Base Neutralizing Capacity of Agricultural Soils in a Quaternary Landscape of North-East Germany and Its Relationship to Best Management Practices in Lime Requirement Determination
Abstract
1. Introduction
- Soil–lime incubations:They involve the mixing of increasing rates of liming material with a fixed weight or volume of soil, equilibrating the soil–lime mixture in a moist state for several weeks or months, and developing a lime–response curve based on the resultant pH changes that are used to determine the lime requirement. This approach is way too time-consuming to be applicable in practical agriculture.
- Soil-base titrations:A soil suspension is titrated or equilibrated with a basic solution, such as Ca(OH)2 or NaOH, and the soil pH is measured after a certain equilibration time of the soil and the basic solution. LR can then be calculated by the conversion factors applied to the added concentration of basic solution. Several techniques have been used to measure soil buffer capacity [10,11,12,13,14,15,16]. As summarized by Robson [1] they differ mainly in: (1) the reagent added to the soil, (2) the conditions of equilibration, and (3) the method of measuring the pH, e.g., the method by Dunn [10] uses Ca(OH)2 and an equilibration time of 3 d. Dunn’s method was often regarded as a reference in comparison with other LR methods (e.g., [17,18]). In contrast, the base neutralizing capacity (BNC) by Meiwes et al. [11] is a discontinuous soil-base titration that only requires an incubation time of 18 h. Compared to the incubation method, titration is much faster but still rather laborious and expensive which prevents its frequent application in routine soil testing.
- Soil-buffer equilibrations:Soil–buffer equilibration is the most widespread approach in the USA to assess the soil’s lime requirement (e.g., Shoemaker-McLean-Pratt (SMP) buffer [19], Adams–Evans buffer [20] or Mehlich buffer [21]). A chemical buffer solution is added to a soil sample, allowing the soil and buffer to equilibrate for a certain period of time. After that, the pH of the soil–buffer mixture is measured. The observed decrease in buffer pH is a measure of the amount of soil acidity to be neutralized by liming in order to raise the soil pH to a target pH [13]. The chemical composition of the buffer solutions is sometimes adapted to the properties of the soils under consideration. Thus, various studies have emphasized the need for a regional calibration of buffer pH methods to verify the suitability of the buffer solution to the range of soil characteristics of a certain geographical region [9,22]. The SMP and Mehlich buffer solutions contain hazardous chemicals that must be handled carefully. In conclusion, the involvement of harmful substances and the lack of universality in the soil–buffer equilibration approach are considered critical [23].
- Estimates based on soil pH and additional soil properties:The estimation methods rely on the measurement of the pH and soil properties that are well correlated with potential acidity (e.g., soil texture, soil organic matter (SOM), soil type or cation-exchange capacity). Based on field trials, empirical relationships have been established between these soil properties and lime requirements. For example, in California (USA), the United Kingdom and in Germany, recommendations for liming are given by defining an optimum pH value needed for the soil and measuring the current soil pH as well as estimating the soil texture (e.g., by hand texturing) and the soil organic matter content (e.g., by loss on ignition). Lime requirement values are listed in look-up tables [24,25]. If data of soil texture and soil organic matter are available from earlier investigations, only the pH has to be measured. This can be done quickly and at low costs, which makes the estimation method very attractive for farmers. However, the estimates might be too rough.
- Characterize the BNC and pH buffer capacity (pHBC) of agricultural land of north-east Germany;
- Analyze the correlation between BNC parameters and BNC-based LR (LRBNC) with soil properties that are well known to affect soil acidity and pHBC (pH, soil texture and SOM); and
- Compare the LRBNC with the LR based on the VDLUFA standard procedure for lime fertilization in Germany (LRVDLUFA).
2. Materials and Methods
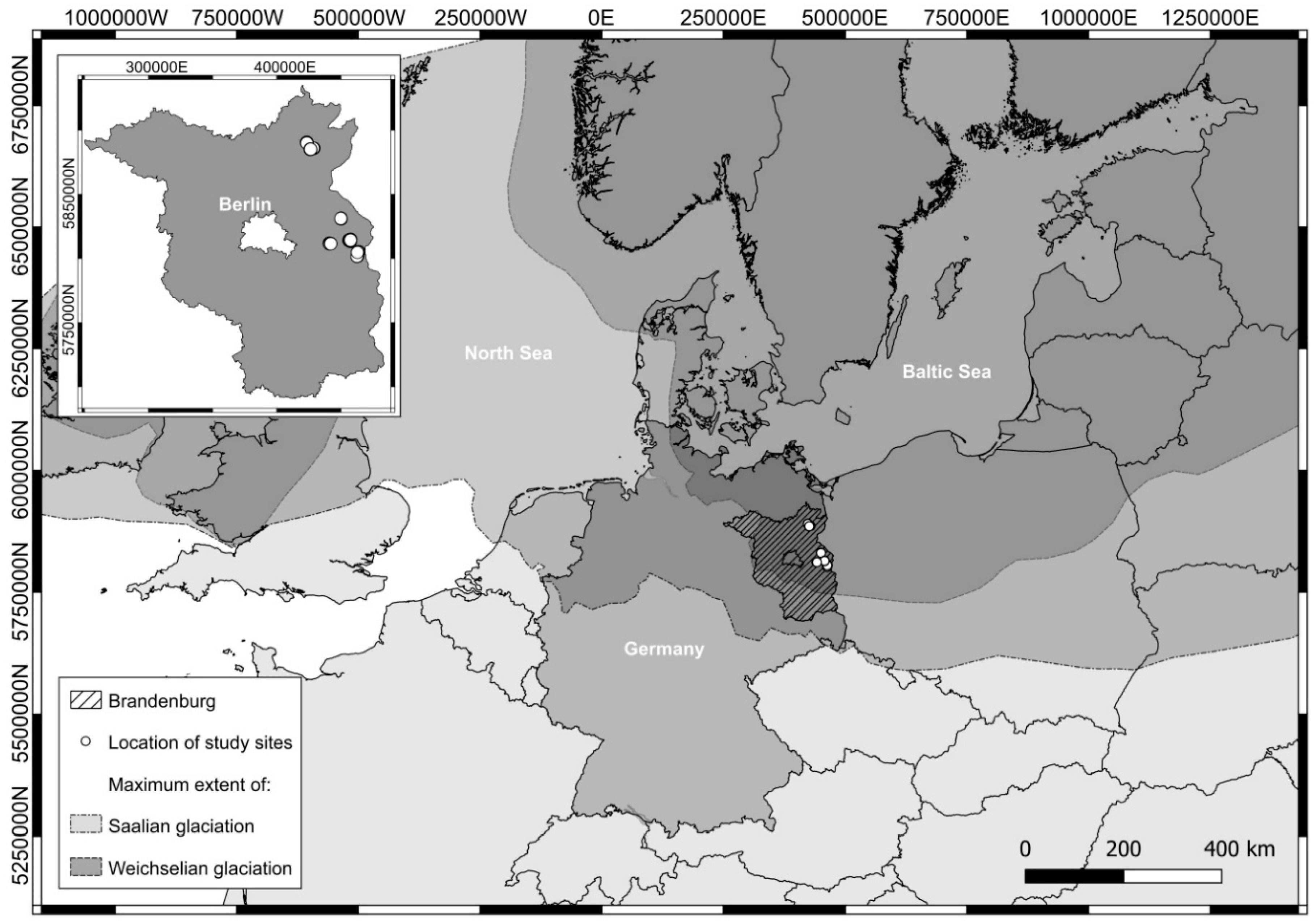
2.1. Site Description
2.2. Standard Laboratory Analyses of Studied Soils
- The soil pH value was measured in 10 g of soil and 25 mL of 0.01 M CaCl2 solution following DIN ISO 10390. The pH was measured with a glass electrode after a reaction time of 60 min;
- The particle distribution of the fraction <2 mm was determined according to the German standard in soil science (DIN ISO 11277) by wet sieving and sedimentation after removal of the organic matter with hydrogen peroxide (H2O2) and dispersal with 0.2 N sodium pyrophosphate (Na4P2O7);
- Soil organic carbon (SOC) was analyzed by elementary analysis using the dry combustion method (DIN ISO 10694) after removing inorganic carbon with hydrochloric acid. Finally, the amount of SOM was calculated following Equation (1) [36]:
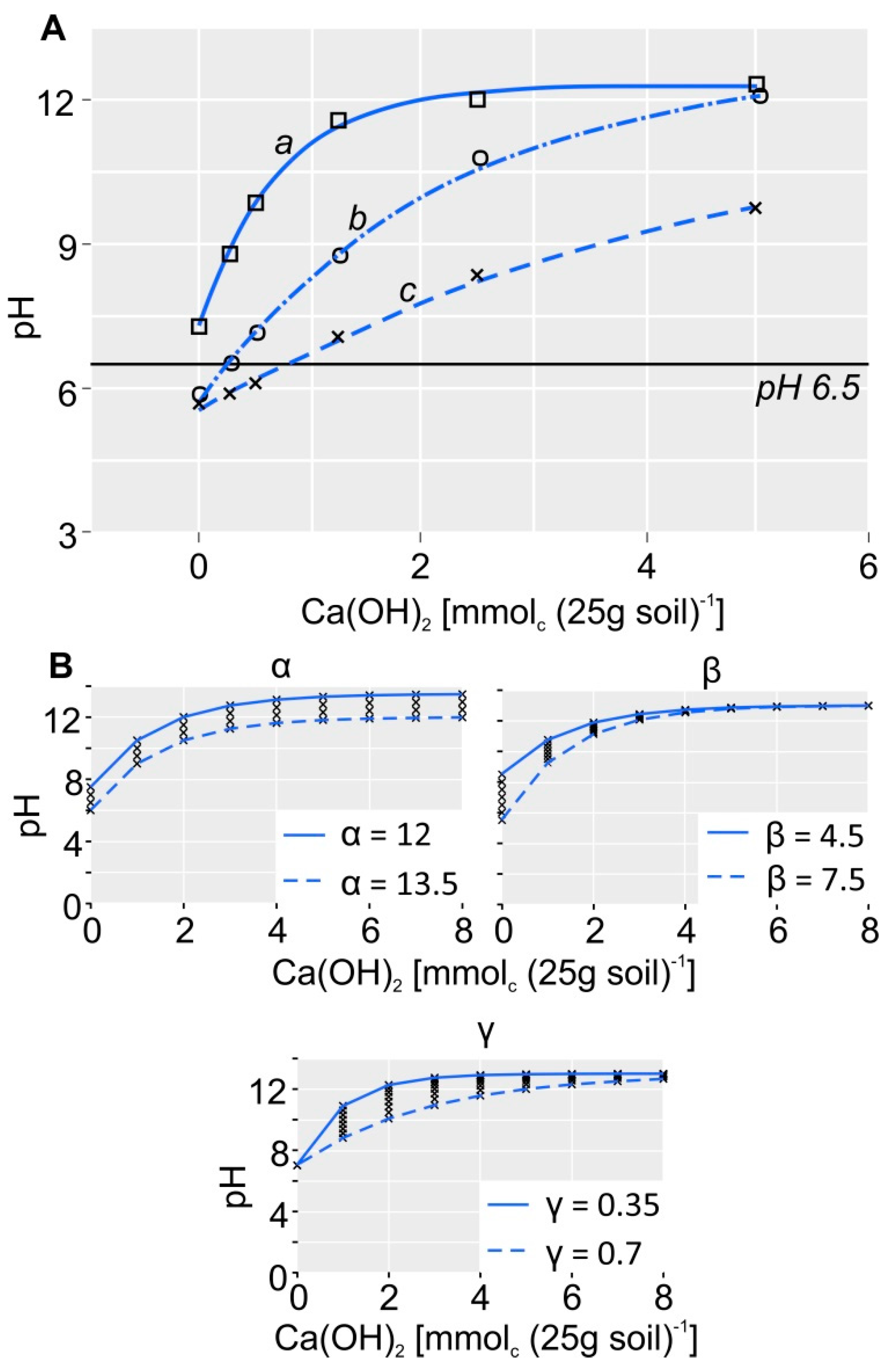
2.3. LR Based on Base Neutralizing Capacity (LRBNC)
2.4. LR Based on VDLUFA Guidelines (LRVDLUFA)
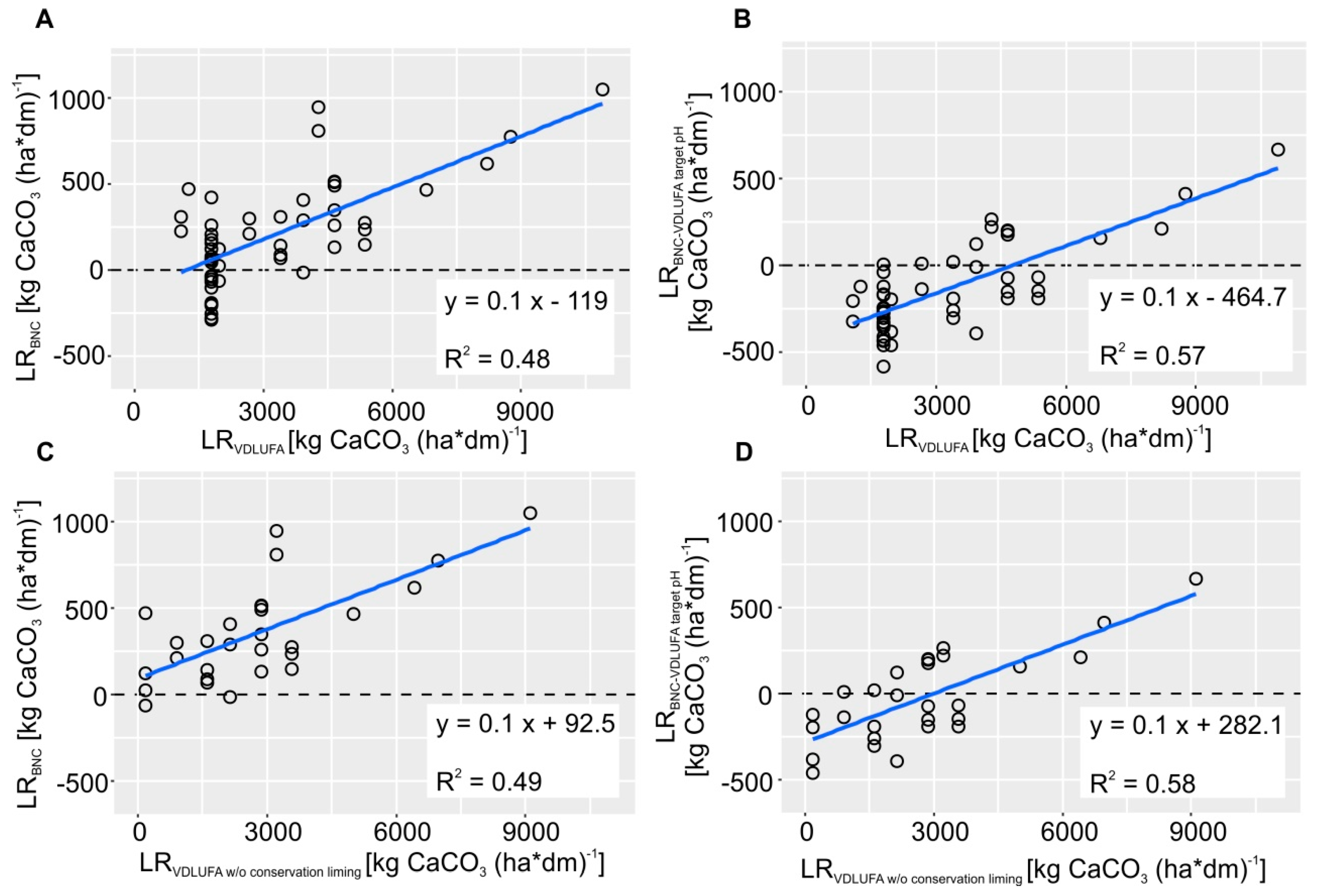
2.5. Comparison between LRBNC and LRVDLUFA
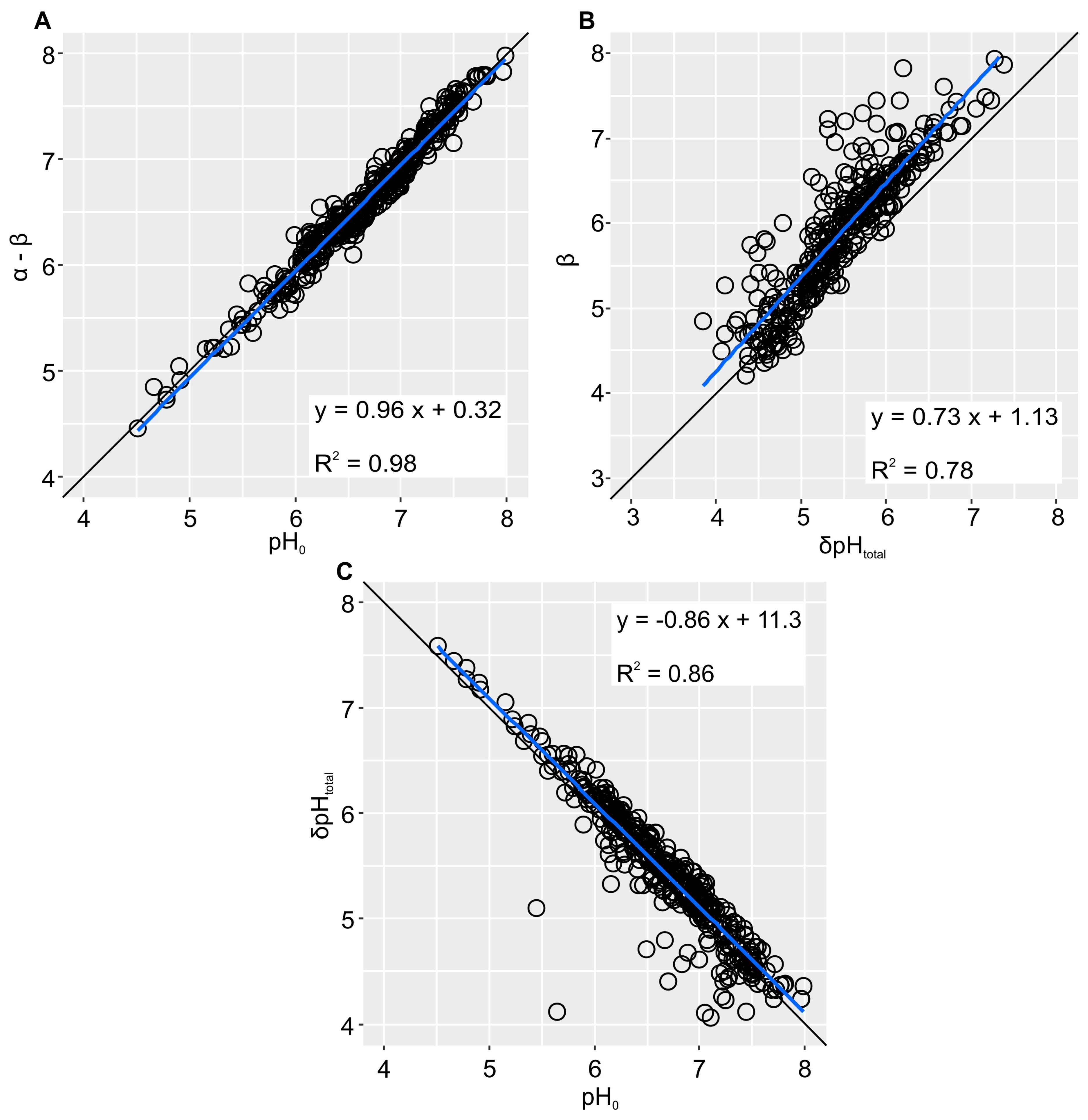
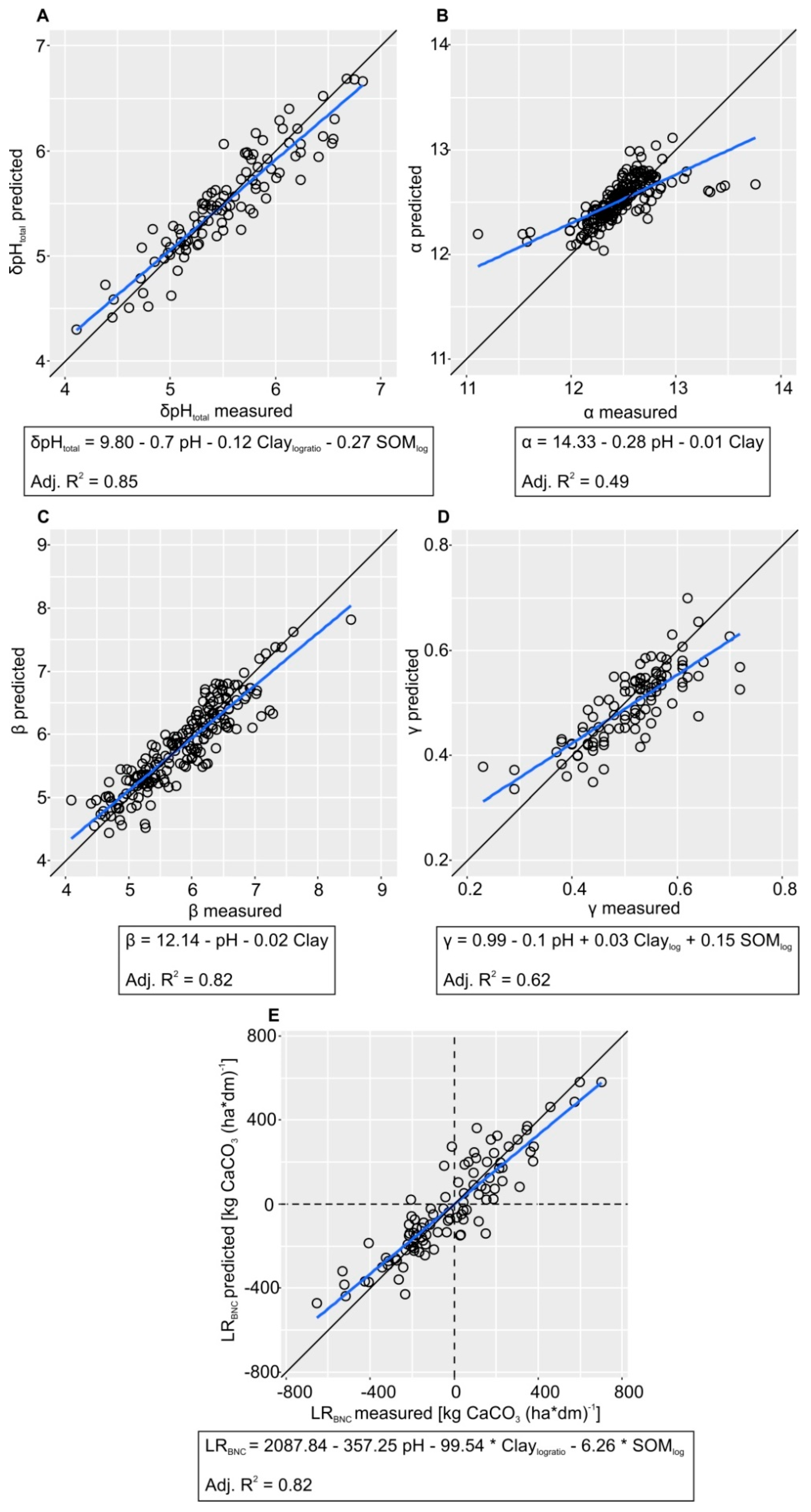
2.6. Titration Curve Fitting and Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Robson, A.D. Soil Acidity and Plant Growth; Academic Press: Sydney, NSW, Australia, 1989. [Google Scholar]
- Mengel, K.; Kirkby, E.A. Principles of Plant Nutrition, 5th ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001. [Google Scholar]
- Epstein, E.; Bloom, A.J. Mineral Nutrition of Plants: Principles and Perspectives, 2nd ed.; Sinauer Associates: Sunderland, MA, USA, 2001. [Google Scholar]
- Blume, H.-P.; Bruemmer, G.W.; Fleige, H.; Horn, R.; Kandeler, E.; Koegel-Knabner, I.; Kretzschmar, R.; Stahr, K.; Wilke, B.-M. Scheffer/Schachtschabel Soil Science; Springer: Heidelberg, Germany, 2016; p. 618. [Google Scholar]
- Pagani, A.; Sawyer, J.E.; Mallarino, A.P.; Moody, L.; Davis, J.; Phillips, S. Site-Specific Nutrient Management For. Nutrient Management Planning To Improve Crop. Production, Environmental Quality, and Economic Return: Module 8—Soil pH and Lime Management; U.S. Department of Agriculture—Natural Resources Conservation Service (USDA-NRCS): Washington, DC, USA, 2017. Available online: http://www.nutrientstewardship.com/training/ (accessed on 6 January 2020).
- Goulding, K.W. Soil acidification and the importance of liming agricultural soils with particular reference to the United Kingdom. Soil Use Manag. 2016, 32, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Holland, J.; Bennett, A.; Newton, A.; White, P.; McKenzie, B.; George, T. Liming impacts on soils, crops and biodiversity in the UK: A review. Sci. Total Environ. 2018, 610, 316–332. [Google Scholar] [CrossRef] [PubMed]
- Mengel, K.; Kirkby, E.A. Principles of Plant Nutrition; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2001; p. 849. [Google Scholar]
- Sims, J.T. Lime Requirement. In Methods of Soil Analysis—Part 3: Chemical Methods; Bigham, J.M., Bartels, J.M., Eds.; Soil Science Society of America Book Series; John Wiley & Sons, Inc.: New York, NY, USA, 1996; Volume 5, p. 670. [Google Scholar]
- Dunn, L.E. Lime-requirement of soils by means of titration curves. Soil Sci. 1943, 56, 341–352. [Google Scholar] [CrossRef]
- Meiwes, K.J.; Koenig, N.; Khana, P.K.; Prenzel, J.; Ulrich, B. Chemical test methods for mineral soils, litter layers and roots to characterize and evaluate acidification in forest soils (In German: Chemische Untersuchungsverfahren für Mineralboden, Auflagehumus und Wurzeln zur Charakterisierung und Bewertung der Versauerung in Waldböden). In Die Erfassung des Stoffkreislaufs in Waldökosystemen—Konzeption und Methodik, Berichte des Forschungszentrums Waldökosysteme/Waldsterben, Bd. 7; Meiwes, K.-J., Hauhs, M., Gerke, H., Asche, N., Matzner, E., Lammersdorf, N., Eds.; Institut für Bodenkunde und Waldernährung der Universität Göttingen: Göttingen, Germany, 1984. [Google Scholar]
- Alley, M.; Zelazny, L. Soil Acidity: Soil pH and Lime Needs. In Soil Testing: Sampling, Correlation, Calibration, and Interpretation, Proceedings of the Annual Meeting of the American Society of Agronomy and Soil Science Society of America, Chicogo, IL, USA, 1–5 December 1985; Brown, J.R., Ed.; Soil Science Society of America, Inc.: Madison, WI, USA, 1987; pp. 65–72. [Google Scholar]
- McLean, E. Principles underlying the practice of determining lime requirements of acid soils by use of buffer methods. Comm. Soil Sci. Plant. Anal. 1978, 9, 699–715. [Google Scholar] [CrossRef]
- Liu, M.; Kissel, D.E.; Cabrera, M.L.; Vendrell, P.F. Soil Lime Requirement by Direct Titration with a Single Addition of Calcium Hyroxide. Soil Sci. Soc. Am. J. 2005, 69, 522–530. [Google Scholar] [CrossRef]
- Kissel, D.E.; Isaac, R.A.; Hitchcock, R.; Sonon, L.S.; Vendrell, P.F. Implementation of soil lime requirement by a single-addition titration method. Commun. Soil Sci. Plant. Anal. 2007, 38, 1341–1352. [Google Scholar] [CrossRef]
- Thompson, J.S.; Kissel, D.E.; Cabrera, M.L.; Sonon, L.S. Equilibration reaction from single addition of base to determine soil lime requirement. Soil Sci. Soc. Am. Proc. 2010, 74, 663–669. [Google Scholar] [CrossRef]
- Alabi, K.E.; Sorensen, R.C.; Knudsen, D.; Rehm, G.W. Comparison of several lime requirement methods on course textured soils of Northeastern Nebraska. Soil Sci. Soc. Am. J. 1986, 50, 937–941. [Google Scholar] [CrossRef]
- Owusu-Bennoah, E.; Acquaye, D.K.; Mahamah, T. Comparative study of selected lime requirement methods for some acid Ghanaian soils. Commun. Soil Sci. Plant. Anal. 1995, 26, 937–950. [Google Scholar] [CrossRef]
- Shoemaker, H.E.; Mclean, E.O.; Pratt, P.F. Buffer methods for determining lime requirement of soils with appreciable amounts of extractable aluminum. Soil Sci. Soc. Am. Proc. 1961, 25, 274–277. [Google Scholar] [CrossRef]
- Adams, F.; Evans, C.E. A rapid method for measuring lime requirement of red-yellow podzolic soils. Soil Sci. Soc. Am. Proc. 1962, 26, 355–357. [Google Scholar] [CrossRef]
- Mehlich, A. New buffer pH method for rapid estimation of exchangeable acidity and lime requirement of soils. Commun. Soil Sci. Plant. Anal. 1976, 7, 637–652. [Google Scholar] [CrossRef]
- Viscarra Rossel, R.A.; McBratney, A.B. A response surface calibration model for rapid and versatile site-specific lime requirement predictions in southeastern Australia. Aust. J. Soil Res. 2001, 39, 185–201. [Google Scholar] [CrossRef]
- Pagani, A.; Mallarino, A.P. Comparison of Methods to Determine Crop Lime Requirement Under Field Conditions. Soil Sci. Soc. Am. J. 2011, 76, 1855–1866. [Google Scholar] [CrossRef]
- Vossen, P. Changing pH in Soil; University of California Cooperative Extension: Davis, CA, USA, 2002. [Google Scholar]
- von Wulffen, U.; Roschke, M.; Kape, H.-E. Target Values for Soil Fertility Analysis and Guidance, as well as for the Implementation of the Fertilization Ordinance; (In German: Richtwerte für die Untersuchung und Beratung sowie zur fachlichen Umsetzung der Düngeverordnung (DüV): Gemeinsame Hinweise der Länder Brandenburg, Mecklenburg-Vorpommern und Sachsen-Anhalt); Landesanstalt für Landwirtschaft, Forsten und Gartenbau des Landes Sachsen-Anhalt (LLFG): Bernburg, Germany; Güterfelde, Germany; Rostock, Germany, 2008. [Google Scholar]
- Kerschberger, M. Ermittlung von Kalkdüngermengen zur Erreichung optimaler pH-Werte des Bodens. Ph.D. Thesis, Institut für Pflanzenernährung Jena der Akademie der Landwirtschaftswissenschaften der DDR, Jena, Germany, 1980; p. 132, Unpublished doctoral dissertation. [Google Scholar]
- Kerschberger, M. Ermittlung Optimaler Bodenreaktion auf dem Ackerland—Sekundarrohstoffe im Stoffkreislauf der Landwirtschaft und Weitere Beiträge aus den Öffentlichen Sitzungen; VDLUFA-Verlag: Darmstadt, Germany, 1996; Volume 44, pp. 591–594. [Google Scholar]
- Kerschberger, M.; Deller, B.; Hege, U.; Heyn, J.; Kape, H.; Krause, O. Bestimmung des Kalkbedarfs von Acker-und Grünlandböden; VDLUFA-Verlag: Darmstadt, Germany, 2000. [Google Scholar]
- Kerschberger, M.; Marks, G. Einstellung und Erhaltung Eines Standorttypischen Optimalen pH-Wertes im Boden—Grundvoraussetzung für Eine Effektive und Umweltverträgliche Pflanzenproduktion; Berichte über Landwirtschaft: Stuttgart, Germany, 2007; Volume 85, pp. 56–77. [Google Scholar]
- Havlin, J.L.; Tisdale, S.L.; Nelson, W.L.; Beaton, J.D. Soil Fertility and Fertilizers: An Introduction to Nutrient Management; Prentice-Hall, Inc.: Upper Saddle River, NJ, USA, 2005; p. 499. [Google Scholar]
- Schilling, G. Pflanzenernährung und Düngung; UTB: Stuttgart, Germany, 2000; p. 464. [Google Scholar]
- Krbetschek, M.R.; Degering, D.; Alexowsky, W. Infrared radiofluorescence ages (IR-RF) of Lower Saalian sediments from Central and Eastern Germany. Zeitschr. Dtsch. Ges. Geowiss. 2008, 133–140. [Google Scholar] [CrossRef]
- Ehlers, J.; Grube, A.; Stephan, H.J.; Wansa, S. Pleistocene glaciations of North Germany e new results. In Quaternary Glaciations e Extent and Chronology e a Closer Look. Developments in Quaternary Science 15; Ehlers, J., Gibbard, P.L., Hughes, P.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2011; pp. 149–162. [Google Scholar]
- Winsemann, J.; Brandes, C.; Polom, U.; Weber, C. Depositional architecture and palaeogeographic significance of Middle Pleistocene glaciolacustrine ice marginal deposits in northwestern Germany: A synoptic overview. E G Quat. Sci. J. 2011, 60, 212–235. [Google Scholar] [CrossRef]
- Moreau, J.; Huuse, M.; Janszen, A.; van der Vegt, P.; Gibbard, P.L.; Moscariello, A. The glaciogenic unconformity of the southern North Sea. In Glaciogenic Reservoirs; Huuse, M., Redfern, J., Le Heron, D.P., Dixon, R.J., Moscariello, A., Craig, J., Eds.; Geological Society of London: London, UK, 2012; pp. 99–110, Special Publication 368. [Google Scholar]
- Peverill, K.; Sparrow, L.; Reuter, D. Soil Analysis: An Interpretation Manual; CSIRO publishing: Collingwood, VIC, Australia, 1999; p. 369. [Google Scholar]
- Utermann, J.; Gorny, A.; Hauenstein, M.; Malessa, V.; Müller, U.; Scheffer, B. Geologisches Jahrbuch Reihe G, Band G 8; Schweizerbart´sche Verlagsbuchhandlung: Stuttgart, Germany, 2000. [Google Scholar]
- Defra. The Fertiliser Manual (RB209); TSO: London, UK, 2010. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: http://www.R-project.org (accessed on 11 November 2019).
- Huang, J.; Subasinghe, R.; Triantafilis, J. Mapping particle-size fractions as a composition using additive log-ratio transformation and ancillary data. Soil Sci. Soc. Am. J. 2014, 78, 1967–1976. [Google Scholar] [CrossRef]
- Muzzamal, M.; Huang, J.; Nielson, R.; Sefton, M.; Triantafilis, J. Mapping soil particle-size fractions using additive log-ratio (ALR) and isometric log-ratio (ILR) transformations and proximally sensed ancillary data. Clays Clay Miner. 2018, 66, 9–27. [Google Scholar] [CrossRef]
- Saltelli, A.; Ratto, M.; Andres, T.; Campolongo, F.; Cariboni, J.; Gatelli, D.; Saisana, M.; Tarantola, S. Global Sensitivity Analysis: The Primer; John Wiley & Sons: Chichester, UK, 2008. [Google Scholar]
- Eckelmann, W.; Sponagel, H.; Grottenthaler, W. Bodenkundliche Kartieranleitung KA5, 5th ed.; Bundesanstalt für Geowissenschaften und Rohstoffe in Cooperation with Staatliche Geologische Dienste: Hanover, Germany, 2005. [Google Scholar]
- Sparks, D. Environmental Soil Chemistry, 2nd ed.; Academic Press, an Imprint of Elsevier: San Diego, CA, USA, 2003. [Google Scholar]
- Sposito, G. The Chemistry of Soils, 3rd ed.; Oxford University Press: New York, NY, USA, 2016. [Google Scholar]
- Weaver, A.R.; Kissel, D.E.; Chen, F.; West, L.T.; Adkins, W.; Rickman, D.; Luvall, J.C. Mapping soil pH buffering capacity of selected fields in the coastal plain. Soil Sci. Soc. Am. J. 2004, 68, 662–668. [Google Scholar] [CrossRef]
- Hoskins, B.R.; Erich, M.S. Modification of the Mehlich lime buff er test. Commun. Soil Sci. Plant. Anal. 2008, 39, 2270–2281. [Google Scholar] [CrossRef]
- Wolf, A.M.; Beegle, D.B.; Hoskins, B. Comparison of Shoemaker–McLean–Pratt and modified Mehlich buffer tests for lime requirement on Pennsylvania soils. Commun. Soil Sci. Plant. Anal. 2008, 39, 1848–1857. [Google Scholar] [CrossRef]
- Süsser, P.; Schwertmann, U. Proton buffering in mineral horizons of some acid forest soils. Geoderma 1991, 49, 63–76. [Google Scholar] [CrossRef]
- Schwertmann, U.; Süsser, P.; Nätscher, L. Proton buffer compounds in soils. J. Plant. Nutr. Soil Sci. 1987, 150, 63–76. [Google Scholar]
- Hue, N.V.; Ikawa, H. Liming acid soils of Hawaii. University of Hawaii. CTAHR Agron. Soils 1994, 1, 1–2. [Google Scholar]
- Nelson, P.N.; Su, N. Soil pH buffering capacity: A descriptive function and its application to some acidic tropical soils. Aust. J. Soil Res. 2010, 48, 201–207. [Google Scholar] [CrossRef]
- Magdoff, F.R.; Bartlett, R.J. Soil pH buffering revisited. Soil Sci. Soc. Am. Proc. 1985, 49, 145–148. [Google Scholar] [CrossRef]
- Bloom, P.R. Soil pH and pH buffering. In Handbook of Soil Science; Sumner, M.E., Li, Y., Huan, P.M., Eds.; CRC Press: Boca Raton, FL, USA, 2000. [Google Scholar]
- Stevenson, F.J. Humus Chemistry, 2nd ed.; Wiley: New York, NY, USA, 1994. [Google Scholar]
- Mikkelsen, R.L. Humic materials for agriculture. Better Crops 2005, 89, 6–10. [Google Scholar]
- Leenen, M.; Welp, G.; Gebbers, R.; Pätzold, S. Rapid determination of lime requirement by mid-infrared spectroscopy: A promising approach for precision agriculture. J. Plant. Nutr. Soil Sci. 2019, 182, 953–963. [Google Scholar] [CrossRef]
- BAD. Nährstoffverluste aus landwirtschaftlichen Betrieben mit einer Bewirtschaftung nach guter fachlicher Praxis; Bundesarbeitskreis Düngung: Frankfurt/Main, Germany, 2003. [Google Scholar]
| pH Class | Lime Supply | Lime Requirement |
|---|---|---|
| A | Very low | Recovery liming |
| B | Low | Buildup liming |
| C | Optimal | Maintenance liming |
| D | High | No liming |
| E | Very high | No liming and no use of fertilizers that react physiologically or chemically alkaline |
| n | Minimum | Mean | Median | Maximum | Standard Deviation | ||
|---|---|---|---|---|---|---|---|
| Standard soil data | pH | 360 | 3.9 | 6.1 | 6.2 | 7.4 | 0.7 |
| SOM * (%) | 100 | 0.8 | 1.6 | 1.4 | 5.6 | 0.8 | |
| Clay (%) | 190 | 1.0 | 12.2 | 9.5 | 46.0 | 8.9 | |
| Silt (%) | 190 | 2.0 | 22.3 | 23.8 | 37.9 | 8.3 | |
| Sand (%) | 190 | 30.0 | 65.5 | 65.6 | 97.0 | 13.5 | |
| BNC data ** | pH0 | 420 | 4.5 | 6.7 | 6.7 | 8.0 | 0.6 |
| pH0.25 | 420 | 5.6 | 7.5 | 7.4 | 9.7 | 0.6 | |
| pH0.5 | 420 | 6.1 | 8.2 | 8.1 | 10.3 | 0.7 | |
| pH1.25 | 420 | 7.0 | 9.8 | 9.8 | 11.6 | 0.7 | |
| pH2.5 | 420 | 8.2 | 11.2 | 11.3 | 12.0 | 0.5 | |
| pH5 | 420 | 9.1 | 12.1 | 12.1 | 12.4 | 0.3 | |
| δpHtotal | 420 | 2.5 | 5.4 | 5.4 | 7.6 | 0.7 | |
| α | 420 | 11.1 | 12.5 | 12.4 | 13.8 | 0.3 | |
| β | 420 | 4.1 | 5.8 | 5.8 | 8.5 | 0.8 | |
| γ | 420 | 0.1 | 0.5 | 0.5 | 0.9 | 0.1 | |
| α | β | γ | |
|---|---|---|---|
| α | 0.85 | 0.57 | |
| β | 0.85 | 0.44 | |
| γ | 0.57 | 0.44 |
| pH | SOM * | Clay ** | Sand ** | |
|---|---|---|---|---|
| δpHtotal ° | −0.91 | −0.31 | −0.22 | −0.14 |
| α | −0.84 | 0.18 | −0.02 | 0.09 |
| β | −0.92 | 0.27 | −0.25 | 0.01 |
| γ | −0.77 | 0.63 | 0.22 | −0.24 |
| LRBNC °° | −0.89 | 0.11 | −0.35 | −0.01 |
| pH | SOM * | Clay ** | Sand ** | |
|---|---|---|---|---|
| δpHtotal | −0.70 | −0.18 | −0.07 | −0.05 |
| α | −0.60 | 0.06 | 0.02 | 0.07 |
| β | −0.82 | 0.07 | −0.10 | −0.01 |
| γ | −0.60 | 0.50 | 0.10 | −0.17 |
| LRBNC | −0.77 | 0.00 | −0.19 | 0.02 |
| Annual Precipitation | ||||
|---|---|---|---|---|
| Soil Texture | Crop Type | Low (<600 mm) | Medium (600–750 mm) | High (>750 mm) |
| Sandy soils | Farmland | 540 | 710 | 890 |
| Grassland | 270 | 450 | 630 | |
| Loamy soils | Farmland | 710 | 890 | 1070 |
| Grassland | 360 | 540 | 710 | |
| Clayey soils | Farmland | 890 | 1070 | 1250 |
| Grassland | 450 | 630 | 800 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vogel, S.; Bönecke, E.; Kling, C.; Kramer, E.; Lück, K.; Nagel, A.; Philipp, G.; Rühlmann, J.; Schröter, I.; Gebbers, R. Base Neutralizing Capacity of Agricultural Soils in a Quaternary Landscape of North-East Germany and Its Relationship to Best Management Practices in Lime Requirement Determination. Agronomy 2020, 10, 877. https://doi.org/10.3390/agronomy10060877
Vogel S, Bönecke E, Kling C, Kramer E, Lück K, Nagel A, Philipp G, Rühlmann J, Schröter I, Gebbers R. Base Neutralizing Capacity of Agricultural Soils in a Quaternary Landscape of North-East Germany and Its Relationship to Best Management Practices in Lime Requirement Determination. Agronomy. 2020; 10(6):877. https://doi.org/10.3390/agronomy10060877
Chicago/Turabian StyleVogel, Sebastian, Eric Bönecke, Charlotte Kling, Eckart Kramer, Katrin Lück, Anne Nagel, Golo Philipp, Jörg Rühlmann, Ingmar Schröter, and Robin Gebbers. 2020. "Base Neutralizing Capacity of Agricultural Soils in a Quaternary Landscape of North-East Germany and Its Relationship to Best Management Practices in Lime Requirement Determination" Agronomy 10, no. 6: 877. https://doi.org/10.3390/agronomy10060877
APA StyleVogel, S., Bönecke, E., Kling, C., Kramer, E., Lück, K., Nagel, A., Philipp, G., Rühlmann, J., Schröter, I., & Gebbers, R. (2020). Base Neutralizing Capacity of Agricultural Soils in a Quaternary Landscape of North-East Germany and Its Relationship to Best Management Practices in Lime Requirement Determination. Agronomy, 10(6), 877. https://doi.org/10.3390/agronomy10060877






