What Has Been Thought and Taught on the Lunar Influence on Plants in Agriculture? Perspective from Physics and Biology
Abstract
1. Introduction
1.1. The Moon
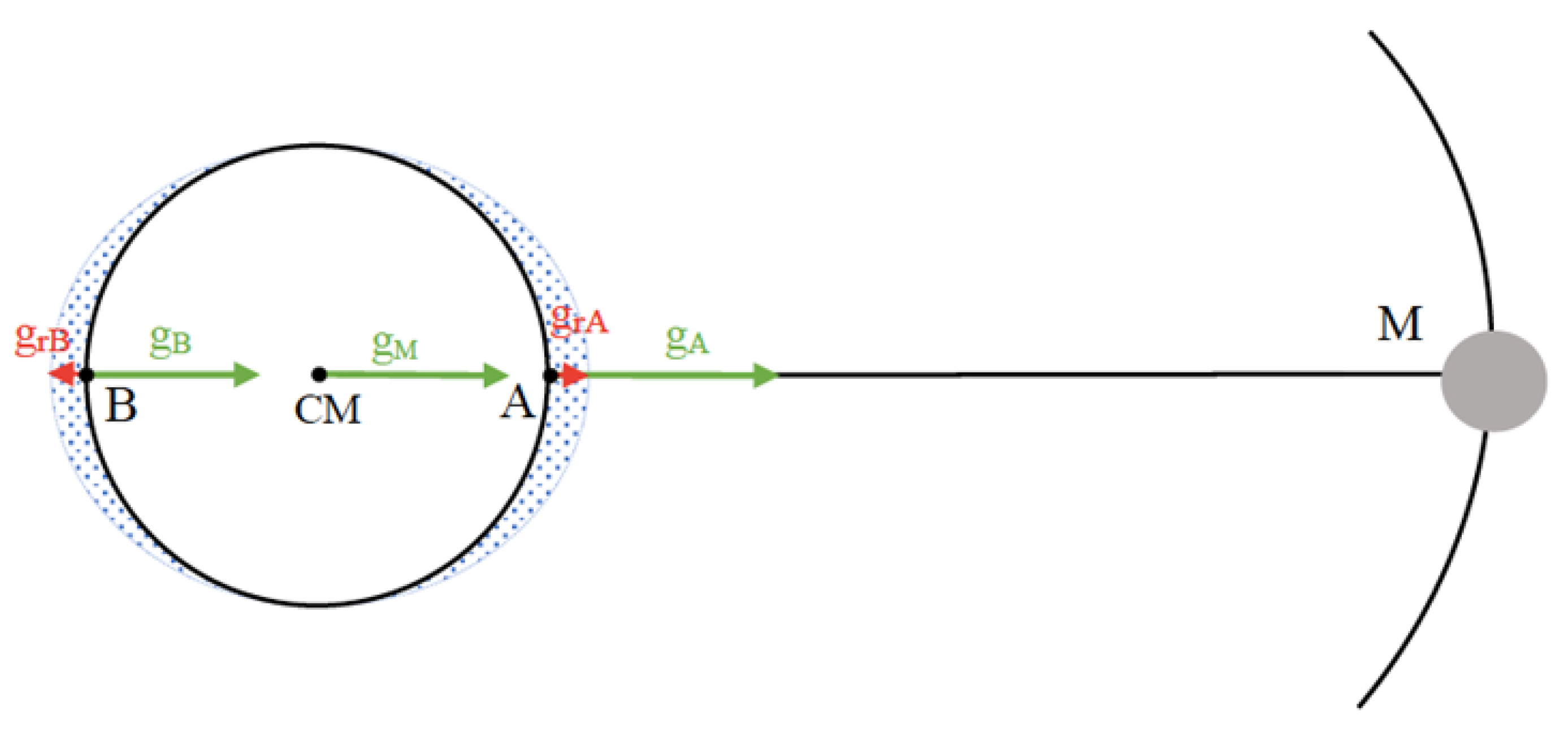
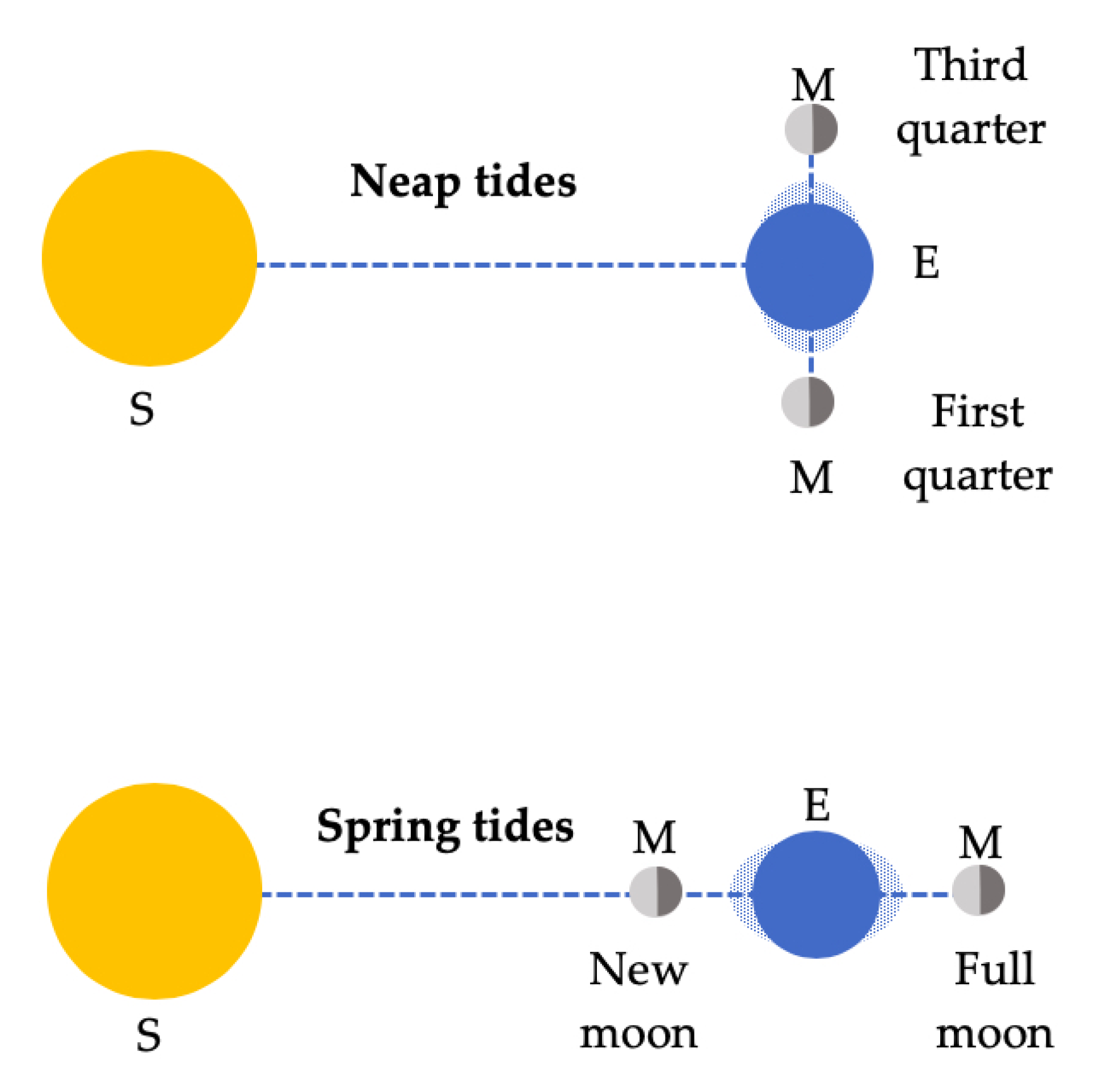
1.1.1. The Gravitational Pull
1.1.2. Illuminance
1.2. Factors Influencing Plant Growth and Development
2. What Has Been Thought on the Influence of the Moon on Plants
2.1. Brief Historical Overview
“Therefore, in certain positions of the Moon, the water from the oceans rises to reach a maximum height, and then goes down to a minimum level, maintaining this oscillation regularly and successively. It has also been checked that this phenomenon makes itself felt in plant sap”.(Translated from Restrepo [29]).
2.2. Agricultural Astronomy Manuals and Websites
“There has been a certain amount of interest in planting according to the phase of the Moon. The basic premise being that ‘above ground crops’ should be planted in the light of the Moon, i.e., on the days between the new moon and the full moon. ‘Below ground crops’ must be planted in the dark of the Moon, that is between the full moon and the next new moon. Refinements on this require that leaf crops are planted at the new moon and fruit crops or flowers planted at the full moon”.
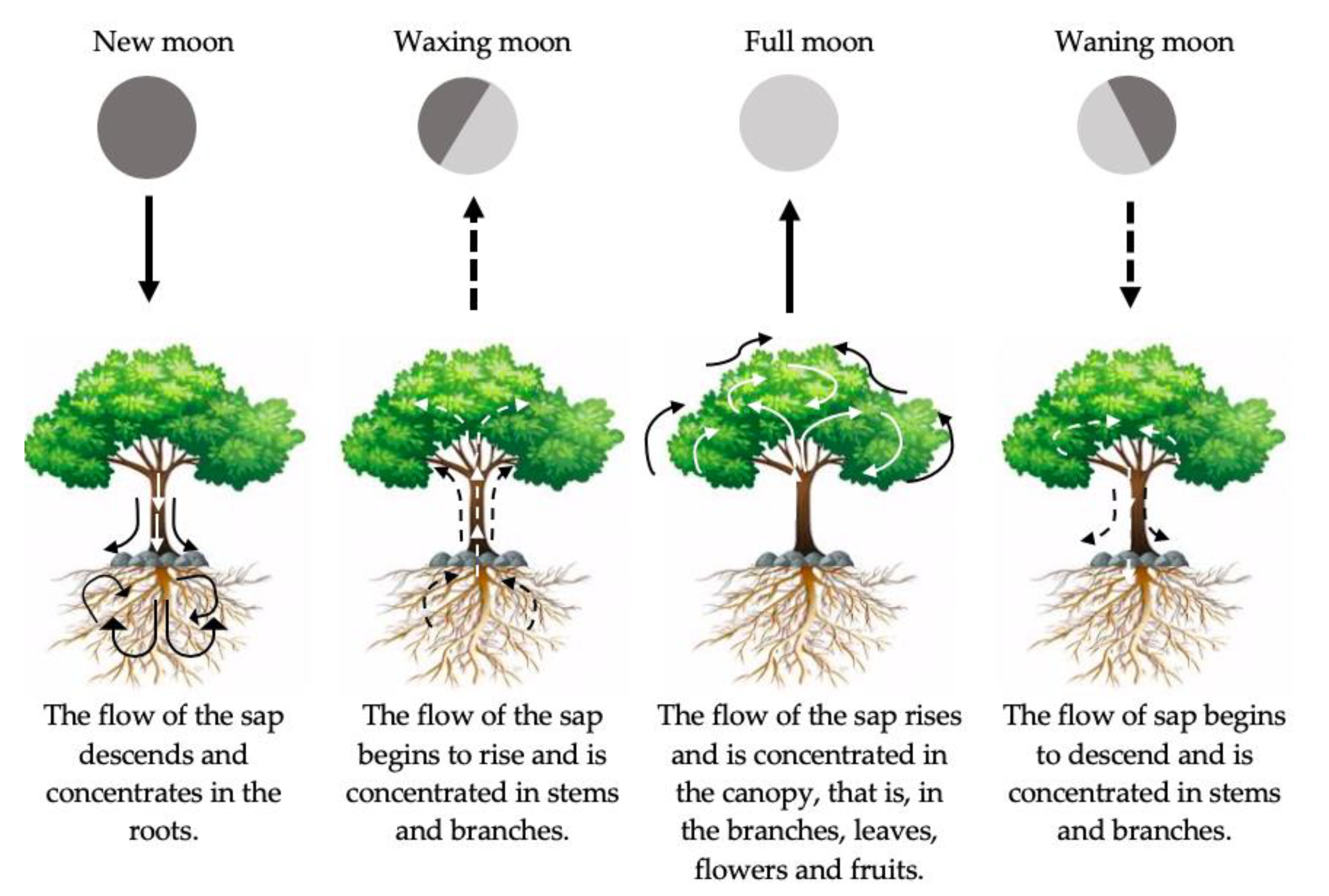
“The best rate of germination is achieved just before a full moon, when moonlight and the Moon’s gravitational pull are both at their maximum, grafting should be done on a waxing moon, because sap rises in plants during this period and this will help a graft to establish, pruning should be done on a waning moon, because the sap is now falling, and this will help cut surfaces to heal quickly and crops for storage should be harvested while the Moon is waning”.
2.3. Biodynamic Agriculture
3. What Has Been Taught on the Influence of the Moon on Plants? Analyses of Handbooks and Scientific Literature
3.1. What Handbooks Say from the Perspective of Physics and Biology
3.2. What Research Papers Say from the Perspective of Physics and Biology
4. Next Steps from the Perspective of Science Teaching
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Martínez, V.J.; Miralles, J.A.; Marco, E.; Galadí-Enríquez, D. Astronomía Fundamental; Universitat de València: Valencia, Spain, 2005. [Google Scholar]
- Morgan, E. The moon and life on earth. Earth Moon Planets 2001, 85–86, 279–290. [Google Scholar]
- Rackham, T. Moon in Focus; Pergamon: Oxford, UK, 1968. [Google Scholar]
- Bakulin, P.I.; Kononovich, E.V.; Moroz, V.I. Curso de Astronomía General; Mir: Moscow, Russia, 1987. [Google Scholar]
- Hewitt, P.G. Conceptual Physics, 9th ed.; Pearson Education: San Francisco, CA, USA, 2002. [Google Scholar]
- Neumann, D. Timing in Tidal, Semilunar, and Lunar Rhythms. In Annual, Lunar and Tidal Clocks: Patterns and Mechanisms of Nature’s Enigmatic Rhythms; Numata, H., Helm, B., Eds.; Springer: Tokyo, Japan, 2014; pp. 3–24. [Google Scholar]
- Adushkin, V.V.; Riabova, S.A.; Spivak, A.A. Lunar–solar tide effects in the Earth’s crust and atmosphere. Izv. Phys. Solid Earth 2017, 53, 565–580. [Google Scholar] [CrossRef]
- Myers, D.E. Gravitational effects of the period of high tides and the new moon on lunacy. Int. J. Emerg. Med. 1995, 13, 529–532. [Google Scholar] [CrossRef]
- RCA Corporation. Electro-Optics Handbook; RCA/Commercial Engineering: Harrison, NJ, USA, 1974. [Google Scholar]
- Schlyter, P. (1997–2017) Radiometry and photometry in astronomy. Available online: http://www.stjarnhimlen.se/comp/radfaq.html#13 (accessed on 29 April 2020).
- Hunt, R. Basic Growth Analysis: Plant Growth Analysis for Beginners; Unwin Hyman: London, UK, 2012. [Google Scholar]
- Arteca, R.N. Plant Growth Substances: Principles and Applications; Springer Science & Business Media: Dordrecht, The Netherlands, 2013. [Google Scholar]
- Evans, G.C. The Quantitative Analysis of Plant Growth; Blackwell Scientific Publications: Oxford, UK, 1972; Volume 1. [Google Scholar]
- Larcher, W. Physiological Plant Ecology: Ecophysiology and Stress Physiology of Functional Groups; Springer-Verlag Berlin Heidelberg: Berlin, Germany, 2003. [Google Scholar]
- Leopold, A.C. Plant Growth and Development; MCGraw-Hill Education: New York, NY, USA, 1964. [Google Scholar]
- Srivastava, L.M. Plant Growth and Development: Hormones and Environment; Elsevier: San Diego, CA, USA, 2002. [Google Scholar]
- Taiz, L.; Zeiger, E.; Møller, I.M.; Murphy, A. Plant Physiology and Development, 6th ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2015. [Google Scholar]
- Bulgari, R.; Franzoni, G.; Ferrante, A. Biostimulants Application in Horticultural Crops under Abiotic Stress Conditions. Agronomy 2019, 9, 306. [Google Scholar] [CrossRef]
- Drobek, M.; Frąc, M.; Cybulska, J. Plant Biostimulants: Importance of the Quality and Yield of Horticultural Crops and the Improvement of Plant Tolerance to Abiotic Stress—A Review. Agronomy 2019, 9, 335. [Google Scholar] [CrossRef]
- Strasburger, E.; Noll, F.; Schenck, H.; Schimper, A. Tratado de Botánica; Omega: Barcelona, Spain, 2004. [Google Scholar]
- Bhatla, S.C.; Lal, M.A. Plant Physiology, Development and Metabolism; Springer: Singapore, 2018. [Google Scholar]
- Brown, F.A., Jr.; Freeland, R.O.; Ralph, C.L. Persistent Rhythms of O2-Consumption in Potatoes, Carrots and the Seaweed, Fucus. Plant Physiol. 1955, 30, 280. [Google Scholar] [CrossRef] [PubMed]
- Ferris, T. Coming of Age in the Milky Way; Anchor Books: Morrow, NY, USA, 1988. [Google Scholar]
- Solbes, J.; Palomar, R. ¿Por qué resulta tan difícil la comprensión de la astronomía a los estudiantes? Didáctica Cienc. Exp. Soc. 2011, 25, 187–211. [Google Scholar]
- Böckler, C.G. Donde Enmudecen las Conciencias: Crepúsculo y Aurora en Guatemala; CIESAS: Mexico City, Mexico, 1986. [Google Scholar]
- Phillips, S. An Encyclopaedia of Plants in Myth, Legend, Magic and Lore; The Crowood Press Ltd.: Marlborough, UK, 2012. [Google Scholar]
- Navazio, J. The Organic Seed Grower: A farmer’s Guide to Vegetable Seed Production; Chelsea Green Publishing Co.: London, UK, 2012. [Google Scholar]
- Anglés Farrerons, J.M. Influencia de la Luna en la Agricultura y Otros Temas de Especial Interés Para el Campesino y Gentes de la Ciudad; Dilagro-Ediciones: Lleida, Spain, 1984. [Google Scholar]
- Restrepo, J. La Luna: El sol Nocturno en los Trópicos y su Influencia en la Agricultura; (No. 630.2233 R436.); Servicio de Información Mesoamericano sobre Agricultura Sostenible: Managua, Nicaragua, 2004. [Google Scholar]
- Tompkins, P.; Bird, C. The Secret Life of Plants; (No. QK50. T65I 1973.); Harper & Row: New York, NY, USA, 1973. [Google Scholar]
- Galston, A.W.; Slayman, C.L. The Not-So-Secret Life of Plants: In which the historical and experimental myths about emotional communication between animal and vegetable are put to rest. Am. Sci. 1979, 67, 337–344. [Google Scholar]
- Horowitz, K.A.; Lewis, D.C.; Gasteiger, E.L. “Plant primary perception”: Electrophysiological unresponsiveness to brine shrimp killing. Science 1975, 189, 478–480. [Google Scholar] [CrossRef][Green Version]
- Bussagli, M. Calendario Lunar de las Siembras y Labores Agrícolas (Pequeñas Joyas); Susaeta: Madrid, Spain, 2019. [Google Scholar]
- Calendario Zaragozano. Calendario Zaragozano. El Firmamento para toda España; Castillo y Ocsiero, M., Ed.; Zaragozano: Madrid, Spain, 2019. [Google Scholar]
- Geiger, P. (Ed.) Farmers’ Almanac for the Year 2020; Almanac Publishing Company: Lewiston, ME, USA, 2019. [Google Scholar]
- Gros, M. Lunario 2020: Calendario Lunar para el Huerto y el Jardín Ecológico y También para Mantener la Salud; Artús Porta Manresa: Manresa, Spain, 2019. [Google Scholar]
- Leendertz, L. The Almanac: A Seasonal Guide to 2020; Mitchell Beazley: London, UK, 2019. [Google Scholar]
- Trédoulat, T. Réussir son Potager Avec la Lune; Rustica éditions: Paris, France, 2020. [Google Scholar]
- Littlewood, M. A Guide to Gardening by the Moon; Gaby Bartai: Glasgow, UK, 2009. [Google Scholar]
- Descubre cómo las fases lunares pueden afectar a tus cultivos. Available online: https://www.todohusqvarna.com/blog/fases-lunares/ (accessed on 19 June 2020).
- Chalker-Scott, L. The science behind biodynamic preparations: A literature review. HortTechnology 2013, 23, 814–819. [Google Scholar] [CrossRef]
- Thun, M.; Thun, M.K. Calendario de Agricultura Biodinámica 2020; Editorial Rudolf Steiner: Madrid, Spain, 2019. [Google Scholar]
- Steiner, R. Agriculture (English translation). 1958. Available online: http://wn.rsarchive.org/Biodynamics/GA327/English/BDA1958/Ag1958_index.html (accessed on 27 April 2020).
- Brock, C.; Geier, U.; Greiner, R.; Olbrich-Majer, M.; Fritz, J. Research in biodynamic food and farming–a review. Open Agric. 2019, 4, 743–757. [Google Scholar] [CrossRef]
- Demeter Association, Inc. Available online: https://www.demeter-usa.org/about-demeter/biodynamic-certification-marks.asp (accessed on 27 April 2020).
- Kirchmann, H. Biological dynamic farming—An occult form of alternative agriculture? J. Agric. Env. Ethics 1994, 7, 173–187. [Google Scholar] [CrossRef]
- Steiner, R. A Modern Art of Education; Anthroposophic Press: Great Barrington, MA, USA, 2004. [Google Scholar]
- Beer, A.; Beer, P. Kepler, Four Hundred Years; Pergammon Press: Oxford, UK, 1975. [Google Scholar]
- Tipler, P.A. Physics for Scientist and Engineers, 3rd ed.; Worth Publishers: New York, NY, USA, 1992. [Google Scholar]
- Feynman, R.P.; Leighton, R.B.; Sands, M. The Feynman Lectures on Physics; Fondo Educativo Interamericano: Bogotá, Colombia, 1971; Volume 1. [Google Scholar]
- Holton, G.; Brush, S. Introduction to Concepts and Theories in Physical Science; Addison Wesley: Reading, MA, USA, 1976. [Google Scholar]
- Gettys, W.E.; Keller, F.J.; Skove, M.J. Physics Classical and Modern; McGraw-Hill: New York, NY, USA, 1989. [Google Scholar]
- Giancoli, D.C. Physics; Prentice-Hall: Englewood Cliffs, NJ, USA, 1985. [Google Scholar]
- Smith, F.G.; Thomson, J.H. Optics; Wiley: Hoboken, NJ, USA, 1989. [Google Scholar]
- Fosket, D.E. Plant Growth and Development: A Molecular Approach; Academic Press Inc.: San Diego, CA, USA, 1994. [Google Scholar]
- Raven, P.H.; Evert, R.F.; Eichhorn, S.E. Biology of Plants; W.H. Freeman and Company: New York, NY, USA, 2005. [Google Scholar]
- Thomas, B.; Vince-Prue, D. Photoperiodism in Plants; Academic Press, Inc.: San Diego, CA, USA, 1996. [Google Scholar]
- Salisbury, F.B. Plant adaptations to the light environment. In Plant Production in the North; Kaurin, A., Junttila, O., Nilsen, J., Eds.; Norwegian University Presss: Tromso, Norway, 1985; pp. 43–61. [Google Scholar]
- Bünning, E.; Moser, I. Interference of moonlight with the photoperiodic measurement of time by plants, and their adaptive reaction. Proc. Natl. Acad. Sci. USA 1969, 62, 1018–1022. [Google Scholar] [CrossRef] [PubMed]
- Kadman-Zahavi, A.; Peiper, D. Effects of moonlight on flower induction in Pharbitis nil, using a single dark period. Ann. Bot. 1987, 60, 621–623. [Google Scholar] [CrossRef]
- Beeson, C.F.C. The moon and plant growth. Nature 1946, 158, 572. [Google Scholar] [CrossRef] [PubMed]
- Brown, F.A., Jr.; Bennett, M.F.; Marguerite Webb, H. Persistent daily and tidal rhythms of O2-consumption in fiddler crabs. J. Cell Comp. Physiol. 1954, 44, 477–505. [Google Scholar] [CrossRef]
- Brown, F.A., Jr.; Webb, H.M.; Bennett, M.F.; Sandeen, M.I. Temperature-independence of the frequency of the endogenous tidal rhythm of Uca. Physiol. Zool. 1954, 27, 345–349. [Google Scholar] [CrossRef]
- Schad, W. Lunar influence on plants. Earth Moon Planets 2001, 85–86, 405–409. [Google Scholar]
- Covington, M.F.; Panda, S.; Liu, X.L.; Strayer, C.A.; Wagner, D.R.; Kay, S.A. ELF3 modulates resetting of the circadian clock in Arabidopsis. Plant Cell 2001, 13, 1305–1316. [Google Scholar] [CrossRef]
- Van Norman, J.M.; Breakfield, N.W.; Benfey, P.N. Intercellular communication during plant development. Plant Cell 2011, 23, 855–864. [Google Scholar] [CrossRef]
- Bitz, D.M.; Sargent, M.L. A failure to detect an influence of magnetic fields on the growth rate and circadian rhythm of Neurospora crassa. Plant Physiol. 1974, 53, 154–157. [Google Scholar] [CrossRef] [PubMed]
- Mironov, V.L.; Kondratev, A.Y.; Mironova, A.V. Growth of Sphagnum is strongly rhythmic: Contribution of the seasonal, circalunar and third components. Physiol. Plant 2020, 168, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Spiess, H. Chronobiological Investigations of Crops Grown under Biodynamic Management. I. Experiments with Seeding Dates to Ascertain the Effects of Lunar Rhythms on the Growth of Winter Rye (Secale cereale cv. Nomaro). Biol. Agric. Hortic. 1990, 7, 165–178. [Google Scholar] [CrossRef]
- Spiess, H. Chronobiological Investigations of Crops Grown under Biodynamic Management. II. Experiments with Seeding Dates to Ascertain the Effects of Lunar Rhythms on the Growth of Little Radish (Raphanus sativus, cv. Parat). Biol. Agric. Hortic. 1990, 7, 179–189. [Google Scholar] [CrossRef]
- Thun, M. Nine years observation of cosmic influences on annual plants. Star Furrow 1964, 22. [Google Scholar]
- Kollerstrom, N.; Staudenmaier, G. Evidence for lunar-sidereal rhythms in crop yield: A review. Biol. Agric. Hortic. 2001, 19, 247–259. [Google Scholar] [CrossRef]
- Chaffey, N.; Volkmann, D.; Baluška, F. The botanical multiverse of Peter Barlow. Comm. Integr. Biol. 2019, 12, 14–30. [Google Scholar] [CrossRef]
- Barlow, P.W.; Klingelé, E.; Klein, G.; Mikulecký Sen, M. Leaf movements of bean plants and lunar gravity. Plant Signal. Behav. 2008, 3, 1083–1090. [Google Scholar] [CrossRef]
- Barlow, P.W. Leaf movements and their relationship with the lunisolar gravitational force. Ann. Bot. 2015, 116, 149–187. [Google Scholar] [CrossRef]
- Fisahn, J.; Klingelé, E.; Barlow, P. Lunar gravity affects leaf movement of Arabidopsis thaliana in the International Space Station. Planta 2015, 241, 1509–1518. [Google Scholar] [CrossRef]
- Zajączkowska, U.; Barlow, P.W. The effect of lunisolar tidal acceleration on stem elongation growth, nutations and leaf movements in peppermint (Mentha × piperita L.). Plant Biol. 2017, 19, 630–642. [Google Scholar] [CrossRef] [PubMed]
- Barlow, P.W.; Mikulecký, M., Sr.; Střeštík, J. Tree-stem diameter fluctuates with the lunar tides and perhaps with geomagnetic activity. Protoplasma 2010, 247, 25–43. [Google Scholar] [CrossRef] [PubMed]
- Barlow, P.W.; Fisahn, J. Lunisolar tidal force and the growth of plant roots, and some other of its effects on plant movements. Ann. Bot. 2012, 110, 301–318. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barlow, P.W.; Fisahn, J.; Yazdanbakhsh, N.; Moraes, T.A.; Khabarova, O.V.; Gallep, C.M. Arabidopsis thaliana root elongation growth is sensitive to lunisolar tidal acceleration and may also be weakly correlated with geomagnetic variations. Ann. Bot. 2013, 111, 859–872. [Google Scholar] [CrossRef] [PubMed]
- Fisahn, J.; Yazdanbakhsh, N.; Klingele, E.; Barlow, P. Arabidopsis thaliana root growth kinetics and lunisolar tidal acceleration. New Phytol. 2012, 195, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Gallep, C.M.; Moraes, T.A.; Červinková, K.; Cifra, M.; Katsumata, M.; Barlow, P.W. Lunisolar tidal synchronism with biophoton emission during intercontinental wheat-seedling germination tests. Plant Signal. Behav. 2014, 9, e28671. [Google Scholar] [CrossRef]
- Gallep, C.M.; Barlow, P.W.; Burgos, R.C.; van Wijk, E.P.R. Simultaneous and intercontinental tests show synchronism between the local gravimetric tide and the ultra-weak photon emission in seedlings of different plant species. Protoplasma 2017, 254, 315–325. [Google Scholar] [CrossRef]
- Moraes, T.A.; Barlow, P.W.; Klingelé, E.; Gallep, C.M. Spontaneous ultra-weak light emissions from wheat seedlings are rhythmic and synchronized with the time profile of the local gravimetric tide. Naturwissenschaften 2012, 99, 465–472. [Google Scholar] [CrossRef]
- Fisahn, J.; Klingelé, E.; Barlow, P. Lunisolar tidal force and its relationship to chlorophyll fluorescence in Arabidopsis thaliana. Plant Signal. Behav. 2015, 10, e1057367. [Google Scholar] [CrossRef]
- Kronenberg, G.H.M.; Kendrick, R.E. The physiology of action. In Photomorphogenesis in Plants; Kendrick, R.E., Kronenberg, G.H.M., Eds.; Nijhoff Publ.: Dordrecht, The Netherlands, 1986; pp. 99–114. [Google Scholar]
- Salisbury, F.B. The Flowering Process; Pergamon Press, Inc.: New York, NY, USA, 1963. [Google Scholar]
- Kolisko, L. The Moon and the Growth of Plants; Bray-on-Thames, Anthroposophical Agricultural Foundation: London, UK, 1936. [Google Scholar]
- Raven, J.A.; Cockell, C.S. Influence on photosynthesis of starlight, moonlight, planetlight, and light pollution (reflections on photosynthetically active radiation in the universe). Astrobiology 2006, 6, 668–675. [Google Scholar] [CrossRef]
- Breitler, J.C.; Djerrab, D.; Leran, S.; Toniutti, L.; Guittin, C.; Severac, D.; Pratlong, M.; Dereeper, A.; Etienne, H.; Bertrand, B. Full moonlight-induced circadian clock entrainment in Coffea arabica. BMC Plant Biol. 2020, 20, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Rydin, C.; Bolinder, K. Moonlight pollination in the gymnosperm Ephedra (Gnetales). Biol. Lett. 2015, 11, 20140993. [Google Scholar] [CrossRef] [PubMed]
- Semmens, E.S. Effect of Moonlight on the Germination of Seeds. Nature 1923, 111, 49–50. [Google Scholar] [CrossRef]
- Semmens, E.S. Hydrolysis in Green Plants by Moonlight. Nature 1932, 130, 243. [Google Scholar] [CrossRef]
- Semmens, E.S. Chemical Effects of Moonlight. Nature 1947, 159, 613. [Google Scholar] [CrossRef]
- Macht, D.I. Concerning the influence of polarized light on the growth of seedlings. J. Gen. Physiol. 1926, 10, 41–52. [Google Scholar] [CrossRef]
- Cronin, T.W.; Marshall, J. Patterns and properties of polarized light in air and water. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2011, 366, 619–626. [Google Scholar] [CrossRef]
- Nowinszky, L.; Szabó, S.; Tóth, G.; Ekk, I.; Kiss, M. The effect of the moon phases and of the intensity of polarized moonlight on the light-trap catches. Zeitschrift Angewandte Entomologie 1979, 88, 337–353. [Google Scholar] [CrossRef]
- Kyba, C.C.; Ruhtz, T.; Fischer, J.; Hölker, F. Lunar skylight polarization signal polluted by urban lighting. J. Geophys. Res. 2011, 116, D24106. [Google Scholar] [CrossRef]
- Begall, S.; Malkemper, E.P.; Cerveny, J.; Nemec, P.; Burda, H. Magnetic alignment in mammals and other animals. Mammal. Biol. 2013, 78, 10–20. [Google Scholar] [CrossRef]
- Ritz, T.; Wiltschko, R.; Hore, P.J.; Rodgers, C.T.; Stapput, K.; Thalau, P.; Timmel, C.R.; Wiltschko, W. Magnetic compass of birds is based on a molecule with optimal directional sensitivity. Biophys. J. 2009, 96, 3451–3457. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Zhang, S.; Chen, P.; Liu, H.; Yin, H.; Li, H. Magnetotactic bacteria, magnetosomes and their application. Microbiol. Res. 2012, 167, 507–519. [Google Scholar] [CrossRef] [PubMed]
- Belyavskaya, N.A. Biological effects due to weak magnetic field on plants. Adv. Space Res. 2004, 34, 1566–1574. [Google Scholar] [CrossRef]
- Galland, P.; Pazur, A. Magnetoreception in plants. J. Plant Res. 2005, 118, 371–389. [Google Scholar] [CrossRef]
- Maffei, M.E. Magnetic field effects on plant growth, development, and evolution. Front. Plant Sci. 2014, 5, 445. [Google Scholar] [CrossRef] [PubMed]
- Nyakane, N.E.; Markus, E.D.; Sedibe, M.M. The effects of magnetic fields on plants growth: A comprehensive review. Int. J. Food Eng. 2019, 5, 79–87. [Google Scholar] [CrossRef]
- Vian, A.; Davies, E.; Gendraud, M.; Bonnet, P. Plant responses to high frequency electromagnetic fields. Biomed. Res. Int. 2016, 2016, 1830262. [Google Scholar] [CrossRef]
- Mighani, S.; Wang, H.; Shuster, D.L.; Borlina, C.S.; Nichols, C.I.; Weiss, B.P. The end of the lunar dynamo. Sci. Adv. 2020, 6, eaax0883. [Google Scholar] [CrossRef]
- Tikoo, S.M.; Weiss, B.P.; Shuster, D.L.; Suavet, C.; Wang, H.; Grove, T.L. A two-billion-year history for the lunar dynamo. Sci. Adv. 2017, 3, e1700207. [Google Scholar] [CrossRef]
- Mitchell, D.L.; Halekas, J.S.; Lin, R.P.; Frey, S.; Hood, L.L.; Acuña, M.H.; Binder, A. Global mapping of lunar crustal magnetic fields by Lunar Prospector. Icarus 2008, 194, 401–409. [Google Scholar] [CrossRef]
- Bevington, M. Lunar biological effects and the magnetosphere. Pathophysiology 2015, 22, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Eve, R.A.; Dunn, D. Psychic powers, astrology and creationism in the classroom? Evidence of pseudoscientific beliefs among high school biology & life science teachers. Am. Biol. Teach. 1990, 52, 10–21. [Google Scholar] [CrossRef]
- Happs, J.C. Challenging pseudoscientific and paranormal beliefs held by some pre-service primary teachers. Res. Sci. Educ. 1991, 21, 171–177. [Google Scholar] [CrossRef]
- Kaplan, A.O. Research on the pseudo-scientific beliefs of preservice science teachers: A sample from astronomy-astrology. J. Balt. Sci. Educ. 2014, 13, 381–393. [Google Scholar]
- Solbes, J.; Palomar, R.; Dominguez-Sales, M.C. To what extent do pseudosciences affect teachers? A look at the mindset of science teachers in training. Mètode Sci. Stud. J. Ann. Rev. 2018, 8, 188–195. [Google Scholar] [CrossRef]
- Halpern, D. Teaching critical thinking for transfer across domains. Am. Psychol. 1998, 53, 449–455. [Google Scholar] [CrossRef]
- Torres, N.; Solbes, J. Contribuciones de una intervención didáctica usando cuestiones sociocientíficas para desarrollar el pensamiento crítico. Enseñanza Cienc. 2016, 34, 43–65. [Google Scholar] [CrossRef]
- Yager, R.E. Science and critical thinking. In Teaching Critical Thinking: Reports from Across the Curriculum; Clarke, J.H., Biddle, A.W., Eds.; Prentice Hall: Englewood Cliffs, NJ, USA, 1993. [Google Scholar]
- Bates, J.; Culpepper, W. Using Pseudoscience to Teach Science: Encouraging Skepticism of Paranormal Powers in the Classroom. J. Coll. Sci. Teach. 1991, 21, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.A. Reducing pseudoscientific and paranormal beliefs in university students through a course in science and critical thinking. Sci. Educ. 2018, 27, 183–210. [Google Scholar] [CrossRef]
- Chinn, C.A.; Malhotra, B.A. Epistemologically authentic inquiry in schools: A theoretical framework for evaluating inquiry tasks. Sci. Educ. 2002, 86, 175–218. [Google Scholar] [CrossRef]
- Lie, D.; Boker, J. Development and validation of the CAM Health Belief Questionnaire (CHBQ) and CAM use and attitudes amongst medical students. BMC Med Educ. 2004, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Pedaste, M.; Mäeots, M.; Siiman, L.A.; De Jong, T.; Van Riesen, S.A.; Kamp, E.T.; Constantinos, C.; Zacharias, M.; Tsourlidaki, E. Phases of inquiry-based learning: Definitions and the inquiry cycle. Educ. Res. Rev. 2015, 14, 47–61. [Google Scholar] [CrossRef]
- Pina, T.; Mayoral, O.; Solbes, J. ¿Influye la Luna en el crecimiento de las plantas? Indagación para favorecer el pensamiento crítico. In Propuestas de Educación Científica Basadas en la Indagación y Modelización en Contexto; Solbes, J., Jiménez, M.R., Pina, T., Eds.; Tirant lo Blanch: Valencia, Spain, 2019; pp. 121–143. [Google Scholar]
- Pina, T.; Mayoral, O.; Solbes, J. Do lunar phases influence the growth of plants? Scientific inquiry to encourage critical thinking in the classroom. In Proceedings of the ESERA conference, Bologna, Italy, 26–30 August 2019. [Google Scholar]
- Dixon, J.A.; Gibbon, D.P.; Gulliver, A. Farming Systems and Poverty: Improving Farmers’ Livelihoods in a Changing World; FAO: Rome, Italy; World Bank: Washington, DC, USA, 2001. [Google Scholar]
- Kuhnlein, H.V.; Erasmus, B.; Spigelski, D. Indigenous Peoples’ Food Systems: The Many Dimensions of Culture, Diversity and Environment for Nutrition and Health; FAO: Rome, Italy, 2009. [Google Scholar]
- Kuhnlein, H.V. Biodiversity and sustainability of indigenous peoples’ foods and diets. Sustainable diets and biodiversity. In Sustainable Diets and Biodiversity: Directions and Solutions for Policy, Research and Action. Proceedings of the International Scientific Symposium, Biodiversity and Sustainable Diets United Against Hunger, Rome, Italy, 3–5 November 2010; Burlingame, B., Dernini, S., Eds.; FAO Headquarters: Rome, Italy, 2012. [Google Scholar]
- Sinclair, F.; Wezel, A.; Mbow, C.; Chomba, S.; Robiglio, V.; Harrison, R. The Contribution of Agroecological Approaches to Realizing Climate-Resilient Agriculture; GCA: Rotterdam, The Netherlands, 2019. [Google Scholar]
- Lambers, H.; Chapin, F.S., III; Pons, T.L. Plant Physiological Ecology; Springer Science + Business Media: Berlin, Germany; LLC: New York, NY, USA, 2008. [Google Scholar]
| Illuminance (lx) | Description |
|---|---|
| 0.001 | Clear night sky, new moon |
| 0.01 | Clear night sky, crescent or waning moon |
| 0.25 | Full moon on a cloudless night |
| 600 | Sunrise or sunset on a cloudless day |
| 32,000 | Sunlight on an average day (minimum) |
| 100,000 | Sunlight on an average day (maximum) |
| New Moon | Waxing Moon | Full Moon | Waning Moon |
|---|---|---|---|
| Crop covering with soil | Prune diseased or fruit trees | Prune | Sow root vegetables |
| Fertilise | Cultivate sandy soils | Plant perennial species | Remove withered leaves |
| Remove weeds | Sow flowers, leafy vegetables | Transplant | Water flowering plants |
| Remove withered leaves | Grafting | Vegetative propagation | Fertilise |
| Sow grass | Avoid watering flowering plants | - | Plant longleaf trees |
| Practice or Product | Organic | Biodynamic |
|---|---|---|
| Crop rotation | x | x |
| Polyculture or intercropping | x | x |
| Cover cropping | x | x |
| Low- or no-till | x | x |
| Green manures | x | x |
| Biological, cultural, mechanical and physical means of pest control | x | x |
| Biodynamic preparations that involve alchemy and homeopathy | x | |
| Lunar and astrological calendars for planting, managing and harvesting | x | |
| Stones used for channelling cosmic energy and radiant fields | x | |
| Burning of pests and weeds (pest ashing) | x | |
| Sensitive testing (including biocrystallization or morphochromatography, among others) | x |
| Book | Issues Regarding the Moon | |||
|---|---|---|---|---|
| Gravity | Tides | Capillarity | Luminosity | |
| Feynman [50] | Law of Gravitation | Qualitative explanation of tides | No | Illuminance I = S/r2 |
| Gettys et al. [52] | Point where gE = gM | Tides in the Bay of Fundy (e.g., resonance) | No | No |
| Giancoli [53] | Law of Gravitation | Tidal energy | Jurin’s Law and negative pressure | No |
| Hewitt [5] | Moon radius, distance Earth–Moon. Law of Gravitation | Compares Sun and Moon tides by distance. It makes approximations to introduce R/r3 Distinguishes between spring and neap tides. Applied to people. Tides in the ionosphere | Qualitative capillarity from surface tension | No |
| Holton and Brush [51] | Law of Gravitation | Qualitative explanation of spring and neap tides | No | No |
| Tipler [49] | Calculation of the gE on the Moon | No | Jurin’s Law | No |
| Handbook | Phase/Process | Endogenous Factors | Exogenous or Environmental Factors | Moon Mention | |
|---|---|---|---|---|---|
| Biotic | Abiotic | ||||
| Arteca [12] | Vital cycle | Endogenous growth substances | No | ||
| Seeds | Water, temp., aeration and light | No | |||
| Flowering | Photoperiod and vernalization | No | |||
| Abscission | Temperature, oxygen and nutrients | No | |||
| Evans [13] | Growth | Environment and ecosystem | No | ||
| Fosket [55] | Development | Genetics | Light versus darkness | No | |
| Embryogenesis, germination and development | No | ||||
| Apical meristems and development | No | ||||
| Plant development | Plant-microbe and symbiotic interactions | No | |||
| Vital cycle | Phototropism and gravitropism | No | |||
| Raven et al. [56] | Biological rhythms | Circadian and biological clocks | Track of daylength by length of darkness | No | |
| Flowering | Daylength as determinant of flowering time | No | |||
| Photoautotrophy | Sunlight | No | |||
| Strasburguer [18] | Growth and differentiation | Phytohormones | Temperature, light, gravity, hydromorphosis | No | |
| Biological rhythms | Circadian rhythms | Photoperiod | No | ||
| Movement | Phototaxis, phototropisms, photonasties | No | |||
| Daylength perception | Circadian rhythms, phytochromes | Light quality, phase setting, interaction of light | No | ||
| Thomas and Vince-Prue [56] | Flower timing | Light quantity, moonlight | Yes | ||
| Flower development | Photoperiodism | No | |||
| Bud dormancy | No | ||||
| Storage and propagation | No | ||||
| Germination | No | ||||
| Stem elongation | No | ||||
| Leaf growth | No | ||||
| Process | Mode of Action 1 | Spectral Range 2 | Fluctuation 3 |
|---|---|---|---|
| Seed germination and bud break | I | R/FR, B | P |
| Stem elongation | Q, F | R/FR | P |
| Stem orientation | Q, F | B | |
| Leaf orientation | Q | R/FR | C |
| Flowering process | I | R/FR | C |
| Development and filling of storage organs | I | R/FR | P |
| Dormancy | I | R/FR | P |
| Enzyme synthesis | I | R/FR | |
| Enzyme activation | I | R/FR | |
| Membrane potentials | I | R/FR |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mayoral, O.; Solbes, J.; Cantó, J.; Pina, T. What Has Been Thought and Taught on the Lunar Influence on Plants in Agriculture? Perspective from Physics and Biology. Agronomy 2020, 10, 955. https://doi.org/10.3390/agronomy10070955
Mayoral O, Solbes J, Cantó J, Pina T. What Has Been Thought and Taught on the Lunar Influence on Plants in Agriculture? Perspective from Physics and Biology. Agronomy. 2020; 10(7):955. https://doi.org/10.3390/agronomy10070955
Chicago/Turabian StyleMayoral, Olga, Jordi Solbes, José Cantó, and Tatiana Pina. 2020. "What Has Been Thought and Taught on the Lunar Influence on Plants in Agriculture? Perspective from Physics and Biology" Agronomy 10, no. 7: 955. https://doi.org/10.3390/agronomy10070955
APA StyleMayoral, O., Solbes, J., Cantó, J., & Pina, T. (2020). What Has Been Thought and Taught on the Lunar Influence on Plants in Agriculture? Perspective from Physics and Biology. Agronomy, 10(7), 955. https://doi.org/10.3390/agronomy10070955








