Durum Wheat Seminal Root Traits within Modern and Landrace Germplasm in Algeria
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Root Phenotyping
2.2.1. Preparation of Seeds
2.2.2. The Rhizo-Slide System
2.2.3. Image Analysis
2.3. Statistical Analysis
3. Results
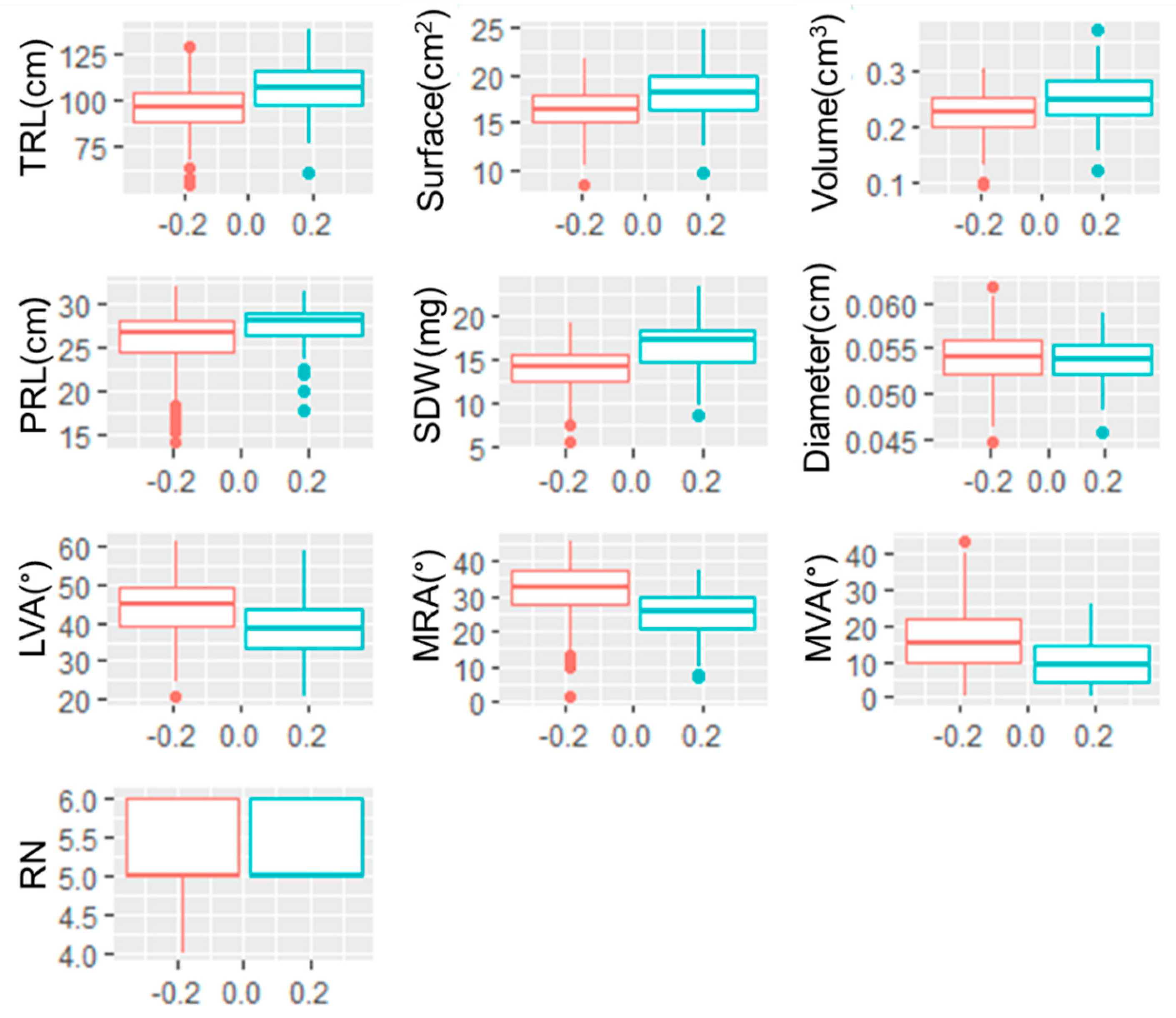
3.1. Genotypic Variability
3.2. Relationships between Traits
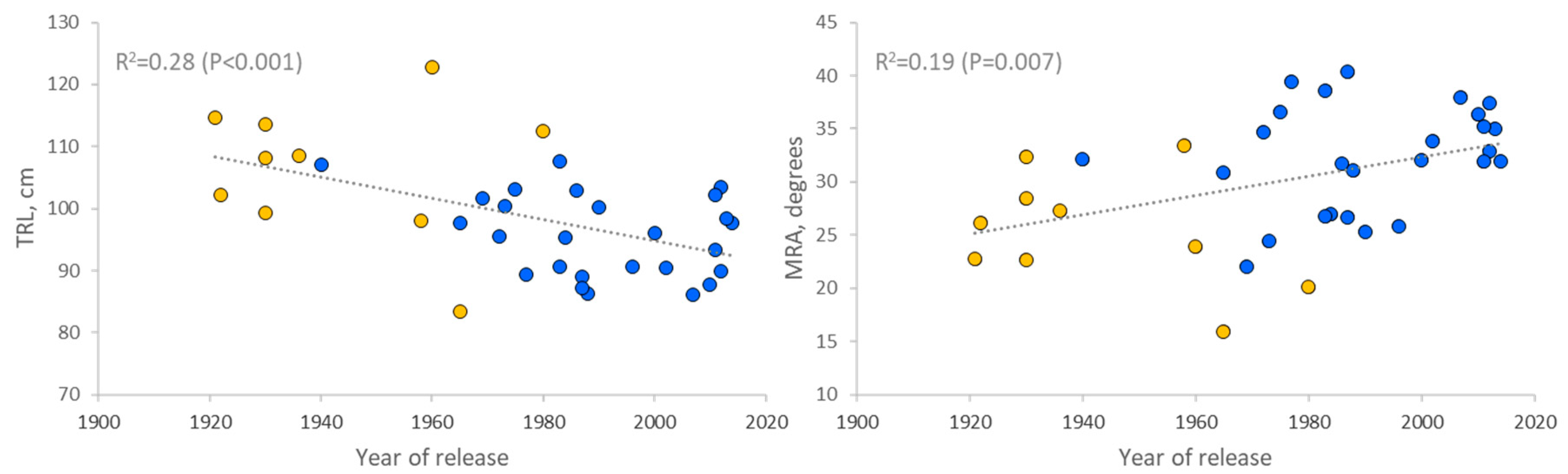
3.3. Time Trends of Root Traits
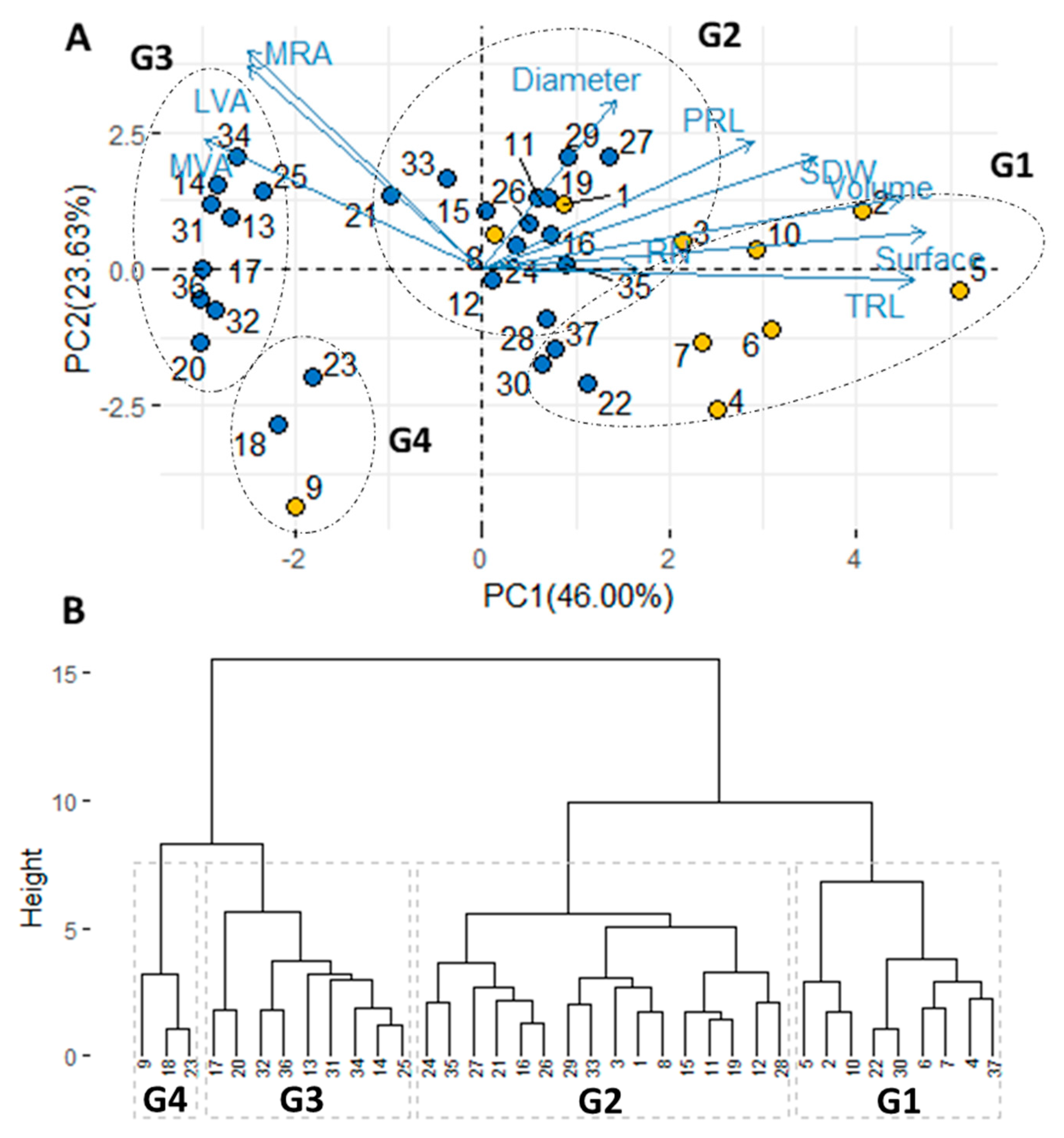
3.4. Grouping of Genotypes According to Root Traits
4. Discussion
4.1. Large Genotypic Variation for Seminal Root Traits
4.2. Classification of Durum Wheat Genotypes According to Root Morphology
4.3. Conclusions and Perspectives
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chochois, V.; Vogel, J.P.; Rebetzke, G.J.; Watt, M. Variation in adult plant phenotypes and partitioning among seed and stem-borne roots across Brachypodium distachyon accessions to exploit in breeding cereals for well-watered and drought environments. Plant Physiol. 2015, 168, 953–967. [Google Scholar] [CrossRef]
- Sinha, S.K.; Rani, M.; Kumar, A.; Kumar, S.; Venkatesh, K.; Mandal, P.K. Natural variation in root system architecture in diverse wheat genotypes grown under different nitrate conditions and root growth media. Theor. Exp. Plant Physiol. 2018, 30, 223–234. [Google Scholar] [CrossRef]
- Araki, H.; Iijima, M. Deep rooting in winter wheat: Rooting nodes of deep roots in two cultivars with deep and shallow root systems. Plant Prod. Sci. 2001, 4, 215–219. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Manske, G.G.B.; Vlek, P.L.G. Root Architecture-Wheat as a Model Plant. In Plant Roots: The Hidden Half, 3rd ed.; Waisel, Y., Eshel, A., Beeckman, T., Kafkafi, U., Eds.; Marcel Dekker: New York, NY, USA, 2002; pp. 249–259. [Google Scholar]
- Maccaferri, M.; El-Feki, W.; Nazemi, G.; Salvi, S.; Canè, M.A.; Colalongo, M.C.; Stefanelli, S.; Tuberosa, R. Prioritizing quantitative trait loci for root system architecture in tetraploid wheat. J. Exp. Bot. 2016, 67, 1161–1178. [Google Scholar] [CrossRef] [PubMed]
- Esau, K. Plant Anatomy, 2nd ed.; John Wiley: New York, NY, USA, 1965; 767p. [Google Scholar]
- Lynch, J. Root architecture and plant productivity. Plant Physiol. 1995, 109, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, J.; Pour-Aboughadareh, A.; Fabriki-Ourang, S.; Mehrabi, A.A.; Siddique, K.H.M. Screening wheat germplasm for seedling root architectural traits under contrasting water regimes: Potential sources of variability for drought adaptation. Arch. Agron. Soil Sci. 2018, 64, 1351–1365. [Google Scholar] [CrossRef]
- York, L.M.; Slack, S.; Bennett, M.J.; Foulkes, M.J. Wheat shovelomics I: A field phenotyping approach for characterizing the structure and function of root systems in tillering species. BioRxiv 2018, 280875. [Google Scholar] [CrossRef]
- Nguyen, V.L.; Stangoulis, J. Variation in root system architecture and morphology of two wheat genotypes is a predictor of their tolerance to phosphorus deficiency. Acta Physiol. Plant. 2019, 41, 109. [Google Scholar] [CrossRef]
- Bengough, A.G.; Gordon, D.C.; Al-Menaie, H.; Ellis, R.P.; Allan, D.; Keith, R.; Thomas, W.T.B.; Forster, B.P. Gel observation chamber for rapid screening of root traits in cereal seedlings. Plant Soil 2004, 262, 63–70. [Google Scholar] [CrossRef]
- Watt, M.; Moosavi, S.; Cunningham, S.C.; Kirkegaard, J.A.; Rebetzke, G.J.; Richards, R.A. A rapid, controlled-environment seedling root screen for wheat correlates well with rooting depths at vegetative, but not reproductive, stages at two field sites. Ann. Bot. 2013, 112, 447–455. [Google Scholar] [CrossRef]
- Richard, C.A.; Hickey, L.T.; Fletcher, S.; Jennings, R.; Chenu, K.; Christopher, J.T. High-throughput phenotyping of seminal root traits in wheat. Plant Methods 2015, 11, 13. [Google Scholar] [CrossRef] [PubMed]
- González, J.M.; Friero, E.; Selfa, L.; Froilán, S.; Jouve, N.A. Comparative study of root system architecture in seedlings of Brachypodium spp. using three plant growth supports. Cereal Res. Commun. 2016, 44, 69–78. [Google Scholar] [CrossRef]
- Adeleke, E.; Millas, R.; McNeal, W.; Faris, J.; Taheri, A. Variation analysis of root system development in wheat seedlings using root phenotyping system. Agronomy 2020, 10, 206. [Google Scholar] [CrossRef]
- Richard, C.; Christopher, J.; Chenu, K.; Borrell, A.; Christopher, M.; Hickey, L. Selection in early generations to shift allele frequency for seminal root angle in wheat. Plant Genome 2018, 11, 170071. [Google Scholar] [CrossRef] [PubMed]
- El Hassouni, K.; Alahmad, S.; Belkadi, B.; Filali-Maltouf, A.; Hickey, L.T.; Bassi, F.M. Root system architecture and its association with yield under different water regimes in durum wheat. Crop Sci. 2018, 58, 2331–2346. [Google Scholar] [CrossRef]
- Bai, C.; Liang, Y.; Hawkesford, M.J. Identification of QTLs associated with seedling root traits and their correlation with plant height in wheat. J. Exp. Bot. 2013, 64, 1745–1753. [Google Scholar] [CrossRef]
- Tuberosa, R.; Salvi, S.; Sanguineti, M.C.; Landi, P.; Maccaferri, M.; Conti, S. Mapping QTLs regulating morpho-physiological traits and yield: Case studies, shortcomings and perspectives in drought-stressed maize. Ann. Bot. 2002, 89, 941–963. [Google Scholar] [CrossRef]
- Tuberosa, R.; Sanguineti, M.C.; Landi, P.; Giuliani, M.M.; Salvi, S.; Conti, S. Identification of QTLs for root characteristics in maize grown in hydroponics and analysis of their overlap with QTLs for grain yield in the field at two water regimes. Plant Mol. Biol. 2002, 48, 697–712. [Google Scholar] [CrossRef]
- Manschadi, A.M.; Christopher, J.; De Voil, P.; Hammer, G.L. The role of root architectural traits in adaptation of wheat to water-limited environments. Funct. Plant Biol. 2006, 33, 823–837. [Google Scholar] [CrossRef]
- Paez-Garcia, A.; Motes, C.M.; Scheible, W.R.; Chen, R.; Blancaflor, E.B.; Monteros, M.J. Root traits and phenotyping strategies for plant improvement. Plants 2015, 4, 334–355. [Google Scholar] [CrossRef]
- Løes, A.K.; Gahoonia, T.S. Genetic variation in specific root length in Scandinavian wheat and barley accessions. Euphytica 2004, 137, 243–249. [Google Scholar] [CrossRef]
- Manschadi, A.M.; Hammer, G.L.; Christopher, J.T.; Devoil, P. Genotypic variation in seedling root architectural traits and implications for drought adaptation in wheat (Triticum aestivum L.). Plant Soil 2008, 303, 115–129. [Google Scholar] [CrossRef]
- Ruiz, M.; Giraldo, P.; González, J.M. Phenotypic variation in root architecture traits and their relationship with eco-geographical and agronomic features in a core collection of tetraploid wheat landraces (Triticum turgidum L.). Euphytica 2018, 214, 54. [Google Scholar] [CrossRef]
- Li, T.; Ma, J.; Zou, Y.; Chen, G.; Ding, P.; Zhang, H.; Yang, C.; Mu, Y.; Tang, H.; Liu, Y.; et al. Quantitative trait loci for seeding root traits and the relationships between root and agronomic traits in common wheat. Genome 2020, 63, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Alahmad, S.; El Hassouni, K.; Bassi, F.M.; Dinglasan, E.; Youssef, C.; Quarry, G.; Able, J.A.; Christopher, J.A. Major root architecture QTL responding to water limitation in durum wheat. Front. Plant Sci. 2019, 10, 436. [Google Scholar] [CrossRef]
- Ali, M.L.; Luetchens, J.; Nascimento, J.; Shaver, T.M.; Kruger, G.R.; Lorenz, A.J. Genetic variation in seminal and nodal root angle and their association with grain yield of maize under water-stressed field conditions. Plant Soil 2015, 397, 213–225. [Google Scholar] [CrossRef]
- Nakamoto, T.; Oyanagi, A. The direction of growth of seminal roots of Triticum aestivum L. and experimental modification thereof. Ann. Bot. 1994, 73, 363–367. [Google Scholar] [CrossRef]
- Masi, C.E.A.; Maranville, J.W. Evaluation of sorghum root branching using fractals. J. Agric. Sci. 1998, 131, 259–265. [Google Scholar] [CrossRef]
- Liao, H.; Rubio, G.; Yan, X.; Cao, A.; Brown, K.M.; Lynch, J.P. Effect of phosphorus availability on basal root shallowness in common bean. Plant Soil 2001, 232, 69–79. [Google Scholar] [CrossRef]
- Lynch, J.P.; Brown, K.M. Topsoil foraging–an architectural adaptation of plants to low phosphorus availability. Plant Soil 2001, 237, 225–237. [Google Scholar] [CrossRef]
- Roselló, M.; Royo, C.; Sanchez-Garcia, M.; Soriano, J.M. Genetic dissection of the seminal root system architecture in Mediterranean durum wheat landraces by genome-wide association study. Agronomy 2019, 9, 364. [Google Scholar] [CrossRef]
- El-Beltagy, A.; Madkour, M. Impact of climate change on arid lands agriculture. Agric. Food Secur. 2012, 1, 3. [Google Scholar] [CrossRef]
- Nazco, R.; Villegas, D.; Ammar, K.; Pena, R.J.; Moragues, M.; Royo, C. Can Mediterranean durum wheat landraces contribute to improved grain quality attributes in modern cultivars? Euphytica 2012, 185, 1–17. [Google Scholar] [CrossRef]
- Lopes, M.S.; El-Basyoni, I.; Baenziger, P.S.; Singh, S.; Royo, C.; Ozbek, K.; Aktas, H.; Ozer, E.; Ozdemir, F.; Manickavelu, A.; et al. Exploiting genetic diversity from landraces in wheat breeding for adaptation to climate change. J. Exp. Bot. 2015, 66, 3477–3486. [Google Scholar] [CrossRef]
- Araujo, S.S.; Beebe, S.; Crespi, M.; Delbreil, B.; González, E.M.; Gruber, V.; Lejeune-Henaut, I.; Link, W.; Monteros, M.J.; Prats, E.; et al. Abiotic stress responses in legumes: Strategies used to cope with environmental challenges. Crit. Rev. Plant Sci. 2015, 34, 237–280. [Google Scholar] [CrossRef]
- Rao, I.M.; Beebe, S.E.; Polania, J.; Grajales, M.; Cajiao, C.; Ricaurte, J.; García, R.; Rivera, M. Evidence for genotypic differences among elite lines of common bean in the ability to remobilize photosynthate to increase yield under drought. J. Agric. Sci. 2017, 155, 857–875. [Google Scholar] [CrossRef]
- Lynch, J.P. Steep, cheap and deep: An ideotype to optimize water and N acquisition by maize root systems. Ann. Bot. 2013, 112, 347–357. [Google Scholar] [CrossRef]
- Aniol, A. Induction of aluminum tolerance in wheat seedlings by low doses of aluminum in the nutrient solution. Plant Physiol. 1984, 75, 551–555. [Google Scholar] [CrossRef]
- Lobet, G.; Pagès, L.; Draye, X. A novel image-analysis toolbox enabling quantitative analysis of root system architecture. Plant Physiol. 2011, 157, 29–39. [Google Scholar] [CrossRef]
- Payne, R.W.; Murray, D.A.; Harding, S.A.; Baird, D.B.; Soutar, D.M. GenStat for Windows Introduction, 12th ed.; VSN International: Hemel Hempstead, UK, 2009. [Google Scholar]
- Le, S.; Josse, J.; Husson, F. FactoMineR: An R Package for Multivariate Analysis. J. Stat. 2008, 25, 18. [Google Scholar]
- Kassambara, A.; Mundt, F. Package ‘factoextra’. Extr. Vis. Results Multivar. Data Anal. 2017, 76. Available online: http://www.sthda.com/english/rpkgs/factoextra (accessed on 2 March 2020).
- Devaiah, B.N.; Nagarajan, V.K.; Raghothama, K.G. Phosphate homeostasis and root development in Arabidopsis is synchronized by the zinc finger transcription factor ZAT6. Plant Physiol. 2007, 145, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, A.; Bruno, I.P.; Franzini, V.I.; Marcante, N.C.; Benitiz, L.; Muraoka, T. Phosphorus uptake efficiency, root morphology and architecture in Brazilian wheat cultivars. J. Radioanal. Nucl. Chem. 2016, 307, 1055–1063. [Google Scholar] [CrossRef]
- Sanguineti, M.C.; Li, S.; Maccaferri, M.; Corneti, S.; Rotondo, F.; Chiari, T.; Tuberosa, R. Genetic dissection of seminal root architecture in elite durum wheat germplasm. Ann. Appl. Biol. 2007, 151, 291–305. [Google Scholar] [CrossRef]
- Cane, M.A.; Maccaferri, M.; Nazemi, G.; Salvi, S.; Francia, R.; Colalongo, C.; Tuberosa, R. Association mapping for root architectural traits in durum wheat seedlings as related to agronomic performance. Mol. Breed. 2014, 34, 1629–1645. [Google Scholar] [CrossRef]
- Robertson, B.M.; Waines, J.G.; Gill, B.S. Genetic variability for seedling root number in wild and domesticated wheats. Crop Sci. 1979, 19, 843–847. [Google Scholar] [CrossRef]
- Grando, S.; Ceccarelli, S. Seminal root morphology and coleoptile length in wild (Hordeum vulgare ssp. spontaneum) and cultivated (Hordeum vulgare ssp. vulgare) barley. Euphytica 1995, 86, 73–80. [Google Scholar]
- O’Brien, L. Genetic variability of root growth in wheat (Triticum aestivum L.). Aust. J. Agric. Res. 1979, 30, 587–595. [Google Scholar] [CrossRef]
- Narayanan, S.; Mohan, A.; Gill, K.S.; Prasad, P.V. Variability of root traits in spring wheat germplasm. PLoS ONE 2014, 9, e100317. [Google Scholar] [CrossRef]
- Bodner, G.; Leitner, D.; Nakhforoosh, A.; Sobotik, M.; Moder, K.; Kaul, H.P. A statistical approach to root system classification. Front. Plant Sci. 2013, 4, 292. [Google Scholar] [CrossRef]
- Royo, C.; Nazco, R.; Villegas, D. The climate of the zone of origin of Mediterranean durum wheat (Triticum durum Desf.) landraces affects their agronomic performance. Genet. Resour. Crop Evol. 2014, 61, 1345–1358. [Google Scholar] [CrossRef]
- Soriano, J.M.; Villegas, D.; Aranzana, M.J.; Del Moral, L.F.G.; Royo, C. Genetic structure of modern durum wheat cultivars and Mediterranean landraces matches with their agronomic performance. PLoS ONE 2016, 11, e0160983. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.H.; Weiner, J.; Yu, M.X.; Li, F.M. Evolutionary agroecology: Trends in root architecture during wheat breeding. Evol. Appl. 2019, 12, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Zhang, D.W.; Li, F.M.; Fan, X.W.; Ma, Q.; Turner, N.C. Drought stress: Soil water availability alters the inter- and intra-cultivar competition of three spring wheat cultivars bred in different eras. J. Agron. Crop Sci. 2010, 196, 323–335. [Google Scholar] [CrossRef]
- De Parseval, H.; Barot, S.; Gignoux, J.; Lata, J.; Raynaud, X. Modelling facilitation or competition within a root system: Importance of the overlap of root depletion and accumulation zones. Plant Soil 2017, 419, 97–111. [Google Scholar] [CrossRef]
- Weiner, J.; Du, Y.L.; Zhang, C.; Qin, X.L.; Li, F.M. Evolutionary agroecology: Individual fitness and population yield in wheat (Triticum aestivum). Ecology 2017, 98, 2261–2266. [Google Scholar] [CrossRef]
- Kembel, S.W.; Cahill, J.F. Independent evolution of leaf and root traits within and among temperate grassland plant communities. PLoS ONE 2011, 6, e19992. [Google Scholar] [CrossRef]
- Ellis, M.H.; Rebetzke, G.J.; Chandler, P.; Bonnett, D.; Spielmeyer, W.; Richards, R.A. The effect of different height reducing genes on the early growth of wheat. Funct. Plant Biol. 2004, 31, 583–589. [Google Scholar] [CrossRef]
| Nº | Genotype | Type/Pedigree | Origin | Year of Release |
|---|---|---|---|---|
| 1 | Beliouni | Landrace | Algeria | 1958 |
| 2 | Bidi 17 | Landrace | Algeria | 1930 |
| 3 | Djenah Khotifa | Landrace | North Africa | 1955 |
| 4 | Gloire de Montgolfier | Landrace | Algeria | 1960 |
| 5 | Guemgoum R’ khem | Landrace | Algeria | 1960 |
| 6 | Hedba 3 | Landrace | Algeria | 1921 |
| 7 | Langlois | Landrace | Algeria | 1930 |
| 8 | Mohammed Ben Bachir (MBB) | Landrace | Algeria | 1930 |
| 9 | Montpellier | Landrace | Algeria | 1965 |
| 10 | Oued Zenati 368 | Landrace | Algeria | 1936 |
| 11 | Acsad 65 | Gerardo-vz-469/3/Jori-1//Nd-61-130/Leeds | ACSAD | 1984 |
| 12 | Altar 84 | Ruff/Flamingo,mex//Mexicali-75/3/Shearwater | CYMMIT | 1984 |
| 13 | Ammar 6 | Lgt3/4/Bicre/3/Ch1//Gaviota/Starke | ICARDA | 2010 |
| 14 | Bousselem | Heider//Martes/Huevos de oro | ICARDA | 2007 |
| 15 | Boutaleb | Hedba 3/Ofanto | Algeria | 2013 |
| 16 | Capeiti | Eiti*6/Senatore-Cappelli | Italy | 1940 |
| 17 | Chen’s | Shearwater(sib)/(sib)Yavaros-79 | CYMMIT | 1983 |
| 18 | Ciccio | Appulo/Valnova(f6)//(f5)Valforte/Patrizio | Italy | 1996 |
| 19 | Cirta | Hedba-3/Gerardo-vz-619 | Algeria | 2000 |
| 20 | Core | Platani/Gianni | Italy | 2008 |
| 21 | GTA Dur | Crane/4/Polonicum PI185309//T.glutin enano/2* Tc60/3/Gll | CIMMYT | 1972 |
| 22 | INRAT 69 | Mahmoudi/(bd-2777)Kyperounda | Tunisia | 1969 |
| 23 | Korifla | Durum-dwarf-s-15/Crane//Geier | ICARDA | 1987 |
| 24 | Mansourah | Bread wheat/MBB | Algeria | 2012 |
| 25 | Massinissa | Ofanto/Bousselem | Algeria | 2012 |
| 26 | Megress | Ofanto/Waha//MBB | Algeria | 2007 |
| 27 | Mexicali 75 | Gerardo-vz-469/3/Jori(sib)//Nd-61-130/Leeds | CIMMYT | 1975 |
| 28 | Ofanto | Ademelio/Appulo | Italy | 1990 |
| 29 | Oued El Berd | Gta dur/Ofanto | Algeria | 2013 |
| 30 | Polonicum | Triticum polinicum/Zenati boulette 1953-58 | France | 1973 |
| 31 | Sahell | Cit”s”/4/Tace/4*tc//2*zb/wls/3/aa”s’’/5/Ruff”s”/Albe”s” | CYMMIT | 1977 |
| 32 | Simeto | Capeiti-8/Valnova | Italy | 1988 |
| 33 | Sitifis | Bousselam/Ofanto | Algeria | 2011 |
| 34 | Vitron | Turkey77/3/Jori/Anhinga//Flamingo | Spain | 1987 |
| 35 | Waha | Plc/Ruff//Gta’s/3/Rolette | ICARDA | 1986 |
| 36 | Wahbi | Bidi 17/Waha//Bidi 17 | Algeria | 2002 |
| 37 | ZB × Fg | Zb/fg‘‘s’’ lk/3/ko 120/4/Ward cs 10604 | Algeria | 1983 |
| Descriptive Statistics | ANOVA | Correlation | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Traits | Min | Mean | Max | CV | h2 | Genotype | Type | TRL | Surface | Volume | Diameter | PRL | SDW | MRA | LVA | MVA | RN |
| TRL (cm) | 54.28 | 98.49 | 137.22 | 13.51 | 0.90 | *** | *** | 1 | *** | *** | ns | *** | *** | *** | ** | *** | *** |
| Surface (cm2) | 8.25 | 16.77 | 24.82 | 14.74 | 0.90 | *** | *** | 0.95 | 1 | *** | *** | *** | *** | *** | ns | *** | *** |
| Volume (cm3) | 0.0962 | 0.2326 | 0.3721 | 17.03 | 0.90 | *** | *** | 0.83 | 0.97 | 1 | *** | *** | *** | *** | ns | ** | *** |
| Diameter (cm) | 0.0447 | 0.0538 | 0.0620 | 5.19 | 0.87 | *** | ns | 0.08 | 0.38 | 0.59 | 1 | *** | *** | *** | * | ** | *** |
| PRL (cm) | 14.32 | 26.51 | 32.06 | 10.76 | 0.90 | *** | *** | 0.55 | 0.55 | 0.52 | 0.17 | 1 | *** | ns | ** | *** | ns |
| SDW (g) | 5.70 | 14.72 | 23.50 | 18.66 | 0.94 | *** | *** | 0.55 | 0.63 | 0.64 | 0.40 | 0.58 | 1 | ns | ** | * | ns |
| MRA (°) | 1.20 | 30.36 | 45.69 | 24.60 | 0.98 | *** | *** | −0.39 | −0.32 | −0.23 | 0.20 | −0.01 | 0.00 | 1 | *** | *** | ns |
| LVA (°) | 20.40 | 42.71 | 61.47 | 17.81 | 0.92 | *** | *** | −0.28 | −0.23 | −0.18 | 0.12 | 0.08 | 0.01 | 0.76 | 1 | *** | ns |
| MVA (°) | 0.00 | 14.10 | 43.49 | 59.82 | 0.80 | *** | *** | −0.25 | −0.20 | −0.15 | 0.12 | −0.27 | −0.13 | 0.62 | 0.25 | 1 | ** |
| RN (no.) | 4.00 | 5.32 | 6.00 | 9.13 | 0.87 | *** | ns | 0.37 | 0.32 | 0.26 | −0.22 | −0.01 | 0.06 | −0.09 | 0.02 | −0.14 | 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boudiar, R.; González, J.M.; Mekhlouf, A.; Casas, A.M.; Igartua, E. Durum Wheat Seminal Root Traits within Modern and Landrace Germplasm in Algeria. Agronomy 2020, 10, 713. https://doi.org/10.3390/agronomy10050713
Boudiar R, González JM, Mekhlouf A, Casas AM, Igartua E. Durum Wheat Seminal Root Traits within Modern and Landrace Germplasm in Algeria. Agronomy. 2020; 10(5):713. https://doi.org/10.3390/agronomy10050713
Chicago/Turabian StyleBoudiar, Ridha, Juan M. González, Abdelhamid Mekhlouf, Ana M. Casas, and Ernesto Igartua. 2020. "Durum Wheat Seminal Root Traits within Modern and Landrace Germplasm in Algeria" Agronomy 10, no. 5: 713. https://doi.org/10.3390/agronomy10050713
APA StyleBoudiar, R., González, J. M., Mekhlouf, A., Casas, A. M., & Igartua, E. (2020). Durum Wheat Seminal Root Traits within Modern and Landrace Germplasm in Algeria. Agronomy, 10(5), 713. https://doi.org/10.3390/agronomy10050713





