Exogenous Serotonin Improves Salt Tolerance in Rapeseed (Brassica napus L.) Seedlings
Abstract
1. Introduction
2. Materials and Methods
2.1. The Materials
2.2. Experimental Methods
2.3. The Measurements
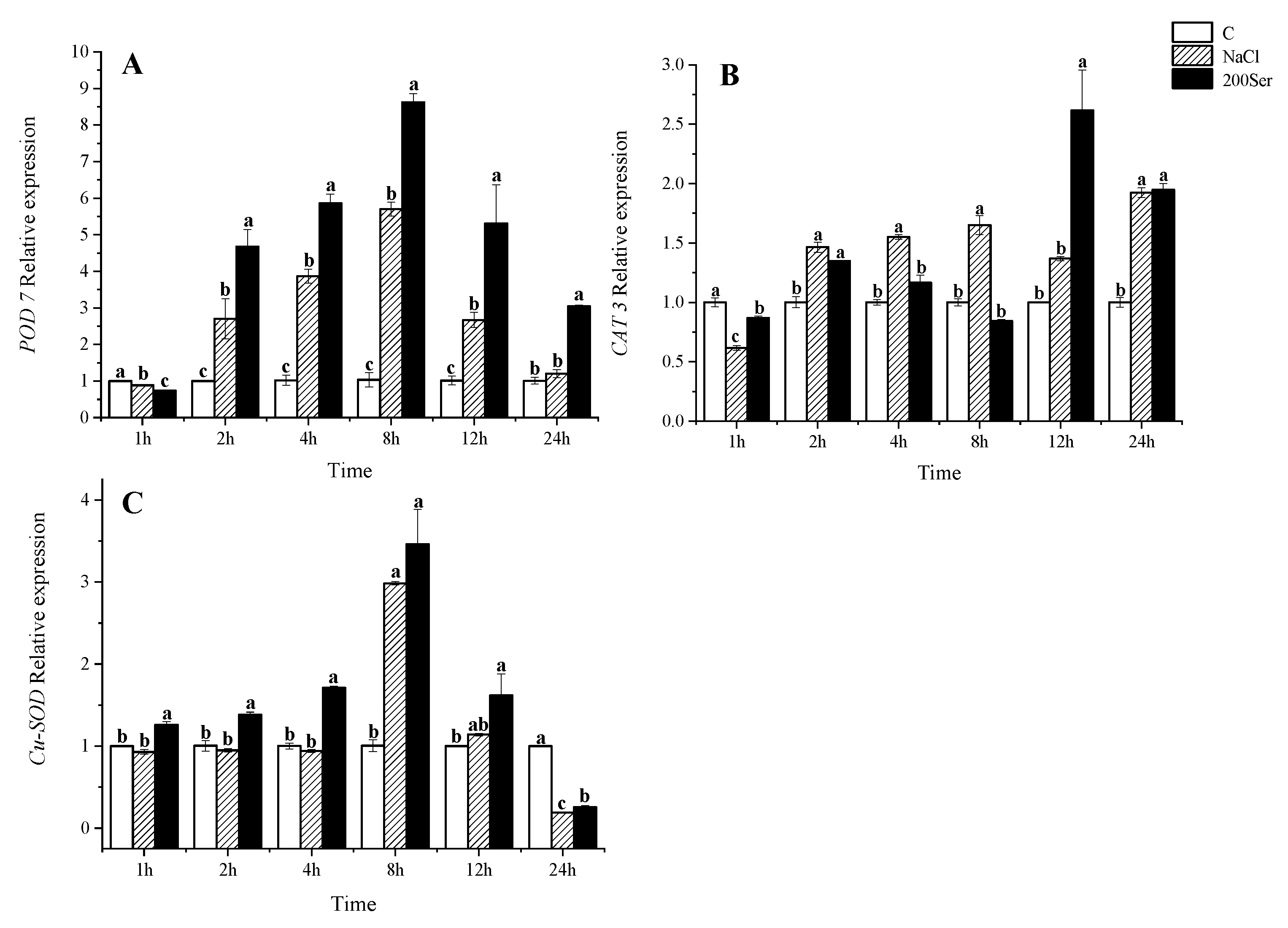
2.4. The RT-qPCR
2.5. Statistical Analysis
3. Results
3.1. Effects of Exogenous Serotonin on Phenotypical of B. napus under Salinity
3.2. Effects of Exogenous Serotonin on the Growth under Salinity
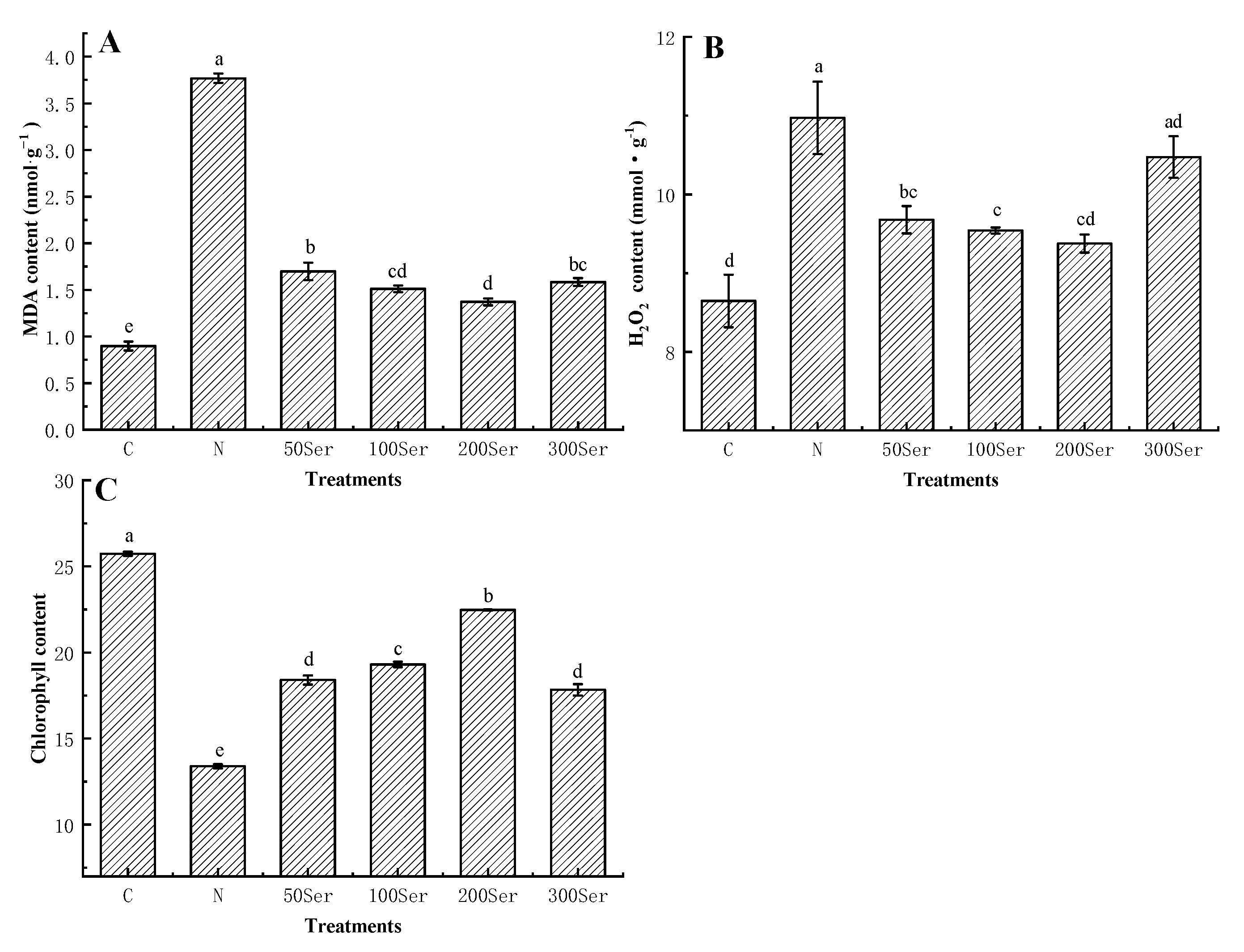
3.3. Effects of Exogenous Serotonin on MDA, H2O2 and Chlorophyll Contents under Salinity
3.4. Effects of Exogenous Serotonin on Osmoregulatory Substances under Salinity
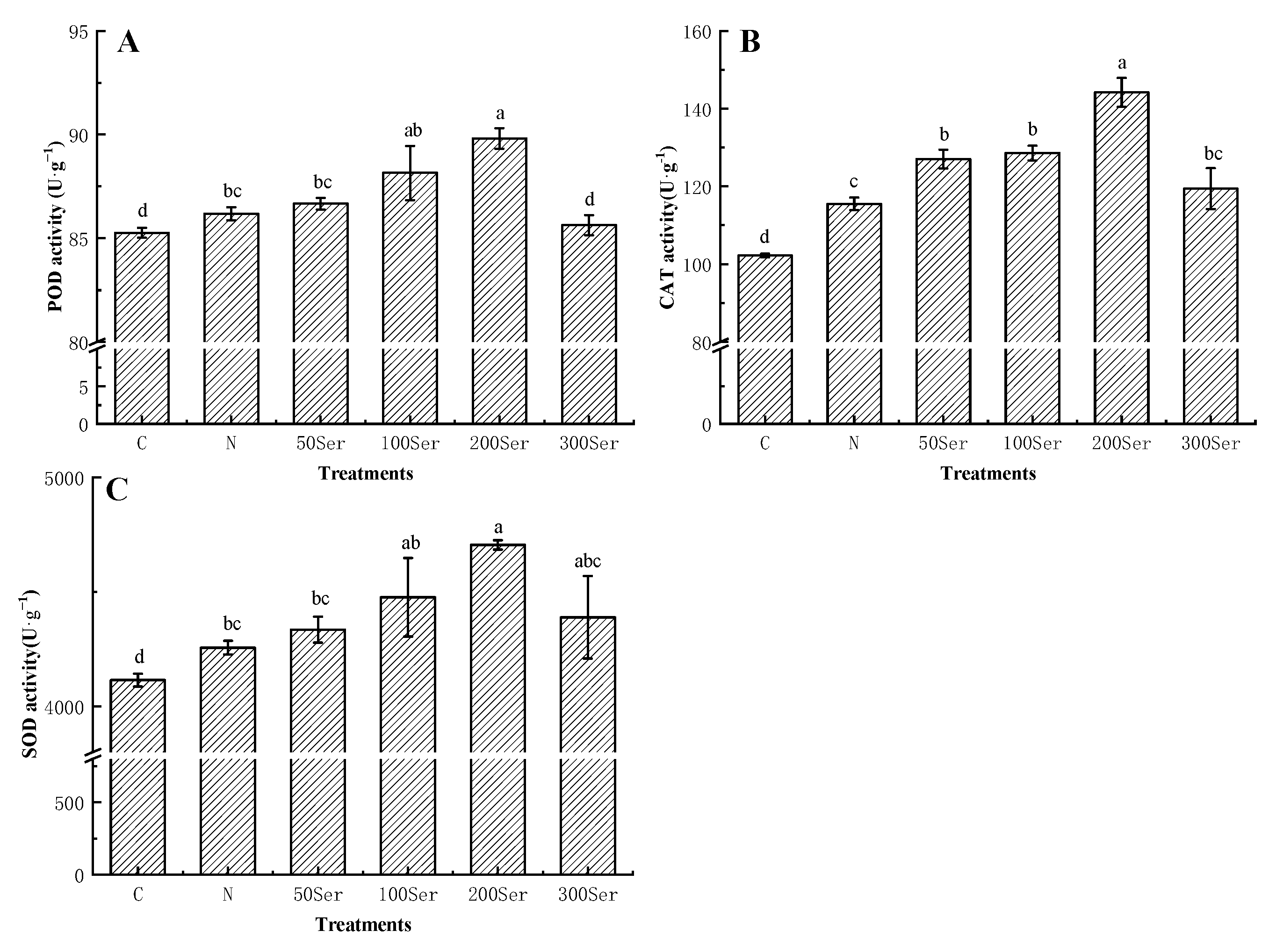
3.5. Effects of Exogenous Serotonin on Antioxidant Enzymes under Salinity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chang, B.; Yang, L.; Cong, W.; Zu, Y.; Tang, Z. The improved resistance to high salinity induced by trehalose is associated with ionic regulation and osmotic adjustment in Catharanthus roseus. Plant Physiol. Biochem. 2014, 77, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Arif, Y.; Singh, P.; Siddiqui, H.; Bajguz, A.; Hayat, S. Salinity induced physiological and biochemical changes in plants: An omic approach towards salt stress tolerance. Plant Physiol. Biochem. 2020, 156, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.P.; Li, S.; Ren, J.; Song, X.S. Role of spermidine and spermine in alleviation of drought-induced oxidative stress and photosynthetic inhibition in Chinese dwarf cherry (Cerasus humilis) seedlings. Plant Growth Regul. 2014, 74, 209–218. [Google Scholar] [CrossRef]
- Koca, H.; Bor, M.; Ozdemir, F.; Turkan, I. The effect of salt stress on lipid peroxidation, antioxidative enzymes and proline content of sesame cultivars. Environ. Exp. Bot. 2007, 60, 344–351. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, Y. Unraveling salt stress signaling in plants. J. Integr. Plant Biol. 2018, 60, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Yan, R.; Chen, F. Effects of different storage temperatures and times on germination and antioxidant responses of Jatropha curcas L. seeds. J. Agric. Sci. Technol. 2015, 17, 1619–1628. [Google Scholar]
- Seleiman, M.; Semida, W.; Rady, M.; Mohamed, G.; Sahmi, A. Sequential application of antioxidants rectifies ion imbalance and strengthens antioxidant systems in salt-stressed cucumber. Plants 2020, 9, 1783. [Google Scholar] [CrossRef]
- Hernandez, J.A.; Jimenez, A.; Mullineaux, P.; Sevilla, F. Tolerance of pea (Pisum sativum L.) to long-term salt stress is associated with induction of antioxidant defences. Plant Cell Environ. 2000, 23, 853–862. [Google Scholar] [CrossRef]
- Hu, T.; Li, H.Y.; Zhang, X.Z.; Luo, H.J.; Fu, J.M. Toxic effect of NaCl on ion metabolism, antioxidative enzymes and gene expression of perennial ryegrass. Ecotoxicol. Environ. Saf. 2011, 74, 2050–2056. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Cai, J.; Li, J.; Lu, G.; Li, C.; Fu, G.; Zhang, X.; Ma, H.; Liu, Q.; Zou, X.; et al. Exogenous application of a low concentration of melatonin enhances salt tolerance in rapeseed (Brassica napus L.) seedlings. J. Integr. Agric. 2018, 17, 328–335. [Google Scholar] [CrossRef]
- Badawi, G.H.; Yamauchi, Y.; Shimada, E.; Sasaki, R.; Kawano, N.; Tanaka, K. Enhanced tolerance to salt stress and water deficit by overexpressing superoxide dismutase in tobacco (Nicotiana tabacum) chloroplasts. Plant Sci. 2004, 166, 919–928. [Google Scholar] [CrossRef]
- Turlejski, K. Evolutionary ancient roles of serotonin: Long-lasting regulation of activity and development. Acta Neurobiol. Exp. 1996, 56, 619. [Google Scholar]
- Pelagio-Flores, R.; Muñoz-Parra, E.; Ortiz-Castro, R.; López-Bucio, J. Melatonin regulates Arabidopsis root system architecture likely acting independently of auxin signaling. J Pineal Res. 2012, 53, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Csaba, G.; Pál, K. Effects of insulin, triiodothyronine, and serotonin on plant seed development. Protoplasma 1982, 110, 20–22. [Google Scholar] [CrossRef]
- Murch, S.J.; Campbell, S.S.B.; Saxena, P.K. The role of serotonin and melatonin in plant morphogenesis: Regulation of auxin-induced root organogenesis in in vitro-cultured explants of st. John’s Wort (Hypericum perforatum L.). In Vitro Cell. Dev. Biol. Plant. 2001, 37, 786–793. [Google Scholar] [CrossRef]
- Kang, K.; Kim, Y.; Park, S.; Back, K. Senescence-Induced Serotonin Biosynthesis and Its Role in Delaying Senescence in Rice Leaves. Plant Physiol. 2009, 150, 1380–1393. [Google Scholar] [CrossRef]
- Pelagio-Flores, R.; Ruiz-Herrera, L.F.; López-Bucio, J. Serotonin modulates Arabidopsis root growth via changes in reactive oxygen species and jasmonic acid-ethylene signaling. Physiol. Plant 2016, 158, 92–105. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin: Plant growth regulator and/or biostimulator during stress? Trends Plant Sci. 2014, 19, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Nawaz, M.A.; Huang, Y.; Bie, Z.; Ahmed, W.; Reiter, R.J.; Niu, M.; Hameed, S. Melatonin: Current status and future perspectives in plant science. Front. Plant Sci. 2016, 6, 1230. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; David, A.; Yadav, S.; Baluška, F.; Bhatla, S.C. Salt stress-induced seedling growth inhibition coincides with differential distribution of serotonin and melatonin in sunflower seedling roots and cotyledons. Physiol. Plant 2014, 152, 714–728. [Google Scholar] [CrossRef] [PubMed]
- Yong, H.Y.; Wang, C.; Bancroft, I.; Li, F.; Wu, X.; Kitashiba, H.; Nishio, T. Identification of a gene controlling variation in the salt tolerance of rapeseed (Brassica napus L.). Planta 2015, 242, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Cui, L.; Feng, X.; Lin, C.J.; Wang, X.; Meng, B.; Wang, X.; Wang, H. Accumulation and translocation of 198Hg in four crop species. Environ. Toxicol. Chem. 2014, 33, 334–340. [Google Scholar] [CrossRef]
- Li, J.; Zeng, L.; Cheng, Y.; Lu, G.; Fu, G.; Ma, H.; Liu, Q.; Zhang, X.; Zou, X.; Li, C. Exogenous melatonin alleviates damage from drought stress in Brassica napus L. (rapeseed) seedlings. Acta Physiol. Plant. 2018, 40, 43. [Google Scholar] [CrossRef]
- Yan, L.; Shah, T.; Cheng, Y.; Lü, Y.; Zou, X. Physiological and molecular responses to cold stress in rapeseed (Brassica napus L.). J. Integr. Agric. 2019, 18, 2742–2752. [Google Scholar] [CrossRef]
- Golldack, D.; Lüking, I.; Yang, O. Plant tolerance to drought and salinity: Stress regulating transcription factors and their functional significance in the cellular transcriptional network. Plant Cell Rep. 2011, 30, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Chen, X.; He, Y.; Kou, X.; Xue, Z. Effects of Exogenous Trehalose on the Metabolism of Sugar and Abscisic Acid in Tomato Seedlings under Salt Stress. Trans. Tianjin Univ. 2019, 25, 451–471. [Google Scholar] [CrossRef]
- Pelagio-Flores, R.; Ortíz-Castro, R.; Méndez-Bravo, A.; Macías-Rodríguez, L.; López-Bucio, J. Serotonin, a tryptophan-derived signal conserved in plants and animals, regulates root system architecture probably acting as a natural auxin inhibitor in Arabidopsis thaliana. Plant Cell Physiol. 2011, 52, 490–508. [Google Scholar] [CrossRef]
- Naeem, M.S.; Warusawitharana, H.; Liu, H.; Liu, D.; Ahmad, R.; Waraich, E.A.; Xu, L.; Zhou, W. 5-Aminolevulinic acid alleviates the salinity-induced changes in Brassica napus as revealed by the ultrastructural study of chloroplast. Plant Physiol. Biochem. 2012, 57, 84–92. [Google Scholar] [CrossRef]
- Muller, O.; Cohu, C.M.; Stewart, J.J.; Protheroe, J.A.; Demmig-Adams, B.; Adams, W.W. Association between photosynthesis and contrasting features of minor veins in leaves of summer annuals loading phloem via symplastic versus apoplastic routes. Physiol. Plant 2014, 152, 174–183. [Google Scholar] [CrossRef]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar] [CrossRef]
- Sairam, R.K.; Srivastava, G.C. Changes in antioxidant activity in sub-cellular fractions of tolerant and susceptible wheat genotypes in response to long term salt stress. Plant Sci. 2002, 162, 897–904. [Google Scholar] [CrossRef]
- Upchurch, R.G. Fatty acid unsaturation, mobilization, and regulation in the response of plants to stress. Biotechnol. Lett. 2008, 30, 967–977. [Google Scholar] [CrossRef]
- Moller, I.M.; Jensen, P.E.; Hansson, A. Oxidative modifications to cellular components in plants. Annu. Rev. Plant Biol. 2007, 58, 459–481. [Google Scholar] [CrossRef]
- Zhang, T.; Shi, Z.; Zhang, X.; Zheng, S.; Wang, J.; Mo, J. Alleviating effects of exogenous melatonin on salt stress in cucumber. Sci. Hortic. 2020, 262, 109070. [Google Scholar] [CrossRef]
- Schutzendubel, A.; Polle, A. Plant responses to abiotic stresses: Heavy metal-induced oxidative stress and protection by mycorrhization. J. Exp. Bot. 2002, 53, 1351–1365. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, N.; Yang, R.; Wang, L.; Sun, Q.; Li, D.; Cao, Y.; Weeda, S.; Zhao, B.; Ren, S.; et al. Melatonin promotes seed germination under high salinity by regulating antioxidant systems, ABA and GA(4) interaction in cucumber (Cucumis sativus L.). J. Pineal Res. 2014, 57, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Hare, P.D.; Cress, W.A.; Staden, J.V. Dissecting the roles of osmolyte accumulation during stress. Plant Cell Environ. 1998, 21, 535–553. [Google Scholar] [CrossRef]
- Zhu, J.K. Plant salt tolerance. Trends Plant Sci. 2001, 6, 66–71. [Google Scholar] [CrossRef]
- Farhangi-Abriz, S.; Torabian, S. Antioxidant enzyme and osmotic adjustment changes in bean seedlings as affected by biochar under salt stress. Ecotoxicol. Environ. Saf. 2017, 137, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.; Harris, P.J.C. Potential biochemical in dicators of salinity tolerance in plants. Plant Sci. 2004, 166, 3–16. [Google Scholar] [CrossRef]
- Fahimirad, S.; Karimzadeh, G.; Ghanati, F. Cold-induced changes of antioxidant enzymes activity and lipid peroxidation in two canola (Brassica napus L.) cultivars. J. Plant Physiol. Breed. 2013, 3, 1–11. [Google Scholar]
- Signorelli, S. The Fermentation Analogy: A Point of View for Understanding the Intriguing Role of Proline Accumulation in Stressed. Front. Plant Sci. 2016, 7, 1339. [Google Scholar] [CrossRef] [PubMed]


| Gene | Accession Number | Forward Primer | Reverse Primer |
|---|---|---|---|
| POD7 | XM_013819255.1 | CCGTGGCTACAGCAACAGTC | CTCACCGCTACTACCAGTCAAGG |
| CAT3 | XM_013786778.1 | GAAGGCTTCGGTGTCCACAC | GGCTCCACCAACCACCTTT |
| Cu-SOD | XM_013808924.1 | GGTAACGGATCTGATTGGACG | ACGGTGGTTCCATCACAAGAA |
| Actin | AF111812 | CTCCTGAAGAGCACCCTG | CCCTCGTAGATTGGCACA |
| Treatment | Root Length(cm) | Shoot Length (cm) | Shoot Fresh Weights (g) | Shoot Dry Weight (g) | Root Fresh Weights (g) | Root Dry Weight (g) |
|---|---|---|---|---|---|---|
| C | 34.50 ± 0.34 a | 3.01 ± 0.04 a | 3.5608 ± 0.1746 a | 0.2290 ± 0.005 a | 0.4006 ± 0.0103 b | 0.0359 ± 0.0027 ab |
| N | 33.18 ± 0.33 b | 2.98 ± 0.05 a | 1.5218 ± 0.301 d | 0.1172 ± 0.003 d | 0.2915 ± 0.003 d | 0.0333 ± 0.0015 b |
| 50 Ser | 30.87 ± 0.38 c | 2.24 ± 0.03 d | 1.8342 ± 0.1344 c | 0.1474 ± 0.0024 c | 0.3786 ± 0.0077 b | 0.0324 ± 0.0007 bc |
| 100 Ser | 28.87 ± 0.5 d | 2.83 ± 0.05 ab | 2.1052 ± 0.0012 bc | 0.1639 ± 0.0046 b | 0.4002 ± 0.004 b | 0.0323 ± 0.0009 bc |
| 200 Ser | 26.75 ± 0.45 e | 2.63 ± 0.03 bc | 2.1825 ± 0.0009 b | 0.1681 ± 0.0025 b | 0.4330 ± 0.011 a | 0.0398 ± 0.0004 a |
| 300 Ser | 24.92 ± 0.26 f | 2.55 ± 0.16 c | 1.8171 ± 0.029 c | 0.1448 ± 0.0023 c | 0.3290 ± 0.0113 c | 0.0285 ± 0.0005 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Ding, X.; Lv, Y.; Cheng, Y.; Li, C.; Yan, L.; Tian, S.; Zou, X. Exogenous Serotonin Improves Salt Tolerance in Rapeseed (Brassica napus L.) Seedlings. Agronomy 2021, 11, 400. https://doi.org/10.3390/agronomy11020400
Liu Y, Ding X, Lv Y, Cheng Y, Li C, Yan L, Tian S, Zou X. Exogenous Serotonin Improves Salt Tolerance in Rapeseed (Brassica napus L.) Seedlings. Agronomy. 2021; 11(2):400. https://doi.org/10.3390/agronomy11020400
Chicago/Turabian StyleLiu, Yue, Xiaoyu Ding, Yan Lv, Yong Cheng, Chunsheng Li, Lei Yan, Shanshan Tian, and Xiling Zou. 2021. "Exogenous Serotonin Improves Salt Tolerance in Rapeseed (Brassica napus L.) Seedlings" Agronomy 11, no. 2: 400. https://doi.org/10.3390/agronomy11020400
APA StyleLiu, Y., Ding, X., Lv, Y., Cheng, Y., Li, C., Yan, L., Tian, S., & Zou, X. (2021). Exogenous Serotonin Improves Salt Tolerance in Rapeseed (Brassica napus L.) Seedlings. Agronomy, 11(2), 400. https://doi.org/10.3390/agronomy11020400






