Response of Two Local Common Bean Ecotypes of “Fagioli di Sarconi” PGI (Phaseolus vulgaris L.) to Seed-Borne Pathogens and Environmental Change
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Isolation of Seed Microflora
2.2.1. Washing Test
2.2.2. Blotter Test
2.2.3. Between Paper Test
2.3. Identification of Pathogens
2.4. Disease Assessment
2.5. Statistical Analysis
3. Results
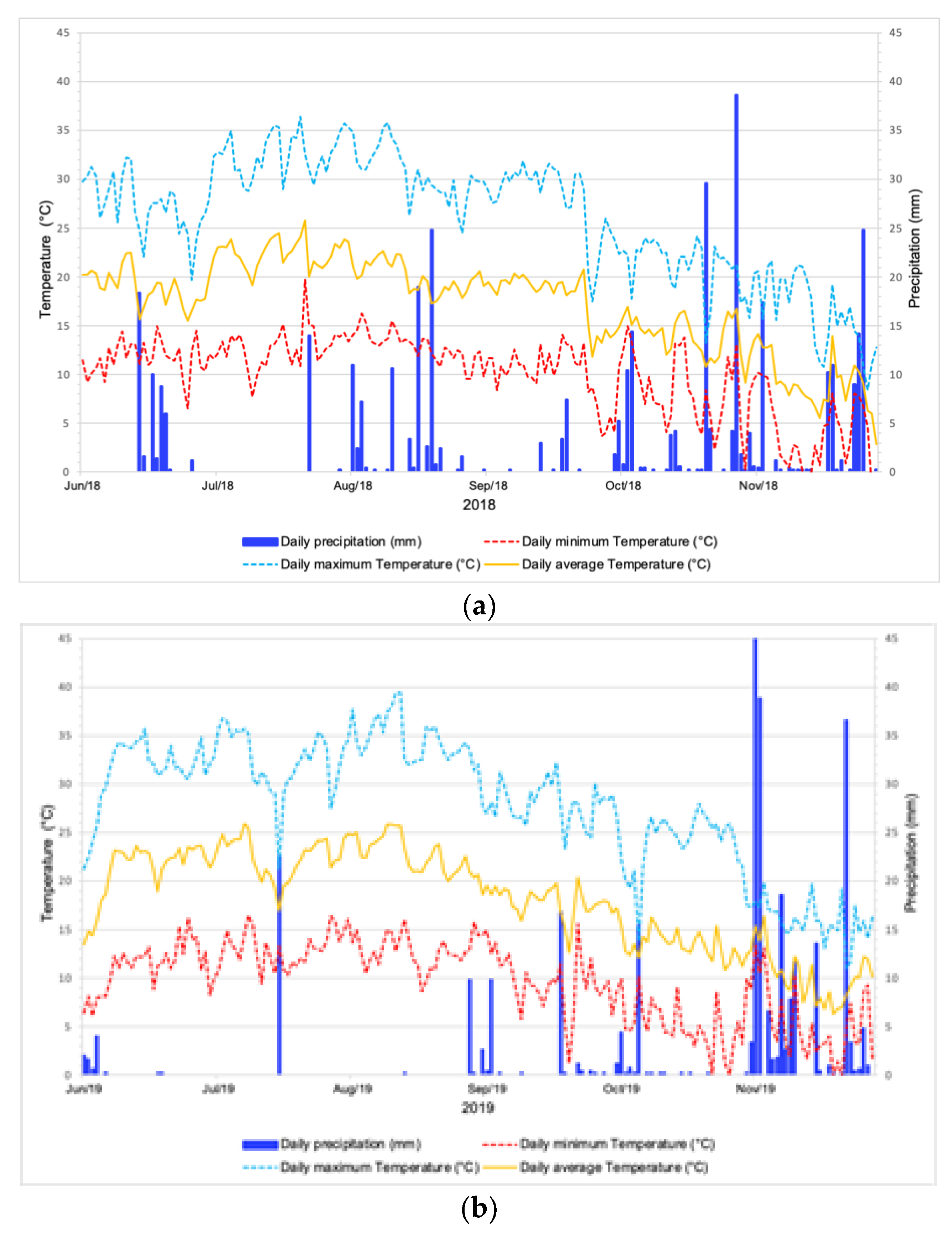
3.1. Meteorological Parameters
3.2. Isolation and Identification of Seed Mycoflora by Washing Test
3.3. Identification and Incidence of R. solani on Treated and Untreated Seeds with Blotter Test, and of C. lindemuthianum, F. oxysporum and Bacterial Diseases Causal Agents with between Paper Test
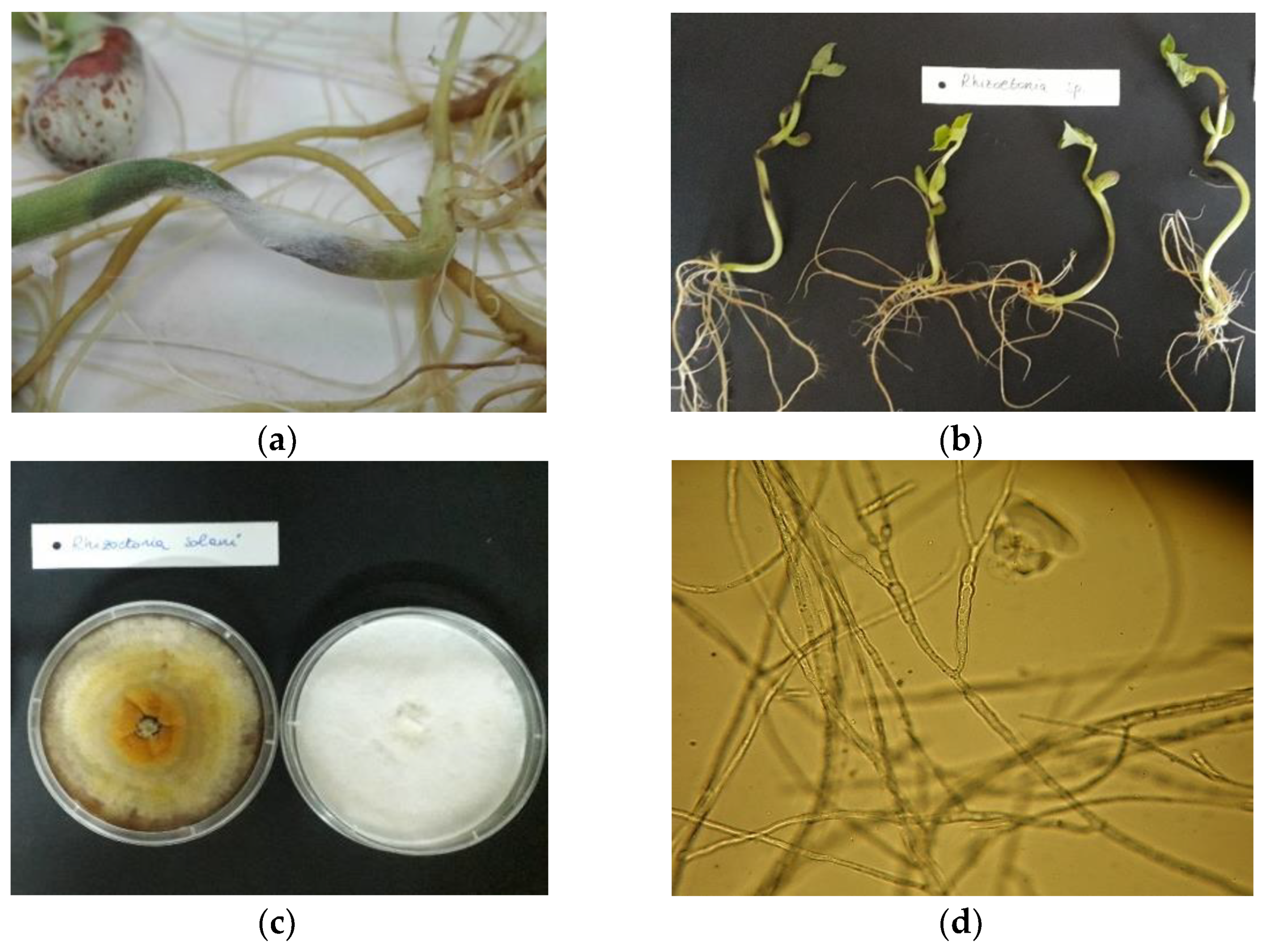
3.4. Macroscopic and Microscopic Characterization of R. solani, C. lindemuthianum and F. oxysporum
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Celmeli, T.; Sari, H.; Canci, H.; Sari, D.; Adak, A.; Eker, T.; Toker, C. The Nutritional Content of Common Bean (Phaseolus vulgaris L.) Landraces in Comparison to Modern Varieties. Agronomy 2018, 8, 166. [Google Scholar] [CrossRef] [Green Version]
- Alvi, G. I Legumi da Granella: 2016 anno Internazionale dei Legumi; Ministero delle Politiche Agricole e Forestali: Rome, Italy, 2016; pp. 1–21.
- Murube, E.; Beleggia, R.; Pacetti, D.; Nartea, A.; Frascarelli, G.; Lanzavecchia, G.; Bellucci, E.; Nanni, L.; Gioia, T.; Marciello, U.; et al. Characterization of Nutritional Quality Traits of a Common Bean Germplasm Collection. Foods 2021, 10, 1572. [Google Scholar] [CrossRef]
- Graham, B.R.; Ranalli, P. Common bean (Phaseolus vulgaris L.). Field Crop. Res. 1997, 53, 131–146. [Google Scholar] [CrossRef]
- Gioia, T.; Logozzo, G.; Attene, G.; Bellucci, E.; Benedettelli, S.; Negri, V.; Papa, R.; Spagnoletti Zeuli, P. Evidence for introduction bottleneck and extensive inter−gene pool (Mesoamerica x Andes) hybridization in the European common bean (Phaseolus vulgaris L.) germplasm. PLoS ONE 2013, 8, e75974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bitocchi, E.; Rau, D.; Bellucci, E.; Rodriguez, M.; Murgia, M.L.; Gioia, T.; Santo, D.; Nanni, L.; Attene, G.; Papa, R. Beans (Phaseolus ssp.) as a Model for Understanding Crop Evolution. Front. Plant. Sci. 2017, 8, 722. [Google Scholar] [CrossRef] [Green Version]
- Istituto Nazionale di Statistica. 2021. Available online: http://dati.istat.it/Index.aspx?DataSetCode=DCSP_COLTIVAZIONI (accessed on 10 August 2021).
- Piergiovanni, A.R.; Laghetti, G. The common bean land−races from Basilicata (Southern Italy): An example of integrated approach applied to genetic resources management. Genet. Resour Crop. Evol. 1999, 46, 47–52. [Google Scholar] [CrossRef]
- Masi, P.; Logozzo, G.; Donini, P.; Spagnoletti Zeuli, P. Analysis of genetic structure in widely distributed common bean (Phaseolus vulgaris L.) landraces with different plant growth habit using SSR and AFLP markers. Crop. Sci. 2009, 49, 187–199. [Google Scholar] [CrossRef]
- Bianco, G.; Buchicchio, A.; Cataldi, T.R.I. Structural characterization of major soyasaponins in traditional cultivars of Fagioli di Sarconi beans investigated by high−resolution tandem mass spectrometry. Anal. Bioanal Chem. 2015, 407, 6381–6389. [Google Scholar] [CrossRef]
- Masi, P.; Figliuolo, G.; Spagnoletti Zeuli, P.L. Landraces of bean (Phaseolus vulgaris L.) collected in Basilicata, Italy. Plant. Genet. Res. Newslett. 1999, 119, 51–55. [Google Scholar]
- Piergiovanni, A.R.; Cerbino, D.; Brandi, M. The common bean populations from Basilicata (Southern Italy). An evaluation of their variation. Genet. Resour. Crop. Evol. 2000, 47, 489–495. [Google Scholar] [CrossRef]
- Lioi, L.; Piergiovanni, A.R.; Pignone, D.; Puglisi, S.; Santantonio, M.; Sonnante, G. Genetic diversity of some surviving on−farm Italian common bean (Phaseolus vulgaris L.) landraces. Plant. Breed. 2005, 124, 576–581. [Google Scholar] [CrossRef]
- Dinelli, G.; Sonetti, A.; Minelli, M.; Marotti, I.; Catione, P.; Mazzanti, A. Content of flavonols in Italian bean (Phaseolus vulgaris L.) ecotypes. Food Chem. 2006, 99, 105–114. [Google Scholar] [CrossRef]
- Piergiovanni, A.R.; Lioi, L. Italian Common Bean Landraces: History, Genetic Diversity and Seed Quality. Diversity 2010, 2, 837–862. [Google Scholar] [CrossRef] [Green Version]
- Marotti, I.; Dinelli, G.; Bregola, V.; Bosi, S. Nutritional characterization of Italian common bean landraces (Phaseolus vulgaris L.): Fatty acid profiles for “genotype−niche diversity” fingerprints. AIMS Agric. Food 2020, 5, 543–562. [Google Scholar] [CrossRef]
- Lo Cantore, P.; Nigro, C.; Castoro, V.; Iacobellis, N.S. Presenza e diffusione delle batteriosi in coltivazioni di fagiolo di Sarconi in Basilicata. J. Plant. Pathol. 2004, 89, 43–44. [Google Scholar]
- Ghangaokar, N.M.; Kshirsagar, A.D. Study of seed borne fungi of different legumes. Trends Life Sci. 2013, 2, 32–35. [Google Scholar]
- Marcenaro, D.; Valkonen, J.P.T. Seedborne Pathogenic Fungi in Common Bean (Phaseolus vulgaris cv. INTA Rojo) in Nicaragua. PLoS ONE 2016, 11, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Hussain, S.; Ajaib, M.; Asghar, R.; Ali, I.; Siddiqui, M.F. Mycoflora associated with Phaseolus vulgaris L. seeds and its impact on seed germination in Azad Jammu & Kashmir. Pak. J. Bot. 2020, 52, 1455–1463. [Google Scholar]
- Singh, S.R.; Singh, N.I. Seed mycoflora of broad bean and its control. Indian Phytopathol. 1986, 39, 541–543. [Google Scholar]
- Kator, L.; Ogo-Oluwa, A.T.; Kemi, A.B. Isolation and Identification of Seed Borne Fungi of Common Bean (Phaseolus vulgaris L.) from Selected Markets in Makurdi. Int. J. Appl Agric. Sci. 2016, 2, 75–78. [Google Scholar] [CrossRef]
- Al-Jaradi, A.; Al-Mahmooli, I.; Janke, R.; Maharachchikumbura, S.; Al-Saady, N.; Al-Sadi, A.M. Isolation and identification of pathogenic fungi and oomycetes associated with beans and cowpea root diseases in Oman. PeerJ 2018, 6, e6064. [Google Scholar] [CrossRef] [Green Version]
- Díaz, G.A.; Cabeza, R.; Amigo, R.; Llancamil, E.; Montenegro, O.; Gonzalez, P.; Valdez, A.V.; Lolas, M.A.; Ferrada, E. Severe outbreak of Fusarium wilt on common beans (Phaseolus vulgaris) caused by Fusarium oxysporum in the Maule Region, central Chile. Plant. Dis. 2021, 3. [Google Scholar] [CrossRef]
- Gargouri, S.; Hajlaoui, M.R.; Abdennadher, M.; Marrakchi, M. Isolation and morphological and identification of Fusarium spp Transmitted by watermelon seeds. Bull. OEPP/EPPO Bull. 2000, 30, 217–222. [Google Scholar] [CrossRef]
- Singh, D.; Mathur, S.B. Location of fungal hyphae in seeds. In Histopathology of Seed-Borne Infections, 1st ed.; Singh, D., Mathur, S.B., Eds.; CRC Press: Boca Raton, FL, USA, 2004; pp. 101–168. [Google Scholar] [CrossRef]
- Francl, L.J. The Disease Triangle: A plant pathological paradigm revisited. Plant. Health Instr. 2001. [Google Scholar] [CrossRef]
- Al−Askar, A.A.; Ghoneem, K.M.; Rashad, Y.M.; Abdulkhair, W.M.; Hafez, E.E.; Shabana, Y.M.; Baka, Z.A. Occurrence and distribution of tomato seed-borne mycoflora in S audi A rabia and its correlation with the climatic variables. Microb. Biotechnol. 2014, 7, 556–569. [Google Scholar] [CrossRef]
- Juroszek, P.; Racca, P.; Link, S.; Farhumand, J.; Kleinhenz, B. Overview on the review articles published during the past 30 years relating to the potential climate change effects on plant pathogens and crop disease risks. Plant. Pathol. 2020, 69, 179–193. [Google Scholar] [CrossRef]
- Crop Ontology for Agricultural Data. Available online: https://www.cropontology.org/terms/CO_335:0000149/ (accessed on 15 April 2019).
- Cortinovis, G.; Opperman, M.; Neumann, K.; Graner, A.; Gioia, T.; Marsella, M.; Alseekh, S.; Fernie, A.R.; Papa, R.; Bellucci, E.; et al. Towards the Development, Maintenance, and Standardized Phenotypic Characterization of Single−Seed−Descent Genetic Resources for Common Bean. Curr. Protoc. 2021, 1, e133. [Google Scholar] [CrossRef]
- Misra, J.K.; Mew, T.W. A Manual of Rice Seed Healt Tasting; International Rice Research Institute (IRRI): Los banõs, Laguna, Philippines, 1994. [Google Scholar]
- International Seed Testing Association. International Rules for Seed Testing 2020: Introduction to the ISTA Rules Chapters 1–19: Including Changes and Editorial Corrections Adopted at the Ordinary General Meeting 2019, Hyderabad, India; International Seed Testing Association: Bassersdorf, Switzerland, 2020. [Google Scholar]
- Miles, S.R. Handbook of tolerances and of measures of precision for seed testing. Proc. Int. Seed Test. Assoc. 1963, 28, 525–686. [Google Scholar]
- Ainsworth, G.C. Introduction and keys to higher taxa. In The Fungi: An advanced Treatise IVB. A Taxonomic Review with Keys: Basidiomycetes and Lower Fungi, 1st ed.; Ainsworth, G.C., Sparrow, F.K., Sussman, A.S., Eds.; Academic Press: New York, NY, USA, 1973; pp. 1–7. [Google Scholar]
- Kulshrestha, D.D.; Mathur, S.B.; Neergard, P. Identification of seed borne species of Colletotrichum. Friesia 1976, 11, 116–125. [Google Scholar]
- Domsch, K.H.; Gams, W.; Anderson, T.H. Compendium of Soil Fungi, 1st ed.; IHW−Verlag: Eching, Germany, 1993. [Google Scholar]
- Dragoni, I.; Cantoni, C.; Papa, A.; Vallone, L. Muffe, Alimenti e Micotossicosi; Città Studi Edizioni: Milano, Italy, 2000; p. 318. [Google Scholar]
- Nipoti, P.; Fantino, M.G.; Filippini, G.; Gennari, S.; Di Pillo, L. Testo−Atlante dei Funghi ad Habitat Terricolo; Zanichelli: Bologna, Italy, 2006; p. 153. [Google Scholar]
- Watanabe, T. Pictorial Atlas of Soil and Seed Fungi: Morphologies of Cultured Fungi and Key to Species, 3rd ed.; CRC Press: London, UK, 2010; p. 486. [Google Scholar]
- Vannacci, G.; Sarrocco, S.; Porta−Puglia, A. Improved Detection and Monitoring of Seed-Borne Fungal Plant Pathogens in Europe. In Plant Pathology in the 21st Century: Global Perspectives on the Healthof Seeds and Plant Propagation Material; Gullino, M.L., Munkvold, G., Eds.; Springer: Dordrecht, The Netherlands, 2014; Volume 6, pp. 67–85. [Google Scholar] [CrossRef]
- Webster, J.; Weber, R. Introduction to Fungi, 3rd ed.; Cambridge University Press: London, UK, 2016; p. 841. [Google Scholar]
- SAS OnDemand for Academics. Available online: https://www.sas.com/it_it/software/on−demand−for−academics.html (accessed on 25 July 2021).
- RStudio Core Team. RStudio: Integrated Development for R. RStudio, PBC, Boston. 2019. Available online: https://www.rstudio.com/ (accessed on 25 July 2021).
- Bevilacqua, V.; Vitti, A.; Logozzo, G.; Marzario, S.; Gioia, T.; Nuzzaci, M. Influence of Cultivation Areas on the Seed-Borne Pathogens on Two Local Common Bean Ecotypes of “Fagioli di Sarconi” PGI (Phaseolus vulgaris L.). Biol. Life Sci. Forum 2020, 1, 15, in press. [Google Scholar]
- Kumar, R.; Gupta, A. Seed-Borne Disease of Agricultural Crops: Detection, Diagnosis e Management; Kumar, R., Gupta, A., Eds.; Springer Nature: Singapore, 2020. [Google Scholar] [CrossRef]
- Harman, G.E.; Howell, C.R.; Viterbo, A.; Cett, I.; Lorito, M. Trichoderma species−opportunistic, avirulent plant symbionts. Nat. Rev. Microbiol. 2004, 2, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Vitti, A.; La Monaca, E.; Sofo, A.; Scopa, A.; Cuypers, A.; Nuzzaci, M. Beneficial effects of Trichoderma harzianum T−22 in tomato seedlings infected by Cucumber Mosaic Virus (CMV). Biocontrol 2015, 60, 135–147. [Google Scholar] [CrossRef]
- Vitti, A.; Pellegrini, E.; Nali, C.; Lovelli, S.; Sofo, A.; Valerio, M.; Scopa, A.; Nuzzaci, M. Trichoderma harzianum T−22 Induces Systemic Resistance in Tomato Infected by Cucumber mosaic virus. Front. Plant. Sci. 2016, 7, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Domsch, K.H.; Gams, W.; Anderson, T.H. Compendium of Soil Fungi, 1st ed.; Academic Press: London, UK, 1980. [Google Scholar]
- Ponti, I.; Laffi, F. Malattie crittogamiche delle piante ortive. In Schede Fitopatologiche; L’Informatore Agrario: Verona, Italy, 2001; p. 289. [Google Scholar]
- Moore, R.T. The Genera of Rhizoctonia—Like Fungi: Ascorhizoctonia, Ceratorhiza gen. nov., Epulorhiza gen. nov., Moniliopsis and Rhizoctonia. Mycotaxon 1987, 29, 91–99. [Google Scholar]
- Ajayi−Oyetunde, O.O.; Bradley, C.A. Rhizoctonia solani: Taxonomy, population biology and management of rhizoctonia seedling disease of soybean. Plant Pathol. 2018, 67, 3–17. [Google Scholar] [CrossRef]
- Disciglio, G.; Nardella, E.; Gatta, G.; Giuliani, M.M.; Tarantino, A. Adaptability of ‘Monti Dauni’ Bean Ecotypes in Plain Areas. WASET 2014, 8, 1118–1162. [Google Scholar]
- Carbonaro, M.; Cappelloni, M.; Nicoli, S.; Lucarini, M.; Carnovale, E. Solubility Digestibility relationship of legume proteins. J. Agric. Food Chem 1997, 45, 3387–3394. [Google Scholar] [CrossRef]
- Lattanzio, V.; Lattanzio, V.M.T.; Cardinali, A. Role of phenolics in the resistance mechanisms of plants against fungal pathogens and insects. In Adv Phytochem; Imperato, F., Ed.; Research Signpost Publisher: Kerala, India, 2006; pp. 23–67. [Google Scholar]
- Wallis, C.M.; Galarneau, E.R.A. Phenolic Compound Induction in Plant−Microbe and Plant−Insect Interactions: A Meta−Analysis. Front. Plant. Sci. 2020, 11, 580753. [Google Scholar] [CrossRef] [PubMed]
- Telles, A.C.; Kupski, L.; Furlong, E.B. Phenolic compound in beans as protection against mycotoxins. Food Chem 2017, 214, 293–299. [Google Scholar] [CrossRef]




| Fagioli di Sarconi PGI Ecotype | Seed Qualitative Trait | ||||||
| Coat Darker Color | Coat Lighter Color | Coat Patter | Longitudinal Shape | Transverse Shape | Veining | Brilliance | |
| Ciuoto | Cream/Brown | Purple/red | Stripe | Oval | Large elliptic | Weak | Medium |
| Cannellino rosso | Whitish | Purple | Spotted bicolor | Kidney shaped | Elliptic | Weak | Medium |
| Fagioli di Sarconi PGI ecotype | Seed Quantitative Trait | ||||||
| Length (mm) | Height (mm) | Width (mm) | Weight (g) | ||||
| Ciuoto | 11.13 ± 1.02 | 7.54 ± 0.62 | 6.53 ± 0.55 | 0.38 ± 0.08 | |||
| Cannellino rosso | 15.91 ± 0.81 | 8.09 ± 0.49 | 6.54 ± 0.48 | 0.63 ± 0.09 | |||
| Meteorological Parameter | 2018 | 2019 | pr (>F) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Min | Max | Range | Mean | Min | Max | Range | Mean | ||
| Rain (mm day−1) | 0.00 | 38.60 | 38.60 | 2.09 | 0.00 | 56.40 | 56.40 | 1.69 | 0.530 |
| Temperature air (°C day−1) | |||||||||
| Minimum | −4.30 | 19.80 | 24.10 | 10.03 | −0.50 | 16.50 | 17.00 | 9.58 | 0.301 |
| Maximum | 8.40 | 36.40 | 28.00 | 26.13 | 11.00 | 39.40 | 28.40 | 27.78 | 0.016 * |
| Average | 2.83 | 25.78 | 22.94 | 17.20 | 6.28 | 25.93 | 19.64 | 18.08 | 0.094 |
| Relative humidity (% day−1) | |||||||||
| Minimum | 19.00 | 90.00 | 71.00 | 42.39 | 13.00 | 91.00 | 78.00 | 37.58 | 0.001 *** |
| Maximum | 67.80 | 100.00 | 32.20 | 96.96 | 82.00 | 99.80 | 17.80 | 96.96 | 0.998 |
| Average | 46.25 | 96.42 | 50.17 | 75.09 | 49.67 | 96.79 | 47.13 | 71.25 | 0.000 *** |
| Evapotranspiration (mm day −1) | 0.76 | 8.13 | 7.37 | 4.44 | 0.90 | 8.08 | 7.18 | 4.87 | 0.049 * |
| 2018 | 2019 | |||||||
|---|---|---|---|---|---|---|---|---|
| Ecotype | Ciuoto | Cannellino Rosso | Ciuoto | Cannellino Rosso | ||||
| Locality | Sarconi | Paterno | Sarconi | Paterno | Sarconi | Paterno | Sarconi | Paterno |
| Fungal microflora | ||||||||
| Alternaria spp. | + | − | − | − | − | + | + | − |
| Alternaria alternata | − | − | − | − | − | + | − | + |
| Aspergillus spp. | − | − | + | + | − | − | − | − |
| Aspergillus flavus | − | − | + | + | + | − | − | − |
| Aspergillus niger | + | − | − | − | − | − | + | − |
| Cladosporium cladosporioides | − | − | − | + | + | + | + | − |
| Botritys spp. | − | − | + | − | − | + | − | + |
| Colletotrichum lindemuthianum | − | + | − | − | − | + | − | − |
| Fusarium oxysporum | − | − | − | − | − | + | − | + |
| Fusarium solani | − | + | − | − | − | + | − | − |
| Mucor hiemalis | + | − | − | − | − | − | + | − |
| Penicillium spp. | − | − | + | + | − | + | + | + |
| Penicillium expansum | − | + | − | − | − | + | − | − |
| Rhizoctonia solani | − | + | − | + | + | + | + | + |
| Rhizophus nigricans | + | − | − | − | − | − | − | − |
| Trichoderma harzianum | − | − | − | + | − | − | − | − |
| Tricoderma viridae | + | − | − | − | − | − | − | − |
| Uromyces appendiculatus | − | − | − | − | − | + | − | + |
| Year | 2018 | 2019 | ||||||
|---|---|---|---|---|---|---|---|---|
| Ecotype | Ciuoto | Cannellino Rosso | Ciuoto | Cannellino Rosso | ||||
| Locality | Sarconi | Paterno | Sarconi | Paterno | Sarconi | Paterno | Sarconi | Paterno |
| Blotter Method | ||||||||
| Rhizoctonia solani | ||||||||
| treated seed | + | + | + | + | + | + | + | + |
| Disease incidence (%) 1 | 27 | 23 | 8 | 15 | 12 | 15 | 4 | 8 |
| untreated seed | + | + | + | + | + | + | + | + |
| Disease incidence (%) 1 | 19 | 35 | 24 | 27 | 11 | 23 | 12 | 24 |
| Between Paper Test | ||||||||
| Fusarium oxysporum | + | + | − | + | − | + | − | + |
| Disease incidence (%) 2 | 1 | 4 | 0 | 2 | 0 | 10 | 0 | 6 |
| C. lindemuthianum | + | + | + | + | + | + | + | + |
| Disease incidence (%) 2 | 4 | 40 | 8 | 20 | 4 | 65 | 4 | 30 |
| Bacterial disease causal agents | ||||||||
| P. syringae pv. phaseolicola | + | + | + | + | + | + | + | − |
| X. campestris pv. phaseoli | + | + | + | − | − | + | − | + |
| Disease incidence (%) 2 | 28 | 30 | 18 | 24 | 12 | 12 | 6 | 16 |
| Development Temperature (°C) | References | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mycoflora | −5 | 0 | 5 | 10 | 15 | 20 | 25 | 30 | 35 | 40 | 45 | 50 | |
| Alternaria spp. | x | x | x | x | [50] | ||||||||
| Alternaria alternata (Fr.) Keissler | x | x | x | x | [51] | ||||||||
| Aspergillus spp. | x | x | x | x | x | x | x | [38] | |||||
| Aspergillus flavus Link | x | x | x | x | x | x | x | [38] | |||||
| Aspergillus niger van Tieghem | x | x | x | x | x | x | [39] | ||||||
| Cladosporium cladosporioides G.A. de Vries | x | x | x | x | x | x | x | x | x | x | [38] | ||
| Botrytis spp. | x | x | x | x | x | x | x | x | x | [38] | |||
| Colletothricum lindemuthianum Briosi e Cavara | x | x | x | x | [42] | ||||||||
| Fusarium oxysporum von Schlechtendal | x | x | x | x | [50] | ||||||||
| Fusarium solani (Mart.) Sacc. | x | x | x | x | [50] | ||||||||
| Mucor hiemalis Wehmer | x | x | x | x | x | x | x | [42] | |||||
| Penicillium expansum Link | x | x | x | x | x | x | x | x | [42] | ||||
| Penicillium spp. | x | x | x | x | x | x | x | [42] | |||||
| Rhizoctonia solani (Cooke) Wint. | x | x | x | x | [52] | ||||||||
| Rhizophus nigricans Vuillemin | x | x | x | x | x | x | x | x | [50] | ||||
| Trichoderma spp. | x | x | [39] | ||||||||||
| Uromyces appendiculatus (Pers.) Link | x | x | x | [51] | |||||||||
| Development Humidity (%) | References | |||
|---|---|---|---|---|
| Mycoflora | <55 | 55 < 75 | >75 | |
| Alternaria spp. | x | [50] | ||
| Alternaria alternata (Fr.) Keissler | x | [51] | ||
| Aspergillus spp. | x | [38] | ||
| Aspergillus flavus Link | x | [38] | ||
| Aspergillus niger van Tieghem | x | [39] | ||
| Cladosporium cladosporioides G.A. de Vries | x | [38] | ||
| Botrytis spp. | x | [38] | ||
| Colletothricum lindemuthianum Briosi e Cavara | x | [42] | ||
| Fusarium oxysporum von Schlechtendal | x | [50] | ||
| Fusarium solani (Mart.) Sacc. | x | [50] | ||
| Mucor hiemalis Wehmer | x | [42] | ||
| Penicillium expansum Link | x | [42] | ||
| Penicillium spp. | x | [42] | ||
| Rhizoctonia solani (Cooke) Wint. | x | [52] | ||
| Rhizophus nigricans Vuillemin | x | [50] | ||
| Trichoderma spp. | x | [39] | ||
| Uromyces appendiculatus (Pers.) Link | x | [51] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vitti, A.; Bevilacqua, V.; Logozzo, G.; Marzario, S.; Gioia, T.; Nuzzaci, M. Response of Two Local Common Bean Ecotypes of “Fagioli di Sarconi” PGI (Phaseolus vulgaris L.) to Seed-Borne Pathogens and Environmental Change. Agronomy 2021, 11, 1924. https://doi.org/10.3390/agronomy11101924
Vitti A, Bevilacqua V, Logozzo G, Marzario S, Gioia T, Nuzzaci M. Response of Two Local Common Bean Ecotypes of “Fagioli di Sarconi” PGI (Phaseolus vulgaris L.) to Seed-Borne Pathogens and Environmental Change. Agronomy. 2021; 11(10):1924. https://doi.org/10.3390/agronomy11101924
Chicago/Turabian StyleVitti, Antonella, Vincenzo Bevilacqua, Giuseppina Logozzo, Stefania Marzario, Tania Gioia, and Maria Nuzzaci. 2021. "Response of Two Local Common Bean Ecotypes of “Fagioli di Sarconi” PGI (Phaseolus vulgaris L.) to Seed-Borne Pathogens and Environmental Change" Agronomy 11, no. 10: 1924. https://doi.org/10.3390/agronomy11101924
APA StyleVitti, A., Bevilacqua, V., Logozzo, G., Marzario, S., Gioia, T., & Nuzzaci, M. (2021). Response of Two Local Common Bean Ecotypes of “Fagioli di Sarconi” PGI (Phaseolus vulgaris L.) to Seed-Borne Pathogens and Environmental Change. Agronomy, 11(10), 1924. https://doi.org/10.3390/agronomy11101924







