Challenges for a Sustainable Food Production System on Board of the International Space Station: A Technical Review
Abstract
1. Introduction
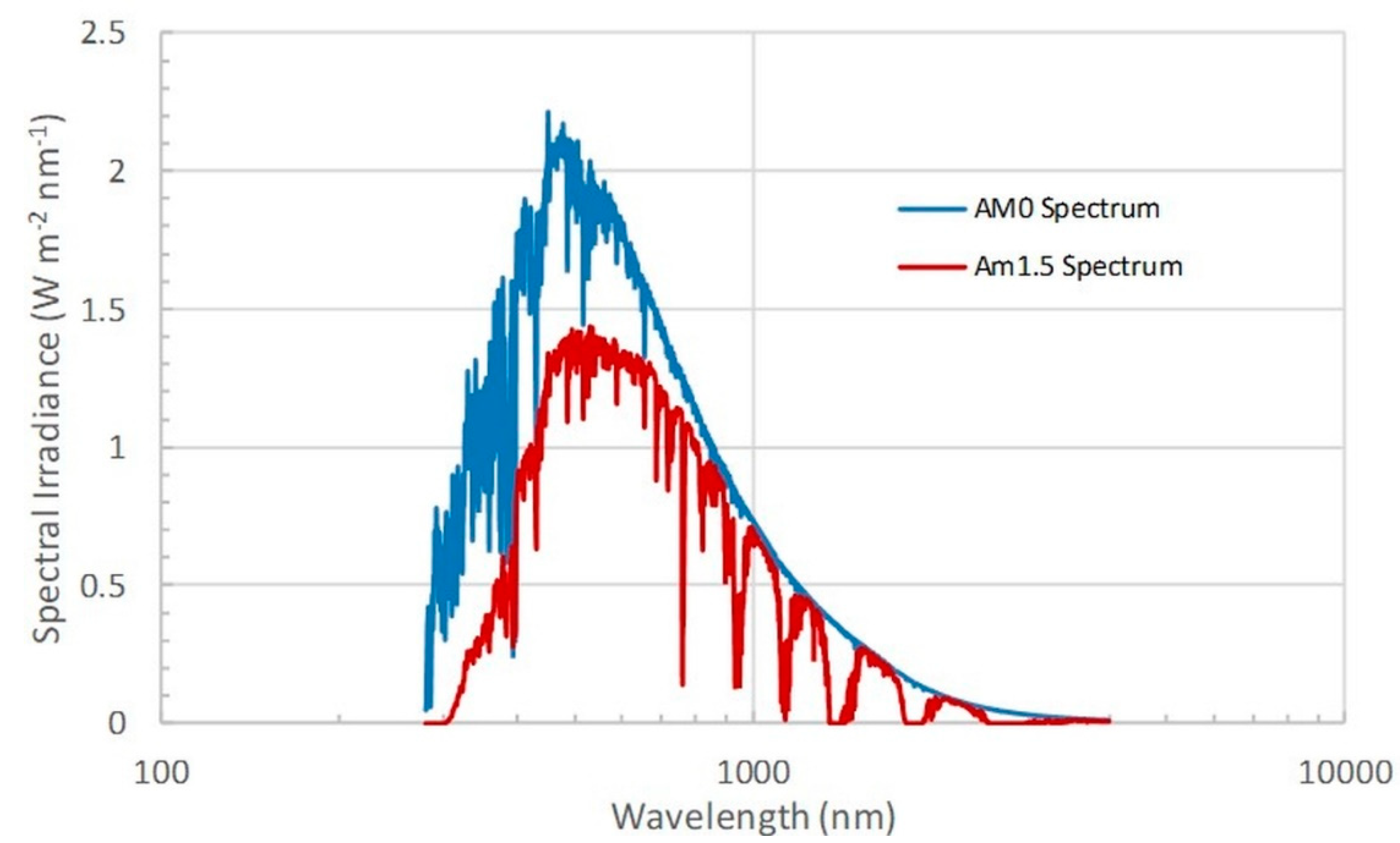
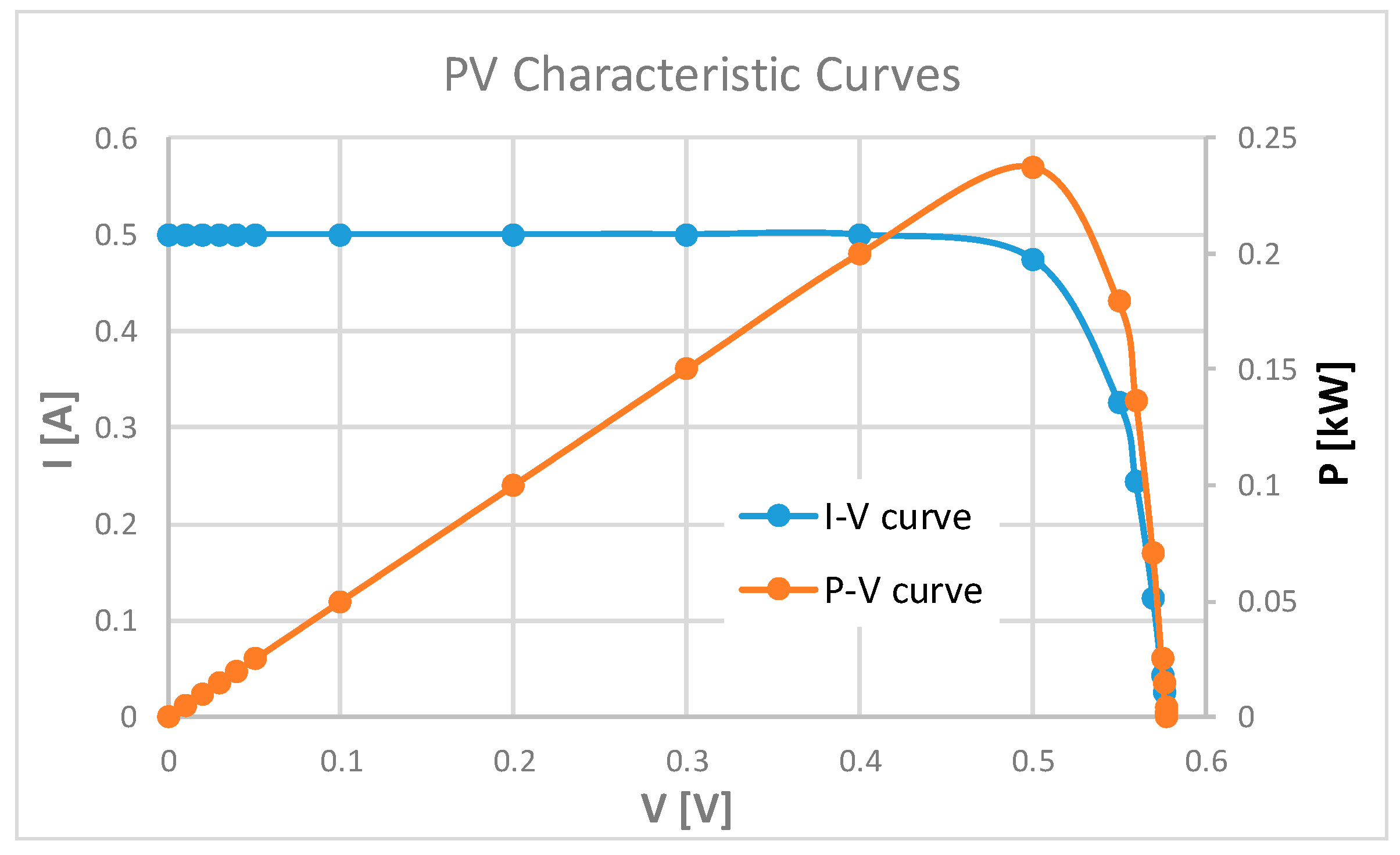
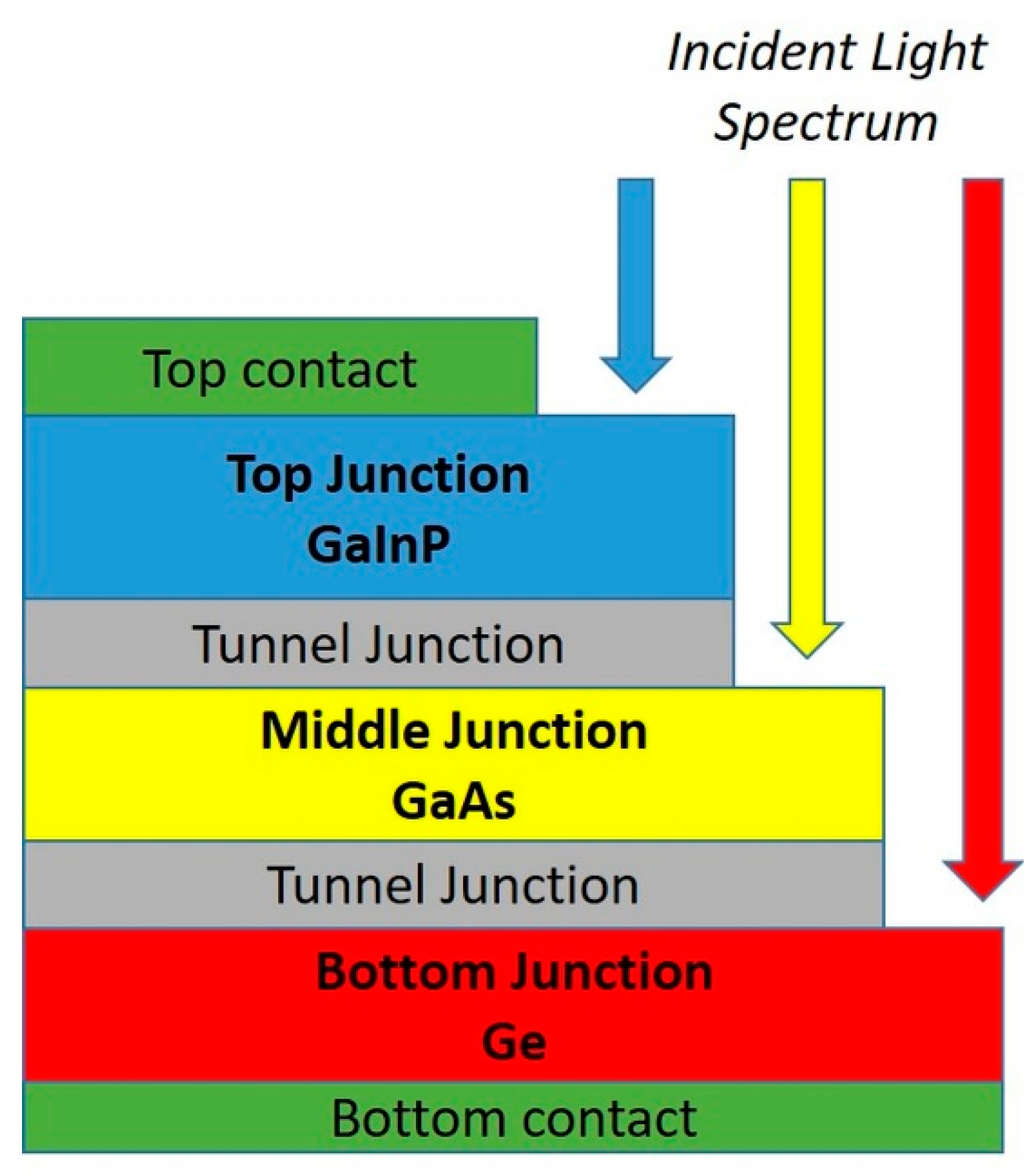
2. Physical Constraints and Energy Requirements for ISS Farming
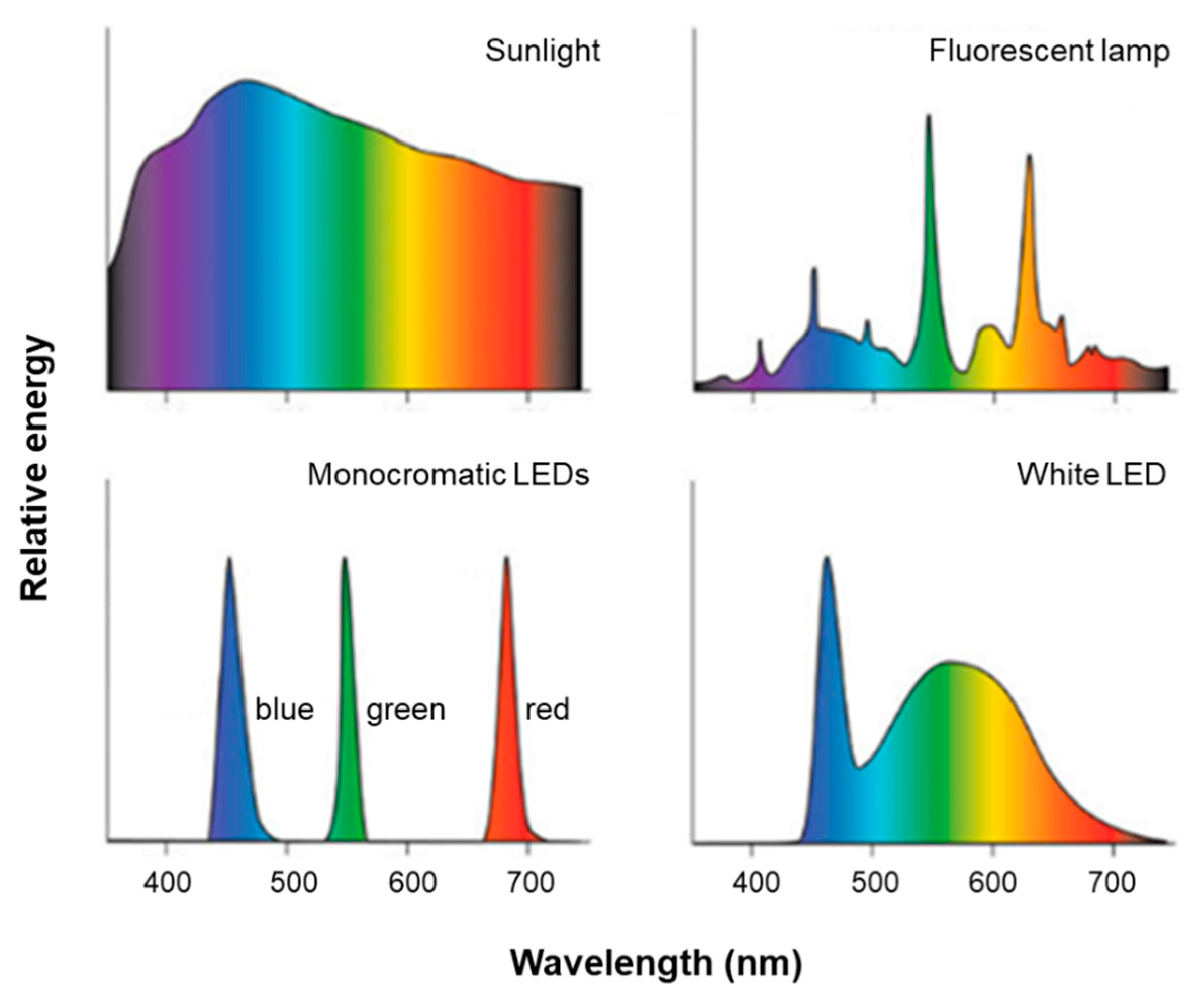
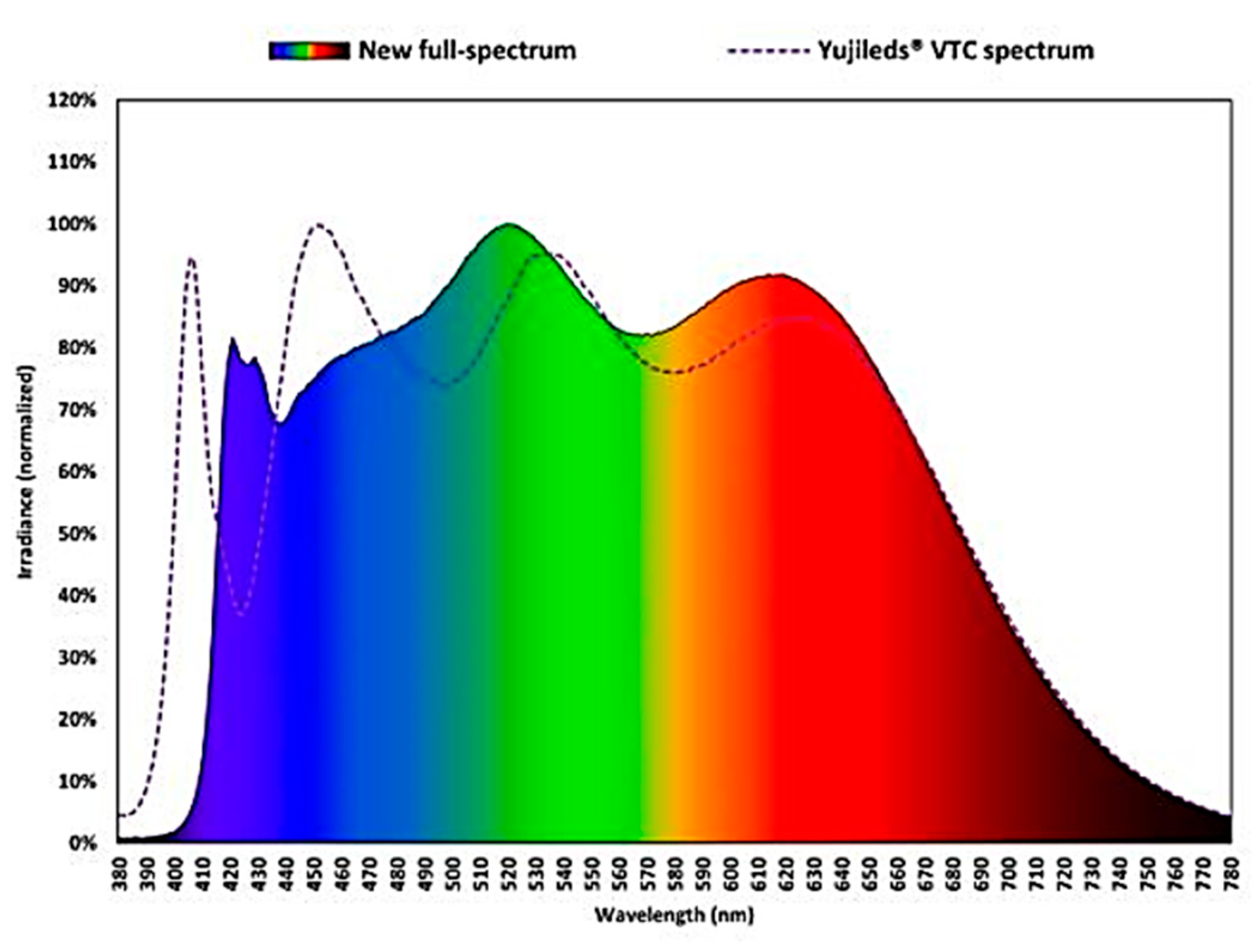
3. Light Sources for Space Farming on ISS
4. Criteria for the Selection of Potential Candidate Crops for ISS Farming
5. Hydroponics for Space Farming on ISS
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wheeler, R. Plants for human life support in space: From Myers to Mars. Gravit. Space Biol. 2011, 23. [Google Scholar]
- Harvey, B.; Zakutnyaya, O. Orbiting Space Stations. In Russian Space Probes: Scientific Discoveries and Future Missions; Praxis: New York, NY, USA, 2011; pp. 301–374. [Google Scholar]
- Zabel, P.; Bamsey, M.; Schubert, D.; Tajmar, M. Review and analysis of over 40 years of space plant growth systems. Life Sci. Space Res. 2016, 10, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Dueck, T.; Kempkes, F.; Meinen, E.; Stanghellini, C. Choosing crops for cultivation in space. ICES 2016, 206, 1–9. [Google Scholar]
- Kyriacou, M.C.; De Pascale, S.; Kyratzis, A.; Rouphael, Y. Microgreens as a Component of Space Life Support Systems: A Cornucopia of Functional Food. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Zwart, S.R. Chapter 3 Nutritional Biochemistry Of Spaceflight. In Advances in Clinical Chemistry; Elsevier: Amsterdam, The Netherlands, 2008; Volume 46, pp. 87–130. [Google Scholar]
- Cooper, M.; Perchonok, M.; Douglas, G.L. Initial assessment of the nutritional quality of the space food system over three years of ambient storage. npj Microgravity 2017, 3, 17. [Google Scholar] [CrossRef]
- Thirsk, R.; Kuipers, A.; Mukai, C.; Williams, D. The space-flight environment: The International Space Station and beyond. CMAJ 2009, 180, 1216–1220. [Google Scholar] [CrossRef]
- Palinkas, L. Psychosocial issues in long-term space flight: Overview. Gravit. Space Biol. Bull. Publ. Am. Soc. Gravit. Space Biol. 2001, 14, 25–33. [Google Scholar]
- Stein, T.P. Space flight and oxidative stress. Nutrition. 2002, 18, 867–871. [Google Scholar] [CrossRef]
- Cooper, M.R. Initial Nutritional Quality of ISS Space Food Provisions; NASA HRP Investigator’s Workshop: Galveston, TX, USA, 2013. [Google Scholar]
- Khodadad, C.L.M.; Hummerick, M.E.; Spencer, L.E.; Dixit, A.R.; Richards, J.T.; Romeyn, M.W.; Smith, T.M.; Wheeler, R.M.; Massa, G.D. Microbiological and Nutritional Analysis of Lettuce Crops Grown on the International Space Station. Front. Plant Sci. 2020, 11. [Google Scholar] [CrossRef]
- ESA/ESOC. Launch list. Space Res. Today 2019, 206, 53–64. [Google Scholar] [CrossRef]
- Genta, G.; Maffione, P.F. A graphical tool to design two-ways human Mars missions. Acta Astronaut. 2019, 154, 301–310. [Google Scholar] [CrossRef]
- Wolff, S.A.; Coelho, L.H.; Karoliussen, I.; Jost, A.-I.K. Effects of the Extraterrestrial Environment on Plants: Recommendations for Future Space Experiments for the MELiSSA Higher Plant Compartment. Life 2014, 4, 189–204. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.S.; Mao, R.X.; Zhang, L.L.; Tang, Y.K.; Li, Y.H. Progress and prospect of research on controlled ecological life support technique. REACH 2017, 6, 1–10. [Google Scholar] [CrossRef]
- Putra, P.A.; Yuliando, H. Soilless Culture System to Support Water Use Efficiency and Product Quality: A Review. Agric. Agric. Sci. Procedia 2015, 3, 283–288. [Google Scholar] [CrossRef]
- Lobascio, C.; Lamantea, M.; Cotronei, V.; Negri, B.; De Pascale, S.; Maggio, A.; Maffei, M.; Foti, M.; Palumberi, S. CAB: The bioregenerative life support system. A feasibility study on the survivability of humans in a long-duration space missions. Acta Hortic. 2008, 801, 465–472. [Google Scholar] [CrossRef]
- Wheeler, R.M. Agriculture for Space: People and Places Paving the Way. In Open Agriculture; De Gruyter: Berlin, Germany; New York, NY, USA, 2017; Volume 2, p. 14. [Google Scholar]
- Walker, J.; Granjou, C. MELiSSA the minimal biosphere: Human life, waste and refuge in deep space. Futures 2017, 92, 59–69. [Google Scholar] [CrossRef]
- Chunxiao, X.; Hong, L. Crop candidates for the bioregenerative life support systems in China. Acta Astronaut. 2008, 63, 1076–1080. [Google Scholar] [CrossRef]
- Narici, L.; Reitz, G.; Lobascio, C. Integrated simulations of Mars flights on the ISS. Adv. Space Res. 2018, 62, 990–996. [Google Scholar] [CrossRef]
- Detrell, G.; Ponsati, E.; Messerschmid, E. ECLSS Reliability Analysis Tool for Long Duration Spaceflight; ICES: Toronto, ON, Canada, 2016; Volume 294, pp. 1–11. [Google Scholar]
- Knox, J.; Campbell, M.; Murdoch, K.; Miller, L.; Jeng, F. Integrated Test and Evaluation of a 4-Bed Molecular Sieve (4BMS) Carbon Dioxide Removal System (CDRA), Mechanical Compressor Engineering Development Unit (EDU), and Sabatier Engineering Development Unit (EDU). SAE Tech. Pap. 2005. [Google Scholar] [CrossRef]
- Knox, J.; Gostowski, R.; Watson, D.; Hogan, J.; King, E.; Thomas, J. Development of Carbon Dioxide Removal Systems for Advanced Exploration Systems. In Proceedings of the 42nd International Conference on Environmental Systems, San Diego, CA, USA, 15–19 July 2012; Volume 3642, pp. 1–13. [Google Scholar] [CrossRef]
- Erden, H. Two-Stage Psa System for CO2 Removal and Concentration During Closed-Loop Human Space Exploration Missions. 2016. Available online: https://scholarcommons.sc.edu/etd/3986 (accessed on 10 May 2020).
- Raftery, M.; Hoffman, J. International Space Station as a base camp for exploration beyond low Earth orbit. Acta Astronaut. 2013, 85, 25–32. [Google Scholar] [CrossRef]
- Lambright, W.H. Administrative Leadership and Long-Term Technology: NASA and the International Space Station. Space Policy 2019, 47, 85–93. [Google Scholar] [CrossRef]
- Burns, J.; Fong, T.; Kring, D.; Pratt, W.; Cichan, T. Science and Exploration at the Moon and Mars Enabled by Surface Telerobotics. In Proceedings of the International Academy of Astronautics 10th IAA Symposium on the Future of Space Exploration: Towards the Moon Village and Beyond Torino, Torino, Italy, 27–29 June 2017; pp. 1–10. [Google Scholar]
- Guardabasso, P.; Artuso, G.; Bigi, G.; Carré, A.; Walewski, A.; Crema, M.; Dragoni, M.; Gaudin, D.; Gollins, N.; Governale, G.; et al. Lunar Outpost Sustaining Human Space Exploration by Utilizing In-Situ Resources with a Focus on Propellant Production. In Proceedings of the 69th International Astronautical Congress (IAC), Bremen, Germany, 1–5 October 2018; pp. 1–3. [Google Scholar]
- Cucinotta, F.A. Space Radiation Risks for Astronauts on Multiple International Space Station Missions. PLoS ONE 2014, 9, e96099. [Google Scholar] [CrossRef] [PubMed]
- Shibata, K.; Isobe, H.; Hillier, A.; Choudhuri, A.R.; Maehara, H.; Ishii, T.T.; Shibayama, T.; Notsu, S.; Notsu, Y.; Nagao, T.; et al. Can Superflares Occur on Our Sun? Publ. Astron. Soc. Jpn. 2013, 65. [Google Scholar] [CrossRef]
- Monje, O.; Stutte, G.W.; Goins, G.D.; Porterfield, D.M.; Bingham, G.E. Farming in space: Environmental and biophysical concerns. Adv. Space Res. 2003, 31, 151–167. [Google Scholar] [CrossRef]
- Kuang, A.; Xiao, Y.; McClure, G.; Musgrave, M.E. Influence of Microgravity on Ultrastructure and Storage Reserves in Seeds of Brassica rapa L. Ann. Bot. 2000, 85, 851–859. [Google Scholar] [CrossRef] [PubMed]
- Rouphael, Y.; Petropoulos, S.A.; El-Nakhel, C.; Pannico, A.; Kyriacou, M.C.; Giordano, M.; Troise, A.D.; Vitaglione, P.; De Pascale, S. Reducing Energy Requirements in Future Bioregenerative Life Support Systems (BLSSs): Performance and Bioactive Composition of Diverse Lettuce Genotypes Grown Under Optimal and Suboptimal Light Conditions. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Berkovich, Y.A.; Chetirkin, P.V.; Wheeler, R.M.; Sager, J.C. Evaluating and optimizing horticultural regimes in space plant growth facilities. Adv. Space Res. 2004, 34, 1612–1618. [Google Scholar] [CrossRef] [PubMed]
- ESA. 7 IntERNATional Space Station—ISS. 2014. Available online: http://wsn.spaceflight.esa.int/docs/EUG2LGPr3/EUG2LGPr3-7-ISS.pdf (accessed on 24 January 2020).
- NASA. Solar Power Technologies for Future Planetary Science Missions. JPL D-101316; 2017. Available online: https://solarsystem.nasa.gov/resources/548/solar-power-technologies-for-future-planetary-science-missions/ (accessed on 10 May 2020).
- Darbali-Zamora, R.; Ortiz-Rivera, E. A State Space Average Model for Dynamic Microgrid Based Space Station Simulations. In Proceedings of the IEEE 44th Photovoltaic Specialist Conference (PVSC), Washington, DC, USA, 25–30 June 2017; pp. 2957–2962. [Google Scholar]
- Hart, M.J.; Kinsey, R.J.; Lee, A.S.; Yoshida, J.S. International Space Station Life Extension. In Proceedings of the IEEE Conference in Aerospace, Big Sky, MT, USA, 6–13 March 2010; pp. 1–15. [Google Scholar]
- NASA. About the Space Station Solar Arrays. 2017. Available online: https://www.nasa.gov/mission_pages/station/structure/elements/solar_arrays-about.html (accessed on 10 May 2020).
- ASTM. American Society for Testing and Materials Standard Extraterrestrial Spectrum Reference; Standard Solar Constant and Air Mass Zero Solar Spectral Irradiance Tables, Standard No. E 490-00a; ASTM: West Conshohocken, PA, USA, 2006. [Google Scholar]
- Please, C.P. An Analysis of Semiconductor P-N Junctions. IMA J. Appl. Math. 1982, 28, 301–318. [Google Scholar] [CrossRef]
- Powalla, M.; Paetel, S.; Hariskos, D.; Wuerz, R.; Kessler, F.; Lechner, P.; Wischmann, W.; Friedlmeier, T.M. Advances in Cost-Efficient Thin-Film Photovoltaics Based on Cu(In,Ga)Se2. Engineering 2017, 3, 445–451. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Takamoto, T.; Araki, K. Super high-efficiency multi-junction and concentrator solar cells. Solar Energy Mater. Solar Cells 2006, 90, 3068–3077. [Google Scholar] [CrossRef]
- Ali, K.; Khalid, A.; Ahmad, M.R.; Khan, H.M.; Ali, I.; Sharma, S.K. Multi-junction (III–V) Solar Cells: From Basics to Advanced Materials Choices. In Solar Cells: From Materials to Device Technology; Sharma, S.K., Ali, K., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 325–350. [Google Scholar]
- Cotal, H.; Fetzer, C.; Boisvert, J.; Kinsey, G.; King, R.; Hebert, P.; Yoon, H.; Karam, N. III–V multijunction solar cells for concentrating photovoltaics. Energy Environ. Sci. 2009, 2, 174–192. [Google Scholar] [CrossRef]
- King, R.R.; Law, D.C.; Edmondson, K.M.; Fetzer, C.M.; Kinsey, G.S.; Yoon, H.; Sherif, R.A.; Karam, N.H. 40% efficient metamorphic GaInP/GaInAs/Ge multijunction solar cells. Appl. Phys. Lett. 2007, 90, 183516. [Google Scholar] [CrossRef]
- Massa, G.; Newsham, G.; Hummerick, M.; Caro, J.L.; Stutte, G.; Morrow, R.; Wheeler, R. Preliminary species and media selection for the Veggie space hardware. Gravit. Space Res. 2013, 1, 95–106. [Google Scholar]
- Morrow, R.C.; Remiker, R.W. A deployable salad crop production system for lunar habitats. SAE Tech. Pap. 2009. No. 2009-01-2382. [Google Scholar]
- TechPort. Vegetable Production System (Veggie). 2018. Available online: https://techport.nasa.gov/view/10498 (accessed on 10 May 2020).
- Massa, G.; Kim, H.H.; Wheeler, R.; Mitchell, C. Plant Productivity in Response to LED Lighting. HortScience 2008, 43, 1951–1956. [Google Scholar] [CrossRef]
- Massa, G.; Wheeler, R.; Morrow, R.; Levine, H. Growth chambers on the International Space Station for large plants. Acta Hortic. 2016, 1134, 215–222. [Google Scholar] [CrossRef]
- Monje, O.; Stutte, G.; Wheeler, R. A Researcher’s Guide to: Plant Science; Jones, J., Ed.; U.S National Aeronautics and Space Administration (NASA): Washington, DC, USA, 2015. Available online: https://www.nasa.gov/connect/ebooks/researchers_guide_plant_science_detail.html (accessed on 10 May 2020).
- Avella Ruiz, J.M.; Souza, T.; Silveira, J. A Comparative Analysis between Fluorescent and LED Ilumination for Improve Energy Efficiency at IPBEN Building. In XI—Latin-American Congress on Electricity Generation and Transmission, 1st ed.; Chapter: TOPIC 24-Efficient Use of Energy/Power Quality; Silveira, J.L., Ed.; FDCT—Fundação para o Desenvolvimento Científico e Tecnológico: Guaratingueta, Brazil, 2015; pp. 1–4. [Google Scholar]
- Ganandran, G.S.B.; Mahlia, T.M.I.; Ong, H.C.; Rismanchi, B.; Chong, W.T. Cost-benefit analysis and emission reduction of energy efficient lighting at the Universiti Tenaga Nasional. Sci. World J. 2014, 2014, 745894. [Google Scholar] [CrossRef]
- Annunziata, M.G.; Apelt, F.; Carillo, P.; Krause, U.; Feil, R.; Mengin, V.; Lauxmann, M.A.; Köhl, K.; Nikoloski, Z.; Stitt, M.; et al. Getting back to nature: A reality check for experiments in controlled environments. J. Exp. Bot. 2017, 68, 4463–4477. [Google Scholar] [CrossRef]
- Garcia-Caparros, P.; Chica, R.M.; Almansa, E.M.; Rull, A.; Rivas, L.A.; García-Buendía, A.; Barbero, F.J.; Lao, M.T. Comparisons of Different Lighting Systems for Horticultural Seedling Production Aimed at Energy Saving. Sustainability 2018, 10, 3351. [Google Scholar] [CrossRef]
- Dutta Gupta, S.; Agarwal, A. Artificial Lighting System for Plant Growth and Development: Chronological Advancement, Working Principles, and Comparative Assessment; Dutta Gupta, S., Ed.; Light Emitting Diodes for Agriculture; Springer: Singapore, 2017; pp. 1–25. [Google Scholar]
- Bingham, G.; Topham, T.; Taylor, A.; Podolskiy, I.; Levinskikh, M.; Sychev, V. Lada: ISS Plant Growth Technology Checkout. SAE Tech. Pap. 2003. [Google Scholar] [CrossRef]
- Sychev, V.N.; Levinskikh, M.A.; Gostimsky, S.A.; Bingham, G.E.; Podolsky, I.G. Spaceflight effects on consecutive generations of peas grown onboard the Russian segment of the International Space Station. Acta Astronaut. 2007, 60, 426–432. [Google Scholar] [CrossRef]
- Bingham, G.; Topham, T.; Mulholland, J.; Podolskiy, I. Lada: The ISS Plant Substrate Microgravity Testbed. SAE Tech. Pap. 2002. [Google Scholar] [CrossRef]
- Zabel, P.; Bamsey, M.; Schubert, D.; Tajmar, M. Review and Analysis of Plant Growth Chambers and Greenhouse Modules for Space; ICES: Toronto, ON, Canada, 2014; Volume 120, pp. 1–17. [Google Scholar]
- Kim, H.-H.; Wheeler, R.; Sager, J.; Yorio, N.; Goins, G. Light-emitting diodes as an illumination source for plants: A review of research at Kennedy Space Center. Habitation 2005, 10, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Viršilė, A. The effects of light-emitting diode lighting on greenhouse plant growth and quality. Agric. Food Sci. 2013, 22, 223–234. [Google Scholar]
- Xin, Z.; He, D.; Niu, G.; Zhengnan, Y.; Song, J. Effects of environment lighting on the growth, photosynthesis, and quality of hydroponic lettuce in a plant factory. Int. J. Agric. Biol. Eng. 2018, 11. [Google Scholar] [CrossRef]
- Avercheva, O.; Berkovich, Y.A.; Smolyanina, S.; Bassarskaya, E.; Pogosyan, S.; Ptushenko, V.; Erokhin, A.; Zhigalova, T. Biochemical, photosynthetic and productive parameters of Chinese cabbage grown under blue–red LED assembly designed for space agriculture. Adv. Space Res. 2014, 53, 1574–1581. [Google Scholar] [CrossRef]
- Cohu, C.M.; Lombardi, E.; Adams, W.W.; Demmig-Adams, B. Increased nutritional quality of plants for long-duration spaceflight missions through choice of plant variety and manipulation of growth conditions. Acta Astronaut. 2014, 94, 799–806. [Google Scholar] [CrossRef]
- Kim, M.J.; Moon, Y.; Tou, J.; Mou, B.; Waterland, N. Nutritional Value, Bioactive Compounds and Health Benefits of Lettuce (Lactuca sativa L.). J. Food Compos. Anal. 2016, 49, 19–34. [Google Scholar] [CrossRef]
- Sean, K. Luminous Intensity of an LED as a Function of Input Power. Int. Sch. Bangk. J. Phys. 2008, 2, 1–4. [Google Scholar]
- Jeong, P.; Lee, C.C. An electrical model with junction temperature for light-emitting diodes and the impact on conversion efficiency. IEEE Electron Device Lett. 2005, 26, 308–310. [Google Scholar] [CrossRef]
- David, A.; Whitehead, L.A. LED-based white light. Comptes Rendus Phys. 2018, 19, 169–181. [Google Scholar] [CrossRef]
- Colla, G.; Battistelli, A.; Proietti, S.; Moscatello, S.; Rouphael, Y.; Cardarelli, M.; Casucci, M. Rocket seedling production on the International Space Station: Growth and nutritional properties. Microgravity Sci. Technol. 2007, 19, 118–121. [Google Scholar] [CrossRef]
- Boeing Company. Investigations of selected higher plants as gas exchange mechanism for closed ecological systems. In Biologistics for Space Systems Symposium, May 1962; AMRL-TDR-62-116; Wright-Patterson Air Force Base: Ohio, OH, USA, 1962. [Google Scholar]
- Silverstone, S.; Nelson, M. Food production and nutrition in Biosphere 2: Results from the first mission September 1991 to September 1993. Adv. Space Res. 1996, 18, 49–61. [Google Scholar] [CrossRef]
- Wheeler, R.M. Horticulture for Mars. Acta Hortic. 2004, 642, 201–215. [Google Scholar] [CrossRef]
- Zabel, P.; Bamsey, M.; Zeidler, C.; Vrakking, V.; Johannes, B.-W.; Rettberg, P.; Schubert, D.; Romberg, O.; Imhof, B.; Davenport, R. Introducing EDEN ISS—A European project on advancing plant cultivation technologies and operations. ICES 2015, 2015–2058. [Google Scholar]
- Bugbee, B.; Koerner, G. Yield comparisons and unique characteristics of the dwarf wheat cultivar ‘USU-Apogee’. Adv. Space Res. 1997, 20, 1891–1894. [Google Scholar] [CrossRef]
- Salisbury, F.B.; Campbell, W.F.; Carman, J.G.; Bingham, G.E.; Bubenheim, D.L.; Yendler, B.; Sytchev, V.; Levinskikh, M.A.; Ivanova, I.; Chernova, L. Plant growth during the greenhouse II experiment on the Mir orbital station. Adv. Space Res. 2003, 31, 221–227. [Google Scholar] [CrossRef][Green Version]
- Salisbury, F.; Bingham, G.; Campbell, W.; Carman, J.; Bubenheim, D.; Yendler, B.; Jahns, G. Growing super-dwarf wheat in Svet on Mir. Life Support Biosph. Sci. Int. J. Earth Space 1995, 2, 31–39. [Google Scholar]
- Wamelink, G.W.W.; Frissel, J.Y.; Krijnen, W.H.J.; Verwoert, M.R.; Goedhart, P.W. Can Plants Grow on Mars and the Moon: A Growth Experiment on Mars and Moon Soil Simulants. PLoS ONE 2014, 9, e103138. [Google Scholar] [CrossRef]
- Salisbury, F.; Bugbee, B. Wheat Farming in a Lunar Base; Mendell, W.W., Ed.; Lunar Bases and Space Activities of the 21st Century; Lunar and Planetary Institute: Houston, TX, USA, 1985; pp. 635–645. [Google Scholar]
- De Micco, V.; Aronne, G.; Colla, G.; Fortezza, R.; De Pascale, S. Agro-biology for bioregenerative Life Support Systems in long-term Space missions: General constraints and the Italian efforts. J. Plant Interact. 2009, 4, 241–252. [Google Scholar] [CrossRef]
- Rivera, C.M.; Battistelli, A.; Moscatello, S.; Proietti, S.; Rouphael, Y.; Cardarelli, M.; Colla, G. Influence of simulated microgravity on growth, yield, and quality of leafy vegetables: Lettuce and rocket. Eur. J. Hortic. Sci. 2006, 71, 45–51. [Google Scholar]
- Colla, G.; Rouphael, Y.; Cardarelli, M.; Mazzucato, A.; Olimpieri, I. Growth, yield and reproduction of dwarf tomato grown under simulated microgravity conditions. Plant Biosyst. Int. J. Deal. All Asp. Plant Biol. 2007, 141, 75–81. [Google Scholar] [CrossRef]
- Poulet, L.; Massa, G.D.; Morrow, R.C.; Bourget, C.M.; Wheeler, R.M.; Mitchell, C.A. Significant reduction in energy for plant-growth lighting in space using targeted LED lighting and spectral manipulation. Life Sci. Space Res. 2014, 2, 43–53. [Google Scholar] [CrossRef]
- El-Nakhel, C.; Giordano, M.; Pannico, A.; Carillo, P.; Fusco, G.M.; De Pascale, S.; Rouphael, Y. Cultivar-Specific Performance and Qualitative Descriptors for Butterhead Salanova Lettuce Produced in Closed Soilless Cultivation as a Candidate Salad Crop for Human Life Support in Space. Life 2019, 9, 61. [Google Scholar] [CrossRef] [PubMed]
- Khandaker, L.; Oba, S. Comparative study on functional components, antioxidant activity and color parameters of selected colored leafy vegetables as affected by photoperiods. J. Food Agric. Environ. 2009, 7, 392–398. [Google Scholar]
- Rouphael, Y.; Kyriacou, M.C.; Carillo, P.; Pizzolongo, F.; Romano, R.; Sifola, M.I. Chemical Eustress Elicits Tailored Responses and Enhances the Functional Quality of Novel Food Perilla frutescens. Molecules 2019, 24, 185. [Google Scholar] [CrossRef]
- Xiao, Z.; Lester, G.E.; Luo, Y.; Wang, Q. Assessment of Vitamin and Carotenoid Concentrations of Emerging Food Products: Edible Microgreens. J. Agric. Food Chem. 2012, 60, 7644–7651. [Google Scholar] [CrossRef]
- Pinto, E.; Almeida, A.A.; Aguiar, A.A.; Ferreira, I.M.P.L.V.O. Comparison between the mineral profile and nitrate content of microgreens and mature lettuces. J. Food Compos. Anal. 2015, 37, 38–43. [Google Scholar] [CrossRef]
- Thornton, W.; Bonato, F. Basic Mechanisms. In The Human Body and Weightlessness: Operational Effects, Problems and Countermeasures; Springer International Publishing: Cham, Switzerland, 2017; pp. 21–30. [Google Scholar]
- Osborn, M. A Strategy for Research in Space Biology and Medicine in The New Century; National Academy Press: Washington, DC, USA, 1998. [Google Scholar]
- Poulet, L.; Fontaine, J.P.; Dussap, C.G. Plant’s response to space environment: A comprehensive review including mechanistic modelling for future space gardeners. Bot. Lett. 2016, 163, 337–347. [Google Scholar] [CrossRef]
- Monje, O.; Nugent, M.; Hummerick, M.; Dreschel, T.; Spencer, L.; Romeyn, M.; Massa, G.; Wheeler, R.; Fritsche, R. New Frontiers in Food Production Beyond LEO; ICES: Toronto, ON, Canada, 2019; Volume 260, pp. 1–6. [Google Scholar]
- Stutte, G.; Wheeler, R.; Morrow, R.; Newsham, G. Operational Evaluation of VEGGIE Food Production System in the Habitat Demonstration Unit. In Proceedings of the 41st International Conference on Environmental Systems, Portland, Oregon, 17–21 July 2011. [Google Scholar] [CrossRef]
- Casado, J. Cultivating the Future—Growing food in space, May 2006. Spaceflight 2006, 180–189. [Google Scholar]
- Morrow, R.; Bula, R.J.; Tibbitts, T.; Dinauer, W. A Matrix-Based Porous Tube Water and Nutrient Delivery System. SAE Tech. Pap. 1992, 1–7. [Google Scholar] [CrossRef]
- Morrow, R.; Bula, R.; Tibbitts, T.; Dinauer, W. The ASTROCULTURE(TM) flight experiment series, validating technologies for growing plants in space. Advances Space Res. 1994, 14, 29–37. [Google Scholar] [CrossRef]
- Ivanova, T.N.; Kostov, P.T.; Sapunova, S.M.; Dandolov, I.W.; Salisbury, F.B.; Bingham, G.E.; Sytchov, V.N.; Levinskikh, M.A.; Podolski, I.G.; Bubenheim, D.B.; et al. Six-month space greenhouse experiments—A step to creation of future biological life support systems. Acta Astronaut. 1998, 42, 11–23. [Google Scholar] [CrossRef]
- Morrow, R.C.; Richter, R.C.J.; Tellez, G. A New Plant Habitat Facility for the ISS. ICES 2016, 2016–2320. [Google Scholar]
- Heinse, R.; Jones, S.; Tuller, M.; Bingham, G.; Podolskiy, I.; Or, D. Providing Optimal Root-Zone Fluid Fluxes: Effects of Hysteresis on Capillary-Dominated Water Distributions in Reduced Gravity. SAE Tech. Pap. 2009, 1–9. [Google Scholar] [CrossRef]
- Heinse, R.; Jones, S.; Steinberg, S.; Tuller, M.; Or, D. Measurements and Modeling of Variable Gravity Effects on Water Distribution and Flow in Unsaturated Porous Media. Vadose Zone J. 2007, 6, 713–724. [Google Scholar] [CrossRef]
- Massa, G.D.; Newsham, G.; Hummerick, M.E.; Morrow, R.C.; Wheeler, R.M. Plant Pillow Preparation for the Veggie Plant Growth System on the International Space Station. Gravit. Space Res. Vol. 2017, 5, 24–34. [Google Scholar]
- Monje, O. Validation of the Advanced Plant Habitat Facility on ISS. 2018. Available online: https://ntrs.nasa.gov/archive/nasa/casi.ntrs.nasa.gov/20180006722.pdf (accessed on 10 May 2020).
- Moffatt, S.A.; Morrow, R.C.; Wetzel, J.P. Astro GardenTM aeroponic plant growth system design evolution. ICES 2019, 195, 1–13. [Google Scholar]
- Morrow, R.C.; Wetzel, J.P.; Richter, C.R.; Crabb, M.T. Evolution of space-based plant growth technologies for Hybrid Life Support Systems. ICES Charleston, SC. ICES 2017, 301, 1–9. [Google Scholar]
- Dreschel, T.; Sager, J. Control of water and nutrients using a porous tube: A method for growing plants in space. Hort Sci. Publ. Am. Soc. Hortic. Sci. 1989, 24, 944–947. [Google Scholar]
- Massa, G.; Dufour, N.; Carver, J.; Hummerick, M.; Wheeler, R.; Morrow, R.; Smith, T.M. VEG-01: Veggie Hardware Validation Testing on the International Space Station. Open Agric. 2017, 2. [Google Scholar] [CrossRef]
- Burgner, S.E.; Mitchell, C.; Massa, G.; Romeyn, M.; Wheeler, M. Troubleshooting performance failures of Chinese Cabbage for Veggie on the ISS. ICES 2019, 2019–2328. [Google Scholar]
- Dreschel, T.W.; Brown, C.S.; Piastuch, W.C.; Hinkle, C.R.; Knott, W.M. Porous tube plant nutrient delivery system development: A device for nutrient delivery in microgravity. Adv. Space Res. 1994, 14, 47–51. [Google Scholar] [CrossRef]
- Hoehn, A.; Scovazzo, P.; Stodieck, L.; Clawson, J.; Kalinowski, W.; Rakow, A.; Simmons, D.; Heyenga, A.; Kliss, M. Microgravity Root Zone Hydration Systems. SAE Tech. Pap. 2000. [Google Scholar] [CrossRef]
- Dreschel, T.; Hall, C.; Foster, T. Demonstration of a Porous Tube Hydroponic System to Control Plant Moisture and Growth; NASA: Washington, DC, USA, 2003. Available online: https://ntrs.nasa.gov/archive/nasa/casi.ntrs.nasa.gov/20050215299.pdf (accessed on 10 May 2020).
- Tibbitts, T.; Cao, W.; Frank, T. Development of a siphon system with porous tubes for maintaining a constant negative water pressure in a rooting matrix. Biotronics 1995, 24, 7–14. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carillo, P.; Morrone, B.; Fusco, G.M.; De Pascale, S.; Rouphael, Y. Challenges for a Sustainable Food Production System on Board of the International Space Station: A Technical Review. Agronomy 2020, 10, 687. https://doi.org/10.3390/agronomy10050687
Carillo P, Morrone B, Fusco GM, De Pascale S, Rouphael Y. Challenges for a Sustainable Food Production System on Board of the International Space Station: A Technical Review. Agronomy. 2020; 10(5):687. https://doi.org/10.3390/agronomy10050687
Chicago/Turabian StyleCarillo, Petronia, Biagio Morrone, Giovanna Marta Fusco, Stefania De Pascale, and Youssef Rouphael. 2020. "Challenges for a Sustainable Food Production System on Board of the International Space Station: A Technical Review" Agronomy 10, no. 5: 687. https://doi.org/10.3390/agronomy10050687
APA StyleCarillo, P., Morrone, B., Fusco, G. M., De Pascale, S., & Rouphael, Y. (2020). Challenges for a Sustainable Food Production System on Board of the International Space Station: A Technical Review. Agronomy, 10(5), 687. https://doi.org/10.3390/agronomy10050687








