Less Is More: Lower Sowing Rate of Irrigated Tef (Eragrostis tef) Alters Plant Morphology and Reduces Lodging
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Experimental Design and Management
2.3. Assessment of Agronomic Traits
2.4. Lodging and Morphological Traits
2.5. Remote Sensing of Lodging
2.6. Statistical Analyses
3. Results
3.1. Plant Performance
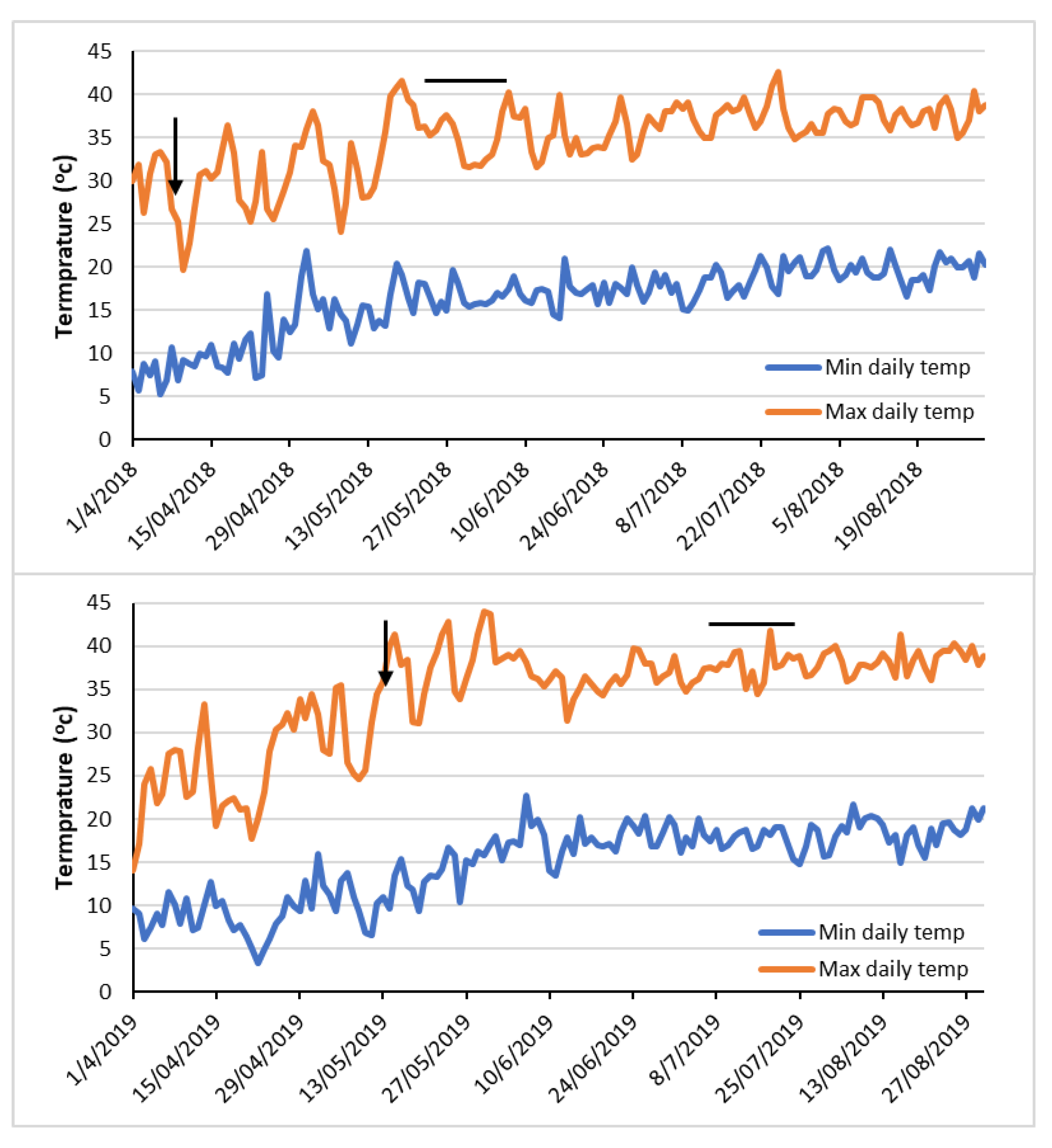
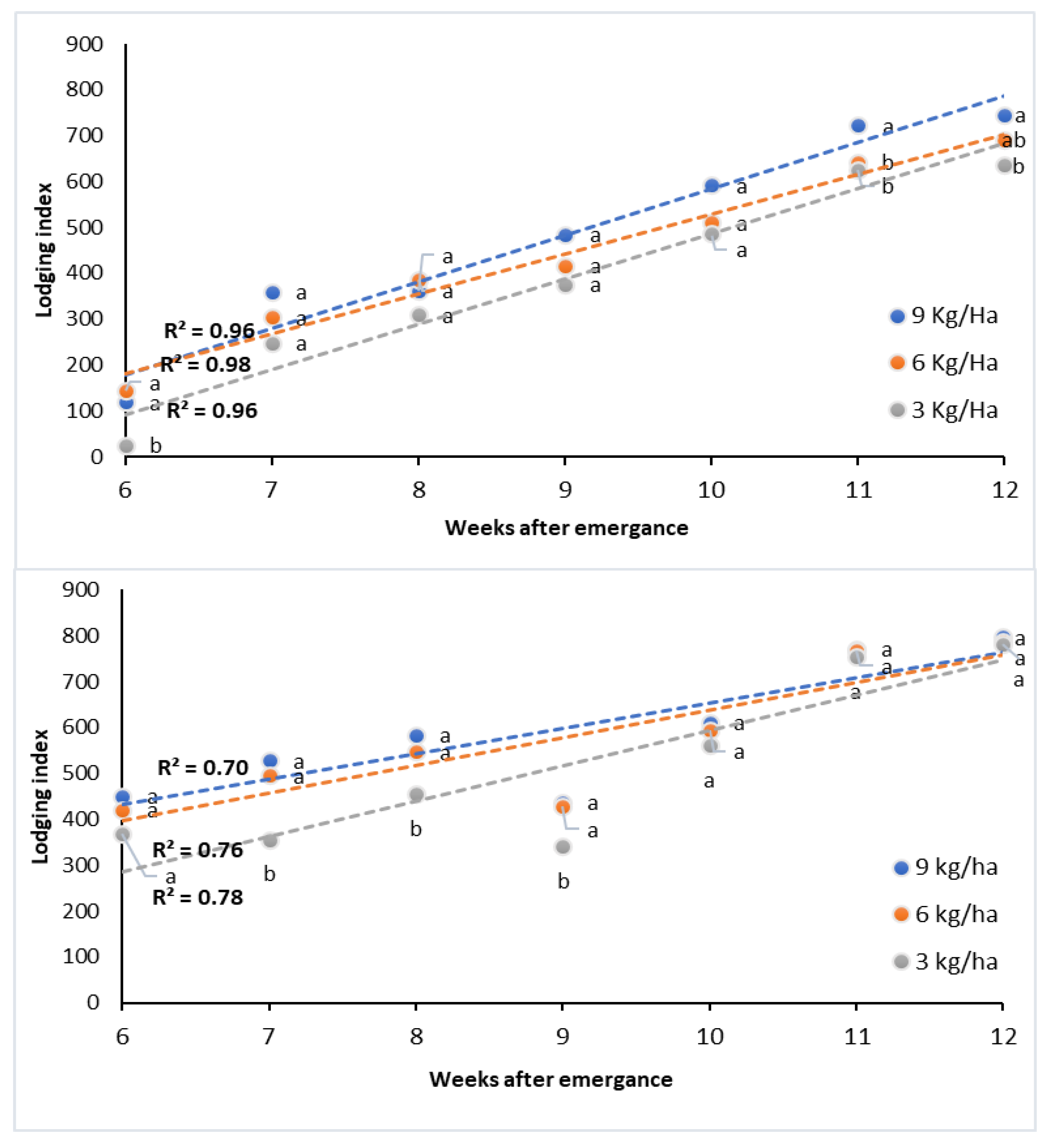
3.2. Lodging Development during the Growing Season
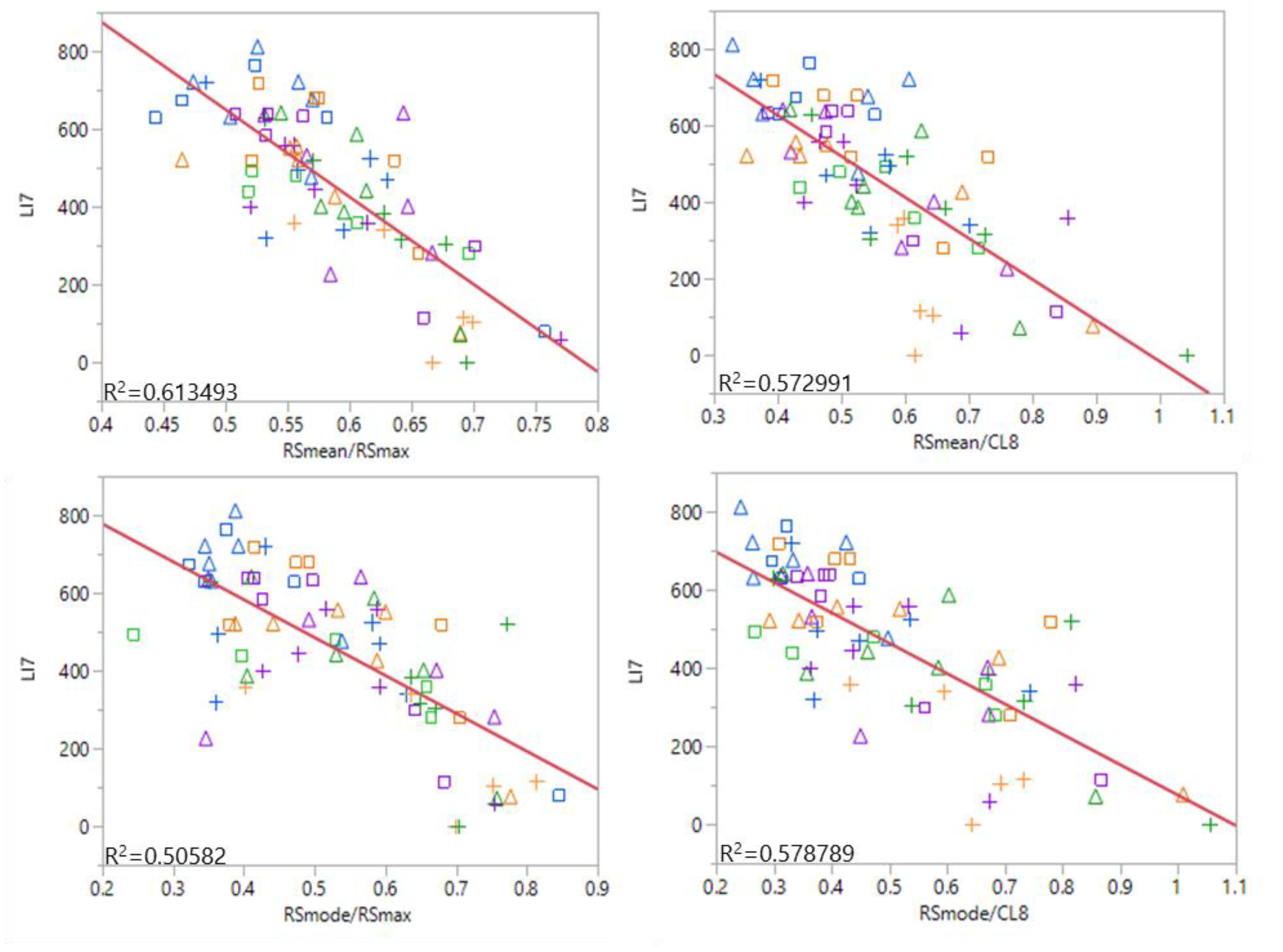
3.3. Remote Sensing of Lodging
3.4. Mid-Season Lodging and Related Traits
4. Discussion
4.1. Effects of Sowing Rate on Tef Productivity
4.2. Lodging
4.3. Remote Sensing of Lodging
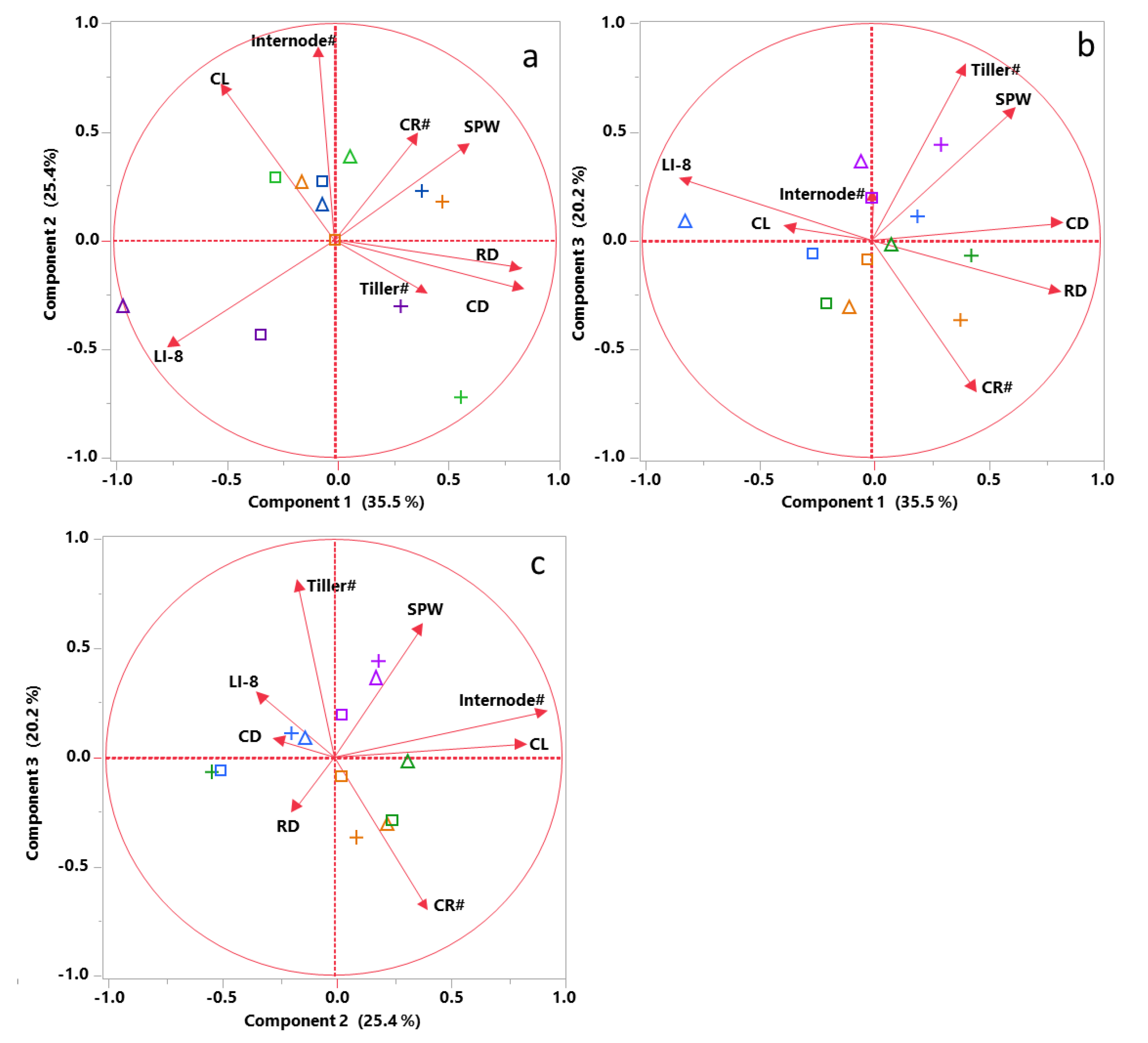
4.4. Lodging-Related Morphological Traits
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yu, J.-K.; Graznak, E.; Breseghello, F.; Tefera, H.; Sorrells, M.E. QTL mapping of agronomic traits in tef [Eragrostis tef (Zucc) Trotter]. BMC Plant Biol. 2007, 7, 2–13. [Google Scholar] [CrossRef]
- Gebre, Y.G.; Bertolini, E.; Pè, M.E.; Zuccolo, A. Identification and characterization of abundant repetitive sequences in Eragrostis tef cv. Enatite genome. BMC Plant Biol. 2016, 16, 39. [Google Scholar] [CrossRef]
- Tatham, A.S.; Fido, R.J.; Moore, C.M.; Kasarda, D.D.; Kuzmicky, D.D.; Keen, J.N.; Shewry, P.R. Characterisation of the major prolamins of tef (Eragrostis tef) and finger millet (Eleusine coracana). J. Cereal Sci. 1996, 24, 65–71. [Google Scholar] [CrossRef]
- Ketema, S. Tef (Eragrostis tef): Breeding, Genetic Resources, Agronomy, Utilization and Role in Ethiopian Agriculture; Institute of Agricultural Research: Adis Ababa, Ethiopia, 1993. [Google Scholar]
- D’Andrea, A.C. T’ef (Eragrostis tef) in ancient agricultural systems of highland Ethiopia. Econ. Bot. 2008, 62, 547–566. [Google Scholar]
- The Federal Democratic Republic of Ethiopia, Central Statistical Agency. Agricultural Sample Survey 2016/2017; Report on area and production of major crops; The Federal Democratic Republic of Ethiopia, Central Statistical Agency: Adis Ababa, Ethiopia, 2017; Volume I. [Google Scholar]
- National Research Council. Lost Crops of Africa: Volume I: Grains, 1st ed.; National Academies Press: Washington, DC, USA, 1996; ISBN 978-0-309-04990-0. [Google Scholar]
- Matthew Davidson, J. Evaluating teff Grass as a Summer Forage; College of Agriculture Manhattan: Kansas, NY, USA, 2018. [Google Scholar]
- Miller, D. Teff Grass: Crop Overview and Forage Production Guide; Cal/West Seed Company: Woodland, CA, USA, 2010. [Google Scholar]
- Paff, K.; Asseng, S. A review of tef physiology for developing a tef crop model. Eur. J. Agron. 2018, 94, 54–66. [Google Scholar] [CrossRef]
- Horowitz, S. New field crops for Palestine. Jewish Agency Palest. Agric. Res. Stn. Rehovot. 1940, 26, 29–38. [Google Scholar]
- Ben-Zeev, S.; Bimro, J.; Barak, V.; Saranga, Y. Phenotypic diversity and heritability in Eragrostis tef under irrigated Mediterranean conditions. Isr. J. Plant Sci. 2018, 65, 222–231. [Google Scholar] [CrossRef]
- Bimro, J. Agronomic Aspects of Teff (Eragrostis tef) Cultivation in Israel; The Hebrew University of Jerusalem: Rehovot, Israel, 2016. [Google Scholar]
- Kebede, H.; Johnson, R.C.; Ferris, D.M. Photosynthetic response of Eragrostis tef to temperature. Physiol. Plant. 1989, 77, 262–266. [Google Scholar] [CrossRef]
- Cohen-Shacham, E.; Dayan, T.; Feitelson, E.; de Groot, R.S. Compromis entre services écologiques dans la gestion de zones humides: Le drainage et la réhabilitation de la Hula, Israël. Hydrol. Sci. J. 2011, 56, 1582–1601. [Google Scholar] [CrossRef]
- Pinthus, M.J. Lodging in Wheat, Barley, and Oats: The Phenomenon, its Causes, and Preventive Measures. Adv. Agron. 1974, 25, 209–263. [Google Scholar]
- Ketema, S. Studies of Lodging Floral Biology and Bredding Techniques in Tef (Eragrostis tef); University of London: London, UK, 1983. [Google Scholar]
- Van Delden, S.H.; Vos, J.; Ennos, A.R.; Stomph, T.J. Analysing lodging of the panicle bearing cereal teff (Eragrostis tef). New Phytol. 2010, 186, 696–707. [Google Scholar] [CrossRef] [PubMed]
- Berry, P.M.; Sterling, M.; Spink, J.H.; Baker, C.J.; Sylvester-Bradley, R.; Mooney, S.J.; Tams, A.R.; Ennos, A.R. Understanding and Reducing Lodging in Cereals. Adv. Agron. 2004, 84, 217–271. [Google Scholar]
- Würschum, T.; Langer, S.M.; Longin, C.F.H.; Tucker, M.R.; Leiser, W.L. A modern Green Revolution gene for reduced height in wheat. Plant J. 2017, 92, 892–903. [Google Scholar]
- Lakew, A.; Berhanu, T. Determination of seeding rate and inter row spacing on the yield of tef (Eragrostis tef Zucc. Trotter) in the dryland areas of Wag Lasta, North Eastern Amhara, Ethiopia. Arch. Agric. Environ. Sci. 2019, 4, 69–74. [Google Scholar] [CrossRef]
- Miller, D. Teff As an Alternative Summer Forage. In Proceedings of the California and Forge Symp and Westren Seed Conference, San Diago, CA, USA, 2–4 December 2008. [Google Scholar]
- Sade, R.; Litaor, M.I.; Shenker, M. Evaluation of groundwater and phosphorus transport in fractured altered wetland soils. J. Hydrol. 2010, 393, 133–142. [Google Scholar] [CrossRef]
- Caldicott, J.J.B.; Nuttall, A.M. A method for the assessment of lodging in cereal crops. J. Natl. Inst. Agric. Bot. 1979, 15, 88–91. [Google Scholar]
- Kaiser, H.F. The Application of Eelectronic Computers to Factor Aalaysis. Educ. Psychol. Meas. 1960, 20, 141–151. [Google Scholar] [CrossRef]
- Negash, W. Effect of Row Spacing on Yield and Yield Components of Teff [Eragrostis tef (Zucc.) Trotter] Varieties in Gonji Kolela District, North Western Ethiopia. J. Biol. Agric. Healthc. 2017, 7, 35–43. [Google Scholar]
- Ketema, S. Eragrostis tef (Zucc.) Trotter: Promoting the Conservation and Use of Underutilized and Neglected Crops; Institute of Plant Genetics and Crop Plant Research, Gatersleben/International Plant Genetic Resources Institute: Rome, Italy, 1997; ISBN 9290433043. [Google Scholar]
- Jabesa, K.B.; Abraham, T. Effect of Different Row Spacing, Levels of Nitrogen and Phosphorus Fertilizers on Yield Attributes, Productivity and Economics of Tef (Eragrostis tef). Int. J. Agric. Environ. Biotechnol. 2017, 10, 603–608. [Google Scholar] [CrossRef]
- Dereje, G.; Adisu, T.; Dimberu, A. Influence of Seed Rate and Row Spacing on Growth and Yield of Tef (Eragrostis tef) Production at Assosa, Benishangul-Gumuz Regional State, Western Ethiopia. Adv. Crop Sci. Technol. 2018, 06, 1–3. [Google Scholar] [CrossRef]
- Shah, L.; Yahya, M.; Shah, S.M.A.; Nadeem, M.; Ali, A.; Wang, J.; Riaz, M.W.; Rehman, S.; Wu, W.; Khan, R.M.; et al. Improving lodging resistance: Using wheat and rice as classical examples. Int. J. Mol. Sci. 2019, 20, 4211. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Xie, R.Z.; Zhang, W.F.; Wang, K.R.; Hou, P.; Ming, B.; Gou, L.; LI, S. Research progress on reduced lodging of high-yield and -density maize. J. Integr. Agric. 2017, 16, 2717–2725. [Google Scholar] [CrossRef]
- Teetor, V.H.; Schmalzel, C.; Ray, D.T. Growing sweet sorghum (Sorghum bicolor [L.] moench) in clumps potentially reduces lodging in the arid-southwestern United States. Ind. Crops Prod. 2017, 107, 458–462. [Google Scholar] [CrossRef]
- Chapman, S.C.; Merz, T.; Chan, A.; Jackway, P.; Hrabar, S.; Dreccer, M.F.; Holland, E.; Zheng, B.; Ling, T.J.; Jimenez-Berni, J. Pheno-copter: A low-altitude, autonomous remote-sensing robotic helicopter for high-throughput field-based phenotyping. Agronomy 2014, 4, 279–301. [Google Scholar] [CrossRef]
- Yang, M.-D.; Huang, K.-S.; Kuo, Y.-H.; Tsai, H.; Lin, L.-M. Spatial and Spectral Hybrid Image Classification for Rice Lodging Assessment through UAV Imagery. Remote Sens. 2017, 9, 583. [Google Scholar] [CrossRef]
- Wilke, N.; Siegmann, B.; Klingbeil, L.; Burkart, A.; Kraska, T.; Muller, O.; van Doorn, A.; Heinemann, S.; Rascher, U. Quantifying Lodging Percentage and Lodging Severity Using a UAV-Based Canopy Height Model Combined with an Objective Threshold Approach. Remote Sens. 2019, 11, 515. [Google Scholar] [CrossRef]
- Assefa, K.; Ketema, S.; Tefera, H.; Kefyalew, T.; Hundera, F. Trait diversity, heritability and genetic advance in selected germplasm lines of tef [Eragrostis tef (Zucc.) Trotter]. Hereditas 2000, 133, 29–37. [Google Scholar] [CrossRef]
- Habteab, G.; Richard, B.; Shimelis, H.; Mark, L. The efficacy of four gametocides for induction of pollen sterility in Eragrostis tef (Zucc.) Trotter. Afr. J. Biotechnol. 2015, 14, 774–780. [Google Scholar] [CrossRef]
- Josst, M.; Esfeld, K.; Burian, A.; Cannarozzi, G.; Chanyalew, S.; Kuhlemeier, C.; Assefa, K.; Tadele, Z. Semi-dwarfism and lodging tolerance in tef (Eragrostis tef) is linked to a mutation in the alpha-Tubulin 1 gene. J. Exp. Bot. 2015, 66, 933–944. [Google Scholar] [CrossRef]
- Snider, J.L.; Raper, R.L.; Schwab, E.B. The effect of row spacing and seeding rate on biomass production and plant stand characteristics of non-irrigated photoperiod-sensitive sorghum (Sorghum bicolor (L.) Moench). Ind. Crops Prod. 2012, 37, 527–535. [Google Scholar] [CrossRef]
- Lafarge, T.A.; Broad, I.J.; Hammer, G.L. Tillering in grain sorghum over a wide range of population densities: Identification of a common hierarchy for tiller emergence, leaf area development and fertility. Ann. Bot. 2002, 90, 87–98. [Google Scholar] [CrossRef]
- Faris, D.G.; De Pauw, R.M. Effect of seeding rate on growth and yield of three spring wheat cultivars. F. Crop. Res. 1980, 3, 289–301. [Google Scholar] [CrossRef]
- Girma, D. The Relationships between Stem Characters and Lodging Tolerance in Tef (Eragrostis tef) Genotypes. Ethiop. J. Agric. Sci. 2019, 29, 59–70. [Google Scholar]
- Zeid, M.; Yu, J.-K.; Assefa, K.; Tefera, H.; Sorrells, M.E.; Belay, G. Breeding tef [Eragrostis tef (Zucc.) trotter]: Conventional and molecular approaches. Plant Breed. 2010, 130, 1–9. [Google Scholar]
- Zeid, M.; Belay, G.; Mulkey, S.; Poland, J.; Sorrells, M.E. QTL mapping for yield and lodging resistance in an enhanced SSR-based map for tef. Theor. Appl. Genet. 2011, 122, 77–93. [Google Scholar] [CrossRef] [PubMed]
- Adnew, T.; Ketema, S.; Tefera, H.; Sridhara, H. Genetic diversity in tef [Eragrostis tef (Zucc.) Trotter] germplasm. Genet. Resour. Crop Evol. 2005, 52, 891–902. [Google Scholar] [CrossRef]
- Berry, P.M.; Griffin, J.M.; Sylvester-Bradley, R.; Scott, R.K.; Spink, J.H.; Baker, C.J.; Clare, R.W. Controlling plant form through husbandry to minimise lodging in wheat. Field Crop. Res. 2000, 67, 59–81. [Google Scholar] [CrossRef]




| Source of Variance | Seedling Density 2018 | Seedling Density 2019 |
|---|---|---|
| Genotype effect (seedlings/m2) | ||
| RTC-2 | 517b | 395b |
| RTC-119 | 409c | 447a |
| RTC-361 | 631a | 452a |
| RTC-400 | 497b | 459a |
| Sowing rate effect (seedlings/m2) | ||
| 3 kg/ha | 270c | 266c |
| 6 kg/ha | 531b | 441b |
| 9 kg/ha | 740a | 607a |
| F ratio | ||
| Genotype (G) (df = 3) | 16.4 *** | 5.77 ** |
| Sowing rate (SR) (df = 2) | 145.2 *** | 248.5 *** |
| G*SR (df = 6) | 2.95 * | 0.81 |
| Block (df = 5) | 1.13 | 1.07 |
| Source of Variance | DPH | CL-12 (cm) | PL (cm) | TDM (g/m2) | GY (g/m2) | TSW (mg) |
|---|---|---|---|---|---|---|
| Genotype effect | ||||||
| RTC-2 | 57.7 | 86.7 | 45.4a | 2307 | 168.9 | 282.4b |
| RTC-119 | 56.7 | 83.0 | 48.6a | 2376 | 138.0 | 312.0a |
| RTC-361 | 56.3 | 81.8 | 40.6b | 2221 | 130.4 | 294.4b |
| RTC-400 | 57.4 | 84.6 | 46.0a | 2478 | 180.9 | 292.1b |
| Sowing rate effect | ||||||
| 3 kg/ha | 56.5 | 84.2 | 47.1a | 2286 | 170.8 | 298.7 |
| 6 kg/ha | 57.1 | 85.6 | 43.1b | 2360 | 142.7 | 293.3 |
| 9 kg/ha | 57.5 | 82.3 | 45.1ab | 2390 | 150.1 | 294.1 |
| F ratio | ||||||
| Genotype (G) (df = 3) | 1.29 | 2.34 | 7.96 *** | 0.62 | 2.44 | 9.8 *** |
| Sowing rate (SR) (df = 2) | 0.94 | 1.92 | 3.65 * | 0.20 | 1.17 | 0.7 |
| G*SR (df = 6) | 1.23 | 1.09 | 0.45 | 1.33 | 0.86 | 1.3 |
| Block (df = 5) | 2.94 * | 5.59 *** | 0.71 | 2.00 | 3.36 * | 1.2 |
| Source of Variance | DPH | CL-12 (cm) | PL (cm) | TDM (g/m2) | GY (g/m2) | TSW (mg) |
|---|---|---|---|---|---|---|
| Genotype effect | ||||||
| RTC-2 | 60.0b | 68.7 | 44.9 | 1855ab | 81.4 | 267.5 |
| RTC-119 | 56.9c | 70.5 | 43.5 | 1709ab | 84.5 | 248.8 |
| RTC-361 | 58.2bc | 72.3 | 41.1 | 1645b | 89.8 | 253.0 |
| RTC-400 | 66.3a | 70.8 | 45.2 | 2018a | 99.8 | 239.6 |
| Sowing rate effect | ||||||
| 3 kg/ha | 59.6 | 71.8 | 45.26 | 1712 | 90.4 | 249.9 |
| 6 kg/ha | 61.2 | 69.6 | 42.45 | 1845 | 92.2 | 249.8 |
| 9 kg/ha | 60.2 | 70.3 | 43.29 | 1864 | 84.0 | 257.0 |
| F ratio | ||||||
| Genotype (df = 3) | 27.38 *** | 1.74 | 1.53 | 2.88 * | 0.54 | 1.2 |
| Sowing rate (df = 2) | 1.52 | 1.45 | 1.32 | 0.99 | 0.20 | 0.2 |
| G*SR (df = 6) | 0.83 | 2.34 * | 1.03 | 0.85 | 1.17 | 0.9 |
| Block (df = 5) | 2.69 * | 2.88 * | 1.70 | 3.47 ** | 3.54 ** | 1.9 |
| Source of Variance | LI-8 | CD (mm) | Tiller # | Internode # | SPW (g) | CL-8 (cm) | RD (mm) | CR# |
|---|---|---|---|---|---|---|---|---|
| Genotype effect | ||||||||
| RTC-2 | 518.4b | 6.36 | 4.47 | 5.33 | 4.09 | 72.06 | 0.64 | 11.30 |
| RTC-119 | 435.7b | 6.16 | 4.24 | 4.98 | 3.42 | 69.10 | 0.65 | 12.88 |
| RTC-361 | 450.5b | 6.50 | 3.65 | 5.20 | 3.32 | 70.10 | 0.67 | 12.62 |
| RTC-400 | 710.9a | 6.17 | 4.17 | 4.94 | 3.14 | 69.77 | 0.63 | 11.11 |
| Sowing rate effect | ||||||||
| 3 kg/ha | 454.8b | 6.80a | 4.33 | 5.01 | 3.74 | 67.61 | 0.68a | 12.06 |
| 6 kg/ha | 541.5a | 6.11b | 4.14 | 5.21 | 3.33 | 72.51 | 0.61b | 11.79 |
| 9 kg/ha | 584.3a | 5.98b | 3.92 | 5.12 | 3.40 | 70.64 | 0.65ab | 12.08 |
| F ratio | ||||||||
| Genotype (df = 3) | 25.2 *** | 0.66 | 1.90 | 2.03 | 2.30 | 0.21 | 1.08 | 1.47 |
| Sowing Rate (df = 2) | 9.4 *** | 6.41 ** | 0.86 | 0.87 | 0.90 | 1.07 | 6.50 ** | 0.06 |
| G*SR (df = 6) | 1.43 | 1.45 | 0.62 | 0.72 | 0.32 | 0.31 | 0.95 | 0.89 |
| Block (df = 5) | 2.24 * | 0.88 | 0.65 | 3.24 * | 0.88 | 1.46 | 0.83 | 0.84 |
| LI-8 | RSmean/RSmax | RSmode/RSmax | RSmean/CL-8 | RSmode/CL-8 | |
|---|---|---|---|---|---|
| Tiller# | −0.09 | 0.17 | 0.07 | 0.22 | 0.11 |
| CL-8 | 0.13 | −0.22 | −0.3 | −0.71 ** | −0.60 * |
| Internode# | −0.23 | 0.11 | 0.06 | −0.37 | −0.25 |
| CR# | −0.67 * | 0.51 | 0.58 * | 0.4 | 0.5 |
| CD | −0.45 | 0.57 * | 0.67 * | 0.51 | 0.62 * |
| RD | −0.58 * | 0.54 | 0.65 * | 0.64 * | 0.66 * |
| SPW | −0.47 | 0.55 | 0.42 | 0.26 | 0.24 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ben-Zeev, S.; Rabinovitz, O.; Orlov-Levin, V.; Chen, A.; Graff, N.; Goldwasser, Y.; Saranga, Y. Less Is More: Lower Sowing Rate of Irrigated Tef (Eragrostis tef) Alters Plant Morphology and Reduces Lodging. Agronomy 2020, 10, 570. https://doi.org/10.3390/agronomy10040570
Ben-Zeev S, Rabinovitz O, Orlov-Levin V, Chen A, Graff N, Goldwasser Y, Saranga Y. Less Is More: Lower Sowing Rate of Irrigated Tef (Eragrostis tef) Alters Plant Morphology and Reduces Lodging. Agronomy. 2020; 10(4):570. https://doi.org/10.3390/agronomy10040570
Chicago/Turabian StyleBen-Zeev, Shiran, Onn Rabinovitz, Valerie Orlov-Levin, Assaf Chen, Nitsan Graff, Yarden Goldwasser, and Yehoshua Saranga. 2020. "Less Is More: Lower Sowing Rate of Irrigated Tef (Eragrostis tef) Alters Plant Morphology and Reduces Lodging" Agronomy 10, no. 4: 570. https://doi.org/10.3390/agronomy10040570
APA StyleBen-Zeev, S., Rabinovitz, O., Orlov-Levin, V., Chen, A., Graff, N., Goldwasser, Y., & Saranga, Y. (2020). Less Is More: Lower Sowing Rate of Irrigated Tef (Eragrostis tef) Alters Plant Morphology and Reduces Lodging. Agronomy, 10(4), 570. https://doi.org/10.3390/agronomy10040570






