Soil Macrofauna: A key Factor for Increasing Soil Fertility and Promoting Sustainable Soil Use in Fruit Orchard Agrosystems
Abstract
1. Introduction
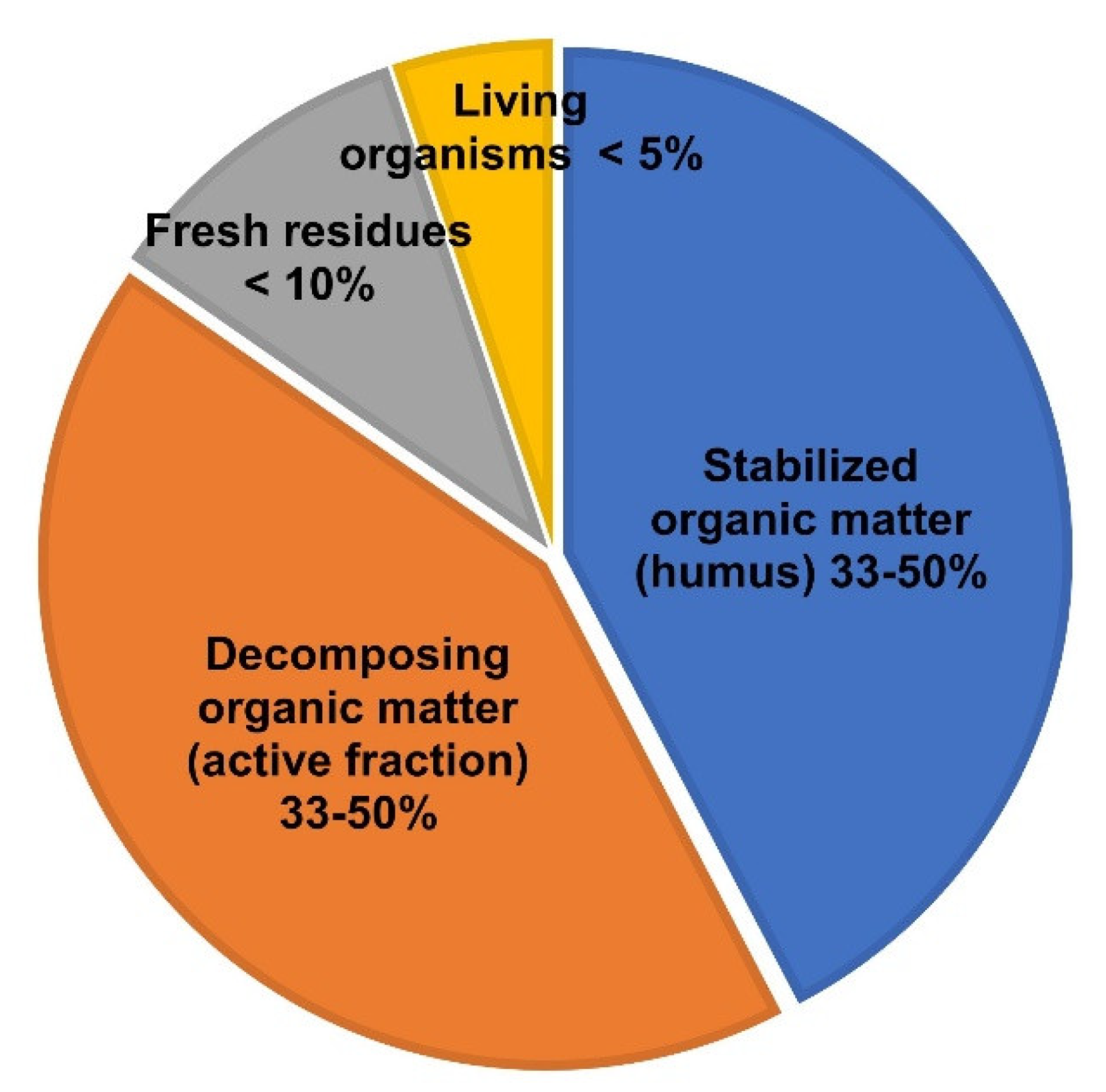
2. The Scenario: Soil Organic Matter
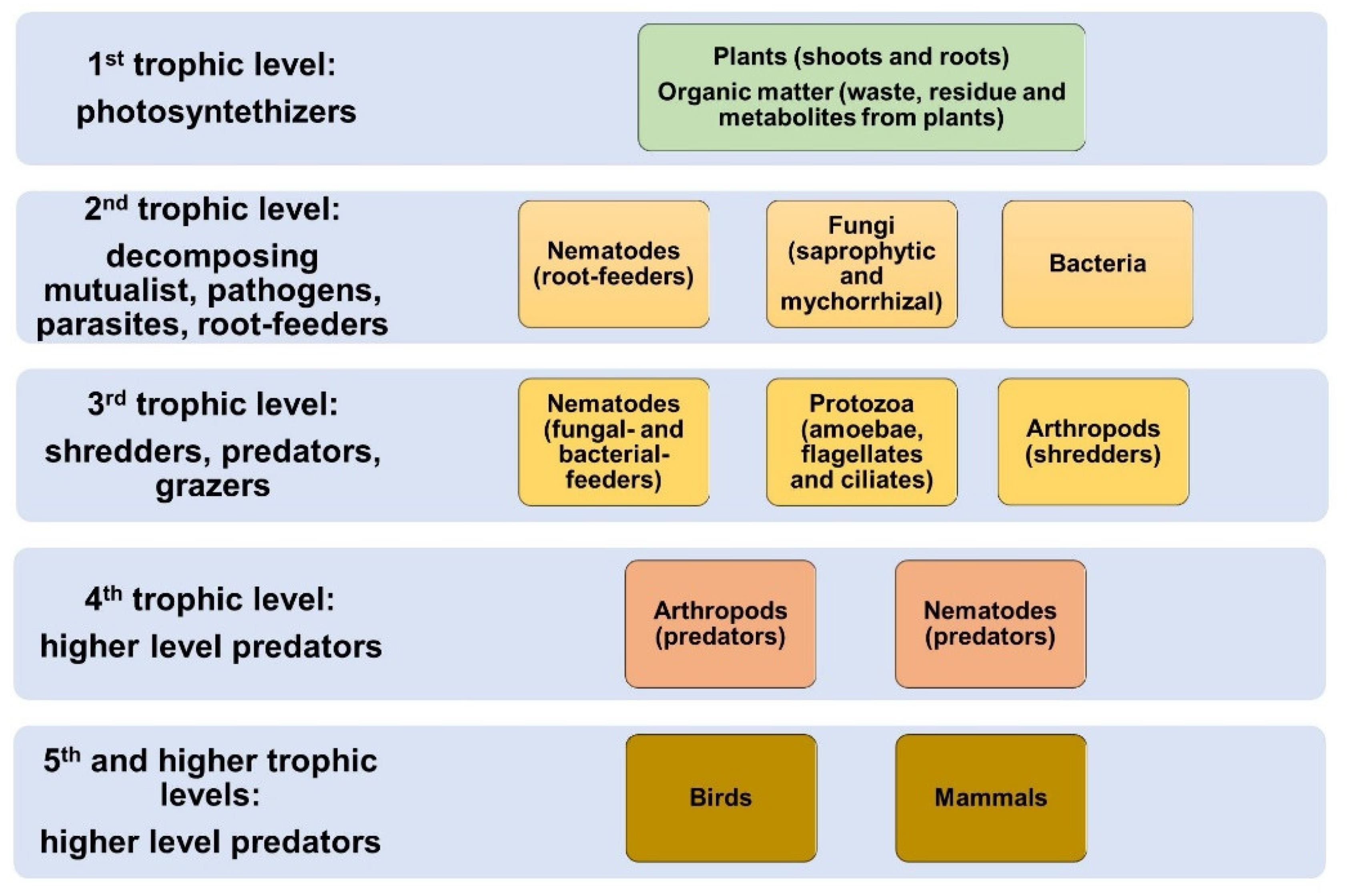
3. The Actors: Soil Biota in Agrosystems
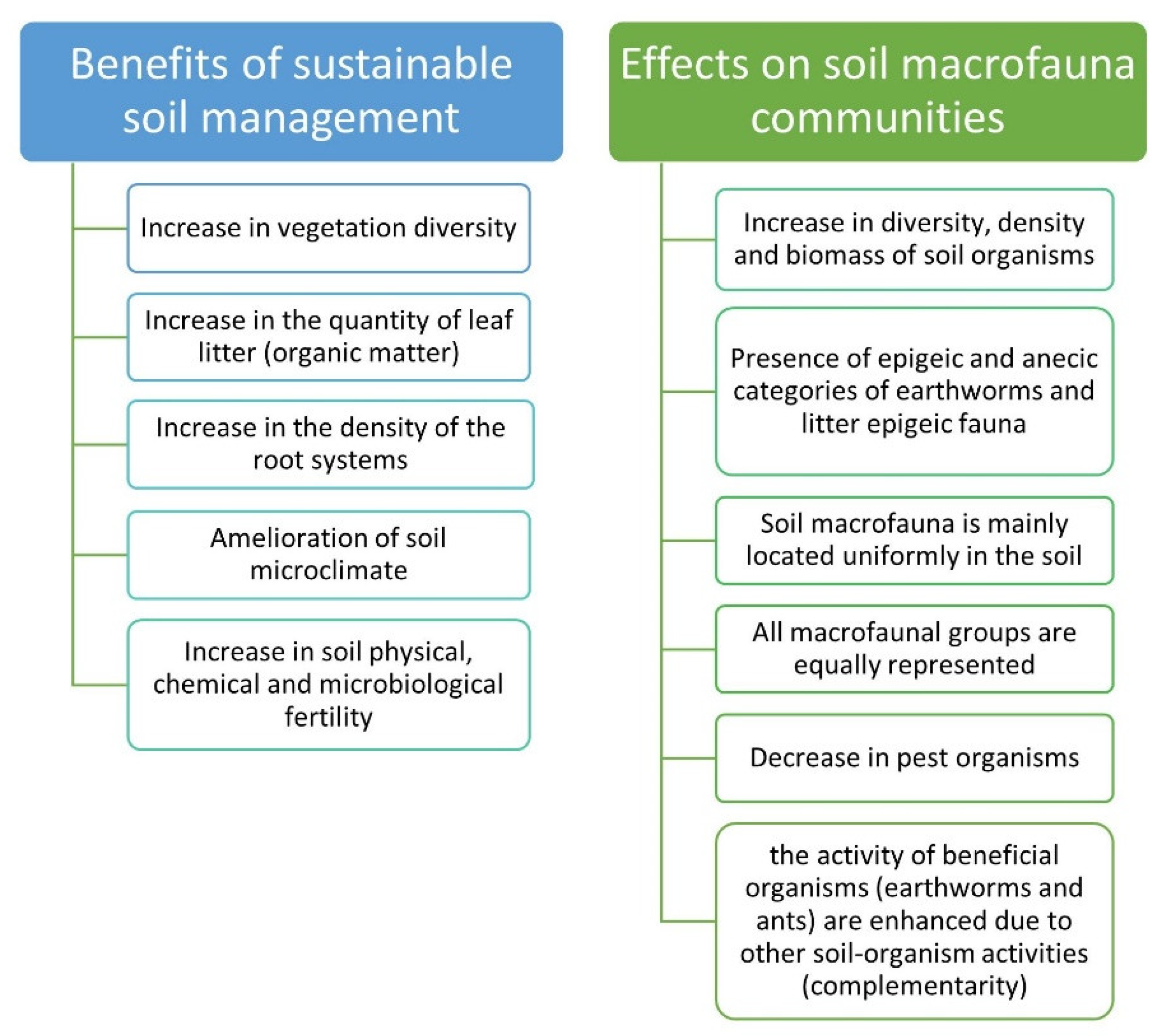
4. Soil Macrofauna Abundance and Activity Are Related to Soil Management
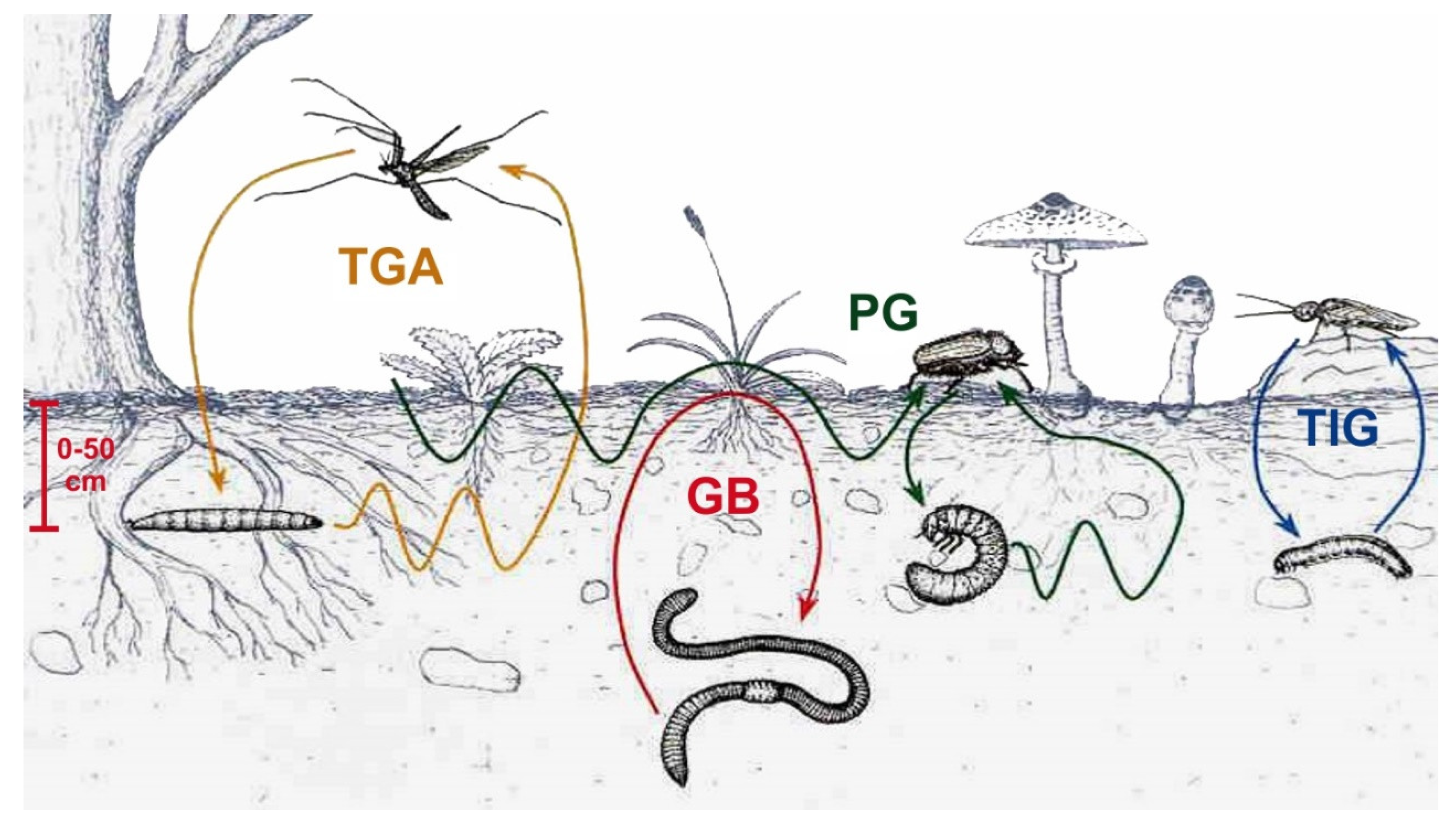
5. Soil Macrofauna in Orchard Agrosystems
5.1. General Aspects
5.2. Earthworms
5.3. Ants
5.4. Termites
5.5. Beetles
6. Cases of Negative Effects of Macrofauna in Orchard Soils
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wiesmeier, M.; Urbanski, L.; Hobley, E.; Lang, B.; von Lützow, M.; Marin-Spiotta, E.; van Wesemael, B.; Rabot, E.; Ließ, M.; García-Franco, N.; et al. Soil organic carbon storage as a key function of soils—A review of drivers and indicators at various scales. Geoderma 2019, 333, 149–162. [Google Scholar] [CrossRef]
- Filser, J.; Faber, J.H.; Tiunov, A.V.; Lijbert Brussaard, L.; Frouz, J.; De Deyn, G.; Uvarov, A.V.; Berg, M.P.; Lavelle, P.; Loreau, M.; et al. Soil fauna: Key to new carbon models. Soil 2016, 2, 565–682. [Google Scholar] [CrossRef]
- Pergola, M.; Favia, M.; Palese, A.M.; Perretti, B.; Xiloyannis, C.; Celano, G. Alternative management for olive orchards grown in semi-arid environments: An energy, economic and environmental analysis. Sci. Hortic. Amst. 2013, 162, 380–386. [Google Scholar] [CrossRef]
- Pascazio, S.; Crecchio, C.; Scagliola, M.; Mininni, A.N.; Dichio, B.; Xiloyannis, C.; Sofo, A. Microbial-based soil quality indicators in dry and wet soil portions of olive and peach Mediterranean orchards. Agric. Water Manag. 2018, 195, 172–179. [Google Scholar] [CrossRef]
- Palese, A.M.; Ringersma, J.; Baartman, J.E.M.; Peters, P.; Xiloyannis, C. Runoff and sediment yield of tilled and spontaneous grass-covered olive groves grown on sloping land. Soil Res. 2015, 53, 542–552. [Google Scholar] [CrossRef]
- Scheu, S.; Ruess, L.; Bonkowski, M. Interactions between microorganisms and soil micro- and mesofauna. In Microorganisms in Soils: Roles in Genesis and Functions; Buscot, F., Varma, A., Eds.; Springer: New York, NY, USA, 2005; pp. 253–278. [Google Scholar]
- Ruiz, N.; Lavelle, P.; Jiménez, J. Effect of land-use and management practices on soil macrofauna. In Soil Macrofauna Field Manual—Technical Level; FAO: Rome, Italy, 2008; pp. 29–36. [Google Scholar]
- Sofo, A.; Palese, A.M.; Casacchia, T.; Celano, G.; Ricciuti, P.; Curci, M.; Crecchio, C.; Xiloyannis, C. Genetic, functional, and metabolic responses of soil microbiota in a sustainable olive orchard. Soil Sci. 2010, 175, 81–88. [Google Scholar] [CrossRef]
- Sofo, A.; Ciarfaglia, A.; Scopa, A.; Camele, I.; Curci, M.; Crecchio, C.; Xiloyannis, C.; Palese, A.M. Soil microbial diversity and activity in a Mediterranean olive orchard using sustainable agricultural practices. Soil Use Manag. 2014, 30, 160–167. [Google Scholar] [CrossRef]
- Sofo, A.; Ricciuti, P.; Fausto, C.; Mininni, A.N.; Crecchio, C.; Scagliola, M.; Malerba, A.D.; Xiloyannis, C.; Dichio, B. The metabolic and genetic diversity of soil bacterial communities is affected by carbon and nitrogen dynamics: A qualitative and quantitative comparison of soils from an olive grove managed with sustainable or conventional approaches. Appl. Soil Ecol. 2019, 137, 21–28. [Google Scholar] [CrossRef]
- Sofo, A.; Tataranni, G.; Xiloyannis, C.; Dichio, B.; Scopa, A. Direct effects of Trichoderma harzianum strain T-22 on micropropagated GiSeLa6® (Prunus cerasus × Prunus canescens) rootstocks. Environ. Exp. Bot. 2012, 76, 33–38. [Google Scholar] [CrossRef]
- Barrios, E. Soil biota, ecosystem services and land productivity. Ecol. Econ. 2014, 64, 269–285. [Google Scholar] [CrossRef]
- FAO—Food and Agriculture Organization of the United Nations. Voluntary Guidelines for Sustainable Soil Management; FAO: Rome, Italy, 2017; Available online: http://www.fao.org/documents/card/en/c/5544358d-f11f-4e9f-90ef-a37c3bf52db7/ (accessed on 20 January 2020).
- Culliney, T.W. Role of arthropods in maintaining soil fertility. Agriculture 2013, 3, 629–659. [Google Scholar] [CrossRef]
- Matson, P.A.; Parton, W.J.; Power, A.G.; Swift, M.J. Agricultural intensification and ecosystem properties. Science 1997, 277, 504–509. [Google Scholar] [CrossRef] [PubMed]
- IPCC—Intergovernmental Panel on Climate Change. Climate Change and Land; IPCC: Geneva, Switzerland, 2019; Available online: https://www.ipcc.ch/srccl-report-download-page/ (accessed on 20 January 2020).
- USDA—United States Department of Agriculture. Natural Resources Conservation Service; USDA: Washington, DC, USA, 2020. Available online: https://www.nrcs.usda.gov/Internet/FSE_MEDIA/nrcs142p2_049823.jpg (accessed on 17 March 2020).
- Fausto, C.; Mininni, A.N.; Sofo, A.; Crecchio, C.; Scagliola, M.; Dichio, B.; Xiloyannis, C. Olive orchard microbiome: Characterisation of bacterial communities in soil-plant compartments and their comparison between sustainable and conventional managements. Plant Ecol. Div. 2018, 11, 597–610. [Google Scholar] [CrossRef]
- Celano, G.; Palese, A.M.; Ciucci, A.; Martorella, E.; Vignozzi, N.; Xiloyannis, C. Evaluation of soil water content in tilled and cover-cropped olive orchards by the geoelectrical technique. Geoderma 2011, 163, 163–170. [Google Scholar] [CrossRef]
- Totsche, K.U.; Amelung, W.; Gerzabek, M.H.; Guggenberger, G.; Klumpp, E.; Knief, C.; Lehndorff, E.; Mikutta, R.; Peth, S.; Prechtel, A.; et al. Microaggregates in soils. J. Plant Nutr. Soil Sci. 2018, 181, 104–136. [Google Scholar] [CrossRef]
- Mininni, A.N.; Dichio, B.; Pascazio, S.; Fausto, C.; Crecchio, C.; Sofo, A.; Xiloyannis, C. Restoration of soil fertility and management of mineral nutrition in a peach orchard under a sustainable farming system in semi-arid conditions. Acta Hortic. 2018, 1217, 257–262. [Google Scholar] [CrossRef]
- Silva, V.; Mol, H.G.J.; Zomer, P.; Tienstra, M.; Ritsema, C.J.; Geissen, V. Pesticide residues in European agricultural soils—A hidden reality. Sci. Total Environ. 2019, 653, 1532–1545. [Google Scholar] [CrossRef]
- Montanaro, G.; Dichio, B.; Briccoli Bati, C.; Xiloyannis, C. Soil management affects carbon dynamics and yield in a Mediterranean peach orchard. Agric. Ecosyst. Environ. 2012, 161, 46–54. [Google Scholar] [CrossRef]
- FAO—Food and Agriculture Organization of the United Nations. The Status of the World’s Soil Resources; FAO: Rome, Italy, 2015; Available online: http://www.fao.org/documents/card/en/c/c6814873-efc3-41db-b7d3-2081a10ede50/ (accessed on 20 January 2020).
- FAO—Food and Agriculture Organization of the United Nations. The 10 Elements of Agroecology: Guiding the Transition to Sustainable Food and Agricultural Systems; FAO: Rome, Italy, 2018; Available online: http://www.fao.org/documents/card/en/c/I9037EN/ (accessed on 20 January 2020).
- Fourati, R.; Scopa, A.; Ben Ahmed, C.; Ben Abdallah, F.; Terzano, R.; Gattullo, C.E.; Allegretta, I.; Galgano, F.; Caruso, M.C.; Sofo, A. Biochemical responses and oil quality parameters in olives plants and fruits exposed to airborne metal pollution. Chemosphere 2017, 168, 514–522. [Google Scholar] [CrossRef]
- Negassa, W.; Sileshi, G.W. Integrated soil fertility management reduces termite damage to crops on degraded soils in western Ethiopia. Agric. Ecosyst. Environ. 2018, 251, 124–131. [Google Scholar] [CrossRef]
- Zimmer, M.; Kautz, G.; Topp, W. Do woodlice and earthworms interact synergistically in leaf litter decomposition? Funct. Ecol. 2005, 19, 7–16. [Google Scholar] [CrossRef]
- Coleman, D.C.; Wall, D.H. Fauna: The engine for microbial activity and transport. In Soil Microbiology, Ecology, and Biochemistry, 3rd ed.; Eldor, A.P., Ed.; Elsevier: Oxford, UK, 2007; pp. 163–194. [Google Scholar]
- Bardgett, R.D.; van der Putten, W.H. Belowground biodiversity and ecosystem functioning. Nature 2014, 525, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Wall, D.H.; Bardgett, R.D.; Kelly, E.F. Biodiversity in the dark. Nat. Geosci. 2010, 3, 297–298. [Google Scholar] [CrossRef]
- Fierer, N. Embracing the unknown: Disentangling the complexities of the soil microbiome. Nat. Rev. Microbiol. 2017, 15, 579–590. [Google Scholar] [CrossRef]
- Semhi, K.; Chaudhuri, S.; Clauer, N.; Boeglin, J.L. Impact of termite activity on soil environment: A perspective from their soluble chemical components. Int. J. Environ. Sci. Technol. 2008, 5, 431–444. [Google Scholar] [CrossRef]
- Wall, D.H.; Nielsen, U.N.; Six, J. Soil biodiversity and human health. Nature 2015, 528, 69–76. [Google Scholar] [CrossRef]
- Williamson, K.E.; Fuhrmann, J.J.; Wommack, K.E.; Radosevich, M. Viruses in soil ecosystems: An unknown quantity within an unknown unexplored territory. Ann. Rev. Virol. 2017, 4, 201–219. [Google Scholar] [CrossRef]
- Orgiazzi, A.; Bardgett, R.; Barrios, E.; Behan-Pelletier, V.; Briones, M.J.I.; Chotte, J.-L.; De Deyn, G.; Eggleton, P.; Fierer, N.; Fraser, T.; et al. Global Soil Biodiversity Atlas; Publications Office of the European Union: Luxembourg, 2016; Available online: https://esdac.jrc.ec.europa.eu/content/global-soil-biodiversity-atlas (accessed on 15 December 2019).
- Six, J.; Bossuyt, H.; Degryze, S.; Denef, K. A history of the link between (micro) aggregates, soil biota and soil organic matter dynamics. Soil Till. Res. 2004, 79, 7–31. [Google Scholar] [CrossRef]
- EASAC—European Academies’ Science Advisory Council. Opportunities for Soil Sustainability in Europe; EASAC Policy Report 36; EASAC: Halle, Germany, 2018; Available online: http://www.easac.eu (accessed on 20 January 2020).
- Ramirez, K.S.; Doring, M.; Eisenhauer, N.; Gardi, C.; Ladau, J.; Leff, J.W.; Lentendu, G.; Lindo, Z.; Rillig, M.C.; Russell, D.; et al. Toward a global platform for linking soil biodiversity data. Front. Ecol. Evol. 2015, 3, 91. [Google Scholar] [CrossRef]
- Tomati, U.; Galli, E. Earthworms, soil fertility and plant productivity. Acta Zool. Fenn. 1995, 186, 11–14. [Google Scholar]
- Li, Y.; Dong, Z.Y.; Pan, D.Z.; Pan, C.H.; Chen, L.H. Effect of termite on soil pH and its application for termite control in Zhejiang province, China. Sociobiology 2017, 64, 317–326. [Google Scholar] [CrossRef]
- Orhue Ehi, R.; Uzu Ogbonnaya, F.; Osaigbovo Ulamen, A. Influence of activities of termites on some physical and chemical properties of soils under different land use patterns: A review. Int. J. Soil Sci. 2007, 2, 1–14. [Google Scholar]
- Farji-Brener, A.G.; Tadey, M. Contributions of leaf-cutting ants to soil fertility; causes and consequences. In Soil Fertility; Lucero, D.P., Boggs, J.E., Eds.; Nova Science Publishers Inc.: Hauppauge, NY, USA, 2009; pp. 81–91. [Google Scholar]
- Farji-Brener, A.G.; Werenkraut, V. The effects of ant nests on soil fertility and plant performance: A meta-analysis. J. Anim. Ecol. 2017, 86, 866–877. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.C.; de Ruiter, P.C. Soil food webs in agricultural ecosystems. In Microbial Ecology in Sustainable Agroecosystems; Cheeke, T.E., Coleman, D.C., Wall, D.H., Eds.; CRC Press: Boca Raton, FL, USA, 2005; pp. 63–88. [Google Scholar]
- Paoletti, M.G.; Sommaggio, D.; Favretto, M.R.; Petruzzelli, G.; Pezzarossa, B.; Barbafieri, M. Earthworms as useful bioindicators of agroecosystem sustainability in orchards and vineyards with different inputs. Appl. Soil Ecol. 1998, 10, 137–150. [Google Scholar] [CrossRef]
- Paoletti, M.G.; D’Incà, A.; Tonin, E.; Tonon, S.; Migliorni, C.; Petruzzelli, G.; Pezzarossa, B.; Gomiero, T.; Sommaggio, D. Soil invertebrates as bio-indicators in a natural area converted from agricultural use: The case study of Vallevecchia-Lugugnana in North-Eastern Italy. J. Sustain. Agric. 2010, 34, 38–56. [Google Scholar] [CrossRef]
- FAO—Food and Agriculture Organization of the United Nations. Integrated Soil and Water Management for Orchard Development. Role and Importance; FAO: Rome, Italy, 2005. Available online: http://www.fao.org/3/a0007e/a0007e00.htm (accessed on 25 March 2020).
- Srivastava, A.K.; Malhotra, S.K. Nutrient use efficiency in perennial fruit crops—A review. J. Plant Nutr. 2017, 40, 1928–1953. [Google Scholar] [CrossRef]
- Hinsinger, P.; Bengough, A.G.; Vetterlein, D.; Young, I. Rhizosphere: Biophysics, biogeochemistry and ecological relevance. Plant Soil 2009, 321, 117–152. [Google Scholar] [CrossRef]
- Wiesmeier, M.; Munro, S.; Barthold, F.; Steffens, M.; Schad, P.; Kögel-Knabner, I. Carbon storage capacity of semi-arid grassland soils and sequestration potentials in Northern China. Glob. Chang. Biol. 2015, 21, 3836–3845. [Google Scholar] [CrossRef]
- Yadav, R.S.; Singh, J. Biodiversity of soil arthropods under mango, Mangifera indica L. orchard ecosystem in Varanasi, Uttar Pradesh. Environ. Ecol. 2009, 27, 1228–1230. [Google Scholar]
- Lavelle, P.; Spain, A.; Blouin, M.; Brown, G.; Decaëns, T.; Grimaldi, M.; Jiménez, J.J.; McKey, D.; Mathieu, J.; Velasquez, E.; et al. Ecosystem engineers in a self-organized soil: A review of concepts and future research questions. Soil Sci. 2016, 181, 91–109. [Google Scholar] [CrossRef]
- Hooper, D.U.; Chapin, F.S., III; Ewel, J.J.; Hector, A.; Inchausti, P.; Lavorel, S.; Lawton, J.H.; Lodge, D.M.; Loreau, M.; Naeem, S.; et al. Effects of biodiversity on ecosystem functioning: A consensus of current knowledge. Ecol. Monogr. 2005, 75, 3–35. [Google Scholar] [CrossRef]
- Wagg, C.; Bender, S.F.; Widmer, F.; van der Heijden, M.G.A. Soil biodiversity and soil community composition determine ecosystem multifunctionality. Proc. Natl. Acad. Sci. USA 2014, 111, 5266–5270. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.G.; Lawton, J.H.; Shachak, M. Organisms as ecosystem engineers. Oikos 1994, 69, 373–386. [Google Scholar] [CrossRef]
- Giller, K.E.; Beare, M.H.; Lavelle, P.; Izac, A.M.; Swift, M.J. Agricultural intensification, soil biodiversity and agroecosystem function. Appl. Soil Ecol. 1997, 6, 3–16. [Google Scholar] [CrossRef]
- Walmsley, A.; Cerdà, A. Soil macrofauna and organic matter in irrigated orchards under Mediterranean climate. Biol. Agric. Hortic. 2017, 33, 247–257. [Google Scholar] [CrossRef]
- Doles, J.L.; Zimmerman, R.J.; Moore, J.C. Soil microarthropod community structure and dynamics in organic and conventionally managed apple orchards in Western Colorado, USA. Appl. Soil Ecol. 2001, 18, 83–96. [Google Scholar] [CrossRef]
- Jerez-Valle, C.; García, P.A.; Campos, M.; Pasqual, F. A simple bioindication method to discriminate olive orchard management types using the soil arthropod fauna. Appl. Soil Ecol. 2014, 76, 42–51. [Google Scholar] [CrossRef]
- Buchholz, J.; Querner, P.; Paredes, D.; Bauer, T.; Strauss, P.; Guernion, M.; Scimia, J.; Cluzeau, D.; Burel, F.; Kratschmer, S.; et al. Soil biota in vineyards are more influenced by plants and soil quality than by tillage intensity or the surrounding landscape. Sci. Rep. 2017, 7, 17445. [Google Scholar] [CrossRef]
- Castro Lopez, J.; Barreal, M.E.; Briones, M.J.I.; Gallego, P.P. Earthworm communities in conventional and organic fruit orchards under two different climates. Appl. Soil Ecol. 2019, 144, 83–91. [Google Scholar] [CrossRef]
- Van Zwieten, L.; Rust, J.; Kingston, T.; Merrington, G.; Morris, S. Influence of copper fungicide residues on occurrence of earthworms in avocado orchard soils. Sci. Total Environ. 2004, 329, 29–41. [Google Scholar] [CrossRef]
- Jongmans, A.G.; Pulleman, M.M.; Balabane, M.; van Oort, F.; Marinissen, J.C.Y. Soil structure and characteristics of organic matter in two orchards differing in earthworm activity. Appl. Soil Ecol. 2003, 24, 219–232. [Google Scholar] [CrossRef]
- Paoletti, M.G. The role of earthworms for assessment of sustainability and as bioindicators. Agric. Ecosyst. Environ. 1999, 74, 137–155. [Google Scholar] [CrossRef]
- Paoletti, M.G.; Schweigl, U.; Favretto, M.R. Soil microinvertebrates, heavy metals and organochlorines in low and high input apple orchards and coppiced woodland. Pedobiologia 1995, 39, 20–33. [Google Scholar]
- Paoletti, M.G. Soil invertebrates in cultivated and uncultivated soils in northest Italy. Redia 1985, 71, 501–563. [Google Scholar]
- Paoletti, M.G.; Iovane, E.; Cortese, M. Pedofauna bioindicators and heavy metals in five agroecosystems in north-east Italy. Rev. Ecol. Biol. Sol. 1988, 25, 33–58. [Google Scholar]
- Curry, J.P. Grassland Invertebrates; Chapman & Hall: London, UK, 1994; pp. 320–354. [Google Scholar]
- Kreis, B.; Edwards, P.; Cuendet, G.; Tarrandellas, J. The dynamics of RCBs between earthworm populations and agricultural soils. Pedobiologia 1987, 30, 379–388. [Google Scholar]
- Reinecke, S.A.; Reinecke, A.J. The impact of organophosphate pesticides in orchards on earthworms in the Western Cape, South Africa. Ecotox. Environ. Saf. 2007, 66, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Haynes, R.J. Effects of soil management practices on soil physical properties, earthworm population and tree root distribution in a commercial apple orchard. Soil Till. Res. 1980, 1, 269–280. [Google Scholar] [CrossRef]
- Pizl, V. Effect of soil compaction on earthworms (Lumbricidae) in apple orchard soil. Soil Biol. Biochem. 1992, 24, 1573–1575. [Google Scholar] [CrossRef]
- Iglesias Briones, M.J.; Barreal, M.E.; Crespo Harrison, A.; Gallego, P.P. Earthworms and nitrogen applications to improve soil health in an intensively cultivated kiwifruit orchard. Appl. Soil Ecol. 2011, 49, 158–166. [Google Scholar] [CrossRef]
- Lardo, E.; Coll, P.; Le Cadre, E.; Palese, A.M.; Villenave, C.; Xiloyannis, C.; Celano, G. Electromagnetic induction (EMI) measurements as a proxy of earthworm presence in Southern French vineyards. Appl. Soil Ecol. 2012, 61, 76–84. [Google Scholar] [CrossRef]
- Goh, K.M.; Pearson, D.R.; Daly, M.J. Effects of apple orchard production systems on some important soil physical, chemical and biological quality parameters. Biol. Agric. Hortic. 2012, 18, 269–292. [Google Scholar] [CrossRef]
- Lardo, E.; Palese, A.M.; Arouss, A.; Ferrazzano, G.; Xiloyannis, C.; Celano, G. Apparent electrical conductivity as a tool for earthworm parameters evaluation in a commercial orchard. Acta Hortic. 2015, 61, 76–84. [Google Scholar] [CrossRef]
- Keller, L.; Gordon, E. La Vie des Fourmis; Odile Jacob: Paris, France, 2006; pp. 152–160. [Google Scholar]
- Diamé, L.; Rey, J.R.; Vayssières, J.F.; Grechi, I.; Chailleux, A.; Diarra, K. Ants: Major functional elements in fruit agro-ecosystems and biological control agents. Sustainability 2017, 10, 23. [Google Scholar] [CrossRef]
- Warren, R.J., II; Bradford, M.A. Ant colonization and coarse woody debris decomposition in temperate forests. Insectes Sociaux 2011, 59, 215–221. [Google Scholar] [CrossRef]
- Cerdà, A.; Jurgensen, M.F. Ant mounds as a source of sediment on citrus orchard plantations in eastern Spain. A three-scale rainfall simulation approach. Catena 2011, 85, 231–236. [Google Scholar] [CrossRef]
- Cerdà, A.; Jurgensen, M.F.; Bodí, M.B. Effects of ants on water and soil losses from organically-managed citrus orchards in Eastern Spain. Biologia 2009, 64, 527–531. [Google Scholar] [CrossRef]
- Stansly, P.A.; Su, N.-Y.; Conner, J.M. Management of subterranean termites, Reticulitermes spp. (Isoptera: Rhinotermitidae) in a citrus orchard with hexaflumuron bait. Crop Prot. 2001, 20, 199–206. [Google Scholar] [CrossRef]
- Coulibaly, T.; Moise Akpesse, A.A.; Boga, J.-P.; Yapi, A.; Kouassi, K.P.; Roisin, Y. Change in termite communities along a chronosequence of mango tree orchards in the north of Côte d’Ivoire. J. Insect Conserv. 2016, 20, 1011–1019. [Google Scholar] [CrossRef]
- Hunt, T.; Bergsten, J.; Levkanicova, Z.; Papadopoulou, A.; John, O.S.; Wild, R.; Hammond, P.M.; Ahrens, D.; Balke, M.; Caterino, M.S.; et al. A comprehensive phylogeny of beetles reveals the evolutionary origins of a superradiation. Science 2007, 318, 1913–1916. [Google Scholar] [CrossRef]
- Marshall, C.B.; Lynch, D.H. Soil microbial and macrofauna dynamics under different green manure termination methods. Appl. Soil Ecol. 2020, 148, 103505. [Google Scholar] [CrossRef]
- Ngosong, C.; Okolle, J.N.; Tening, A.S. Mulching: A sustainable option to improve soil health. In Soil Fertility Management for Sustainable Development; Panpatte, D., Jhala, Y., Eds.; Springer: Singapore, 2019; pp. 231–249. [Google Scholar]
- Epstein, D.L.; Zack, R.S.; Brunner, J.F.; Gut, L.; Brown, J.J. Ground beetle activity in apple orchards under reduced pesticide management regimes. Biol. Control 2001, 21, 97–104. [Google Scholar] [CrossRef]
- Vignozzi, V.; Agnelli, A.E.; Brandi, G.; Cagnarli, E.; Goggioli, D.; Lagomarsino, A.; Pellegrino, S.; Simoncini, S.; Simoni, S.; Velboa, G.; et al. Soil ecosystem functions in a high-density olive orchard managed by different soil conservation practices. Appl. Soil Ecol. 2019, 134, 64–76. [Google Scholar] [CrossRef]
- Balog, A.; Markó, V.; Imre, A. Farming system and habitat structure effects on rove beetles (Coleoptera: Staphylinidae) assembly in Central European apple and pear orchards. Biologia 2009, 64, 343–349. [Google Scholar] [CrossRef]
- Hedde, M.; Mazzia, C.; Decaëns, T.; Nahmani, J.; Pey, B.; Thénard, J.; Capowiez, Y. Orchard management influences both functional and taxonomic ground beetle (Coleoptera, Carabidae) diversity in South-East France. Appl. Soil Ecol. 2015, 88, 26–31. [Google Scholar]
- Cotes, B.; Castro, J.; Cárdenas, M.; Campos, M. Responses of epigeal beetles to the removal of weed cover crops in organic olive orchards. Bull. Insectol. 2009, 62, 47–52. [Google Scholar]
- Kromp, B. Carabid beetles in sustainable agriculture: A review on pest control efficacy, cultivation impacts and enhancement. Agric. Ecosyst. Environ. 1999, 74, 187–228. [Google Scholar] [CrossRef]
- Miñarro, M.; Dapena, E. Effects of groundcover management on ground beetles (Coleoptera: Carabidae) in an apple orchard. Appl. Soil Ecol. 2003, 23, 111–117. [Google Scholar] [CrossRef]
- Balog, A.; Markó, V. Rove beetles (Coleoptera: Staphylinidae) in Central European apple and pear orchards—Comparative studies of species richness, abundance and diversity. J. Plant Prot. Res. 2007, 47, 309–320. [Google Scholar]
- Honěk, A.; Kocian, M.; Martinová, Z. Rove beetles (Coleoptera: Staphylinidae) in an apple orchard. Plant Prot. Sci. 2012, 48, 116–122. [Google Scholar] [CrossRef]
- Nietupski, M.; Kosewska, A.; Markuszewski, B.; Sądej, W. Soil management system in hazelnut groves (Corylus sp.) versus the presence of ground beetles (Coleoptera: Carabidae). J. Plant Prot. Res. 2015, 55, 26–34. [Google Scholar] [CrossRef]
- David, J.-F. The role of litter-feeding macroarthropods in decomposition processes: A reappraisal of common views. Soil Biol. Biochem. 2014, 76, 109–118. [Google Scholar] [CrossRef]
- Lubbers, I.M.; van Groenigen, K.J.; Brussaard, L.; van Groenigen, J.W. Reduced greenhouse gas mitigation potential of no-tillage soils through earthworm activity. Sci. Rep. 2015, 5, 13787. [Google Scholar] [CrossRef] [PubMed]
- Lubbers, I.M.; van Groenigen, K.J.; Fonte, S.J.; Six, J.; Brussaard, L.; van Groenigen, J.W. Greenhouse-gas emissions from soils increased by earthworms. Nat. Clim. Chang. 2013, 3, 187–194. [Google Scholar] [CrossRef]
- Cross, J.; Fountain, M.; Marko, V.; Nagy, C. Arthropod ecosystem services in apple orchards and their economic benefits. Ecol. Entomol. 2015, 40, 82–96. [Google Scholar] [CrossRef]
- Mody, K.; Spoerndli, C.; Dorn, S. Within-orchard variability of the ecosystem service ‘parasitism’: Effects of cultivars, ants and tree location. Basic Appl. Ecol. 2011, 12, 456–465. [Google Scholar] [CrossRef]
- Jouquet, P.; Bottinelli, N.; Shanbhag, R.R.; Bourguignon, T.; Traoré, S.; Abbasi, S.A. Termites: The neglected soil engineers of tropical soils. Soil Sci. 2016, 181, 157–165. [Google Scholar] [CrossRef]
- Rogers, D.J.; Cole, L.M.; Delate, K.M.; Walker, J.T.S. Managing bronze beetle, Eucolaspis Brunnea, in organic apple orchards. N. Z. Plant Prot. 2006, 59, 57–62. [Google Scholar] [CrossRef]
- Rogers, D.J.; Cole, L.M.; Fraser, T.M.; Walker, J.T.S. Managing bronze beetle (Eucolaspsis sp.) in hawke’s bay organic apple orchards using soil cultivation. N. Z. Plant Prot. 2009, 62, 285–290. [Google Scholar]
- Xiloyannis, C.; Montanaro, G.; Mininni, A.N.; Dichio, B. Sustainable production systems in fruit tree orchards. Acta Hortic. ISHS 2005, 1099, 319–324. [Google Scholar] [CrossRef]
| Authors | Year | Fruit Orchard | Agricultural Management | Results | Reference |
|---|---|---|---|---|---|
| Haynes | 1980 1981 | Apple | Retention and removal of grass (either by cultivation or by use of herbicides) |
| [72] |
| Pizl | 1992 | Apple | Intensively managed soils (trafficked and untrafficked plots) |
| [73] |
| Iglesias Briones et al. | 2011 | Kiwifruit | Conventional practices with the introduction of anecic earthworm species in combination with nitrogen applications, such as cow manure or planting N-fixing legume | Only manure applications promoted:
| [74] |
| Lardo et al. | 2012 | Vineyards | Grass cover, chemical weeding or tillage | Lowest values of earthworm density and biomass, and selective action towards earthworm categories in tillage treatments | [75] |
| Goh et al. | 2012 | Apple |
| In both cases (a,b), marked increases in the total number of epigeic earthworm species, fresh biomass and species composition occurred in organic systems, related to high SOM input through grass covering and to the absence of biocides | [76] |
| Lardo et al. | 2015 | Peach | Commercial orchard with a clay loam managed by fertigation | Abundance and biomass of endogeic and anecic earthworms were significantly higher in inter-rows covered by permanent spontaneous grass cover (no-tillage, no mineral fertilization) than in rows weeded with glyphosate | [77] |
| Authors | Year | Fruit Orchard | Agricultural Management | Results | Reference |
|---|---|---|---|---|---|
| Cerdà et al. | 2009 | Citrus | Organic orchards |
| [82] |
| Farji-Brener and Tadey | 2009 | Ecosystem soil | Organically-managed soils | The building, enlargement, and maintenance of nests ants affected soil structure, porosity and density | [43] |
| Cerdà and Jurgensen | 2011 | Citrus | Intensively-managed orchard |
| [81] |
| Farji-Brener and Werenkraut | 2017 | Ecosystem soil | Organically- managed soils |
| [44] |
| Authors | Year | Fruit Orchard | Agricultural Management | Results | Reference |
|---|---|---|---|---|---|
| Stansly et al. | 2001 | Citrus | Baiting with hexaflumuron bait |
| [83] |
| Coulibaly et al. | 2016 | Mango | Chronosequence of tree orchards (from young to old) with higher management in young compared to old (tillage; weeding; application of pesticides) |
| [84] |
| Authors | Year | Fruit Orchard | Agricultural Management | Results | Reference |
|---|---|---|---|---|---|
| Epstein et al. | 2001 | Apple | Conventionally managed orchards vs. no neural-active insecticides addition | Orchard blocks under conventionally managed regimes had significantly lower populations of ground beetles compared with orchard blocks managed without broad-spectrum insecticides | [88] |
| Miñarro and Dapena | 2003 | Cider-apple orchard | Six different groundcover management systems | The groundcover management affected the activity, density and diversity of ground beetles that contribute to the natural control of pests | [94] |
| Balog and Markó | 2007 | Apple, pear | Conventionally treated orchards and abandoned orchards |
| [95] |
| Balog et al. | 2009 | Apple, pear | Different environmental conditions (agricultural lowland environment; flooded areas; woodland areas of medium height mountains) | The cumulative effects of environmental conditions and soil modified the activity and density of Staphylinidae communities | [90] |
| Cotes et al. | 2009 | Olive | Organically and conventionally-managed soil |
| [92] |
| Honěk et al. | 2012 | Apple | Organic vs. conventional apple orchard | Positive role of rove beetles in the biological control of agricultural arthropod pests | [96] |
| Hedde et al. | 2015 | Apple | Conventionally-managed orchards vs. lower use of synthetic chemical compounds | The activity-density of ground beetle communities was solely influenced by season and species richness and by orchard management | [91] |
| Nietupski et al. | 2015 | Hazelnut | Different soil cultivation methods | Optimal soil tillage system promoted the presence of beetles in soil kept fallow with machines or chemicals, and in soil covered with manure | [97] |
| Marshall and Lynch | 2020 | Apple | No-till green cover vs. tilled soil | The density of some species of beetle (Harpalus spp.) increased with green cover | [86] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sofo, A.; Mininni, A.N.; Ricciuti, P. Soil Macrofauna: A key Factor for Increasing Soil Fertility and Promoting Sustainable Soil Use in Fruit Orchard Agrosystems. Agronomy 2020, 10, 456. https://doi.org/10.3390/agronomy10040456
Sofo A, Mininni AN, Ricciuti P. Soil Macrofauna: A key Factor for Increasing Soil Fertility and Promoting Sustainable Soil Use in Fruit Orchard Agrosystems. Agronomy. 2020; 10(4):456. https://doi.org/10.3390/agronomy10040456
Chicago/Turabian StyleSofo, Adriano, Alba Nicoletta Mininni, and Patrizia Ricciuti. 2020. "Soil Macrofauna: A key Factor for Increasing Soil Fertility and Promoting Sustainable Soil Use in Fruit Orchard Agrosystems" Agronomy 10, no. 4: 456. https://doi.org/10.3390/agronomy10040456
APA StyleSofo, A., Mininni, A. N., & Ricciuti, P. (2020). Soil Macrofauna: A key Factor for Increasing Soil Fertility and Promoting Sustainable Soil Use in Fruit Orchard Agrosystems. Agronomy, 10(4), 456. https://doi.org/10.3390/agronomy10040456







