Johnsongrass (Sorghum halepense (L.) Pers.) Interference, Control and Recovery under Different Management Practices and its Effects on the Grain Yield and Quality of Maize Crop
Abstract
1. Introduction
2. Materials and Methods
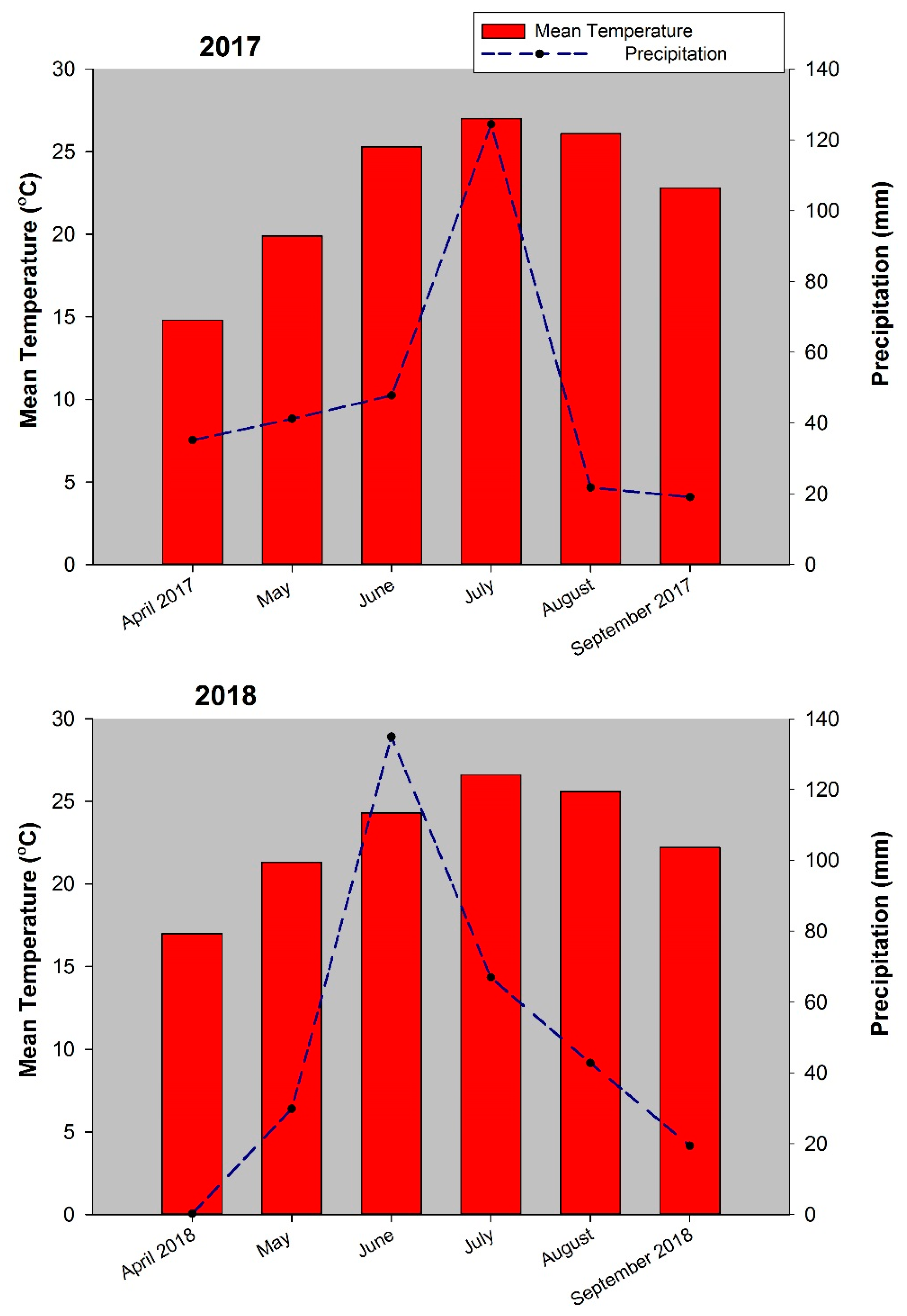
2.1. Study Site
2.2. Experimental Design
2.3. Data Collection
2.4. Statistical Analyses
3. Results
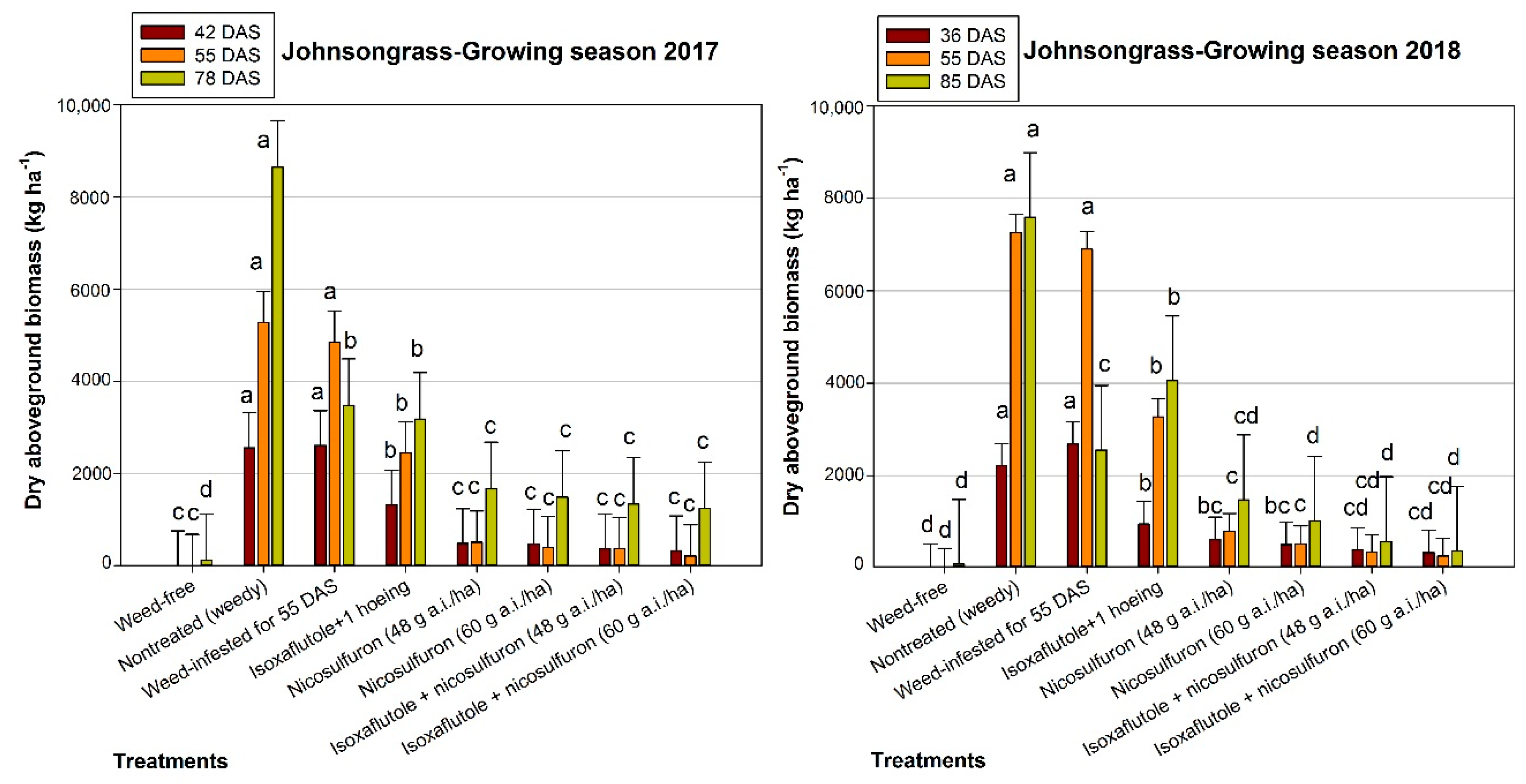
3.1. Johnsongrass Aboveground Biomass
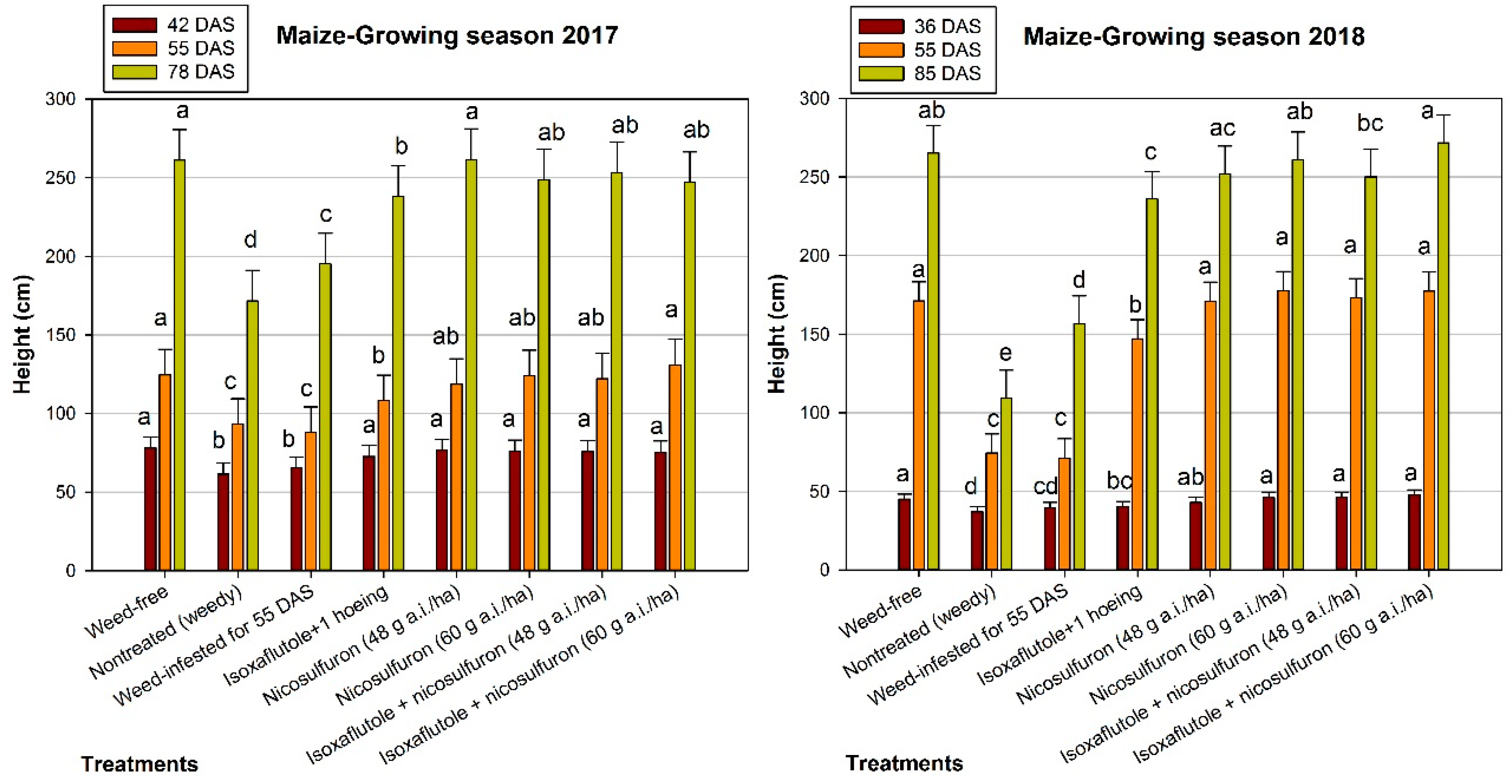
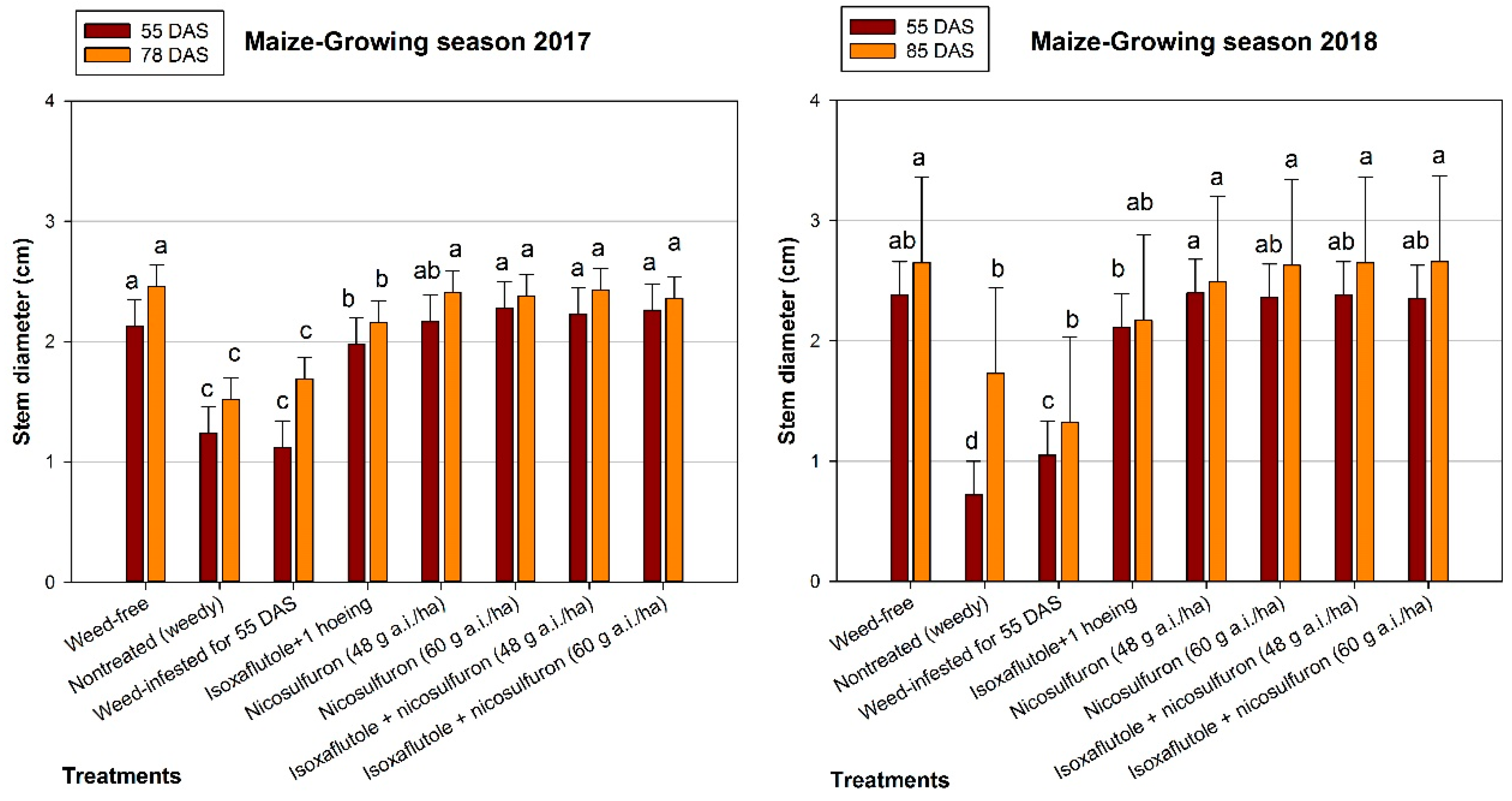
3.2. Maize Growth
3.3. Relative Chlorophyll Content
3.4. Maize Yield and Quality
4. Discussion
4.1. Johnsongrass Control
4.2. Maize Growth
4.3. Maize Yield and Quality
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAO (Food and Agriculture Organization of the United Nations). FAOSTAT Database. Available online: www.fao.org/faostat (accessed on 2 January 2019).
- Sekhar, J.C.; Karjagi, C.G.; Kumar, B.; Rakshit, S.; Soujanya, L.; Kumar, P.; Singh, K.P.; Dhandapani, A.; Dass, S.; Kumar, R.S. Genetics of resistance to Sesamia inferens infestation and its correlation with yield in maize. Plant Breed. 2015, 134, 394–399. [Google Scholar] [CrossRef]
- Marković, M.; Josipović, M.; Šoštarić, J.; Jambrović, A.; Brkić, A. Response of maize (Zea mays L.) grain yield and yield components to irrigation and nitrogen fertilization. J. Cent. Eur. Agric. 2017, 18, 55–72. [Google Scholar] [CrossRef][Green Version]
- Assefa, Y.; Carter, P.; Hinds, M.; Bhalla, G.; Schon, R.; Jeschke, M.; Paszkiewicz, S.; Smith, S.; Ciampitti, I.A. Analysis of long term study indicates both agronomic optimal plant density and increase maize yield per plant contributed to yield gain. Sci. Rep. 2018, 8, 4937. [Google Scholar] [CrossRef] [PubMed]
- Hussain, H.A.; Men, S.; Hussain, S.; Chen, Y.; Ali, S.; Zhang, S.; Zhang, K.; Li, Y.; Xu, Q.; Liao, C.; et al. Interactive effects of drought and heat stresses on morpho-physiological attributes, yield, nutrient uptake and oxidative status in maize hybrids. Sci. Rep. 2019, 9, 3890. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, G.R.; Mozafaril, S.; Najaphy, A.; Ghobadi, M.E. Corn (Zea mays L.) seed vigour and quality as influenced by weed interference and living mulch. Adv. Environ. Biol. 2012, 6, 1026–1031. [Google Scholar]
- Lehoczky, É.; Márton, L.; Nagy, P. Competition for nutrients between cold-tolerant maize and weeds. Commun. Soil Sci. Plant Anal. 2013, 44, 526–534. [Google Scholar] [CrossRef]
- Takim, F. Weed competition in maize (Zea mays L.) as a function of the timing of hand-hoeing weed control in the southern Guinea savanna zone of Nigeria. Acta Agron. Hung. 2012, 60, 257–264. [Google Scholar] [CrossRef]
- Andújar, D.; Barroso, J.; Fernández-Quintanilla, C.; Dorado, J. Spatial and temporal dynamics of Sorghum halepense patches in maize crops. Weed Res. 2012, 52, 411–420. [Google Scholar] [CrossRef]
- Follak, S.; Essl, F. Spread dynamics and agricultural impact of Sorghum halepense, an emerging invasive species in Central Europe. Weed Res. 2013, 53, 53–60. [Google Scholar] [CrossRef]
- Damalas, C.A.; Gitsopoulos, T.K.; Koutroubas, S.D.; Alexoudis, C.; Georgoulas, I. Weed control and selectivity in maize (Zea mays L.) with tembotrione mixtures. Int. J. Pest Manag. 2018, 64, 11–18. [Google Scholar] [CrossRef]
- Chiriţa, R.; Grozea, I.; Sarpe, N.; Lauer, K.F. Control of Sorghum halepense (L.) species in western part of Romania. Commun. Agric. Appl. Biol Sci. 2008, 73, 959–964. [Google Scholar] [PubMed]
- Barroso, J.; Maxwell, B.D.; Dorado, J.; Andújar, D.; San Martín, C.; Fernández-Quintanilla, C. Response of Sorghum halepense demographic processes to plant density and rimsulfuron dose in maize. Weed Res. 2016, 56, 304–312. [Google Scholar] [CrossRef]
- Scopel, A.L.; Ballare, C.L.; Ghersa, C.M. Role of seed reproduction in the population ecology of Sorghum halepense in maize crops. J. Appl. Ecol. 1988, 25, 951–962. [Google Scholar] [CrossRef]
- Atwater, D.Z.; Kim, W.; Tekiela, D.R.; Barney, J.N. Competition and propagule density affect sexual and clonal propagation of a weed. Invasive Plant Sci. Manag. 2017, 10, 17–25. [Google Scholar] [CrossRef]
- Vasilakoglou, I.; Dhima, K.; Eleftherohorinos, I. Allelopathic potential of bermudagrass and johnsongrass and their interference with cotton and corn. Agron. J. 2005, 97, 303–313. [Google Scholar]
- Horowitz, M. Spatial growth of Sorghum halepense (L.) Pers. Weed Res. 1973, 13, 200–208. [Google Scholar] [CrossRef]
- Ghosheh, H.Z.; Holshouser, D.L.; Chandler, J.M. Influence of density on johnsongrass (Sorghum halepense) interference in field corn (Zea mays). Weed Sci. 1996, 44, 879–883. [Google Scholar] [CrossRef]
- Mitskas, M.B.; Tsolis, C.E.; Eleftherohorinos, I.G.; Damalas, C.A. Interference between corn and johnsongrass (Sorghum halepense) from seed or rhizomes. Weed Sci. 2003, 51, 540–545. [Google Scholar] [CrossRef]
- Karkanis, A.; Alexiou, A.; Katsaros, C.; Petropoulos, S. Allelopathic activity of spearmint (Mentha spicata L.) and peppermint (Mentha x piperita L.) reduces yield, growth, and photosynthetic rate in a succeeding crop of maize (Zea mays L.). Agronomy 2019, 9, 461. [Google Scholar] [CrossRef]
- Stephenson, D.O.; Bond, J.A. Evaluation of thiencarbazone-methyl and isoxaflutole-based herbicide programs in corn. Weed Technol. 2012, 26, 37–42. [Google Scholar] [CrossRef]
- Ortiz, A.; Martínez, L.; Quintana, Y.; Pérez, P.; Fischer, A. Resistance of johnsongrass [Sorghum halepense (L.) Pers.] to herbicides nicosulfuron and foramsulfuron+iodosulfuron in Venezuela. Bioagro 2014, 26, 71–78. [Google Scholar]
- Rosales-Robles, E.; Chandler, J.M.; Senseman, S.A.; Prostko, E.P. Influence of growth stage and herbicide rate on postemergence johnsongrass (Sorghum halepense) control. Weed Technol. 1999, 13, 525–529. [Google Scholar] [CrossRef]
- Johnson, W.G.; Jianmei, L.I.; Wait, J.D. Johnsongrass control, total nonstructural carbohydrates in rhizomes, and regrowth after application of herbicides used in herbicide-resistant corn (Zea mays). Weed Technol. 2003, 17, 36–41. [Google Scholar] [CrossRef]
- Torma, M.; Kazinczi, G.; Hódi, L. Postemergence herbicide treatments in maize against difficult to control weeds in Hungary. J. Plant Dis. Prot. 2006, 20, 781–786. [Google Scholar]
- Hernández, M.J.; León, R.; Fischer, A.J.; Gebauer, M.; Galdames, R.; Figueroa, R. Target-site resistance to nicosulfuron in johnsongrass (Sorghum halepense) from Chilean corn fields. Weed Sci. 2015, 63, 631–640. [Google Scholar] [CrossRef]
- Panozzo, S.; Milani, A.; Scarabel, L.; Balogh, Á.; Dancza, I.; Sattin, M. Occurrence of different resistance mechanisms to acetolactate synthase inhibitors in European Sorghum halepense. J. Agric. Food Chem. 2017, 65, 7320–7327. [Google Scholar] [CrossRef]
- Eleftherohorinos, I.G.; Kotoula-Syka, E. Influence of herbicide application rate and timings for post-emergence control of Sorghum halepense (L.) Pers. in maize. Weed Res. 1995, 35, 99–103. [Google Scholar] [CrossRef]
- Djurkić, M.; Knežević, M.; Ostojić, Z. Effect of rimsulfuron application on weeds in maize inbred lines in Croatia. Cereal Res. Commun. 1997, 25, 203–209. [Google Scholar] [CrossRef]
- Johnson, D.B.; Norsworthy, J.K. Johnsongrass (Sorghum halepense) management as influenced by herbicide selection and application timing. Weed Technol. 2014, 28, 142–150. [Google Scholar] [CrossRef]
- Gubbiga, N.G.; Worsham, A.D.; Corbin, F.T. Investigations into the growth suppressing effect of nicosulfuron-treated johnsongrass (Sorghum halepense) on corn (Zea mays). Weed Sci. 1996, 44, 640–644. [Google Scholar] [CrossRef]
- Labuschagne, M.; Phalafala, L.; Osthoff, G.; van Biljon, A. The influence of storage conditions on starch and amylose content of South African quality protein maize and normal maize hybrids. J. Stored Prod. Res. 2014, 56, 16–20. [Google Scholar] [CrossRef]
- Silva, P.R.F.; Strieder, M.L.; Coser, R.P.S.; Rambo, L.; Sangoi, L.; Argenta, G.; Forsthofer, E.L.; Silv, A.A. Grain yield and kernel crude protein content increases of maize hybrids with late nitrogen side-dressing. Sci. Agric. 2005, 62, 487–492. [Google Scholar] [CrossRef]
- Karkanis, A.; Vellios, E.; Grigoriou, F.; Gkrimpizis, T.; Giannouli, P. Evaluation of efficacy and compatibility of herbicides with fungicides in durum wheat (Triticum durum Desf.) under different environmental conditions: Effects on grain yield and gluten content. Not. Bot. Horti Agrobo. 2018, 46, 601–607. [Google Scholar] [CrossRef]
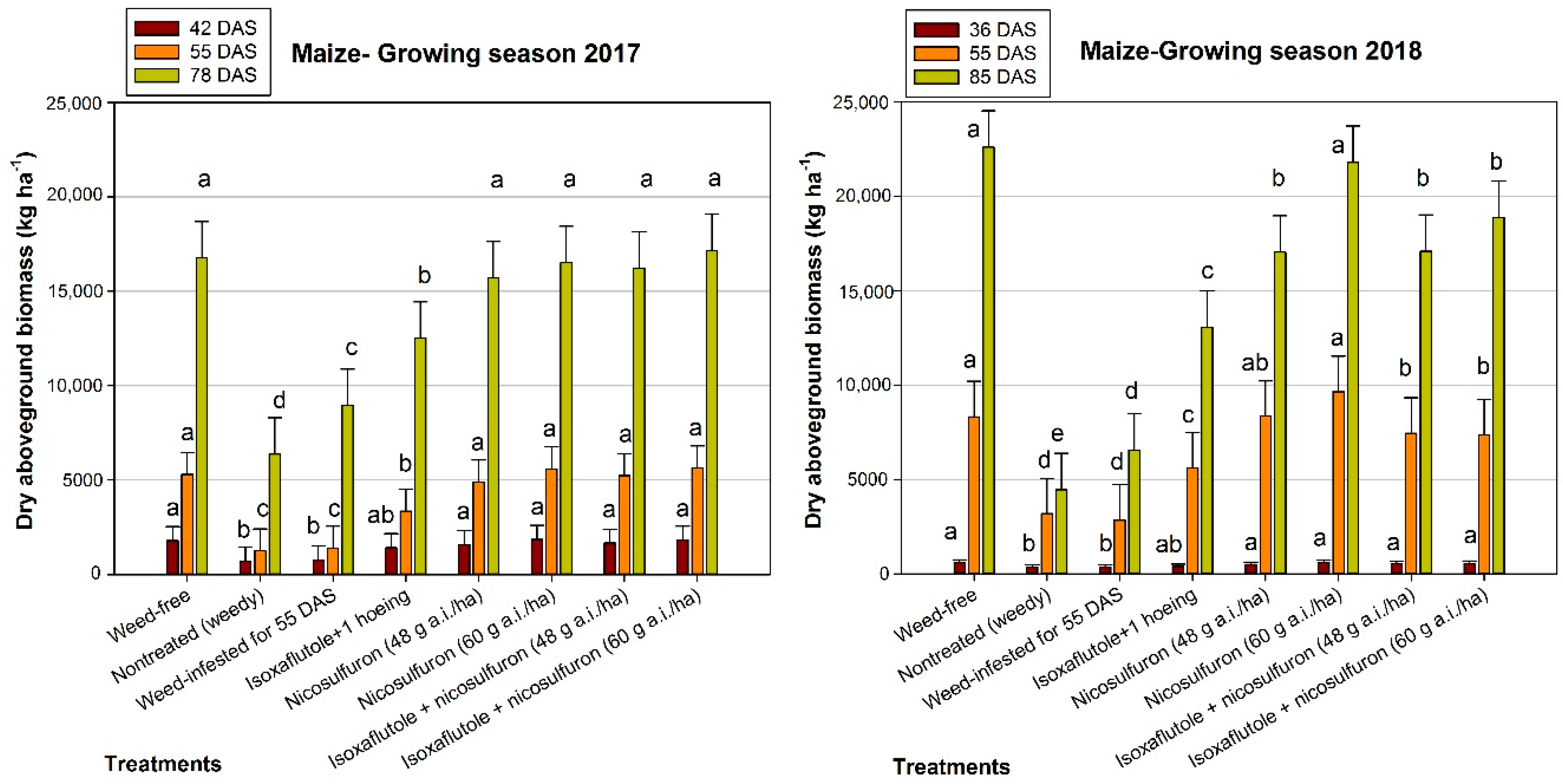
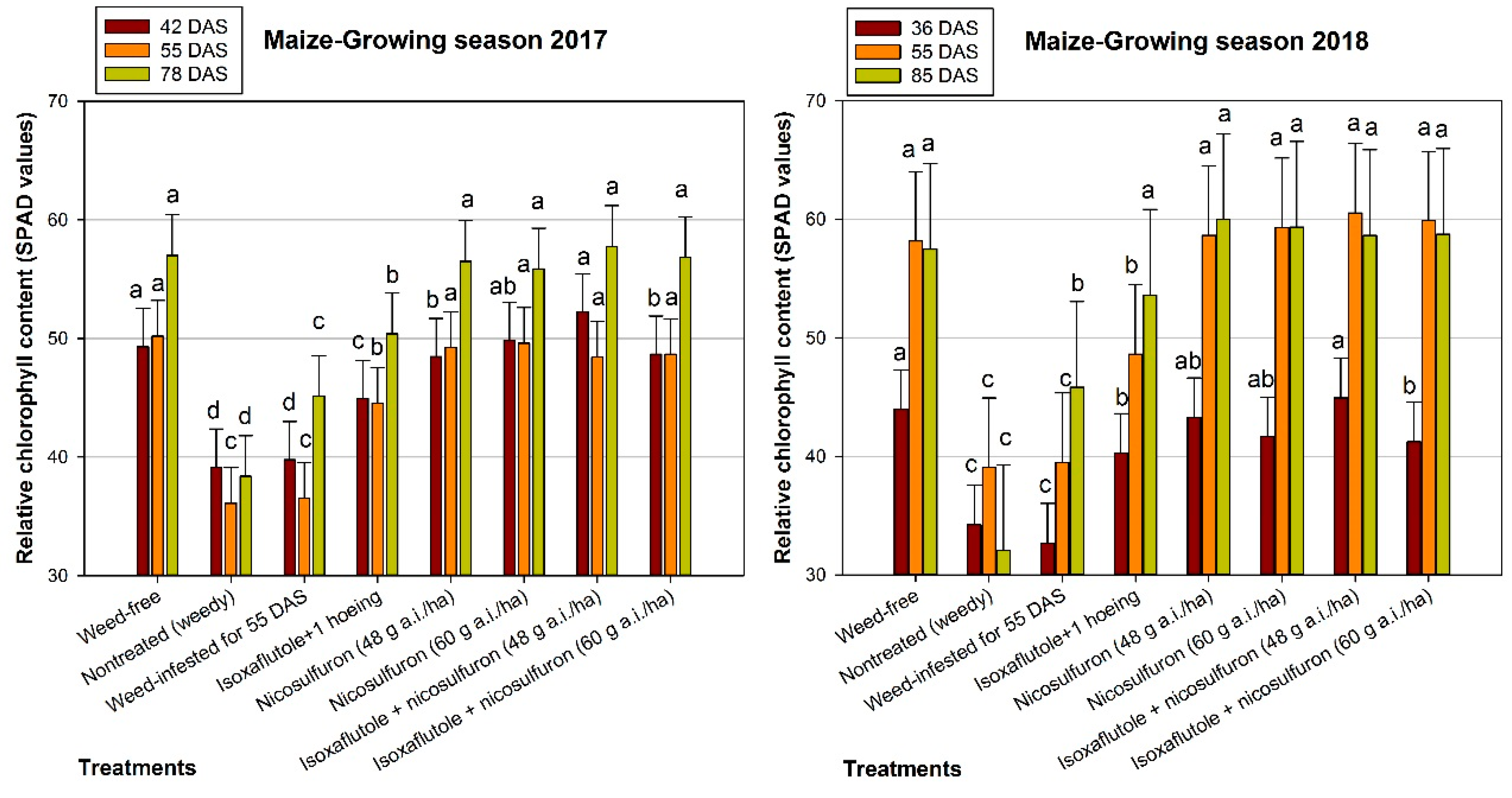
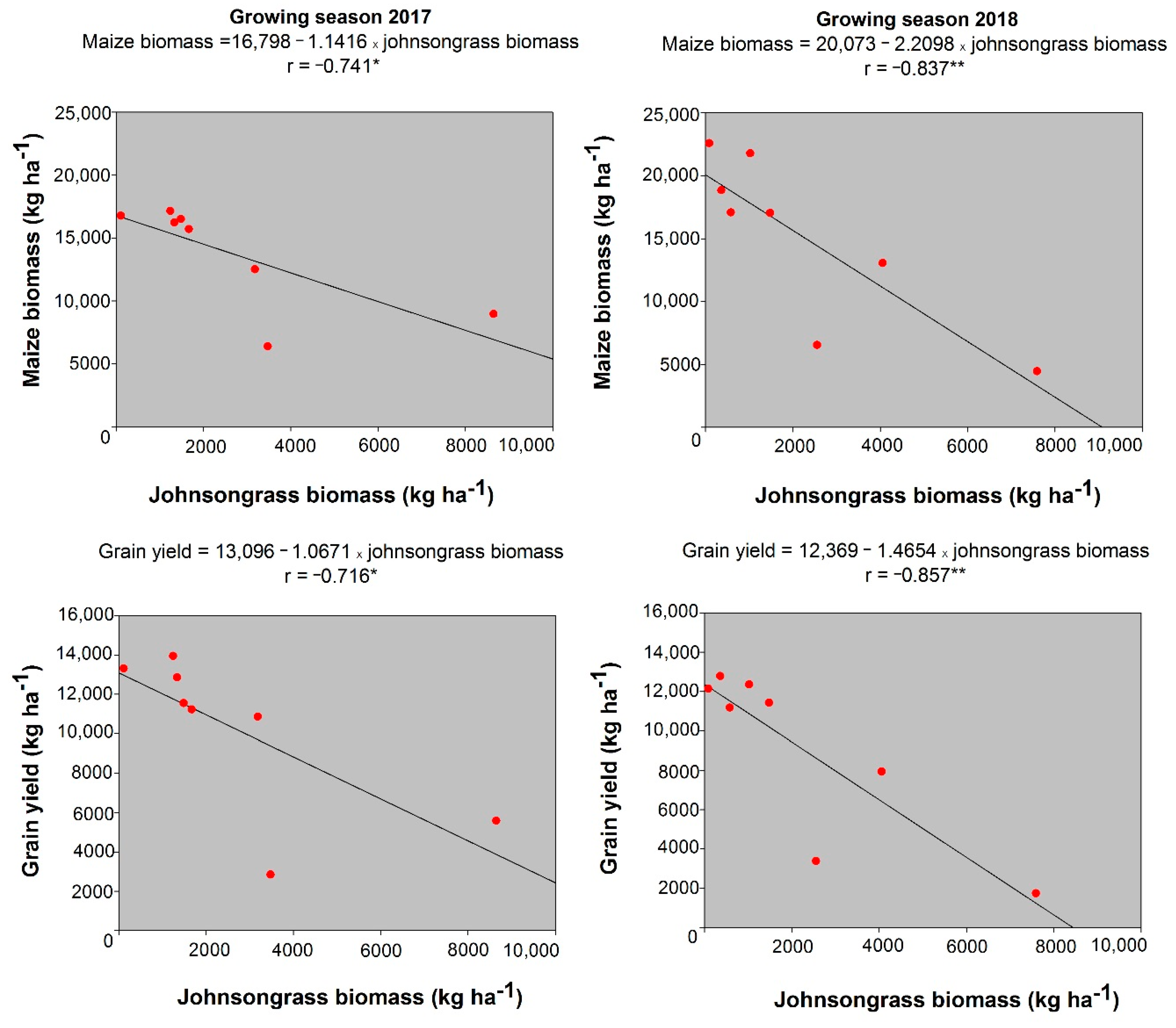
| 2017 Treatments | Ear Length (cm) | 1000 Grain-Weight (g) | Grain Yield (kg ha−1) |
|---|---|---|---|
| Weed-free | 22.3 ab | 423.6 a | 13,320 a |
| Nontreated (weedy) | 10.7 f | 288.0 e | 2841 d |
| Weed-infested for 55 DAS | 13.9 e | 320.0 d | 5582 c |
| Isoxaflutole + 1 hoeing | 19.6 d | 361.0 c | 8983 b |
| Nicosulfuron (48 g a.i. ha−1) | 21.3 bc | 401.3 b | 11,899 a |
| Nicosulfuron (60 g a.i. ha−1) | 22.6 ac | 408.3 ab | 12,220 a |
| Isoxaflutole + nicosulfuron (48 g a.i. ha−1) | 21.2 b | 407.3 ab | 12,861 a |
| Isoxaflutole + nicosulfuron (60 g a.i. ha−1) | 22.7 a | 421.3 a | 13,949 a |
| LSD5% | 1.30 | 18.81 | 2325 |
| 2018 Treatments | Ear Length (cm) | 1000 Grain-Weight (g) | Grain Yield (kg ha−1) |
| Weed-free | 21.7 a | 369.9 a | 12,141ac |
| Nontreated (weedy) | 11.9 d | 280.7 d | 1743 f |
| Weed-infested for 55 DAS | 13.8 c | 302.9 c | 3391 e |
| Isoxaflutole + 1 hoeing | 18.3 b | 331.3 b | 7938 d |
| Nicosulfuron (48 g a.i. ha−1) | 20.8 a | 362.1 a | 11,432 bc |
| Nicosulfuron (60 g a.i. ha−1) | 21.9 a | 372.5 a | 12,359 a |
| Isoxaflutole + nicosulfuron (48 g a.i. ha−1) | 21.8 a | 365.7 a | 11,184 b |
| Isoxaflutole + nicosulfuron (60 g a.i. ha−1) | 22.2 a | 367.2 a | 12,788 a |
| LSD5% | 1.47 | 15.79 | 945.51 |
| Treatments | 2017 | 2018 | ||
|---|---|---|---|---|
| Protein Content (%) | Starch Content (%) | Protein Content (%) | Starch Content (%) | |
| Weed-free | 8.19 a | 64.70 a | 7.73 a | 64.10 ab |
| Nontreated (weedy) | 7.27 c | 62.07 c | 6.70 c | 63.00 c |
| Weed-infested for 55 DAS | 7.12 c | 62.77 b | 6.91 c | 62.87 c |
| Isoxaflutole + 1 hoeing | 7.78 b | 64.07 a | 7.37 b | 63.60 bc |
| Nicosulfuron (48 g a.i. ha−1) | 8.06 a | 64.83 a | 7.68 a | 64.57 a |
| Nicosulfuron (60 g a.i. ha−1) | 8.16 a | 64.73 a | 7.73 a | 64.30 ab |
| Isoxaflutole + nicosulfuron (48 g a.i. ha−1) | 8.10 a | 64.63 a | 7.75 a | 64.23 ab |
| Isoxaflutole + nicosulfuron (60 g a.i. ha−1) | 8.04 a | 64.80 a | 7.83 a | 64.07 ab |
| LSD5% | 0.151 | 1.085 | 0.229 | 0.802 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karkanis, A.; Athanasiadou, D.; Giannoulis, K.; Karanasou, K.; Zografos, S.; Souipas, S.; Bartzialis, D.; Danalatos, N. Johnsongrass (Sorghum halepense (L.) Pers.) Interference, Control and Recovery under Different Management Practices and its Effects on the Grain Yield and Quality of Maize Crop. Agronomy 2020, 10, 266. https://doi.org/10.3390/agronomy10020266
Karkanis A, Athanasiadou D, Giannoulis K, Karanasou K, Zografos S, Souipas S, Bartzialis D, Danalatos N. Johnsongrass (Sorghum halepense (L.) Pers.) Interference, Control and Recovery under Different Management Practices and its Effects on the Grain Yield and Quality of Maize Crop. Agronomy. 2020; 10(2):266. https://doi.org/10.3390/agronomy10020266
Chicago/Turabian StyleKarkanis, Anestis, Despoina Athanasiadou, Kyriakos Giannoulis, Konstantina Karanasou, Spyridon Zografos, Spyridon Souipas, Dimitrios Bartzialis, and Nicholaos Danalatos. 2020. "Johnsongrass (Sorghum halepense (L.) Pers.) Interference, Control and Recovery under Different Management Practices and its Effects on the Grain Yield and Quality of Maize Crop" Agronomy 10, no. 2: 266. https://doi.org/10.3390/agronomy10020266
APA StyleKarkanis, A., Athanasiadou, D., Giannoulis, K., Karanasou, K., Zografos, S., Souipas, S., Bartzialis, D., & Danalatos, N. (2020). Johnsongrass (Sorghum halepense (L.) Pers.) Interference, Control and Recovery under Different Management Practices and its Effects on the Grain Yield and Quality of Maize Crop. Agronomy, 10(2), 266. https://doi.org/10.3390/agronomy10020266






