Evaluation of Fungicides and Management Strategies against Cercospora Leaf Spot of Olive Caused by Pseudocercospora cladosporioides
Abstract
1. Introduction
2. Materials and Methods
2.1. Fungicides
2.2. In Vitro Evaluation of Fungicides
2.2.1. Inoculum Sources
2.2.2. Effect of Fungicides on Mycelial Growth
2.2.3. Effect of Fungicides on Conidial Germination
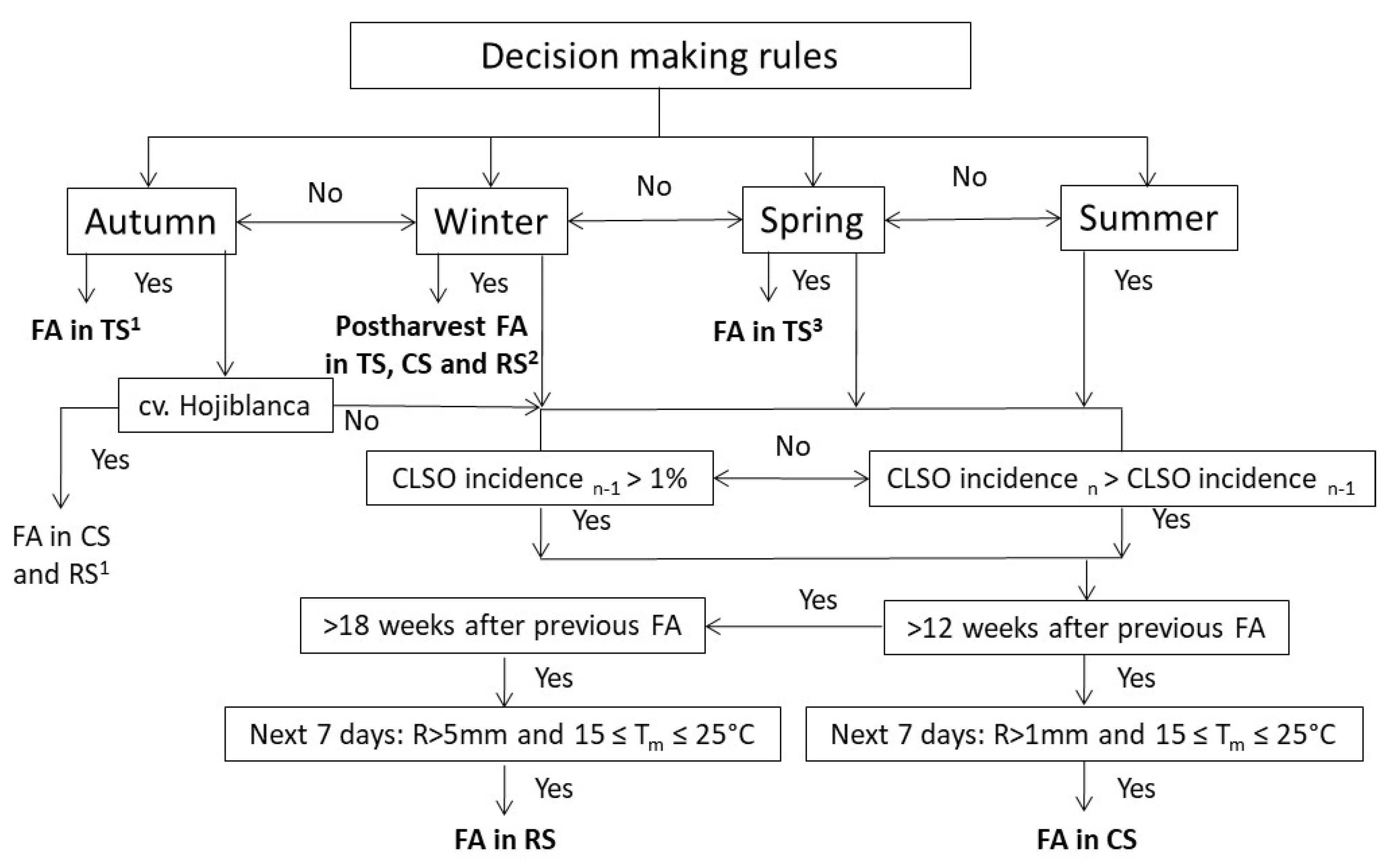
2.3. Effect of Fungicides and Annual Timing on Disease Incidence in a Naturally Infected Orchard
2.4. Comparative Effectiveness of the Three Management Strategies for Disease Control
2.4.1. Management Strategies
2.4.2. Experimental Design
2.5. Data Analyses
3. Results
3.1. In Vitro Evaluation of Fungicides
3.1.1. Effect of Fungicides on Mycelial Growth
3.1.2. Effect of Fungicides on Conidial Germination
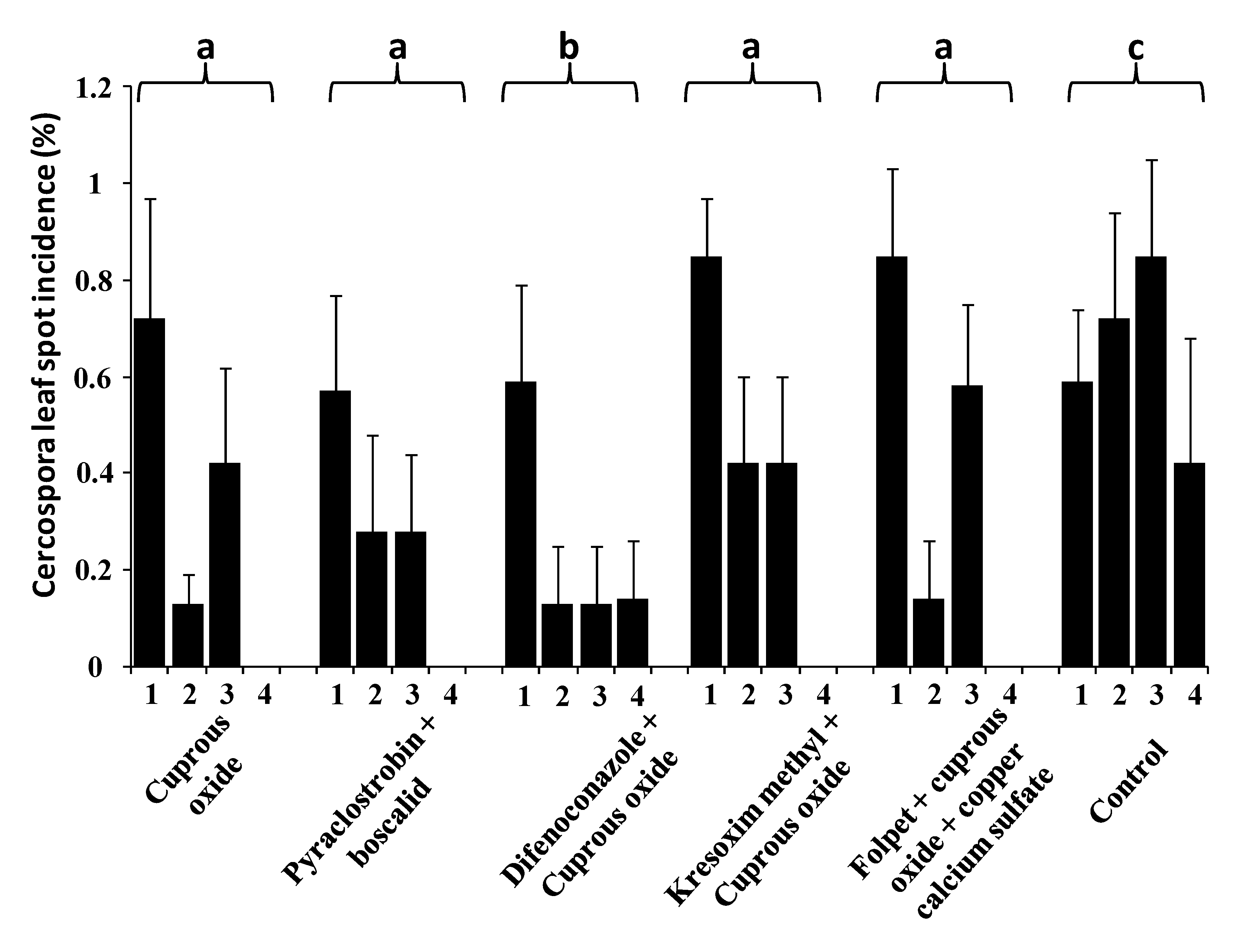
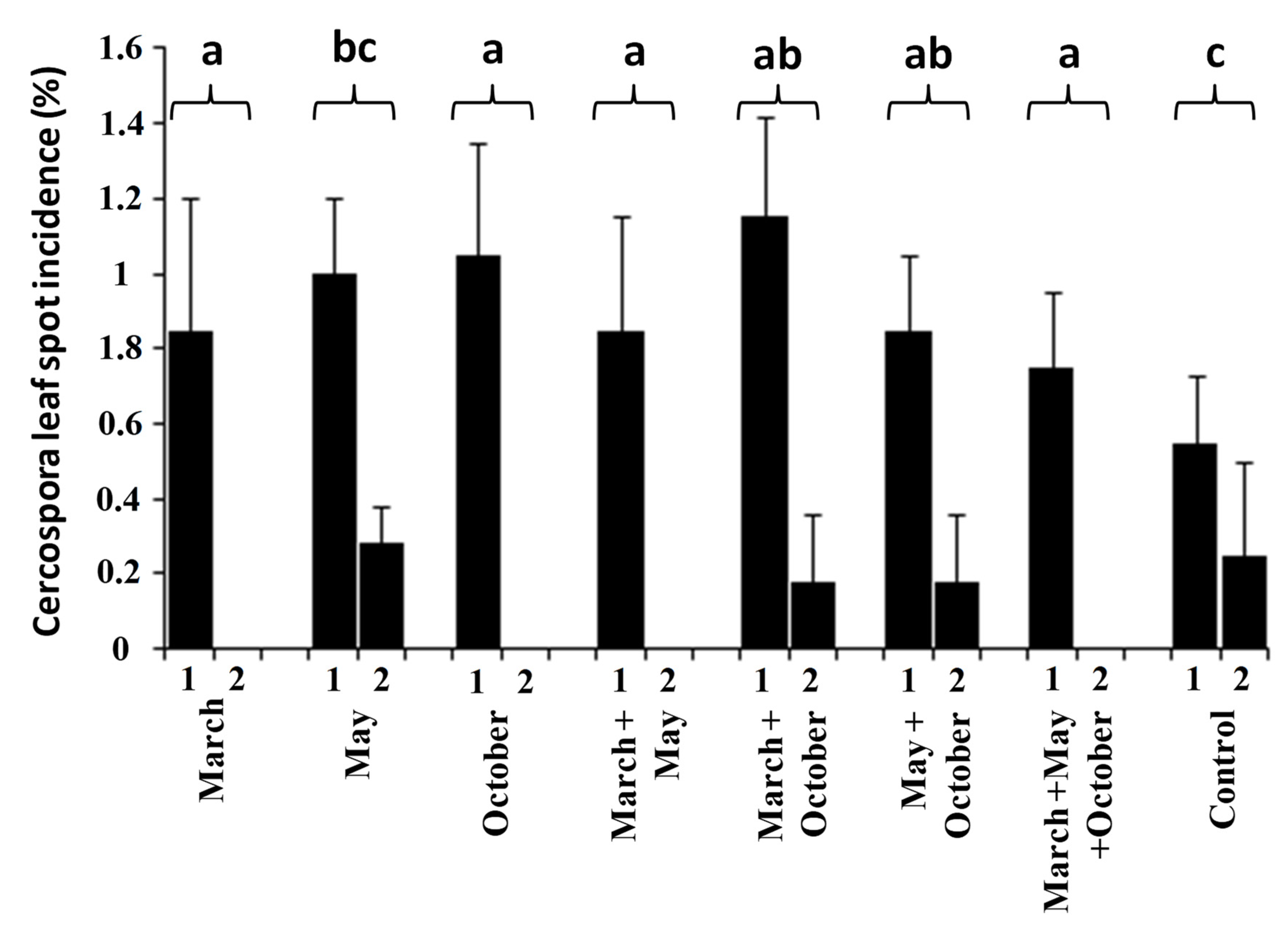
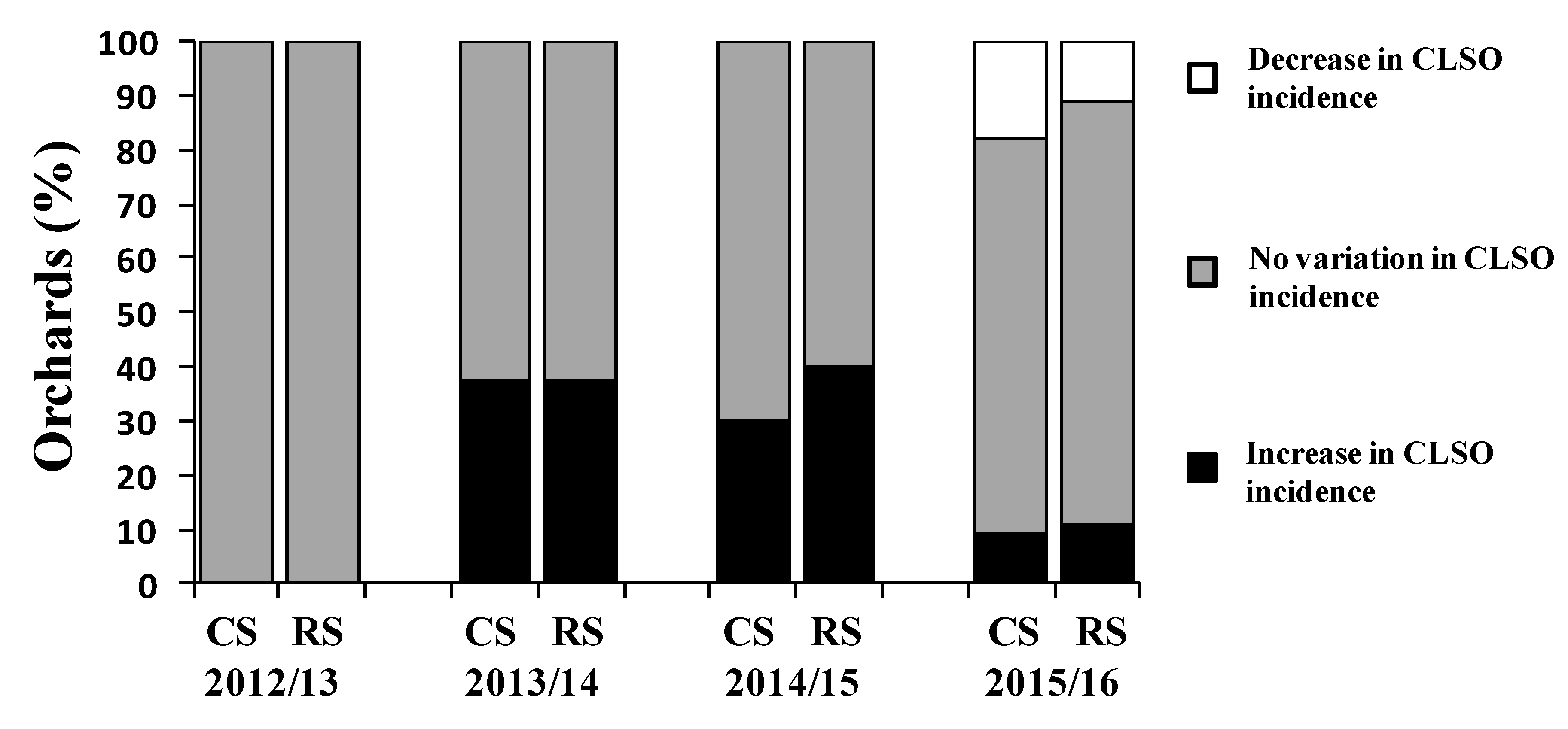
3.2. Effect of Fungicides and Application Times in a Naturally Infected Orchard
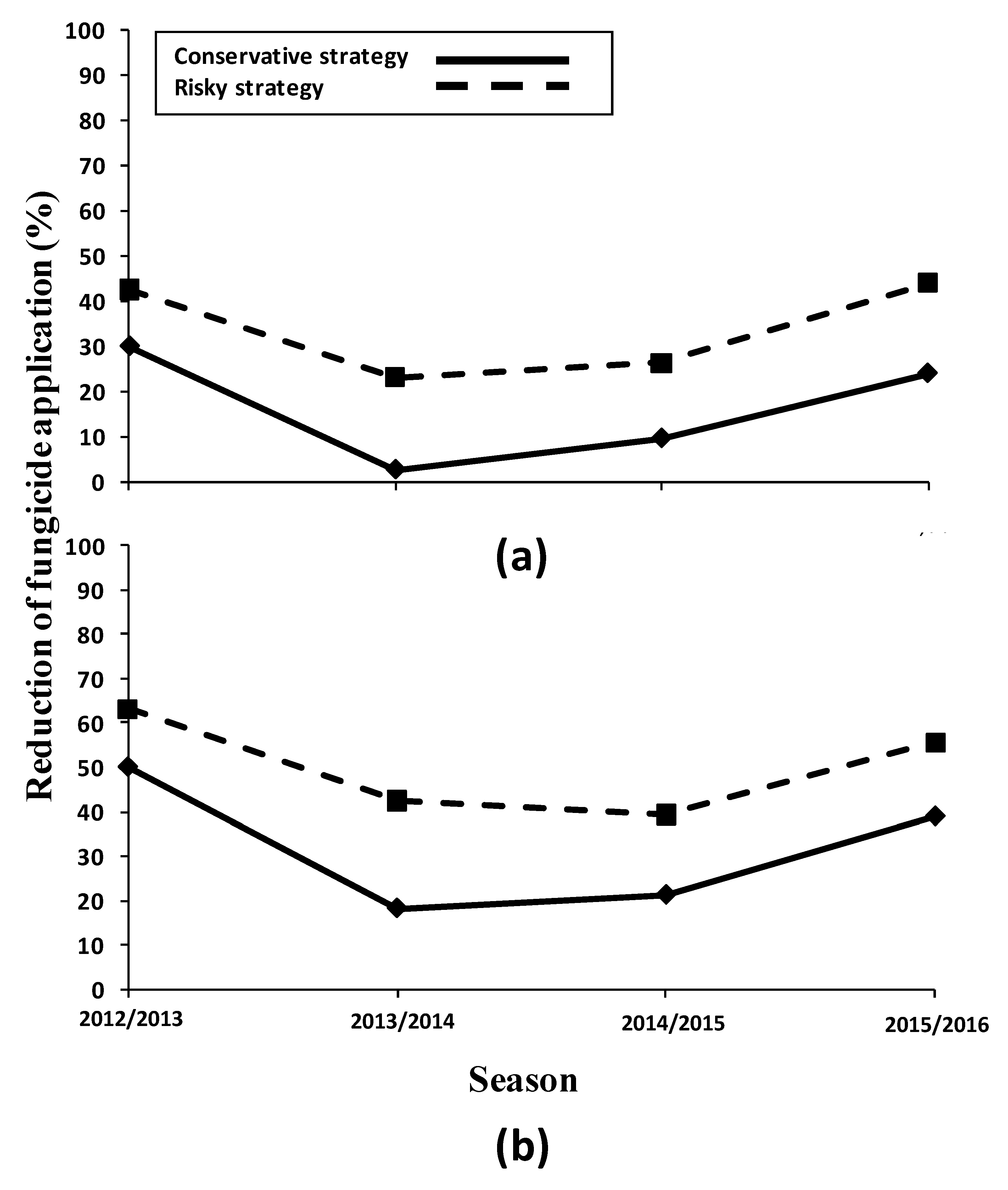
3.3. Effectiveness of Management Strategies against P. cladosporioides
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Trapero, A.; López-Escudero, F.J.; Blanco, M.A. Enfermedades. In El Cultivo del Olivo, 7th ed.; Mundi-Prensa: Madrid, Spain, 2015; pp. 733–798. ISBN 9788484767145. [Google Scholar]
- Ministerio de Agricultura. Pesca y Alimentación (MAPA). 2016. Available online: http://www.magrama.gob.es/es/agricultura/temas/producciones-agricolas/aceite-oliva-y-aceituna-mesa/aceituna.aspx#para2 (accessed on 9 September 2019).
- Del Moral, J.; Medina, D. El “repilo plomizo” del olivo causado por Cercospora cladosporioides Sacc., enfermedad presente en España. Bol. San. Veg. Plagas 1985, 12, 31–35. [Google Scholar]
- Ávila, A.; Groenewald, J.; Trapero, A.; Crous, P. Characterisation and epitypification of Pseudocercospora cladosporioides, the causal organism of Cercospora leaf spot of olives. Mycol. Res. 2005, 109, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Ávila, A.; Romero, J.; Agustí-Brisach, C.; Roca, L.F.; Benali, A.; Trapero, A. Phenotypic and pathogenic characterization of Pseudocercospora cladosporioides, causal agent of cercospora leaf spot of olives. Eur. J. Plant Pathol. 2019, 156, 45–65. [Google Scholar] [CrossRef]
- González-Fragoso, R. Botánica Criptogámica Agrícola, 1st ed.; Espasa-Calpe: Madrid, Spain, 1927; p. 321. [Google Scholar]
- García-Figueres, F. Repilos del olivo: Ataque en fruto. Phytoma Esp. 1991, 25, 31–36. [Google Scholar]
- Pappas, A.C. Mycocentrospora cladosporioides on olive in Greece. EPPO Bull. 1993, 23, 405–409. [Google Scholar] [CrossRef]
- Hansen, H.N.; Rawlins, T.E. Cercospora fruit and leaf spot of olive. Phytopathologia 1944, 34, 257–259. [Google Scholar]
- Bottalico, A.; Corda, P. Mycocentrospora cladosporioides from olive drupes in Sardinia. Plant Dis. 1995, 79, 320. [Google Scholar] [CrossRef]
- Sergeeva, V.; Braun, U.; Spooner-Hart, R.; Nair, N. First report of Pseudocercospora cladosporioides on olive (Olea europaea) berries in Australia. Australas. Plant Dis. Notes 2008, 3, 24. [Google Scholar] [CrossRef]
- Saccardo, P.A. Sylloge fungorum. Sylloge Fungorum 1886, 4, 1–87. [Google Scholar]
- Farr, D.F.; Rossman, A.Y. Fungal Databases, U.S. National Fungus Collections, ARS, USDA. Available online: https://nt.ars-grin.gov/fungaldatabases/ (accessed on 2 May 2019).
- Viruega, J.R.; Moral, J.; Roca, L.F.; Navarro, N.; Trapero, A. Spilocaea oleagina in olive groves of southern Spain: Survival, inoculum, production, and dispersal. Plant Dis. 2013, 97, 1549–1556. [Google Scholar] [CrossRef][Green Version]
- Viruega, J.R.; Roca, L.F.; Moral, J.; Trapero, A. Factors affecting infection and disease development on olive leaves inoculated with Fusicladium oleagineum. Plant Dis. 2011, 95, 1139–1146. [Google Scholar] [CrossRef][Green Version]
- Triki, M.A.; Rhouma, A. First report of Pseudocercospora cladosporioides, the causal agent of Cercospora leaf spot of olive trees, in Tunisia. Phytopathol. Mediterr. 2008, 47, 262–265. [Google Scholar]
- Nigro, F.; Sion, V. Integrated control of aerial fungal diseases of olive. In Proceedings of the 8th International Olive Symposium, Split, Croatia, 10–14 October 2016; Acta Hortic: Brussels, Belgium, 2018. [Google Scholar]
- Obanor, F.O.; Walter, M.; Jones, E.E.; Jaspers, M.V. Greenhouse and field evaluation of fungicides for control of olive leaf spot in New Zealand. Crop Prot. 2008, 27, 1335–1342. [Google Scholar] [CrossRef]
- Viruega, J.R.; Trapero, A.; Moreno, S. Efficacy of kresoxim-methyl against olive leaf spot caused by Spilocaea oleagina. Acta Hortic. 2002, 586, 801–804. [Google Scholar] [CrossRef]
- Roca, L.F.; Viruega, J.R.; Avila, A.; Moral, J.; Marchal, F.; Romero, J.; Agustí- Brisach, C.; Trapero, A. El cobre: Materia activa básica en el control de las enfermedades del olivo. In Proceedings of the 14th Symposium de Sanidad Vegetal, Sevilla, Spain, 25–27 January 2017; COITAND: Sevilla, Spain, 2017. [Google Scholar]
- Cacciola, S.O.; Faedda, R.; Sinatra, F.; Agosteo, G.E.; Schena, L.; Frisullo, S.; di San Lio, G.M. Olive anthracnose. J. Plant Pathol. 2012, 94, 29–44. [Google Scholar]
- Vitanovic, E. Use of Cu fungicides in vineyards and olive groves. In Fungicides for Plant and Animal Diseases, 1st ed.; Intech Open: London, UK, 2012; pp. 279–298. ISBN 9789533078045. [Google Scholar]
- Sanz-Cortés, F.; Martinez-Calvo, J.; Badenes, M.L.; Bleiholder, H.; Hack, H.; Yacer, G.; Meier, V. Phenological growth stages of olive trees (Olea europaea). Ann. Appl. Biol. 2002, 140, 151–157. [Google Scholar] [CrossRef]
- Pérez-Rodríguez, P.; Paradelo, M.; Soto-Gömez, D.; Fernández-Calviño, D.; López-Periago, J.E. Modeling losses of copper-based fungicide foliar sprays in wash-off under simulated rain. Int. J. Environ. Sci. Technol. 2015, 12, 661–672. [Google Scholar] [CrossRef][Green Version]
- Vicent, A.; Armengol, J.; García-Jiménez, J. Rain fastness and persistence of fungicides for control of Alternaria brown spot of citrus. Plant Dis. 2007, 91, 393–399. [Google Scholar] [CrossRef]
- Borkow, G.; Gabbay, J. Copper, an ancient remedy returning to fight microbial, fungal and viral infections. Curr. Chem. Biol. 2009, 3, 272–278. [Google Scholar]
- Mackie, K.A.; Müller, T.; Kandeler, E. Remediation of copper in vineyards—A mini review. Environ. Pollut. 2012, 167, 16–26. [Google Scholar] [CrossRef]
- Alavanja, M.C.R.; Hoppin, J.A.; Kamel, F. Health effects of chronic pesticide exposure: Cancer and neurotoxicity. Annu. Rev. Public Health 2004, 25, 155–197. [Google Scholar] [CrossRef]
- Epstein, L. Fifty years since silent spring. Annu. Rev. Phytopathol. 2014, 52, 377–402. [Google Scholar] [CrossRef]
- Gorlach, J.; Volrath, S.; Knauf-Beiter, G.; Hengy, G.; Beckhove, U.; Kogel, K.H.; Oostendorp, M.; Staub, T.; Ward, E.; Kessmann, H.; et al. Benzathiadiazole, a novel class of inducers of systemic acquired resistance, activates gene expression and disease resistance in wheat. Plant Cell 1996, 8, 629–643. [Google Scholar]
- Rossi, V.; Caffi, T. Fungicide models are key components of multiple modeling approaches for decision-making in crop protection. Phytopathol. Mediterr. 2018, 57, 153–169. [Google Scholar]
- Dhingra, O.D.; Sinclair, J.B. Basic Plant Pathology Methods, 2nd ed.; CRC Press: Boca Raton, FL, USA, 1995; p. 448. ISBN 978-0873716383. [Google Scholar]
- Moral, J.; Trapero, A. Assessing the susceptibility of olive cultivars to anthracnose caused by Colletotrichum acutatum. Plant Dis. 2009, 93, 1028–1036. [Google Scholar] [CrossRef]
- Moral, J.; Jurado-Bello, J.; Sánchez, M.I.; Oliveira, R.; Trapero, A. Effect of temperature, wetness duration, and planting density on olive anthracnose caused by Colletotrichum spp. Phytopathology 2012, 102, 974–981. [Google Scholar] [CrossRef]
- Romero, J.; Agustí-Brisach, C.; Roca, L.F.; Moral, J.; González-Domínguez, E.; Rossi, V.; Trapero, A. A long-term study on the effect of agroclimatic variables on olive scab in Spain. Crop Prot. 2018, 114, 239–243. [Google Scholar] [CrossRef]
- Barranco, D.; Fernández-Escobar, R.; Rallo, L. El Cultivo del Olivo, 7th ed.; Ediciones Mundi-Prensa: Madrid, Spain, 2017; p. 1008. ISBN 9788484767145. [Google Scholar]
- Teviotdale, B.L.; Sibbet, G.S.; Harper, D.H. Several copper fungicides control olive leaf spot. Calif. Agric. 1989, 43, 30–31. [Google Scholar]
- Steel, R.G.D.; Torrie, J.H. Bioestadística: Principios y Procedimientos, 2nd ed.; MacGraw-Hill: Bogotá, Colombia, 1985; p. 622. ISBN 9684514956. [Google Scholar]
- Rossi, V.; Caffi, T.; Salinari, F. Helping farmers face the increasing complexity of decision-making for crop protection. Phytopathol. Mediterr. 2012, 51, 457–479. [Google Scholar]
- Ciccarone, A. Uno sguardo alla patologia dell’olivo nei paesi mediterranei. Options Mediterr. 1986, 24, 71–79. [Google Scholar]
- Gatica De Mathey, M.; Oriolani, E.J.A. Cercospora del Olivo, 1st ed.; INTA Estación Experimental Regional Agropecuaria Mendoza: Lujan de Cuyo, Argentina, 1983; p. 8. [Google Scholar]
- Graniti, A.; Laviola, C. Sguardo generale alle malattie parassitarie dell’olivo. Inf. Fitopatol. 1981, 31, 77–92. [Google Scholar]
- Sarasola, A.A. Nueva enfermedad del olivo en la Argentina causada por Cercospora cladosporioides Sacc. Rev. Fac. Agron. La Plata 1951, 28, 41–47. [Google Scholar]
- Evans, E. Enfermedades de las Plantas y su Control Químico, 1st ed.; Labor: Barcelona, Spain, 1973; p. 307. ISBN 978-84-335-5809-1. [Google Scholar]
- Moral, J.; Agustí-Brisach, C.; Agalliu, G.; Rodrigues de Oliveira, M.; Pérez- Rodríguez, M.; Roca, L.F.; Romero, J.; Trapero, A. Preliminary selection and evaluation of fungicides and natural compounds to control olive anthracnose caused by Colletotrichum species. Crop Prot. 2018, 114, 167–176. [Google Scholar] [CrossRef]
- Sanchez-Pacheco, N. Evaluación In Vitro de Fungicidas Contra Spilocaea oleagina, Agente del Repilo del Olivo, Trabajo Fin de Carrera; ETSIAM, Universidad de Córdoba: Córdoba, Spain, 1999. [Google Scholar]
- Campillo, R. Los productos cúpricos en la olivicultura actual. Phytoma Esp. 1998, 102, 159–167. [Google Scholar]
- Cirulli, M.; Laviola, C.; Roberti, D. Avversità e difesa. In L’Olivo. Frutticoltura Anni 80; REDA: Roma, Italy, 1981; pp. 142–187. [Google Scholar]
- Nigro, F.; Ippolito, A.; Gallone, P.; Carmignano, P.; Romanazi, G. Cercosporiosis of olive in Apulia and attempts to control the disease. Acta Hortic. 2002, 586, 773–776. [Google Scholar] [CrossRef]
- Agrios, G.N. Plant Pathology, 5th ed.; Elsevier Academic Press: San Diego, CA, USA, 2005; p. 952. ISBN 9780080473789. [Google Scholar]
- Yang, C.; Hamel, C.; Vujanovic, V.; Gan, Y. Fungicide: Modes of action and possible impact on non target microorganisms. ISRN Ecol. 2011, 1, 1–8. [Google Scholar]
- Hunt, T.E.; Wright, R.J.; Hein, G.L. Economic Threshold for Today’s Commodity Values; Adapted from Proceeding Crop Production Clinics 2019; University of Nebraska-Lincoln: Lincoln, NE, USA, 2009; pp. 93–96. [Google Scholar]
- Savary, S.; Teng, P.S.; Willocquet, L.; Nutter, F.W. Quantificacion and modelling of crop losses: A review of purposes. Annu. Rev. Phytopathol. 2006, 44, 89–112. [Google Scholar] [CrossRef]
- Chen, G.; Pham, T.T. Introduction to Fuzzy Sets, Fuzzy Logic, and Fuzzy Control Systems; CRC Press: Boca Ratón, FL, USA, 2000; p. 328. ISBN 9780367397883. [Google Scholar]

| Tradename 1 | Active Ingredient-Formulation 3 | Class (FRAC Number) 4 | Systemic Action | Doses (mg L−1) | |||
|---|---|---|---|---|---|---|---|
| Company 2 | Mycelial Growth | Conidial Germination | Manufacturer Recommended | ||||
| Caldo Bordelés Vallés | Ind. Químicas del Vallés | Copper calcium sulfate 20% WP | Inorganic (M1) | No | 50; 150; 300; 600 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 5000 |
| Copper sulphate | Merk Lab | Copper sulphate 25% Cu WP | Inorganic (M1) | No | 50; 150; 300; 600 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 2500 |
| Cuproflow Caffaro Blue | Caffaro (Isagro) | Copper oxychloride 38% Cu WP | Inorganic (M1) | No | 50; 150; 300; 600 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 4000 |
| Cuprosan * | Bayer CropScience | Copper oxychloride 50% Cu WG | Inorganic (M1) | No | - | - | 4000 |
| Funguran-OH 50 PH | Nufarm España | Copper hydroxide 50% WP | Inorganic (M1) | No | 50; 150; 300; 600 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 4000 |
| Nordox super 75 * | Nordox | Cuprous oxide 75% WP | Inorganic (M1) | No | 50; 150; 300; 600 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 2000 |
| Benomilo 50 | Aragonesas Agro | Benomyl 50% WP | ß-tubulin inhibitor (B) (3) | Yes | 0.001;0.01;0.1;1;10 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 900 |
| Bellis * | Basf | Pyraclostrobin 10% + Boscalid 20% WG | QoI 5 (11)-SDHI 6 (7) | Yes | 0.001;0.01;0.1;1;10 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01; 0.001; 0.0001 | 1000 |
| Flint Max * | Bayer CropScience | Trifloxystrobin 25% + Tebuconazole 50% WG | QoI (11)/ DMI-triazole (3) | Yes | - | - | 200 |
| Score 25 * | Novartis (Syngenta) | Difenoconazole 25% EC | DMI 7-triazole (3) | Yes | 0.001;0.01;0.1;1;10 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 150 |
| Stroby * | Basf | Kresoxim- methyl 50% WG | QoI (11) | Yes | 0.001;0.01;0.1;1;10 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 750 |
| Belpron F 50 | Probelte | Folpet 50% WP | Phtalimide (M4) | No | 0.001;0.01;0.1;1;10 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 3000 |
| Belpron M 80 | Probelte | Maneb 80% WP | Carbamate (EBDC) 7(M3) | No | 50; 150; 300; 600 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 2500 |
| Captazel | Zeneca Agro (Syngenta) | Captan 50% WP | Phtalimide (M4) | No | 50; 150; 300; 600 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 3000 |
| Cobreline Folpet * | C. Q. Masso | Folpet 30% + Cuprous oxide 10% + Copper calcium sulfate 10% WP | Phtalimide (M4)- Inorganic (M1)- Inorganic (M1) | No | 0.001;0.01;0.1;1;10 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 2500 |
| Dithane M45 | Rohm and Haas (Dow) | Mancozeb 80% WP | Carbamate (EBDC) 8 (M3) | No | 50; 150; 300; 600 | 32;16;8;4;2;1;0.5;0.25;0.12;0.06;0.03;0.01 | 4000 |
| Orchard | Location | UTM 1 | Farming System 2 | Cultivar | Risk Factor 3 | Period of Evaluation | |
|---|---|---|---|---|---|---|---|
| 1 | El Cañuelo | Córdoba (Córdoba) | 37.763166; −4.670026 | Low-density/Organic | Picual | Low | 2012/2016 |
| 2 | El Realengo 1 4 | Antequera (Málaga) | 37.188711; −4.573885 | High-density/IPM | Arbequina | Mild | 2012/2016 |
| 3 | El Realengo 2 | Antequera (Málaga) | 37.191442; −4.576739 | High-density/IPM | Hojiblanca | Mild | 2012/2016 |
| 4 | El Realengo 3 | Antequera (Málaga) | 37.,191959; −4.576605 | High-density/IPM | Picual | Mild | 2012/2016 |
| 5 | Fonte dos Frades | Beja (Portugal) | 38.021307; −7.747067 | High-density/IPM | Hojiblanca | High | 2012/2013 |
| 6 | La Veguilla | Córdoba (Córdoba) | 37.820535; −4.896576 | High-density/IPM | Hojiblanca | Low | 2012/2016 |
| 7 | Linares | Linares (Jaén) | 38.097234; −3.709578 | High-density/IPM | Picual | Mild | 2012/2016 |
| 8 | Los Cansinos | Córdoba (Córdoba) | 37.892221; −4.596169 | Super-High-density/IPM | Arbequina | Low | 2012/2015 |
| 9 | Malena de Castro | Linares (Jaén) | 38.113009; −3.590850 | High-density/IPM | Picual | Low | 2012/2016 |
| 10 | Naranjilla | Carmona (Sevilla) | 37.409752; −5.807546 | High-density/IPM | Manzanilla de Sevilla | High | 2012/2015 |
| 11 | Ribera Alta | Córdoba (Córdoba) | 37.953271; −4.624638 | High-density/IPM | Picual | High | 2012/2016 |
| 12 | Todolivo | Pedro Abad (Córdoba) | 37.959700; −4.466828 | Super-High-density/IPM | Arbequina | Mild | 2012/2015 |
| 13 | Aljarafe | Bollullos de la Mitación (Sevilla) | 37.356450; −6.164204 | High-density/Organic | Manzanilla de Sevilla | High | 2013/2016 |
| 14 | Los Ballesteros | El Cuervo (Sevilla) | 36.795540; −5.971842 | High-density/IPM | Hojiblanca | High | 2013/2016 |
| 15 | Cortijo de Guadiana 1 4 | Úbeda (Jaén) | 37.914793; −3.232109 | High-density/IPM | Picual | Mild | 2014/2016 |
| 16 | Cortijo de Guadiana 2 | Úbeda (Jaén) | 37.902171; −3.237424 | High-density/Organic | Picual | Low | 2014/2016 |
| Fungicides 1 | Inhibition | |||
|---|---|---|---|---|
| Mycelial Growth | Conidial Germination | |||
| EC50 2 | EC90 3 | EC50 | EC90 | |
| Copper hydroxide 50% Cu WP | 111.82 b 4 | 400.41 a | 0.85 cd | 15.03 a |
| Copper oxychloride 38% Cu WP | 210.39 a | >600 | 0.75 cd | 13.05 a |
| Cuprous oxide 75% WP | 172.14 ab | >600 | 0.36 e | 3.32 bc |
| Copper calcium sulfate 20% WP | 223.15 a | >600 | 0.80 cd | 5.38 b |
| Copper sulphate 25% Cu WP | >600 | >600 | 0.08 gh | 0.65 e |
| Captan 50% WP | >600 | >600 | 0.48 de | 1.35 de |
| Maneb 80% WP | 42.58 c | >600 | 1.21 c | 27.52 a |
| Mancozeb 80% WP | 60.61 c | 327.27 a | 6.74 b | >32 |
| Folpet 50% WP | 2.73 e | >10 | 0.10 fg | 0.59 e |
| Folpet 30% + Cuprous oxide 10% + Copper calcium sulfate 10% WP | 8.17 d | >10 | 0.14 f | 1.77 cd |
| Difenoconazole 25% EC | 2.14 e | >10 | 17.01 a | >32 |
| Kresoxim- methyl 50% WG | 0.54 f | >10 | 0.004 i | 0.04 g |
| Pyraclostrobin 10% + Boscalid 20% WG | 0.12 g | >10 | 0.002 j | 0.01 h |
| Benomyl 50% WP | 0.11 g | 0.89 b | 0.05 h | 0.15f |
| Orchard | Strategy | FA 1 | CLSO Incidence (%) 2 | IFAE 3 | RIFAE 4 | |
|---|---|---|---|---|---|---|
| 1 | El Cañuelo | Traditional | 1.75 | 0.49 b5 | 56.86 | |
| Conservative | 1.50 | 1.04 a | 65.97 | 1.16 | ||
| Risky | 1.50 | 0.87 a | 66.09 | 1.16 | ||
| 2 | El Realengo | Traditional | 2.25 | 1.87 a | 43.61 | |
| Conservative | 2.25 | 1.87 a | 43.61 | 1.00 | ||
| Risky | 1.75 | 1.87 a | 56.07 | 1.29 | ||
| 3 | El Realengo | Traditional | 2.00 | 1.85 b | 49.08 | |
| Conservative | 1.75 | 3.36 a | 55.22 | 1.13 | ||
| Risky | 1.00 | 2.74 ab | 97.26 | 1.98 | ||
| 4 | El Realengo | Traditional | 3.67 | 0.84 a | 27.02 | |
| Conservative | 3.33 | 0.76 a | 29.80 | 1.10 | ||
| Risky | 2.00 | 1.74 a | 49.13 | 1.81 | ||
| 5 | Fonte Dos Frades | Traditional | 5.00 | 1.22 a | 19.76 | |
| Conservative | 4.00 | 1.22 a | 24.70 | 1.25 | ||
| Risky | 2.00 | 1.22 a | 49.39 | 2.50 | ||
| 6 | La Veguilla | Traditional | 2.50 | 1.14 b | 39.54 | |
| Conservative | 1.75 | 2.19 a | 55.89 | 1.41 | ||
| Risky | 1.25 | 2.07 a | 78.34 | 1.97 | ||
| 7 | Linares | Traditional | 2.50 | 0.61 b | 39.76 | |
| Conservative | 2.50 | 0.62 b | 39.75 | 1.00 | ||
| Risky | 1.75 | 1.87 a | 56.07 | 1.41 | ||
| 8 | Los Cansinos | Traditional | 3.33 | 0.04 a | 30.02 | |
| Conservative | 2.00 | 0.04 a | 49.98 | 1.67 | ||
| Risky | 1.33 | 0.04 a | 75.16 | 2.50 | ||
| 9 | Malena de Castro | Traditional | 2.50 | 0.70 b | 39.72 | |
| Conservative | 2.25 | 2.06 a | 43.53 | 1.10 | ||
| Risky | 2.00 | 2.53 a | 48.74 | 1.23 | ||
| 10 | Naranjilla | Traditional | 2.33 | 0.84 b | 42.56 | |
| Conservative | 2.67 | 2.03 a | 36.69 | 0.86 | ||
| Risky | 2.33 | 1.02 b | 42.48 | 1.00 | ||
| 11 | Ribera Alta | Traditional | 3.25 | 2.50 a | 30.00 | |
| Conservative | 3.25 | 2.89 a | 29.88 | 1.00 | ||
| Risky | 2.50 | 2.81 a | 38.88 | 1.29 | ||
| 12 | Todolivo | Traditional | 3.00 | 0.04 a | 33.32 | |
| Conservative | 2.00 | 0.04 a | 49.98 | 1.50 | ||
| Risky | 1.67 | 0.04 a | 59.86 | 1.80 | ||
| 13 | Aljarafe | Traditional | 4.33 | 0.81 b | 22.91 | |
| Conservative | 3.33 | 1.48 a | 29.59 | 1.29 | ||
| 14 | Los Ballesteros | Traditional | 5.00 | 0.81 a | 19.84 | |
| Conservative | 5.00 | 0.60 a | 19.88 | 1.00 | ||
| Risky | 3.33 | 0.51 a | 29.88 | 1.51 | ||
| 15 | Cortijo de Guadiana | Traditional | 2.50 | 0.21 a | 39.92 | |
| Conservative | 2.00 | 0.28 a | 49.86 | 1.25 | ||
| Risky | 1.50 | 0.18 a | 66.55 | 1.67 | ||
| 16 | Cortijo de Guadiana | Traditional | 2.00 | 0.21 a | 49.90 | |
| Conservative | 0.50 | 0.31 a | 199.38 | 4.00 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romero, J.; Ávila, A.; Agustí-Brisach, C.; Roca, L.F.; Trapero, A. Evaluation of Fungicides and Management Strategies against Cercospora Leaf Spot of Olive Caused by Pseudocercospora cladosporioides. Agronomy 2020, 10, 271. https://doi.org/10.3390/agronomy10020271
Romero J, Ávila A, Agustí-Brisach C, Roca LF, Trapero A. Evaluation of Fungicides and Management Strategies against Cercospora Leaf Spot of Olive Caused by Pseudocercospora cladosporioides. Agronomy. 2020; 10(2):271. https://doi.org/10.3390/agronomy10020271
Chicago/Turabian StyleRomero, Joaquín, Arantxa Ávila, Carlos Agustí-Brisach, Luis F. Roca, and Antonio Trapero. 2020. "Evaluation of Fungicides and Management Strategies against Cercospora Leaf Spot of Olive Caused by Pseudocercospora cladosporioides" Agronomy 10, no. 2: 271. https://doi.org/10.3390/agronomy10020271
APA StyleRomero, J., Ávila, A., Agustí-Brisach, C., Roca, L. F., & Trapero, A. (2020). Evaluation of Fungicides and Management Strategies against Cercospora Leaf Spot of Olive Caused by Pseudocercospora cladosporioides. Agronomy, 10(2), 271. https://doi.org/10.3390/agronomy10020271







