CO2 Emission and Change in the Fertility Parameters of a Calcareous Soil Following Annual Applications of Deinking Paper Sludge (The Case of Tunisia)
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
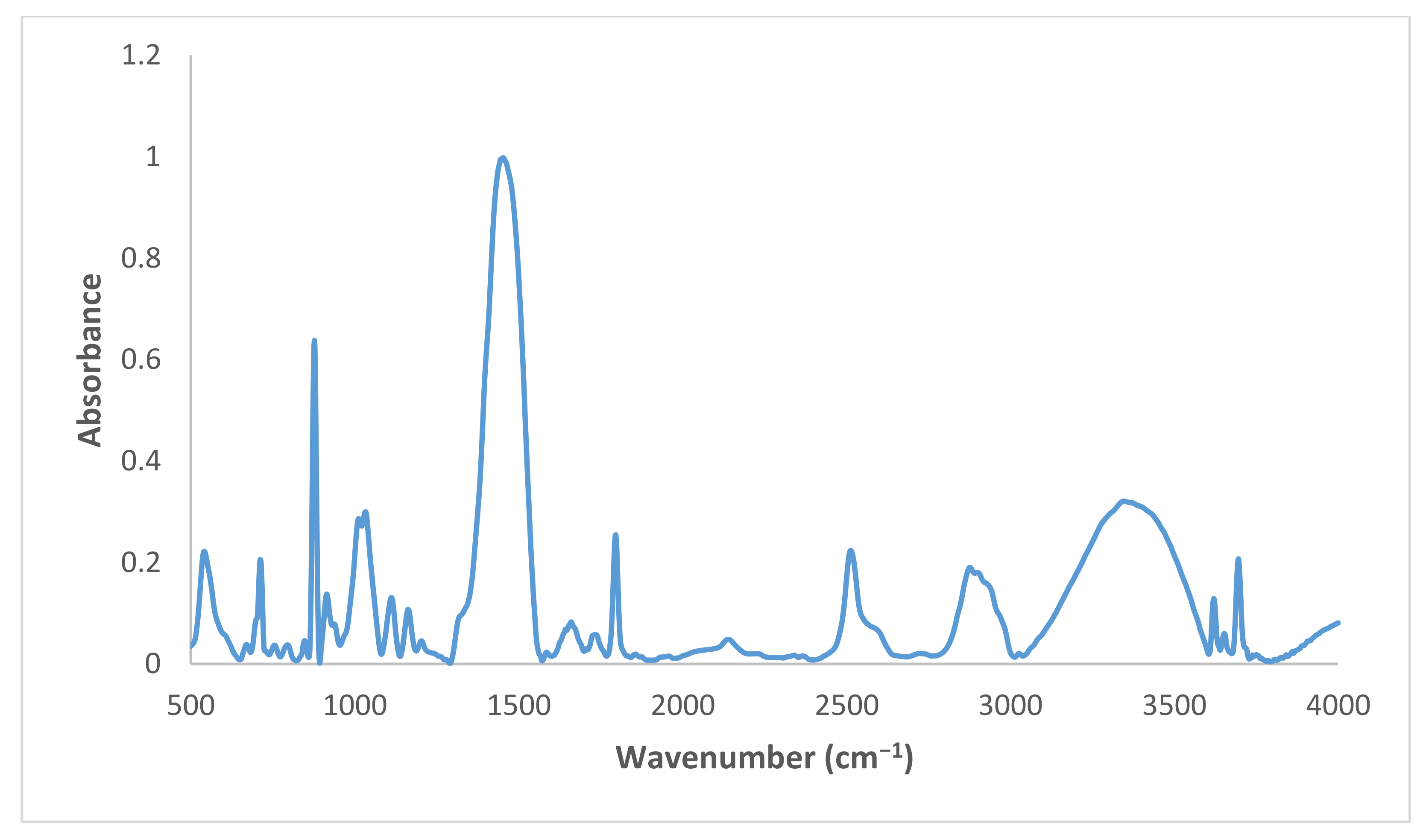
2.2. Characterization of DPS
2.3. Soil Analyses
2.3.1. Soil Physical and Chemical Analyses
2.3.2. Soil Biological Analysis
Determination of Microbial Biomass
Carbon Mineralization of DPS-Amended Soil
2.3.3. Microbial Metabolic Quotient
2.4. Statistical Analyses
3. Results
3.1. Characterization of DPS
3.2. Effect of DPS Application on Soil Chemical Properties
3.2.1. Effect on Soil pH
3.2.2. Effect on Soil Electrical Conductivity
3.2.3. Effect on Soil OM Content
3.2.4. Effect on the Total Soil N Content
3.2.5. Effect on Soil Available P
3.2.6. Effect on Soil Exchangeable Bases (K+, Ca2+, and Na+)
3.3. Effect of DPS on Soil Physical Properties
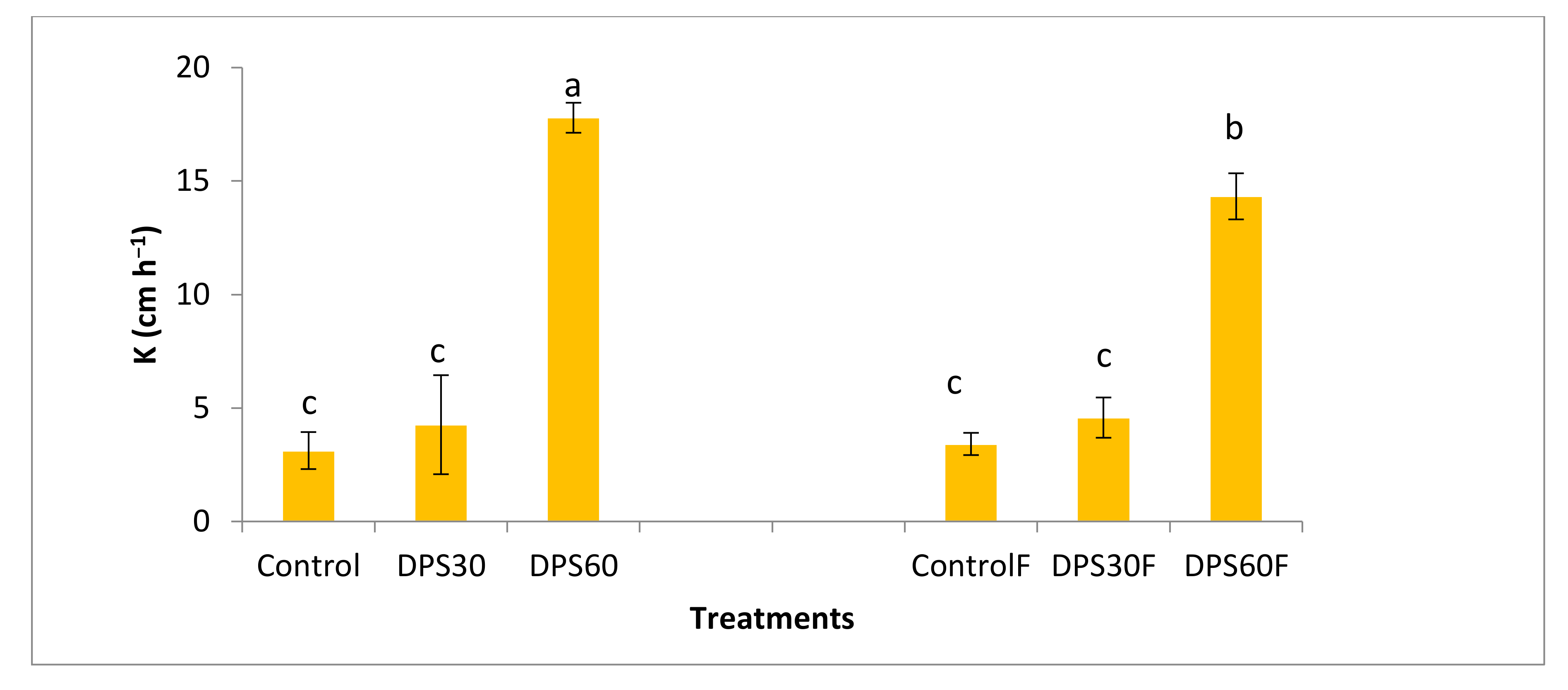
3.3.1. Effect on Soil Permeability
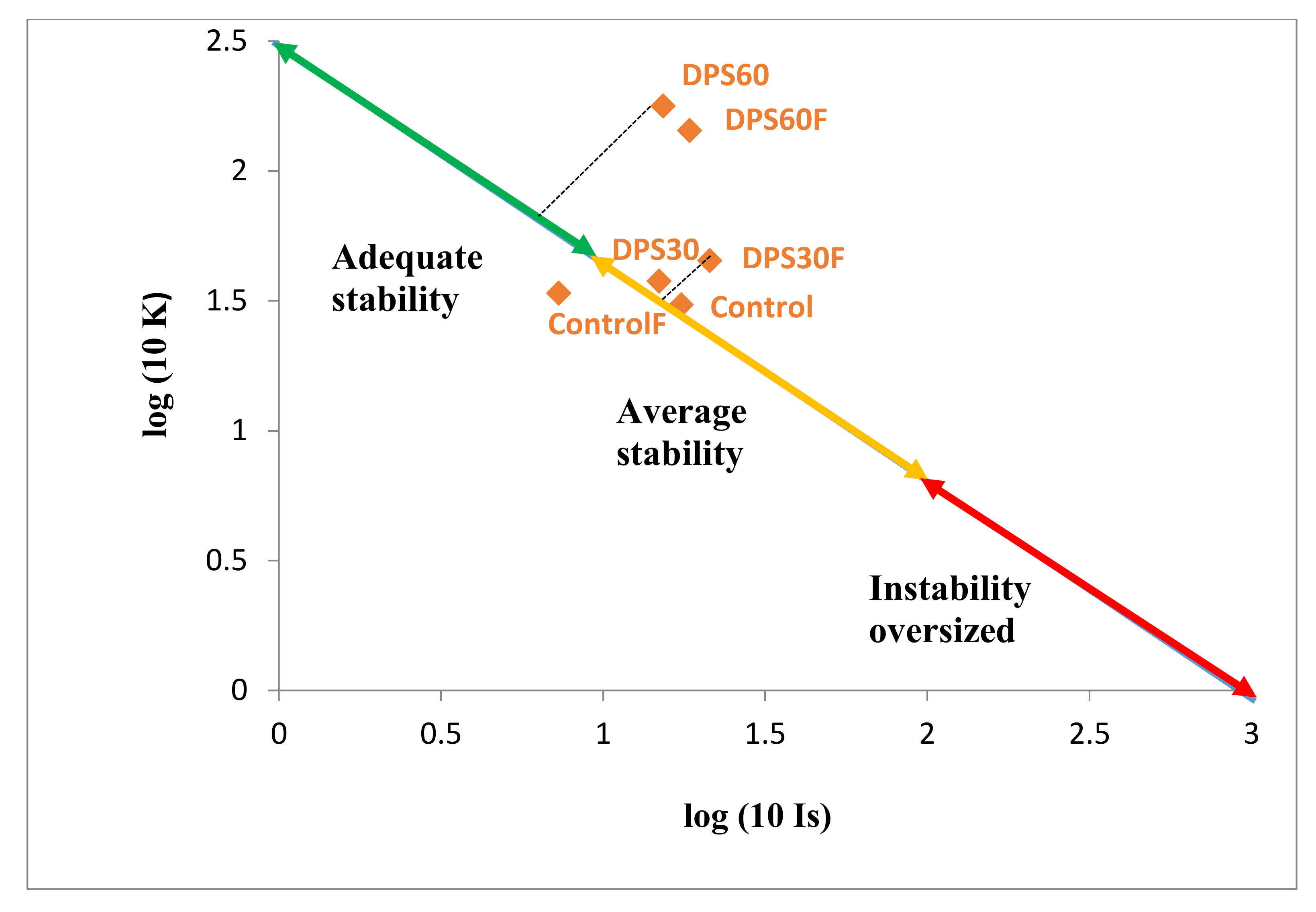
3.3.2. Effect on Structural Stability
3.4. Effect on Soil Biological Parameters
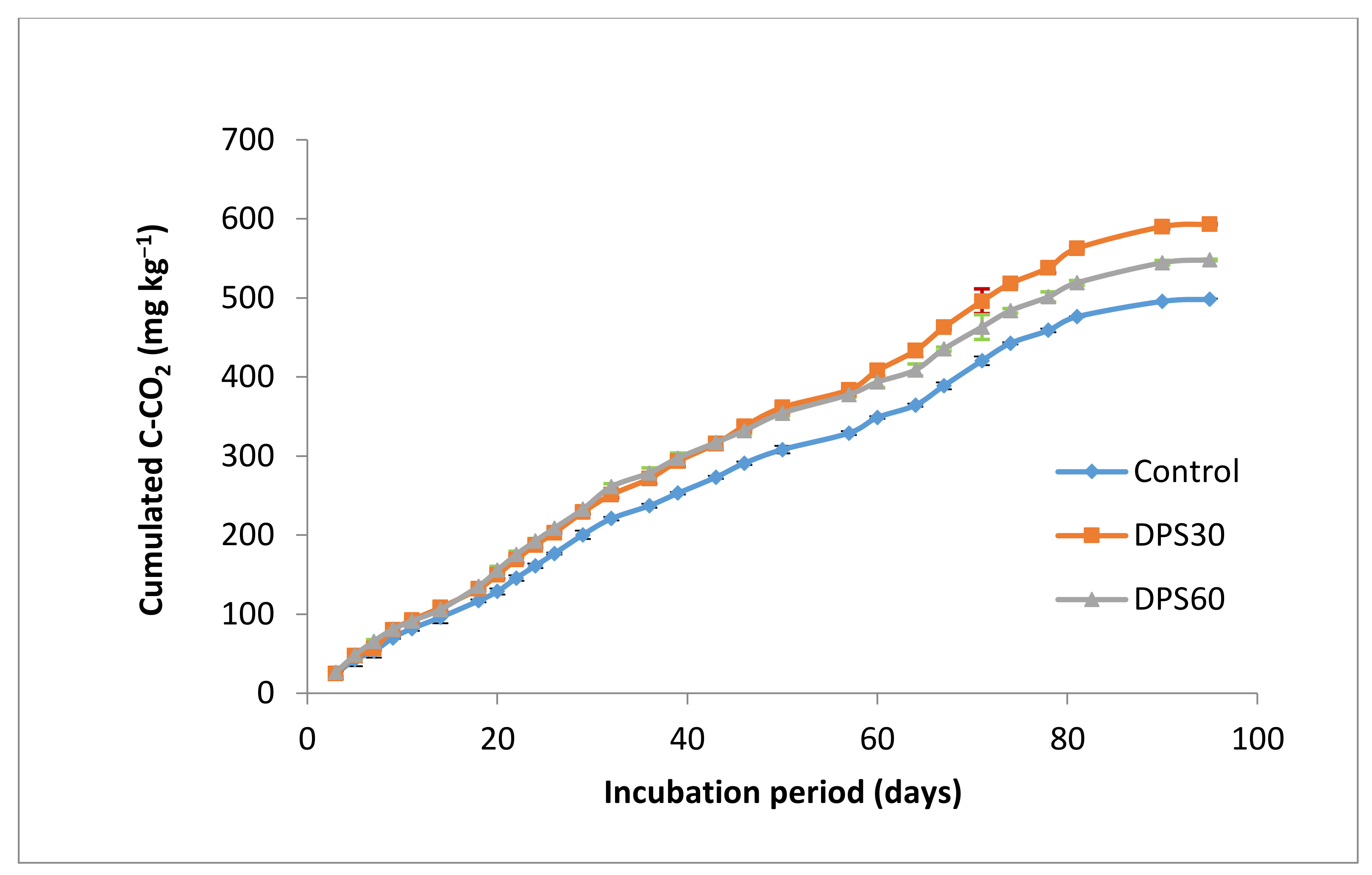
3.4.1. Effect on Mineralization Parameters
3.4.2. Effect on Microbial Biomass
3.5. Soil Trace Element Content
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Verdi, L.; Napoli, M.; Santoni, M.; Dalla Marta, A.; Ceccherini, M.T. So il carbon dioxide emission flux from organic and conventional farming in a long term experiment in Tuscany. In Proceedings of the 2019 IEEE International Workshop on Metrology for Agriculture and Forestry (MetroAgriFor), Portici, Italy, 24–26 October 2019; pp. 85–89. [Google Scholar]
- Masunga, R.H.; Uzokwe, V.N.; Mlay, P.D.; Odeh, I.; Singh, A.; Buchan, D.; De Neve, S. Nitrogen mineralization dynamics of different valuable organic amendments commonly used in agriculture. Appl. Soil Ecol. 2016, 101, 185–193. [Google Scholar] [CrossRef]
- Mohamed, E.-M.; El-Naggar, A.H.; Usman, A.R.; Al-Wabel, M. Dynamics of CO2 emission and biochemical properties of a sandy calcareous soil amended with Conocarpus waste and biochar. Pedosphere 2015, 25, 46–56. [Google Scholar]
- Chantigny, M. Émissions de Protoxyde d’azote (N2O) en Agriculture, Contribution des Amendements Organiques, des Fertilisants Minéraux et du Labour. Available online: https://www.agrireseau.net/agroenvironnement/documents/chantigny.pdf (accessed on 26 January 2020).
- Chantigny, M.H.; Pelster, D.E.; Perron, M.-H.; Rochette, P.; Angers, D.A.; Parent, L.-É.; Massé, D.; Ziadi, N. Nitrous oxide emissions from clayey soils amended with paper sludges and biosolids of separated pig slurry. J. Environ. Qual. 2013, 42, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Royer-Tardif, S.; Whalen, J.; Rivest, D. Can alkaline residuals from the pulp and paper industry neutralize acidity in forest soils without increasing greenhouse gas emissions? Sci. Total Environ. 2019, 663, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Bispo, A.; Andersen, L.; Angers, D.A.; Bernoux, M.; Brossard, M.; Cécillon, L.; Comans, R.N.; Harmsen, J.; Jonassen, K.; Lamé, F. Accounting for carbon stocks in soils and measuring GHGs emission fluxes from soils: Do we have the necessary standards? Front. Environ. Sci. 2017, 5, 41. [Google Scholar] [CrossRef]
- Nunes, J.R.; Cabral, F.; López-Piñeiro, A. Short-term effects on soil properties and wheat production from secondary paper sludge application on two Mediterranean agricultural soils. Bioresour. Technol. 2008, 99, 4935–4942. [Google Scholar] [CrossRef]
- Rashid, M.; Barry, D.; Goss, M. Paper mill biosolids application to agricultural lands: Benefits and environmental concerns with special reference to situation in Canada. Soil Environ. 2006, 25, 85–98. [Google Scholar]
- Gagnon, B.; Ziadi, N. Papermill biosolids and alkaline residuals affect crop yield and soil properties over nine years of continuous application. Can. J. Soil Sci. 2012, 92, 917–930. [Google Scholar] [CrossRef]
- Marouani, E.; Benzina, N.K.; Ziadi, N.; Bouslimi, B.; Abouda, A.; Koubaa, A. Deinking sludge compost stability and maturity assessment using Fourier transform infrared spectroscopy and thermal analysis. Waste Manag. Res. 2019, 37, 1043–1057. [Google Scholar] [CrossRef]
- Hébert, M. Guide sur le Recyclage des Matières Résiduelles Ferilisantes:critères de Référence et Normes Réglementaires; Ministry of Sustainable Development, Environment, and Fight Against Climate Change: Quebec, QC, Canada; Available online: http://www.environnement.gouv.qc.ca/matieres/mat_res/fertilisantes/critere/guide-mrf.pdf (accessed on 26 June 2020).
- Brouillette, F. Valorisation des boues de désencrage par combustion. Ph.D. Thesis, Université du Québec à Trois-Rivières, Trois-Rivières, QC, Canada, 1996. Available online: http://depot-e.uqtr.ca/id/eprint/4981/1/000623588.pdf (accessed on 20 January 2020).
- Smith, P.; Bustamante, M.; Ahammad, H.; Clark, H.; Dong, H.; Elsiddig, E.A.; Haberl, H.; Harper, R.; House, J.; Jafari, M. Agriculture, forestry and other land use (AFOLU). In Climate Change 2014: Mitigation of Climate Change. Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2014; pp. 811–922. [Google Scholar]
- Ziadi, N.; Gagnon, B.; Nyiraneza, J. Crop yield and soil fertility as affected by papermill biosolids and liming by-products. Can. J. Soil Sci. 2013, 93, 319–328. [Google Scholar] [CrossRef]
- Filiatrault, P.; Camiré, C.; Norrie, J.P.; Beauchamp, C.J. Effects of de-inking paper sludge on growth and nutritional status of alder and aspen. Resour. Conserv. Recycl. 2006, 48, 209–226. [Google Scholar] [CrossRef]
- Tian, G.; Chiu, C.Y.; Franzluebbers, A.J.; Oladeji, O.O.; Granato, T.C.; Cox, A.E. Biosolids amendment dramatically increases sequestration of crop residue-carbon in agricultural soils in western Illinois. Appl. Soil Ecol. 2015, 85, 86–93. [Google Scholar]
- Fierro, A.; Angers, D.A.; Beauchamp, C.J. Restoration of ecosystem function in an abandoned sandpit: Plant and soil responses to paper de-inking sludge. J. Appl. Ecol. 1999, 36, 244–253. [Google Scholar] [CrossRef]
- Fierro, A.; Angers, D.; Beauchamp, C. Decomposition of paper de-inking sludge in a sandpit minesoil during its revegetation. Soil Biol. Biochem. 2000, 32, 143–150. [Google Scholar] [CrossRef]
- Camberato, J.; Gagnon, B.; Angers, D.; Chantigny, M.; Pan, W. Pulp and paper mill by-products as soil amendments and plant nutrient sources. Can. J. Soil Sci. 2006, 86, 641–653. [Google Scholar] [CrossRef]
- Annabi, M.; Bahri, H.; Latiri, K. Statut organique et respiration microbienne des sols du nord de la Tunisie. Biotechnol. Agron. Soc. Environ. 2009, 13, 401. [Google Scholar]
- Barriga, S.; Méndez, A.; Cámara, J.; Guerrero, F.; Gascó, G. Agricultural valorisation of de-inking paper sludge as organic amendment in different soils. J. Therm. Anal. Calorim. 2010, 99, 981–986. [Google Scholar] [CrossRef]
- Faubert, P.; Barnabé, S.; Bouchard, S.; Côté, R.; Villeneuve, C. Pulp and paper mill sludge management practices: What are the challenges to assess the impacts on greenhouse gas emissions? Resour. Conserv. Recycl. 2016, 108, 107–133. [Google Scholar] [CrossRef]
- Bouajila, K.; Jeddi, F.B.; Sanaa, M. Effet de la décomposition des résidus de sulla (Hedysarum coronarium L.) sur l’évolution des caractéristiques chimiques de deux types de sol en climat aride Tunisien (Decomposition’s Effect of sulla residues (Hedysarum coronarium L.) on the chemical evolution characteristics of two soil’s types under Tunisian arid climate). J. Mater. Environ. Sci. 2014, 5, 723–730. [Google Scholar]
- Waksman, S.A.; Hutchings, I.J. Associative and antagonistic effects of microorganisms: III. Associative and antagonistic relationships in the decomposition of plant residues. Soil Sci. 1937, 43, 77–92. [Google Scholar] [CrossRef]
- Legrain, X.; Berding, F.; Dondeyne, S.; Schad, P.; Chapelle, J. Base de référence mondiale pour les ressources en sols 2014. Système international de classification des sols pour nommer les sols et élaborer des légendes de cartes pédologiques. Available online: http://www.fao.org/3/i3794fr/I3794FR.pdf (accessed on 26 June 2020).
- Richards, J.E. Chemical characterization of plant tissue. Soil Sampl. Methods Anal. 1993, 15, 115–139. [Google Scholar]
- Parkinson, J.; Allen, S. A wet oxidation procedure suitable for the determination of nitrogen and mineral nutrients in biological material. Commun. Soil Sci. Plant Anal. 1975, 6, 1–11. [Google Scholar] [CrossRef]
- Barnhisel, R.; Bertsch, P.M. Aluminum. In Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties; American Society of Agronomy, Inc.: Madison, WI, USA, 1983; Available online: https://doi.org/10.2134/agronmonogr9.2.2ed.c16 (accessed on 20 June 2020).
- Pauwels, J.; Van Ranst, E.; Verloo, M.; Mvondo Ze, A. Manuel de Laboratoire de Pédologie-méthodes d’analyses de sols et de plantes; equipment et gestion des stocks de verrerie et de produits chimiques. Available online: http://hdl.handle.net/1854/LU-223183. (accessed on 20 June 2020).
- Du, C.; Zhou, J. Application of infrared photoacoustic spectroscopy in soil analysis. Appl. Spectrosc. Rev. 2011, 46, 405–422. [Google Scholar] [CrossRef]
- Olsen, S.R. Estimation of Available Phosphorus in Soils by Extraction with Sodium Bicarbonate; United States Department Of Agriculture: Washington, DC, USA, 1954. [Google Scholar]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Hénin, S. Cours de physique du sol: Texture-structure-aération. Available online: https://www.documentation.ird.fr/hor/fdi:01944. (accessed on 20 June 2020).
- Vance, E.; Brookes, P.; Jenkinson, D. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Helmi, H.; Hechmi, S.; Khelil, M.N.; Zoghlami, I.R.; Benzarti, S.; Mokni-Tlili, S.; Hassen, A.; Jedidi, N. Repetitive land application of urban sewage sludge: Effect of amendment rates and soil texture on fertility and degradation parameters. Catena 2019, 172, 11–20. [Google Scholar]
- Jenkinson, D.; Powlson, D. The effects of biocidal treatments on metabolism in soil—I. Fumigation with chloroform. Soil Biol. Biochem. 1976, 8, 167–177. [Google Scholar] [CrossRef]
- Shi, W.; Yao, H.; Bowman, D. Soil microbial biomass, activity and nitrogen transformations in a turfgrass chronosequence. Soil Biol. Biochem. 2006, 38, 311–319. [Google Scholar] [CrossRef]
- Zhou, H.; Zhang, D.; Wang, P.; Liu, X.; Cheng, K.; Li, L.; Zheng, J.; Zhang, X.; Zheng, J.; Crowley, D. Changes in microbial biomass and the metabolic quotient with biochar addition to agricultural soils: A Meta-analysis. Agric. Ecosyst. Environ. 2017, 239, 80–89. [Google Scholar] [CrossRef]
- SAS Institute Inc (Cary, NC, USA). 2008. Available online: https://support.sas.com/resources/papers/proceedings/pdfs/sgf2008/190-2008.pdf. (accessed on 20 June 2020).
- Jedidi, N. Minéralisation et Humification des Amendements Organiques dans un sol Limono-argileux Tunisien. Ph.D. Thesis, Universiteit Gent, Gent, Belgium, 1998. [Google Scholar]
- Béjaoui, I. Pour une Valorisation Agronomique du Phosphogypse en Tunisie.I mpact sur le sol et la Plante et Évaluation du Transfert et de la Biodisponibilité du Cadmium et du Plomb. Ph.D. Thesis, Institut National Agronomique De Tunisie, Tunis, Tunisia, 2016; p. 207. [Google Scholar]
- Pandey, K.; Pitman, A. FTIR studies of the changes in wood chemistry following decay by brown-rot and white-rot fungi. Int. Biodeterior. Biodegrad. 2003, 52, 151–160. [Google Scholar] [CrossRef]
- Simard, R.R.; Baziramakenga, R.; Yelle, S.; Coulombe, J. Effects of de-inking paper sludges on soil properties and crop yields. Can. J. Soil Sci. 1998, 78, 689–697. [Google Scholar] [CrossRef]
- Cabral, F.; Vasconcelos, E. Agricultural use of combined primary/secondary pulpmill sludge. Agrochimica 1993, 37, 409–417. [Google Scholar]
- Tisdale, S.L.; Nelson, W.L.; Beaton, J.D. Soil Fertility and Fertilizers; Collier Macmillan Publishers: New York City, NY, USA, 1985. [Google Scholar]
- Xu, J.; Tang, C.; Chen, Z.L. The role of plant residues in pH change of acid soils differing in initial pH. Soil Biol. Biochem. 2006, 38, 709–719. [Google Scholar] [CrossRef]
- Haynes, R.J.; Naidu, R. Influence of lime, fertilizer and manure applications on soil organic matter content and soil physical conditions: A review. Nutr. Cycl. Agroecosyst. 1998, 51, 123–137. [Google Scholar] [CrossRef]
- Diehl, R.; Carpentier, C.-D. The structural chemistry of indium phosphorus chalcogenides. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1978, 34, 1097–1105. [Google Scholar] [CrossRef]
- Sumner, M.E. Sodic soils-New perspectives. Soil Res. 1993, 31, 683–750. [Google Scholar] [CrossRef]
- Trepanier, L.; Gallichand, J.; Caron, J.; Theriault, G. Environmental effects of deinking sludge application on soil and soilwater quality. Trans. ASAE 1998, 41, 1279. [Google Scholar] [CrossRef]
- Nemati, M.; Caron, J.; Gallichand, J. Using Paper De-inking Sludge to Maintain Soil Structural Form Field Measurements. Soil Sci. Soc. Am. J. 2000, 64, 275–285. [Google Scholar] [CrossRef]
- Chan, K.; Heenan, D. The influence of crop rotation on soil structure and soil physical properties under conventional tillage. Soil Tillage Res. 1996, 37, 113–125. [Google Scholar] [CrossRef]
- Chantigny, M.; Angers, D. Activités microbiologiques et qualité des sols: Quoi de neuf sous nos pieds. In Proceedings of the Colloque en Environnement:«des Outils d’intervention à notre Échelle», Centre de Référence en Agriculture et Agroalimentaire du Québec, Drummondville, QC, Canada, 23 February 2005. [Google Scholar]
- Lévesque, V.; Rochette, P.; Ziadi, N.; Dorais, M.; Antoun, H. Mitigation of CO2, CH4 and N2O from a fertigated horticultural growing medium amended with biochars and a compost. Appl. Soil Ecol. 2018, 126, 129–139. [Google Scholar] [CrossRef]
| DPS | FLc Soil | |
|---|---|---|
| Texture | - | Clayey silt |
| Sand (%) | - | 6.2 |
| Clay (%) | - | 51.6 |
| Loam (%) | - | 42.2 |
| pH-water | 7.9 | 7.8 |
| EC (µS cm−1) | 204.3 | 175.6 |
| OM (%) | 60.0 | 1.1 |
| TOC (%) | 31.5 | 0.6 |
| Total nitrogen (%) | 0.29 | 0.16 |
| C:N | 108.6 | 3.8 |
| Potassium (mg kg−1) | 400.9 | 1.4 |
| Calcium (g kg−1) | 270.0 | 5.11 |
| Magnesium (g kg−1) | 4.5 | - |
| Total phosphorus (mg kg−1) | 350.2 | - |
| Available phosphorus (mg kg−1) | 56.8 | 18.7 |
| C:P | 900.0 | - |
| Ca:Mg | 60.1 | |
| θcc | - | 26.1 |
| Total CaCO3 | - | 25.5 |
| Treatments | pH | EC (µS cm−1) | OM (%) | Total N (g kg−1) | P (mg kg−1) | K+ (g kg−1) | Ca2+ (g kg−1) | Na+ (mg kg−1) |
|---|---|---|---|---|---|---|---|---|
| Control | 7.8 bc (0.1) | 204.3 d (7.9) | 1.1 c (0.2) | 1.6 ab (0.1) | 40.0 d (2.7) | 1.3 b (0.01) | 5.1 d (0.1) | 620.0 a (20) |
| DPS30 | 7.9 ab (0.1) | 209.5 d (2.7) | 1.5 b (0.2) | 1.4 b (0.1) | 51.7 bc (3.4) | 1.3 b (0.01) | 5.3 b (0.02) | 633.3 a (30.6) |
| DPS60 | 8.0 a (0.1) | 217.3 d (3.6) | 1.9 a (0.2) | 1.2 c (0.1) | 63.1 a (10.3) | 1.3 b (0.01) | 5.4 a (0.03) | 640.0 a (34.6) |
| ControlF | 7.7 c (0.2) | 247.8 b (4.3) | 1.1 c (0.1) | 1.7 a (0.1) | 40.7 cd (6.5) | 1.3 b (0.04) | 5.2 d (0.03) | 213.3 c (11.6) |
| DPS30F | 7.9 ab (0.0) | 260.3 a (29.7) | 1.3 bc (0.1) | 1.5 ab (0.0) | 49.9 bc (3.3) | 1.4 a (0.01) | 5.3 b (0.06) | 293.3 b (3.1) |
| DPS60F | 7.9 ab (0.0) | 231.7 c (4.0) | 1.5 b (0.1) | 1.5 ab (0.0) | 55.1 ab (1.3) | 1.4 a (0.01) | 5.2 c (0.02) | 320.0 b (34.6) |
| Effect | p-Value | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DF | pH | CE | OM | Total | Na | Ca | K | Available | Permeability | SS 3 | ||
| N | C | P | ||||||||||
| Treatments 1 | 2 | 0.09 | 0.0002 | 0.007 | 0.04 | 0.007 | 0.039 | 0.002 | 0.01 | 0.009 | <0.0001 | 0.001 |
| Fertilization 2 | 1 | 0.03 | <0.0001 | 0.029 | 0.03 | 0.029 | <0.0001 | 0.039 | 0.32 | 0.40 | 0.12 | 0.87 |
| Interaction T × F | 2 | 0.33 | <0.0001 | 0.11 | 0.21 | 0.11 | 0.08 | 0.009 | 0.29 | 0.24 | 0.04 | 0.52 |
| Treatment (1) | C0 (2) (mg C kg−1) | k (3) (day−1) | C0 * k | R | TMR (4) (%) |
|---|---|---|---|---|---|
| Control | 1234.4 | 0.00578 | 7.1 | 0.99 | 4.3 |
| PS30 | 1774.1 | 0.00454 | 8.1 | 0.99 | 4.6 |
| DPS60 | 985.4 | 0.00890 | 8.7 | 0.99 | 4.1 |
| Treatment 1 | qCO2 2 | MBC (mg kg−1) | MBN (mg kg−1) | MBC: MBN |
|---|---|---|---|---|
| Control | 3.3 a | 152.4 b | 16.5 b | 10.2 b |
| DPS30 | 3.5 a | 167.6 b | 19.1 b | 8.9 b |
| DPS60 | 0.6 b | 914.3 a | 30.2 a | 19.4 a |
| Parameters | Control | DPS30 | DPS60 | Regulations CCME and EN13650 |
|---|---|---|---|---|
| Cd | 2.2 b * | 2.2 b | 2.4 a | 3 |
| Co | 10.2 a | 10.1 a | 9.9 b | 40 |
| Cu | 51.9 a | 49.7 b | 49.4 b | 63 |
| Fe | 16,646.4 a | 16,584.7 a | 16,317.3 b | |
| Mn | 293.2 a | 287.8 a | 284.9 b | |
| Mo | 85.7 a | 85.0 a | 85.2 a | |
| Pb | 27.1 a | 25.6 ab | 26.1 ab | 180 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marouani, E.; Kolsi Benzina, N.; Ziadi, N.; Bouslimi, B.; Abida, K.; Tlijani, H.; Koubaa, A. CO2 Emission and Change in the Fertility Parameters of a Calcareous Soil Following Annual Applications of Deinking Paper Sludge (The Case of Tunisia). Agronomy 2020, 10, 956. https://doi.org/10.3390/agronomy10070956
Marouani E, Kolsi Benzina N, Ziadi N, Bouslimi B, Abida K, Tlijani H, Koubaa A. CO2 Emission and Change in the Fertility Parameters of a Calcareous Soil Following Annual Applications of Deinking Paper Sludge (The Case of Tunisia). Agronomy. 2020; 10(7):956. https://doi.org/10.3390/agronomy10070956
Chicago/Turabian StyleMarouani, Emna, Naïma Kolsi Benzina, Noura Ziadi, Besma Bouslimi, Khouloud Abida, Hanen Tlijani, and Ahmed Koubaa. 2020. "CO2 Emission and Change in the Fertility Parameters of a Calcareous Soil Following Annual Applications of Deinking Paper Sludge (The Case of Tunisia)" Agronomy 10, no. 7: 956. https://doi.org/10.3390/agronomy10070956
APA StyleMarouani, E., Kolsi Benzina, N., Ziadi, N., Bouslimi, B., Abida, K., Tlijani, H., & Koubaa, A. (2020). CO2 Emission and Change in the Fertility Parameters of a Calcareous Soil Following Annual Applications of Deinking Paper Sludge (The Case of Tunisia). Agronomy, 10(7), 956. https://doi.org/10.3390/agronomy10070956







