1. Introduction
Weeds reduce crop yield by competing for nutrients, light, and moisture [
1]. Sustainable weed management is one of the main challenges for both conventional and organic horticulture [
2]. Current weed control in horticultural production includes synthetic herbicides and physical methods [
3].
Recently, bioherbicides have been developed from allelochemicals [
4]. Allelochemicals, such as saponins, tannins, flavonoids, terpenoids, and lactones, are plant secondary metabolites that can be found in different plant organs, such as in the leaves, stems, roots, seeds, fruits, flowers, and pollen [
5,
6]. These molecules are produced by plants and are released in the surrounding environment to affect the growth, development, survival, and reproduction of neighboring plants, directly or indirectly; this phenomenon is known as allelopathy [
7,
8]. Their use is of interest in weed management as they are biodegradable, have structural complexity, and a wide variety of sites of action. Additionally, they are not halogenated and are generally safer for non-target organisms [
9,
10,
11,
12]. For this purpose, several studies have aimed to identify species with phytotoxic activity [
13]. Rice (
Oryza sativa L.) hull extracts inhibit
Echinochloa crus-galli (L.) Beauv. germination, seedling growth, and weight [
14], and plant extracts of
Everniastrum sorocheilum (Vain.) Hale ex Sipman,
Usnea roccellina Motyka, and
Cladonia confusa R. Sant. inhibit the germination and root growth of
Trifolium pratense L. [
15]. In addition, seed extracts from
Sicyos angulatus L. inhibit the germination of
Lactuca sativa L. [
16].
Ailanthus altissima (Mill.) Swingle is a perennial invasive species known to cause major negative impacts on human activities, especially in urban areas. Since 1959, it has been known that
A. altissima produces phytotoxic compounds and that the major identified toxin is in the quassinoid ailanthone (Ail), firstly isolated by Heisey [
17] from bark and foliage. Quassinoids are highly oxygenated triterpenes, which have been isolated as bitter principles from the plants of the Simaroubaceae family. Their synthesis has attracted much attention because of the wide spectrum of their biological properties. The extracts obtained from different parts of
A. altissima have been reported to exhibit diverse biological activities, such as antiproliferative, central nervous depressant, antimicrobial, and antioxidant activities [
18,
19,
20,
21,
22,
23,
24]. Ail phytotoxic activity has been observed both in the pre-emergence and the post-emergence stages of several species, showing a broad spectrum of herbicidal effects on monocots and dicots under controlled conditions [
25,
26]. More recently, Demasi et al. [
12,
27] tested Ail in a growth chamber and controlled cultivation conditions, highlighting its extremely efficient phytotoxicity on the seed germination of
Lepidium sativum L. “Inglese” and
Raphanus sativus L. “Tondo Rosso BIO”.
Nevertheless, as well as other natural compounds, several constraints have so far prevented the commercial development of natural Ail-based herbicides [
24,
28,
29,
30,
31,
32,
33]. The main ailanthone drawbacks are its high extraction and purification costs and its short-term efficacy in soil, demonstrated both in greenhouses [
24,
34] and fields [
25]. As emerges from the literature, ailanthone is not the only molecule with phytotoxic activity present in the tissues of
A. altissima, but it acts better in synergy with other compounds that differ on the basis of the used tissue [
35].
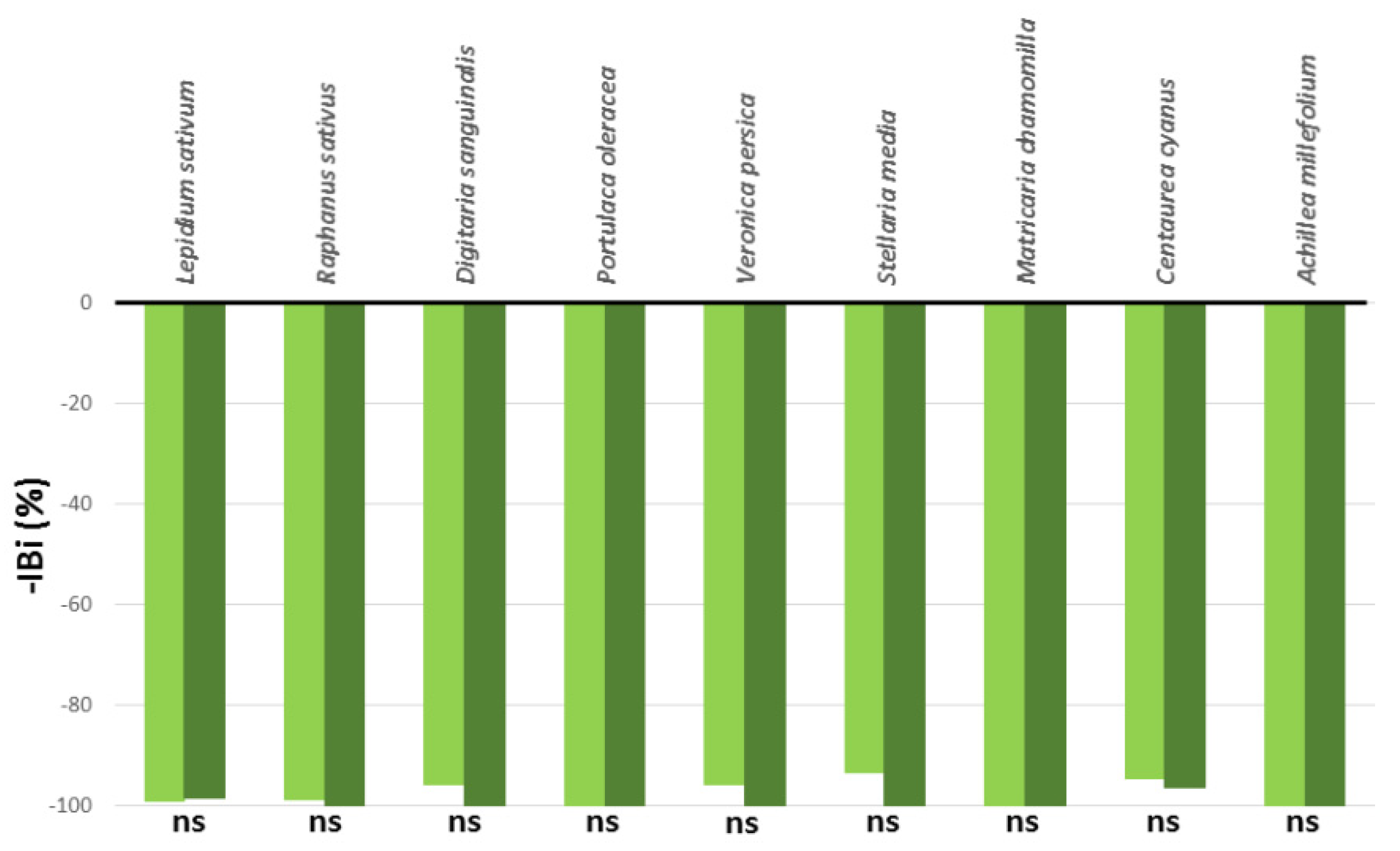
Thus, this study aimed to provide new insights into the phytotoxicity of A. altissima extracts obtained from the leaves, samaras, rachises, and secondary roots in pre-emergence and to assess their application potential for weed control in horticulture. Four extracts at different dilutions were evaluated by a combination of in vitro and in vivo bioassays on two indicator species (i.e., Lepidium sativum L. and Raphanus sativus L.), seven common weeds (i.e., Achillea millefolium L., Centaurea cyanus L., Matricaria chamomilla L., Portulaca oleracea L., Stellaria media (L.) Vill., Digitaria sanguinalis (L.) Scop., and Veronica persica Poir.), and during the cultivation of three horticulture crops (i.e., Salvia officinalis L., Salvia rosmarinus (L.) Schleid., and Dianthus caryophyllus L.).
4. Discussion
Ailanthus altissima water extracts were used to simulate the natural release of water-soluble phytotoxins into the environment. The measurement of the physiochemical properties of aqueous extracts, such as pH or conductivity, is commonly explored, since they can cause changes in cellular processes that could be mistaken as possible phytotoxic effects [
38]. Taking into account that the used extracts present pH values around 4.5 and electrical conductivity (EC) values below 0.02 dS m
−1 (data not shown), the inhibitory effects observed in the bioassays could be attributed a priori to the phytotoxins present in the extracts.
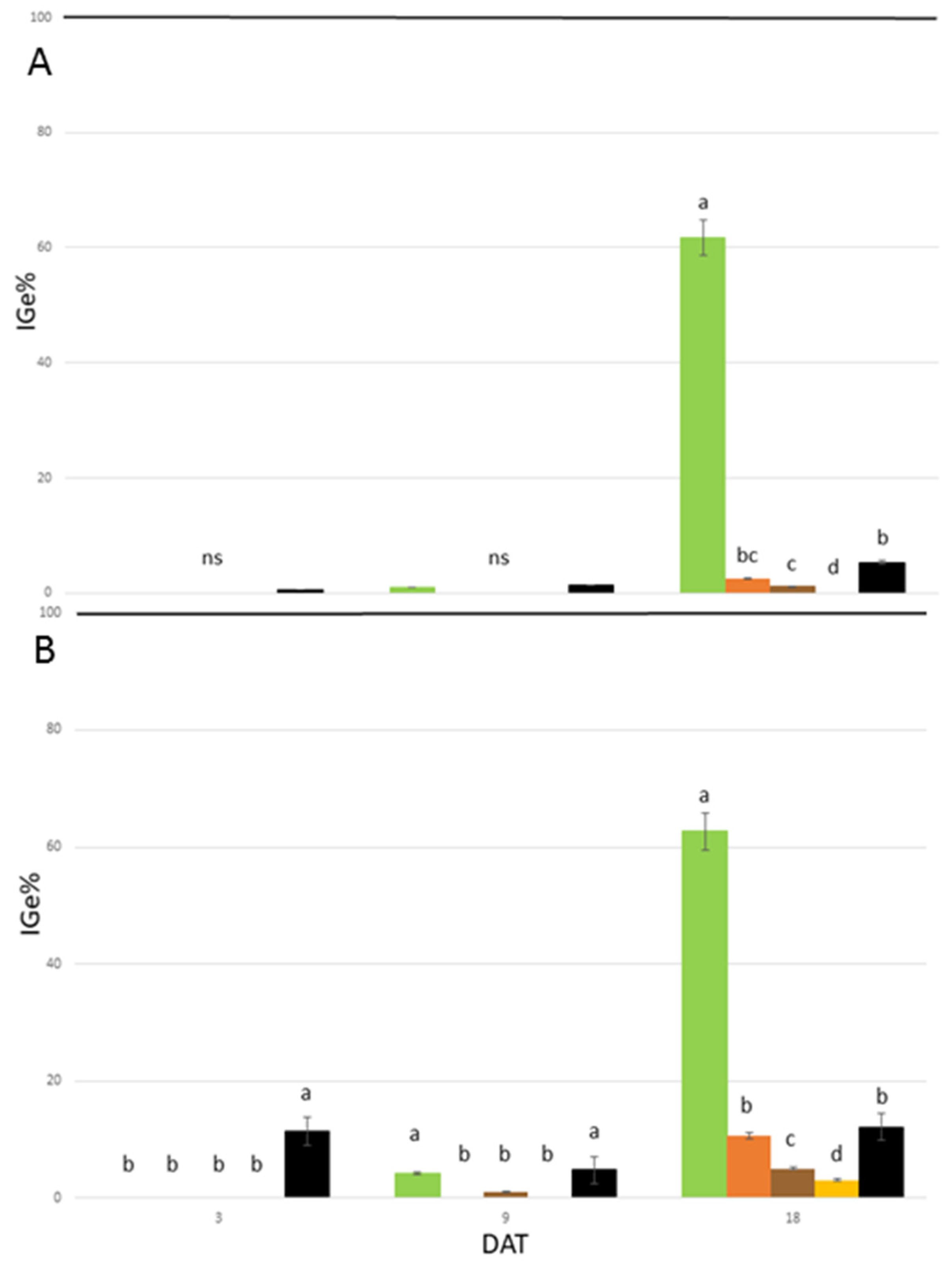
In the in vitro phytotoxicity tests, among the measured macroscopic traits, seed germination and root growth are considered as the most sensitive criteria to detect the toxicity of allelopathic compounds [
39,
40]. In turn, the seed index of germination (IGe%) is directly affected by root growth and germination and may thus contribute to the unraveling of the herbicidal effects of the studied extracts. In the present study, all the applied
A. altissima extracts showed a marked inhibition in the seed germination and root growth of the indicator plants
L. sativum and
R. sativus during the growth chamber assay. Thus, this indicates that they could be a promising natural herbicide. The inhibition of IGe% was observed in the seeds of all the treated species at different concentrations and during the entire experiment, with some limitations at 1.8 mg L
−1 Ail, particularly at the end of the trial. However, some differences among extract types were observed. Among them, the secondary root-derived extract presents the most promising efficacy in reducing seed germination and root growth under in vitro germination conditions. In addition, the leaf, samara, and rachis extracts presented results of interest, albeit to a lesser extent. The concentration of 25 mg L
−1 Ail was found to be the most promising, both in terms of its efficacy and persistence for all extracts. As shown in
Figure 2, by comparing the effects of the extracts used in this study with the results obtained by Demasi et al. [
12,
27] with pure Ail, there are significant differences between the treatments at the concentration of 25 mg L
−1 Ail.
In R. sativus, the studied extracts affected IGe% as well as pure Ail (IGe% values ranged between −98.60% and −99.75%) without significant differences up to 9 DAT. At 18 DAT, the leaf extract was the least effective, while the rachis and secondary root extracts significantly reduced IGe% in comparison to pure Ail. In L. sativum, the tested extracts were significantly more effective than the pure Ail up to 9 DAT, with the exception of the leaf extract (9 DAT). At the end of the experiment, a pattern similar to R. sativus was observed.
Although the leaf extract was found to be less effective and less persistent than the other extracts tested in the growth chamber assay, it was nevertheless used for the greenhouse and nursery assays, because leaf biomass is more available and easy to collect and process. This result implies that plant extracts obtained by the applied multistep solvent extraction method can be successfully used without incurring huge purification costs, as for quassinoids such as Ail. These findings are in agreement with the known pronounced phytotoxic activity of the outer tissues of
A. altissima, such as the trunk bark of the rachis and, in particular, of the roots [
25,
34]. Only few and ambiguous reports on the phytotoxic activity of
A. altissima extracts under in vitro conditions are present in the literature. Tsao et al. [
41] highlighted that the secondary metabolite extracts from the leaves of
A. altissima have inhibitory effects on the seed germination and plant growth of
Medicago sativa L. El Ayeb-Zakhama et al. [
42] evaluated the phytotoxicity of
A. altissima essential oils obtained from different parts of the plant. In agreement with our results, these authors observed a dose-dependent inhibitory effect on
L. sativa seed germination and growth (range between −50% and −85%), with the essential oil obtained from the roots as the most effective. In contrast, Bagheri and Cici [
20] showed no statistical differences among the leaf, stem, root, bark, and fruit extracts on
L. sativum seed germination, with only limited inhibition (circa −5%). However, the same authors indicated that a marked reduction in radicle length was observed, with the highest activity from the bark and root (circa −65%) extracts under greenhouse conditions. De Feo et al. [
35] indicated that water extracts from the roots and leaves resulted in the inhibition of circa 80%, 70%, and 50% of the germination of
L. sativum,
R. sativus, and
P. oleracea, respectively, markedly less than our studied extracts. Our extracts were more effective than that, reported in the abovementioned studies. The effectiveness of a phytoextract depends on the extraction procedure used. Specifically, at the moment there are no studies in the literature that have used the method proposed in this work. The advantage in utilizing water extracts rather than a single purified compound is suggested by many authors, demonstrating that a mixture of compounds can act synergistically and could be more phytotoxic than their respective individual compounds [
2]. Furthermore, the applied water extracts could help in reducing the dose of the application of synthetic herbicides, if applied in combination, serving as an effective management strategy in controlling weeds without causing much harm to the environment quality [
43]. Jabran et al. [
44] reported a reduction in weed population by using a mixture of water extracts of
Helianthus annuus L. and
Oryza sativa L. when applied in combination, with a significant reduction in the dose of pendimethalin. Accurate scientific studies must, however, be conducted to evaluate the environmental impact of
A. altissima water extracts, for any other pesticides or biologically active compounds used in agriculture.
Based on these promising results, further in vivo experiments were conducted, this time both under greenhouse and nursery conditions by testing more seed species belonging to the weed and horticulture sector. Although the leaf extract was found to be less effective and less persistent than the other extracts tested, it was nevertheless selected for this purpose. A higher amount of this extract can be obtained because leaf biomass is more abundant than the other organs and is easier to collect and process, leading to lower production costs. Moreover, considering that an organic substrate is buffered due to its physico-chemical properties, higher concentrations (50, 100, and 200 mg L−1 Ail) were evaluated compared to the growth chamber assay, where filter paper was used.
In the greenhouse experiment, all of the treated weed species were strongly inhibited by the used leaf extract, without differences among extract concentrations, suggesting the possibility to further reduce the concentration of this leaf extract. In the literature, it is reported that the influence of phytotoxic compounds on seed germination also depends on the size and permeability of the seed integument [
45,
46], and that seeds of weeds tend to be less sensitive to the action of phytochemicals because the phytotoxic effects can be ameliorated due to lower absorption and translocation or faster degradation of phytotoxins [
47].
In the present assay, as also observed in the laboratory trial, only a residual number of roots emerged from the seeds, but their development was inhibited early, resulting in senescence and rotting. This can be explained by the fact that the secondary root surfaces are more permeable to our extracts in comparison to hypocotyl surfaces due to the presence of a less pronounced protective cuticle layer, which can result in a greater penetration and concentration of these compounds in root tissues [
48]. However, in this regard, little is known about the mode of action of
A. altissima natural compounds in inhibiting seed germination. Dayan et al. [
49] and Duke et al. [
29] suggested that ailanthone, similar to other quassinoids, might act as a mitosis inhibitor. The strong inhibitory activity of quassinoids, particularly on seed germination and early root growth, corroborates previous studies that investigated the inhibitory effects of other terpenoids obtained from
Ageratina adenophora (Spreng.) R.M. King & H. Rob. and
Drimys brasiliensis Miers on the early growth of
O. sativa,
Barbarea verna (Mill.) Asch.,
Echinochola crus-galli (L.) P. Beauv., and
Ipomea grandiflora (Dammer) O’Donnell [
50,
51]. This has already been interpreted as a result of the loss of mitotic activity, capable of reducing both the germination and seedling growth of several seed species, such as
Amaranthus retroflexus L. and
Setaria viridis (L.) P. Beauv., treated with different plant extracts and essential oils [
52]. Very little information about
A. altissima extracts in pot cultivation under greenhouse conditions has been reported. Bagheri and Cici [
20] affirmed that the root extract had no effect on
E. crus-galli growth, while it significantly affected the dry biomass of different weed species, such as
A. retroflexus,
Abutilion theophrasti Medik., and
Carthamus tinctorius L. However, the indicated reduction was lower than in this experiment, settling in a range between −34% and −80%.
Ailanthone stability in a sterile environment has already been demonstrated by previous tests performed by Heisey [
24], in which biological activity was detected on sterile substrates for up to 21 days, versus 3–5 days on non-sterile substrates. More recently, Demasi et al. [
12,
27] reported that Ail in paper was extremely active at low doses (i.e., 7.5 mg L
−1) up to 30 DAT on radish and 20 DAT on garden cress, while on cultivation substrate, it lasted 30 DAT on radish when using a concentration of 60 mg L
−1. These authors also indicated that Ail remained highly toxic up to 21 DAT if the soil was sterilized. Here, the assays in the growth chamber and the greenhouse showed that the studied extracts are able to maintain their phytotoxic activity for a period ranging between 18 and 30 DAT, both in sterile and, as is particularly evident, in non-sterile substrates.
Further details highlighting the biological effects and the persistence of the leaf extract were provided by the two experiments performed in the nursery, which, to the best of our knowledge, is the first time such effects have been noted. In this context, the leaf extract showed a potential herbicidal effect in pre-emergence for the whole duration of both experiments, particularly in the first. In this experiment, the substrate contained only a few weed seeds (1.3–3.1 weeds per pot germinated); therefore, it was not possible to evaluate the susceptibility of different weed species to the studied extracts, as was done in the greenhouse assay. For the same reason, the parameter “weed presence per pot (%)” was used instead of the commonly used “weed density.” The lower phytotoxic activity highlighted in the second experiment could be related to the higher temperatures of late spring (mean temperature ranging between 11.2 and 17.2 °C during experiment 1, and between 20.5 and 24.1 °C during experiment 2), which more likely increased the number and the growth of weeds.
Bioherbicides are a future target for development in weed management, although research in this area has been somewhat slow. As reported by Seiber et al. [
53], the toxicity of non-target species is one of the main problems that limit the use of natural compounds as herbicides. Here, during the first experiment in the nursery, the leaf extract negatively affected the plant growth and health status of the cultivated crops, particularly in
S. officinalis and slightly less in
S. rosmarinus. The senescence and the presence of wilted leaves in the treated crops might indicate a reduction in chlorophyll content and the inhibition of the photosynthesis [
54]. Such observations could reflect different modes of action, depending on the site of phytotoxin entry, with the growth and morphological parameters being affected early by spraying on the canopy. The phytotoxic effect on
D. caryophyllus was markedly less intense. This behavior could be due to the selectivity of the phytocomplex contained within the extract. However, at present, it is not yet possible to accurately explain the selectivity of these complexes. Meanwhile, the indirect effects on the growth and quality of the untreated
S. officinalis and
S. rosmarinus plants can be supposed as the effect of the volatilization of some herbicide-carrying molecules during or after treatment, although there is currently no bibliographical evidence. Mastelic and Jerkovic [
55] characterized the volatiles produced by
A. altissima plants and listed several oxygenated aliphatic compounds and terpenes that are commonly known for their phytotoxicity. On the other hand, during the second experiment, the highest leaf extract concentration promoted the growth of plants. Evidence of the growth stimulation of crops by plant-released compounds is available in the literature [
56,
57]. However, this phenomenon, called hormesis [
58], is usually related to lower doses of phytotoxins, not as high as in this study, with the leaf extract at 200 mg L
−1 Ai eq. Regarding this topic, Hussain et al. [
59] espoused that a foliar spray containing 3% aqueous extract of
Moringa oleifera Lam.,
Sorghum vulgare Pers., and
Brassica rapa L. increases maize grain yield by circa 50%. Unfortunately, the literature is lacking data on these aspects, and further studies are needed to elucidate them.