Assessing the Potential of Jellyfish as an Organic Soil Amendment to Enhance Seed Germination and Seedling Establishment in Sand Dune Restoration
Abstract
1. Introduction
2. Material and Methods
2.1. Petri Plate Experiment
2.2. Pot Experiment
2.3. Statistical Analysis
3. Results
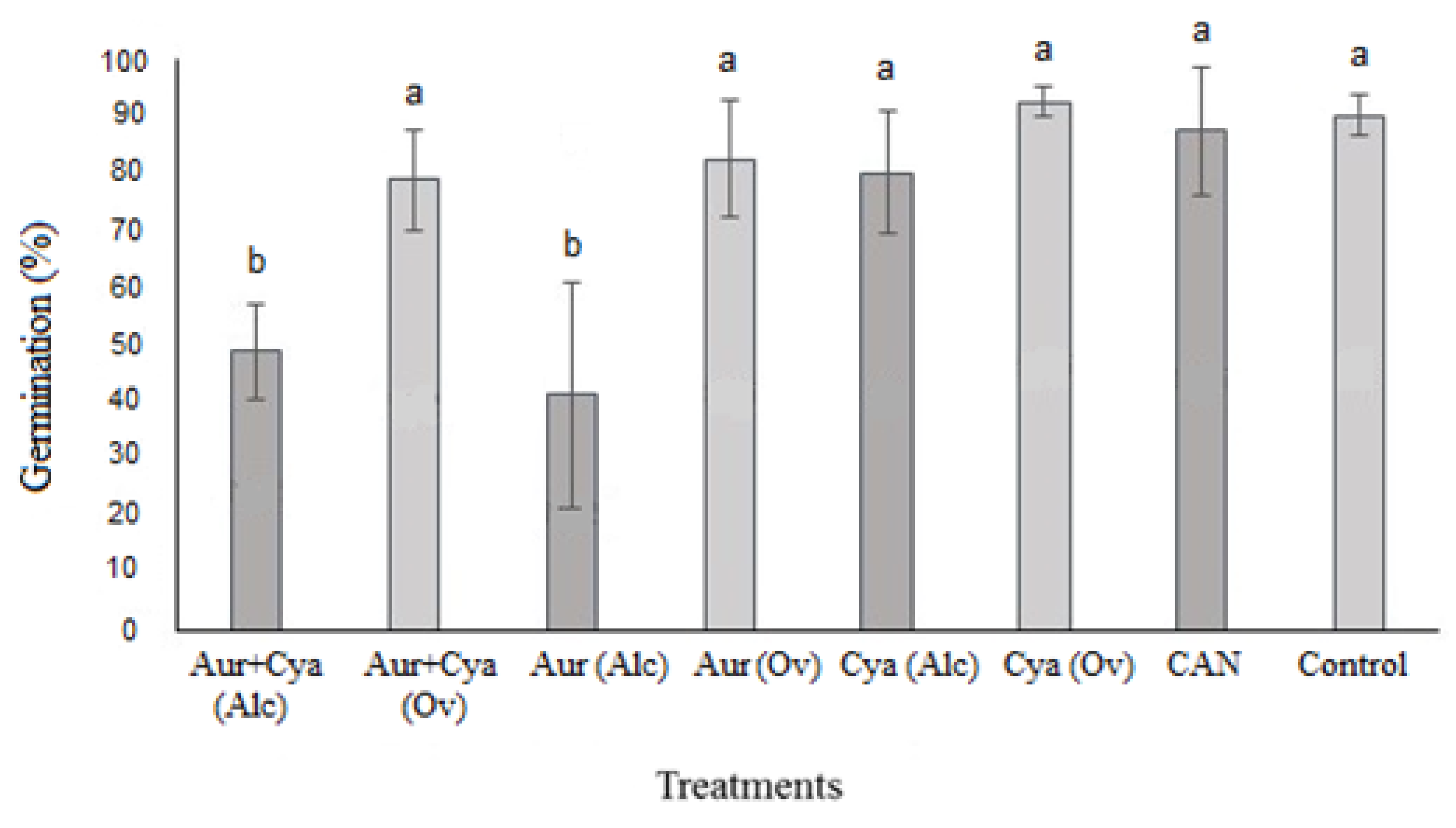
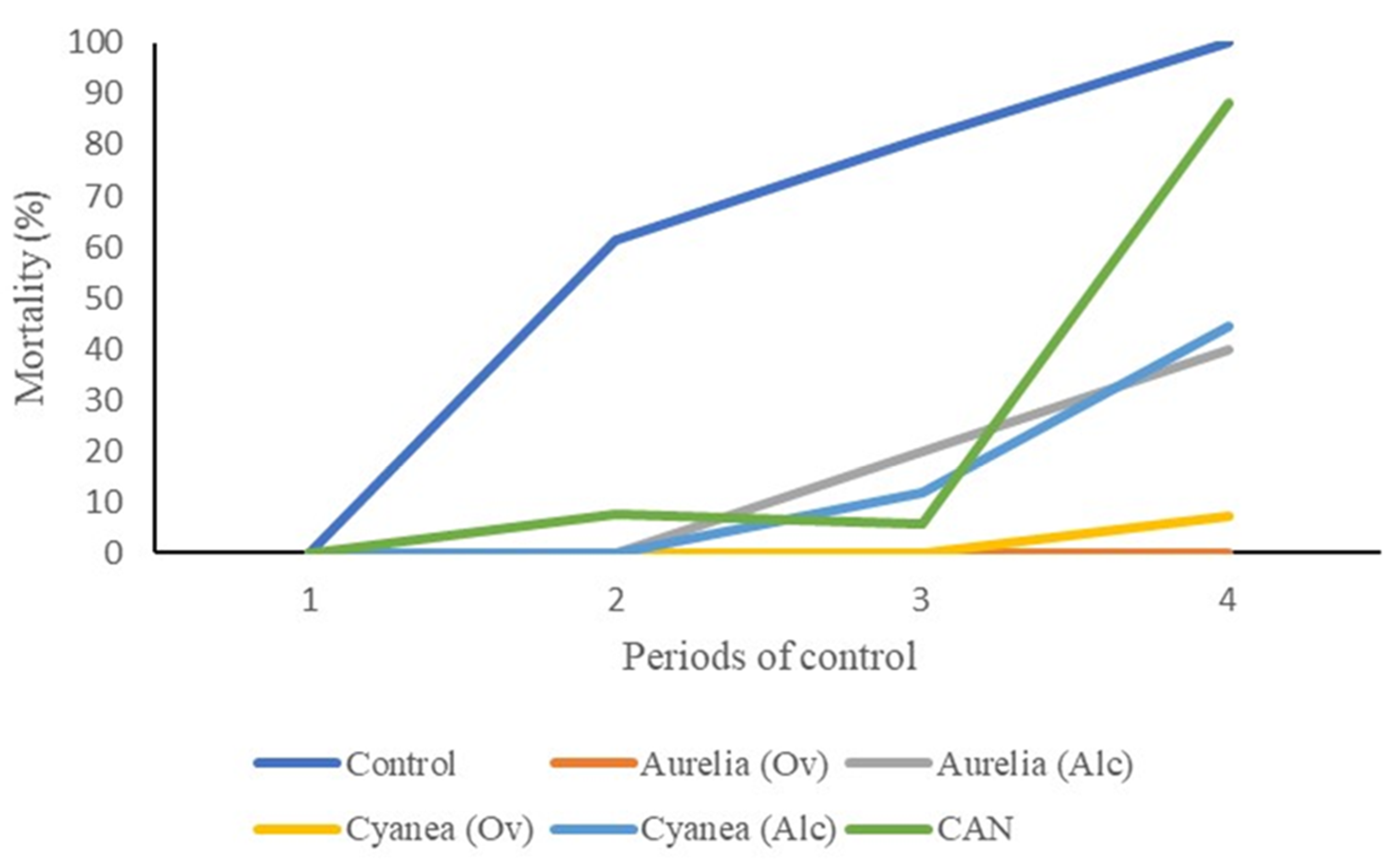
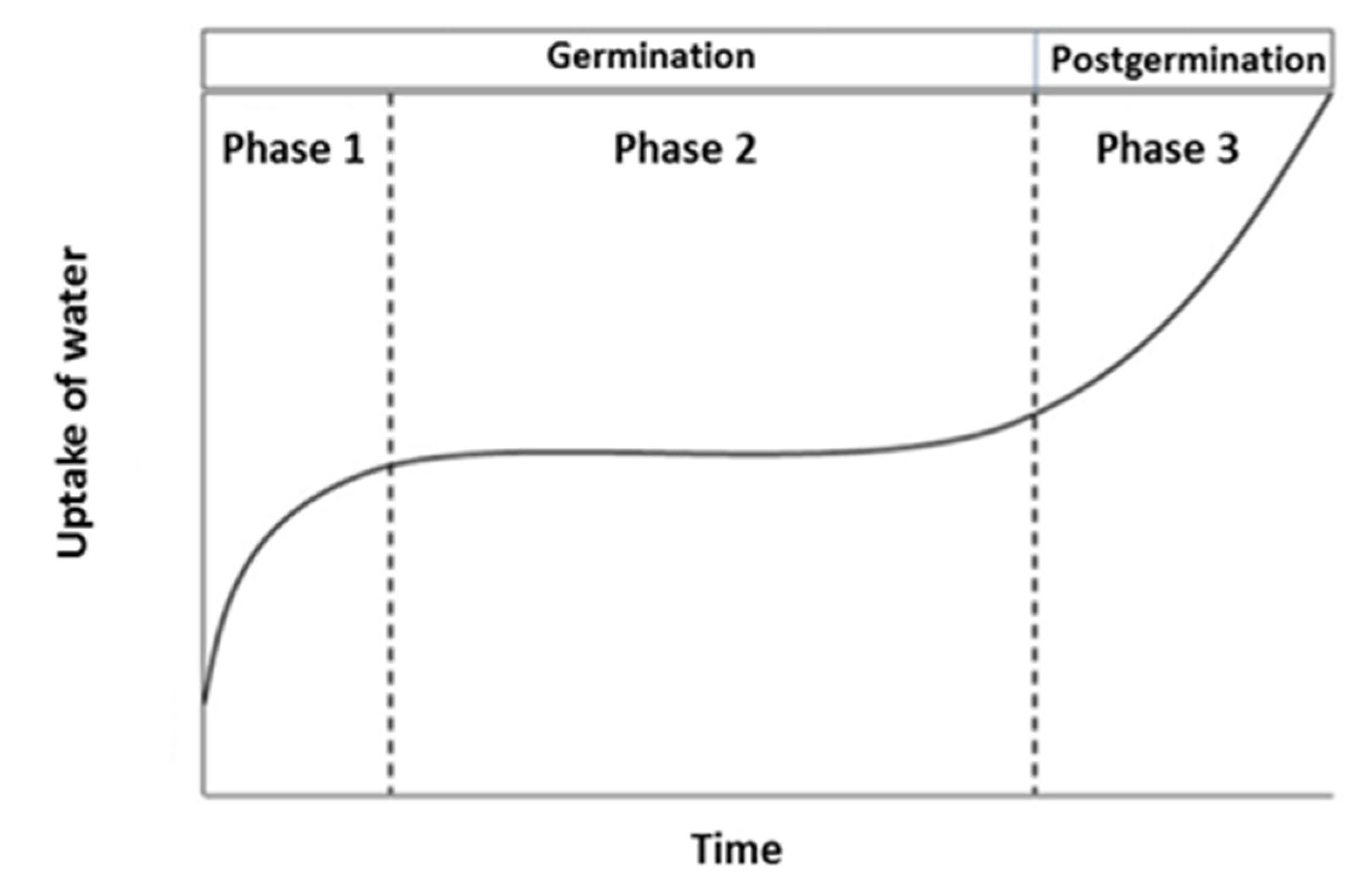
3.1. Petri Plate Experiment
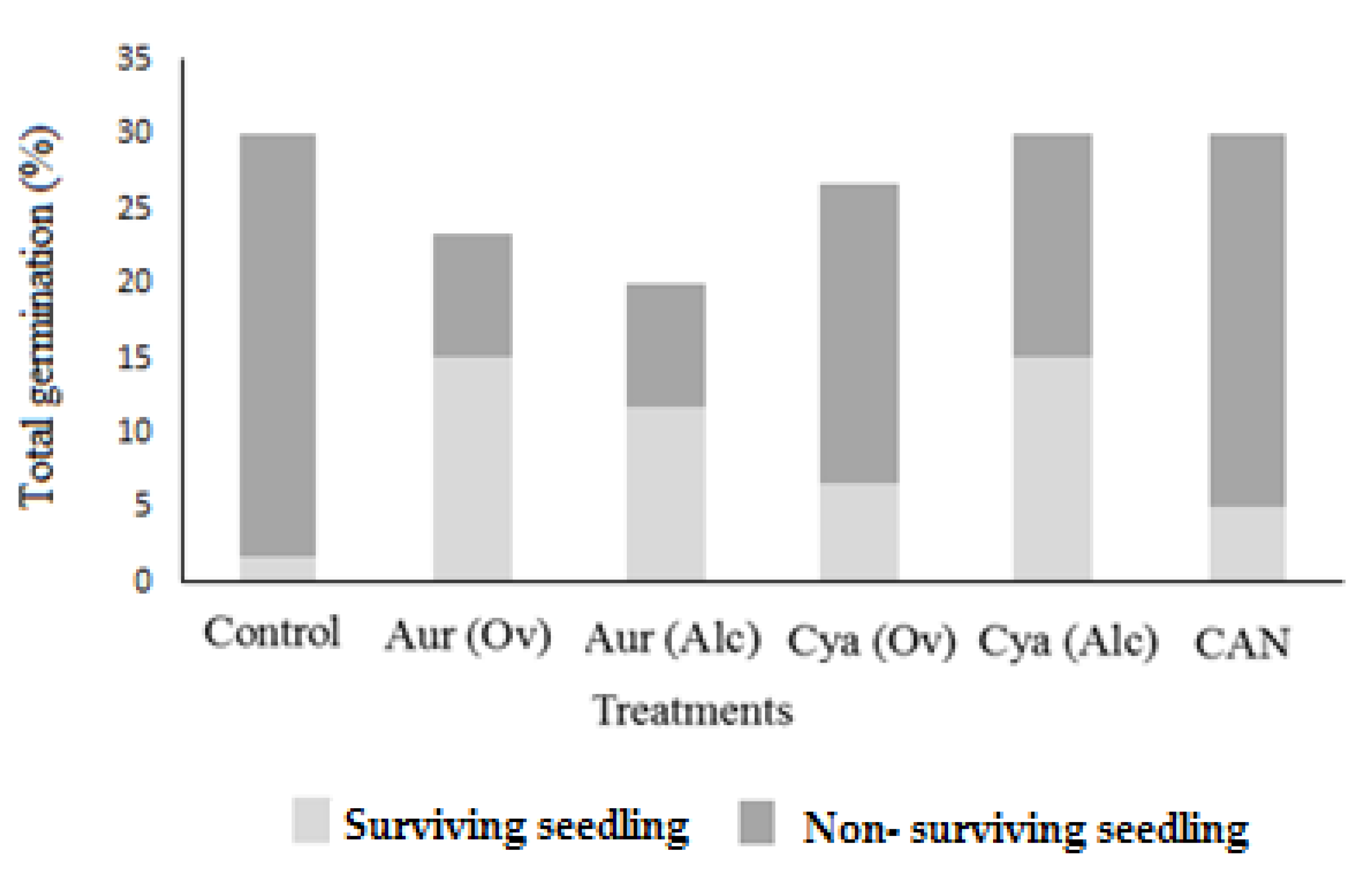
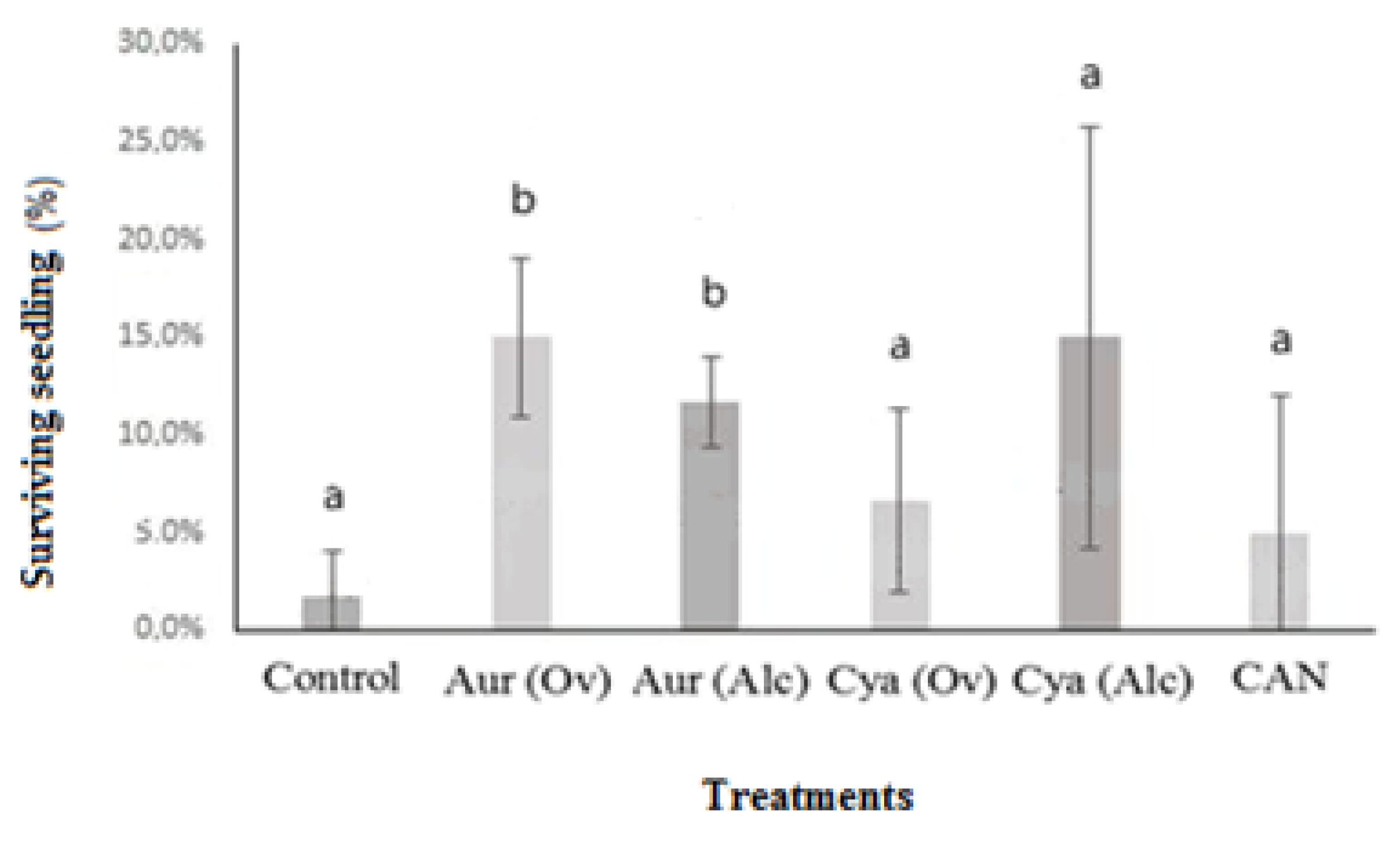
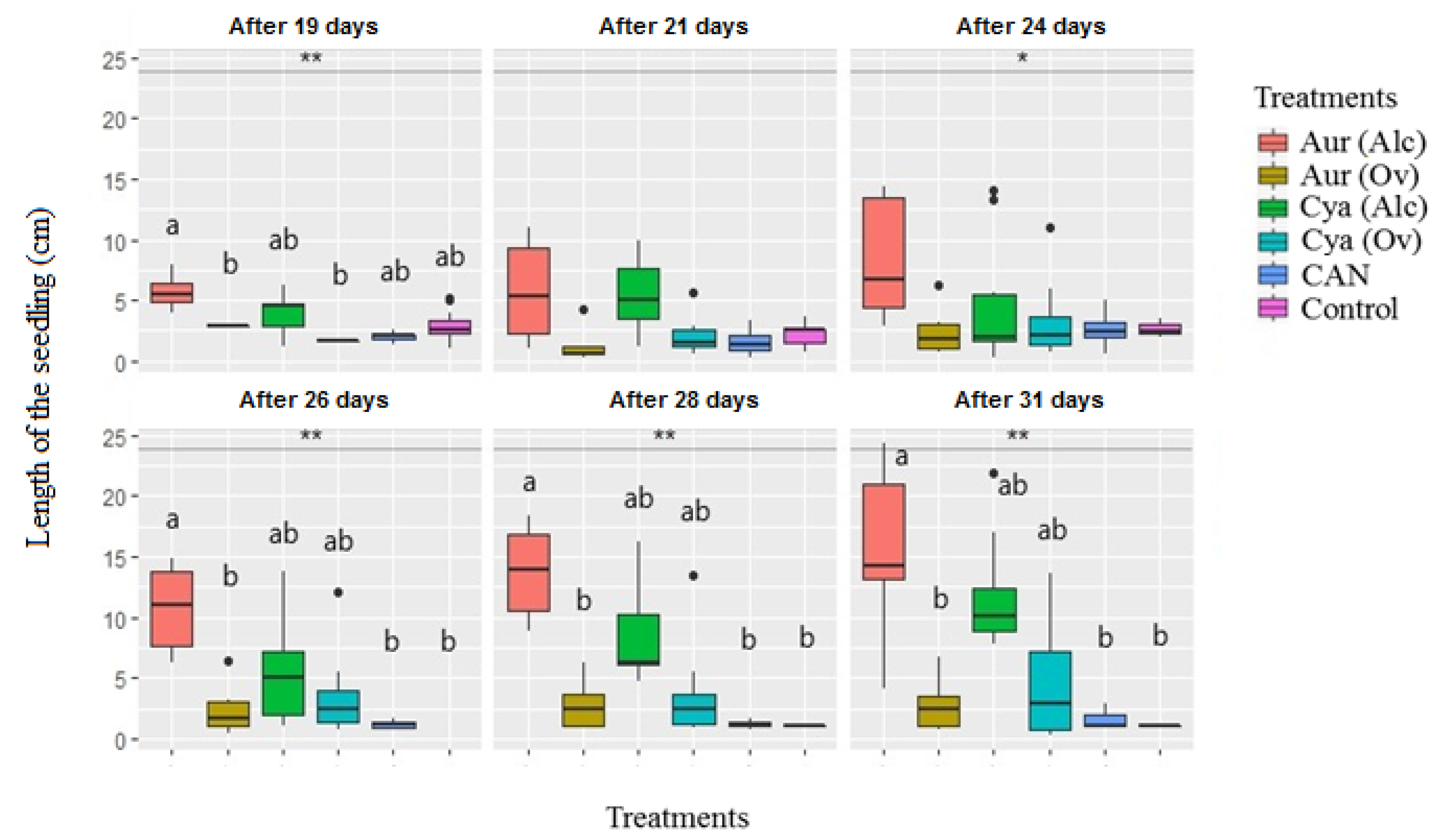
3.2. Pot Experiment
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lithgow, D.; Martinez, M.L.; Gallego-Fernandez, J.B.; Hesp, P.A.; Flores, P.; Gachuz, S.; Rodríguez-Revelo, N.; Jiménez-Orocio, O.; Mendoza-González, G.; Álvarez-Molina, L.L. Linking restoration ecology with coastal dune restoration. Geomorphology 2013, 199, 214–224. [Google Scholar] [CrossRef]
- Torretta, V.; Katsoyiannis, I.; Collivignarelli, M.C.; Bertanza, G.; Xanthopoulou, M. Water reuse as a secure pathway to deal with water scarcity. MATEC Web Conf. 2020, 305, 1–6. [Google Scholar] [CrossRef][Green Version]
- Kim, J.H.; Lim, S.D.; Jang, C.S. Oryza sativa drought-, heat-, and salt-induced RING finger protein 1 (OsDHSRP1) negatively regulates abiotic stress-responsive gene expression. Plant Mol. Biol. 2020. [Google Scholar] [CrossRef] [PubMed]
- De Lillis, M.; Costanzo, L.; Bianco, P.M.; Tinelli, A. Sustainability of sand dune restoration along the coast of the Tyrrhenian sea. J. Coast. Conserv. 2004, 10, 93–100. [Google Scholar] [CrossRef]
- Spano, C.; Balestri, M.; Bottega, S.; Grilli, I.; Forino, L.M.C.; Ciccarelli, D. Anthemis maritima L. in different coastal habitats: A tool to explore plant plasticity. Estuar. Coast. Shelf Sci. 2013, 129, 105–111. [Google Scholar] [CrossRef]
- Donohue, K.; de Casas, R.R.; Burghardt, L.; Kovach, K.; Willis, C.G. Germination, postgermination adaptation, and species ecological ranges. Annu. Rev. Ecol. Evol. Syst. 2010, 41, 293–319. [Google Scholar] [CrossRef]
- Yuan, X.; Wen, B. Seed germination response to high temperature and water stress in three invasive Asteraceae weeds from Xishuangbanna, SW China. PLoS ONE 2018. [Google Scholar] [CrossRef]
- Bewley, J.D. Seed germination and dormancy. Plant Cell 1997, 9, 1055–1066. [Google Scholar] [CrossRef]
- Kildisheva, O.A.; Kingsley, W.; Dixon, K.W.; Silveira, F.A.O.; Chapman, T.; Sacco, A.D.; Mondoni, A.; Turner, S.R.; Cross, A.T. Dormancy and germination: Making every seed count in restoration. Restor. Ecol. 2020. [Google Scholar] [CrossRef]
- Forcella, F.; Benech, A.R.L.; Sanchez, R.; Ghersa, C.M. Modeling seedling emergence. Field Crop. Res. 2000, 67, 123–139. [Google Scholar] [CrossRef]
- Padilla, F.M.; Pugnaire, F.I. Rooting depth and soil moisture control Mediterranean woody seedling survival during drought. Funct. Ecol. 2007, 21, 489–495. [Google Scholar] [CrossRef]
- Manz, B.; Muller, K.; Kucera, B.; Volke, F.; Leubner-Metzger, G. Water uptake and distribution in germinating tobacco seeds investigated in vivo by nuclear magnetic resonance imaging. Plant Physiol. 2005, 138, 1538–1551. [Google Scholar] [CrossRef]
- Bewley, J.D.; Bradford, K.J.; Hilhorst, H.W.M.; Nonogaki, H. Seeds: Physiology of Development, Germination and Dormancy; Springer: New York, NY, USA, 2013. [Google Scholar]
- Emadodin, I.; Reinsch, T.; Taube, F. Potential of jellyfish as a seed germination promoter. Jahrestag. Arbeitsgem. Grünl. Futterb. 2019. Available online: https://www.lfl.bayern.de/mam/cms07/ipz/dateien/aggf_2019_emadodin_et_al.pdf (accessed on 6 May 2019).
- Condon, R.H.; Steinberg, D.K.; del Giorgio, P.A.; Bouvier, T.C.; Bronk, D.A.; Graham, W.M.; Ducklow, H.W. Jellyfish blooms result in a major microbial respiratory sink of carbon in marine systems. Proc. Natl. Acad. Sci. USA 2011, 108, 10225–10230. [Google Scholar] [CrossRef] [PubMed]
- Emadodin, I.; Reinsch, T.; Rotter, A.; Orlando-Bonaca, M.; Taube, F.; Javidpour, J. A perspective on the potential of using marine organic fertilizers for the sustainable management of coastal ecosystem services. Environ. Sustain. 2020. [Google Scholar] [CrossRef]
- Uye, S. Blooms of the giant jellyfish Nemopilema nomurai: A threat to the fisheries sustainability of the East Asian Marginal Seas. Plankton Benthos Res. 2008, 3, 125–131. [Google Scholar] [CrossRef]
- Cruz-Rivera, E.; El-Regal, M.A. A bloom of an edible scyphozoan jellyfish in the Red Sea. Mar. Biodivers. 2015, 46, 515–519. [Google Scholar] [CrossRef]
- Boero, F. Review of jellyfish blooms in the Mediterranean and Black Sea; Studies and Reviews; General Fisheries Commission for the Mediterranean; FAO: Rome, Italy, 2013; p. 53. [Google Scholar]
- Fukushi, K.; Ishio, N.; Tsujimoto, J.; Yokota, K.; Hamatake, T.; Sogabe, H.; Toriya, K.; Nimomiya, T. Preliminary Study on the Potential usefulness of Jellyfish fertilizer. J. Bull. Soc. Sea Water Sci. 2003, 2, 209–217. [Google Scholar]
- Hossain, S.T.; Sugimoto, H.; Asagi, N.; Araki, T.; Ueno, H.; Morokuma, M.; Kato, H. The use of desalinated-dried jellyfish and rice bran for controlling weeds and rice yield. J. Org. Syst. 2013, 8, 28–37. [Google Scholar]
- Hussein, O.S.; Sayed, R.M.; Saleh, O.I. Uses of jellyfish in pre-sowing seeds treatment and pest control. Am. J. Exp. Agric. 2015, 5, 60–69. [Google Scholar] [CrossRef]
- Pedersen, M.T.; Brewer, J.R.; Duelund, L.; Hansen, P.L. On the gastrophysics of jellyfish preparation. Int. J. Gastron. Food Sci. 2017, 9, 34–38. [Google Scholar] [CrossRef]
- Esechie, H.A.; Al-Saidi, A.; Al-Khanjari, S. Effect of sodium chloride salinity on seedling emergence in chickpea. J. Agron. Crop Sci. 2002, 188, 155–160. [Google Scholar] [CrossRef]
- Welch, B.L. The generalization of "Student′s" problem when several different population variances are involved. Biometrika 1947, 34, 28–35. [Google Scholar] [PubMed]
- Baath, G.S.; Shukla, M.K.; Bosland, P.W.; Steiner, R.L.; Walker, S.J. Irrigation water salinity influences at various growth stages of Capsicum annuum. Agric. Water Manag. 2017, 179, 246–253. [Google Scholar] [CrossRef]
- Killham, K. Soil Ecology; Cambridge University Press: Cambridge, UK, 1994; p. 242. [Google Scholar]
- Smith, J.L.; Doran, J.W. Measurement and use of pH and electrical conductivity for soil quality analysis. In Methods for assessing Soil Quality; Doran, J.W., Jones, A.J., Eds.; Soil Science Society of America Journal, SSSA: Madison, WI, USA, 1996; p. 49. [Google Scholar]
- Rawls, W.J.; Pachepsky, Y.A.; Ritchie, J.C.; Sobecki, T.M.; Bloodworth, H. Effect of soil organic carbon on soil water retention. Geoderma 2003, 116, 61–76. [Google Scholar] [CrossRef]
- Carter, O.G. The effect of chemical fertilizers on seedling establishment. Aust. J. Exp. Agric. Anim. Husb. 1967, 7, 174–180. [Google Scholar] [CrossRef]
- Aulakh, M.; Khera, T.; Doran, J. Mineralization and denitrification in upland, nearly saturated and flooded subtropical soil II. Effect of organic manures varying in N content and C:N ratio. Biol. Fertil. Soils 2000, 31, 168–174. [Google Scholar] [CrossRef]
| Elements in Dry Matter | Cyanea capillata % per Dry Mass | Aurelia aurita % per Dry Mass | ||
|---|---|---|---|---|
| Alcohol Dried | Oven Dried | Alcohol Dried | Oven Dried | |
| N | 4.4 | 2.9 | 7.8 | 0.7 |
| C | 15.5 | 10.5 | 27.3 | 3.1 |
| P | 0.22 | 0.8 | 1.0 | 0.2 |
| Ca | 0.75 | 0.77 | 0.75 | 0.84 |
| Mg | 2 | 1.8 | 1.0 | 1.6 |
| Na | 17.8 | 19.5 | 9.2 | 33.4 |
| K | 0.16 | 1.2 | 0.44 | 0.96 |
| Mn | <0.01 | <0.01 | <0.01 | <0.01 |
| Cu | <0.01 | <0.01 | <0.01 | <0.01 |
| Zn | 0.01 | 0.02 | 0.04 | <0.01 |
| C:N | 3.5 | 3.6 | 3.5 | 4.4 |
| Nr. | Treatments | Abbreviation |
|---|---|---|
| 1 | Distilled water (Control) | Control |
| 2 | Aurelia aurita (Oven-dried) | Aur (Ov) |
| 3 | Aurelia aurita (Alcohol-dried) | Aur (Alc) |
| 4 | Cyanea capillata (Oven-dried) | Cya (Ov) |
| 5 | Cyanea capillata (Alcohol-dried) | Cya (Alc) |
| 6 | Aurelia aurita and Cyanea capillata (Oven-dried) | Aur + Cya (Ov) |
| 7 | Aurelia aurita and Cyanea capillata (Alcohol-dried) | Aur + Cya (Alc) |
| 8 | Calcium ammonium nitrate | CAN |
| Treatments | pH | EC [µS/cm] |
|---|---|---|
| Control | 6.1 | 0 |
| Aurelia (Ov) | 7.5 | 19.00 |
| Aurelia (Alc) | 7.42 | 5.06 |
| Cyanea (Ov) | 6.6 | 13.33 |
| Cyanea (Alc) | 6.83 | 2.89 |
| Aur + Cya (Ov) | 6.9 | 14.8 |
| Aur + Cya (Alc) | 7.2 | 4.23 |
| CAN | 7.15 | 15.21 |
| Nr. | Treatments | Abbreviation |
|---|---|---|
| 1 | Distilled water (Control) | Control |
| 2 | Aurelia aurita (Oven-dried) | Aur (Ov) |
| 3 | Aurelia aurita (Alcohol-dried) | Aur (Alc) |
| 4 | Cyanea capillata (Oven-dried) | Cya (Ov) |
| 5 | Cyanea capillata (Alcohol-dried) | Cya (Alc) |
| 6 | Calcium ammonium nitrate | CAN |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Emadodin, I.; Reinsch, T.; Ockens, R.-R.; Taube, F. Assessing the Potential of Jellyfish as an Organic Soil Amendment to Enhance Seed Germination and Seedling Establishment in Sand Dune Restoration. Agronomy 2020, 10, 863. https://doi.org/10.3390/agronomy10060863
Emadodin I, Reinsch T, Ockens R-R, Taube F. Assessing the Potential of Jellyfish as an Organic Soil Amendment to Enhance Seed Germination and Seedling Establishment in Sand Dune Restoration. Agronomy. 2020; 10(6):863. https://doi.org/10.3390/agronomy10060863
Chicago/Turabian StyleEmadodin, Iraj, Thorsten Reinsch, Raffaele-Romeo Ockens, and Friedhelm Taube. 2020. "Assessing the Potential of Jellyfish as an Organic Soil Amendment to Enhance Seed Germination and Seedling Establishment in Sand Dune Restoration" Agronomy 10, no. 6: 863. https://doi.org/10.3390/agronomy10060863
APA StyleEmadodin, I., Reinsch, T., Ockens, R.-R., & Taube, F. (2020). Assessing the Potential of Jellyfish as an Organic Soil Amendment to Enhance Seed Germination and Seedling Establishment in Sand Dune Restoration. Agronomy, 10(6), 863. https://doi.org/10.3390/agronomy10060863






