Abstract
The aim of this study was to determine the effects of new Micro Carbon Technology (MCT®) fertilizers based on humic acids biologically digested on the growth and development of pepper plants. In this work, the biostimulant effect of MCT® fertilizers was compared to conventional mineral fertilizers. In order to evaluate MCT® fertilizers, a previous chemical characterization (infrared spectroscopy, liquid chromatography and mass spectrometry) of seven MCT® fertilizers was performed. Two fertilization tests of pepper plants were carried out in hydroponic conditions, where the fertilization and the age of the plants were studied in order to evaluate the specific effects on roots and leaves. Plant weight and foliar analysis (chlorophyll indices and nutrients) have been determined. Transmission electron microscopy was used to visualize the morphological differences in the root and leaf cells. Comparison between conventional and MCT® based fertilizers showed that, with the MCT® fertilizers, the plant is exposed to the presence of free amino acids (Glycine and Alanine), polyphenols and humic substances. Although no significant differences were found in plant mass production, the plants fertilized with MCT® products presented better nutritional status than plants treated with conventional fertilization in terms of nutrient content in leaves. Important morphological differences in root cells were found. A large central vacuole that represented the 68–83% of the total root cell area was shown if the MCT® products were used, suggesting significant changes of membrane permeability in terms of water adsorption and consequently nutrient storage. The morphological differences observed in the root cells were more noticeable in adult plants.
1. Introduction
Agriculture nowadays has to face several problems of surface water contamination and water overuse due to the irrational use and consumption of fertilizers in intensive agriculture, caused by the increasing demand for food due to the world population increase [1,2]. Fertilizers based on organic matter (OM) are intended to replace conventional fertilizers by having greater efficiency and being more respectful of the environment [3]. This will result in an increase in the demand for food, number of producers and area of land organically grown [4], as well as the demand for organic fertilizers or humic substances (HS) [5]. HS are natural substances which proceed from the biochemical transformation of organic materials by decomposition processes, microbial synthesis, polymerization and condensation reactions [6]. They are composed of condensed aromatic structures, aliphatic structures and highly functional carbon biopolymers, linked by different types of hydrogen bonds, π bonds and hydrophobic interactions that provide them a high stability [7]. However, HS physical and chemical characteristics depend on many factors, among which is the type of original organic material [8]. HS have positive effects on crops by increasing the available nutrients [9], organic carbon levels [10] or improving the photosynthetic efficiency of crops [11]. For instance, bioactive molecules like plant growth hormones can be incorporated into HS structures and thus influence the growth and development of plants in a combined hormonal/humic effect [12]. Other proposed mechanisms consist of an “indirect action” on the metabolism of the microbial population and the physical conditions of the soil [13], meaning better absorption of nutrients [14]. Ansari et al. [1] showed that the application of several organic and bio-organic fertilizers based on the consortia Rhizobium sp. and municipal waste significantly improved peroxidase activity and nitrate reductase activity, increasing the soil microbial population.
Knowledge of the molecular size, weight and structural disposition of the functional groups of humic acids is required for an adequate understanding of their role in environmental processes [15]. However, despite many years of research, the chemical structure of humic macromolecules, their molecular weight and their genesis are still unknown due to their complexity [15]. In fact, the current scale of HS manufacturing is insufficient because there is a lack on the effects of these substances have, an absence of necessary information about their structures and physico-chemical characteristics that has led to no effective industrial process for its production [16].
The main objective of this work was to evaluate the effectiveness of Micro Carbon Technology (MCT®) fertilizers with expected biostimulant activity in comparison with conventional mineral fertilizers. After chemical characterization by elemental analysis, infrared spectroscopy (FTIR), high performance liquid chromatography with photodiode array detector (HPLC-PDA) for amino acids and polyphenols of MCT® fertilizers was conducted. The agronomical comparison between MCT® and conventional fertilizers was based on two hydroponic assays where plant growth, foliar mineral analysis, chlorophyll indices and morphological visualization of root cells with transmission electronic microscopy (TEM) were performed to determine possible effects on pepper plants (Capsicum annuum L.).
2. Materials and Methods
2.1. Description of Micro Carbon Technology (MCT®) Fertilizers
The Micro Carbon Technology (MCT®) products used in this work belong to the range of HUMA GRO products generated by the company Bio Huma Netics, Inc. (Gilbert, Arizona, USA). The new fertilizers generated are concentrated liquids produced with inorganic salts and humic acid derivatives from the biological digestion of a specific leonardite from a company-owned mine located in Southwestern Idaho (USA). This digestion breaks down the humic substances and generates fractions of lower molecular weight (carbon molecules of 1 to 3 rings of benzene with high active surface) creating the base of MCT®.
All the target products evaluated were liquid formulations based on MCT® combined with inorganic fertilizers: X_Tend (TX) (6-3-0) ureic nitrogen and 6.02% of OM; UltraNitro-12% (TN) (12-0-0) N nitric; Phos-Max (TP) (0-50-0) and 6% of OM; Super K (TK) (0-0-40) and 8.5% O.M; Calcium (TC) (8-0-0 and 15% Ca) N nitric and 0.4% OM; 44 Mag (TM) (8% Mg and 14% S) and 0.6% OM; Max Pak TS (10% S; 0.5% B; 0.05% Co; 1% Cu; 2% Fe; 1% Mn; 0.05% Mo and 4% Zn) and 8.5% OM. Of all these products, the TX is the product that has a higher concentration of MCT® fractions and it is most similar to what is used as a base to introduce the MCT® into the other products.
For the design of conventional fertilizer solutions based only on inorganic salts, the following Panreac (Barcelona, Spain) analytical grade products were used: Ca(NO3)2·4H2O, KH2PO4, K2SO4 and MgSO4·7H2O as macronutrients and (NH4)6Mo7O24·4H2O, CuSO4·5H2O, ZnSO4·H2O, H3BO3 and MnSO4·H2O as micronutrients. The KNO3 used was from Merck & Co., Inc. (Kenilworth, New Jersey, USA) and Fe-EDDHA from Quimioprox (Barcelona, Spain).
2.2. Chemical Characterization of Fertilizers
Elemental analysis for determining total carbon (C), nitrogen (N), hydrogen (H) and sulfur (S) was carried out with LECO Element Analyzer CHNS-932 (St. Joseph, Michigan, USA) in the solid product obtained after the evaporation of the liquid at room temperature.
The solid residue was subjected to Fourier-transform infrared spectroscopy (FTIR) using a Bruker IFS66v spectrometer (Billerica, MA, USA); readings were obtained in arbitrary units of diffuse reflectance. Samples (1 mg) were diluted in 99 mg of KBr before the analysis. Spectra were obtained by accumulating 250 scans at a resolution of 4 cm−1 in a spectral range of 450–4000 cm−1.
Mass spectrometry with inductive coupling plasma (ICP-MS NexION 300XX, Perkin-Elmer, Waltham, MA, USA) was used for the determination of trace elements in the fertilizers.
Free amino acids and polyphenols were determined by HPLC-PDA. The HPLC used for both analyses was composed of a 2695 separation module (Waters, Milford, MA, USA) equipped with a 996 photodiode array detector (Waters 996 PDA, Milford, MA, USA) and a Waters AccQ·TagTM amino acid analysis column (4 μm Nova-PakTM C18 column) for the amino acid analysis and a Waters Spherisorb® S5 OD52 5 μm separation column for polyphenol analysis.
The analysis of amino acids was performed by HPLC-PDA after derivatization of amino acids with 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate. Briefly, the mobile phase was sodium acetate 140 mM with triethylamine 17 mM at pH 5.05 (A), acetonitrile (B) and Milli-Q water (C). The gradient elution program was a linear gradient elution from 100% A at 0 min to 83% A at 30 min followed by isocratic elution for 10 min of 60% B and 40% C, at flow rate of 1 mL/min, column temperature 37 °C. The chromatogram was monitored at 254 nm [17]. The quantification of polyphenols was carried out following the method proposed by Fabrés-Cerrián et al. [18], with modifications in the extraction phase, which was carried out using a 1:1 ratio of the samples to be analyzed and extracting solution (79.5% methanol + 0.5% hydrochloric acid + 20% water). The mobile phase was 0.1% formic acid in water (D) and acetonitrile (E). The gradient elution program was a linear gradient elution from 85% D and 15% E at 0 min to 50% for both at 40 min, at a flow rate of 1 mL/min, column temperature 35 °C. The chromatogram was monitored at 280 nm.
2.3. Nutritional Efficacy and Biostimulant Effects of MCT® Fertilizers
2.3.1. Biostimulant Effect of MCT® Fertilizers on Same Age Pepper Plants
The first agronomic assay was carried out to test the efficiency of the MCT® fertilizers compared to conventional inorganic fertilizers on pepper plants (Capsicum annuum L.cv. Brocanto). Three different nutrient solutions were tested:
CF: conventional fertilization using only inorganic fertilizers.
HF: nutrient solution made with the MCT® fertilizers: TP, TM, TC, TN, TK, TS.
HFX: nutrient solution made with the same MCT® fertilizers of HF plus the TX fertilizer.
The nutrient concentrations of the all solutions were the following for macronutrients (mmolc/L): N 15, P 2, K 9, Ca 10, Mg 3 and the following for micronutrients (mg/L): Fe 2; Mn 1; Cu 0.1; Zn 4; B 0.5; Mo 0.05 [19]. The quantitative analysis of the nutrients was carried out according to the methodologies described in the Spanish official methods of analysis [20]. The pH was quantified with a Crison GLP 21-pH meter (Hach Lange, Barcelona, Spain) and electrical conductivity (EC) with a Crison micro CM 2200 conductivity meter (Hach Lange, Barcelona, Spain). Nitrates were measured with a Thermo Scientific Orion VersaStar ion selective electrode. NH4+-N was quantified by distillation with the J. P. Selecta ProNitro S (Barcelona, Spain) equipment, phosphorus by the Duval method (measurement of absorbance at 660 nm) by spectrophotometry (Thermo Scientific Genesys 10 uV Scanning). Ca, Mg and Fe were quantified by flame atomic absorption spectrometry, and K was determined by atomic emision (AAnalyst 800, Perkin-Elmer) at the wavelengths of 422.7, 285.2, 248.3 and 766.5 nm, respectively.
The pH levels of the solutions were in the range 6.05–6.90 and EC was in the range of 2.66–3.00 dS/m, values which are suitable for the growth and development of the pepper crop [19]. The higher values of pH and EC of HF and HFX could be explained by the presence of abundant ions (sodium and potassium mostly) bound to the organic matter as counterions of the acid and basic groups.
Pepper plants were grown from 45 days old seedlings provided by a commercial greenhouse (Surinver Coop. V., Murcia, Spain). The seedlings were individually placed in Riviera type containers using perlite as an inert substrate in the research greenhouse of the Autonomous University of Madrid, where a maximum temperature of 28 °C and a minimum of 19 °C were programmed. The three nutrient solution treatments (CF, HF and HFX) were supplied by filling the tank, depending on the crop demand. The deposit was located at the base of the containers, from which it rises by capillarity to the area where the root is located. The assay was performed in triplicate with one plant per container. After 87 days of cultivation, the plants were sampled and analyzed.
2.3.2. Biostimulant Effect of MCT® Fertilizers on Different Age Pepper Plants
A second agronomic assay was carried out to evaluate the effects of MCT® products on the root cell morphology of plants of different ages. Two different nutrient solutions were produced at nutrient concentrations equal as the ones described above.
CF: conventional fertilization using only inorganic fertilizers.
CFX: conventional fertilization using inorganic fertilizers plus the MCT® fertilizer TX.
The nutritional content of the solutions was confirmed by the procedures described in the previous section.
Nine plants from the first batch (used in Section 2.3.1) were kept for 3 months in the culture chamber, with water and conventional nutrient solution. These plants were labeled adult plants. Nine plants freshly taken from a second consignment from the same seed company (Surinver Coop. V., Murcia, Spain) were used as young plants. The two treatments were applied (CF and CFX) by fertigation using 4 L/h drippers through submersible water pumps. The duration of the assay was 60 days and was carried out entirely in a culture chamber where the temperature of 28 and 19 °C and relative humidity of 40% and 60% were controlled, with a day/night cycle of 14 and 10 h, respectively.
2.4. Crop Analysis
2.4.1. Plant Growth Analysis
To quantify plant growth and development of agronomic assays, the weights of plant organs were used as well as the chlorophyll activities. The nitrogen balance index (NBI) (ratio: chorophyll/flavonols activities) obtained by means of the DUALEX Scientific+TM meter was able to provide a rapid and reasonably accurate estimate of leaf chlorophyll content [21] and was measured on the fully developed adult leaves. The other indexes measured were total chlorophylls (Chl), flavonols (Flav) and anthocyanins (Anth).
The nutritional status of the plant was assessed by foliar analysis. Leaves were dried in a forced air oven at 65 °C for 3 days and weighted and analyzed for mineral concentration after dry digestion at 480 °C for 2 h and acid digestion for ash solubilization with HCl 6 M at 90 °C. The analysis of the elements in the pepper leaves was carried out by ICP-MS (NexION 300XX, Perkin-Elmer, Waltham, MA, USA). Foliar nitrogen was quantified by colorimetry (Berthelot’s method), with spectrophotometry at 660 (Thermo Scientific Genesys 10 uV) after Kjeldahl digestion of leaves. The leaf organic matter percentage was done by calcination in a muffle furnace at 450 °C for 4 h.
2.4.2. Root Microscopic Analysis
Images of the pepper plants’ roots were obtained by a transmission electron microscope (TEM). The equipment used were the JEOL JEM1010 Electronic Transmission Microscope (100 kV), the Leica Ultracut S Ultramicrotome and the 1000 Series Vibratomo. The microscopic preparations were made on the fresh roots of the pepper plants. Samples were subjected to cutting processes in 1 cm2 fragments, fixation using 1% glutaraldehyde and 4% formaldehyde in 0.1 M sodium cacodylate at pH 7.4, followed by degassing and treatment with 1% osmium tetroxide, dehydration with increasing concentrations of ethanol and finally acetone and inclusion by infiltration in Durcupan epoxy resin, followed by polymerization in an oven 48 h at 60 °C. Once the sample preparation was carried out, it was cut into sections approximately 60 nm thick and finally subjected to a process of contrast of the sample sections with heavy metals. The analysis of the images was conducted with the image processing program ImageJ v. 1.52.
2.5. Statistical Analysis
The data were statistically evaluated by means of the analysis of the variance with ANOVA one-way followed by the Duncan or Games–Howell post-hoc test (according to Levene variance homogeneity test), with a level of significance of 95% (p ≤ 0.05), using the software IBM SPSS Statistical package for the Social Sciences v20.
3. Results and Discussion
3.1. Chemical Characterization of MCT® Fertilizers
Of all the MCT® fertilizers analyzed, the one with the highest percentage of carbon was the TX (Table 1), which is the product that is most similar to the base used for the addition of the MCT® compounds into the other MCT® fertilizers. Nitrogen appears in greater percentages in the product TX, as well as in the fertilizers with NO3− (TN and TC). Obviously, the highest percentages of sulfur were observed in the products with sulfates in their composition (TM and TS).

Table 1.
Percentages of carbon (C), hydrogen (H), nitrogen (N) and sulfur (S) of the MCT® fertilizers tested. Data were represented as the mean ± standard deviation (n = 4).
Coherent values of other macro- and micronutrients were observed with those that appear in the labeling of the products for the fertilizers used in the design of the nutritive solutions (Table 2). The content of micronutrients was low or very low for many products, except TM and TS. The higher concentration in TS was because this product was fortified with these nutrients.

Table 2.
Concentration (mg/L) of micronutrients in the MCT® fertilizer products.
Figure 1 shows the IR spectra of MCT® fertilizers. The assignation of bands of IR spectra was performed according to previous works on humic substances derived from leonardite and lignite [22,23]. The fertilizer TX is the product that is most similar to the base used for the addition of the MCT® compounds into the other MCT® fertilizers; hence, it was considered as a reference. The most significant bands of the fertilizer TX were at 2700–3750, 1680–1620, 1445–1410, 1045 and 610–560 cm−1. The absorption bands in 3000–4000 cm−1 are due to stretching vibrations of –OH or –NH groups, with varying degrees of hydrogen bonding. The absorption bands at 2920 and 2850 cm−1 were assigned to stretching vibrations of aliphatic –CH, –CH2 and –CH3 side chain groups of aromatic nuclei. The bands at 1410 cm−1 corresponded to bending motions in aliphatic groups. The band at 1450 cm−1 that appears in these products seems to be a typical C=C aromatic compound tension of aromatic rings, although the degree of substitution that they had cannot be determined [24]. The TX product showed a well-defined peak at 1035 cm−1 due to C-O stretching vibrations of ethers and phenols [23]. The double band at 1680–1620 was assigned to amides, ketones or aldehydes and C=C stretching vibration in aromatic compounds.
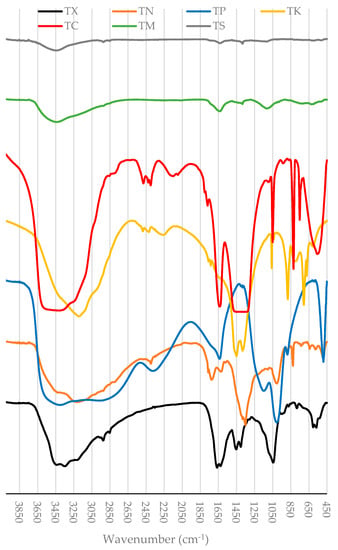
Figure 1.
IR spectra of the MCT® fertilizers used in the work.
IR spectra of the other MCT® fertilizers showed clear differences between products despite their similar origin (TX). One plausible possible explanation of this fact is the interactions between organic compounds and the inorganic fertilizers added to increase the nutrient content of the final products. In this respect, the IR profile of TN and TP showed common modifications, such as the increment of width of stretching vibrations of –OH or –NH groups centered in 3100 cm−1, probably related to the formation of hydrogen bonds [22], and the shift of the peak at 1045 cm−1 to a lower wavenumber, 1000 cm−1. However, clear differences were detected between TN and TP IR spectra. TN showed a peak at 1355 cm−1 that is absent in TP, and the peak at 1645 cm−1 in TP was less intense than the double peak of TN. The fertilizers TK and TC, with fortification with K and Ca, respectively, showed a strong increment of absorbance in the 2700–3750 cm−1 band and the double peak at 1450 cm−1 and new groups of bands centered at 2450 cm−1. However, the IR spectrum of TC conserved the original band of TX at 1635 cm−1 that did not appear in TK. The IR spectra of TM and TS registered very low intensity in comparison with the other products. Probably, the main reason for this was the high concentration of metals, including Mg, in these fertilizers and the high effective charge of metals to form complexes between themselves and the functional groups of organic carbon. The resulting complexes have higher rigidity than the parent organic compounds, reducing the possibility of vibration and flexion of bonds and therefore minimizing the intensity in the IR spectrum.
Free amino acids were only detected and quantified in TX fertilizer (Figure 2). The detected amino acids were glycine and alanine at concentrations of 0.18 and 711 mg L−1, respectively. Free amino acids are easily degradable by physical, biological and chemical factors [25]. Therefore, in the TX product, the organic matter or the metal cations that it contained could be protecting the amino acids from degradation, supplying nutrients to the plants [26]. The amino acid alanine is used by plants as nitrogen storage [27]; meanwhile, glycine is believed to enhance flowering and maintain the water balance between the plant cell and its environment [28].
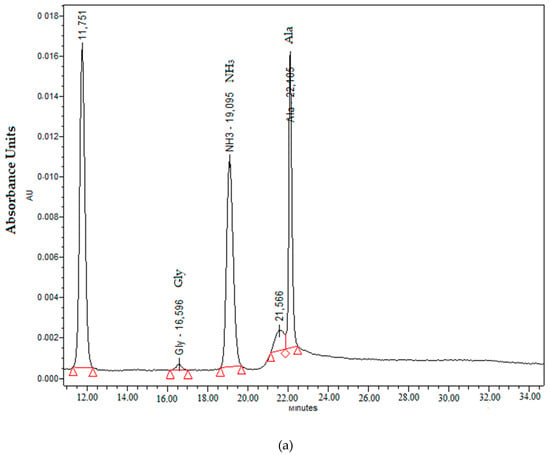
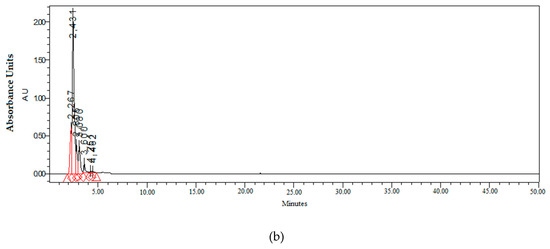
Figure 2.
Chromatogram profiles of the amino acids (a) and polyphenols (b) detected in the TX fertilizer.
The chromatogram profile of polyphenols from TX fertilizer (Figure 2) showed peaks with a low retention time, which meant that they were organic compounds of polyphenolic nature (polyphenol selective column) of reduced molecular size. This was justified by the low retention times that present a rapid elution by the column, demonstrating the existence of molecules of low molecular weight, which is the base of the MCT®, and indicating the degradation of the raw material (leonardite) in portions of low molecular weight. Polyphenols are known for the health benefits that they present, like antioxidant and radical scavenging properties, which could likely contribute to the prevention of oxidative DNA damage [29].
3.2. Nutritional Efficacy and Biostimulant Effect of MCT® Fertilizers
There were no significant differences between CF and fertilization treatments based on MCT® products (HF and HFX) for leaves, stem, root and fruit mass production (Figure 3). Hence, the MCT® products did not stimulate the plant growth in terms of mass production. The literature describes the positive effect of biostimulants on the growth of fruits and vegetables. At the same time, there are studies in which no effect of biostimulants on fruit size or plant growth was found. The lack of biostimulant effects could be explained by the use of a biostimulant that is unsuitable for the tested cultivar or by the lack of the soil factor since biostimulants supported metabolic processes in both soil and plants [30].
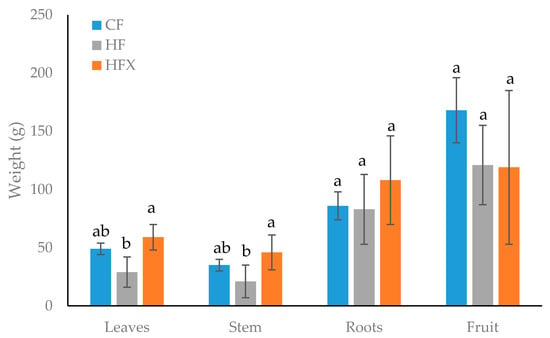
Figure 3.
Fresh weight of leaves, stem and roots of pepper plants fertigated with nutrient solutions made with conventional fertilization (CF), MCT® fertilizers (HF) and MCT® fertilizers plus TX (HFX). Data are represented as the mean ± standard deviation. Different letters indicate significant differences between treatments (p < 0.05, n = 3).
The values of the chlorophyll index NBI (Table 3) followed a similar pattern, in general, to the fresh weights of the plants since both were related [21]. No significant differences were found between CF and HFX treatments for NBI. However, HF treatment reached significantly lower NBI than CF and HFX. These data were in agreement with Karakurt et al. [31], who also observed that the application of humic acids, via foliar or edaphic, influenced significantly the total chlorophyll content. The differences observed in the clorophyll activitites could be related to a higher amount of cloroplasts since biostimulants increase the colour of leaves by stimulating chlorophyll biosynthesis [32]. Guilayn et al. [33] showed a significant effect of biostimulation in roots and less significant for the aerial part, where the morphological effect of humic substances could be less impactful overall. The application of humic substances increases the net photosynthesis of plants under water stress by increasing the rate of gas exchange and electron transport flow in plants [11], improving the responses to water stress by reducing the moisture loss [34] and increasing the number of flowers and fruits [35]. They are able to increase the content of antioxidant compounds such as total content of carotenoids, xanthophylls, beta-carotene, lycopene and chlorogenic acid [36]. Its use is also associated with lower concentrations of sucrose in the roots and leaves (increased glycolysis activity to support metabolic processes) that lead to greater plant growth [12]. However, there is currently a limited understanding of the underlying mechanisms for crop responses after the application of humic substances since more work is needed in field conditions, especially to determine the modifying effects on humic product’s efficacy of environmental and management factors [37].

Table 3.
Dualex indexes of nitrogen balance index (NBI), total chlorophylls (Chl), flavonols (Flav) and anthocyanins (Anth) measured in the fresh leaves of pepper plants fertigated with nutrient solutions made with conventional fertilization (CF), MCT® fertilizers (HF) and MCT® fertilizers plus TX (HFX). Data are represented as the mean ± standard deviation. Different letters indicate significant differences between treatments (p < 0.05, n = 4).
As can be seen in Table 4, significant differences in the content of the macronutrients N, P, Ca and Mg were found, depending on the fertilization treatment used. The treatment HFX implied significantly higher values of N and P in the leaves than the conventional fertilization treatment (CF). However, HF reached a lower N concentration than CF or HFX. This result was in agreement with the leaf NBI values, the CF and HFX treatments being the ones with the highest values (Table 3). Vujinović et al. [38] reported that humic substances caused an enhancement of nitrate uptake rates in plant roots and modulated several genes involved in nitrogen acquisition. The noticeable increment of molecules from the degradation of humic substances in HFX treatment could be the reason behind the highest concentration of foliar N.

Table 4.
Foliar analysis of pepper plants fertigated with nutrient solutions made with conventional fertilization (CF), MCT® fertilizers (HF) and MCT® fertilizers plus TX (HFX). Data are represented as the mean ± standard deviation. Different letters indicate significant differences between treatments (p < 0.05, n = 3).
The use of MCT® fertilizers implied significantly higher concentrations of some micronutrients in leaves than CF (Table 4). HF treatment reached higher concentrations of Mn, B, Mo, Ni, Si and Co than CF. However, a significantly lower concentration of Fe was observed in HF with respect to CF. Similar results were obtained for the HFX treatment. The foliar concentrations of Mn, Zn, Cu, B, Si and Co were significantly higher than CF. However, in this case, no significant differences were found between HFX and CF in the Fe foliar concentration. Previous works reported variations in nutrient concentrations in leaves for macro- [39] and micronutrients [40] in response to biostimulant applications. Polyphenols present in MCT® after humic substance digestion could have improved plant nutrition by metal complexation, promoting the physiological mechanisms involved in nutrient acquisition and inducing changes in root morphology to modulate plant membrane activities [41].
The percentages of foliar organic matter revealed significantly higher values for the HFX treatment (80.7 ± 0.4% a) than CF and HF treatments (77.9 ± 0.6% b and 77 ± 1% b, respectively). These results agree with the better nutrient statuses of plants fertilized with HFX and are in line with Dinçsoy et al. [42], who probed the positive effects of humic acids on plant dry weight, nutrient content and intake and seed germination.
The biostimulant effect of the MCT® fertilizers was clearly shown in the treatment HFX, which improved the foliar nutritional status and foliar organic matter with respect to CF. However, this better nutritional status was not reflected in the plants’ growth maybe related with a not enough duration of the experiment. The biostimulant effect seemed to be related to the use of the TX fertilizer. This product was the MCT® fertilizer with the highest C and N contents and the only one among the fertilizers tested that contained amino acids.
Figure 4 shows the structure of the root cell of pepper fertilized with CF, HF and HFX. The cells of the pepper roots nourished with the MCT® (HF and HFX) products developed a large central vacuole (clearly seen at 1500 and 4000 magnifications in Figure 4) that displaces and compresses the rest of the cell organelles to the cellular membrane. The vacuoles represented 15% of the total root cell area at 4000 magnifications for the CF treatment, 77% for HF and 68% for HFX. Hence, the MCT® fertilizers caused increases in cell membrane permeability and water storage. The existence of this large vacuole meant that the plant could store more water and solutes. This fact could explain the greater resistance to drought, since biostimulants like humic substances and concomitant compounds produced after their digestion are able to improve plant tolerance to abiotic stresses [30] like high temperatures [43] or drought [44] by the modification of the membrane structures of all the subcellular compartments and changes in the composition of the membrane’s lipids [45].
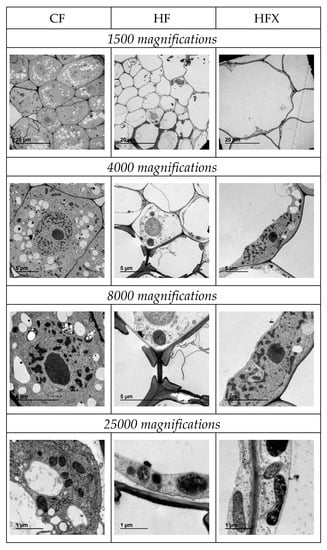
Figure 4.
Transversal images of the roots of pepper plants fertigated with CF, HF and HFX nutrient solutions at 1500, 4000, 8000 and 25,000 magnifications obtained by transmission electron microscopy (TEM).
Leonardite humic acids have positive effects on the architecture of the treated root by stimulating the elongation and proliferation of secondary roots, improving the absorption of nutrients and the efficiency of their use, influencing the metabolism of nitrogen, by increasing the activity of the GS and GOGAT enzymes and therefore the production of proteins [22].
HS are able to improve root and shoot growth through an interconnected action on signaling pathways related to auxin and abscisic acid in roots and to cytokinins in the shoot. This action is hypothesized to be carried out by the release of phytoregulators embedded in the organic matter structure and/or by a mild, beneficial stress associated with the accumulation of humic molecules on the root surface and the partial fouling of cell wall pores on the root surface [46]. They enhanced the activity of nitrate reductase, phosphoglucose isomerase, pyruvate kinase and glycolysis by potentially altering the root membrane permeability [47] through physicochemical interactions with weakly bound supramolecular structures, releasing hormone-like molecules capable of initiating bioactivity in close proximity to root membranes [48].
In the second assay, the effects of CF and CFX at root cell level were assessed in young and adult plants. TEM images of root cells are shown in Figure 5. The inclusion of TX in conventional fertilizer (CFX treatment) generated a large central vacuole in young and adult plants. The percentages of the vacuole areas measured with the ImageJ program were 28% and 83% of the total cells of the young plant roots treated with CF and CFX, respectively, and for the adult plants, the vacuoles constituted 13% and the 98% of the cells for CF and CFX. Hence, the results of the first assay were confirmed, relating the use of the MCT® fertilizers with biostimulant activity with changes in the root morphology. This effect was observed to be enhanced the older the plant was, because there was not such a marked difference in cell morphology in young plants. This is an interesting result because the induction of vacuole growth can be induced in adult crops. Therefore, the use of MCT® fertilizers could be applied once the crop was established and potentially protected from external stress such as extreme temperatures or drought periods. The production of a large vacuole in the root could explain the positive effects of HS towards root weight or tolerance to water stress, as previously reported [30,34].
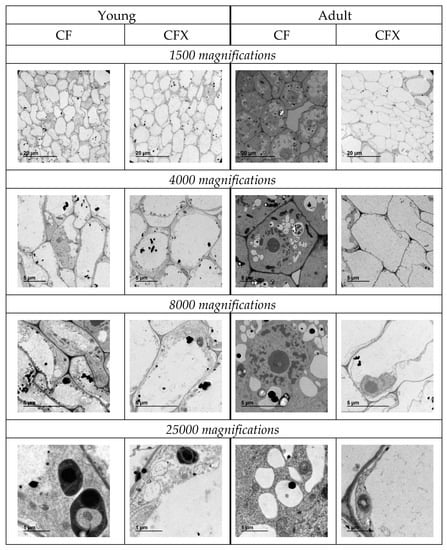
Figure 5.
Transverse photographs of the roots of pepper plants fertigated with conventional fertilization (CF) and conventional fertilization plus TX (CFX) grown in hydroponics at 1500, 4000, 8000 and 25,000 magnifications obtained by transmission electron microscopy (TEM).
The inclusion of biostimulant products such as MCT® fertilizers in modern agriculture can produce multiple advantages in the face of current agricultural threats such as arable land reduction and severe drought, deficit of irrigation and the necessity to reduce mineral fertilization to prevent water pollution [49,50]. Fertilizers which can help the plants to retain more water will propose a good alternative in the future, especially taking into account that plant biomass is closely linked to transpiration [51]. Svietlova et al. [45] reported the modification of the membrane structures of all subcellular compartments and changes in lipid components occurring in plants to ensure the maintenance of optimal nutrient concentrations, increasing the concentration of abscisic acid and the hydraulic conductivity of the root [52].
The existence of clear morphological and structural differences in root cells could suppose under stress conditions an improvement in health and crop production. The future experiments to be carried out will be aimed at solving and studying the parameters of water stress (such as humidity in different parts of the plant) and fruit yield in the long term.
4. Conclusions
MCT® fertilizers have bioactive components of organic nature due to the infrared spectra profile and percentages of carbon found in the products. Foliar analysis indicated the better nutrient status of pepper plants treated with TX MCT® fertilizer than mineral fertilization; but did not affect the plant growth or pepper yield in the short term. It was in the root where the use of MCT® fertilizers showed a clear difference compared to conventional fertilizers due to the change in the root’s organelles size. MCT® fertilizers induce the formation of a large vacuole in root cells, with the dimension reaching almost the complete cell volume. Those morphological differences observed in roots were more noticeable in adult plants. Although more research is needed, the fertilization with biostimulants can clearly improve the water storage of plants grown hydroponically, although the mechanisms of action of these compounds should be clarified. The effectiveness of these products should be tested also in a soil-plant scenario to test the additional effects that they could present if the soil is present.
Author Contributions
R.A.-H. carried out the set-up of the experiment, wrote the original draft and reviewed and edited the final manuscript. C.G.-D. performed data curation, prepared the original draft, contributed to the writing of several sections of the manuscript and edited the final manuscript. B.M. performed analysis of the data and the interpretation of the statistical analysis results. R.C.-A. chemical analyses and the methodology used. E.E. obtained funding, conceive, administered and supervised the project and reviewed and edited the final manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This study is a part of the research agreement “Comparison of agronomic efficiency between Huma Gro® products and mineral conventional fertilizers”, funded by the company Bio Huma Netics, Inc. (Gilbert, Arizona, USA).
Acknowledgments
We acknowledge Bio Huma Netics, Inc., for the supply of the products used. We also thank for their support in the TEM analysis Francisco R. Urbano Olmos and Covadonga Aguado Ballano of the Interdepartmental Research Service, Autonomous University of Madrid.
Conflicts of Interest
This article is the result of a research project funded by the company Bio Huma Netics Inc., although the company did not participate in the experimental procedures or in the evaluation of the results.
References
- Ansari, R.A.; Mahmood, I. Optimization of organic and bio-organic fertilizers on soil properties and growth of pigeon pea. Sci. Hortic. 2017, 226, 1–9. [Google Scholar] [CrossRef]
- Parihar, C.M.; Jat, S.L.; Singh, A.K.; Majumdar, K.; Jat, M.L.; Saharawat, Y.S.; Pradhan, S.; Kuri, B.R. Bio-energy, water-use efficiency and economics of maize-wheat-mungbean system under precision-conservation agriculture in semi-arid agro-ecosystem. Energy 2017, 119, 245–256. [Google Scholar] [CrossRef]
- Michelsen-Correa, S.; Harrison, R.; Dietzen, C. Enhanced efficiency fertilizers (EEFs) do not increase nitrogen retention in Pacific Northwest Douglas-fir forest soils four weeks post-fertilization. For. Ecol. Manag. 2018, 427, 317–324. [Google Scholar] [CrossRef]
- Laursen, K.H.; Mihailova, A.; Kelly, S.D.; Epov, V.N.; Bérail, S.; Schjoerring, J.K.; Donard, O.F.X.; Larsen, E.H.; Pedentchouk, N.; Marca-bell, A.D.; et al. Is it really organic?—Multi-isotopic analysis as a tool to discriminate between organic and conventional plants. Food Chem. 2013, 141, 2812–2820. [Google Scholar] [CrossRef] [PubMed]
- De Pascale, S.; Rouphael, Y.; Colla, G. Plant biostimulants: Innovative tool for enhancing plant nutrition in organic farming. Eur. J. Hortic. Sci. 2017, 82, 277–285. [Google Scholar] [CrossRef]
- López-Salazar, R.; González-Cervantes, G.; Vázquez-Alvarado, R.E.; Olivares-Sáenz, E.; Vidales-Contreras, J.A.; Carranza de la Rosa, R.; Ortega-Escobar, M. Humic and fulvic acid extraction method and characterization by infrared spectrophotometry. Rev. Mex. Cienc. Agrícolas 2010, 5, 1397–1407. [Google Scholar]
- Martínez, C.X.; Bravo, I.; Martin, F.J. Molecular composition of humic acids evaluated by pyrolysis-gas chromatography-mass spectrometry and thermally assisted hydrolysis and methylation in high Colombian Andean soils. Rev. Colomb. Quím. 2013, 42, 31–46. [Google Scholar]
- Dores-Silva, P.R.; Landgraf, M.D.; Rezende, M.O.O. Humification process in different kinds of organic residue by composting and vermicomposting: Have microbioreactors really accelerated the process? Environ. Sci. Pollut. Res. 2018, 25, 17490–17498. [Google Scholar] [CrossRef]
- Quintero, J.; Bonilla, L.J. Obtención de ácidos húmicos por oxidación de carbón con permanganato de potasio y su efecto sobre la disponibilidad de fósforo proveniente de roca fosfórica. Rev. Col. Quím. 1991, 20, 9–14. [Google Scholar]
- Hu, J.; Wu, J.; Qu, X.; Li, J. Effects of Organic Wastes on Structural Characterizations of Humic Acid in Semiarid Soil under Plastic Mulched Drip Irrigation. Chemosphere 2018, 200, 313–321. [Google Scholar] [CrossRef]
- Lotfi, R.; Kalaji, H.M.; Valizadeh, G.R.; Khalilvand Behrozyar, E.; Hemati, A.; Gharavi-Kochebagh, P.; Ghassemi, A. Effects of humic acid on photosynthetic efficiency of rapeseed plants growing under different watering conditions. Photosynthetica 2018, 56, 962–970. [Google Scholar] [CrossRef]
- Conselvan, G.B.; Fuentes, D.; Merchant, A.; Peggion, C.; Francioso, O.; Carletti, P. Effects of humic substances and indole-3-acetic acid on Arabidopsis sugar and amino acid metabolic profile. Plant Soil 2018, 426, 17–32. [Google Scholar] [CrossRef]
- Kobierski, M.; Kondratowicz-Maciejewska, K.; Banach-Szott, M.; Wojewódzki, P.; Peñas Castejón, J.M. Humic substances and aggregate stability in rhizospheric and non-rhizospheric soil. J. Soils Sediments 2018, 18, 2777–2789. [Google Scholar] [CrossRef]
- Tavarini, S.; Passera, B.; Martini, A.; Avio, L.; Sbrana, C.; Giovannetti, M.; Angelini, L.G. Plant growth, steviol glycosides and nutrient uptake as affected by arbuscular mycorrhizal fungi and phosphorous fertilization in Stevia rebaudiana Bert. Ind. Crops Prod. 2018, 111, 899–907. [Google Scholar] [CrossRef]
- Tahiri, A.; Richel, A.; Destain, J.; Druart, P.; Thonart, P.; Ongena, M. Comprehensive comparison of the chemical and structural characterization of landfill leachate and leonardite humic fractions. Anal. Bioanal. Chem. 2016, 408, 1917–1928. [Google Scholar] [CrossRef]
- Pavlovich, L.B.; Strakhov, V.M. Effect of Humic Fertilizers from Brown Coal on the Mineral Composition of Vegetable Crops. Solid Fuel Chem. 2018, 52, 206–210. [Google Scholar] [CrossRef]
- Cooper, C.; Packer, N.; Williams, K. Amino Acid Analysis Protocols; Cooper, C., Ed.; Humana Press: Totowa, NJ, USA, 2001; Volume 159, ISBN 0896036561. [Google Scholar]
- Farrés-Cebrián, M.; Seró, R.; Saurina, J.; Núñez, O. HPLC-UV Polyphenolic Profiles in the Classification of Olive Oils and Other Vegetable Oils via Principal Component Analysis. Separations 2016, 3, 33. [Google Scholar] [CrossRef]
- Cadahía, C.; Eymar, E. Fertirrigación: Cultivos Hortícolas y Ornamentales; Mundi-Prensa: Madrid, Spain, 2000; ISBN 8484762475. [Google Scholar]
- Vallejo, J.M.; Heredia, F.; Blazquez, R.; Cadahía, C.; Gamboa, A.; Guardiola, J.L.; Lachica, M.; Lopez, J.; Pozuelo, J.M.; Arroyo, C. Métodos Oficiales de Análisis, Métodos de Análisis de Productos Orgánicos Fetilizantes; Ministerio de Agricultura, Pesca y Alimentación: Madrid, Spain, 1994; ISBN 847479532X.
- Kalaji, H.M.; Dąbrowski, P.; Cetner, M.D.; Samborska, I.A.; Łukasik, I.; Brestic, M.; Zivcak, M.; Tomasz, H.; Mojski, J.; Kociel, H.; et al. A comparison between different chlorophyll content meters under nutrient deficiency conditions. J. Plant Nutr. 2017, 40, 1024–1034. [Google Scholar] [CrossRef]
- Conselvan, G.B.; Pizzeghello, D.; Francioso, O.; Di Foggia, M.; Nardi, S.; Carletti, P. Biostimulant activity of humic substances extracted from leonardites. Plant Soil 2017, 420, 119–134. [Google Scholar] [CrossRef]
- Nasir, S.; Sarfaraz, T.B.; Verheyen, T.V.; Chaffee, A.L. Structural elucidation of humic acids extracted from Pakistani lignite using spectroscopic and thermal degradative techniques. Fuel Process. Technol. 2011, 92, 983–991. [Google Scholar] [CrossRef]
- Fujitake, N.; Kusumoto, A.; Yanagi, Y.; Suzuki, T.; Otsuka, H. Properties of soil humic substances in fractions obtained by sequential extraction with pyrophosphate solutions at different pHs. Soil Sci. Plant Nutr. 2003, 49, 347–353. [Google Scholar] [CrossRef]
- Devadason, I.P.; Anjaneyulu, A.S.R.; Mendirtta, S.K.; Murthy, T.R.K. Quality and shelf life of buffalo meat blocks processed in retort pouches. J. Food Sci. Technol. 2014, 51, 3991–3997. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, S.; Khoshgoftarmanesh, A.H.; Hadadzadeh, H.; Jafari, M. Synthesis of Iron-Amino Acid Chelates and Evaluation of Their Efficacy as Iron Source and Growth Stimulator for Tomato in Nutrient Solution Culture. J. Plant Growth Regul. 2012, 31, 498–508. [Google Scholar] [CrossRef]
- Carillo, P.; Colla, G.; Fusco, G.M.; Dell’Aversana, E.; El-Nakhel, C.; Giordano, M.; Pannico, A.; Cozzolino, E.; Mori, M.; Reynaud, H.; et al. Morphological and physiological responses induced by protein hydrolysate-based biostimulant and nitrogen rates in greenhouse spinach. Agronomy 2019, 9, 450. [Google Scholar] [CrossRef]
- Khan, N.; Ali, S.; Zandi, P.; Mehmood, A.; Ullah, S.; Ikram, M.; Ismail; Shahid, M.A.; Babar, A. Role of sugars, amino acids and organic acids in improving plant abiotic stress tolerance. Pakistan J. Bot. 2020, 52, 355–363. [Google Scholar] [CrossRef]
- Di Sotto, A.; Vecchiato, M.; Abete, L.; Toniolo, C.; Maria, A.; Mannina, L.; Locatelli, M.; Nicoletti, M.; Di, S. Capsicum annuum L. var. Cornetto di Pontecorvo PDO: Polyphenolic profile and in vitro biological activities. J. Funct. Foods 2018, 40, 679–691. [Google Scholar] [CrossRef]
- Drobek, M.; Frąc, M.; Cybulska, J. Plant biostimulants: Importance of the quality and yield of horticultural crops and the improvement of plant tolerance to abiotic stress-a review. Agronomy 2019, 9, 335. [Google Scholar] [CrossRef]
- Karakurt, Y.; Unlu, H.; Unlu, H.; Padem, H. The influence of foliar and soil fertilization of humic acid on yield and quality of pepper. Acta Agric. Scand. 2009, 59, 233–237. [Google Scholar] [CrossRef]
- Bulgari, R.; Franzoni, G.; Ferrante, A. Biostimulants application in horticultural crops under abiotic stress conditions. Agronomy 2019, 9, 306. [Google Scholar] [CrossRef]
- Guilayn, F.; Benbrahim, M.; Rouez, M.; Crest, M.; Patureau, D.; Jimenez, J. Humic-like substances extracted from different digestates: First trials of lettuce biostimulation in hydroponic culture. Waste Manag. 2020, 104, 239–245. [Google Scholar] [CrossRef]
- Qin, K.; Leskovar, D.I. Lignite-derived humic substances modulate pepper and soil-biota growth under water deficit stress. J. Plant Nutr. Soil Sci. 2018, 181, 655–663. [Google Scholar] [CrossRef]
- Arancon, N.Q.; Edwards, C.A.; Lee, S.; Byrne, R. Effects of humic acids from vermicomposts on plant growth. Eur. J. Soil Biol. 2006, 42, 65–69. [Google Scholar] [CrossRef]
- Karakurt, Y.; Ozdamar-Unlu, H.; Unlu, H.; Tonguc, M. Antioxidant compounds and activity in cucumber fruit in response to foliar and soil humic acid application. Eur. J. Hortic. Sci. 2015, 80, 76–80. [Google Scholar] [CrossRef]
- Olk, D.C.; Dinnes, D.L.; Rene Scoresby, J.; Callaway, C.R.; Darlington, J.W. Humic products in agriculture: Potential benefits and research challenges—A review. J. Soils Sediments 2018, 18, 2881–2891. [Google Scholar] [CrossRef]
- Vujinović, T.; Zanin, L.; Venuti, S.; Contin, M.; Ceccon, P.; Tomasi, N.; Pinton, R.; Cesco, S.; De Nobili, M. Biostimulant Action of Dissolved Humic Substances From a Conventionally and an Organically Managed Soil on Nitrate Acquisition in Maize Plants. Front. Plant Sci. 2019, 10, 1652. [Google Scholar] [CrossRef]
- Szczepanek, M.; Siwik-Ziomek, A. P and K accumulation by rapeseed as affected by biostimulant under different NPK and S fertilization doses. Agronomy 2019, 9, 477. [Google Scholar] [CrossRef]
- Soppelsa, S.; Kelderer, M.; Casera, C.; Bassi, M.; Robatscher, P.; Matteazzi, A.; Andreotti, C. Foliar applications of biostimulants promote growth, yield and fruit quality of strawberry plants grown under nutrient limitation. Agronomy 2019, 9, 483. [Google Scholar] [CrossRef]
- Zanin, L.; Tomasi, N.; Cesco, S.; Varanini, Z.; Pinton, R. Humic substances contribute to plant iron nutrition acting as chelators and biostimulants. Front. Plant Sci. 2019, 10, 675. [Google Scholar] [CrossRef]
- Dinçsoy, M.; Sönmez, F. The effect of potassium and humic acid applications on yield and nutrient contents of wheat (Triticum aestivum L. var. Delfii) with same soil properties. J. Plant Nutr. 2019, 42, 2757–2772. [Google Scholar] [CrossRef]
- Francesca, S.; Arena, C.; Hay Mele, B.; Schettini, C.; Ambrosino, P.; Barone, A.; Rigano, M.M. The use of a plant-based biostimulant improves plant performances and fruit quality in tomato plants grown at elevated temperatures. Agronomy 2020, 10, 363. [Google Scholar] [CrossRef]
- Petropoulos, S.A.; Fernandes, Â.; Plexida, S.; Chrysargyris, A.; Tzortzakis, N.; Barreira, J.C.M.; Barros, L.; Ferreira, I.C.F.R. Biostimulants application alleviates water stress effects on yield and chemical composition of greenhouse green bean (phaseolus vulgaris l.). Agronomy 2020, 10, 182. [Google Scholar] [CrossRef]
- Svietlova, N.; Sytar, O.; Volkogon, M. Remodeling of the composition of the membrane’s lipids of buckwheat plants (Fagopyrum esculentum Moench.) under conditions of phosphorous deficiency and seed bacterization with phosphate solubilizing microorganisms. J. Cent. Eur. Agric. 2017, 18, 879–888. [Google Scholar] [CrossRef]
- Olaetxea, M.; De Hita, D.; Garcia, C.A.; Fuentes, M.; Baigorri, R.; Mora, V.; Garnica, M.; Urrutia, O.; Erro, J.; Zamarreño, A.M.; et al. Hypothetical framework integrating the main mechanisms involved in the promoting action of rhizospheric humic substances on plant root-and shoot-growth. Appl. Soil Ecol. 2018, 123, 521–537. [Google Scholar] [CrossRef]
- Palumbo, G.; Schiavon, M.; Nardi, S.; Ertani, A.; Celano, G.; Colombo, C.M. Biostimulant Potential of Humic Acids Extracted From an Amendment Obtained via Combination of Olive Mill Wastewaters (OMW) and a Pre-treated Organic Material Derived From Municipal Solid Waste (MSW). Front. Plant Sci. 2018, 9, 1028. [Google Scholar] [CrossRef]
- Bento, L.R.; Melo, C.A.; Ferreira, O.P.; Moreira, A.B.; Mounier, S.; Piccolo, A.; Spaccini, R.; Bisinoti, M.C. Humic extracts of hydrochar and Amazonian Dark Earth: Molecular characteristics and effects on maize seed germination. Sci. Total Environ. 2020, 708, 135000. [Google Scholar] [CrossRef]
- Wang, Y.; Janz, B.; Engedal, T.; de Neergaard, A. Effect of irrigation regimes and nitrogen rates on water use efficiency and nitrogen uptake in maize. Agric. Water Manag. 2017, 179, 271–276. [Google Scholar] [CrossRef]
- Hou, X.; Zhang, W.; Du, T.; Kang, S.; Davies, W.J. Responses of Water Accumulation and Solute Metabolism in Tomato Fruit to Water Scarcity and Implications for Main Fruit Quality Variables. J. Exp. Bot. 2020, 71, 1249–1264. [Google Scholar] [CrossRef]
- Blum, A. Effective use of water (EUW) and not water-use efficiency (WUE) is the target of crop yield improvement under drought stress. F. Crop. Res. 2009, 112, 119–123. [Google Scholar] [CrossRef]
- Olaetxea, M.; Mora, V.; Bacaicoa, E.; Garnica, M.; Fuentes, M.; Casanova, E.; Zamarreño, A.M.; Iriarte, J.C.; Etayo, D.; Ederra, I.; et al. Abscisic Acid Regulation of Root Hydraulic Conductivity and Aquaporin Gene Expression Is Crucial to the Plant Shoot Growth Enhancement Caused by Rhizosphere Humic Acids. Plant Physiol. 2015, 169, 2587–2596. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).