High Soybean Yield and Drought Adaptation Being Associated with Canopy Architecture, Water Uptake, and Root Traits
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Growth Conditions
2.2. Water Extraction/Use
2.3. Roots Sampling at R2 Stage Under TDS Condition
2.4. Flower and Pod Tagging
2.5. Harvest
2.6. Statistical Analysis
3. Results
3.1. Yield Performance
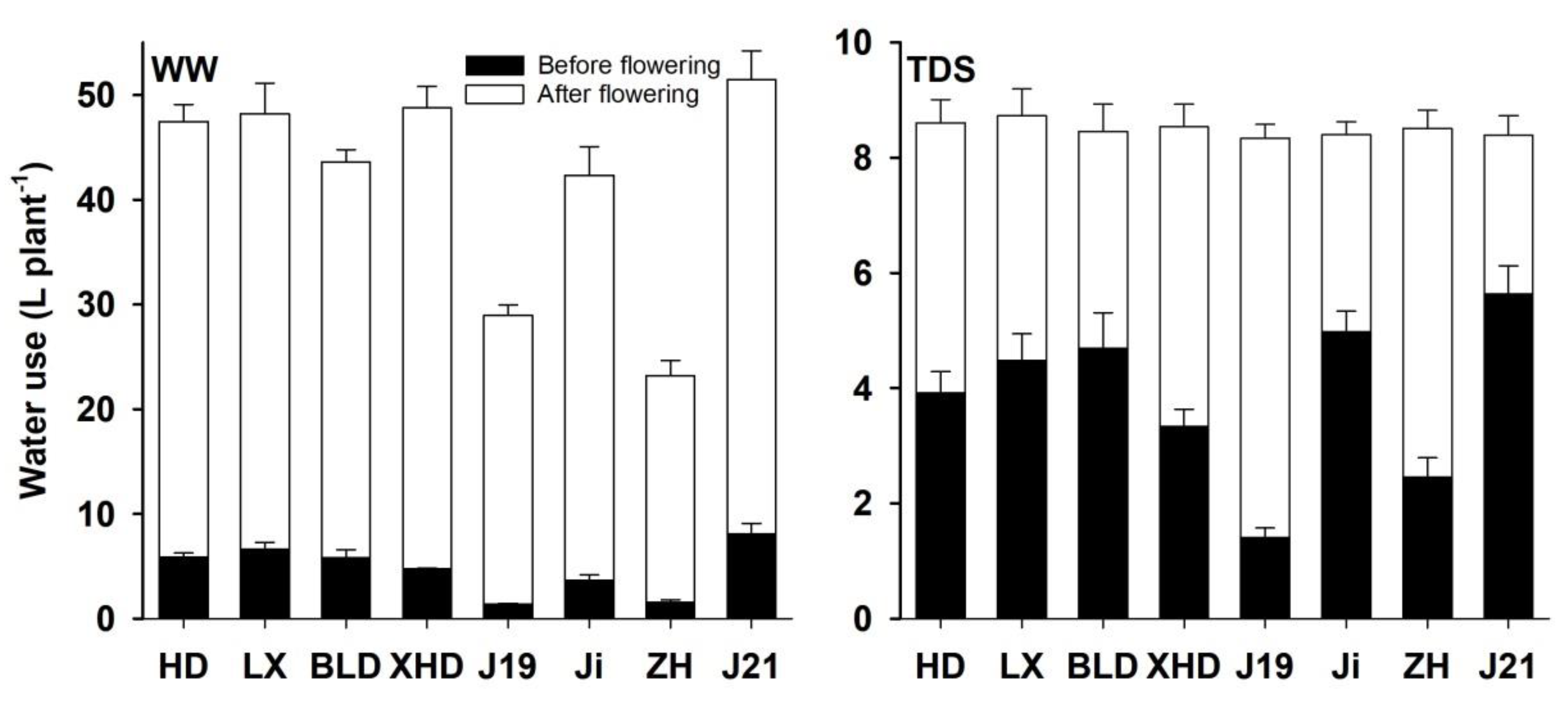
3.2. Water Use Under Two Water Regimes
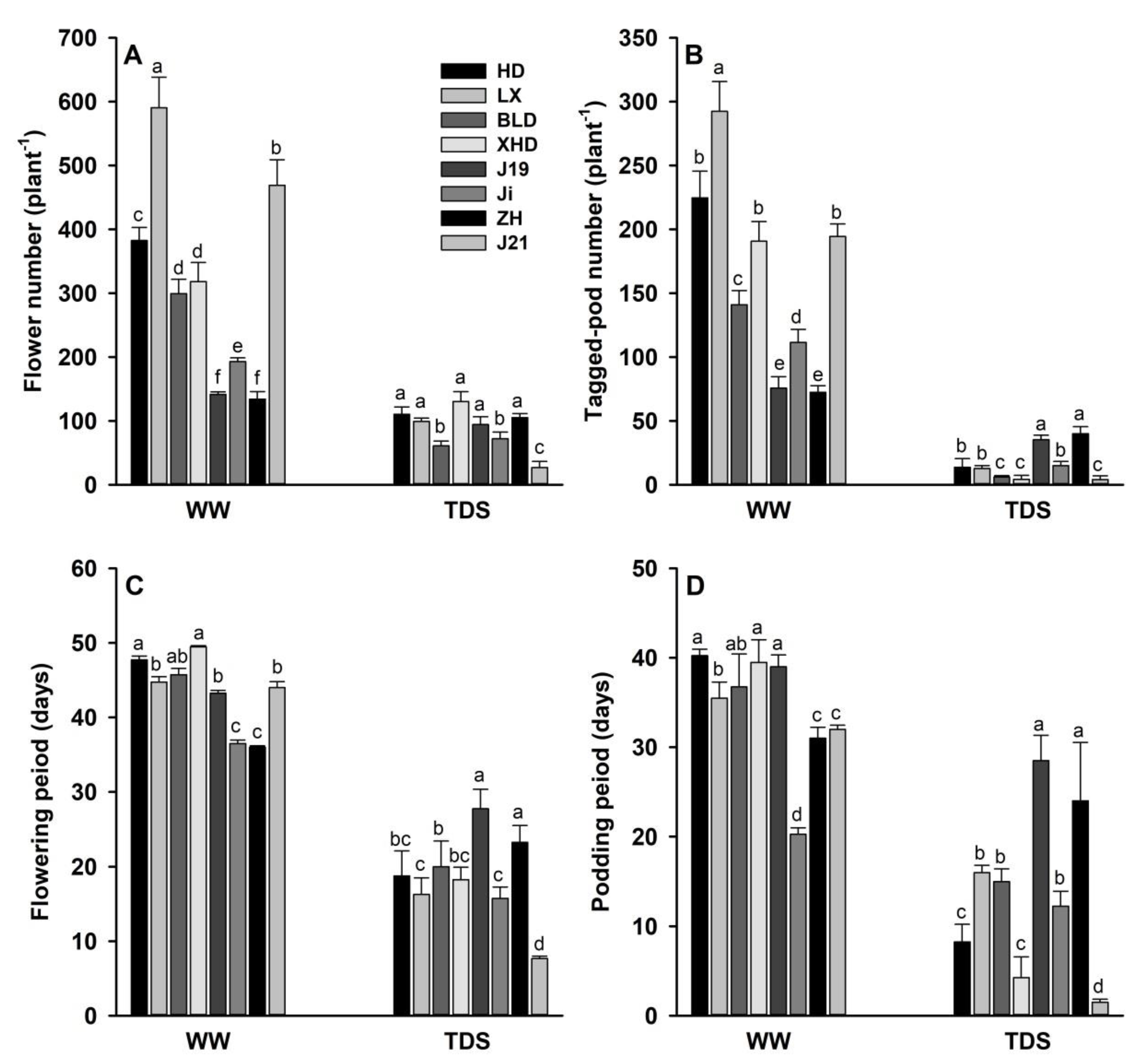
3.3. Flower and Pod Number, and Flowering and Podding Period Under WW and TDS
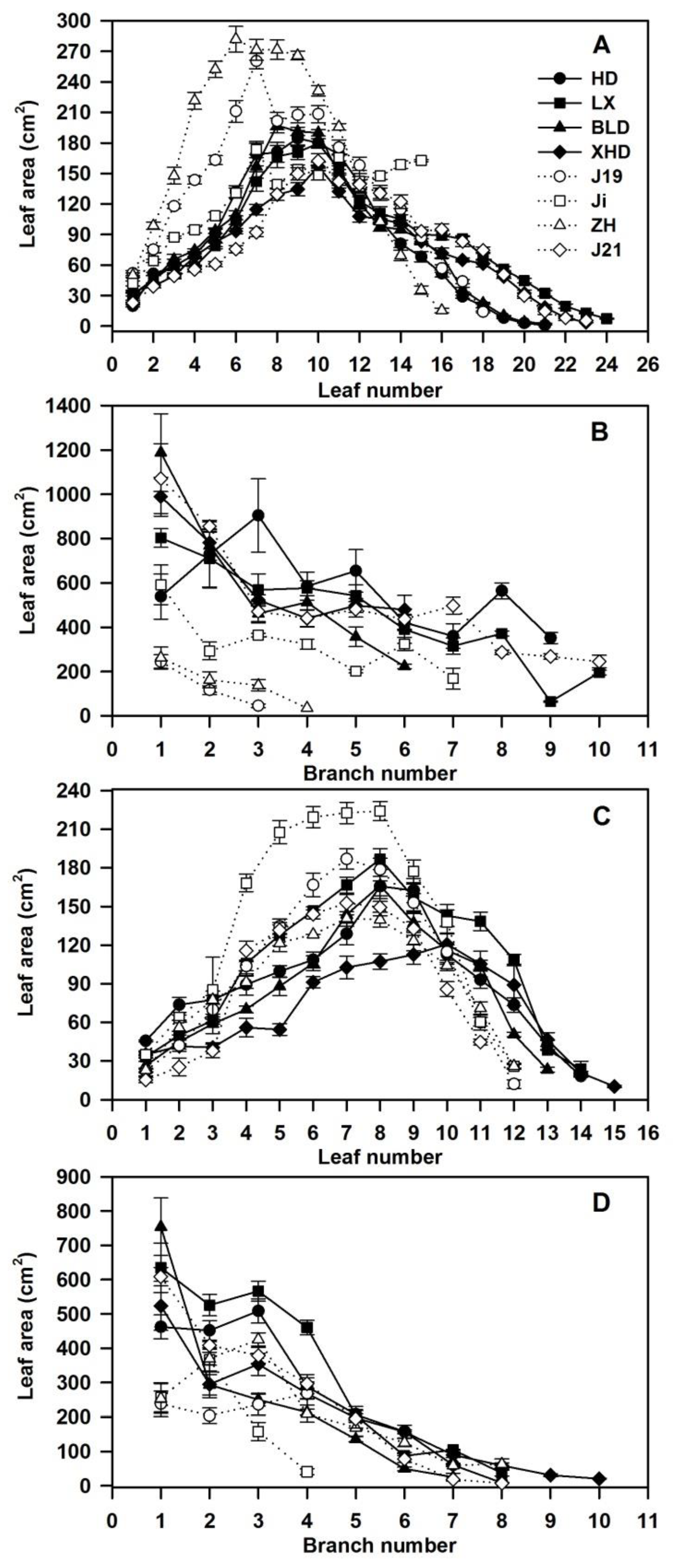
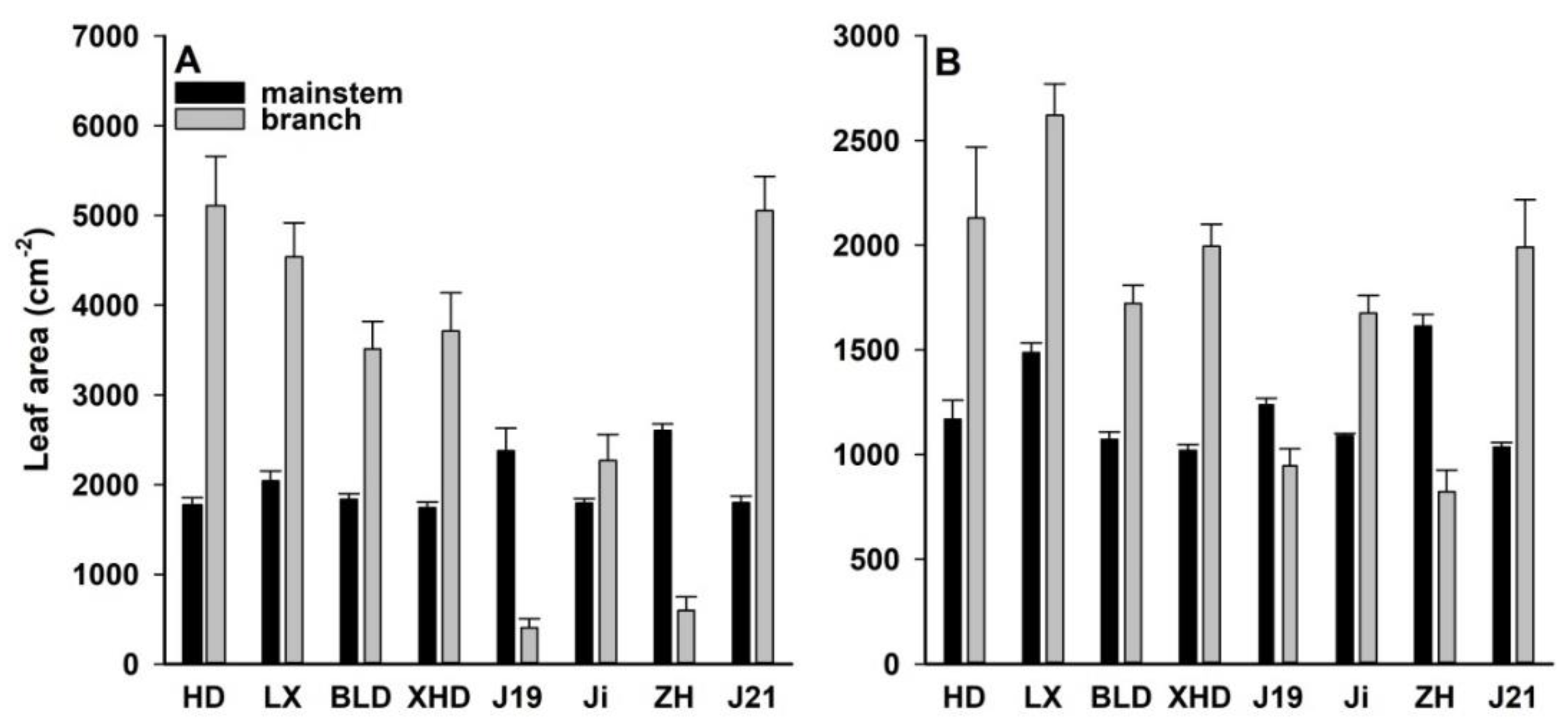
3.4. Canopy Architecture Under Both Water Regimes at Harvest
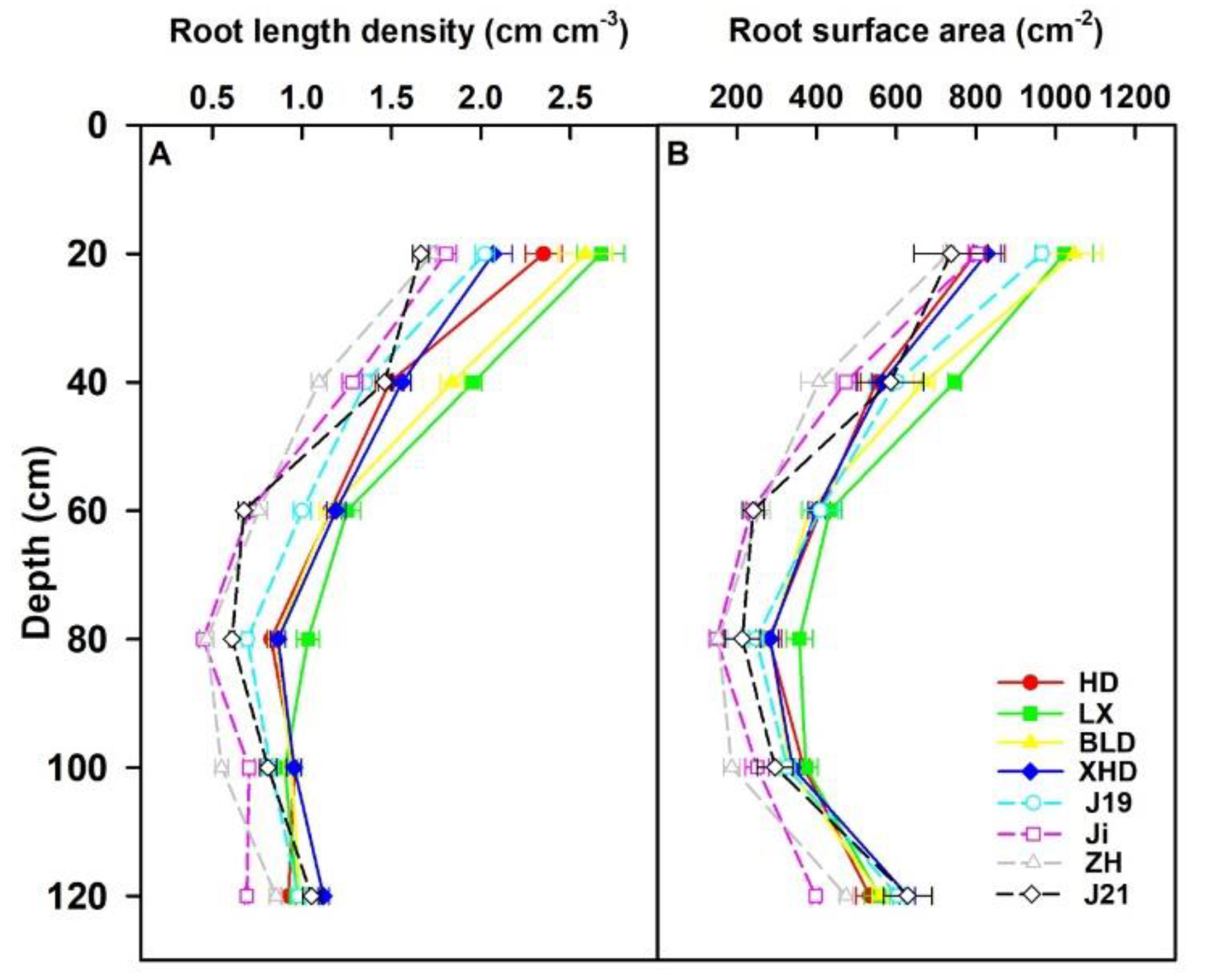
3.5. Root Morphology at R2 stage
4. Discussion
4.1. Root Size, Canopy Architecture, and Water Use
4.2. Water Use, Flower and Pod Development, and Yield and Drought Adaptation
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Manavalan, L.P.; Guttikonda, S.K.; Tran, L.S.P.; Nguyen, H.T. Physiological and molecular approaches to improve drought resistance in Soybean. Plant Cell Physiol. 2009, 50, 1260–1276. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.C.; Wright, G.C.; Siddique, K.H.H. Adaptation of grain legumes (pulses) to water-limited environments. Adv. Agron. 2001, 71, 193–231. [Google Scholar]
- Merah, O. Potential importance of water status traits for durum wheat improvement under Mediterranean conditions. J. Agric. Sci. 2001, 137, 139–145. [Google Scholar] [CrossRef]
- Koester, R.P.; Skoneczka, J.A.; Cary, T.R.; Diers, B.W.; Ainsworth, E.A. Historical gains in soybean (Glycine max Merr.) seed yield are driven by linear increases in light interception, energy conversion, and partitioning efficiencies. J. Exp. Bot. 2014, 65, 3311–3321. [Google Scholar] [CrossRef]
- Leport, L.; Turner, N.C.; Davies, S.L.; Siddique, K.H.M. Variation in pod production and abortion among chickpea cultivars under terminal drought. Eur. J. Agron. 2006, 24, 236–246. [Google Scholar] [CrossRef]
- Fang, X.W.; Turner, N.C.; Yan, G.J.; Li, F.M.; Siddique, K.H.M. Flower numbers, pod production, pollen viability, and pistil function are reduced and flower and pod abortion increased in chickpea (Cicer arietinum L.) under terminal drought. J. Exp. Bot. 2010, 61, 335–345. [Google Scholar] [CrossRef]
- Kholova, J.; Hash, C.T.; Kakkera, A.; Kocova, M.; Vadez, V. Constitutive water-conserving mechanisms are correlated with the terminal drought tolerance of pearl millet [Pennisetum glaucum (L.) R. Br.]. J. Exp. Bot. 2010, 61, 369–377. [Google Scholar] [CrossRef]
- Kholova, J.; Hash, C.T.; Kumar, P.L.; Yadav, R.S.; Kocova, M.; Vadez, V. Terminal drought-tolerant pearl millet [Pennisetum glaucum (L.) R. Br.] have high leaf ABA and limit transpiration at high vapour pressure deficit. J. Exp. Bot. 2010, 61, 1431–1440. [Google Scholar] [CrossRef]
- Zaman-Allah, M.; Jenkinson, D.M.; Vadez, V. A conservative pattern of water use, rather than deep or profuse rooting, is critical for the terminal drought tolerance of chickpea. J. Exp. Bot. 2011, 62, 4239–4252. [Google Scholar] [CrossRef]
- Belko, N.; Zaman-Allah, M.; Cisse, N.; Diop, N.N.; Zombre, G.; Ehlers, J.D.; Vadez, V. Lower soil moisture threshold for transpiration decline under water deficit correlates with lower canopy conductance and higher transpiration efficiency in drought-tolerant cowpea. Funct. Plant Biol. 2012, 39, 306–322. [Google Scholar] [CrossRef]
- Vadez, V.; Kholova, J.; Yadav, R.S.; Hash, C.T. Small temporal differences in water uptake among varieties of pearl millet (Pennisetum glaucum (L.) R. Br.) are critical for grain yield under terminal drought. Plant Soil 2013, 371, 447–462. [Google Scholar] [CrossRef]
- Borrell, A.K.; Mullet, J.E.; George-Jaeggli, B.; van Oosterom, E.J.; Hammer, G.L.; Klein, P.E.; Jordan, D.R. Drought adaptation of stay-green sorghum is associated with canopy development, leaf anatomy, root growth, and water uptake. J. Exp. Bot. 2014, 65, 6251–6263. [Google Scholar] [CrossRef] [PubMed]
- Van Oosterom, E.J.; Borrell, A.K.; Deifel, K.S.; Hammer, G.L. Does increased leaf appearance rate enhance adaptation to postanthesis drought stress in sorghum? Crop Sci. 2011, 51, 2728–2740. [Google Scholar] [CrossRef]
- Liu, Y.; Gai, J.Y.; Lu, H.; Wang, Y.J.; Chen, S.Y. Identification of drought tolerant germplasm and inheritance and QTL mapping of related root traits in soybean [Glycine max (L.) Merr.]. Acta Genet. Sin. 2005, 32, 855–863. [Google Scholar] [PubMed]
- Wang, M.; Zhang, C.; Ma, T. studies in the drought resistance of seedings in soybean. Chin. J. Oil Sci. 2004, 26, 29–32. [Google Scholar]
- Garay, A.; Wilhelm, W. Root system characteristics of two soybean isolines undergoing water stress conditions. Agron. J. 1983, 75, 973–977. [Google Scholar] [CrossRef]
- He, J.; Du, Y.L.; Wang, T.; Turner, N.C.; Yang, R.P.; Jin, Y.; Xi, Y.; Zhang, C.; Cui, T.; Fang, X.W.; et al. Conserved water use improves the yield performance of soybean (Glycine max (L. Merr.)) under drought. Agric. Water Manag. 2017, 179, 236–245. [Google Scholar] [CrossRef]
- Fehr, W.; Caviness, C.; Burmood, D.; Pennington, J. Stage of development descriptions for soybeans, Glycine max (L.). Merrill. Crop Sci. 1971, 11, 929–931. [Google Scholar] [CrossRef]
- Subbarao, G.V.; Johansen, C.; Slinkard, A.E.; Rao, R.C.N.; Saxena, N.P.; Chauhan, Y.S. Strategies for improving drought resistance in grain legumes. Crit. Rev. Plant Sci. 1995, 14, 469–523. [Google Scholar] [CrossRef]
- Kashiwagi, J.; Krishnamurthy, L.; Upadhyaya, H.D.; Krishna, H.; Chandra, S.; Vadez, V.; Serraj, R. Genetic variability of drought-avoidance root traits in the mini-core germplasm collection of chickpea (Cicer arietinum L.). Euphytica 2005, 146, 213–222. [Google Scholar] [CrossRef]
- Sinclair, T.R.; Messina, C.D.; Beatty, A.; Samples, M. Assessment across the United States of the benefits of altered soybean drought traits. Agron. J. 2010, 102, 475. [Google Scholar] [CrossRef]
- Kim, H.K.; Luquet, D.; van Oosterom, E.; Dingkuhn, M.; Hammer, G. Regulation of tillering in sorghum: Genotypic effects. Ann. Bot. 2010, 106, 69–78. [Google Scholar] [CrossRef]
- Borrell, A.K.; Hammer, G.L.; Douglas, A.C. Does maintaining green leaf area in sorghum improve yield under drought? I. Leaf growth and senescence. Crop Sci. 2000, 40, 1026–1037. [Google Scholar] [CrossRef]
- He, J.; Du, Y.L.; Wang, T.; Turner, N.C.; Xi, Y.; Li, F.M. Old and new cultivars of soya bean (Glycine max L.) subjected to soil drying differ in abscisic acid accumulation, water relations characteristics and yield. J. Agron. Crop Sci. 2016, 202, 372–383. [Google Scholar] [CrossRef]
- He, J.; Jin, Y.; Turner, N.C.; Li, F.M. Irrigation during flowering improves subsoil water uptake and grain yield in rainfed soybean. Agronomy 2020, 10, 120. [Google Scholar] [CrossRef]

| Cultivars | Location | Breeding Year | Days To Maturity | Characteristics |
|---|---|---|---|---|
| Huangsedadou (HD) | Gansu | Landrace variety | 147 | small seed, low yield |
| Longxixiaohuangpi (LX) | Gansu | Landrace variety | 149 | small seed, low yield |
| Bailudou (BLD) | Shanxi | Landrace variety | 151 | small seed, low yield |
| Xiaoheidou (XHD) | Shanxi | Landrace variety | 148 | small seed, low yield |
| Jindou 21 (J21) | Shanxi | 1999 | 145 | medium seed, high yield |
| Jindou 19 (J19) | Shanxi | 2003 | 140 | large seed, high yield |
| Jidou 12 (Ji) | Hebei | 2006 | 151 | large seed, high yield |
| Zhonghuang 30 (ZH) | Beijing | 2006 | 140 | large seed, high yield |
| Month | Monthly Mean Temperature (°C) | Monthly Precipitation (mm) |
|---|---|---|
| April | 14.1 | 1.2 |
| May | 13.9 | 32.6 |
| June | 18.5 | 38.2 |
| July | 19.3 | 42.5 |
| August | 19.4 | 42.8 |
| September | 13.4 | 54.6 |
| WT | cv. | PW | GN | GY | HGW | WUEG |
|---|---|---|---|---|---|---|
| WW | HD | 29.6 | 185.8 | 15.3 | 8.2 | 0.31 |
| LX | 27.5 | 217.3 | 16.0 | 7.3 | 0.32 | |
| BLD | 22.9 | 142.3 | 13.5 | 9.6 | 0.30 | |
| XHD | 27.8 | 247.3 | 15.5 | 6.3 | 0.31 | |
| J19 | 28.9 | 111.5 | 19.6 | 17.6 | 0.63 | |
| Ji | 33.0 | 117.0 | 19.3 | 16.6 | 0.44 | |
| ZH | 25.4 | 111.3 | 16.6 | 15.1 | 0.70 | |
| J21 | 32.9 | 189.3 | 19.4 | 10.3 | 0.37 | |
| TDS | HD | 0.0 | 0.0 | 0.0 | 0.0 | 0 |
| LX | 0.0 | 0.0 | 0.0 | 0.0 | 0 | |
| BLD | 0.1 | 0.5 | 0.05 | 0.9 | 0 | |
| XHD | 0.0 | 0.0 | 0.0 | 0.0 | 0 | |
| J19 | 6.8 | 40.0 | 3.6 | 8.8 | 0.43 | |
| Ji | 0.9 | 2.5 | 0.1 | 4.8 | 0.01 | |
| ZH | 4.6 | 31.5 | 2.4 | 7.6 | 0.27 | |
| J21 | 0.0 | 0.0 | 0.0 | 0.0 | 0 | |
| LSD0.05 | 3.6 | 25.0 | 2.1 | 2.4 | 0.11 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, X.-B.; Guo, C.; Li, F.-M.; Li, M.; He, J. High Soybean Yield and Drought Adaptation Being Associated with Canopy Architecture, Water Uptake, and Root Traits. Agronomy 2020, 10, 608. https://doi.org/10.3390/agronomy10040608
Gao X-B, Guo C, Li F-M, Li M, He J. High Soybean Yield and Drought Adaptation Being Associated with Canopy Architecture, Water Uptake, and Root Traits. Agronomy. 2020; 10(4):608. https://doi.org/10.3390/agronomy10040608
Chicago/Turabian StyleGao, Xiu-Bing, Can Guo, Feng-Min Li, Ming Li, and Jin He. 2020. "High Soybean Yield and Drought Adaptation Being Associated with Canopy Architecture, Water Uptake, and Root Traits" Agronomy 10, no. 4: 608. https://doi.org/10.3390/agronomy10040608
APA StyleGao, X.-B., Guo, C., Li, F.-M., Li, M., & He, J. (2020). High Soybean Yield and Drought Adaptation Being Associated with Canopy Architecture, Water Uptake, and Root Traits. Agronomy, 10(4), 608. https://doi.org/10.3390/agronomy10040608






