Response of Photosynthetic Performance to Drought Duration and Re-Watering in Maize
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Growth Conditions
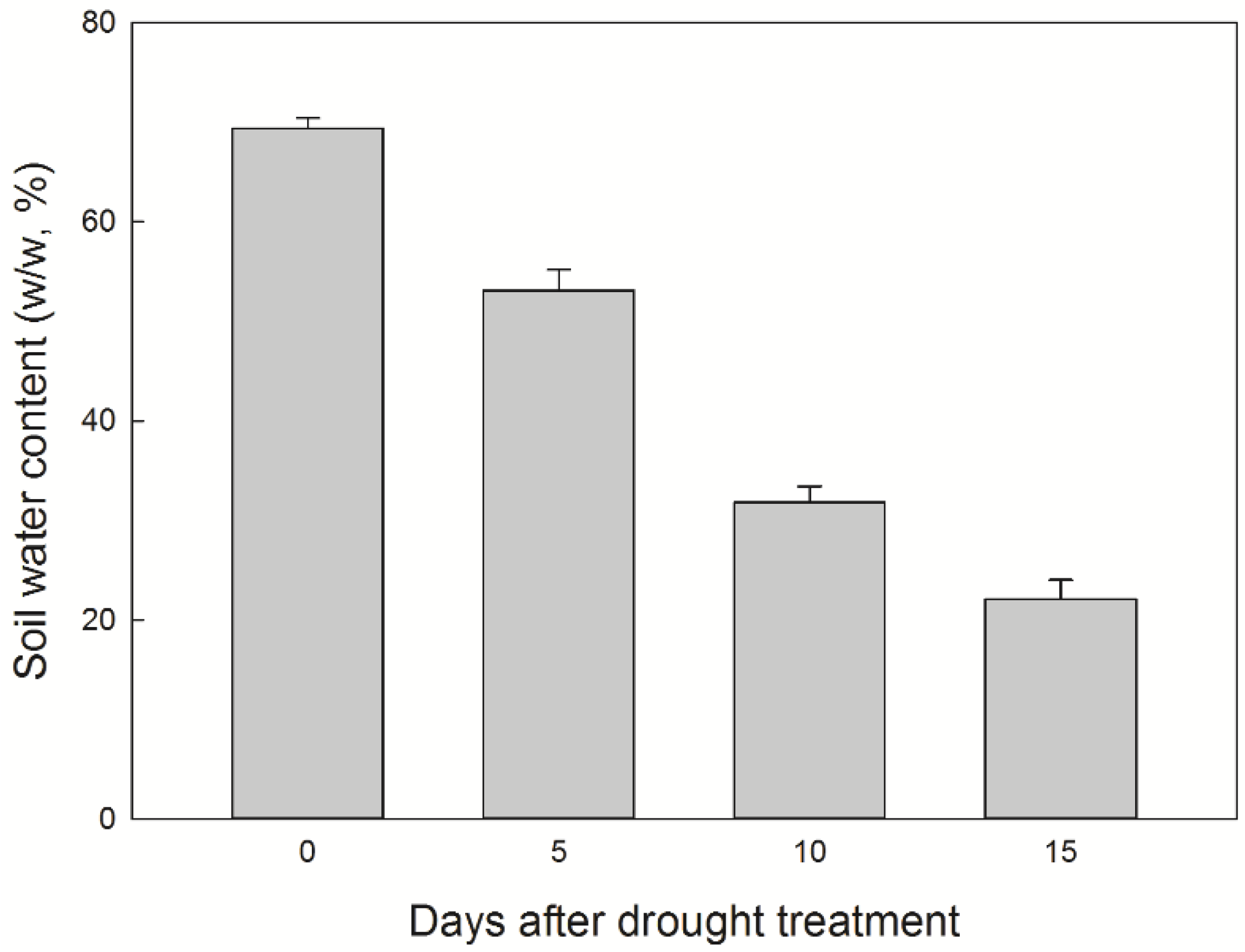
2.2. Soil Water Content
2.3. Gas Exchange Parameters
2.4. Slow Chlorophyll Fluorescence Kinetics
2.5. Chlorophyll a Fluorescence Transient
2.6. Measurement of H2O2 Content, MDA Content, and Antioxidant Enzyme Activity
2.7. Data Analysis
3. Results
3.1. Soil Water Content
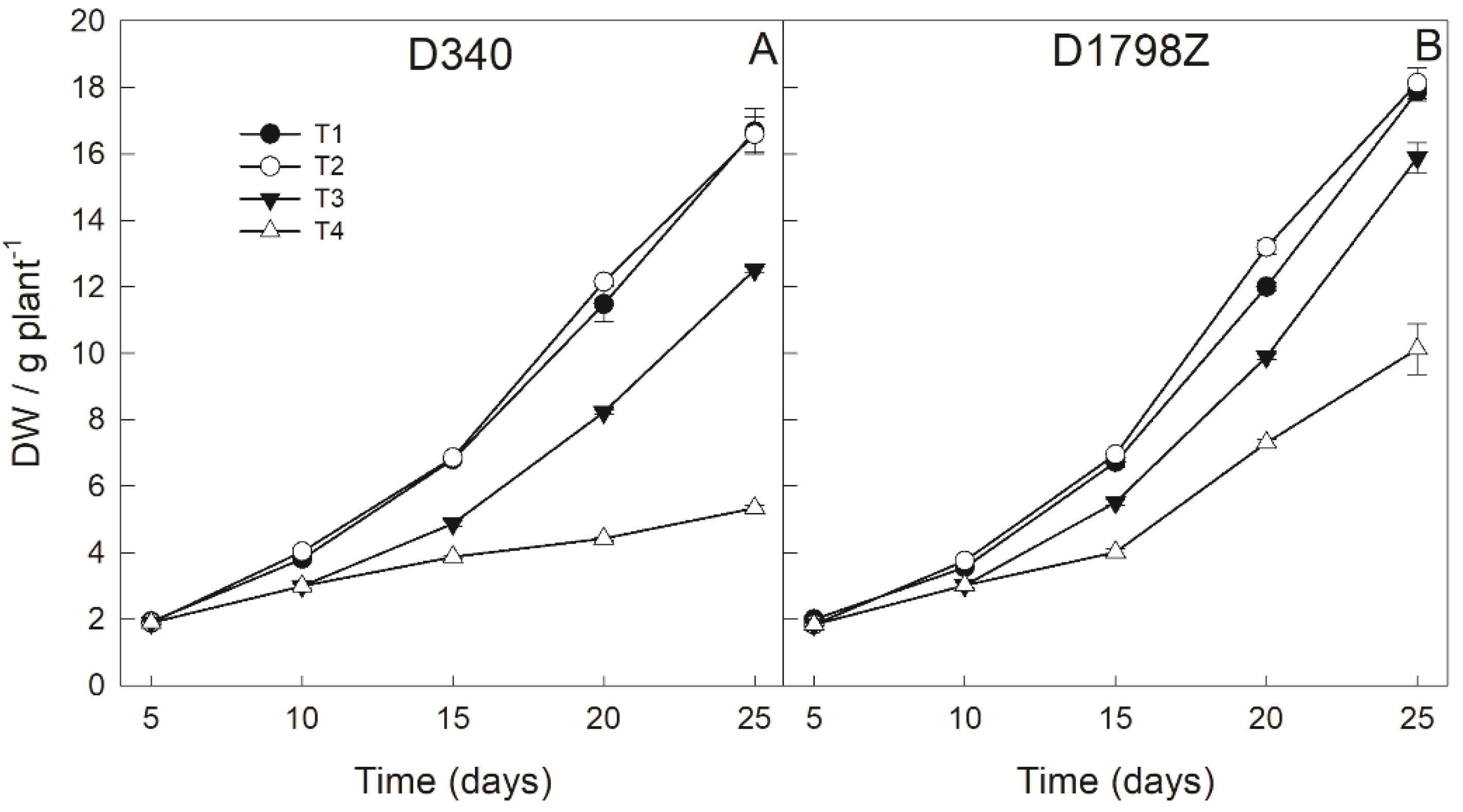
3.2. Plant Growth
3.3. Gas Exchange
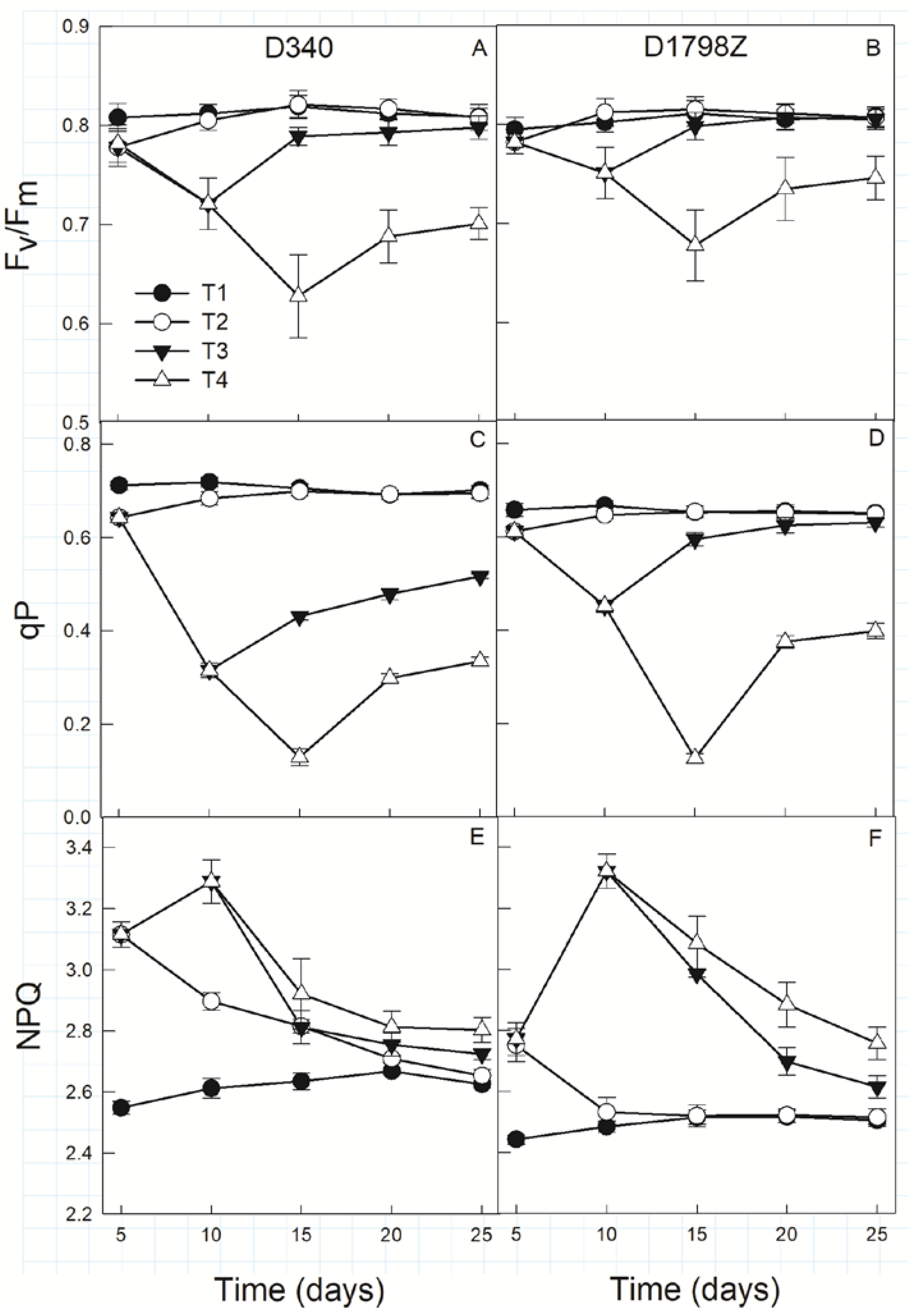
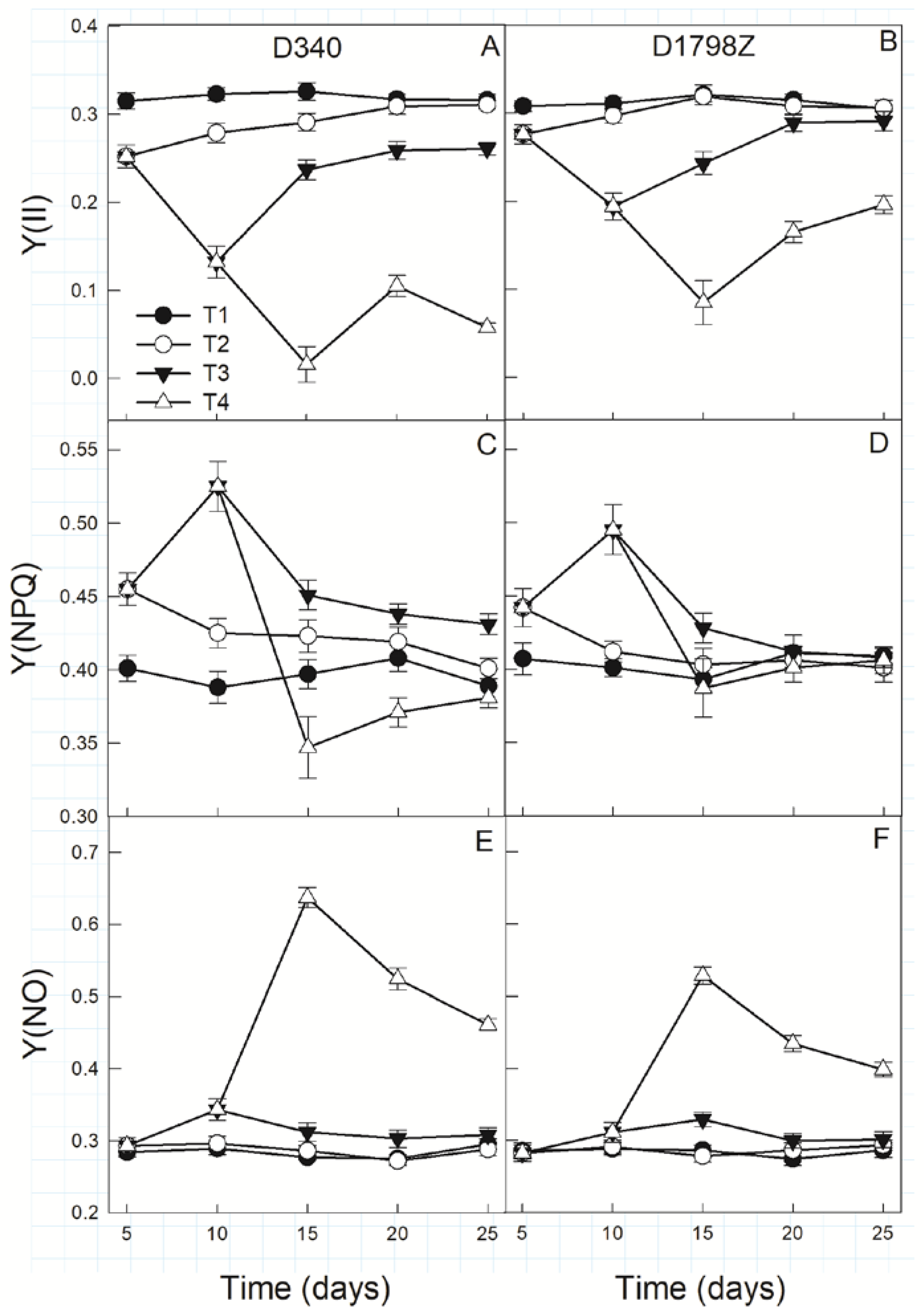
3.4. Chlorophyll Fluorescence
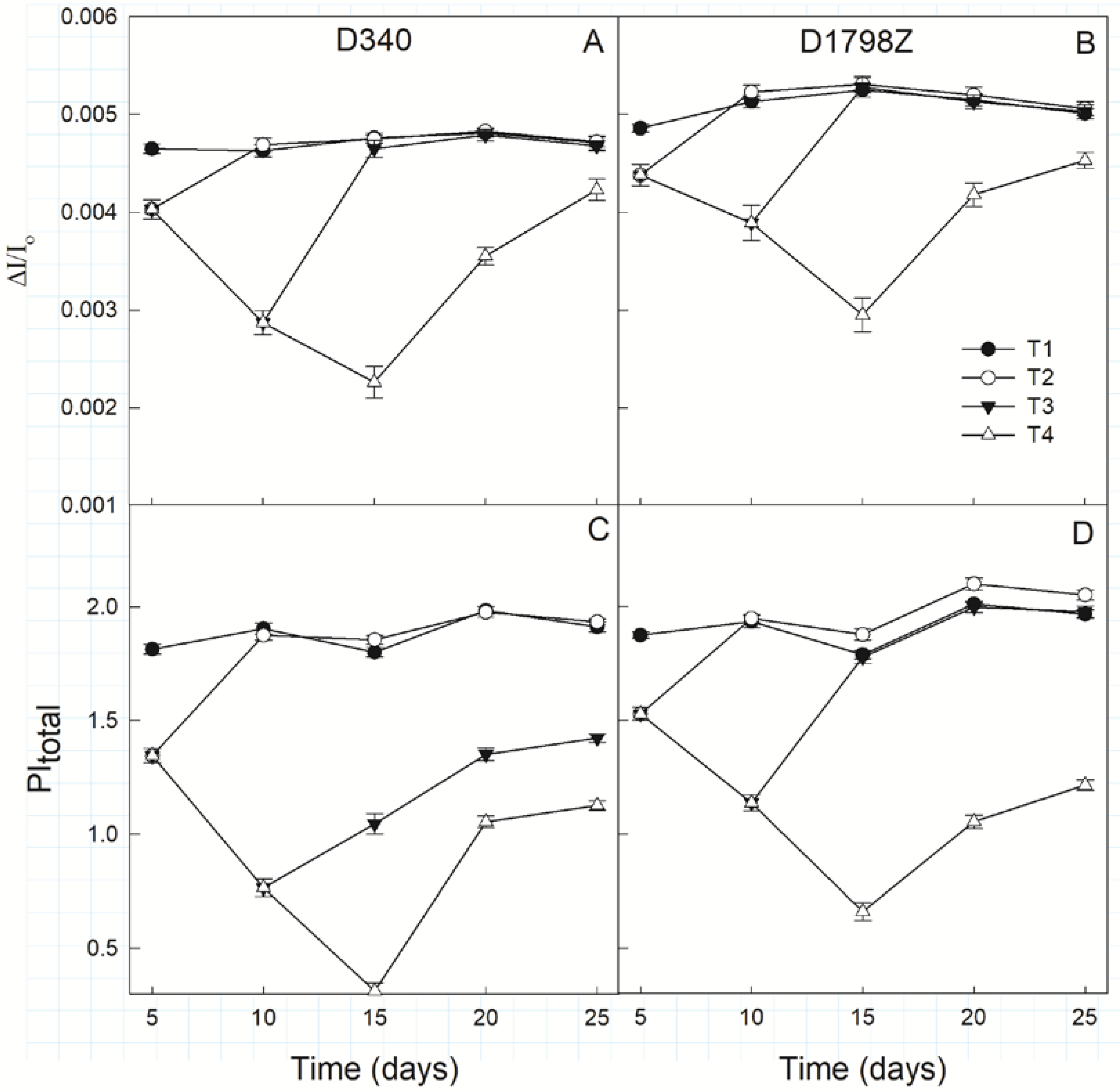
3.5. Fluorescence Parameters from OJIP Transients
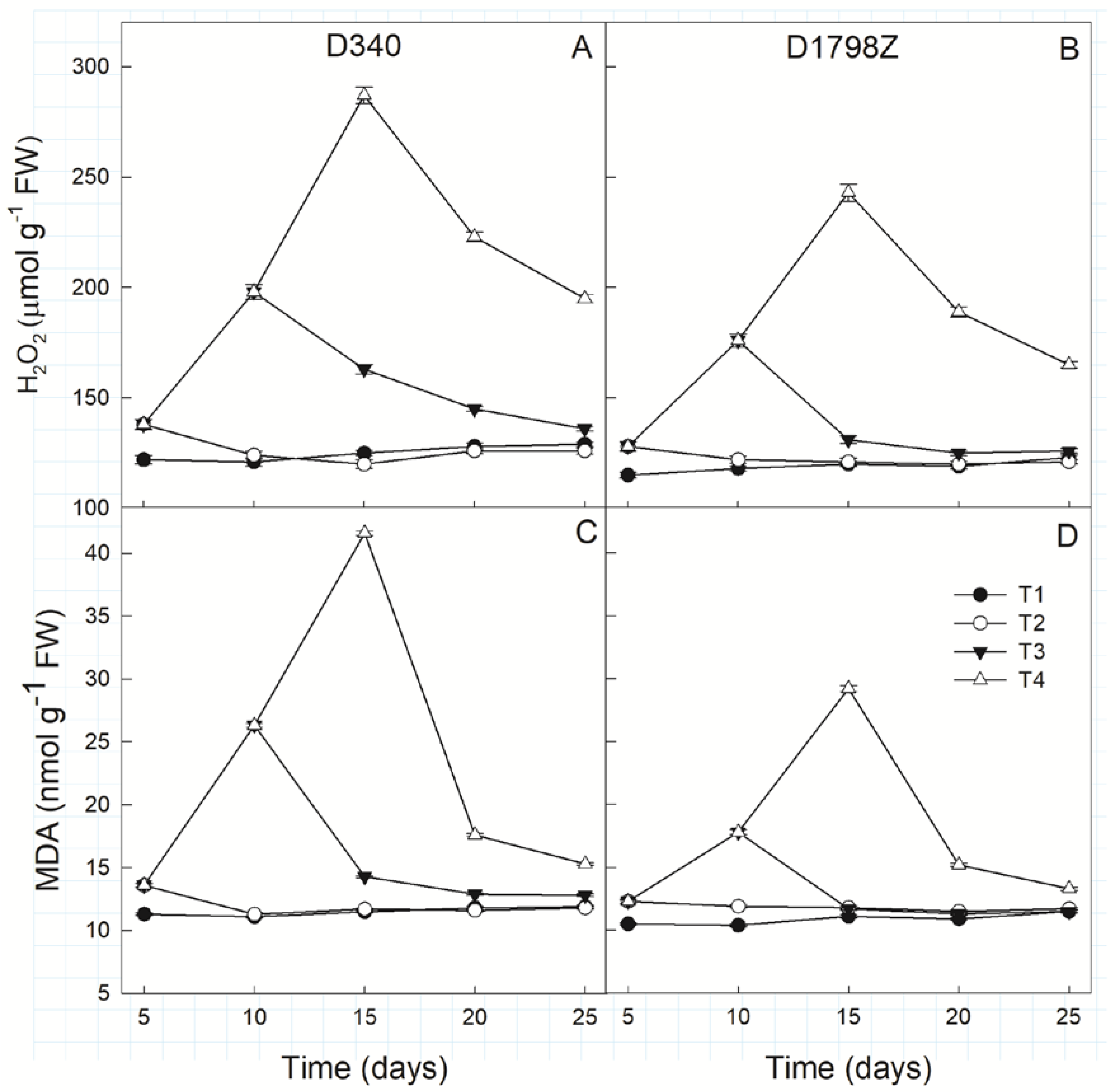
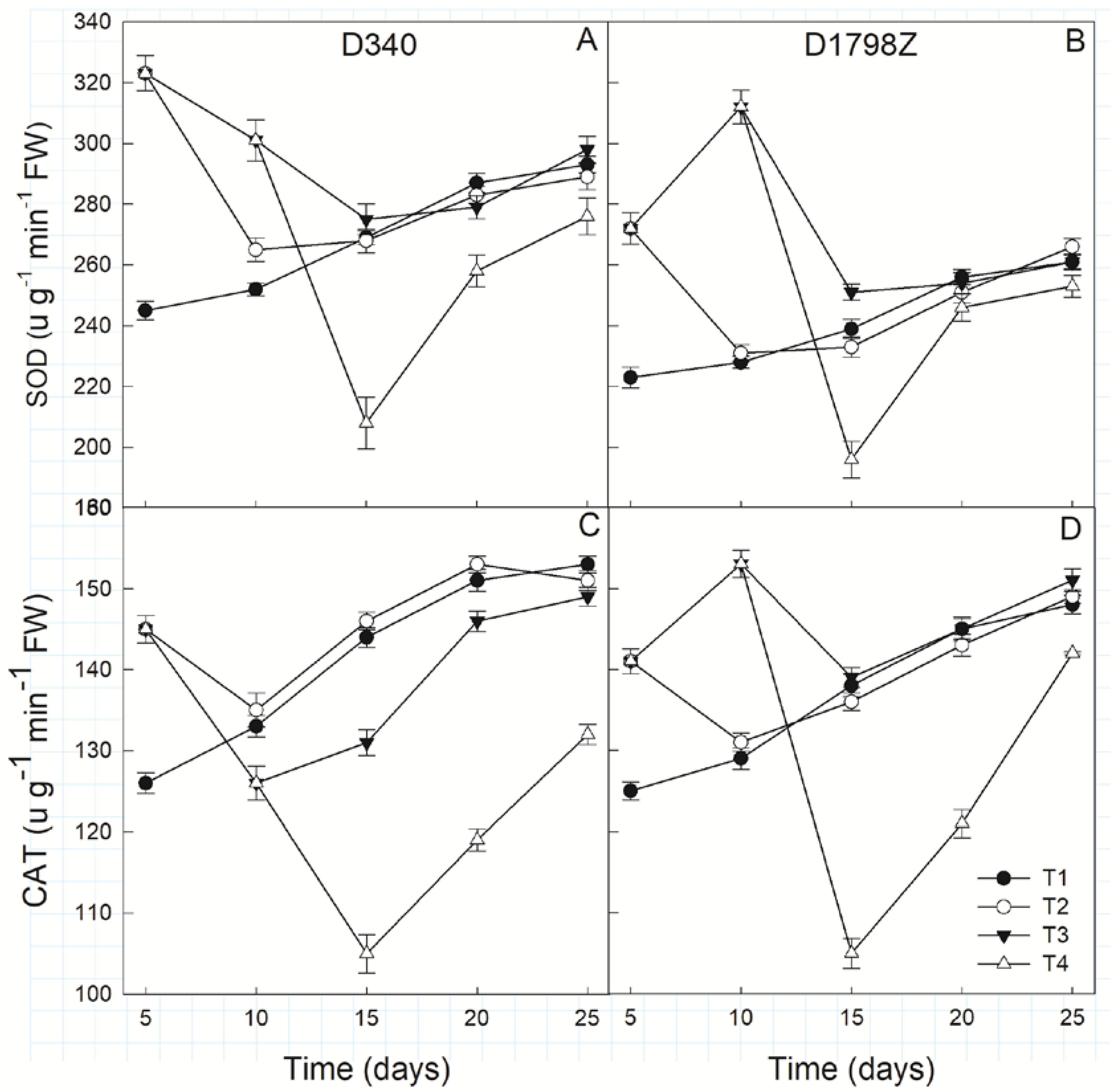
3.6. ROS Production and Scavenging
4. Discussion
4.1. The Response of Leaf Functionality to Different Drought Duration
4.2. The Recovery of Leaf Functionality to Different Drought Duration
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Field, C.D.; Barros, V.R.; Dokken, D.J.; Mach, K.J.; Mastrandrea, M.D.; Bilir, T.E.; Chatterjee, M.; Ebi, K.L.; Estrada, Y.O.; Genova, R.C.; et al. Climate Change 2014: Impacts, Adaptation, and Vulnerability, Part A. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2014; pp. 1–1132. [Google Scholar]
- Mancosu, N.; Snyder, R.L.; Kyriakakis, G.; Spano, D. Water Scarcity and Future Challenges for Food Production. Water 2015, 7, 975–992. [Google Scholar] [CrossRef]
- Hussain, M.; Farooq, S.; Hasan, W.; Ul-Allah, S.; Tanveer, M.; Farooq, M.; Nawaz, A. Drought stress in sunflower: Physiological effects and its management through breeding and agronomic alternatives. Agric. Water Manag. 2018, 201, 152–166. [Google Scholar] [CrossRef]
- Ogbaga, C.; Stepien, P.; Johnson, G. Sorghum (Sorghum bicolor) varieties adopt strongly contrasting strategies in response to drought. Physiol. Plant. 2014, 152, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wilkinson, S.; Shen, J.; Forde, B.; Davies, W.J. Stomatal and growth responses to hydraulic and chemical changes induced by progressive soil drying. J. Exp. Bot. 2017, 68, 5883–5894. [Google Scholar] [CrossRef] [PubMed]
- Tuberosa, R.; Salvi, S.; Giuliani, S.; Sanguineti, M.C.; Bellotti, M.; Conti, S.; Landi, P. Genome-wide Approaches to Investigate and Improve Maize Response to Drought. Crop. Sci. 2007, 47, 120–141. [Google Scholar] [CrossRef]
- Neto, M.L.; Cerqueira, J.V.A.; Cunha, J.R.; Ribeiro, R.V.; Silveira, J.A.G. Cyclic electron flow, NPQ and photorespiration are crucial for the establishment of young plants of Ricinus communisandJatropha curcasexposed to drought. Plant Boil. 2017, 19, 650–659. [Google Scholar] [CrossRef]
- Xu, Z.; Zhou, G. Responses of leaf stomatal density to water status and its relationship with photosynthesis in a grass. J. Exp. Bot. 2008, 59, 3317–3325. [Google Scholar] [CrossRef]
- Lawson, T.; Blatt, M.R. Stomatal Size, Speed, and Responsiveness Impact on Photosynthesis and Water Use Efficiency1. Plant Physiol. 2014, 164, 1556–1570. [Google Scholar] [CrossRef]
- Goltsev, V.; Zaharieva, I.; Chernev, P.; Kouzmanova, M.; Kalaji, H.M.; Yordanov, I.; Krasteva, V.; Aleksandrov, V.; Stefanov, D.; Allakhverdiev, S.I.; et al. Drought-induced modifications of photosynthetic electron transport in intact leaves: Analysis and use of neural networks as a tool for a rapid non-invasive estimation. Biochim. Biophys. Acta (BBA) Bioenerg. 2012, 1817, 1490–1498. [Google Scholar] [CrossRef]
- Zhou, R.; Kan, X.; Chen, J.; Hua, H.; Li, Y.; Ren, J.; Feng, K.; Liu, H.; Deng, D.; Xu, C. Drought-induced changes in photosynthetic electron transport in maize probed by prompt fluorescence, delayed fluorescence, P700 and cyclic electron flow signals. Environ. Exp. Bot. 2019, 158, 51–62. [Google Scholar] [CrossRef]
- Osakabe, Y.; Osakabe, K.; Shinozaki, K.; Tran, L.-S.P. Response of plants to water stress. Front. Plant Sci. 2014, 5, 86. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-E.; Liu, W.-J.; Su, Y.-Q.; Cui, J.-M.; Zhang, Z.-W.; Yuan, M.; Zhang, H.-Y.; Yuan, S. Different response of photosystem II to short and long-term drought stress inArabidopsis thaliana. Physiol. Plant. 2016, 158, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Sousa, C.A.F.; De Paiva, D.S.; Casari, R.A.D.C.N.; De Oliveira, N.G.; Molinari, H.B.C.; Kobayashi, A.K.; Magalhães, P.C.; Gomide, R.L.; Souza, M.T., Jr. A procedure for maize genotypes discrimination to drought by chlorophyll fluorescence imaging rapid light curves. Plant Methods 2017, 13, 61. [Google Scholar] [CrossRef]
- Oukarroum, A.; Schansker, G.; Strasser, R.J. Drought stress effects on photosystem I content and photosystem II thermotolerance analyzed using Chlafluorescence kinetics in barley varieties differing in their drought tolerance. Physiol. Plant. 2009, 137, 188–199. [Google Scholar] [CrossRef] [PubMed]
- Kalaji, H.M.; Kalaji, H.M.; Oukarroum, A.; Brestič, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Łukasik, I.; Goltsev, V.; Ladle, R.J. Chlorophyll a fluorescence as a tool to monitor physiological status of plants under abiotic stress conditions. Acta Physiol. Plant. 2016, 38, 102. [Google Scholar] [CrossRef]
- Yao, X.; Zhou, H.; Zhu, Q.; Li, C.; Zhang, H.; Wu, J.-J.; Xie, F. Photosynthetic Response of Soybean Leaf to Wide Light-Fluctuation in Maize-Soybean Intercropping System. Front. Plant Sci. 2017, 8, 8. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Račková, L.; Paganová, V.; Swoczyna, T.; Rusinowski, S.; Sitko, K. Can chlorophyll-a fluorescence parameters be used as bio-indicators to distinguish between drought and salinity stress in Tilia cordata Mill? Environ. Exp. Bot. 2018, 152, 149–157. [Google Scholar] [CrossRef]
- Lima, C.S.; Ferreira-Silva, S.L.; Carvalho, F.E.L.; Neto, M.L.; Aragão, R.M.; Silva, E.N.; Sousa, R.M.J.; Silveira, J.A.G. Antioxidant protection and PSII regulation mitigate photo-oxidative stress induced by drought followed by high light in cashew plants. Environ. Exp. Bot. 2018, 149, 59–69. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. Is Phytomelatonin a New Plant Hormone? Agronomy 2020, 10, 95. [Google Scholar] [CrossRef]
- Arnao, M.B.; Hernández-Ruiz, J. Melatonin: A New Plant Hormone and/or a Plant Master Regulator? Trends Plant Sci. 2019, 24, 38–48. [Google Scholar] [CrossRef]
- Zhang, R.H.; Zhang, X.H.; Camberato, J.J.; Xue, J.Q. Photosynthetic performance of maize hybrids to drought stress. Russ. J. Plant Physiol. 2015, 62, 788–796. [Google Scholar] [CrossRef]
- Zong, Y.Z.; Wang, W.F.; Xue, Q.; Shangguan, Z. Interactive effects of elevated CO2 and drought on photosynthetic capacity and PSII performance in maize. Photosynthetica 2014, 52, 63–70. [Google Scholar] [CrossRef]
- Vitale, L.; Di Tommasi, P.; Arena, C.; Fierro, A.; De Santo, A.V.; Magliulo, V. Effects of water stress on gas exchange of field grown Zea mays L. in Southern Italy: An analysis at canopy and leaf level. Acta Physiol. Plant. 2007, 29, 317–326. [Google Scholar] [CrossRef]
- Tůmová, L.; Tarkowská, D.; Rehorova, K.; Marková, H.; Kočová, M.; Rothová, O.; Čečetka, P.; Holá, D. Drought-tolerant and drought-sensitive genotypes of maize (Zea mays L.) differ in contents of endogenous brassinosteroids and their drought-induced changes. PLoS ONE 2018, 13, e0197870. [Google Scholar] [CrossRef]
- Strasser, R.J.; Tsimilli-Michael, M.; Srivastava, A. Analysis of the Chlorophyll a Fluorescence Transient. Plant Cell Monographs 2004, 19, 321–362. [Google Scholar]
- Strasser, R.J.; Tsimilli-Michael, M.; Qiang, S.; Goltsev, V. Simultaneous in vivo recording of prompt and delayed fluorescence and 820-nm reflection changes during drying and after rehydration of the resurrection plant Haberlea rhodopensis. Biochim. Biophys. Acta (BBA) Gen. Subj. 2010, 1797, 1313–1326. [Google Scholar] [CrossRef]
- Guo, Y.Y.; Tian, S.S.; Liu, S.S.; Wang, W.Q.; Sui, N. Energy dissipation and antioxidant enzyme system protect photosystem II of sweet sorghum under drought stress. Photosynthetica 2017, 56, 861–872. [Google Scholar] [CrossRef]
- Spicher, L.; Glauser, G.; Kessler, F. Lipid Antioxidant and Galactolipid Remodeling under Temperature Stress in Tomato Plants. Front. Plant Sci. 2016, 7, 726. [Google Scholar] [CrossRef]
- Samantary, S. Biochemical responses of Cr-tolerant and Cr-sensitive mung bean cultivars grown on varying levels of chromium. Chemosphere 2002, 47, 1065–1072. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Methods Enzym. 1984, 105, 121–126. [Google Scholar]
- Sengupta, D.; Guha, A.; Reddy, A.R. Interdependence of plant water status with photosynthetic performance and root defense responses in Vigna radiata (L.) Wilczek under progressive drought stress and recovery. J. Photochem. Photobiol. B Boil. 2013, 127, 170–181. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, H.; Li, Y.; Zhang, S. Improving water-use efficiency by decreasing stomatal conductance and transpiration rate to maintain higher ear photosynthetic rate in drought-resistant wheat. Crop. J. 2017, 5, 231–239. [Google Scholar] [CrossRef]
- Ashraf, M.; Harris, P.J.C. Photosynthesis under stressful environments: An overview. Photosynthetica 2013, 51, 163–190. [Google Scholar] [CrossRef]
- Singh, R.; Naskar, J.; Pathre, U.V.; Shirke, P.A. Reflectance and Cyclic Electron Flow as an Indicator of Drought Stress in Cotton (Gossypium hirsutum). Photochem. Photobiol. 2013, 90, 544–551. [Google Scholar] [CrossRef]
- Gill, S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Rangani, J.; Panda, A.; Patel, M.; Parida, A.K. Regulation of ROS through proficient modulations of antioxidative defense system maintains the structural and functional integrity of photosynthetic apparatus and confers drought tolerance in the facultative halophyte Salvadora persica L. J. Photochem. Photobiol. B Boil. 2018, 189, 214–233. [Google Scholar] [CrossRef]
- Xu, Z.; Zhou, G.; Shimizu, H. Plant responses to drought and rewatering. Plant Signal. Behav. 2010, 5, 649–654. [Google Scholar] [CrossRef]
- Abid, M.; Tian, Z.; Ata-Ul-Karim, S.T.; Wang, F.; Liu, Y.; Zahoor, R.; Jiang, N.; Dai, T. Adaptation to and recovery from drought stress at vegetative stages in wheat (Triticum aestivum) cultivars. Funct. Plant Boil. 2016, 43, 1159. [Google Scholar] [CrossRef]
- Song, H.; Li, Y.; Zhou, L.; Xu, Z.; Zhou, G. Maize leaf functional responses to drought episode and rewatering. Agric. For. Meteorol. 2018, 249, 57–70. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, Y.; Xiao, W.; Ye, Y.; Wang, X.; Liu, X.; Wang, G.; Li, G.; Wang, Y. Response of Photosynthetic Performance to Drought Duration and Re-Watering in Maize. Agronomy 2020, 10, 533. https://doi.org/10.3390/agronomy10040533
Jia Y, Xiao W, Ye Y, Wang X, Liu X, Wang G, Li G, Wang Y. Response of Photosynthetic Performance to Drought Duration and Re-Watering in Maize. Agronomy. 2020; 10(4):533. https://doi.org/10.3390/agronomy10040533
Chicago/Turabian StyleJia, Yuying, Wanxin Xiao, Yusheng Ye, Xiaolin Wang, Xiaoli Liu, Guohong Wang, Gang Li, and Yanbo Wang. 2020. "Response of Photosynthetic Performance to Drought Duration and Re-Watering in Maize" Agronomy 10, no. 4: 533. https://doi.org/10.3390/agronomy10040533
APA StyleJia, Y., Xiao, W., Ye, Y., Wang, X., Liu, X., Wang, G., Li, G., & Wang, Y. (2020). Response of Photosynthetic Performance to Drought Duration and Re-Watering in Maize. Agronomy, 10(4), 533. https://doi.org/10.3390/agronomy10040533



