Facing Climate Change: Application of Microbial Biostimulants to Mitigate Stress in Horticultural Crops
Abstract
1. Introduction
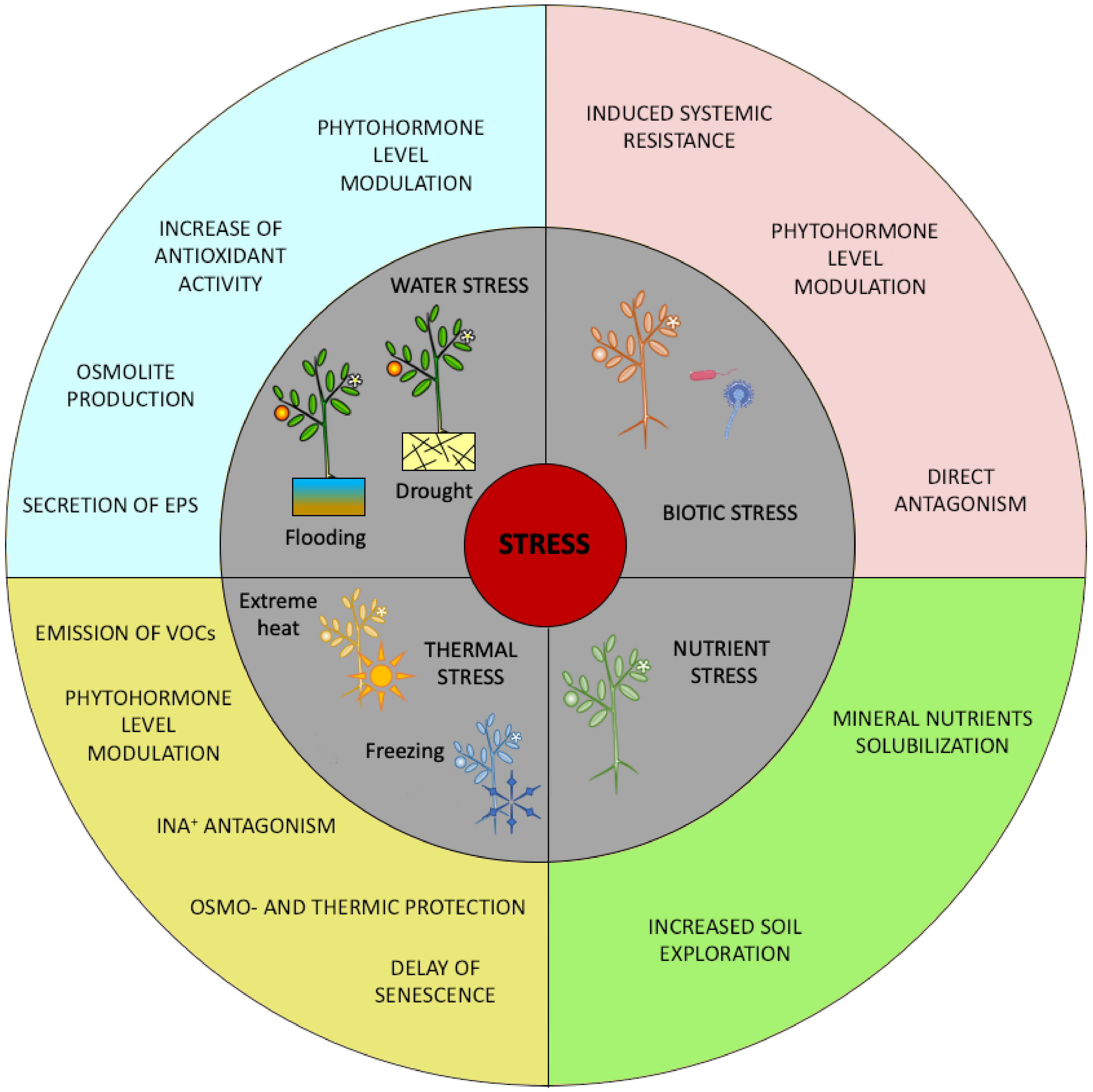
2. Effect of Climate Change-Induced Stress on Plant and Role of Microbial Biostimulants
2.1. Stress Induced by Extreme Thermal Events
2.1.1. High Temperature and Heat Waves
2.1.2. Low Temperature and Frost
2.2. Stress Induced by Water Scarcity or Waterlogging
2.2.1. Drought and Salinity Stress
2.2.2. Heavy Rainfall, Flooding and Water Stagnation
3. Role of Microbial Biostimulants in Response to Biotic Stresses
4. Limitations and Future Perspectives in the Application of Microbial Biostimulants
- Soil and crop characteristics: no microorganisms can be universally applied in any ecosystem [196] or on any vegetable host [197], thus, choosing a particular strain for a biostimulant product needs consideration of soil properties and specific crop requirements, in order to select microbial strains with the best adaptation to each particular condition [198].
- Mode of application of the microbial biostimulant, that should reduce microorganisms dispersion or death due to abiotic factors (UV, temperature) [203].
- Integration of microbial and plant genetic resources: future crop breeding programs should consider the plant’s capacity to establish stable symbiotic relationships with useful microorganisms as a highly desirable trait, closely linked to stress resistance, productivity, and resilience. Concurrently, the deeper characterization of microbial functions and mechanisms of interaction may enable the selection of specific biostimulants for a particular crop/cultivar in a given cultural condition.
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lotze-Campen, H.; Müller, C.; Popp, A.; Füssel, H.-M. Food security in a changing climate. In Climate Change, Justice and Sustainability: Linking Climate and Development Policy; Edenhofer, O., Wallacher, J., Lotze-Campen, H., Reder, M., Knopf, B., Müller, J., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 33–43. [Google Scholar]
- De Wolf, D.; Isard, S.A. Disease cycle approach to plant disease prediction. Annu. Rev. Phytopathol. 2007, 45, 203–220. [Google Scholar] [CrossRef] [PubMed]
- Garrett, K.A.; Nita, M.; De Wolf, E.D.; Esker, P.D.; Gomez-Montano, L.; Sparks, A.H. Plant pathogens as indicators of climate change. In Climate Change, 2nd ed.; Letcher, T.M., Ed.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 325–338. [Google Scholar]
- Kumar, A.; Verma, J.P. Does plant–Microbe interaction confer stress tolerance in plants: A review? Microbiol. Res. 2018, 207, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Wild, A. Soils, Land and Food: Managing the Land During the Twenty-First Century; Cambridge University Press: Cambridge, UK, 2003. [Google Scholar]
- Byerlee, D.; Stevenson, J.; Villoria, N. Does intensification slow crop land expansion or encourage deforestation? Glob. Food Secur. 2014, 3, 92–98. [Google Scholar] [CrossRef]
- Calvo, P.; Nelson, L.; Kloepper, J.W. Agricultural uses of plant biostimulants. Plant Soil 2014, 383, 3–41. [Google Scholar] [CrossRef]
- Yakhin, O.I.; Lubyanov, A.A.; Yakhin, I.A.; Brown, P.H. Biostimulants in plant science: A global perspective. Front. Plant Sci. 2017, 7, 2049. [Google Scholar] [CrossRef]
- Hayat, R.; Ali, S.; Amara, U.; Khalid, R.; Ahmed, I. Soil beneficial bacteria and their role in plant growth promotion: A review. Ann. Microbiol. 2010, 60, 579–598. [Google Scholar] [CrossRef]
- European Biostimulants Industry Council. Available online: http://www.biostimulants.eu/ (accessed on 30 March 2020).
- Du Jardin, P. Plant biostimulants: Definition, concept, main categories and regulation. Sci. Hortic. 2015, 19, 3–14. [Google Scholar] [CrossRef]
- Vandenkoornhuyse, P.; Quaiser, A.; Duhamel, M.; Le Van, A.; Dufresne, A. The importance of the microbiome of the plant holobiont. N. Phytol. 2015, 206, 1196–1206. [Google Scholar] [CrossRef]
- Selosse, M.A.; Le Tacon, F. The land flora: A phototroph–fungus partnership? Trends Ecol. Evol. 1998, 13, 15–20. [Google Scholar] [CrossRef]
- Wagg, C.; Bender, S.F.; Widmer, F.; van der Heijden, M.G.A. Soil biodiversity and soil community composition determine ecosystem multifunctionality. Proc. Natl. Acad. Sci. USA 2014, 111, 5266–5270. [Google Scholar] [CrossRef]
- Bhattacharyya, P.N.; Jha, D.K. Plant growth-promoting rhizobacteria (PGPR): Emergence in agriculture. World J. Microb. Biot. 2012, 28, 1327–1350. [Google Scholar] [CrossRef] [PubMed]
- Adnan, M.; Islam, W.; Shabbir, A.; Khan, K.A.; Ghramh, H.A.; Huang, Z.; Lu, G.D. Plant defense against fungal pathogens by antagonistic fungi with Trichoderma in focus. Microb. Pathog. 2019, 129, 7–18. [Google Scholar] [CrossRef] [PubMed]
- Szabó, M.; Csepregi, K.; Gálber, M.; Virányi, F.; Fekete, C. Control plant-parasitic nematodes with Trichoderma species and nematode-trapping fungi: The role of chi18-5 and chi18-12 genes in nematode egg-parasitism. Biol. Control 2012, 63, 121–128. [Google Scholar] [CrossRef]
- Szczałba, M.; Kopta, T.; Gąstoł, M.; Sękara, A. Comprehensive insight into arbuscular mycorrhizal fungi, Trichoderma spp. and plant multilevel interactions with emphasis on biostimulation of horticultural crops. J. Appl. Microbiol. 2019, 127, 630–647. [Google Scholar] [CrossRef] [PubMed]
- Preininger, C.; Sauer, U.; Bejarano, A.; Berninger, T. Concepts and applications of foliar spray for microbial inoculants. Appl. Microbiol. Biot. 2018, 102, 7265–7282. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.; Lee, S.H.; Kim, K.M.; Ryu, C.M. Foliar application of the leaf-colonizing yeast Pseudozyma churashimaensis elicits systemic defense of pepper against bacterial and viral pathogens. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Sarabia, M.; Cazares, S.; González-Rodríguez, A.; Mora, F.; Carreón-Abud, Y.; Larsen, J. Plant growth promotion traits of rhizosphere yeasts and their response to soil characteristics and crop cycle in maize agroecosystems. Rhizosphere 2018, 6, 67–73. [Google Scholar] [CrossRef]
- Bonaldi, M.; Chen, X.; Kunova, A.; Pizzatti, C.; Saracchi, M.; Cortesi, P. Colonization of lettuce rhizosphere and roots by tagged Streptomyces. Front. Microbiol. 2015, 6, 25. [Google Scholar] [CrossRef]
- Farrar, K.; Bryant, D.; Cope-Selby, N. Understanding and engineering beneficial plant–microbe interactions: Plant growth promotion in energy crops. Plant Biotechnol. J. 2014, 12, 1193–1206. [Google Scholar] [CrossRef]
- Yang, J.; Kloepper, J.W.; Ryu, C.M. Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci. 2009, 14, 1–4. [Google Scholar] [CrossRef]
- Pieterse, C.M.; Zamioudis, C.; Berendsen, R.L.; Weller, D.M.; Van Wees, S.C.; Bakker, P.A. Induced systemic resistance by beneficial microbes. Ann. Rev. Phytopathol. 2014, 52, 347–375. [Google Scholar] [CrossRef] [PubMed]
- EU Pesticides Database. Available online: https://ec.europa.eu/food/plant/pesticides/eu-pesticides-database (accessed on 22 May 2020).
- Microbe-Containing Bioproducts Database. The Ohio State University. Available online: https://u.osu.edu/vegprolab/microbe-containing-bioproducts/ (accessed on 22 May 2020).
- What are Climate and Climate Change? Available online: www.nasa.gov/audience/forstudents/5-8/features/nasa-knows/what-is-climate-change-58.html (accessed on 13 February 2020).
- Munns, R. Comparative physiology of salt and water stress. Plant Cell Environ. 2002, 25, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, S.K. Horticultural crops and climate change—A review. Indian J. Agric. Sci. 2017, 87, 12–22. [Google Scholar]
- Jordan, D.B.; Ogren, W.L. The CO2/O2 specificity of ribulose 1,5-bisphosphate carboxylase/oxygenase. Planta 1984, 161, 308–313. [Google Scholar] [CrossRef]
- Ku, S.B.; Edwards, G.E. Oxygen inhibition of photosynthesis. I. temperature dependence and relation to O2/CO2 solubility ratio. Plant Physiol. 1977, 598, 986–990. [Google Scholar] [CrossRef]
- Lawlor, D.W.; Fock, H. Water stress induced changes in the amounts of some photosynthetic assimilation products and respiratory metabolites of sunflower leaves. J. Exp. Bot. 1977, 288, 329–337. [Google Scholar] [CrossRef]
- Bhattacharya, A. Effect of high temperature on carbohydrate metabolism in plants. In Effect of High Temperature on Crop Productivity and Metabolism of Macro Molecules; Bhattacharya, A., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 115–216. [Google Scholar]
- Maltby, J.E. Tomatoes. In Horticulture Australia; Coombes, B., Ed.; Morescope Publishing: Hawthorn, Australia, 1995. [Google Scholar]
- Pearson, S.; Wheeler, T.R.; Hadley, P.; Wheldon, A.E. A validated model to predict the effects of environment on the growth of lettuce (Lactuca sativa L.): Implications for climate change. J. Hort. Sci. 1997, 72, 503–517. [Google Scholar] [CrossRef]
- Bisbis, M.B.; Nazim, G.; Blanke, M. Potential impacts of climate change on vegetable production and product quality—A review. J. Clean. Prod. 2018, 170, 1602–1620. [Google Scholar] [CrossRef]
- Wheeler, T.R.; Hadley, P.; Ellis, R.H.; Morison, J.I.L. Changes in growth and radiation use by lettuce crops in relation to temperature and ontogeny. Agr. Forest Meteorol. 1993, 66, 173–186. [Google Scholar] [CrossRef]
- Wien, H.C. The Physiology of Vegetable Crops; Cab International: Wallingford, UK, 1997. [Google Scholar]
- Hazarika, T.K. Climate change and Indian horticulture: Opportunities, challenges and mitigation strategies. Int. J. Environ. Eng. Manag. 2013, 4, 629–630. [Google Scholar]
- Funes, I.; Aranda, X.; Biel, C.; Carbó, J.; Camps, F.; Molina, A.J.; Savé, R. Future climate change impacts on apple flowering date in a Mediterranean subbasin. Agr. Water Manag. 2016, 164, 19–27. [Google Scholar] [CrossRef]
- Luedeling, E. Climate change impacts on winter chill for temperate fruit and nut production: A review. Sci. Hortic. 2012, 144, 218–229. [Google Scholar] [CrossRef]
- Sunley, R.J.; Atkinson, C.J.; Jones, H.G. Chill unit models and recent changes in the occurrence of winter chill and spring frost in the United Kingdom. J. Hort. Sci. Biotechnol. 2006, 81, 949–958. [Google Scholar] [CrossRef]
- Webb, L.; Darbyshire, R.; Goodwin, I. Climate change: Horticulture. In Encyclopedia of Agriculture and Food Systems; Van Alfen, N.K., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 266–283. [Google Scholar]
- Gornall, J.; Betts, R.; Burke, E.; Clark, R.; Camp, J.; Willett, K.; Wiltshire, A. Implications of climate change for agricultural productivity in the early twenty-first century. Philos. T. R. Soc. B 2010, 365, 2973–2989. [Google Scholar] [CrossRef] [PubMed]
- Kotak, S.K.; Larkindale, J.; Lee, U.; von Koskull-Do, P.; Vierling, E.; Scharf, K.D. Complexity of the heat stress response in plants. Curr. Opin. Plant Biol. 2007, 10, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Shulaev, V.; Cortes, D.; Miller, G.; Mittler, R. Metabolomics for plant stress response. Physiol. Plant 2008, 132, 199–208. [Google Scholar] [CrossRef]
- Qu, A.L.; Ding, Y.F.; Jiang, Q.; Zhu, C. Molecular mechanisms of the plant heat stress response. Biochem. Biophys. Res. Commun. 2013, 432, 203–207. [Google Scholar] [CrossRef]
- Dubois, M.; Van den Broeck, L.; Inzé, D. The pivotal role of ethylene in plant growth. Trends Plant Sci. 2018, 23, 311–323. [Google Scholar] [CrossRef]
- Duc, N.H.; Csintalan, Z.; Posta, K. Arbuscular mycorrhizal fungi mitigate negative effects of combined drought and heat stress on tomato plants. Plant Physiol. Biochem. 2018, 13, 297–307. [Google Scholar] [CrossRef]
- Bensalim, S.; Nowak, J.; Asiedu, S.K. A plant growth promoting rhizobacterium and temperature effects on performance of 18 clones of potato. Am. J. Potato Res. 1998, 75, 145–152. [Google Scholar] [CrossRef]
- Vitasse, Y.; Rebetez, M. Unprecedented risk of spring frost damage in Switzerland and Germany in 2017. Clim. Change 2018, 149, 233–246. [Google Scholar] [CrossRef]
- De Pascale, S.; Inglese, P.; Tagliavini, M. Harvesting the Sun Italy; Italian Society for Horticultural Science: Firenze, Italy, 2018; pp. 1–4. [Google Scholar]
- Unterberger, C.; Brunner, L.; Nabernegg, S.; Steininger, K.W.; Steiner, A.K.; Stabentheiner, E.; Monschein, E.; Truhetz, H. Spring frost risk for regional apple production under a warmer climate. PLoS ONE 2018, 13, e0200201. [Google Scholar] [CrossRef] [PubMed]
- Huner, N.P.; Öquist, G.; Hurry, V.M.; Krol, M.; Falk, S.; Griffith, M. Photosynthesis, photoinhibition and low temperature acclimation in cold tolerant plants. Photosynth. Res. 1993, 37, 19–39. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.K.; Bisht, S.C.; Bisht, J.K.; Bhatt, J.C. Cold-tolerant PGPRs as bioinoculants for stress management. In Bacteria in Agrobiology: Stress Management; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 95–118. [Google Scholar]
- Lindow, S.E. The role of bacterial ice nucleation in frost injury to plants. Annu. Rev. Phytopathol. 1983, 21, 363–384. [Google Scholar] [CrossRef]
- Lee, M.R.; Lee, R.E.; Strong-Gunderson, J.M.; Minges, S.R. Isolation of ice-nucleating active bacteria from the freeze-tolerant frog, Rana sylvatica. Cryobiology 1995, 32, 358–365. [Google Scholar] [CrossRef]
- Pouleur, S.; Richard, C.; Martin, J.-G.; Antoun, H. Ice nucleation activity in Fusarium acuminatum and Fusarium avenaceum. Appl. Environ. Microbiol. 1992, 58, 2960–2964. [Google Scholar] [CrossRef]
- Arny, D.C.; Lindow, S.E.; Upper, C.D. Frost sensitivity of Zea mays increased by application of Pseudomonas syringae. Nature 1976, 262, 282–284. [Google Scholar] [CrossRef]
- Anderson, J.A.; Buchanan, D.W.; Stall, R.E.; Hall, C.B. Frost injury of tender plants increased by Pseudomonas syringae van Hall. J. Am. Soc. Hort. Sci. 1982, 107, 123–125. [Google Scholar]
- Lindow, S.E.; Arny, D.C.; Upper, C.D. Erwinia herbicola: A bacterial ice nucleus active in increasing frost injury to corn. Phytopathology 1978, 68, 523–527. [Google Scholar] [CrossRef]
- Kim, H.K.; Orser, C.; Lindow, S.E.; Sands, D.C. Xanthomonas campestris pv. translucens strains active in ice nucleation. Plant Dis. 1987, 71, 994–997. [Google Scholar]
- Failor, K.C.; Schmale, D.G.; Vinatzer, B.A.; Monteil, C.L. Ice nucleation active bacteria in precipitation are genetically diverse and nucleate ice by employing different mechanisms. ISME J. 2017, 11, 2740–2753. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Griffith, M.; Patten, C.L.; Glick, B.R. Isolation and characterization of an antifreeze protein with ice nucleation activity from the plant growth promoting rhizobacterium Pseudomonas putida GR12-2. Can. J. Microbiol. 1998, 44, 64–73. [Google Scholar] [CrossRef]
- Skirvin, R.M.; Kohler, E.; Steiner, H.; Ayers, D.; Laughnan, A.; Norton, M.A.; Warmund, M. The use of genetically engineered bacteria to control frost on strawberries and potatoes. Whatever happened to all of that research? Sci. Hortic. 2000, 84, 179–189. [Google Scholar] [CrossRef]
- Wilson, M.; Lindow, S.E. Interactions between the biological control agent Pseudomonas fluorescens A506 and Erwinia amylovora in pear blossoms. Phytopathology 1993, 83, 117–123. [Google Scholar] [CrossRef]
- Wilson, M.; Lindow, S.E. Coexistence among epiphytic bacterial populations mediated through nutritional resource partitioning. Appl. Environ. Microbiol. 1994, 60, 4468–4477. [Google Scholar] [CrossRef]
- Selvakumar, G.; Panneerselvam, P.; Ganeshamurthy, A.N. Bacterial mediated alleviation of abiotic stress in crops. In Bacteria in Agrobiology: Stress Management; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 205–224. [Google Scholar]
- Lindow, S.E.; Brandl, M.T. Microbiology of the phyllosphere. Appl. Environ. Microbiol. 2003, 69, 1875–1883. [Google Scholar] [CrossRef]
- Amara, U.; Rabia, K.; Hayat, R. Soil bacteria and phytohormones for sustainable crop production. In Bacterial Metabolites in Sustainable Agroecosystem. Sustainable Development and Biodiversity; Maheshwari, D., Ed.; Springer: Cham, Switzerland, 2015; Volume 12, pp. 87–103. [Google Scholar]
- Selvakumar, G.; Kundu, S.; Joshi, P.; Nazim, S.; Gupta, A.D.; Mishra, P.K.; Gupta, H.S. Characterization of a cold-tolerant plant growth-promoting bacterium Pantoea dispersa 1A isolated from a sub-alpine soil in the North Western Indian Himalayas. World J. Microb. Biot. 2008, 24, 955–960. [Google Scholar] [CrossRef]
- Selvakumar, G.; Mohan, M.; Kundu, S.; Gupta, A.D.; Joshi, P.; Nazim, S.; Gupta, H.S. Cold tolerance and plant growth promotion potential of Serratia marcescens strain SRM (MTCC 8708) isolated from flowers of summer squash (Cucurbita pepo). Lett. Appl. Microbiol. 2008, 46, 171–175. [Google Scholar] [CrossRef]
- Mishra, P.K.; Mishra, S.; Selvakumar, G.; Bisht, S.C.; Bisht, J.K.; Kundu, S.; Gupta, H.S. Characterisation of a psychrotolerant plant growth promoting Pseudomonas sp. strain PGERs17 (MTCC 9000) isolated from North Western Indian Himalayas. Ann. Microbiol. 2008, 58, 561–568. [Google Scholar] [CrossRef]
- Glick, B.R. Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol. Res. 2014, 169, 30–39. [Google Scholar] [CrossRef]
- Barka, E.A.; Nowak, J.; Clément, C. Enhancement of chilling resistance of inoculated grapevine plantlets with a plant growth-promoting rhizobacterium, Burkholderia phytofirmans strain PsJN. Appl. Environ. Microbiol. 2006, 72, 7246–7252. [Google Scholar] [CrossRef] [PubMed]
- Theocharis, A.; Bordiec, S.; Fernandez, O.; Paquis, S.; Dhondt-Cordelier, S.; Baillieul, F.; Barka, E.A. Burkholderia phytofirmans PsJN primes Vitis vinifera L. and confers a better tolerance to low nonfreezing temperatures. MPMI 2012, 25, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Tiryaki, D.; Aydın, I.; Atıcı, O. Psychrotolerant bacteria isolated from the leaf apoplast of cold-adapted wild plants improve the cold resistance of bean (Phaseolus vulgaris L.) under low temperature. Cryobiology 2019, 86, 111–119. [Google Scholar] [CrossRef] [PubMed]
- El-Daim, I.A.; Bejai, S.; Meijer, J. Improved heat stress tolerance of wheat seedlings by bacterial seed treatment. Plant Soil 2014, 379, 337–350. [Google Scholar] [CrossRef]
- Park, Y.G.; Mun, B.G.; Kang, S.M.; Hussain, A.; Shahzad, R.; Seo, C.W.; Kim, A.Y.; Lee, S.U.; Oh, K.Y.; Lee, D.Y.; et al. Bacillus aryabhattai SRB02 tolerates oxidative and nitrosative stress and promotes the growth of soybean by modulating the production of phytohormones. PLoS ONE 2017, 12, e0173203. [Google Scholar] [CrossRef]
- Ali, S.Z.; Sandhya, V.; Grover, M.; Kishore, N.; Rao, L.V.; Venkateswarlu, B. Pseudomonas sp. strain AKM-P6 enhances tolerance of sorghum seedlings to elevated temperatures. Biol. Fert. Soils 2009, 46, 45–55. [Google Scholar] [CrossRef]
- Ali, S.Z.; Sandhya, V.; Grover, M.; Linga, V.R.; Bandi, V. Effect of inoculation with a thermotolerant plant growth promoting Pseudomonas putida strain AKMP7 on growth of wheat (Triticum spp.) under heat stress. J. Plant Interact. 2011, 6, 239–246. [Google Scholar] [CrossRef]
- Sansavini, S.; Costa, G.; Gucci, R.; Inglese, P.; Ramina, A.; Xiloyannis, C. Arboricoltura Generale; Patron Editore: Bologna, Italy, 2012. [Google Scholar]
- Wetherald, R.T.; Manabe, S. Simulation of hydrologic changes associated with global warming. J. Geophys. Res. Atmos. 2002, 107, D19. [Google Scholar] [CrossRef]
- Trenberth, K.E.; Dai, A.; Van Der Schrier, G.; Jones, P.D.; Barichivich, J.; Briffa, K.R.; Sheffield, J. Global warming and changes in drought. Nat. Clim. Chang. 2014, 4, 17. [Google Scholar] [CrossRef]
- Othman, Y.; Al-Karaki, G.; Al-Tawaha, A.R.; Al-Horani, A. Variation in germination and ion uptake in barley genotypes under salinity conditions. WJAS 2006, 2, 11–15. [Google Scholar]
- Sequi, P. Fondamenti di Chimica del Suolo, 2nd ed.; Pàtron Editore: Bologna, Italy, 2006. [Google Scholar]
- Rasool, S.; Hameed, A.; Azooz, M.M.; Siddiqi, T.O.; Ahmad, P. Salt stress: Causes, types and responses of plants. In Ecophysiology and Responses of Plants Under Salt Stress; Prasad, M.N.V., Ahmad, P., Eds.; Springer: New York, NY, USA, 2013; pp. 1–24. [Google Scholar]
- Vicente-Serrano, S.M.; Quiring, S.M.; Peña-Gallardo, M.; Yuan, S.; Domínguez-Castro, F. A review of environmental droughts: Increased risk under global warming? Earth Sci. Rev. 2020, 201, 102953. [Google Scholar] [CrossRef]
- Daliakopoulos, I.N.; Tsanis, I.K.; Koutroulis, A.; Kourgialas, N.N.; Varouchakis, A.E.; Karatzas, G.P.; Ritsema, C.J. The threat of soil salinity: A European scale review. Sci. Total Environ. 2016, 573, 727–739. [Google Scholar] [CrossRef] [PubMed]
- Shukla, P.R.; Skea, J.; Slade, R.; van Diemen, R.; Haughey, E.; Malley, J.; Pathak, M.; Portugal Pereira, J. Technical Summary, 2019. In Climate Change and Land: An IPCC Special Report on Climate Change, Desertification, Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems; Shukla, P.R., Skea, J., Calvo Buendia, E., Masson-Delmotte, V., Portner, H.-O., Roberts, D.C., Zhai, P., Slade, R., Connors, S., van Diemen, R., et al., Eds.; UN Intergovernmental Panel on Climate Change: Geneva, Switzerland, in press.
- Huang, J.; Zhang, G.; Zhang, Y.; Guan, X.; Wei, Y.; Guo, R. Global desertification vulnerability to climate change and human activities. Land Degrad. Dev. 2020, 1–12. [Google Scholar] [CrossRef]
- Smirnoff, N. The role of active oxygen in the response of plants to water deficit and desiccation. N. Phytol. 1993, 125, 27–58. [Google Scholar] [CrossRef]
- Ali, S.; Charles, T.C.; Glick, B.R. Amelioration of high salinity stress damage by plant growth-promoting bacterial endophytes that contain ACC deaminase. Plant Physiol. Biochem. 2014, 80, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Rouphael, Y.; Cardarelli, M.; Schwarz, D.; Franken, P.; Colla, G. Effects of drought on nutrient uptake and assimilation in vegetable crops. In Plant Responses to Drought Stress; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 171–195. [Google Scholar]
- Grover, M.; Ali, S.Z.; Sandhya, V.; Rasul, A.; Venkateswarlu, B. Role of microorganisms in adaptation of agriculture crops to abiotic stresses. World J. Microb. Biot. 2011, 27, 1231–1240. [Google Scholar] [CrossRef]
- Nwodo, U.U.; Green, E.; Okoh, A.I. Bacterial exopolysaccharides: Functionality and prospects. Int. J. Mol. Sci. 2012, 13, 14002–14015. [Google Scholar] [CrossRef]
- Tewari, S.; Arora, N.K. Multifunctional exopolysaccharides from Pseudomonas aeruginosa PF23 involved in plant growth stimulation, biocontrol and stress amelioration in sunflower under saline conditions. Curr. Microbiol. 2014, 69, 484–494. [Google Scholar] [CrossRef]
- Sandhya, V.Z.A.S.; Grover, M.; Reddy, G.; Venkateswarlu, B. Alleviation of drought stress effects in sunflower seedlings by the exopolysaccharides producing Pseudomonas putida strain GAP-P45. Biol. Fert. Soils 2009, 46, 17–26. [Google Scholar] [CrossRef]
- Barzana, G.; Aroca, R.; Paz, J.A.; Chaumont, F.; Martinez-Ballesta, M.C.; Carvajal, M.; Ruiz-Lozano, J.M. Arbuscular mycorrhizal symbiosis increases relative apoplastic water flow in roots of the host plant under both well-watered and drought stress conditions. Ann. Bot. 2012, 109, 1009–1017. [Google Scholar] [CrossRef]
- Khan, A.L.; Hussain, J.; Al-Harrasi, A.; Al-Rawahi, A.; Lee, I.J. Endophytic fungi: Resource for gibberellins and crop abiotic stress resistance. Crit. Rev. Biotechnol. 2015, 35, 62–74. [Google Scholar] [CrossRef] [PubMed]
- Cavagnaro, T.R.; Bender, S.F.; Asghari, H.R.; van der Heijden, M.G. The role of arbuscular mycorrhizas in reducing soil nutrient loss. Trends Plant Sci. 2015, 20, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Gong, M.; Tang, M.; Chen, H.; Zhang, Q.; Feng, X. Effects of two Glomus species on the growth and physiological performance of Sophora davidii seedlings under water stress. N. Forest 2013, 44, 399–408. [Google Scholar] [CrossRef]
- Wu, Q.S.; Xia, R.X.; Zou, Y.N. Improved soil structure and citrus growth after inoculation with three arbuscular mycorrhizal fungi under drought stress. Eur. J. Soil Biol. 2008, 44, 122–128. [Google Scholar] [CrossRef]
- Bray, E.A. Response to abiotic stress. In Biochemistry and Molecular Biology of Plants; Buchanan, B.B., Gruissem, W., Jones, R.L., Eds.; ASPP: Rockville, MD, USA, 2000; pp. 1158–1203. [Google Scholar]
- Aroca, R.; Rosa, P.; Ruiz-Lozano, J.M. How does arbuscular mycorrhizal symbiosis regulate root hydraulic properties and plasma membrane aquaporins in Phaseolus vulgaris under drought, cold or salinity stresses? N. Phytol. 2007, 173, 808–816. [Google Scholar] [CrossRef]
- Li, T.; Hu, Y.J.; Hao, Z.P.; Li, H.; Chen, B.D. Aquaporin genes GintAQPF1 and GintAQPF2 from Glomus intraradices contribute to plant drought tolerance. Plant Signal. Behav. 2013, 8, e24030. [Google Scholar] [CrossRef]
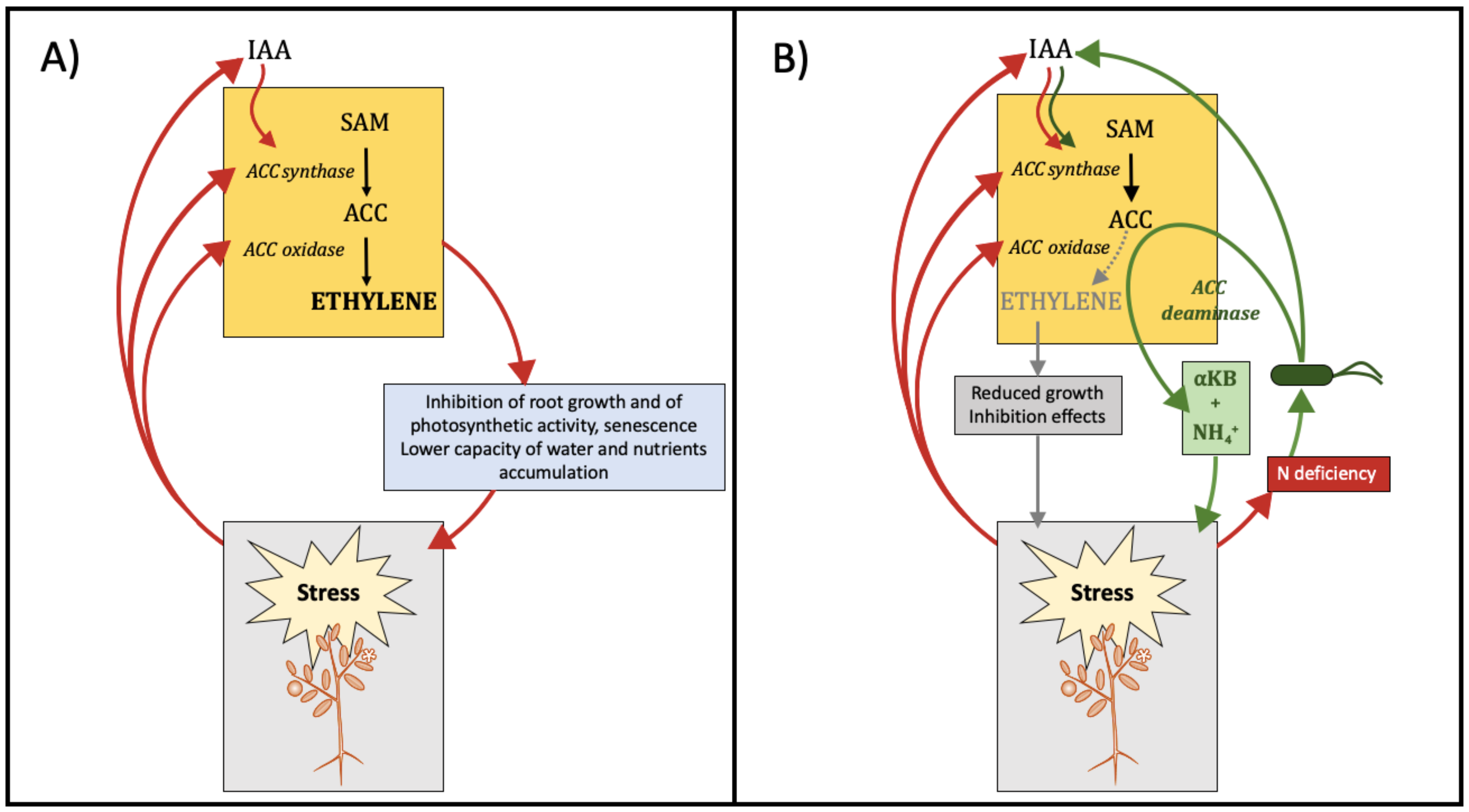
- Dimkpa, C.; Weinand, T.; Asch, F. Plant–rhizobacteria interactions alleviate abiotic stress conditions. Plant Cell Environ. 2009, 32, 1682–1694. [Google Scholar] [CrossRef]
- Paul, D.; Lade, H. Plant-growth-promoting rhizobacteria to improve crop growth in saline soils: A review. Agron. Sustain. Dev. 2014, 34, 737–752. [Google Scholar] [CrossRef]
- Kavamura, V.N.; Santos, S.N.; da Silva, J.L.; Parma, M.M.; Ávila, L.A.; Visconti, A.; Zucchi, T.D.; Taketani, R.G.; Andreote, F.D.; de Melo, I.S. Screening of Brazilian cacti rhizobacteria for plant growth promotion under drought. Microbiol. Res. 2013, 168, 183–191. [Google Scholar] [CrossRef]
- Kaushal, M.; Wani, S.P. Plant-growth-promoting rhizobacteria: Drought stress alleviators to ameliorate crop production in drylands. Ann. Microbiol. 2016, 66, 35–42. [Google Scholar] [CrossRef]
- Sharifi, R.; Ryu, C.M. Sniffing bacterial volatile compounds for healthier plants. Curr. Opin. Plant Biol. 2018, 44, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, L.M.; Pei, H.Y.; Xu, Z.H. Low nitrogen stress stimulating the indole-3-acetic acid biosynthesis of Serratia sp. ZM is vital for the survival of the bacterium and its plant growth-promoting characteristic. Arch. Microbiol. 2017, 199, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Yuan, C.-L.; Mou, C.X.; Wu, W.L.; Guo, Y.B. Effect of different fertilization treatments on indole-3-acetic acid producing bacteria in soil. J. Soil Sediment 2011, 11, 322–329. [Google Scholar] [CrossRef]
- Egamberdieva, D. Pseudomonas chlororaphis: A salt-tolerant bacterial inoculant for plant growth stimulation under saline soil conditions. Acta Physiol. Plant 2012, 34, 751–756. [Google Scholar] [CrossRef]
- Liu, C.Y.; Zhang, F.; Zhang, D.J.; Srivastava, A.K.; Wu, Q.S.; Zou, Y.N. Mycorrhiza stimulates root-hair growth and IAA synthesis and transport in trifoliate orange under drought stress. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Arkhipova, T.N.; Prinsen, E.; Veselov, S.U.; Martinenko, E.V.; Melentiev, A.I.; Kudoyarova, G.R. Cytokinin producing bacteria enhance plant growth in drying soil. Plant Soil 2007, 292, 305–315. [Google Scholar] [CrossRef]
- Liu, F.; Xing, S.; Ma, H.; Du, Z.; Ma, B. Cytokinin-producing, plant growth-promoting rhizobacteria that confer resistance to drought stress in Platycladus orientalis container seedlings. Appl. Microbiol. Biotechnol. 2013, 97, 9155–9164. [Google Scholar] [CrossRef]
- Kang, S.M.; Radhakrishnan, R.; You, Y.H.; Khan, A.L.; Park, J.M.; Lee, S.M.; Lee, I.J. Cucumber performance is improved by inoculation with plant growth-promoting microorganisms. Acta Agr. Scand. B-S P 2015, 65, 36–44. [Google Scholar] [CrossRef]
- Kang, S.M.; Radhakrishnan, R.; Khan, A.L.; Kim, M.J.; Park, J.M.; Kim, B.R.; Shin, D.H.; Lee, I.J. Gibberellin secreting rhizobacterium, Pseudomonas putida H-2-3 modulates the hormonal and stress physiology of soybean to improve the plant growth under saline and drought conditions. Plant Physiol. Biochem. 2014, 84, 115–124. [Google Scholar] [CrossRef]
- Jahromi, F.; Aroca, R.; Porcel, R.; Ruiz-Lozano, J.M. Influence of salinity on the in vitro development of Glomus intraradices and on the in vivo physiological and molecular responses of mycorrhizal lettuce plants. Microb. Ecol. 2008, 55, 45. [Google Scholar] [CrossRef]
- Mayak, S.; Tirosh, T.; Glick, B.R. Plant growth-promoting bacteria that confer resistance to water stress in tomatoes and peppers. Plant Sci. 2004, 166, 525–530. [Google Scholar] [CrossRef]
- Saravanakumar, D.; Samiyappan, R. ACC deaminase from Pseudomonas fluorescens mediated saline resistance in groundnut (Arachis hypogea) plants. J. Appl. Microbiol. 2007, 102, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Vurukonda, S.S.K.P.; Vardharajula, S.; Shrivastava, M.; SkZ, A. Enhancement of drought stress tolerance in crops by plant growth promoting rhizobacteria. Microbiol. Res. 2016, 184, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Heidari, M.; Golpayegani, A. Effects of water stress and inoculation with plant growth promoting rhizobacteria (PGPR) on antioxidant status and photosynthetic pigments in basil (Ocimum basilicum L.). J. Saudi Soc. Agr. Sci. 2012, 11, 57–61. [Google Scholar] [CrossRef]
- Morgan, J.M. Osmoregulation and water stress in higher plants. Annu. Rev. Plant Physiol. 1984, 35, 299–319. [Google Scholar] [CrossRef]
- Paul, M.J.; Primavesi, L.F.; Jhurreea, D.; Zhang, Y. Trehalose metabolism and signaling. Annu. Rev. Plant Biol. 2008, 59, 417–441. [Google Scholar] [CrossRef]
- Sanders, G.J.; Arndt, S.K. Osmotic adjustment under drought conditions. In Plant Responses to Drought Stress; Hernandez-Ros, R.A., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 199–229. [Google Scholar]
- Shintu, P.V.; Jayaram, K.M. Phosphate solubilising bacteria (Bacillus polymyxa)-An effective approach to mitigate drought in tomato (Lycopersicon esculentum Mill.). Trop. Plant Res. 2015, 2, 17–22. [Google Scholar]
- Zhang, H.; Kim, M.-S.; Krishnamachari, V.; Payton, P.; Sun, Y.; Grimson, M.; Farag, M.A.; Ryu, C.-M.; Allen, R.; Melo, I.S.; et al. Rhizobacterial volatile emissions regulate auxin homeostasis and cell expansion in Arabidopsis. Planta 2007, 226, 839–851. [Google Scholar] [CrossRef]
- Bailly, A.; Groenhagen, U.; Schulz, S.; Geisler, M.; Eberl, L.; Weisskopf, L. The inter-kingdom volatile signal indole promotes root development by interfering with auxin signalling. Plant J. 2014, 80, 758–771. [Google Scholar] [CrossRef]
- Cho, S.M.; Kang, B.R.; Han, S.H.; Anderson, A.J.; Park, J.-Y.; Lee, Y.-H.; Cho, B.H.; Yang, K.-Y.; Ryu, C.-M.; Kim, Y.C. 2R,3R-butanediol, a bacterial volatile produced by Pseudomonas chlororaphis O6, is involved in induction of systemic tolerance to drought in Arabidopsis thaliana. MPMI 2008, 21, 1067–1075. [Google Scholar] [CrossRef]
- Zhang, H.; Murzello, C.; Sun, Y.; Kim, M.-S.; Xie, X.; Jeter, R.M.; Zak, J.C.; Dowd, S.E.; Pare, P.W. Choline and osmotic-stress tolerance induced in Arabidopsis by the soil microbe Bacillus subtilis (GB03). MPMI 2010, 23, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Ryu, C.-M.; Farag, M.A.; Hu, C.-H.; Reddy, M.S.; Wei, H.-X.; Paré, P.W.; Kloepper, J.W. Bacterial volatiles promote growth in Arabidopsis. Proc. Natl. Acad. Sci. USA 2003, 100, 4927–4932. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, D.; Garladinne, M.; Lee, Y.H. Volatile indole produced by rhizobacterium Proteus vulgaris jbls202 stimulates growth of Arabidopsis thaliana through auxin, cytokinin, and brassinosteroid pathways. J. Plant Growth Regul. 2015, 34, 158–168. [Google Scholar] [CrossRef]
- Tahir, H.A.S.; Gu, Q.; Wu, H.; Raza, W.; Hanif, A.; Wu, L.; Colman, M.V.; Gao, X. Plant growth promotion by volatile organic compounds produced by Bacillus subtilis SYST2. Front. Microbiol. 2017, 8, 171. [Google Scholar] [CrossRef] [PubMed]
- Ledger, T.; Rojas, S.; Timmermann, T.; Pinedo, I.; Poupin, M.J.; Garrido, T.; Richter, P.; Tamayo, J.; Donoso, R. Volatile-mediated effects predominate in Paraburkholderia phytofirmans growth promotion and salt stress tolerance of Arabidopsis thaliana. Front. Microbiol. 2016, 7, 1838. [Google Scholar] [CrossRef]
- Gutiérrez-Luna, F.M.; López-Bucio, J.; Altamirano-Hernández, J.; Valencia-Cantero, E.; de la Cruz, H.R.; Macías-Rodríguez, L. Plant growth-promoting rhizobacteria modulate root-system architecture in Arabidopsis thaliana through volatile organic compound emission. Symbiosis 2010, 51, 75–83. [Google Scholar] [CrossRef]
- Meehl, G.A.; Stocker, T.F.; Collins, W.D.; Friedlingstein, P.; Gaye, A.T.; Gregory, J.M.; Kitoh, A.; Knutti, R.; Murphy, J.M.; Noda, A.; et al. Global climate projections. In Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Solomon, S., Qin, D.H., Manning, M., Marquis, M., Averyt, K., Tignor, M.M.B., Miller, H.L., Chen, Z.L., Eds.; IPCC: Cambridge, UK, 2007. [Google Scholar]
- Foster, G.; Rahmstorf, S. Global temperature evolution 1979–2010. Environ. Res. Lett. 2011, 6, 044022. [Google Scholar] [CrossRef]
- Stevenson, S.; Fox-Kemper, B.; Jochum, M.; Neale, R.; Deser, C.; Meehl, G. Will there be a significant change to El Nino in the 21st century? J. Climate 2011, 25, 2129–2145. [Google Scholar] [CrossRef]
- Cramer, G.R.; Urano, K.; Delrot, S.; Pezzotti, M.; Shinozaki, K. Effects of abiotic stress on plants: A systems biology perspective. BMC Plant Biol. 2011, 11, 163. [Google Scholar] [CrossRef]
- Bradford, K.J.; Yang, S.F. Xylem transport of 1-aminocyclopropane-1-carboxylic acid, an ethylene precursor, in waterlogged tomato plants. Plant Physiol. 1980, 65, 322–326. [Google Scholar] [CrossRef]
- Else, M.A.; Jackson, M.B. Transport of 1-aminocyclopropane-1-carboxylic acid (ACC) in the transpiration stream of tomato (Lycopersicon esculentum) in relation to foliar ethylene production and petiole epinasty. Funct. Plant Biol. 1998, 25, 453–458. [Google Scholar] [CrossRef]
- Grichko, V.P.; Glick, B.R. Amelioration of flooding stress by ACC deaminase-containing plant growth-promoting bacteria. Plant Physiol. Biochem. 2001, 39, 11–17. [Google Scholar] [CrossRef]
- Li, J.; McConkey, B.J.; Cheng, Z.; Guo, S.; Glick, B.R. Identification of plant growth-promoting bacteria-responsive proteins in cucumber roots under hypoxic stress using a proteomic approach. J. Proteom. 2013, 84, 119–131. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Kim, W.C. Plant growth promotion under water: Decrease of waterloggining-induced ACC and ethylene levels by ACC deaminase-producing bacteria. Front. Microbiol. 2018, 9, 1096. [Google Scholar] [CrossRef] [PubMed]
- Ratchaniwan, J.; Jantasuriyarat, C.; Thamchaipenet, A. Positive role of 1-aminocyclopropane-1-carboxylate deaminase-producing endophytic Streptomyces sp. GMKU 336 on flooding resistance of mung bean. Agric. Nat. 2018, 52, 330–334. [Google Scholar]
- Lim, J.H.; Kim, S.D. Induction of drought stress resistance by multi-functional PGPR Bacillus licheniformis K11 in pepper. Plant Pathol. J. 2013, 29, 201–208. [Google Scholar] [CrossRef]
- Zahir, Z.A.; Munir, A.; Asghar, H.N.; Shaharoona, B.; Arshad, M. Effectiveness of rhizobacteria containing ACC deaminase for growth promotion of peas (Pisum sativum) under drought conditions. J. Microbiol. Biotechnol. 2008, 18, 958–963. [Google Scholar]
- Porcel, R.; Aroca, R.; Azcon, R.; Ruiz-Lozano, J.M. PIP aquaporin gene expression in arbuscular mycorrhizal Glycine max and Lactuca sativa plants in relation to drought stress tolerance. Plant Mol. Biol. 2006, 60, 389–404. [Google Scholar] [CrossRef]
- Porcel, R.; Ruiz-Lozano, J.M. Arbuscular mycorrhizal influence on leaf water potential, solute accumulation, and oxidative stress in soybean plants subjected to drought stress. J. Exp. Bot. 2004, 55, 1743–1750. [Google Scholar] [CrossRef]
- Baltes, N.J.; Gil-Humanes, J.; Voytas, D.F. Genome engineering and agriculture: Opportunities and challenges. In Progress in Molecular Biology and Translational Science; Weeks, D.P., Yang, B., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 1–26. [Google Scholar]
- Logan, J.A.; Régnière, J.; Powell, J.A. Assessing the impacts of global warming on forest pest dynamics. Front. Ecol. Environ. 2003, 1, 130–137. [Google Scholar] [CrossRef]
- Coakley, S.M.; Scherm, H.; Chakraborty, S. Climate change and plant disease management. Annu. Rev. Phytopathol. 1999, 37, 399–426. [Google Scholar] [CrossRef] [PubMed]
- Velásquez, A.C.; Danve, C.; Castroverde, M.; He, S.Y. Plant-pathogen warfare under changing climate conditions. Curr. Biol. 2018, 28, 619–634. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Tripathi, D.K.; Chauhan, D.K.; Kumar, N.; Singh, G.S. Paradigms of climate change impacts on some major food sources of the world: A review on current knowledge and future prospects. Agr. Ecosyst. Environ. 2016, 216, 356–373. [Google Scholar] [CrossRef]
- Ferrante, P.; Scortichini, M. Frost promotes the pathogenicity of Pseudomonas syringae pv. actinidiae in Actinidia chinensis and A. deliciosa plants. Plant Pathol. 2014, 63, 12–19. [Google Scholar]
- Delcour, I.; Spanoghe, P.; Uyttendaele, M. Impact of climate change on pesticide use. Food Res. Int. 2015, 68, 7–15. [Google Scholar] [CrossRef]
- Bloomfield, J.P.; Williams, R.J.; Gooddy, D.C.; Cape, J.N.; Guha, P. Impacts of climate change on the fate and behaviour of pesticides in surface and groundwater—A UK perspective. Sci. Total Environ. 2006, 369, 163–177. [Google Scholar] [CrossRef]
- Noyes, P.D.; McElwee, M.K.; Miller, H.D.; Clark, B.W.; Van Tiem, L.A.; Walcott, K.C.; Erwin, K.N.; Levin, E.D. The toxicology of climate change: Environmental contaminants in a warming world. Environ. Int. 2009, 35, 971–986. [Google Scholar] [CrossRef]
- Garrett, K.A.; Forbes, G.A.; Savary, S.; Skelsey, P.; Sparks, A.; Valdivia, C.; van Bruggen, A.H.C.; Willocquet, L.; Djurle, A.; Duveiller, E.; et al. Complexity in climate-change impacts: An analytical framework for effects mediated by plant disease. Plant Pathol. 2011, 60, 15–30. [Google Scholar] [CrossRef]
- Durrant, W.E.; Dong, X. Systemic acquired resistance. Annu. Rev. Phytopathol. 2004, 4, 185–209. [Google Scholar] [CrossRef]
- Van Oosten, V.R.; Bodenhausen, N.; Reymond, P.; Van Pelt, J.A.; Van Loon, L.C.; Dicke, M.; Pieterse, C.M. Differential effectiveness of microbially induced resistance against herbivorous insects in Arabidopsis. MPMI 2008, 21, 919–930. [Google Scholar] [CrossRef]
- De Vleesschauwer, D.; Höfte, M. Rhizobacteria-induced systemic resistance. Adv. Bot. Res. 2009, 51, 223–281. [Google Scholar]
- Kloepper, J.W.; Ryu, C.M.; Zhang, S. Induced systemic resistance and promotion of plant growth by Bacillus spp. Phytopathology 2004, 94, 1259–1266. [Google Scholar] [CrossRef] [PubMed]
- Bakker, P.A.H.M.; Pieterse, C.; Van Loon, L.C. Induced systemic resistance by fluorescent Pseudomonas spp. Phytopathology 2007, 97, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Ryu, C.M.; Farag, M.A.; Hu, C.H.; Reddy, M.S.; Kloepper, J.W.; Paré, P.W. Bacterial volatiles induce systemic resistance in Arabidopsis. Plant Physiol. 2004, 134, 1017–1026. [Google Scholar] [CrossRef]
- Schuhegger, R.; Ihring, A.; Gantner, S.; Bahnweg, G.; Knappe, C.; Vogg, G.; Hutzler, P.; Schmid, M.; Van Breusegem, F.; Eberl, L.; et al. Induction of systemic resistance in tomato by N-acyl-L-homoserine lactone-producing rhizosphere bacteria. Plant Cell Environ. 2006, 29, 909–918. [Google Scholar] [CrossRef]
- Iavicoli, A.; Boutet, E.; Buchala, A.; Métraux, J.P. Induced systemic resistance in Arabidopsis thaliana in response to root inoculation with Pseudomonas fluorescens CHA0. MPMI 2003, 16, 851–858. [Google Scholar] [CrossRef]
- Ongena, M.; Jacques, P. Bacillus lipopeptides: Versatile weapons for plant disease biocontrol. Trends Microbiol. 2008, 16, 115–125. [Google Scholar] [CrossRef]
- Lugtemberg, B.; Kamilova, F. Plant growth-promoting rhizobacteria. Annu. Rev. Microbiol. 2009, 63, 541–556. [Google Scholar] [CrossRef]
- Pieterse, C.M.J.; Van Pelt, J.A.; Ton, J.; Parchmann, S.; Mueller, M.J.; Buchala, A.J.; Metraux, J.-P.; Van Loon, L.C. Rhizobacteria-mediated induced systemic resistance (ISR) in Arabidopsis requires sensitivity to jasmonate and ethylene but is not accompanied by an increase in their production. Physiol. Mol. Plant Pathol. 2000, 57, 123–134. [Google Scholar] [CrossRef]
- Verhagen, B.W.M.; Glazebrook, J.; Zhu, T.; Chang, H.-S.; Van Loon, L.C.; Pieterse, C.M.J. The transcriptome of rhizobacteria-induced systemic resistance in Arabidopsis. MPMI 2004, 17, 895–908. [Google Scholar] [CrossRef]
- Perpetuini, G.; Donati, I.; Cellini, A.; Orrù, L.; Giongo, L.; Farneti, B.; Spinelli, F. Genetic and functional characterization of the bacterial community on fruit of three raspberry (Rubus idaeus) cultivars. J. Berry Res. 2019, 9, 227–247. [Google Scholar] [CrossRef]
- Van der Ent, S.; Verhagen, B.W.; Van Doorn, R.; Bakker, D.; Verlaan, M.G.; Pel, M.J.; Joosten, R.G.; Proveniers, M.C.G.; Van Loon, L.C.; Ton, J.; et al. MYB72 is required in early signaling steps of rhizobacteria-induced systemic resistance in Arabidopsis. Plant Physiol. 2008, 146, 1293–1304. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.S.; Lakshmanan, V.; Caplan, J.L.; Powell, D.; Czymmek, K.J.; Levia, D.F.; Bais, H.P. Rhizobacteria Bacillus subtilis restricts foliar pathogen entry through stomata. Plant J. 2012, 72, 694–706. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.Y.; Park, E.M.; Glick, B.R. 1-Aminocyclopropane-1-carboxylate deaminase from Pseudomonas putida UW4 facilitates the growth of canola in the presence of salt. Can. J. Microbiol. 2007, 53, 912–918. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Reddy, M.S.; Ryu, C.M.; McInroy, J.A.; Wilson, M.; Kloepper, J.W. Induced systemic protection against tomato late blight elicited by plant growth-promoting rhizobacteria. Phytopathology 2002, 92, 1329–1333. [Google Scholar] [CrossRef]
- Park, K.; Kloepper, J.W.; Ryu, C.M. Rhizobacterial exopolysaccharides elicit induced resistance on cucumber. J. Microbiol. Biotechnol. 2008, 18, 1095–1100. [Google Scholar]
- Tran, H.; Ficke, A.; Asiimwe, T.; Höfte, M.; Raaijmakers, J.M. Role of the cyclic lipopeptide massetolide A in biological control of Phytophthora infestans and in colonization of tomato plants by Pseudomonas fluorescens. N. Phytol. 2007, 175, 731–742. [Google Scholar] [CrossRef]
- Leeman, M.; Van Pelt, J.A.; Hendrickx, M.J.; Scheffer, R.J.; Bakker, P.A.H.M.; Schippers, B. Biocontrol of Fusarium wilt of radish in commercial greenhouse trials by seed treatment with Pseudomonas fluorescens WCS374. Phytopathology 1995, 85, 1301–1305. [Google Scholar] [CrossRef]
- Reitz, M.; Oger, P.; Farrand, S.; Hallmann, J.; Meyer, A.; Sikora, R.; Niehaus, K. Importance of the O-antigen, core-region and lipid A of rhizobial lipopolysaccharides for the induction of systemic resistance in potato to Globodera pallida. Nematology 2002, 4, 73–79. [Google Scholar] [CrossRef]
- Reitz, M.; Rudolph, K.; Schröder, I.; Hoffmann-Hergarten, S.; Hallmann, J.; Sikora, R.A. Lipopolysaccharides of Rhizobium etli strain G12 act in potato roots as an inducing agent of systemic resistance to infection by the cyst nematode Globodera pallida. Appl. Environ. Microbiol. 2000, 66, 3515–3518. [Google Scholar] [CrossRef]
- Ran, L.X.; Van Loon, L.C.; Bakker, P.A.H.M. No role for bacterially produced salicylic acid in rhizobacterial induction of systemic resistance in Arabidopsis. Phytopathology 2005, 95, 1349–1355. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhao, L.J.; Yang, X.N.; Li, X.Y.; Wei, M.U.; Feng, L.I.U. Antifungal, insecticidal and herbicidal properties of volatile components from Paenibacillus polymyxa strain BMP-11. Agr. Sci. China 2011, 10, 728–736. [Google Scholar] [CrossRef]
- Kishimoto, K.; Matsui, K.; Ozawa, R.; Takabayashi, J. Volatile 1-octen-3-ol induces a defensive response in Arabidopsis thaliana. J. Gen. Plant Pathol. 2007, 73, 35–37. [Google Scholar] [CrossRef]
- Choi, H.K.; Song, G.C.; Yi, H.S.; Ryu, C.M. Field evaluation of the bacterial volatile derivative 3-pentanol in priming for induced resistance in pepper. J. Chem. Ecol. 2014, 40, 882–892. [Google Scholar] [CrossRef]
- Szczech, M.; Nawrocka, J.; Felczyński, K.; Małolepsza, U.; Sobolewski, J.; Kowalska, B.; Maciorowski, R.; Jas, K.; Kancelista, A. Trichoderma atroviride TRS25 isolate reduces downy mildew and induces systemic defence responses in cucumber in field conditions. Sci. Hortic. 2017, 224, 17–26. [Google Scholar] [CrossRef]
- Tonelli, M.L.; Furlan, A.; Taurian, T.; Castro, S.; Fabra, A. Peanut priming induced by biocontrol agents. Physiol. Mol. Plant Pathol. 2011, 75, 100–105. [Google Scholar] [CrossRef]
- Ghazalibiglar, H.; Hampton, J.G.; de Jong, E.V.Z.; Holyoake, A. Is induced systemic resistance the mechanism for control of black rot in Brassica oleracea by a Paenibacillus sp.? Biol. Control 2016, 92, 195–201. [Google Scholar] [CrossRef]
- Backer, R.; Rokem, J.S.; Ilangumaran, G.; Lamont, J.; Praslickova, D.; Ricci, E.; Subramanian, S.; Smith, D.L. Plant growth-promoting rhizobacteria: Context, mechanisms of action, and roadmap to commercialization of biostimulants for sustainable agriculture. Front. Plant Sci. 2018, 9, 1473. [Google Scholar] [CrossRef]
- Bashan, Y.; de-Bashan, L.E.; Prabhu, S.R.; Hernandez, J.P. Advances in plant growth-promoting bacterial inoculant technology: Formulations and practical perspectives (1998–2013). Plant Soil 2014, 37, 1–33. [Google Scholar] [CrossRef]
- Tabassum, B.; Khan, A.; Tariq, M.; Ramzan, M.; Khan, M.S.I.; Shahid, N.; Aaliya, K. Bottlenecks in commercialisation and future prospects of PGPR. Appl. Soil Ecol. 2017, 121, 102–117. [Google Scholar] [CrossRef]
- Nadeem, S.M.; Ahmad, M.; Zahir, Z.A.; Javaid, A.; Ashraf, M. The role of mycorrhizae and plant growth promoting rhizobacteria (PGPR) in improving crop productivity under stressful environments. Biotechnol. Adv. 2014, 32, 429–448. [Google Scholar] [CrossRef] [PubMed]
- Adesemoye, A.O.; Torbert, H.A.; Kloepper, J.W. Plant growth-promoting rhizobacteria allow reduced application rates of chemical fertilizers. Microb. Ecol. 2009, 58, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Finkel, O.M.; Castrillo, G.; Paredes, S.H.; González, I.S.; Dangl, J.L. Understanding and exploiting plant beneficial microbes. Curr. Opin. Plant Biol. 2017, 38, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Ipek, M.; Pirlak, L.; Esitken, A.; Figen Dönmez, M.; Turan, M.; Sahin, F. Plant growth-promoting rhizobacteria (PGPR) increase yield, growth and nutrition of strawberry under high-calcareous soil conditions. J. Plant Nutr. 2014, 37, 990–1001. [Google Scholar] [CrossRef]
- Jefwa, J.; Vanlauwe, B.; Coyne, D.; Van Asten, P.; Gaidashova, S.; Rurangwa, E.; Mwashasha, M.; Elsen, A. Benefits and potential use of arbuscular mycorrhizal fungi (AMF) in banana and plantain (Musa spp.) systems in Africa. Acta Hortic. 2010, 879, 479–486. [Google Scholar] [CrossRef]
- Ruzzi, M.; Aroca, R. Plant growth-promoting rhizobacteria act as biostimulants in horticulture. Sci. Hortic. 2015, 196, 124–134. [Google Scholar] [CrossRef]
- Bashan, Y.; de-Bashan, L.E.; Prabhu, S.R. Superior polymeric formulations and emerging innovative products of bacterial inoculants for sustainable agriculture and the environment. In Agriculturally Important Microorganisms; Singh, H., Sarma, B., Keswani, C., Eds.; Springer: Singapore, 2016; pp. 15–46. [Google Scholar]
- Strigul, N.S.; Kravchenko, L.V. Mathematical modeling of PGPR inoculation into the rhizosphere. Environ. Modell. Softw. 2006, 21, 1158–1171. [Google Scholar] [CrossRef]
- Vejan, P.; Abdullah, R.; Khadiran, T.; Ismail, S.; Nasrulhaq Boyce, A. Role of plant growth promoting rhizobacteria in agricultural sustainability—A review. Molecules 2016, 21, 573. [Google Scholar] [CrossRef]
- Etesami, H.; Maheshwari, D.K. Use of plant growth promoting rhizobacteria (PGPRs) with multiple plant growth promoting traits in stress agriculture: Action mechanisms and future prospects. Ecotox. Environ. Safe 2018, 156, 225–246. [Google Scholar] [CrossRef]
- Mazzola, M.; Freilich, S. Prospects for biological soilborne disease control: Application of indigenous versus synthetic microbiomes. Phytopathology 2017, 107, 256–263. [Google Scholar] [CrossRef]
- Purahong, W.; Orrù, L.; Donati, I.; Perpetuini, G.; Cellini, A.; Lamontanara, A.; Michelotti, V.; Tacconi, G.; Spinelli, F. Plant microbiome and its link to plant health: Host species, organs and Pseudomonas syringae pv. actinidiae infection shaping bacterial phyllosphere communities of kiwifruit plants. Front. Plant Sci. 2018, 9, 1563. [Google Scholar] [PubMed]
- Berendsen, R.L.; Pieterse, C.M.J.; Bakker, P.A.H.M. The rhizosphere microbiome and plant health. Trends Plant Sci. 2012, 17, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Lemanceau, P.; Blouin, M.; Muller, D.; Moënne-Loccoz, Y. Let the core microbiota be functional. Trends Plant Sci. 2017, 22, 583–595. [Google Scholar] [CrossRef] [PubMed]
- Bouffaud, M.L.; Renoud, S.; Dubost, A.; Moënne-Loccoz, Y.; Muller, D. 1-Aminocyclopropane-1-carboxylate deaminase producers associated to maize and other Poaceae species. Microbiome 2018, 6, 114. [Google Scholar] [CrossRef] [PubMed]
- Almario, J.; Moënne-Loccoz, Y.; Muller, D. Monitoring of the relation between 2, 4-diacetylphloroglucinol-producing Pseudomonas and Thielaviopsis basicola populations by real-time PCR in tobacco black root-rot suppressive and conducive soils. Soil Biol. Biochem. 2013, 57, 144–155. [Google Scholar] [CrossRef]
- Ahkami, A.H.; White, R.A., III; Handakumbura, P.P.; Jansson, C. Rhizosphere engineering: Enhancing sustainable plant ecosystem productivity. Rhizosphere 2017, 3, 233–243. [Google Scholar] [CrossRef]
- Ricci, M.; Tilbury, L.; Daridon, B.; Sukalac, K. General principles to justify plant biostimulant claims. Front. Plant Sci. 2019, 10, 494. [Google Scholar] [CrossRef]
| Microorganism | Crop Plant | Mode of Action | Ref. |
|---|---|---|---|
| Protection from High Temperatures | |||
| Paraburkholderia phytofirmans | Potato | ACC deaminase production | [51] |
| Mycorrhizae | Tomato | Reduction in lipid peroxidation and H2O2, higher ROS scavenging activity in leaves and roots | [50] |
| Bacillus amyloliquefaciens, Azospirillum brasilense | Wheat | ROS reduction, pre-activation of heat shock proteins | [79] |
| Bacillus aryabhatthai SRBO2 | Soy | ABA production | [80] |
| Pseudomonas putida AKMP7, Pseudomonas sp. AKMP6 | Wheat, sorghum | ROS reduction, increase in proline, chlorophyll, sugar, starch, amino acid and protein content, production of phytohormones | [81,82] |
| Protection from Cold and Frost | |||
| Ps. fluorescens A506 | Apple and pear | Competition with INA+ bacteria | [70] |
| Pantoea dispersa 1A, Serratia marcescens SRM, Pseudomonas spp. PGERs17, NARs9 | Wheat | ACC deaminase production | [56,72,73,74] |
| Paraburkholderia phytofirmans | Grapevine | ACC deaminase production | [76,77] |
| Microorganism | Crop Plant | Mode of Action | Ref. |
|---|---|---|---|
| Phoma glomerata, Penicillium sp., Exophiala sp., Paecilomyces formosus, Glomus intraradices | Cucumber, bean | Greater soil exploration by roots or by fungal hyphae and better water root conductivity | [101,106] |
| Pseudomonas chlororaphis TSAU13, Funneliformis mosseae | Tomato, cucumber, orange | IAA production | [115,116] |
| Burkholderia, Promicromonospora, Acinetobacter, Pseudomonas spp. | Cucumber | Gibberellin production | [119] |
| Bacillus subtilis | Lettuce | Cytokinin production | [117] |
| Ps. putida H-2-3 | Soybean | ABA production | [120] |
| Achromobacter piechaudii ARV8, B. licheniformis K11, Pseudomonas spp., Ps. fluorescens TDK1 | Tomato, pepper, pea, peanut | ACC deaminase | [122,123,149,150] |
| Glomus intraradices | Carrot, soybean, lettuce | Increased aquaporin activity | [107,151] |
| Pseudomonas sp. | Basil | Increased antioxidant protection | [125] |
| Ps. putida, Ps. aeruginosa PF23, Glomus mosseae, G. versiforme, G. diaphanum | Sunflower, orange | EPS production | [98,99,104] |
| Bacillus polymyxa, Glomus intraradices, G. versiforme | Tomato, soybean, tangerine | Osmolyte production | [104,124,129,152] |
| Microorganism | Plant/Pathogen(s) | Microbial Elicitor | Signaling Pathway | Ref. |
|---|---|---|---|---|
| Bacillus pumilus SE34 | Tomato/Phytophtora infestans | depending on ET/JA; SA independent | [179] | |
| B. subtilis S499 | Bean/Botritis cinerea | Cyclic lipopeptides (surfactin and fengicine) | [163] | |
| Burkholderia gladioli | Cucumber/Colletotrichum orbiculare | Exopolysaccharides | [180] | |
| Pseudomonas fluorescens SS101 | Tomato/Phytophtora infestans | Cyclic lipopeptides | SA-independent | [181] |
| Ps. fluorescens WCS374 | Radicchio/Fusarium oxysporum f. sp. raphani | Pigment (pseudobactin), lipopolysaccharides | [182] | |
| Rhizobium etli G12 | Potato/Globodera pallida | lipopolysaccharides | [183,184] | |
| Ps. fluorescens WCS417 | Eucalyptus/Ralstonia solanacearum | Lipopolysaccharides and siderophores | [185] | |
| Serratia liquefaciens MG1 | Tomato/Alternaria alternata | N-acyl homoserine lactones | Probably depending on SA and ET | [161] |
| B. amyloliquefaciens IN937a | Arabidopsis/Erwinia carotovora | VOCs (2R,3R-butanediol) | depending on ET/JA; NPR1/SA-independent | [160] |
| Paenibacillus polymixa BMP-11 | Arabidopsis/Phytophthora capsici, Alternaria brassicicola, Botrytis cinerea, Colletotrichum capsici, Fusarium oxysoprum | VOCs (1-octen-3-ol) | [186,187] | |
| B. amyloliquefaciens IN937a | Pepper, cucumber/Xanthomonas axonopodis pv. vesicatoria | VOCs (3-pentanol) | [188] | |
| Trichoderma atroviride TRS25 | Cucumber/Pseudoperonospora cubensis | Probably depending on SA and JA/ET | [189] | |
| Bacillus sp. CHEP5 e Pseudomonas sp. BREN6 | Peanut/Sclerotium rolfsii | Increased ACC conversion capacity | [190] | |
| Paenibacillus P16 | Cabbage/Xanthomonas campestris | [191] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sangiorgio, D.; Cellini, A.; Donati, I.; Pastore, C.; Onofrietti, C.; Spinelli, F. Facing Climate Change: Application of Microbial Biostimulants to Mitigate Stress in Horticultural Crops. Agronomy 2020, 10, 794. https://doi.org/10.3390/agronomy10060794
Sangiorgio D, Cellini A, Donati I, Pastore C, Onofrietti C, Spinelli F. Facing Climate Change: Application of Microbial Biostimulants to Mitigate Stress in Horticultural Crops. Agronomy. 2020; 10(6):794. https://doi.org/10.3390/agronomy10060794
Chicago/Turabian StyleSangiorgio, Daniela, Antonio Cellini, Irene Donati, Chiara Pastore, Claudia Onofrietti, and Francesco Spinelli. 2020. "Facing Climate Change: Application of Microbial Biostimulants to Mitigate Stress in Horticultural Crops" Agronomy 10, no. 6: 794. https://doi.org/10.3390/agronomy10060794
APA StyleSangiorgio, D., Cellini, A., Donati, I., Pastore, C., Onofrietti, C., & Spinelli, F. (2020). Facing Climate Change: Application of Microbial Biostimulants to Mitigate Stress in Horticultural Crops. Agronomy, 10(6), 794. https://doi.org/10.3390/agronomy10060794







