Sensory Analyses and Nutritional Qualities of Wheat Population Varieties Developed by Participatory Breeding
Abstract
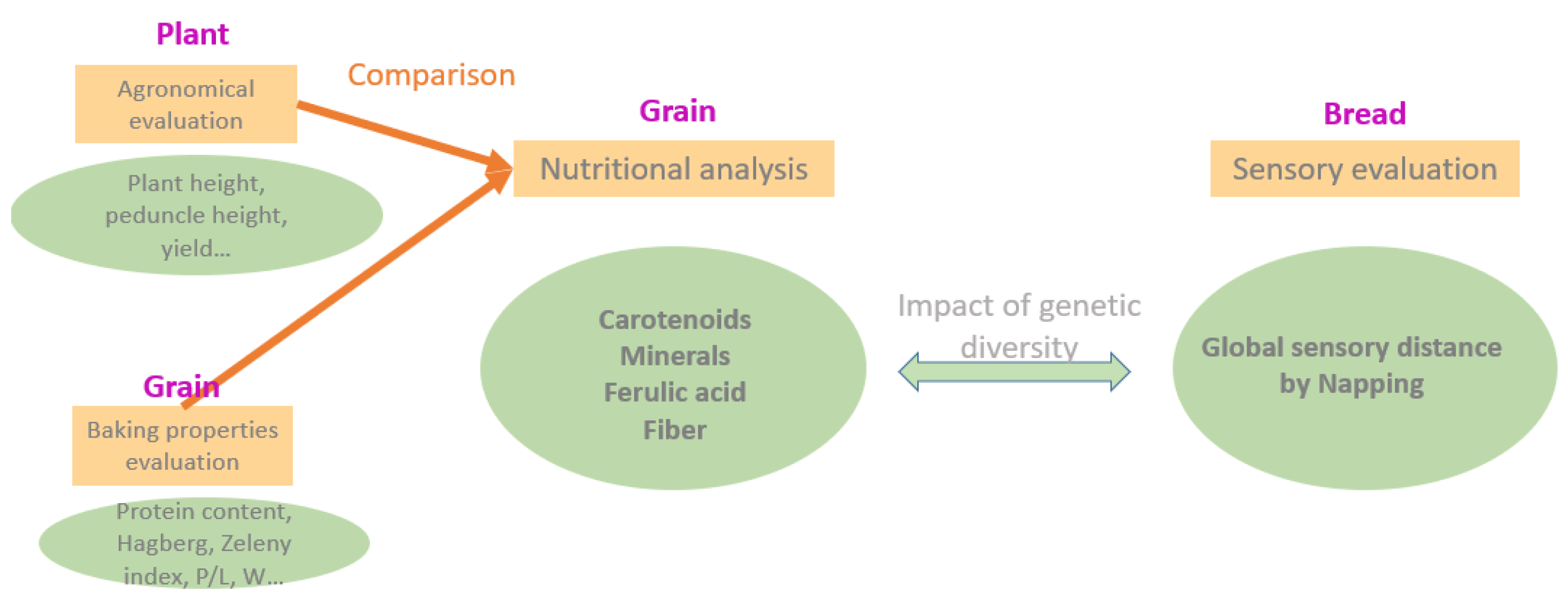
:1. Introduction
2. Materials and Methods
2.1. Wheat PPB Populations and Commercial Varieties Studied
2.2. Dough and Baking Properties Evaluation
2.3. Nutritional Analyses
2.4. Sensory Evaluation
2.4.1. Bread Making
2.4.2. Napping Method
2.5. Statistical Analyses
2.5.1. Technological and Nutritional Characterization
2.5.2. Napping Test
2.5.3. Correlation between Nutritional, Technologic, and Agronomic Properties
3. Results
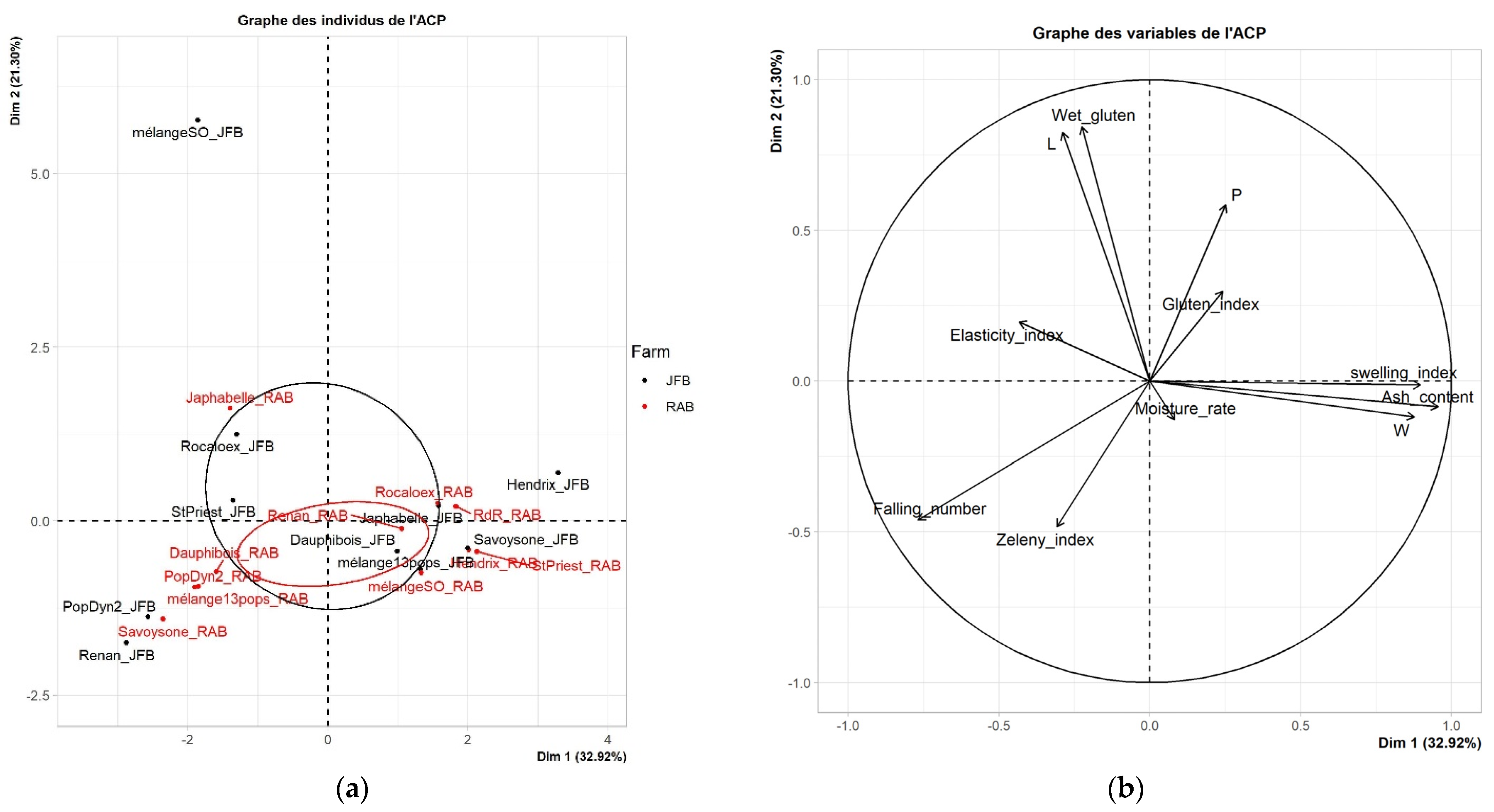
3.1. Technological Characterization
3.2. Nutritional Analyses
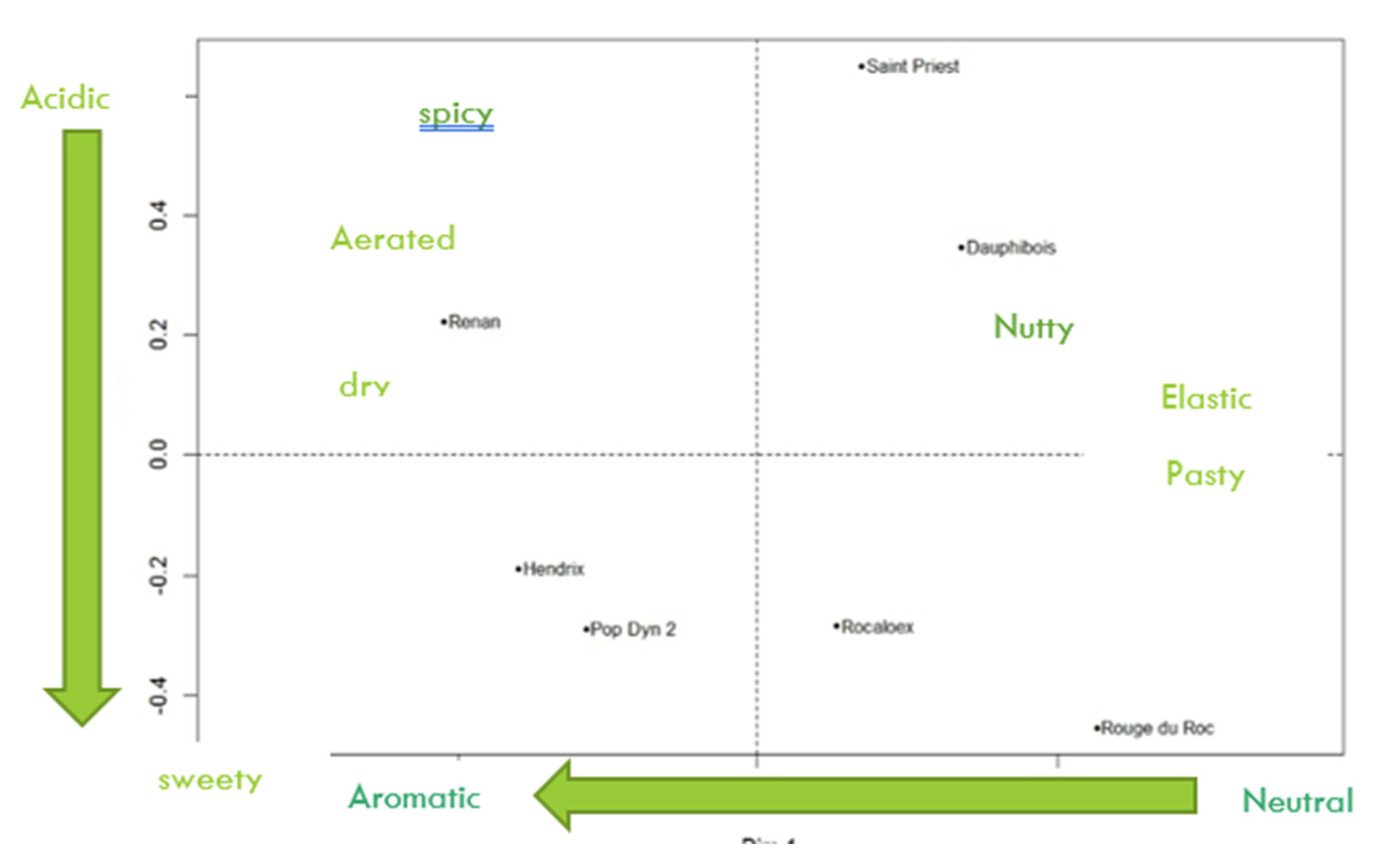
3.3. Napping Test
3.4. Correlation between Nutritional, Technologic, and Agronomic Properties
4. Discussion
4.1. Nutritional and Sensory Quality of PPB Varieties
4.2. Correlation between Agronomic and Nutritionnal Traits
4.3. Fine Tuning of Bread Making Process to Compare Diverse Varieties
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Appendix A. Nutritional Content of the 22 Wheat Samples
| Sample | Varieties | Farm | Ca | Mg | K | Cu | Zn | Se | Fibers | Lutein |
|---|---|---|---|---|---|---|---|---|---|---|
| Dauphibois_JFB | Dauphibois | JFB | 41.8 | 120 | 434.3 | 0.5 | 3.6 | 163 | 1.3 | 1.2 |
| Japhabelle_JFB | Japhabelle | JFB | 45.5 | 126.6 | 447 | 0.5 | 3.8 | 0 | 3.1 | 1.3 |
| mélange13pops_JFB | mélange13pops | JFB | 40.4 | 121.2 | 434.5 | 0 | 3.6 | 51 | 3.1 | 1.2 |
| mélangeSO_JFB | mélangeSO | JFB | 42.2 | 126.5 | 444.6 | 0 | 4 | 67 | 3 | 1.3 |
| PopDyn2_JFB | PopDyn2 | JFB | 42.2 | 131.5 | 431.4 | 0 | 4.2 | 158 | 2 | 1.1 |
| RdR_JFB | RdR | JFB | 71 | 240.7 | 745.6 | 0.7 | 7.7 | 50 | 1.8 | 1.3 |
| Rocaloex_JFB | Rocaloex | JFB | 43.6 | 126.5 | 450.3 | 0.5 | 4.1 | 0 | 1.7 | 1.2 |
| Savoysone_JFB | Savoysone | JFB | 55.8 | 136.1 | 487.8 | 0.5 | 4 | 0 | 2.1 | 1.2 |
| StPriest_JFB | StPriest | JFB | 37.37 | 133 | 369.6 | 0.5 | 4.9 | 0 | 1.8 | 0 |
| Hendrix_JFB | Hendrix | JFB | 49.4 | 105.1 | 453.3 | 0 | 2.7 | 205 | 2.6 | 1.3 |
| Renan_JFB | Renan | JFB | 42.8 | 115 | 484.6 | 0 | 3.8 | 53 | 1.8 | 1.2 |
| Dauphibois_RAB | Dauphibois | RAB | 39.7 | 133.1 | 393.6 | 0.6 | 4 | 169 | 2 | 1.3 |
| Japhabelle_RAB | Japhabelle | RAB | 39.5 | 136.1 | 374.8 | 0.5 | 4.6 | 0 | 2.2 | 1.4 |
| mélange13pops_RAB | mélange13pops | RAB | 37.2 | 128 | 388.6 | 0.5 | 4 | 0 | 2.5 | 1.3 |
| mélangeSO_RAB | mélangeSO | RAB | 38.5 | 143 | 427.5 | 0.5 | 4.8 | 0 | 2.2 | 1.4 |
| PopDyn2_RAB | PopDyn2 | RAB | 33.5 | 127.9 | 348 | 0.5 | 4.4 | 0 | 2 | 1.4 |
| RdR_RAB | RdR | RAB | 44.7 | 150.2 | 376.5 | 0.6 | 5 | 0 | 2.4 | 1.4 |
| Rocaloex_RAB | Rocaloex | RAB | 43.3 | 124.5 | 357.9 | 0.5 | 3.9 | 0 | 2 | 1.3 |
| Savoysone_RAB | Savoysone | RAB | 42 | 119 | 413.5 | 0.6 | 3.9 | 0 | 2.4 | 1.5 |
| StPriest_RAB | StPriest | RAB | 33.6 | 138.8 | 361 | 0.5 | 5.2 | 0 | 2.2 | 1.2 |
| Hendrix_RAB | Hendrix | RAB | 34.9 | 105.1 | 380.2 | 0 | 3.1 | 0 | 2.2 | 1.6 |
| Renan-RAB | Renan | RAB | 39.3 | 120.6 | 380.4 | 0.5 | 3.6 | 0 | 2 | 1.5 |

Appendix B
| Dimension 1 | Dimension 2 | Dimension 3 | ||||||
|---|---|---|---|---|---|---|---|---|
| Variable | Correlation | p-Value | Variable | Correlation | p-Value | Variable | Correlation | p-Value |
| Grain yield | 5.91 × 10−1 | 4.75 × 10−3 | Ash content | 7.85 × 10−1 | 2.46 × 10−5 | Curve | 7.33 × 10−1 | 1.56 × 10−4 |
| Potassium | 5.44 × 10−1 | 1.07 × 10−2 | W | 7.66 × 10−1 | 5.08 × 10−5 | % Sterile kernels | 6.51 × 10−1 | 1.38 × 10−3 |
| Awns | 5.12 × 10−1 | 1.75 × 10−2 | Swelling index | 7.31 × 10−1 | 1.66 × 10−4 | P | 6.44 × 10−1 | 1.64 × 10−3 |
| P | 4.59 × 10−1 | 3.63 × 10−2 | Spike weight | 4.57 × 10−1 | 3.71 × 10−2 | Colors | 5.24 × 10−1 | 1.47 × 10−2 |
| Selenium | 4.54 × 10−1 | 3.87 × 10−2 | Ferulic acid | −4.81 × 10−1 | 2.74 × 10−2 | Nb of spikelet per spike | 5.21 × 10−1 | 1.56 × 10−2 |
| Calcium | 4.45 × 10−1 | 4.29 × 10−2 | Hagberg | −7.17 × 10−1 | 2.51 × 10−4 | Wet gluten | 4.51 × 10−1 | 4.00 × 10−2 |
| Protein content | −4.82 × 10−1 | 2.68 × 10−2 | ||||||
| Copper | −6.67 × 10−1 | 9.64 × 10−4 | ||||||
| Colors | −6.75 × 10−1 | 7.81 × 10−4 | ||||||
| Plant height | −7.7 × 10−1 | 4.46 × 10−5 | ||||||
| Magnesium | −8.00 × 10−1 | 1.33 × 10−5 | ||||||
| LLSD | −8.50 × 10−1 | 1.08 × 10−6 | ||||||
| zinc | −9.02 × 10−1 | 2.40 × 10−6 | ||||||
| Variable | R2 | p-value | Variable | R2 | p-value | Variable | R2 | p-value |
| Farm | 2.05 × 10−1 | 3.95 × 10−2 | Farm | 2.36 × 10−1 | 2.53 × 10−2 | |||
| JFB farm | 6.18 × 10−1 | 3.95 × 10−3 | RAB farm | 5.18 × 10−1 | 2.53 × 10−2 | |||
| Hendrix | 2.85 | 9.72 × 10−4 | ||||||
| St Priest | −1.79 | 4.58 × 10−2 | ||||||
References
- Dinu, M.; Whittaker, A.; Pagliai, G.; Benedettelli, S.; Sofi, F. Ancient wheat species and human health: Biochemical and clinical implications. J. Nutr. Biochem. 2018, 52, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, S.L.; van Bueren, E.T.L.; Ceccarelli, S.; Grando, S.; Upadhyaya, H.D.; Ortiz, R. Diversifying Food Systems in the Pursuit of Sustainable Food Production and Healthy Diets. Trends Plant Sci. 2017, 22, 842–856. [Google Scholar] [CrossRef] [Green Version]
- Fardet, A.; Leenhardt, F.; Lioger, D.; Scalbert, A.; Rémésy, C. Parameters controlling the glycaemic response to breads. Nutr. Res. Rev. 2006, 19, 18–25. [Google Scholar] [CrossRef]
- Kasarda, D.D. Can an Increase in Celiac Disease Be Attributed to an Increase in the Gluten Content of Wheat as a Consequence of Wheat Breeding? J. Agric. Food Chem. 2013, 61, 1155–1159. [Google Scholar] [CrossRef]
- Leenhardt, F.; Lyan, B.; Rock, E.; Boussard, A.; Potus, J.; Chanliaud, E.; Remesy, C. Genetic variability of carotenoid concentration, and lipoxygenase and peroxidase activities among cultivated wheat species and bread wheat varieties. Eur. J. Agron. 2006, 25, 170–176. [Google Scholar] [CrossRef]
- Durazzo, A.; Zaccaria, M.; Polito, A.; Maiani, G.; Carcea, M. Lignan Content in Cereals, Buckwheat and Derived Foods. Foods 2013, 2, 53–63. [Google Scholar] [CrossRef]
- Tian, S.; Sun, Y.; Chen, Z.; Yang, Y.; Wang, Y. Functional Properties of Polyphenols in Grains and effects of Physiochemical Processing on Polyphenols. J. Food Qual. 2019, 2019, 8. [Google Scholar] [CrossRef]
- Oury, F.-X.; Leenhardt, F.; Rémésy, C.; Chanliaud, E.; Duperrier, B.; Balfourier, F.; Charmet, G. Genetic variability and stability of grain magnesium, zinc and iron concentrations in bread wheat. Eur. J. Agron. 2006, 25, 177–185. [Google Scholar] [CrossRef]
- Luo, C.; Branlard, G.; Griffin, W.; McNeil, D. The Effect of Nitrogen and Sulphur Fertilisation and their Interaction with Genotype on Wheat Glutenins and Quality Parameters. J. Cereal Sci. 2000, 31, 185–194. [Google Scholar] [CrossRef]
- Ames, N.P.; Clarke, J.M.; Marchylo, B.A.; Dexter, J.E.; Woods, S.M. Effect of Environment and Genotype on Durum Wheat Gluten Strength and Pasta Viscoelasticity. Cereal Chem. J. 1999, 76, 582–586. [Google Scholar] [CrossRef]
- Katina, K.; Heiniö, R.-L.; Autio, K.; Poutanen, K. Optimization of sourdough process for improved sensory profile and texture of wheat bread. LWT Food Sci. Technol. 2006, 39, 1189–1202. [Google Scholar] [CrossRef]
- Khilberg, I.; Johansson, L.; Kohler, A.; Risvik, E. Sensory qualities of whole wheat pan bread—Influence of farming system, milling and baking technique. J. Cereal Sci. 2004, 39, 67–84. [Google Scholar] [CrossRef]
- Rémésy, C.; Leenhardt, F.; Fardet, A. Donner un nouvel avenir au pain dans le cadre d’une alimentation durable et préventive. Cah. De Nutr. Et De Diététique 2015, 50, 39–46. [Google Scholar] [CrossRef]
- van Bueren, E.T.L.; Jones, S.S.; Tamm, L.; Murphy, K.M.; Myers, J.R.; Leifert, C.; Messmer, M.M. The need to breed crop varieties suitable for organic farming, using wheat, tomato and broccoli as examples: A review. NJAS Wagening. J. Life Sci. 2011, 58, 193–205. [Google Scholar] [CrossRef]
- de Vallavieille-Pope, C.; Belhai Fraj, M.; Mille, B.; Meynard, J.M. Les associations de variétés: Accroître la biodiversité pour mieux maîtriser les maladies. Les Doss. De L’environnement De L’inra 2006, 30, 101–109. [Google Scholar]
- Di Silvestro, R.; Marotti, I.; Bosi, S.; Bregola, V.; Carretero, A.S.; Sedej, I.; Mandic, A.; Sakac, M.; Benedettelli, S.; Dinelli, G. Health-promoting phytochemicals of Italian common wheat varieties grown under low-input agricultural management: Bioactive compounds of different common wheat varieties. J. Sci. Food Agric. 2012, 92, 2800–2810. [Google Scholar] [CrossRef] [PubMed]
- Call, L.; Kapeller, M.; Grausgruber, H.; Reiter, E.; Schoenlechner, R.; D’Amico, S. Effects of species and breeding on wheat protein composition. J. Cereal Sci. 2020, 93, 102974. [Google Scholar] [CrossRef]
- Roussel, V.; Leisova, L.; Exbrayat, F.; Stehno, Z.; Balfourier, F. SSR allelic diversity changes in 480 European bread wheat varieties released from 1840 to 2000. Theor. Appl. Genet. 2005, 111, 162–170. [Google Scholar] [CrossRef] [Green Version]
- Bencze, S.; Makádi, M.; Aranyos, T.J.; Földi, M.; Hertelendy, P.; Mikó, P.; Bosi, S.; Negri, L.; Drexler, D. Re-Introduction of Ancient Wheat Cultivars into Organic Agriculture—Emmer and Einkorn Cultivation Experiences under Marginal Conditions. Sustainability 2020, 12, 1584. [Google Scholar] [CrossRef] [Green Version]
- Murphy, K.M.; Reeves, P.G.; Jones, S.S. Relationship between yield and mineral nutrient concentrations in historical and modern spring wheat cultivars. Euphytica 2008, 163, 381–390. [Google Scholar] [CrossRef]
- Fan, M.-S.; Zhao, F.-J.; Fairweather-Tait, S.J.; Poulton, P.R.; Dunham, S.J.; McGrath, S.P. Evidence of decreasing mineral density in wheat grain over the last 160 years. J. Trace Elem. Med. Biol. 2008, 22, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Chable, V.; Dawson, J.; Bocci, R.; Goldringer, I. Seeds for Organic Agriculture: Development of Participatory Plant Breeding and Farmers’ Networks in France. In Organic Farming, Prototype for Sustainable Agricultures; Bellon, S., Penvern, S., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 383–400. [Google Scholar]
- van Frank, G.; Rivière, P.; Pin, S.; Baltassat, R.; Berthellot, J.F.; Caizergues, F.; Dalmasso, C.; Gascuel, J.S.; Hyacinthe, A.; Mercier, F.; et al. Genetic Diversity and Stability of Performance of Wheat Population Varieties Developed by Participatory Breeding. Sustainability 2020, 12, 384. [Google Scholar] [CrossRef] [Green Version]
- Cooper, R. Re-discovering ancient wheat varieties as functional foods. J. Tradit. Complement. Med. 2015, 5, 138–143. [Google Scholar] [CrossRef] [Green Version]
- Kohajdova, Z.; Karovicova, J. Nutritional value and baking applications of spelt wheat. Acta Sci. Pol. Technol. Aliment. 2008, 7, 5–14. [Google Scholar]
- Abdel-Aal, E.S.M.; Young, J.C.; Rabalski, I.; Hucl, P.; Fregeau-Reid, J. Identification and Quantification of Seed Carotenoids in Selected Wheat Species. J. Agric. Food Chem. 2007, 55, 787–794. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.; Benbrook, C.; Davis, D.R. With the Grain, a Closer Look at the Nutrient Quality of Grain, Grained-Based Products, and the Role of Organic Agriculture; The Organic Center: Washington, DC, USA, 2012. [Google Scholar]
- Hammes, W.P.; Brandt, M.J.; Francis, K.L.; Rosenheim, J.; Seitter, M.F.H.; Vogelmann, S.A. Microbial ecology of cereal fermentations. Trends Food Sci. Technol. 2005, 16, 4–11. [Google Scholar] [CrossRef]
- Schnürer, J.; Magnusson, J. Antifungal lactic acid bacteria as biopreservatives. Trends Food Sci. Technol. 2005, 16, 70–78. [Google Scholar] [CrossRef]
- Chavan, R.S.; Chavan, S.R. Sourdough Technology—A Traditional Way for Wholesome Foods: A Review. Compr. Rev. Food Sci. Food Saf. 2011, 10, 169–182. [Google Scholar] [CrossRef]
- Sacchi, G.; Cei, L.; Stefani, G.; Lombardi, G.V.; Rocchi, B.; Belletti, G.; Padel, S.; Sellars, A.; Gagliardi, E.; Nocella, G.; et al. A Multi-Actor Literature Review on Alternative and Sustainable Food Systems for the Promotion of Cereal Biodiversity. Agriculture 2018, 8, 173. [Google Scholar] [CrossRef] [Green Version]
- Moreira, P.M.R.M.; Pêgo, S.E.; Vaz Patto, C.; Hallauer, A.R. Comparison of selection methods on ‘Pigarro’, a Portuguese improved maize population with fasciation expression. Euphytica 2008, 163, 481–499. [Google Scholar] [CrossRef]
- Wolfe, M.S.; Baresel, J.P.; Desclaux, D.; Goldringer, I.; Hoad, S.; Kovacs, G.; Löschenberger, F.; Miedaner, T.; Østergård, H.; van Bueren, E.T.L. Developments in breeding cereals for organic agriculture. Euphytica 2008, 163, 323–346. [Google Scholar] [CrossRef] [Green Version]
- Murphy, K.; Lammer, D.; Lyon, S.; Carter, B.; Jones, S.S. Breeding for organic and low-input farming systems: An evolutionary–participatory breeding method for inbred cereal grains. Renew. Agric. Food Syst. 2005, 20, 48–55. [Google Scholar] [CrossRef]
- Goldringer, I.; van Frank, G.; d’Yvoire, C.B.; Forst, E.; Galic, N.; Garnault, M.; Locqueville, J.; Pin, S.; Bailly, J.; Baltassat, R.; et al. Agronomic Evaluation of Bread Wheat Varieties from Participatory Breeding: A Combination of Performance and Robustness. Sustainability 2019, 12, 128. [Google Scholar] [CrossRef] [Green Version]
- Kucek, L.K.; Dyck, E.; Russell, J.; Clark, L.; Hamelman, J.; Burns-Leader, S.; Senders, S.; Jones, J.; Benscher, D.; Davis, M.; et al. Evaluation of wheat and emmer varieties for artisanal baking, pasta making, and sensory quality. J. Cereal Sci. 2017, 74, 19–27. [Google Scholar] [CrossRef] [Green Version]
- Rivière, P.; Pin, S.; Galic, N.; De Oliviera, Y.; Davis, O.; Dawson, J.; Wanner, A.; Heckmann, R.; Obbeliane, S.; Ronnot, B.; et al. Mise en place d’une méthodologie de sélection participative sur le blé en France. Innov. Agron. 2013, 32, 427–441. [Google Scholar]
- Pagès, J. Recueil direct de distances sensorielles: Application à l’évaluation de dix vins blancs du Val-de-Loire. Sci. Aliment. 2003, 23, 679–688. [Google Scholar] [CrossRef]
- Ares, G.; Bruzzone, F.; Giménez, A. Is a consumer panel able to reliably evaluate the texture of dairy desserts using unstructured intensity scales? Evaluation of global and individual performance: Is a consumer panel reiable? J. Sens. Stud. 2011, 26, 363–370. [Google Scholar] [CrossRef]
- Risvik, E.; McEwan, J.A.; Colwill, J.S.; Rogers, R.; Lyon, D.H. Projective mapping: A tool for sensory analysis and consumer research. Food Qual. Prefer. 1994, 5, 263–269. [Google Scholar] [CrossRef]
- Kennedy, J. Evaluation of Replicated Projective Mapping of Granola Bars: Replicated Projective Mapping. J. Sens. Stud. 2010, 25, 672–684. [Google Scholar] [CrossRef]
- Lê, S.; Josse, J.; Husson, F. FactomineR: An R Package for Multivariate Analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Pagès, J. Collection and analysis of perceived product inter-distances using multiple factor analysis: Application to the study of 10 white wines from the Loire Valley. Food Qual. Prefer. 2005, 16, 642–649. [Google Scholar] [CrossRef]
- Lê, S.; Lê, T.M.; Cadoret, M. Napping and sorted Napping as a sensory profiling technique. In Rapid Sensory Profiling Techniques; Woodhead Publishing: Sawston, UK, 2015; pp. 197–213. [Google Scholar]
- Vindras-Fouillet, C.; Ranke, O.; Anglade, J.P.; Taupier-Letage, B.; Chable, V.; Goldringer, I. Sensory Analyses and Nutritional Qualities of Hand-Made Breads with Organic Grown Wheat Bread Populations. Food Nutr. Sci. 2014, 5, 1860–1874. [Google Scholar] [CrossRef] [Green Version]
- Ortiz-Monasterio, J.I.; Palacios-Rojas, N.; Meng, E.; Pixley, K.; Trethowan, R.; Peña, R.J. Enhancing the mineral and vitamin content of wheat and maize through plant breeding. J. Cereal Sci. 2007, 46, 293–307. [Google Scholar] [CrossRef]
- Moreira-Ascarrunz, S.; Larsson, H.; Prieto-Linde, M.; Johansson, E. Mineral Nutritional Yield and Nutrient Density of Locally Adapted Wheat Genotypes under Organic Production. Foods 2016, 5, 89. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michel, E.; Monfort, C.; Deffrasnes, M.; Guezenec, S.; Lhomme, E.; Barret, M.; Sicard, D.; Dousset, X.; Onno, B. Characterization of relative abundance of lactic acid bacteria species in French organic sourdough by cultural, qPCR and MiSeq high-throughput sequencing methods. Int. J. Food Microbiol. 2016, 239, 35–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]





| Variety Name | Origin (Farmer, Location) | Development Process |
|---|---|---|
| Saint-Priest | FLM, Maine-et-Loire | Derived from a Swedish variety registered in 1942 (Progress) |
| Rouge du Roc | JFB, Lot-et-Garonne | Population derives from a mass selection within a landrace |
| Pop Dynamique 2 | FLM, Maine-et-Loire | Mixture of 3 landraces and 2 recent varieties |
| Mélange du Sud-Ouest | JFB, Lot-et-Garonne | Mixture of about 18 landraces |
| Savoysone | RAB, Haute-Savoie | Population derived from a cross between two landraces |
| Rocaloex | RAB, Haute-Savoie | Mixture of 11 crosses |
| Mélanges1 13 Pops | BER, Côte d’Or | Mixture of 13 populations derived from crosses |
| Dauphibois | CHD, Isère | Mixtures of 26 landraces and crosses |
| Japhabelle | JFB, Lot-et-Garonne | Mixture of around 25 populations derived from crosses and selected on farm |
| Renan | INRAE | Pure-line commercial variety, registered in 1989 |
| Hendrix | INRAE | Pure-line commercial variety for organic agriculture, registered in 2013 |
| Lutein | Se | Cu | Ferulic Acid | Ca | Mg | K | Zn | Fibers | |
|---|---|---|---|---|---|---|---|---|---|
| JFB | 1.1 * | 69.7. | 0.25 * | 716.78. | 44.11 ** | 124.15 | 443.74 *** | 3.87 | 2.25 |
| RAB | 1.39 * | 16.9. | 0.47 * | 614.1. | 38.15 ** | 127.61 | 382.55 *** | 4.15 | 2.17 |
| Dauphibois | 1.25 | 166 | 0.55 | 720 | 40.75 | 126.55 *(ab) | 413.95 | 3.8 **(ab) | 1.65 |
| Japhabelle | 1.35 | 0 | 0.5 | 645.5 | 42.5 | 131.35 *(ab) | 410.9 | 4.2 **(bc) | 2.65 |
| Mélange13pops | 1.25 | 25.5 | 0.25 | 597.5 | 38.8 | 124.6 *(ab) | 411.55 | 3.8 **(ab) | 2.8 |
| MélangeSO | 1.35 | 33.5 | 0.25 | 633.5 | 40.35 | 134.75 *(b) | 436.05 | 4.4 **(bc) | 2.6 |
| PopDyn2 | 1.25 | 79 | 0.25 | 805.5 | 37.85 | 129.7 *(ab) | 389.7 | 4.3 **(bc) | 2 |
| Rocaloex | 1.25 | 0 | 0.5 | 632 | 43.45 | 125.5 *(ab) | 404.1 | 4 **(ac) | 1.85 |
| Savoysone | 1.35 | 0 | 0.55 | 767.5 | 48.9 | 127.55 *(ab) | 450.65 | 4 **(ac) | 2.25 |
| StPriest | 0.6 | 0 | 0.5 | 575 | 35.485 | 135.9 *(b) | 365.3 | 5.1 **(c) | 2 |
| Hendrix | 1.45 | 102.5 | 0 | 651 | 42.15 | 105.1 *(a) | 416.75 | 2.9 **(a) | 2.4 |
| Renan | 1.35 | 26.5 | 0.25 | 628.5 | 41.05 | 117.8 *(ab) | 432.5 | 3.7 **(ab) | 1.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vindras-Fouillet, C.; Goldringer, I.; Frank, G.; Dewalque, M.; Colin, A.; Montaz, H.; Berthellot, J.-F.; Baltassat, R.; Dalmasso, C. Sensory Analyses and Nutritional Qualities of Wheat Population Varieties Developed by Participatory Breeding. Agronomy 2021, 11, 2117. https://doi.org/10.3390/agronomy11112117
Vindras-Fouillet C, Goldringer I, Frank G, Dewalque M, Colin A, Montaz H, Berthellot J-F, Baltassat R, Dalmasso C. Sensory Analyses and Nutritional Qualities of Wheat Population Varieties Developed by Participatory Breeding. Agronomy. 2021; 11(11):2117. https://doi.org/10.3390/agronomy11112117
Chicago/Turabian StyleVindras-Fouillet, Camille, Isabelle Goldringer, Gaëlle van Frank, Marc Dewalque, Axel Colin, Hélène Montaz, Jean-François Berthellot, Raphaël Baltassat, and Christian Dalmasso. 2021. "Sensory Analyses and Nutritional Qualities of Wheat Population Varieties Developed by Participatory Breeding" Agronomy 11, no. 11: 2117. https://doi.org/10.3390/agronomy11112117
APA StyleVindras-Fouillet, C., Goldringer, I., Frank, G., Dewalque, M., Colin, A., Montaz, H., Berthellot, J.-F., Baltassat, R., & Dalmasso, C. (2021). Sensory Analyses and Nutritional Qualities of Wheat Population Varieties Developed by Participatory Breeding. Agronomy, 11(11), 2117. https://doi.org/10.3390/agronomy11112117






