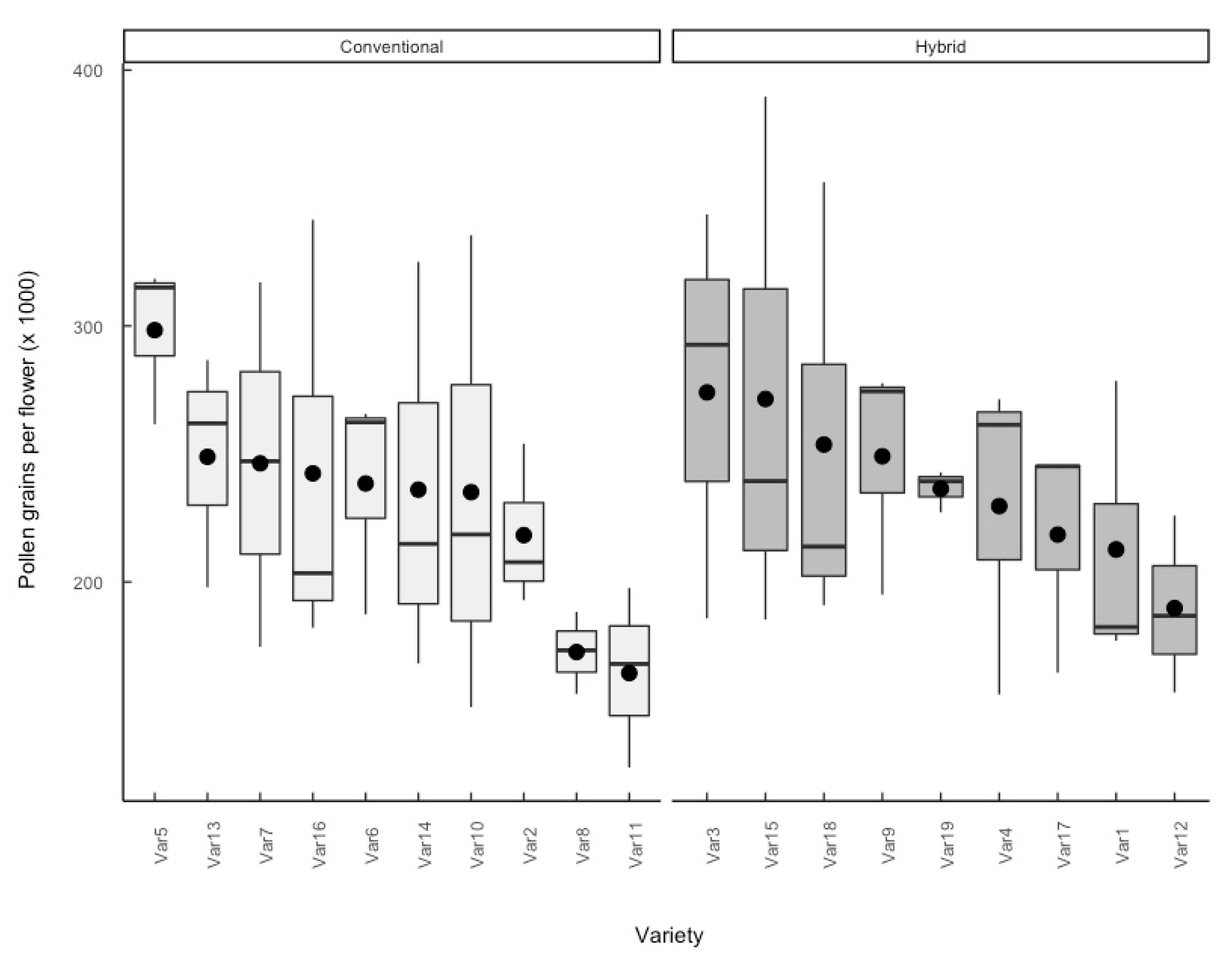
3.3. Pollen Grains per Flower
3.4. Prediction Models
3.4.1. Sugar per Flower
3.4.2. Pollen per Flower
4. Discussion
4.1. Overview
4.2. Impact of Environmental Factors on Nectar Resources
4.3. Impact of Breeding System on Floral Resources
4.4. Impact of Agronomic Traits on Floral Resources
4.5. Model Application
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Biesmeijer, J.C.; Roberts, S.P.M.; Reemer, M.; Ohlemüller, R.; Edwards, M.; Peeters, T.; Schaffers, A.P.; Potts, S.G.; Kleukers, R.; Thomas, C.D.; et al. Parallel Declines in Pollinators and Insect-Pollinated Plants in Britain and the Netherlands. Science 2006, 313, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global Pollinator Declines: Trends, Impacts and Drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Cameron, S.A.; Lozier, J.D.; Strange, J.P.; Koch, J.B.; Cordes, N.; Solter, L.F.; Griswold, T.L. Patterns of Widespread Decline in North American Bumble Bees. Proc. Natl. Acad. Sci. USA 2011, 108, 662–667. [Google Scholar] [CrossRef]
- Carvalheiro, L.G.; Kunin, W.E.; Keil, P.; Aguirre-Gutiérrez, J.; Ellis, W.N.; Fox, R.; Groom, Q.; Hennekens, S.; Van Landuyt, W.; Maes, D.; et al. Species Richness Declines and Biotic Homogenisation Have Slowed down for NW-European Pollinators and Plants. Ecol. Lett. 2013, 16, 870–878. [Google Scholar] [CrossRef]
- Vanbergen, A.J.; Initiative, T.I.P. Threats to an Ecosystem Service: Pressures on Pollinators. Front. Ecol. Environ. 2013, 11, 251–259. [Google Scholar] [CrossRef]
- Ollerton, J.; Winfree, R.; Tarrant, S. How Many Flowering Plants Are Pollinated by Animals? Oikos 2011, 120, 321–326. [Google Scholar] [CrossRef]
- Klein, A.-M.; Vaissière, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of Pollinators in Changing Landscapes for World Crops. Proc. R. Soc. B Boil. Sci. 2007, 274, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Blackstock, T.H.; Rimes, C.A.; Stevens, D.P.; Jefferson, R.G.; Robertson, H.J.; Mackintosh, J.; Hopkins, J.J. Hopkins the Extent of Semi-natural Grassland Communities in Lowland England and Wales: A Review of Conservation Surveys 1978–1996. Grass Forage Sci. 1999, 54, 1–18. [Google Scholar] [CrossRef]
- Robinson, R.A.; Sutherland, W.J. Post-war Changes in Arable Farming and Biodiversity in Great Britain. J. Appl. Ecol. 2002, 39, 157–176. [Google Scholar] [CrossRef]
- Potts, S.G.; Imperatriz-Fonseca, V.; Ngo, H.T.; Aizen, M.A.; Biesmeijer, J.C.; Breeze, S.G.P.T.D.; Dicks, L.V.; Garibaldi, L.A.; Hill, R.; Settele, J.; et al. Safeguarding Pollinators and Their Values to Human Well-Being. Nat. Cell Biol. 2016, 540, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Goulson, D.; Nicholls, E.; Botías, C.; Rotheray, E.L. Bee Declines Driven by Combined Stress from Parasites, Pesticides, and Lack of Flowers. Science 2015, 347, 1255957. [Google Scholar] [CrossRef] [PubMed]
- USDA. Oilseeds: World markets and trade. Available online: https://www.fas.usda.gov/data/oilseeds-world-markets-and-trade (accessed on 12 February 2021).
- Bommarco, R.; Marini, L.; Vaissière, B.E. Insect Pollination Enhances Seed Yield, Quality, and Market Value in Oilseed Rape. Oecologia 2012, 169, 1025–1032. [Google Scholar] [CrossRef]
- Morandin, L.A.; Winston, M.L. Pollinators Provide Economic Incentive to Preserve Natural Land in Agroecosystems. Agric. Ecosyst. Environ. 2006, 116, 289–292. [Google Scholar] [CrossRef]
- Bartomeus, I.; Potts, S.G.; Steffan-Dewenter, I.; Vaissière, B.E.; Woyciechowski, M.; Krewenka, K.M.; Tscheulin, T.; Roberts, S.P.; Szentgyörgyi, H.; Westphal, C.; et al. Contribution of Insect Pollinators to Crop Yield and Quality Varies with Agricultural Intensification. PeerJ 2014, 2, e328. [Google Scholar] [CrossRef] [PubMed]
- Sabbahi, R.; De Oliveira, D.; Marceau, J. Influence of Honey Bee (Hymenoptera: Apidae) Density on the Production of Canola (Crucifera: Brassicacae). J. Econ. Èntomol. 2005, 98, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Jauker, F.; Wolters, V. Hover Flies Are Efficient Pollinators of Oilseed Rape. Oecologia 2008, 156, 819–823. [Google Scholar] [CrossRef]
- Fairhurst, S.M. Oilseed Rape and Pollinators: The Impact of Variety on Resource Availability and Pollination Resilience. Ph.D. Thesis, University of Edinburgh, Edinburgh, UK, 2020. [Google Scholar]
- Steffan-Dewenter, I. Seed Set of Male-Sterile and Male-Fertile Oilseed Rape (Brassica napus) in Relation to Pollinator Density. Apidologie 2003, 34, 227–235. [Google Scholar] [CrossRef][Green Version]
- Garibaldi, L.A.; Aizen, M.A.; Klein, A.M.; Cunningham, S.A.; Harder, L.D. Global growth and stability of agricultural yield decrease with pollinator dependence. Proc. Natl. Acad. Sci. USA 2011, 108, 5909–5914. [Google Scholar] [CrossRef]
- Breeze, T.D.; Boreux, V.; Cole, L.; Dicks, L.; Klein, A.; Pufal, G.; Balzan, M.V.; Bevk, D.; Bortolotti, L.; Petanidou, T.; et al. Linking Farmer and Beekeeper Preferences with Ecological Knowledge to Improve Crop Pollination. People Nat. 2019, 1, 562–572. [Google Scholar] [CrossRef]
- Conner, J.K.; Proctor, M.; Yeo, P.; Lack, A. The Natural History of Pollination. Ecology 1997, 78, 327. [Google Scholar] [CrossRef]
- Roulston, T.H.; Cane, J.H. Pollen Nutritional Content and Digestibility for Animals. Plant Syst. Evol. 2000, 222, 187–209. [Google Scholar] [CrossRef]
- Bowers, M.A. Resource Availability and Timing of Reproduction in Bumble Bee Colonies (Hymenoptera: Apidae). Environ. Èntomol. 1986, 15, 750–755. [Google Scholar] [CrossRef]
- Albrecht, M.; Duelli, P.; Müller, C.; Kleijn, D.; Schmid, B. The Swiss Agri-Environment Scheme Enhances Pollinator Diversity and Plant Reproductive Success in Nearby Intensively Managed Farm-Land. J. Appl. Ecol. 2007, 44, 813–822. [Google Scholar] [CrossRef]
- Seeley, T.D. The Wisdom of the Hive: The Social Physiology of Honey Bee Colonies; Harvard University Press: Cambridge, MA, USA, 2009. [Google Scholar]
- Stanley, D.A.; Stout, J.C. Quantifying the Impacts of Bioenergy Crops on Pollinating Insect Abundance and Diversity: A Field-Scale Evaluation Reveals Taxon-Specific Responses. J. Appl. Ecol. 2013, 50, 335–344. [Google Scholar] [CrossRef]
- Gill, K.A.; O’Neal, M.E. Survey of Soybean Insect Pollinators: Community Identification and Sampling Method Analysis. Environ. Èntomol. 2015, 44, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Requier, F.; Odoux, J.-F.; Tamic, T.; Moreau, N.; Henry, M.; Decourtye, A.; Bretagnolle, V. Honey Bee Diet in Intensive Farmland Habitats Reveals an Unexpectedly High Flower Richness and a Major Role of Weeds. Ecol. Appl. 2015, 25, 881–890. [Google Scholar] [CrossRef] [PubMed]
- Westphal, C.; Steffan-Dewenter, I.; Tscharntke, T. Mass Flowering Crops Enhance Pollinator Densities at a Landscape Scale. Ecol. Lett. 2003, 6, 961–965. [Google Scholar] [CrossRef]
- Knight, E.M.; Osborne, J.L.; Sanderson, R.A.; Hale, R.J.; Martin, A.P.; Goulson, D. Bumblebee Nest Density and the Scale of Available Forage in Arable Landscapes. Insect Conserv. Divers. 2009, 2, 116–124. [Google Scholar] [CrossRef]
- Holzschuh, A.; Dormann, C.F.; Tscharntke, T.; Steffan-Dewenter, I. Mass-Flowering Crops Enhance Wild Bee Abundance. Oecologia 2012, 172, 477–484. [Google Scholar] [CrossRef]
- Westphal, C.; Steffan-Dewenter, I.; Tscharntke, T. Mass Flowering Oilseed Rape Improves Early Colony Growth but Not Sexual Reproduction of Bumblebees. J. Appl. Ecol. 2009, 46, 187–193. [Google Scholar] [CrossRef]
- Jauker, F.; Peter, F.; Wolters, V.; Diekötter, T. Early Reproductive Benefits of Mass-Flowering Crops to the Solitary Bee Osmia Rufa Outbalance Post-flowering Disadvantages. Basic Appl. Ecol. 2012, 13, 268–276. [Google Scholar] [CrossRef]
- Timberlake, T.P.; Vaughan, I.P.; Memmott, J. Phenology of Farmland Floral Resources Reveals Seasonal Gaps in Nectar Availability for Bumblebees. J. Appl. Ecol. 2019, 56, 1585–1596. [Google Scholar] [CrossRef]
- Carvell, C.; Meek, W.R.; Pywell, R.F.; Goulson, D.; Nowakowski, M. Comparing the Efficacy of Agri-Environment Schemes to Enhance Bumble Bee Abundance and Diversity on Arable Field Margins. J. Appl. Ecol. 2006, 44, 29–40. [Google Scholar] [CrossRef]
- Ouvrard, P.; Transon, J.; Jacquemart, A.-L. Flower-Strip Agri-Environment Schemes Provide Diverse and Valuable Summer Flower Resources for Pollinating Insects. Biodivers. Conserv. 2018, 27, 2193–2216. [Google Scholar] [CrossRef]
- Friedt, W.; Snowdon, R. Oilseed Rape. In Oil Crops; Friedt, W.; Snowdon, R. Springer: New York, NY, USA, 2009; pp. 91–126. [Google Scholar]
- AHDB. AHDB Recommended Lists for Cereals and Oilseeds 2016–2021. Available online: https://cereals.ahdb.org.uk/publications/2016/july/01/ahdb-recommended-lists-for-cereals-and-oilseeds-2016–2021.aspx (accessed on 23 August 2020).
- AHDB. AHDB Recommended Lists—Winter Oilseed Rape 2019/20. Available online: https://ahdb.org.uk/rl (accessed on 18 September 2020).
- Pelletier, G.; Primard, C.; Renard, M.; Pellan-Delourme, R.; Mesquida, J. Molecular, Phenotypic and Genetic Characterization of Mitochondrial Recombinants in Rapeseed. In Proceedings of the 7th GCIRC Rapeseed Congress, Poznan, Poland, 11–14 May 1987. pp. 113–118. [Google Scholar]
- Mesquida, J.; Pham Delegue, M.H.; Marilleau, R.; Le Metayer, M.; Renard, M. The Floral Nectar Secretion in Male Sterile Cybrid of Winter Rapeseed (Brassica napus L.). Agronomie 1991, 11, 217–227. [Google Scholar] [CrossRef]
- Bertazzini, M.; Forlani, G. Intraspecific Variability of Floral Nectar Volume and Composition in Rapeseed (Brassica napus L. Var. Oleifera). Front. Plant Sci. 2016, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Carruthers, J.M.; Cook, S.M.; Wright, G.A.; Osborne, J.L.; Clark, S.J.; Swain, J.L.; Haughton, A.J. Oilseed Rape (Brassica napus) as a Resource for Farmland Insect Pollinators: Quantifying Floral Traits in Conventional Varieties and Breeding Systems. GCB Bioenergy 2017, 9, 1370–1379. [Google Scholar] [CrossRef]
- Ouvrard, P.; Quinet, M.; Jacquemart, A.-L. Breeding System and Pollination Biology of Belgian Oilseed Rape Cultivars (Brassica napus). Crop. Sci. 2017, 57, 1455–1463. [Google Scholar] [CrossRef]
- Baude, M.; Kunin, W.E.; Boatman, N.D.; Conyers, N.D.B.S.; Davies, N.; Gillespie, W.E.K.M.A.K.; Morton, R.D.; Smart, R.D.M.S.M.; Memmott, M.B.N.D.J. Historical Nectar Assessment Reveals the Fall and Rise of Floral Resources in Britain. Nat. Cell Biol. 2016, 530, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Cruden, R.W. Pollen Grains: Why so Many? Plant Syst. Evol. 2000, 222, 143–165. [Google Scholar] [CrossRef]
- Southwick, E.E. Photosynthate Allocation to Floral Nectar: A Neglected Energy Investment. Ecology 1984, 65, 1775–1779. [Google Scholar] [CrossRef]
- Búrquez, A.; Corbet, S.A. Dynamics of Production and Exploitation of Nectar: Lessons from Impatiens Glandulifera Royle. In Nectary Biology; Bahadur, B., Ed.; Dattsons: Nagpur, India, 1998; pp. 130–152. [Google Scholar]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop Evapotranspiration (Guidelines for Computing Crop Water Requirements); Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 1998. [Google Scholar]
- Corbet, S.A. Nectar Sugar Content: Estimating Standing Crop and Secretion Rate in the Field. Apidologie 2003, 34, 1–10. [Google Scholar] [CrossRef]
- Cruden, R.W.; Hermann, S.M. Studying Nectar? Some Observations on the Art. In The Biology of Nectaries; Bentley, B., Elias, T., Eds.; Columbia University Press: New York, NY, USA, 1983; pp. 223–241. [Google Scholar]
- Bolten, A.B.; Feinsinger, P.; Baker, H.G.; Baker, I. On the Calculation of Sugar Concentration in Flower Nectar. Oecologia 1979, 41, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Galetto, L.; Bernardello, G. Nectar. In Practical Pollination Biology; Dafni, A., Kevan, P.G., Husband, B.C., Eds.; Enviroquest Ltd.: Ontario, ON, Canada, 2005; pp. 261–313. [Google Scholar]
- Zuur, A.F.; Ieno, E.N.; Elphick, C.S. A Protocol for Data Exploration to Avoid Common Statistical Problems: Data Exploration. Methods Ecol. Evol. 2010, 1, 3–14. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna. 2018. Available online: https://www.R-project.org (accessed on 25 February 2021).
- Villarreal, A.G.; Freeman, C.E. Effects of Temperature and Water Stress on Some Floral Nectar Characteristics in Ipomopsis longiflora (Polemoniaceae) under Controlled Conditions. Int. J. Plant Sci. 1990, 151, 5–9. [Google Scholar] [CrossRef]
- Takkis, K.; Tscheulin, T.; Petanidou, T. Differential Effects of Climate Warming on the Nectar Secretion of Early-and Late-Flowering Mediterranean Plants. Front. Plant Sci. 2018, 9, 874. [Google Scholar] [CrossRef] [PubMed]
- Corbet, S.A.; Unwin, D.M.; Prŷs-Jones, O.E. Humidity, Nectar and Insect Visits to Flowers, with Special Reference to Crataegus, Tilia and Echium. Ecol. Èntomol. 1979, 4, 9–22. [Google Scholar] [CrossRef]
- Jakobsen, H.B.; Kritjánsson, K. Influence of Temperature and Floret Age on Nectar Secretion in Trifolium repens L. Ann. Bot. 1994, 74, 327–334. [Google Scholar] [CrossRef]
- Pernal, S.F.; Currie, R.W. Nectar Quality in Open-Pollinated, Pol CMS hybrid, And Dominant SI Hybrid Oilseed Summer Rape. Can. J. Plant Sci. 1998, 78, 79–89. [Google Scholar] [CrossRef]
- Pierre, J.; Mesquida, J.; Marilleau, R.; Pham-Delegue, M.H.; Renard, M. Nectar Secretion in Winter Oilseed Rape, Brassica Napus- Quantitative and Qualitative Variability among 71 Genotypes. Plant Breed. 1999, 118, 471–476. [Google Scholar] [CrossRef]
- Christen, O.; Friedt, W. Winter Rape. The Handbook for Professionals; Winterraps, Das Handbuch fuer Profis; DLG-Verlag: Frankfurt, Germany, 2007. [Google Scholar]
- Ludlow, M.; Muchow, R. A Critical Evaluation of Traits for Improving Crop Yields in Water-Limited Environments. Adv. Agron. 1990, 43, 107–153. [Google Scholar] [CrossRef]
- Passioura, J.; Angus, J. Improving Productivity of Crops in Water-Limited Environments. Adv. Agron. 2010, 106, 37–75. [Google Scholar] [CrossRef]
- Nicolson, S.W.; Thornburg, R.W. Nectar Chemistry. In Nectaries and Nectar; Nicolson, S.W., Nepi, M., Pacini, E., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 215–264. [Google Scholar]
- Limagrain UK Ltd. Boost Bonuses with High Oil Content OSR Varieties. Available online: https://www.lgseeds.co.uk/news/boost-bonuses-with-high-oil-content-osr-varieties/ (accessed on 12 February 2020).
- Doust, J.L. Plant Reproductive Strategies and Resource Allocation. Trends Ecol. Evol. 1989, 4, 230–234. [Google Scholar] [CrossRef]
- Kozłowski, J. Optimal Allocation of Resources to Growth and Reproduction: Implications for Age and Size at Maturity. Trends Ecol. Evol. 1992, 7, 15–19. [Google Scholar] [CrossRef]
- Filipiak, M. Key Pollen Host Plants Provide Balanced Diets for Wild Bee Larvae: A Lesson for Planting Flower Strips and Hedgerows. J. Appl. Ecol. 2019, 56, 1410–1418. [Google Scholar] [CrossRef]
- Lindström, S.A.M.; Herbertsson, L.; Rundlöf, M.; Smith, H.G.; Bommarco, R. Large-Scale Pollination Experiment Demonstrates the Importance of Insect Pollination in Winter Oilseed Rape. Oecologia 2015, 180, 759–769. [Google Scholar] [CrossRef]
- Stanley, D.A.; Gunning, D.; Stout, J.C. Pollinators and Pollination of Oilseed Rape Crops (Brassica napus L.) in Ireland: Ecological and Economic Incentives for Pollinator Conservation. J. Insect Conserv. 2013, 17, 1181–1189. [Google Scholar] [CrossRef]
- Scheper, J.; Bommarco, R.; Holzschuh, A.; Potts, S.G.; Riedinger, V.; Roberts, S.P.M.; Rundlöf, M.; Smith, H.G.; Steffan-Dewenter, I.; Wickens, J.B.; et al. Local and Landscape-Level Floral Resources Explain Effects of Wildflower Strips on Wild Bees across Four European Countries. J. Appl. Ecol. 2015, 52, 1165–1175. [Google Scholar] [CrossRef]
| Variables | Description |
|---|---|
| Sugar quantity | Sugar mass per flower: mg sugar μL−1 |
| Pollen quantity | Number of pollen grains per flower |
| Agronomic traits | |
| Gross output | Tonnes per hectare |
| Seed yield | Tonnes per hectare |
| Oil content | Oil content % |
| Early vigour | Competition with weeds: 1–9 (1 very weak, 9 very strong) |
| Emergence | Date of full emergence: 1–9 (1 very slow, 9 very fast) |
| Establishment | Plants per m2 following emergence: 1–9 (1 very thin, 9 very thick) |
| Lodging | Resistance to lodging (flowering): 1–9 (1 all plants lodged, 9 no lodging) |
| Stem stiffness | Resistance to lodging (maturity): 1–9 (1 all plants lodged, 9 no lodging) |
| Height | Average plant height at end of flowering: measured in centimetres |
| Earliness of flowering | Start of flowering: 1–9 (1 latest flowering plot, 9 earliest flowering plot) |
| Earliness of maturity | Canopy senescence before harvest: 1–9 (1 very late, 9 very early) |
| Winter hardiness | Survival rates throughout winter: 1–9 (1 complete loss, 9 no damage) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fairhurst, S.M.; Cole, L.J.; Kocarkova, T.; Jones-Morris, C.; Evans, A.; Jackson, G. Agronomic Traits in Oilseed Rape (Brassica napus) Can Predict Foraging Resources for Insect Pollinators. Agronomy 2021, 11, 440. https://doi.org/10.3390/agronomy11030440
Fairhurst SM, Cole LJ, Kocarkova T, Jones-Morris C, Evans A, Jackson G. Agronomic Traits in Oilseed Rape (Brassica napus) Can Predict Foraging Resources for Insect Pollinators. Agronomy. 2021; 11(3):440. https://doi.org/10.3390/agronomy11030440
Chicago/Turabian StyleFairhurst, Stacey M., Lorna J. Cole, Tereza Kocarkova, Catherine Jones-Morris, Andy Evans, and Gail Jackson. 2021. "Agronomic Traits in Oilseed Rape (Brassica napus) Can Predict Foraging Resources for Insect Pollinators" Agronomy 11, no. 3: 440. https://doi.org/10.3390/agronomy11030440
APA StyleFairhurst, S. M., Cole, L. J., Kocarkova, T., Jones-Morris, C., Evans, A., & Jackson, G. (2021). Agronomic Traits in Oilseed Rape (Brassica napus) Can Predict Foraging Resources for Insect Pollinators. Agronomy, 11(3), 440. https://doi.org/10.3390/agronomy11030440






