Allelopathic Efficiency of Plant Extracts to Control Cyanobacteria in Hydroponic Culture
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Aqueous Extracts
2.2. Algae Culture
2.3. Hydroponic Culture
2.4. Measurement of Cyanobacteria and Plant Growth Parameters
2.5. Determination of Tannic Acid Using HPLC
2.6. Statistical Analyses
3. Results and Discussion
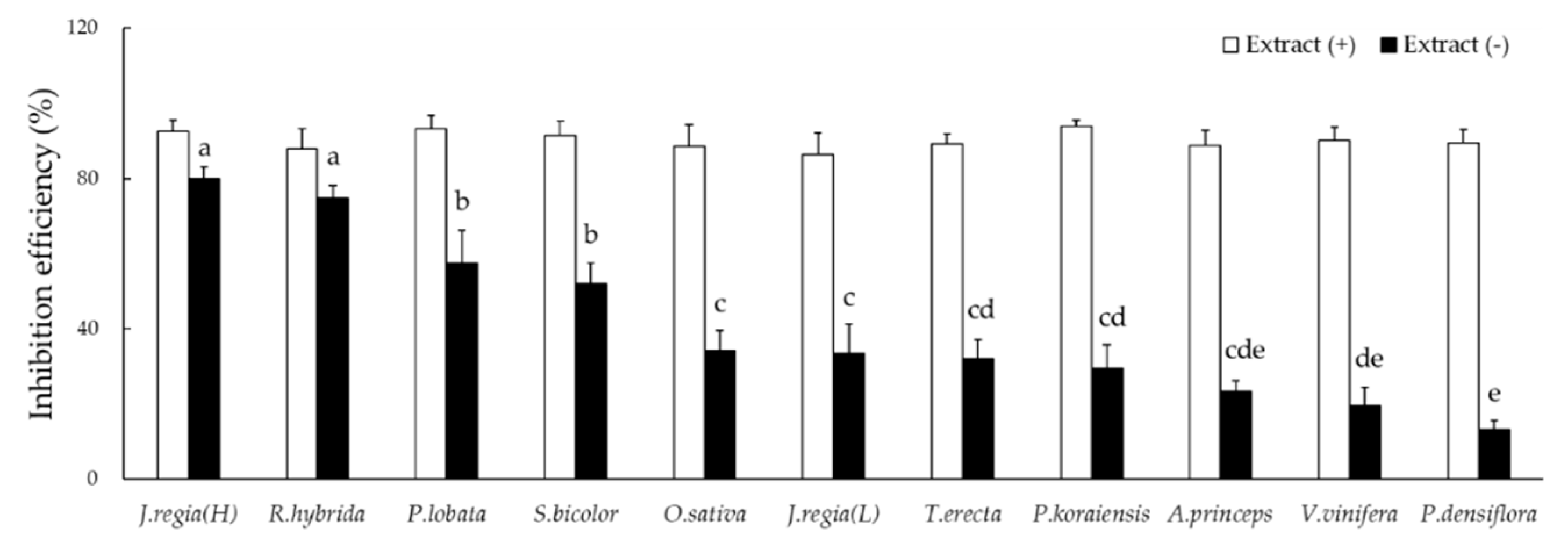
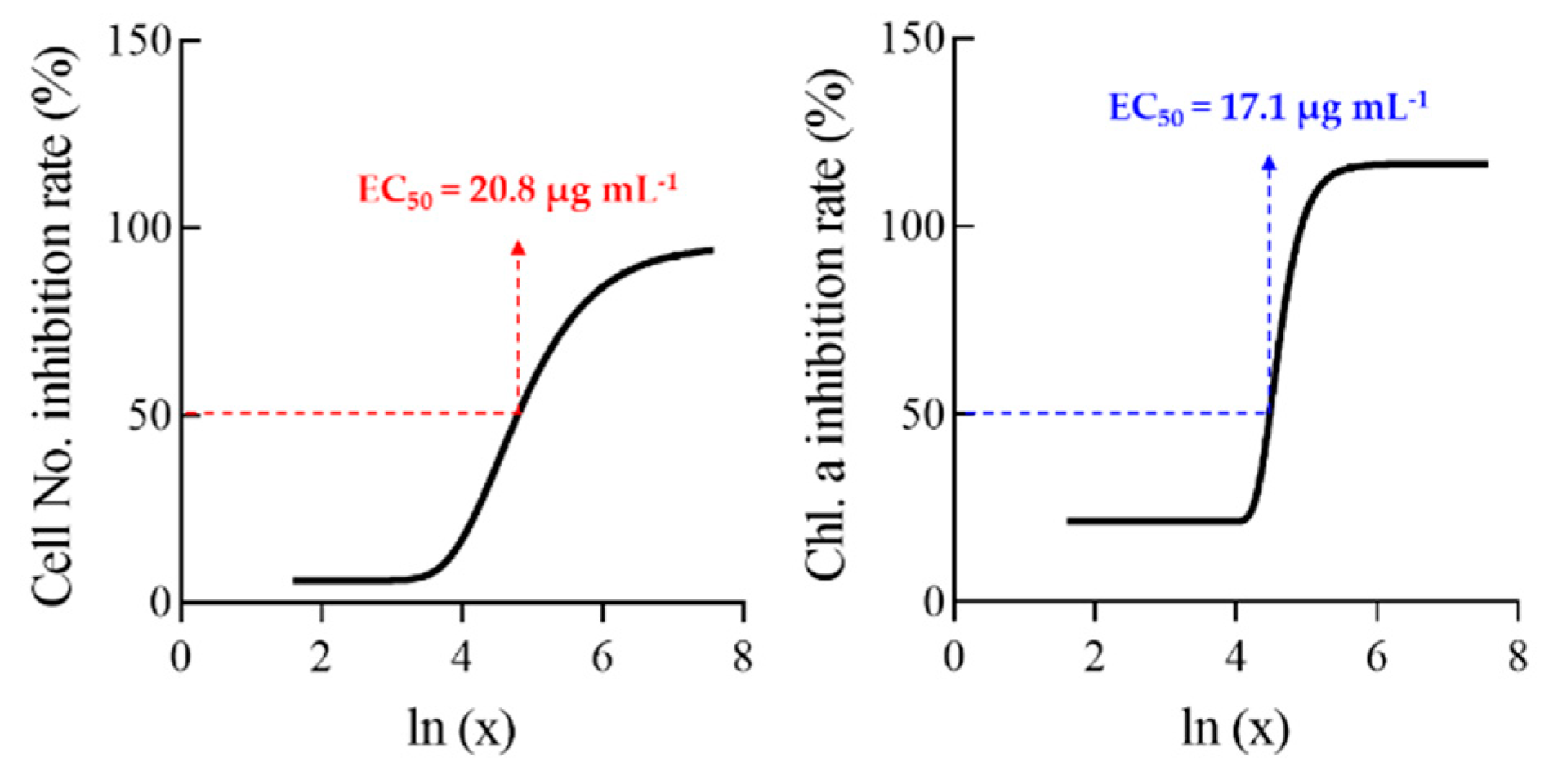
3.1. Effects of Allelopathic Extracts on Algal Inhibition
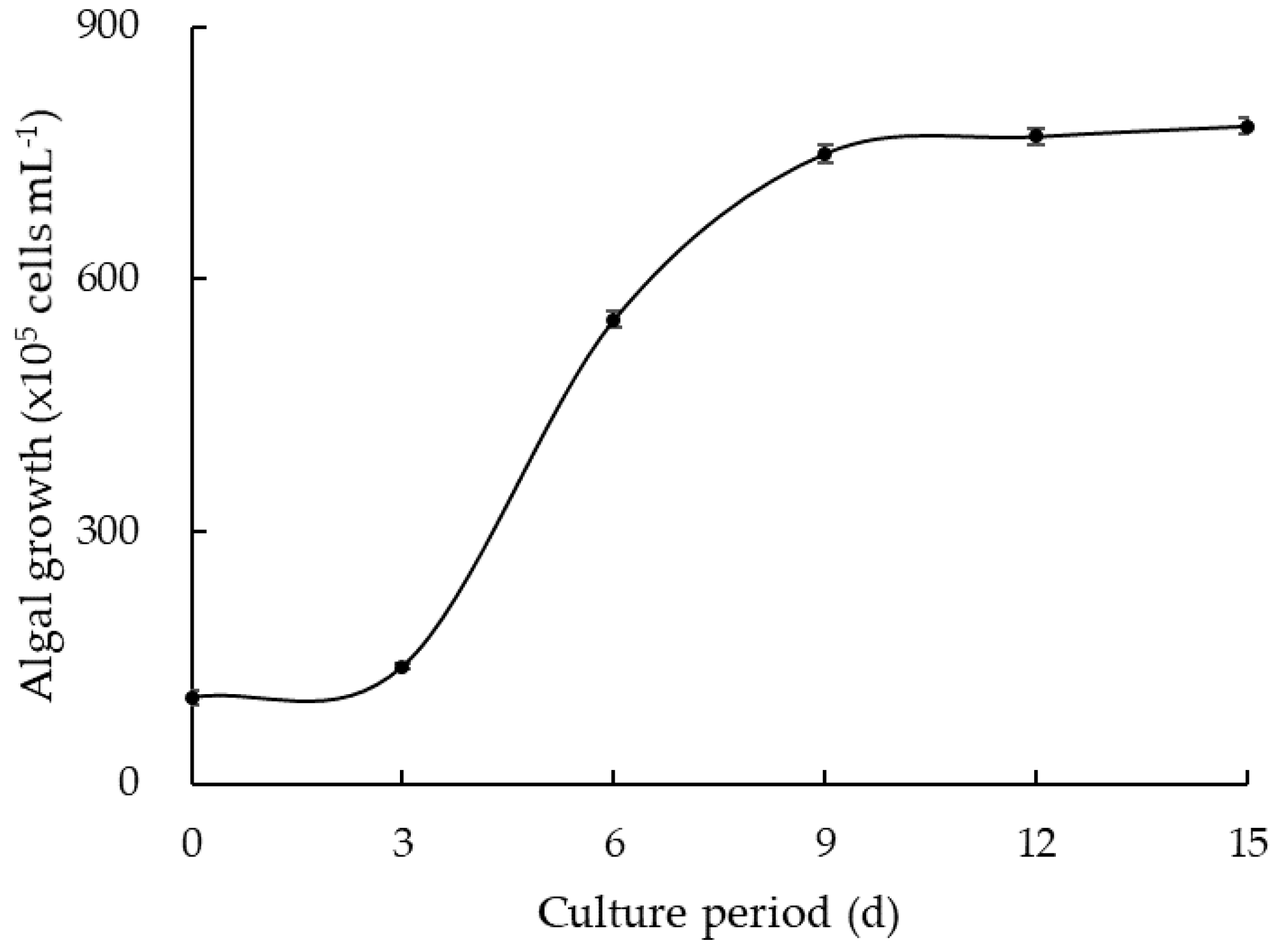
3.2. Algal Regrowth
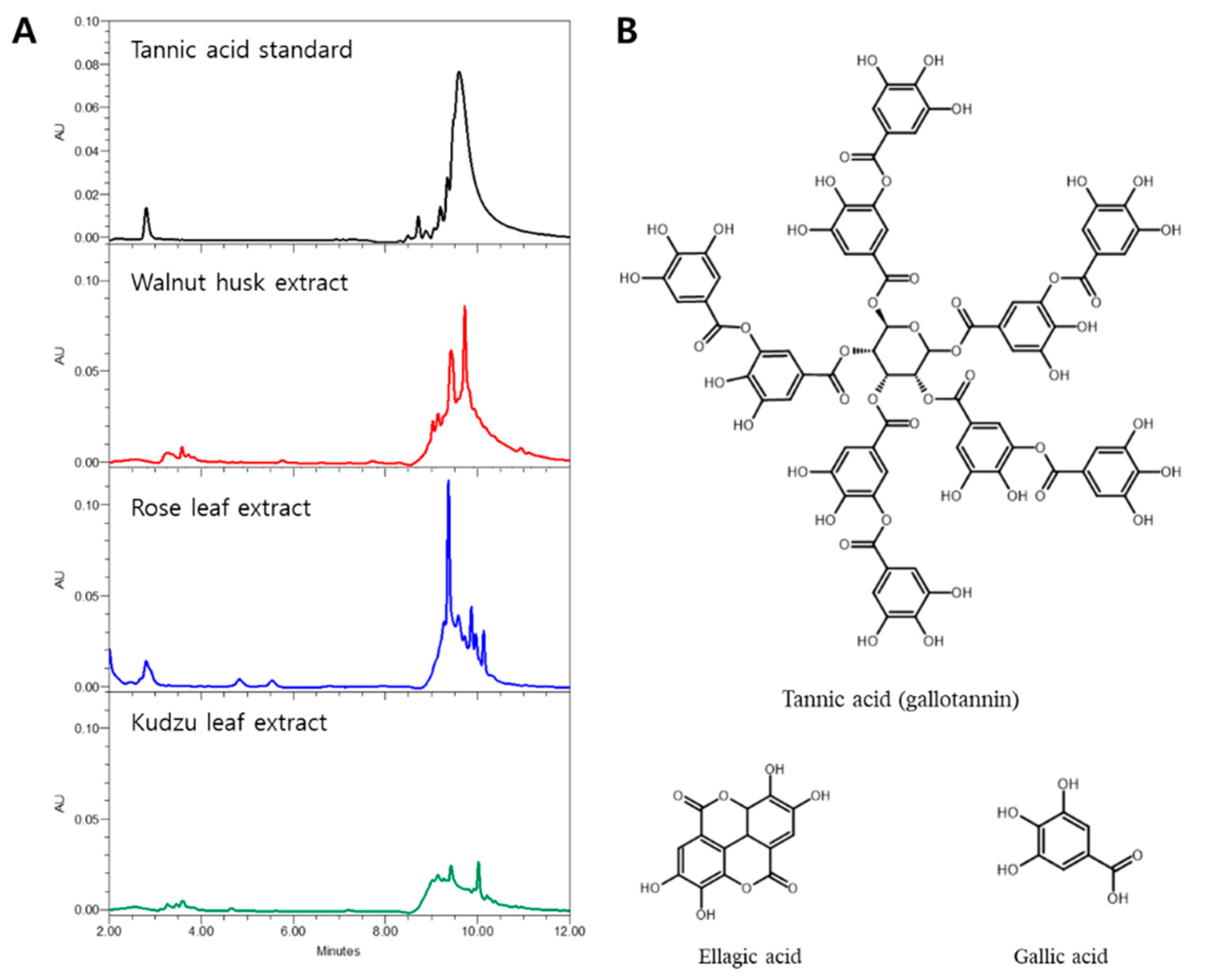
3.3. HPLC Profiles and Algae Inhibitory Effect of Tannic Acid
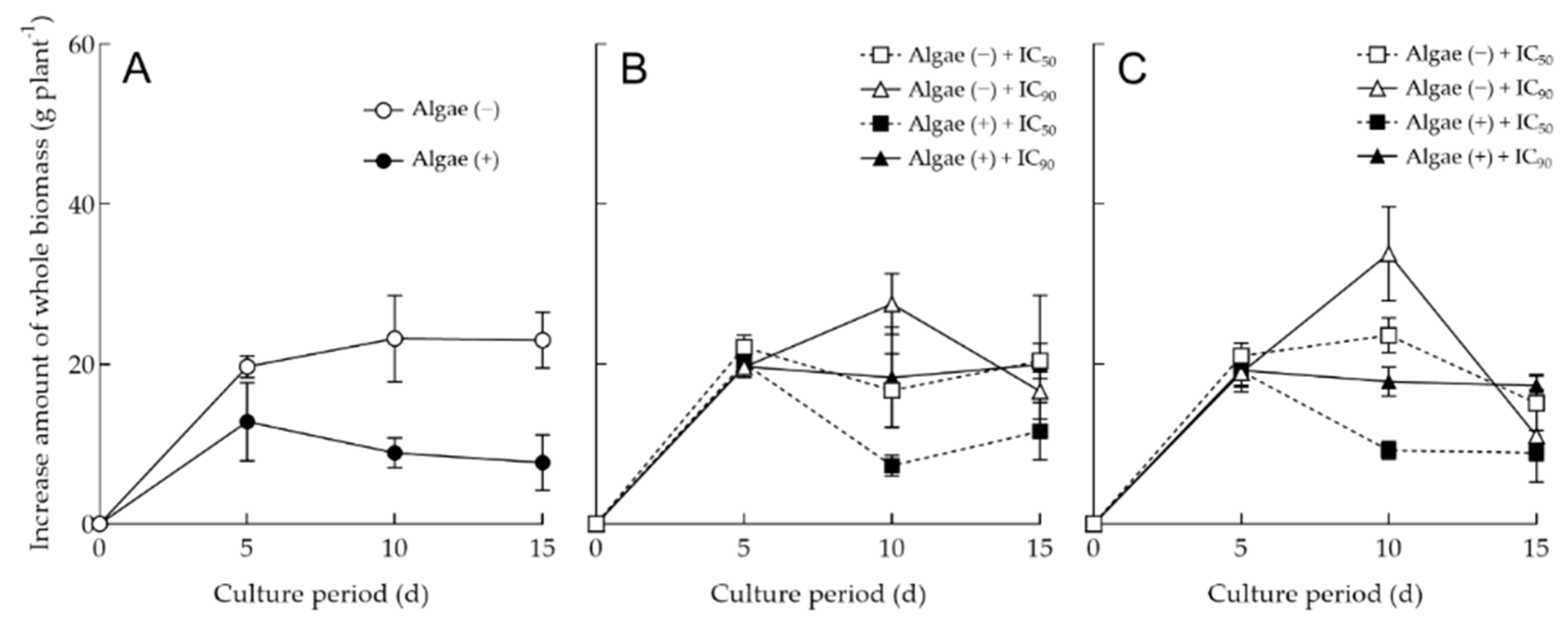
3.4. Application of Allelopathic Extracts in Hydroponic Culture
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Huo, S.; Liu, J.; Addy, M.; Chen, P.; Necas, D.; Cheng, P.; Li, K.; Chai, H.; Liu, Y.; Ruan, R. The influence of microalgae on vegetable production and nutrient removal in greenhouse hydroponics. J. Clean. Prod. 2019, 243, 118563. [Google Scholar] [CrossRef]
- Wada, T. Theory and Technology to Control the Nutrient Solution of Hydroponics. In Plant Factory using Artificial Light; Elsevier: Amsterdam, The Netherlands, 2019; pp. 5–14. [Google Scholar] [CrossRef]
- Hua, Q.; Liu, Y.-G.; Yan, Z.-L.; Zeng, G.-M.; Liu, S.-B.; Wang, W.-J.; Tan, X.; Deng, J.-Q.; Tang, X.; Wang, Q.-P. Allelopathic effect of the rice straw aqueous extract on the growth of Microcystis aeruginosa. Ecotoxicol. Environ. Saf. 2018, 148, 953–959. [Google Scholar] [CrossRef]
- Abdel-Raouf, N.; Al-Homaidan, A.A.; Ibraheem, I.B.M. Microalgae and wastewater treatment. Saudi J. Biol. Sci. 2012, 19, 257–275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Wang, X.; Zhou, Q. Co-cultivation of Chlorella spp and tomato in a hydroponic system. Biomass Bioenergy 2016, 97, 132–138. [Google Scholar] [CrossRef]
- Yang, J.; Deng, X.; Xian, Q.; Qian, X.; Li, A. Allelopathic effect of Microcystis aeruginosa on Microcystis wesenbergii: Microcystin-LR as a potential allelochemical. Hydrobiologia 2013, 727, 65–73. [Google Scholar] [CrossRef]
- Iredale, R.S.; McDonald, A.T.; Adams, D.G. A series of experiments aimed at clarifying the mode of action of barley straw in cyanobacterial growth control. Water Res. 2012, 46, 6095–6103. [Google Scholar] [CrossRef]
- Wang, J.; Peng, Y.; Wang, Z.; Zou, W.; Peng, X.; Song, Q. Transcriptional response of Microcystis aeruginosa to the recruitment promoting-benthic bacteria. J. Oceanol. Limnol. 2021, 1–10. [Google Scholar] [CrossRef]
- Li, J.; Hu, J.; Cao, L.; Yuan, Y. Growth, physiological responses and microcystin-production/-release dynamics of Microcystis aeruginosa exposed to various luteolin doses. Ecotoxicol. Environ. Saf. 2020, 196, 110540. [Google Scholar] [CrossRef]
- Jančula, D.; Maršálek, B. Critical review of actually available chemical compounds for prevention and management of cyanobacterial blooms. Chemosphere 2011, 85, 1415–1422. [Google Scholar] [CrossRef]
- Eom, S.H.; Yang, H.S.; Weston, L.A. An Evaluation of the Allelopathic Potential of Selected Perennial Groundcovers: Foliar Volatiles of Catmint (Nepeta × faassenii) Inhibit Seedling Growth. J. Chem. Ecol. 2006, 32, 1835–1848. [Google Scholar] [CrossRef]
- Maksimovi, T.; Hasanagi, D.; Sciences, N.; Luka, B.; Stojanovi, M.; Luka, B. Allelopathic influence of Juglans Regia L. aqueous extract on the germination and the growth of lettuce and tomato. J. Agri. Food Environ. Sci. 2020, 74, 5–12. [Google Scholar]
- Chung, I.-M.; Park, S.-K.; Ali, M.; Prabakaran, M.; Oh, Y.-T.; Kim, S.-H.; Siddiqui, N.A.; Ahmad, A. Flavonoid glycosides from leaves and straw of Oryza sativa and their effects of cytotoxicity on a macrophage cell line and allelopathic on weed germination. Saudi Pharm. J. 2018, 26, 375–387. [Google Scholar] [CrossRef]
- Kobayashi, K.; Sasamoto, H.; Sasamoto, Y.; Sugiyama, A.; Fujii, Y. Evaluation of Isoflavones as Allelochemicals with Strong Allelopathic Activities of Kudzu Using Protoplast Co-Culture Method with Digital Image Analysis. Am. J. Plant Sci. 2021, 12, 376–393. [Google Scholar] [CrossRef]
- Weston, L.A.; Alsaadawi, I.S.; Baerson, S.R. Sorghum Allelopathy—From Ecosystem to Molecule. J. Chem. Ecol. 2013, 39, 142–153. [Google Scholar] [CrossRef]
- Arora, K.; Batish, D.R.; Kohli, R.K.; Singh, H.P. Allelopathic impact of essential oil of Tagetes minuta on common agricultural and wasteland weeds. Innovare J. Agric. Sci. 2017, 5, 1–4. [Google Scholar]
- Neilen, A.D.; Hawker, D.W.; O’Brien, K.R.; Burford, M.A. Phytotoxic effects of terrestrial dissolved organic matter on a freshwater cyanobacteria and green algae species is affected by plant source and DOM chemical composition. Chemosphere 2017, 184, 969–980. [Google Scholar] [CrossRef]
- Yuan, R.; Li, Y.; Li, J.; Ji, S.; Wang, S.; Kong, F. The allelopathic effects of aqueous extracts from Spartina alterniflora on controlling the Microcystis aeruginosa blooms. Sci. Total Environ. 2019, 712, 136332. [Google Scholar] [CrossRef]
- Wu, L.; Guo, X.; Harivandi, M. Allelopathic effects of phenolic acids detected in buffalograss (Buchloe dactyloides) clippings on growth of annual bluegrass (Poa annua) and buffalograss seedlings. Environ. Exp. Bot. 1998, 39, 159–167. [Google Scholar] [CrossRef]
- López, M.L.; Bonzani, N.E.; Zygadlo, J.A. Allelopathic potential of Tagetes minuta terpenes by a chemical, anatomical and phytotoxic approach. Biochem. Syst. Ecol. 2008, 36, 882–890. [Google Scholar] [CrossRef]
- de Arruda, G.L.; de Moraes, G.K.A.; Junior, A.F.C.; Araujo, A.R.; Chapla, V.M. Aromatic compounds from the endophytic fungus Asordaria conoidea and their allelochemical property using OSMAC strategy. Nat. Prod. Res. 2021, 1–4. [Google Scholar] [CrossRef]
- Yu, S.; Li, C.; Xu, C.; Effiong, K.; Xiao, X. Understanding the inhibitory mechanism of antialgal allelochemical flavonoids from genetic variations: Photosynthesis, toxin synthesis and nutrient utility. Ecotoxicol. Environ. Saf. 2019, 177, 18–24. [Google Scholar] [CrossRef]
- Ogunsusi, M.; Akinlalu, A.O.; Komolafe, I.J.; Oyedapo, O.O. Allelopathic effects of alkaloid fraction of Crotalaria retusa Linn on growth and some biochemical parameters of bean seedlings (Phaseolus vulgaris). Int. J. Plant Physiol. Biochem. 2018, 10, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Patiño, R.; Rashel, R.H.; Rubio, A.; Longing, S. Growth-suppressing and algicidal properties of an extract from Arundo donax, an invasive riparian plant, against Prymnesium parvum, an invasive harmful alga. Harmful Algae 2018, 71, 1–9. [Google Scholar] [CrossRef]
- Tebaa, L.; Douma, M.; Tazart, Z.; Manaut, N.; Mouhri, K.H.; Loudiki, M. Assessment of the potentially algicidal effects of Thymus satureioides Coss. and Artemisia herba-alba L. against Microcystis Aeruginosa. Appl. Ecol. Environ. Res. 2018, 16, 903–912. [Google Scholar] [CrossRef]
- Rodolfi, L.; Chini Zittelli, G.; Bassi, N.; Padovani, G.; Biondi, N.; Bonini, G.; Tredici, M.R. Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol. Bioeng. 2009, 102, 100–112. [Google Scholar] [CrossRef]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. Circ. Calif. Agric. Exp. Stn. 1950, 347, 32. [Google Scholar]
- Nguyen, T.; Yeom, M.-S.; Oh, M.-M. Effect of a Newly-Developed Nutrient Solution and Electrical Conductivity on Growth and Bioactive Compounds in Perilla frutescens var. crispa. Agronomy 2021, 11, 932. [Google Scholar] [CrossRef]
- Zhao, W.; Zheng, Z.; Zhang, J.; Roger, S.-F.; Luo, X. Allelopathically inhibitory effects of eucalyptus extracts on the growth of Microcystis aeruginosa. Chemosphere 2019, 225, 424–433. [Google Scholar] [CrossRef]
- Lim, Y.J.; Eom, S.H. Effects of different light types on root formation of Ocimum basilicum L. cuttings. Sci. Hortic. 2013, 164, 552–555. [Google Scholar] [CrossRef]
- Gu, H.-F.; Li, C.-M.; Xu, Y.-J.; Hu, W.-F.; Chen, M.-H.; Wan, Q.-H. Structural features and antioxidant activity of tannin from persimmon pulp. Food Res. Int. 2008, 41, 208–217. [Google Scholar] [CrossRef]
- Kwak, H.; Hegeman, A.; Park, S. Seasonal changes in metabolic profiles of galls and leaves of Rhus chinensis using gas chromatography mass spectrometry and liquid chromatography quadrupole time-of-flight mass spectrometry. J. Plant Biol. 2014, 57, 127–135. [Google Scholar] [CrossRef]
- Shen, X.; Su, Y.-C.; Liu, C.; Oscar, T.; DePaola, A. Efficacy of Vibrio parahaemolyticus depuration in oysters (Crassostrea gigas). Food Microbiol. 2018, 79, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Canton, M.C.; Holguin, F.O.; Boeing, W.J. Alkaloid gramine to control algal invaders: Algae inhibition and gramine persistence. Bioresour. Technol. Rep. 2019, 7, 100304. [Google Scholar] [CrossRef]
- Jose, S.; Gillespie, A.R. Allelopathy in black walnut (Juglans nigra L.) alley cropping. I. Spatio-temporal variation in soil juglone in a black walnut–corn (Zea mays L.) alley cropping system in the midwestern USA. Plant Soil 1998, 203, 191–197. [Google Scholar] [CrossRef]
- Terzi, I. Allelopathic effects of juglone and decomposed walnut leaf juice on muskmelon and cucumber seed germination and seedling growth. Afr. J. Biotechnol. 2008, 7, 1870–1874. [Google Scholar]
- Jahanban-Esfahlan, A.; Ostadrahimi, A.; Tabibiazar, M.; Amarowicz, R.; Esfahlan, J. A Comprehensive Review on the Chemical Constituents and Functional Uses of Walnut (Juglans spp.) Husk. Int. J. Mol. Sci. 2019, 20, 3920. [Google Scholar] [CrossRef] [Green Version]
- Švanys, A.; Eigemann, F.; Grossart, H.-P.; Hilt, S. Microcystins do not necessarily lower the sensitivity of Microcystis aeruginosato tannic acid. FEMS Microbiol. Lett. 2015, 363, fnv227. [Google Scholar] [CrossRef] [Green Version]
- Zhao, G.; Watson, J.; Crowder, C.; Stevens, J.S.E. Changes in biological production of the cyanobacterium, Nostoc sp. strain MAC, under subinhibitory concentrations of tannic acid and related compounds. J. Appl. Phycol. 1998, 10, 1–7. [Google Scholar] [CrossRef]
- Romani, A.; Campo, M.; Pinelli, P. HPLC/DAD/ESI-MS analyses and anti-radical activity of hydrolyzable tannins from different vegetal species. Food Chem. 2012, 130, 214–221. [Google Scholar] [CrossRef]
- Soong, Y.; Barlow, P. Quantification of gallic acid and ellagic acid from longan (Dimocarpus longan Lour.) seed and mango (Mangifera indica L.) kernel and their effects on antioxidant activity. Food Chem. 2006, 97, 524–530. [Google Scholar] [CrossRef]
- Laue, P.; Bährs, H.; Chakrabarti, S.; Steinberg, C.E. Natural xenobiotics to prevent cyanobacterial and algal growth in freshwater: Contrasting efficacy of tannic acid, gallic acid, and gramine. Chemosphere 2014, 104, 212–220. [Google Scholar] [CrossRef]
- Oliveira, I.; Sousa, A.; Ferreira, I.; Bento, A.A.; Estevinho, M.L.M.F.; Pereira, J.A. Total phenols, antioxidant potential and antimicrobial activity of walnut (Juglans regia L.) green husks. Food Chem. Toxicol. 2008, 46, 2326–2331. [Google Scholar] [CrossRef]
- Carvalho, M.; Ferreira, P.J.; Mendes, V.S.; Silva, R.; Pereira, J.A.; Jeronimo, C.; Silva, B.M. Human cancer cell antiproliferative and antioxidant activities of Juglans regia L. Food Chem. Toxicol. 2010, 48, 441–447. [Google Scholar] [CrossRef]
- Maleita, C.; Esteves, I.; Chim, R.; Fonseca, L.; Braga, M.E.M.; Abrantes, I.; de Sousa, H.C. Naphthoquinones from Walnut Husk Residues Show Strong Nematicidal Activities against the Root-knot Nematode Meloidogyne hispanica. ACS Sustain. Chem. Eng. 2017, 5, 3390–3398. [Google Scholar] [CrossRef]
- Cunja, V.; Mikulic-Petkovsek, M.; Stampar, F.; Schmitzer, V. Compound Identification of Selected Rose Species and Cultivars: An Insight to Petal and Leaf Phenolic Profiles. J. Am. Soc. Hortic. Sci. 2014, 139, 157–166. [Google Scholar] [CrossRef] [Green Version]
- Riffault, L.; Destandau, E.; Pasquier, L.; André, P.; Elfakir, C. Phytochemical analysis of Rosa hybrida cv. ‘Jardin de Granville’ by HPTLC, HPLC-DAD and HPLC-ESI-HRMS: Polyphenolic fingerprints of six plant organs. Phytochemistry 2014, 99, 127–134. [Google Scholar] [CrossRef]
- Jakopic, J.; Colaric, M.; Veberic, R.; Hudina, M.; Solar, A.; Stampar, F. How much do cultivar and preparation time influence on phenolics content in walnut liqueur? Food Chem. 2007, 104, 100–105. [Google Scholar] [CrossRef]
- Bayona-Morcillo, P.J.; Plaza, B.M.; Gómez-Serrano, C.; Rojas, E.; Jiménez-Becker, S. Effect of the foliar application of cyanobacterial hydrolysate (Arthrospira platensis) on the growth of Petunia x hybrida under salinity conditions. J. Appl. Phycol. 2020, 32, 4003–4011. [Google Scholar] [CrossRef]
- Castro-Castellon, A.T.; Hughes, J.M.R.; Read, D.S.; Azimi, Y.; Chipps, M.J.; Hankins, N.P. The role of rhizofiltration and allelopathy on the removal of cyanobacteria in a continuous flow system. Environ. Sci. Pollut. Res. 2021, 28, 27731–27741. [Google Scholar] [CrossRef]
- Supraja, K.V.; Behera, B.; Balasubramanian, P. Performance evaluation of hydroponic system for co-cultivation of microalgae and tomato plant. J. Clean. Prod. 2020, 272, 122823. [Google Scholar] [CrossRef]
- Schwarz, D.; Gross, W. Algae affecting lettuce growth in hydroponic systems. J. Hortic. Sci. Biotechnol. 2004, 79, 554–559. [Google Scholar] [CrossRef]
- Kraus, T.E.C.; Dahlgren, R.; Zasoski, R.J. Tannins in nutrient dynamics of forest ecosystems—A review. Plant Soil 2003, 256, 41–66. [Google Scholar] [CrossRef]
- Rudrappa, T.; Bonsall, J.; Gallagher, J.L.; Seliskar, D.M.; Bais, H.P. Root-secreted Allelochemical in the Noxious Weed Phragmites australis Deploys a Reactive Oxygen Species Response and Microtubule Assembly Disruption to Execute Rhizotoxicity. J. Chem. Ecol. 2007, 33, 1898–1918. [Google Scholar] [CrossRef]
- Kinraide, T.B.; Hagermann, A.E. Interactive intoxicating and ameliorating effects of tannic acid, aluminum (Al3+), copper (Cu2+), and selenate (SeO42−) in wheat roots: A descriptive and mathematical assessment. Physiol. Plant. 2010, 139, 68–79. [Google Scholar] [CrossRef]
- Arowosegbe, S.; Wintola, O.A.; Afolayan, A.J. Phytochemical constituents and allelopathic effect of Aloe ferox Mill. root extract on tomato. J. Med. Plants Res. 2012, 6, 2094–2099. [Google Scholar] [CrossRef]

| Species | Common Name | Organ Used |
|---|---|---|
| Artemisia princeps | Mugwort | Leaf |
| Juglans regia | Walnut | Leaf (L) |
| Juglans regia | Walnut | Green husk (H) |
| Oryza sativa | Rice | Leaf |
| Pinus densiflora | Pine | Leaf |
| Pinus koraiensis | Nut pine | Leaf |
| Pueraria lobate | Kudzu | Leaf |
| Rosa hybrida | Rose | Leaf |
| Sorghum bicolor | Sorghum | Leaf |
| Tagetes erecta | Marigold | Leaf |
| Vitis vinifera | Grape | Leaf |
| Species | IC50 (g L−1) | IC90 (g L−1) | Equation 1 | r 2 |
|---|---|---|---|---|
| R. hybrida | 0.6 ± 0.04 d2 | 3.5 ± 0.13 d | y = 84.66e−0.61x | 0.977 |
| J. regia (L) | 1.2 ± 0.03 c | 4.1 ± 0.11 cd | y = 94.13e−0.51x | 0.973 |
| P. densiflora | 1.3 ± 0.06 c | 4.3 ± 0.23 cd | y = 101.02e−0.53x | 0.949 |
| J. regia (H) | 1.4 ± 0.07 c | 4.6 ± 0.22 bcd | y = 106.11e−0.5x | 0.960 |
| T. erecta | 1.5 ± 0.10 bc | 5.0 ± 0.31 bc | y = 101.56e−0.45x | 0.972 |
| A. princeps | 1.5 ± 0.01 bc | 5.1 ± 0.14 bc | y = 100.14e−0.45x | 0.964 |
| V. vinifera | 1.4 ± 0.06 c | 5.2 ± 0.23 bc | y = 101.43e−0.74x | 0.971 |
| O. sativa | 1.4 ± 0.07 c | 5.3 ± 0.26 bc | y = 86.79e−0.41x | 0.971 |
| S. bicolor | 1.7 ± 0.05 b | 5.7 ± 0.24b | y = 101.38e−0.40 x | 0.986 |
| P. koraiensis | 2.2 ± 0.07 a | 7.4 ± 0.34 a | y = 97.84e−0.31x | 0.971 |
| P. lobata | 2.4 ± 0.08 a | 7.7 ± 0.21 a | y = 104.64e−0.31x | 0.983 |
| Extract | Concentration (g L−1) | Algae Inoculation | Algae Harvest 1 (×105 cells mL−1) | Leaf Biomass (g DW leaf−1) | Chlorophyll Content (mg g−1 DW) |
|---|---|---|---|---|---|
| Non-treatment | 0 | − | 0 | 0.52 ± 0.00 abc 2 | 10.09 ± 0.28 abc |
| 0 | + | 579 ± 1.93 a | 0.38 ± 0.06 d | 8.67 ± 0.54 de | |
| Rose leaf | 0.6 (IC50) | − | 0 | 0.56 ± 0.01 a | 10.72 ± 0.41 a |
| 0.6 (IC50) | + | 263 ± 1.59 b | 0.42 ± 0.01 cd | 10.13 ± 0.16 ab | |
| 3.5 (IC90) | − | 0 | 0.54 ± 0.03 ab | 10.72 ± 0.34 a | |
| 3.5 (IC90) | + | 103 ± 2.19 d | 0.49 ± 0.04 abc | 9.34 ± 0.20 bcd | |
| Walnut husk | 1.4 (IC50) | − | 0 | 0.46 ± 0.03 abcd | 9.31 ± 0.21 bcd |
| 1.4 (IC50) | + | 172 ± 1.37 c | 0.51 ± 0.01 abc | 8.81 ± 0.74 cde | |
| 4.6 (IC90) | − | 0 | 0.45 ± 0.04 bcd | 7.94 ± 0.15e | |
| 4.6 (IC90) | + | 105 ± 1.39 d | 0.45 ± 0.03 bcd | 6.52 ± 0.21 f |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gil, C.S.; Duan, S.; Kim, J.H.; Eom, S.H. Allelopathic Efficiency of Plant Extracts to Control Cyanobacteria in Hydroponic Culture. Agronomy 2021, 11, 2350. https://doi.org/10.3390/agronomy11112350
Gil CS, Duan S, Kim JH, Eom SH. Allelopathic Efficiency of Plant Extracts to Control Cyanobacteria in Hydroponic Culture. Agronomy. 2021; 11(11):2350. https://doi.org/10.3390/agronomy11112350
Chicago/Turabian StyleGil, Chan Saem, Shucheng Duan, Ji Hye Kim, and Seok Hyun Eom. 2021. "Allelopathic Efficiency of Plant Extracts to Control Cyanobacteria in Hydroponic Culture" Agronomy 11, no. 11: 2350. https://doi.org/10.3390/agronomy11112350
APA StyleGil, C. S., Duan, S., Kim, J. H., & Eom, S. H. (2021). Allelopathic Efficiency of Plant Extracts to Control Cyanobacteria in Hydroponic Culture. Agronomy, 11(11), 2350. https://doi.org/10.3390/agronomy11112350






