Abstract
The interest in using plant-growth-promoting bacteria (PGPB) as biopesticides is significantly growing as a result of the discovery of new properties of certain beneficial microbes in protecting agricultural crops. While several rhizobial species have been widely exploited for their ability to optimize plant use of environmental resources, now the focus is shifted to species that are additionally capable of improving plant health and conferring resistance to abiotic stress and deleterious biotic agents. In some cases, PGPB species may directly act against plant pathogens and parasites through a variety of mechanisms, including competition, protective biofilm formation, and the release of bioactive compounds. The use of this type of bacteria is in line with the principles of ecosustainability and integrated pest management, including the reduction of employing chemical pesticides. Several strains of Bacillus, Paenibacillus, Brevibacillus, Pseudomonas, Serratia, Burkholderia, and Streptomyces species have been the subject of specific studies in this direction and are under evaluation for further development for their use in biological control. Accordingly, specific case studies are presented and discussed.
1. Introduction
The evolving interactions between microorganisms and plants have led to the establishment of intimate relationships supporting plant growth and promoting their health and access to nutrients from the external environment. In this context, a role of primary importance is played by several bacterial species which, in particular, establish a direct relationship with the root system. Among these, several rhizobacteria have evolved the ability to perform biochemical reactions in the rhizosphere, converting nutrients present in the external environment into forms directly usable by plants. Nutrients in the soil are often found in forms not easily available to plants, but optimization of their intake may result from chemical conversions operated by soil microbes. This includes vital elements like iron, phosphorus, and nitrogen, whose soil bioavailability can thus be improved [1]. Accordingly, siderophores produced by special bacteria solubilize iron, while immobilized phosphorus can be solubilized and mineralized by mycorrhizae and some soil bacteria [2]. Nitrogen is fixed by the enzymatic action of rhizobia, establishing a symbiotic relationship with leguminous plants roots [3] and other bacteria, such as Azospirillum, Herbaspirillum, Azotobacter, and Agrobacterium [4]. In addition to supporting the optimization of the use of soil resources like nutrients and water, soil bacteria may further promote plant growth by interacting with plant physiology through the production of phytohormones involved in plant response to stress [5].
These microbes therefore play a supportive role in fostering plant health, both by assisting the nutrient acquisition process and through indirect mechanisms that protect plants from potential pathogens and parasites (Figure 1). Among the protection mechanisms are both the induction of resistance towards the action of biotic adversities and the direct action of inhibition towards them [4].
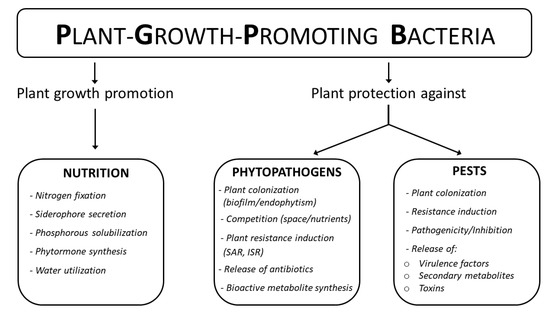
Figure 1.
Representative scheme of the multiple utilities of some soil-dwelling bacterial species and their main mechanisms of action.
Beneficial bacteria and their plant hosts have developed biochemical adaptations, including a key role of microbial molecules in regulating interactions within the ecosystem. A growing scientific and industrial interest is fostering studies aiming at understanding these mechanisms, with a view to identifying microbial strains with the dual aptitude of plant growth promoters and direct or indirect biological control agents. Such an approach aligns with the modern integrated protection management (IPM) principles of reducing the release of chemical residues in agroecosystems [6].
The present review focuses on the potential of some plant-growth-promoting bacteria (PGPB) species as biological control agents against noxious insects, with some mention of other invertebrate pests, such as nematodes. The purpose is to give a concise overview of a rapidly evolving and still understudied subject, whose interest is significantly growing for innovative and ecosustainable application opportunities in agriculture.
2. Potential of Plant-Growth-Promoting Bacterial Species as Invertebrate Pest Control Agents
Several species included among plant-growth-promoting bacteria for the abovementioned mechanisms have been shown to have additional antagonistic properties against plant pathogens and invertebrate pests, such as insects and nematodes.
Common mechanisms by which soil bacteria protect plants include the competition for space and nutrients with phytopathogens, often associated with the formation of biofilm on the root surface [7], and more rarely with the penetration of the root by endophytic species [8]. However, some plant-growth-promoting bacterial species may also be able to trigger plant immune systems by induction mechanisms, leading to variable levels of systemic resistance [9]. In addition to these indirect mechanisms, plant-associated bacteria can exert a direct antimicrobial action against phytopathogens, which may depend on the production of antimicrobial compounds, enzymes, and toxins. A representative list of bioactive compounds produced by bacteria against plant pathogens is shown in Table 1 [10].

Table 1.
Representative bioactive compounds produced by different plant-growth-promoting bacteria (PGPB) species against phytopathogens.
Certain bacteria that are recognized as plant growth promoters may also play a role in protecting plants against invertebrate pests through diverse mechanisms, including pathogenesis and the production of a variety of insecticidal toxins, virulence factors, and metabolites (Table 2). The mode of action is complex and, in many cases, not fully clarified. However, recent studies are highlighting the involvement of diverse bioactive compounds. Among them are specific protein toxins often acting in the midgut, enzymes like chitinases and proteases degrading external barriers or the peritrophic matrix in the midgut, and several secondary metabolites targeting different tissues. This is the case in species in the genera Bacillus, Brevibacillus, Paenibacillus, Pseudomonas, Serratia, Burkholderia, and Streptomyces. Accordingly, specific case studies are here presented and discussed.

Table 2.
Representative bioactive compounds produced by some PGPB species against invertebrate pests.
3. Case Studies
3.1. Bacilli
3.1.1. Bacillus firmus
The plant growth promoter Bacillus firmus (Bredemann and Werner) was found to show a significant biocontainment effect on soil nematodes in the Meloidogyne genus in experiments with cucumber and tomato [11]. Bionematicidal formulations containing the bacterium have consequently been developed and commercialized. The promising results obtained in laboratory experiments were confirmed by field trials carried out on tomato plants employing some B. firmus strains against the root-knot nematode Meloidogyne incognita (Kofold and White) (Chitwood) (Tylenchida: Heteroderidae) [12]. Serine proteases (Sep1) with nematicidal activity were found to be produced by specific B. firmus strains [13]. The involvement of this enzyme was demonstrated in studies employing transformed Escherichia coli expressing the Sep1 protein in bioassays with nematodes. This extracellular protein was observed to act on the nematode physical barrier, degrading its surface and mouth parts covered by the cuticle. In addition to this enzymatic action by contact, there is an effect by ingestion that determines damages to the intestinal epithelial tissues [13]. Although it is thought that there are also other not yet discovered virulence factors produced by the bacterium, the enzymatic action is expected to be effective against different species of nematodes and other invertebrates, such as insects, which have in common the characteristic of having a body covered by a cuticle.
3.1.2. Bacillus subtilis
Though it is more famous for its plant growth promotion and antimicrobial properties [14], Bacillus subtilis (Ehrenberg) (Cohn) has several strains that show bioinsecticidal potential, often associated with the production and release of specific bioactive compounds in the rhizosphere.
Part of the insecticidal activity derives from the production of biosurfactants causing histopathological changes in the midgut of treated insects [15]. Ultrastructural changes observed in experiments with Ephestia kuehniella (Zeller) (Lepidoptera: Pyralidae) larvae in the midgut were similar to those caused by Cry toxins produced by Bacillus thuringiensis (Berliner) and included loosening of the columnar cells, cytoplasm vacuolization, microvilli damage, and the release of cytoplasm into the lumen [16].
Some strains are also able to produce extracellular chitinases acting in the gut of lepidopteran larvae (i.e., Spodoptera spp.). Experiments with Spodoptera litura (Fabricius) (Lepidoptera: Noctuidae) showed that, after ingestion, chitinases decreased the activities of gut enzymes and caused alterations to the peritrophic membrane and epithelial cells [17].
3.1.3. Brevibacillus laterosporus
This ubiquitous bacterium is easily found in soil and plant rhizosphere, where, in addition to playing a role in supporting plant nutrition, it can exert a protective action in favor of plant health [18]. The effects against plant phytopathogens are related to the high antimicrobial potential associated with the bioactive molecules Brevibacillus laterosporus (Laubach) produces. These include antibiotics and several enzymes like chitinases and various peptides [19,20]. The insecticidal properties against a wide range of Coleoptera, Lepidoptera, and Diptera have been observed in several strains of this entomopathogenic species and were found to be related with well-conserved genomic traits [21]. Among major insecticidal toxins and virulence factors, there are Cry toxin homologues [22], proteins associated with the typical canoe-shaped parasporal body [23], and enzymes like chitinases and proteases [24]. The action of these enzymes is exerted on several invertebrate pests, including insects, nematodes, and molluscs, through the degradation of external barriers consisting of tissues containing chitin and various protein components. In addition, the enzymatic action may result in the degradation of the intestinal peritrophic membrane. This bacterium normally acts by ingestion through its protein toxins, determining the disruption of the midgut epithelial cells, before insect paralysis and death [25]. The generally recognized safety for nontarget insects, including honeybees [26], hymenopteran parasitoids [27], and lacewings [28], makes this species particularly promising as a biocontrol agent.
3.1.4. Paenibacillus spp.
The genus Paenibacillus includes a variety of species involved in crop growth promotion through different mechanisms, such as the fixation of nitrogen, the solubilization of phosphate, the release of siderophores, and the production of phytohormones [29].
Several Paenibacillus species show significant capability to protect plants against phytopathogens and pests. Among these, a considerable number of studies have been conducted with Paenibacillus polymyxa, and some other species including Paenibacillus alvei (Cheshire and Cheyne) (Ash et al.) [30], Paenibacillus dendritiformis (Ash et al.) [31], Paenibacillus lentimorbus (Dutky) (Pettersson et al.) [32], Paenibacillus macerans (Schardinger) (Ash et al.) [33], and Paenibacillus thiaminolyticus (Nakamura) (Shida et al.) [34]. The mechanisms of action comprise the induction of systemic resistance and the production of a variety of antimicrobials, like polymyxin and insecticidal compounds [35].
The specificity by which species of the genus Paenibacillus establish pathogenic relationships with insects is well represented by Paenibacillus larvae subsp. Larvae, the etiological agent of the American Foulbrood (AFB), a disease affecting the honeybee [36]. Other insect pathogenic species include the milky disease causative agents Paenibacillus popilliae (Dutky) (Pettersson et al.) and P. lentimorbus, acting against coleopteran pests as a result of a combination of a toxin-mediated process and vegetative-cell-caused septicemia [37].
3.2. Gammaproteobacteria
3.2.1. Pseudomonas spp.
Many plant-associated pseudomonads play a significant role in promoting plant growth promotion and crop protection against phytopathogens and invertebrate pests, which has important practical implications to enhance crop productivity [38]. Among the different species in this genus, at least Pseudomonas putida (Trevisan) (Migula), Pseudomonas aeruginosa (Schroeter) (Migula), and Pseudomonas fluorescens (Flügge) (Migula) deserve to be mentioned. Numerous isolates belonging to these species have been the subject of in-depth studies on the mechanisms of beneficial action in favor of plants [39].
Of particular scientific and industrial interest is the P. fluorescens species group that is characterized by the production of fluorescent siderophores [40]. This group includes the recently identified novel species Pseudomonas protegens (Ramette et al.), whose properties range from root colonization to growth stimulation, competition, and antibiosis against phytopathogens [41]. Fluorescent pseudomonad biofertilization capabilities relate to nitrogen fixation, phosphate mobilization, and the secretion of siderophores improving iron availability in the soil [14]. In addition, some plant-colonizing species may induce systemic resistance against pathogenic actions [42]. On the other hand, root colonization is, per se, a mechanism of protection due to the competition for space and nutrients these bacteria establish with soil phytopathogens. A more direct action of plant protection is related to the release of bioactive compounds, such as hydrogen cyanide (HCN), pyoluteorin, toxoflavin, orfamide A, 2,4-diacetylphloroglucinol (DAPG), pyoluteorin, pyrrolnitrin, toxoflavin, orfamide A, and several enzymes including chitinases and various proteases [43]. Some of these secondary metabolites are also implied in inhibitory or toxic actions against invertebrate pests. Specific insecticidal toxin genes were also found in the genome of some of the species in the P. fluorescens group, such as P. protegens that produces a fluorescent insecticidal toxin (Fit) secretion system, including a high molecular weight protein, FitD, whose toxicity against insects was demonstrated by injection and ingestion assays, even if the mechanism of interaction with susceptible insect cells is still not completely understood [44].
Among other members of the Pseudomonas genus active against invertebrates, Pseudomonas entomophila (Mulet et al.) is a species acting by ingestion on susceptible insects and causing histopathological changes in the midgut epithelium, as a result of a toxin-mediated process [45].
3.2.2. Serratia spp.
Several Serratia species are implied in a wide diversity of mutualistic and antagonistic associations favouring plant growth and health [46]. Some species, like Serratia marcescens (Bizio) [47] and Serratia plymuthica (Lehmann and Neumann) (Breed et al.) [48], have found interest as biological control agents against plant phytopathogens. On the other hand, the antagonistic relationship of this bacterial genus with invertebrates is well documented [49,50]. The biocontrol potential relies on the production of diverse virulence factors and, in particular, on toxin complexes (Tc) homologous to those produced by the entomopathogenic nematode symbiotic bacteria belonging to the genera Photorhabdus and Xenorhabdus. This is the case with Sep proteins produced by Serratia entomophila (Grimont et al.), affecting the grass grub Costelytra zealandica (White) (Coleoptera: Scarabaeidae) [51]. The sep gene cluster includes three genes (sepA, sepB, and sepC) expressed in the insect gut and causing its clearance and consequent bacterial septicaemia. A second gene cluster (afp) is responsible for an antifeeding effect. Other insect pathogenic compounds produced by Serratia strains include a variety of extracellular enzymes, such as chitinases, proteases, and lipases [52]. Among other virulence factors, there is a serralysin metalloprotease secreted by S. marcescens, which is involved in insect cellular immunity suppression [53].
3.3. Betaproteobacteria
Burkholderia spp.
The Burkholderia genus includes a wide range of species with high biology diversity, some of which are involved in plant growth promotion and biocontrol. Among them, species in the Burkholderia cepacia complex have been exploited in agriculture for their beneficial properties [54]. On the other hand, some Burkholderia species are pathogenic to plants [55].
Most of the known relationships between invertebrates and Burkholderia spp. are mutualistic and often involve symbiotic bacteria associated with the insect gut [56] or may play a role in insect development [57]. Recently isolated Burkholderia strains have been reported to produce bioactive compounds with potential against invertebrates [58], and an isolate of the newly discovered species Burkholderia rinojensis sp. nov. was found to act against insect pests and mites, which led to the development of new biopesticidal products [59].
3.4. Actinobacteria
Streptomyces spp.
Streptomyces species represent a rich resource of secondary metabolites that find use as commercially available antimicrobial and antiparasitic active substances. The association of some streptomycetes with plants may also lead to plant growth improvement and protection against pests through a variety of mechanisms [60]. These actinobacteria typically colonize the rhizosphere and rhizoplane, influencing the microbial community composition in the soil–root system [61]. In certain cases, they behave as endophytes establishing a more intimate relationship with plant tissues [62]. The mechanisms of plant growth promotion include biofertilization and biostimulation effects, while bioprotection relies on competition mechanisms and the production of secondary metabolites [60]. Among the latter, there are several compounds with insecticidal properties, such as antimycin A [63], flavensomycin [64], macrotetralides [65], piericidins [66], and prasinons [67]. Very effective and commercially successful metabolites produced by streptomycetes are represented by avermectins, macrocyclic lactone derivatives affecting the insect nervous system. Avermectins interact with the receptors of gamma-aminobutyric acid (GABA), generating a cascade of events leading to neurotransmission inhibition and consequent neuromuscular paralysis and insect death [68].
4. Industrial Interest and Future Prospects
The need to feed a constantly increasing human population on earth is accompanied by an increment of agricultural production. Accordingly, the whole global market for pesticides is growing at an estimated compound annual growth rate (CAGR) of 5.3% [69]. The biopesticide market segment, on the other hand, is growing at a significantly higher rate of 15.99% [70], as a result of the need to reduce chemical inputs into the agroecosystem. This includes different product types, like bioinsecticides, biofungicides, and bionematicides based on diverse active substances deriving from microbials, plant extracts, and beneficial arthropods. Microbial products are mostly obtained from fungi, bacteria, viruses, and nematodes [2].
Another area of application of microorganisms in agriculture is represented by plant growth promoters, such as biofertilizers, which may include species having biocontrol potential against pests, and which therefore overlap with the biopesticide segment. This market segment is worth nearly US$2 billion and is estimated to grow at a 14.3% rate [71].
All these positive trends align with a current legislative framework requiring the use of environmentally safer bioproducts in respect to conventional synthetic substances for crop protection and nutrition.
Although biofertilizers and microbiological control agents are treated separately by regulations concerning the pre-market authorization of new products, some of the plant-growth-promoting bacteria, including those mentioned in this review, associate a nutritional role with plant protection functions. Such a feature is of considerable practical utility and is attracting the interest of scientists, industry, and end-users [10]. While the use and the commercialization of certain PGPB species, such as strains of Azospirillum, Azobacter, Bacillus, Burkholderia, Enterobacter, Pseudomonas, Serratia, and Xanthomonas, are well established, other strains and species are emerging because of their dual aptitude to support the use of nutritional resources in the soil and protect plants from pathogens and pests. The possibility of exploiting this type of microbial products allows for multiple functionality in favor of cultivation, which translates into practical and economic advantages that are in line with the principles of integrated agroecosystem management, limiting inputs from outside. From the point of view of the market, a single product that meets different crop needs has a significant competitive advantage. For these reasons, research and development in this field are experiencing a significantly growing interest. While most plant-associated microbials are generally regarded as safe, safety evaluation of new strains is another important issue that deserves specific investigations.
5. Conclusions
The use of PGPB species has historically represented an important resource to naturally support plant nutrition, and this has led to the development of a specific research area and to applications in agriculture. More recent advancements in knowledge of the biology of a variety of soil-dwelling species is leading to an increased interest for their potential application for both plant growth promotion and protection against pathogens and parasites. Among the latter, several invertebrate pests, especially insects and nematodes, can be inhibited through direct and indirect mechanisms, thus helping to maintain crop health and productivity. Studies aiming at screening bacterial strains with such properties not only increase our understanding of the ecological significance of the bacterial community associated with plant roots but also create concrete opportunities of application in agricultural contexts, in line with the principles of integrated pest management and ecosustainability.
Funding
This research was funded by the Italian Ministry of Education, University and Research (MIUR) (PRIN project 2015 “BIOPIC”, 2015BABFCF) and by the University of Sassari (Fondo di Ateneo per la Ricerca).
Conflicts of Interest
The author declares no conflicts of interest.
References
- Gamalero, E.; Glick, B.R. Mechanisms used by plant growth-promoting bacteria. In Bacteria in Agrobiology: Plant Nutrient Management; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 17–46. [Google Scholar]
- Bolan, N.S. A critical review on the role of mycorrhizal fungi in the uptake of phosphorus by plants. Plant Soil 1991, 134, 189–207. [Google Scholar] [CrossRef]
- Somasegaran, P.; Hoben, H.J. Handbook for Rhizobia: Methods in Legume-Rhizobium Technology; Springer Science & Business Media: New York, NY, USA, 2012. [Google Scholar]
- Glick, B.R. Plant growth-promoting bacteria: Mechanisms and applications. Scientifica 2012, 2012, 963401. [Google Scholar] [CrossRef]
- Glick, B.R. Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol. Res. 2014, 169, 30–39. [Google Scholar] [CrossRef]
- Ruiu, L. Microbial Biopesticides in Agroecosystems. Agronomy 2018, 8, 235. [Google Scholar] [CrossRef]
- Kloepper, J.W.; Rodriguez-Kabana, R.; Zehnder, A.W.; Murphy, J.F.; Sikora, E.; Fernandez, C. Plant root-bacterial interactions in biological control of soilborne diseases and potential extension to systemic and foliar diseases. Australas. Plant Pathol. 1999, 28, 21–26. [Google Scholar] [CrossRef]
- Gray, E.J.; Smith, D.L. Intracellular and extracellular PGPR: Commonalities and distinctions in the plant–bacterium signaling processes. Soil Boil. Biochem. 2005, 37, 395–412. [Google Scholar] [CrossRef]
- Pieterse, C.M.; Zamioudis, C.; Berendsen, R.L.; Weller, D.M.; Van Wees, S.C.; Bakker, P.A. Induced systemic resistance by beneficial microbes. Annu. Rev. Phytopathol. 2014, 52, 347–375. [Google Scholar] [CrossRef]
- Ruiu, L. Plant growth-promoting bacteria (PGPBs) as biocontrol agents against invertebrate pests. In Biopesticides for Sustainable Agriculture; Birch, N., Glare, T., Eds.; Burleigh Dodds Science Publishing Limited: Cambridge, UK, 2020; pp. 151–166. [Google Scholar]
- Keren-Zur, M.; Antonov, J.; Bercovitz, A.; Feldman, K.; Husid, A.; Kenan, G.; Marcov, N.; Rebhun, M. Bacillus firmus formulations for the safe control of root-knot nematodes. In Proceedings of the BCPC Conference Pests and Diseases, Brighton, UK, 13–16 November 2000; British Crop Protection Council: Hampshire, UK, 2000; Volume 1, pp. 47–52. [Google Scholar]
- Terefe, M.; Tefera, T.; Sakhuja, P.K. Effect of a formulation of Bacillus firmus on root-knot nematode Meloidogyne incognita infestation and the growth of tomato plants in the greenhouse and nursery. J. Invertebr. Pathol. 2009, 100, 94–99. [Google Scholar] [CrossRef]
- Geng, C.; Nie, X.; Tang, Z.; Zhang, Y.; Lin, J.; Sun, M.; Peng, D. A novel serine protease, Sep1, from Bacillus firmus DS-1 has nematicidal activity and degrades multiple intestinal-associated nematode proteins. Sci. Rep. 2016, 6, 25012. [Google Scholar] [CrossRef]
- Sivasakthi, S.; Usharani, G.; Saranraj, P. Biocontrol potentiality of plant growth promoting bacteria (PGPR)-Pseudomonas fluorescens and Bacillus subtilis: A review. Afr. J. Agric. Res. 2014, 9, 1265–1277. [Google Scholar]
- Assie, L.K.; Deleu, M.; Arnaud, L.; Paquot, M.; Thonart, P.; Gaspar, C.H.; Haubruge, E. Insecticide activity of surfactins and iturins from a biopesticide Bacillus subtilis Cohn (S499 strain). Mededelingen (Rijksuniversiteit te Gent. Fakulteit van de Landbouwkundige en Toegepaste Biologische Wetenschappen) 2002, 67, 647–655. [Google Scholar]
- Ghribi, D.; Elleuch, M.; Abdelkefi-Mesrati, L.; Boukadi, H.; Ellouze-Chaabouni, S. Histopathological effects of Bacillus subtilis SPB1 biosurfactant in the midgut of Ephestia kuehniella (Lepidoptera: Pyralidae) and improvement of its insecticidal efficiency. J. Plant Dis. Protect. 2012, 119, 24–29. [Google Scholar] [CrossRef]
- Chandrasekaran, R.; Revathi, K.; Thanigaivel, A.; Kirubakaran, S.A.; Senthil-Nathan, S. Bacillus subtilis chitinase identified by matrix-assisted laser desorption/ionization time-of flight/time of flight mass spectrometry has insecticidal activity against Spodoptera litura Fab. Pestic. Biochem. Physiol. 2014, 116, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ruiu, L. Brevibacillus laterosporus, a pathogen of invertebrates and a broad-spectrum antimicrobial species. Insects 2013, 4, 476–492. [Google Scholar] [CrossRef] [PubMed]
- Prasanna, L.; Eijsink, V.G.; Meadow, R.; Gåseidnes, S. A novel strain of Brevibacillus laterosporus produces chitinases that contribute to its biocontrol potential. Appl. Microbiol. Biotechnol. 2013, 97, 1601–1611. [Google Scholar] [CrossRef]
- Zhao, J.; Guo, L.; Zeng, H.; Yang, X.; Yuan, J.; Shi, H.; Xiong, Y.; Chen, M.; Han, L.; Qiu, D. Purification and characterization of a novel antimicrobial peptide from Brevibacillus laterosporus strain A60. Peptides 2012, 33, 206–211. [Google Scholar] [CrossRef]
- Glare, T.R.; Durrant, A.; Berry, C.; Palma, L.; Ormskirk, M.M.; Cox, M.P. Phylogenetic determinants of toxin gene distribution in genomes of Brevibacillus laterosporus. Genomics 2020, 112, 1042–1053. [Google Scholar] [CrossRef]
- Bowen, D.J.; Chay, C.A.; Flasinski, S.; Yin, Y. Novel Insect Inhibitory Proteins. Monsanto Technology LLC. U.S. Patent Application No. A120170044568, 16 February 2017. [Google Scholar]
- Marche, M.G.; Mura, M.E.; Falchi, G.; Ruiu, L. Spore surface proteins of Brevibacillus laterosporus are involved in insect pathogenesis. Sci. Rep. 2017, 7, 43805. [Google Scholar] [CrossRef]
- Marche, M.G.; Camiolo, S.; Porceddu, A.; Ruiu, L. Survey of Brevibacillus laterosporus insecticidal protein genes and virulence factors. J. Invertebr. Pathol. 2018, 155, 38–43. [Google Scholar] [CrossRef]
- Ruiu, L.; Satta, A.; Floris, I. Observations on house fly larvae midgut ultrastructure after Brevibacillus laterosporus ingestion. J. Invertebr. Pathol. 2012, 111, 211–216. [Google Scholar] [CrossRef]
- Ruiu, L.; Satta, A.; Floris, I. Susceptibility of the house fly pupal parasitoid Muscidifurax raptor (Hymenoptera: Pteromalidae) to the entomopathogenic bacteria Bacillus thuringiensis and Brevibacillus laterosporus. Biol. Control 2007, 43, 188–194. [Google Scholar] [CrossRef]
- Marche, M.G.; Mura, M.E.; Ruiu, L. Brevibacillus laterosporus inside the insect body: Beneficial resident or pathogenic outsider? J. Invertebr. Pathol. 2016, 137, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Ruiu, L.; Lazzeri, A.M.; Nuvoli, M.T.; Floris, I.; Satta, A. Safety evaluation of the entomopathogenic bacterium Brevibacillus laterosporus for the green lacewing Chrysoperla agilis (Neuroptera: Chrysopidae). J. Invertebr. Pathol. 2020, 169, 107281. [Google Scholar] [CrossRef] [PubMed]
- Grady, E.N.; MacDonald, J.; Liu, L.; Richman, A.; Yuan, Z.C. Current knowledge and perspectives of Paenibacillus: A review. Microb. Cell Fact. 2016, 15, 203. [Google Scholar] [CrossRef] [PubMed]
- Tjamos, S.E.; Flemetakis, E.; Paplomatas, E.J.; Katinakis, P. Induction of resistance to Verticillium dahliae in Arabidopsis thaliana by the biocontrol agent K-165 and pathogenesis-related proteins gene expression. Mol. Plant Microbe Interact. 2005, 18, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Lapidot, D.; Dror, R.; Vered, E.; Mishli, O.; Levy, D.; Helman, Y. Disease protection and growth promotion of potatoes (Solanum tuberosum L.) by Paenibacillus dendritiformis. Plant Pathol. 2015, 64, 545–551. [Google Scholar] [CrossRef]
- Montealegre, J.R.; Herrera, R.; Velásquez, J.C.; Silva, P.; Besoaín, X.; Pérez, L.M. Biocontrol of root and crown rot in tomatoes under greenhouse conditions using Trichoderma harzianum and Paenibacillus lentimorbus: Additional effect of solarization. Electron. J. Biotechnol. 2005, 8. [Google Scholar] [CrossRef]
- Gardener, B.B.M. Ecology of Bacillus and Paenibacillus spp. in agricultural systems. Phytopathology 2004, 94, 1252–1258. [Google Scholar] [CrossRef]
- Huang, E.; Yousef, A.E. The lipopeptide antibiotic paenibacterin binds to the bacterial outer membrane and exerts bactericidal activity through cytoplasmic membrane damage. Appl. Environ. Microbiol. 2014, 80, 2700–2704. [Google Scholar] [CrossRef]
- Zhao, L.J.; Yang, X.N.; Li, X.Y.; Wei, M.U.; Feng, L.I.U. Antifungal, insecticidal and herbicidal properties of volatile components from Paenibacillus polymyxa strain BMP-11. Agric. Sci. China 2011, 10, 728–736. [Google Scholar] [CrossRef]
- Davidson, E.A. Ultrastructures of American foulbrood disease pathogenesis in larvae of the worker honey bee Apis mellifera. J. Invertebr. Pathol. 1973, 21, 53–61. [Google Scholar] [CrossRef]
- Zhang, J.; Hodgman, T.C.; Krieger, L.; Schnetter, W.; Schairer, H.U. Cloning and analysis of the cry gene from Bacillus popilliae. J. Bacteriol. 1997, 179, 4336–4341. [Google Scholar] [CrossRef] [PubMed]
- Lucy, M.; Reed, E.; Glick, B.R. Applications of free living plant growth-promoting rhizobacteria. Antonie van Leeuwenhoek 2004, 86, 1–25. [Google Scholar] [CrossRef]
- Dueholm, M.S.; Søndergaard, M.T.; Nilsson, M.; Christiansen, G.; Stensballe, A.; Overgaard, M.T.; Givskov, M.; Tolker-Nielsen, T.; Otzen, D.E.; Nielsen, P.H. Expression of Fap amyloids in Pseudomonas aeruginosa, P. fluorescens, and P. putida results in aggregation and increased biofilm formation. Microbiologyopen 2013, 2, 365–382. [Google Scholar] [CrossRef] [PubMed]
- Raaijmakers, J.M.; Paulitz, T.C.; Steinberg, C.; Alabouvette, C.; Moënne-Loccoz, Y. The rhizosphere: A playground and battlefield for soilborne pathogens and beneficial microorganisms. Plant Soil 2009, 321, 341–361. [Google Scholar] [CrossRef]
- Ramette, A.; Frapolli, M.; Saux, M.F.-L.; Gruffaz, C.; Meyer, J.M.; Défago, G.; Sutra, L.; Moënne-Loccoz, Y. Pseudomonas protegens sp. nov., widespread plant-protecting bacteria producing the biocontrol compounds 2, 4-diacetylphloroglucinol and pyoluteorin. Syst. Appl. Microbiol. 2011, 34, 180–188. [Google Scholar] [CrossRef]
- Ramamoorthy, V.; Viswanathan, R.; Raguchander, T.; Prakasam, V.; Samiyappan, R. Induction of systemic resistance by plant growth promoting rhizobacteria in crop plants against pests and diseases. Crop Prot. 2001, 20, 1–11. [Google Scholar] [CrossRef]
- Philmus, B.; Shaffer, B.T.; Kidarsa, T.A.; Yan, Q.; Raaijmakers, J.M.; Begley, T.P.; Loper, J.E. Investigations into the biosynthesis, regulation, and self-resistance of toxoflavin in Pseudomonas protegens Pf-5. ChemBioChem 2015, 16, 1782–1790. [Google Scholar] [CrossRef]
- Flury, P.; Aellen, N.; Ruffner, B.; Péchy-Tarr, M.; Fataar, S.; Metla, Z.; Dominguez-Ferreras, A.; Bloemberg, G.; Frey, J.; Goesmann, A.; et al. Insect pathogenicity in plant-beneficial pseudomonads: Phylogenetic distribution and comparative genomics. ISME J. 2016, 10, 2527–2542. [Google Scholar] [CrossRef]
- Vodovar, N.; Vallenet, D.; Cruveiller, S.; Rouy, Z.; Barbe, V.; Acosta, C.; Cattolico, L.; Jubin, C.; Lajus, A.; Segurens, B.; et al. Complete genome sequence of the entomopathogenic and metabolically versatile soil bacterium Pseudomonas entomophila. Nat. Biotechnol. 2006, 24, 673–679. [Google Scholar] [CrossRef]
- Petersen, L.M.; Tisa, L.S. Friend or foe? A review of the mechanisms that drive Serratia towards diverse lifestyles. Can. J. Microbiol. 2013, 59, 627–640. [Google Scholar] [CrossRef] [PubMed]
- Lavania, M.; Chauhan, P.S.; Chauhan, S.V.S.; Singh, H.B.; Nautiyal, C.S. Induction of plant defense enzymes and phenolics by treatment with plant growth–promoting rhizobacteria Serratia marcescens NBRI1213. Curr. Microbiol. 2006, 52, 363–368. [Google Scholar] [CrossRef] [PubMed]
- De Vleesschauwer, D.; Höfte, M. Using Serratia plymuthica to control fungal pathogens of plants. CAB Rev. 2003, 2, 046. [Google Scholar] [CrossRef]
- Abebe, E.; Abebe-Akele, F.; Morrison, J. An insect pathogenic symbiosis between a Caenorhabditis and Serratia. Virulence 2011, 2, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.-X.; Yang, S.-Y.; Xu, M.-X.; Sun, J.; Liu, H.; Liu, J.R.; Liu, H.; Kan, F.; Sun, J.; Lai, R.; et al. Serratia nematodiphila sp. nov., associated symbiotically with the entomopathogenic nematode Heterorhabditidoides chongmingensis (Rhabditida: Rhabditidae). Int. J. Syst. Evol. Microbiol. 2000, 59, 1603–1608. [Google Scholar] [CrossRef]
- Tan, B.; Jackson, T.A.; Hurst, M.R. Virulence of Serratia strains against Costelytra zealandica. Appl. Environ. Microbiol. 2006, 72, 6417–6418. [Google Scholar] [CrossRef]
- Kwak, Y.; Khan, A.R.; Shin, J.H. Genome sequence of Serratia nematodiphila DSM 21420(T), a symbiotic bacterium from entomopathogenic nematode. J. Biotechnol. 2015, 193, 1–2. [Google Scholar] [CrossRef]
- Ishii, K.; Adachi, T.; Hamamoto, H.; Sekimizu, K. Serratia marcescens suppresses host cellular immunity via the production of an adhesion-inhibitory factor against immunosurveillance cells. J. Biol. Chem. 2014, 289, 5876–5888. [Google Scholar] [CrossRef]
- Coenye, T.; Vandamme, P. Diversity and significance of Burkholderia species occupying diverse ecological niches. Environ. Microbiol. 2003, 5, 719–729. [Google Scholar] [CrossRef]
- Stoyanova, M.; Pavlina, I.; Moncheva, P.; Bogatzevska, N. Biodiversity and incidence of Burkholderia species. Biotechnol. Biotechnol. Equip. 2007, 21, 306–310. [Google Scholar] [CrossRef]
- Kim, J.K.; Kim, N.H.; Jang, H.A.; Kikuchi, Y.; Kim, C.H.; Fukatsu, T.; Lee, B.L. Specific midgut region controlling the symbiont population in an insect-microbe gut symbiotic association. Appl. Environ. Microbiol. 2013, 79, 7229–7233. [Google Scholar] [CrossRef] [PubMed]
- Kil, Y.J.; Seo, M.J.; Kang, D.K.; Oh, S.N.; Cho, H.S.; Youn, Y.N.; Yasunaga-Aoki, C.; Yu, Y.M. Effects of Enterobacteria (Burkholderia sp.) on development of Riptortus pedestris. J. Fac. Agric. Kyushu Univ. 2014, 59, 77–84. [Google Scholar]
- He, H.; Ratnayake, A.S.; Janso, J.E.; He, M.; Yang, H.Y.; Loganzo, F.; Shor, B.; O’Donnell, C.J.; Koehn, F.E. Cytotoxic spliceostatins from Burkholderia sp. and their semisynthetic analogues. J. Nat. Prod. 2014, 77, 1864–1870. [Google Scholar] [CrossRef] [PubMed]
- Cordova-Kreylos, A.L.; Fernandez, L.E.; Koivunen, M.; Yang, A.; Flor-Weiler, L.; Marrone, P.G. Isolation and characterization of Burkholderia rinojensis sp. nov., a non-Burkholderia cepacia complex soil bacterium with insecticidal and miticidal activities. Appl. Environ. Microbiol. 2013, 79, 7669–7678. [Google Scholar] [CrossRef] [PubMed]
- de Jesus Sousa, J.A.; Olivares, F.L. Plant growth promotion by streptomycetes: Ecophysiology, mechanisms and applications. Chem. Biol. Technol. Agric. 2016, 3, 24. [Google Scholar] [CrossRef]
- Hartmann, A.; Rothballer, M.; Schmid, M. Lorenz Hiltner, a pioneer in rhizosphere microbial ecology and soil bacteriology research. Plant Soil 2008, 312, 7–14. [Google Scholar] [CrossRef]
- Qin, S.; Xing, K.; Jiang, J.H.; Xu, L.H.; Li, W.J. Biodiversity, bioactive natural products and biotechnological potential of plant-associated endophytic actinobacteria. Appl. Microbiol. Biotechnol. 2011, 89, 457–473. [Google Scholar] [CrossRef]
- Kido, G.S.; Spyhalski, E. Antimycin A, an antibiotic with insecticidal and miticidal properties. Science 1950, 112, 172–173. [Google Scholar] [CrossRef]
- Craveri, R.; Giolitti, G. An antibiotic with fungicidal and insecticidal activity produced by Streptomyces. Nature 1957, 179, 1307. [Google Scholar] [CrossRef]
- Oishi, H.; Sugawa, T.; Okutomi, T.; Suzuki, K.; Hayashi, T.; Sawada, M.; Ando, K. Insecticidal activity of macrotetrolide antibiotics. J. Antibiot. 1970, 23, 105–106. [Google Scholar] [CrossRef]
- Takahaski, N.; Suzuki, A.; Kimura, Y.; Miyamoto, S.; Tamura, S.; Mitsui, T.; Fukami, J. Isolation, structure and physiological activities of piericidin B, natural insecticide produced by a Streptomyces. Agric. Biol. Chem. 1968, 32, 1115–1122. [Google Scholar]
- Box, S.J.; Cole, M.; Yeoman, G.H. Prasinons A and B: Potent insecticides from Streptomyces prasinus. Appl. Microbiol. 1973, 29, 699–704. [Google Scholar] [CrossRef]
- Turner, M.J.; Schaeffer, J.M. Mode of action of ivermectin. In Ivermectin and Abamectin; Cambell, W.C., Ed.; Springer: New York, NY, USA, 1989; pp. 73–88. [Google Scholar]
- BCC Research LLC. Biopesticides: Global Markets to 2022; Report CHM029G; BCC Publishing: Wellesley, MA, USA, 2018. [Google Scholar]
- Markets and Markets. Biopesticides Market by Type (Bioinsecticides, Biofungicides, and Bionematicides), Source (Microbials, Plant Extracts, and Beneficial Insects), Mode of Application, Formulation, Crop Application, and Region—Global Forecast to 2023; Report 4759718; Research and Markets: Dublin, Ireland, 2019. [Google Scholar]
- Research and Markets. Biofertilizer Market: Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2019–2024. 2019. Available online: https://www.researchandmarkets.com/r/ucz1gg (accessed on 10 May 2020).
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).