Effects of Mycorrhizae on Physiological Responses and Relevant Gene Expression of Peach Affected by Replant Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Set-Up
2.2. Determinations of Variables
2.3. Statistical Analysis
3. Results
3.1. AMF Colonization, Total Plant Biomass, and Root Morphology
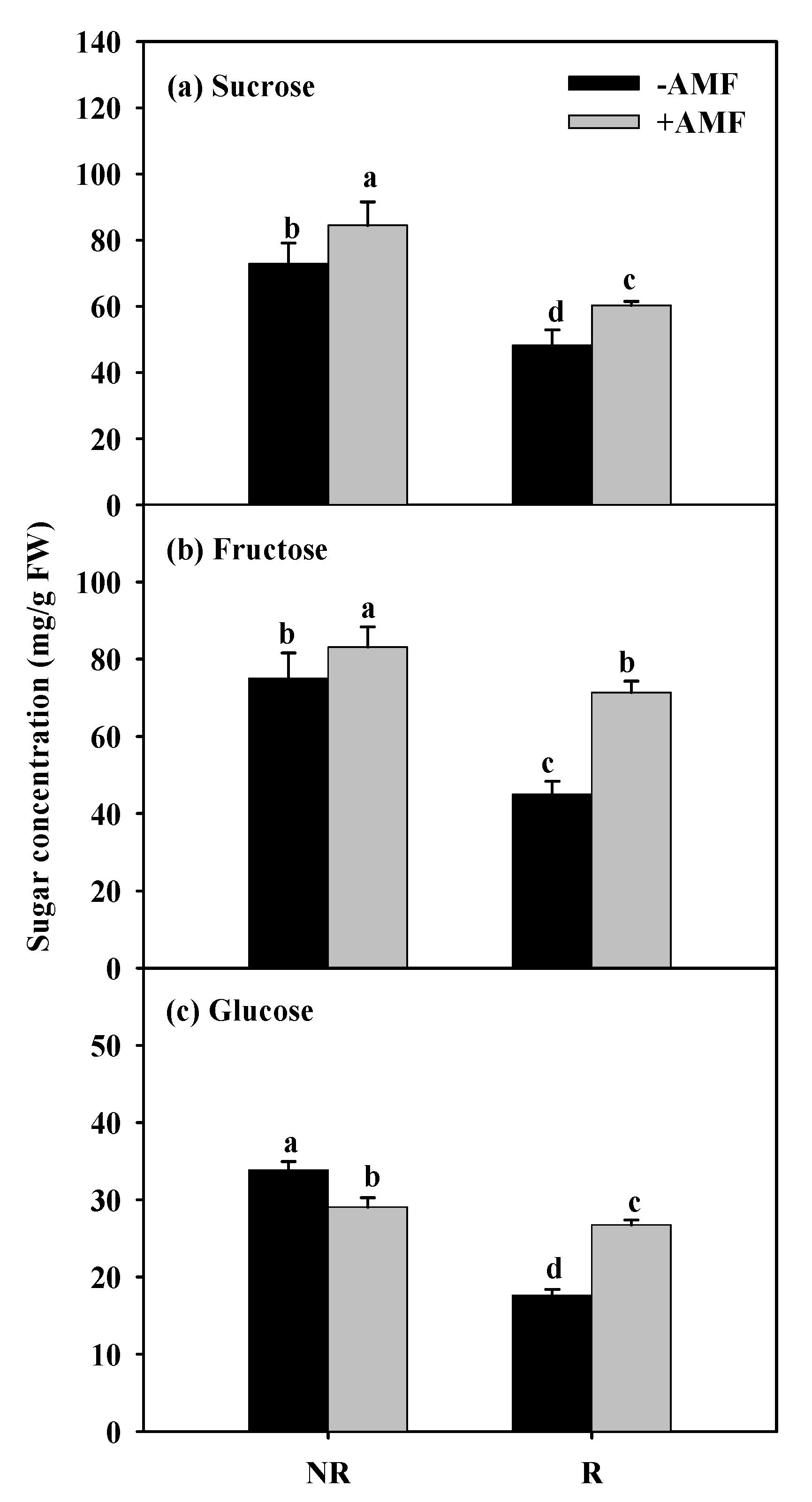
3.2. Changes in Root Carbohydrate Concentrations
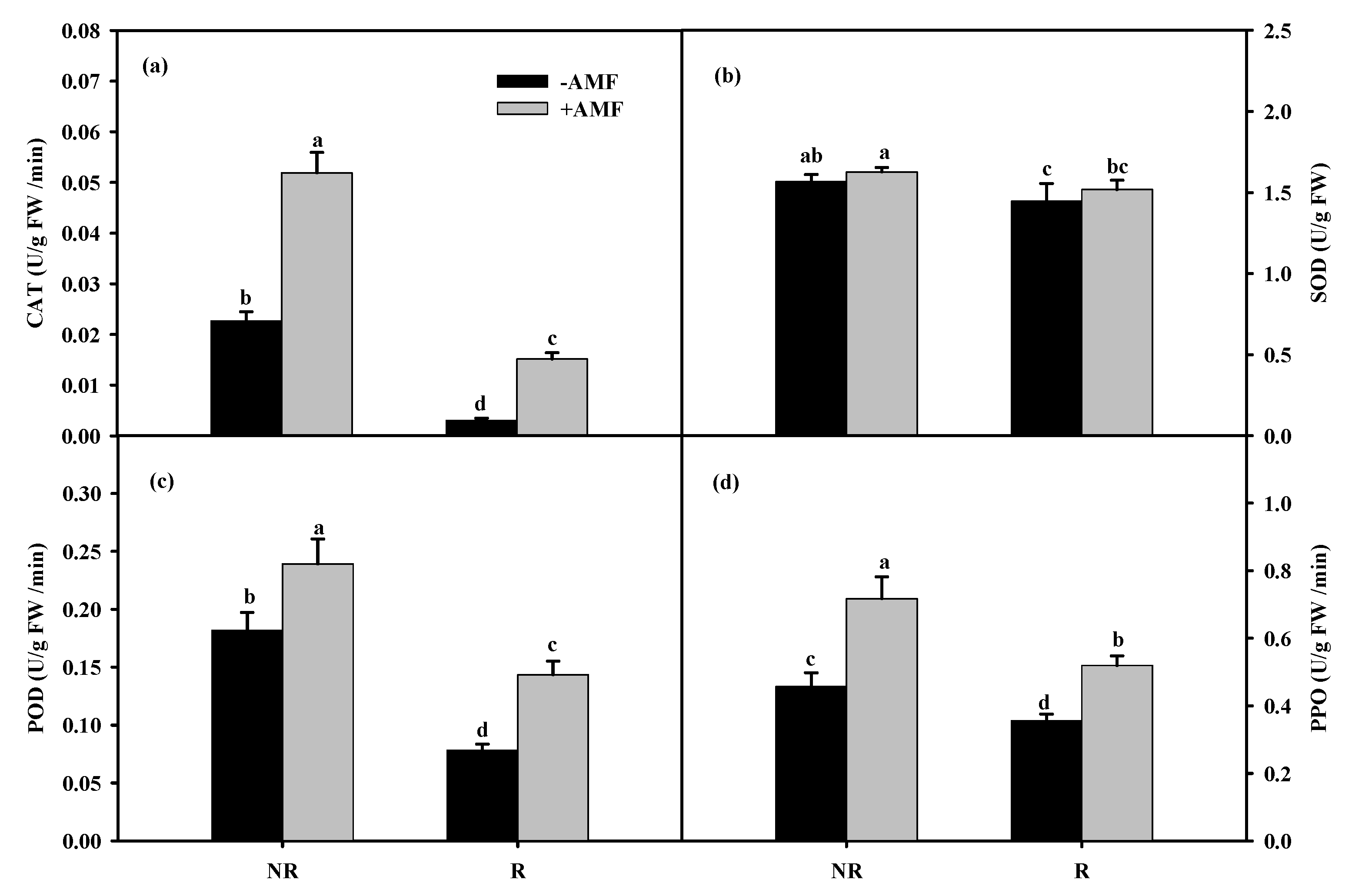
3.3. Changes in Root Antioxidant Enzyme Activities
3.4. Root Physiological Responses
3.5. Changes in Relative Expression Levels of Genes
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tewoldemedhin, Y.T.; Mazzola, M.; Labuschagne, I.; Mcleod, A. A multi-phasic approach reveals that apple replant disease is caused by multiple biological agents, with some agents acting synergistically. Soil Biol. Biochem. 2011, 43, 1917–1927. [Google Scholar] [CrossRef]
- Lü, L.H.; Srivastava, A.K.; Shen, Y.L.; Wu, Q.S. A negative feedback regulation of replanted soil microorganisms on plant growth and soil properties of peach. Not. Bot. Horti Agrobot. 2019, 47, 255–261. [Google Scholar] [CrossRef]
- Lü, L.H.; Wu, Q.S. Mitigation of replant disease by mycorrhization in horticultural plants: A review. Folia Hortic. 2018, 30, 269–282. [Google Scholar] [CrossRef]
- Spies, C.F.J.; Mazzola, M.; Mcleod, A. Characterisation and detection of Pythium and Phytophthora species associated with grapevines in South Africa. Eur. J. Plant Pathol. 2011, 131, 103–119. [Google Scholar] [CrossRef]
- Wu, Q.S.; He, J.D.; Srivastava, A.K.; Zou, Y.N.; Kuča, K. Mycorrhizas enhance drought tolerance of citrus by altering root fatty acid compositions and their saturation levels. Tree Physiol. 2019, 39, 1149–1158. [Google Scholar] [CrossRef]
- Zhang, Z.Z.; Lou, Y.G.; Deng, D.J.; Rahman, M.M.; Wu, Q.S. Effects of common mycorrhizal network on plant carbohydrates and soil properties in trifoliate orange–white clover association. PLoS ONE 2015, 10, e0142371. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Cai, B.Y. Advances in effects of AM fungi on metabolites of continuous cropping roots. Chin. Agric. Sci. Bull. 2018, 34, 35–39. [Google Scholar]
- Volpin, H.; Phillips, D.A.; Okon, Y.; Kapulnik, Y. Suppression of an isoflavonoid phytoalexin defense response in mycorrhizal alfalfa roots. Plant Physiol. 1995, 108, 1449–1454. [Google Scholar] [CrossRef]
- Čatska, V. Interrelationships between vesicular-arbuscular mycorrhiza and rhizosphere microflora in apple replant disease. Biol. Plant. 1994, 36, 99–104. [Google Scholar] [CrossRef]
- Mehta, P.; Bharat, N.K. Effect of indigenous arbuscular-mycorrhiza (Glomus spp.) on apple (Malus domestica) seedlings grown in replant disease soil. Indian J. Agric. Sci. 2013, 83, 1173–1178. [Google Scholar]
- Guo, X.W.; Li, K.; Guo, Y.S.; Zhang, L.H.; Sun, Y.N.; Xie, H.G. Effect of arbuscular mycorrhizal fungi (AMF) strains on growth and root exudation characteristics of grapevine. J. Shenyang Agric. Univ. 2009, 40, 392–395. [Google Scholar]
- Felton, G.W.; Korth, K.L. Trade-offs between pathogen and herbivore resistance. Curr. Opin. Plant Biol. 2000, 3, 309–314. [Google Scholar] [CrossRef]
- Song, Y.Y.; Zeng, R.S.; Xu, J.F.; Shen, X.; Yihdego, W.G. Interplant communication of tomato plants through underground common mycorrhizal networks. PLoS ONE 2010, 5, e13324. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.C.; Zou, Y.N.; Liu, L.P.; Wu, Q.S. Common mycorrhizal networks activate salicylic acid defense responses of trifoliate orange (Poncirus trifoliata). J. Integr. Plant Biol. 2019, 61, 1099–1111. [Google Scholar] [CrossRef]
- Vergne, E.; Grand, X.; Ballini, E.; Chalvon, V.; Saindrenan, P.; Tharreau, D. Preformed expression of defense is a hallmark of partial resistance to rice blast fungal pathogen Magnaporthe oryzae. BMC Plant Biol. 2010, 10, 206. [Google Scholar] [CrossRef]
- Bol, J.F.; van Kan, J.A. The synthesis and possible functions of virus-induced proteins in plants. Microbiol. Sci. 1988, 5, 47. [Google Scholar]
- Zhang, Y.C.; Liu, L.P.; Zou, Y.N.; Liu, C.Y.; Wu, Q.S. Responses of signal substances to canker in trifoliate orange roots trough mycorrhizal hyphal bridge. Mycosystema 2017, 36, 1028–1036. [Google Scholar]
- Phillips, J.M.; Hayman, D.S. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc. 1970, 55, 158–161. [Google Scholar] [CrossRef]
- Wu, Q.S.; Lou, Y.G.; Li, Y. Plant growth and tissue sucrose metabolism in the system of trifoliate orange and arbuscular mycorrhizal fungi. Sci. Hortic. 2015, 181, 189–193. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro on antioxidative defenses. Methods Enzymol. 1984, 105, 121–127. [Google Scholar]
- Beauchamp, C.; Fridovich, I. Superoxide dismutase: Improved assays and an assay applicable to acrylamide gels. Anal. Biochem. 1971, 44, 276–287. [Google Scholar] [CrossRef]
- Lurie, S.; Fallik, E.; Handros, A.; Shapira, R. The possible involvement of peroxidase in resistance to botrytis cinerea in heat treated tomato fruit. Physiol. Mol. Plant Pathol. 1997, 50, 141–149. [Google Scholar] [CrossRef]
- Aquino-Bolaños, E.N.; Mercado-Silva, E. Effects of polyphenol oxidase and peroxidase activity, phenolics and lignin content on the browning of cut jicama. Postharvest Biol. Technol. 2004, 33, 275–283. [Google Scholar] [CrossRef]
- Wang, X.K.; Huang, J.L. Principles and Techniques of Plant Physiological Biochemical Experiment; Higher Education Press: Beijing, China, 2015. [Google Scholar]
- Segarrad, G.; Jáuregui, O.; Casanova, E.; Trillas, I. Simultaneous quantitative LC-ESI-MS/MS analyses of salicylic acid and jasmonic acid in crude extracts of Cucumis sativus under biotic stress. Phytochemistry 2006, 67, 395–401. [Google Scholar] [CrossRef]
- Rodriguez-Kabana, R.; Godoy, G.; Morgan-Jones, G.; Shelby, R.A. The determination of soil chitinase activity: Conditions for assay and ecological studies. Plant Soil 1983, 75, 95–106. [Google Scholar] [CrossRef]
- Pedersen, J.F.; Vogel, K.P.; Funnell, D.L. Impact of reduced lignin on plant fitness. Crop Sci. 2005, 45, 812–819. [Google Scholar] [CrossRef]
- Kenneth, J.L.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and 2−ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar]
- Gao, L.; Wang, Y.T.; Li, Z.; Zhang, H.; Ye, J.L.; Li, G.H. Gene expression changes during the gummosis development of peach shoots in response to Lasiodiplodia theobramae infection using RNA-seq. Front. Physiol. 2016, 7, 170. [Google Scholar] [CrossRef]
- Tong, Z.G.; Gao, Z.H.; Wang, F.; Zhou, J.; Zhang, Z. Selection of reliable reference genes for gene expression studies in peach using real-time PCR. BMC Mol. Biol. 2009, 10, 71. [Google Scholar] [CrossRef]
- Zhang, Z.Z.; Wu, Q.S.; Li, G.H. Effects of arbuscular mycorrhizal fungi on growth and rhizospheric soil structure of Prunus persica. South. Chin. Fruits 2014, 43, 14–17. [Google Scholar]
- Zhang, Z.Z.; Srivastava, A.K.; Wu, Q.S.; Li, G.H. Growth performance and rhizospheric traits of peach (Prunus persica) in response to mycorrhization on replant versus non-replant soil. Indian J. Agric. Sci. 2015, 85, 125–130. [Google Scholar]
- Trindade, A.V.; Siqueira, J.O.; Stürmer, S.L. Arbuscular mycorrhizal fungi in papaya plantations of Espírito Santo and Bahia, Brazil. Braz. J. Microbiol. 2006, 37, 283–289. [Google Scholar] [CrossRef]
- Qi, G.H.; Chen, G.L.; Lv, G.Y.; Nie, L.C.; Ding, P.H. Effects of arbuscular mycorrhizal fungi on the yield and quality of strawberry grown in replanted soil. J. Fruit Sci. 2001, 18, 341–344. [Google Scholar]
- Gastoł, M.; Domagałaświatkiewicz, I. Mycorrhizal inoculation of apple in replant soils-Enhanced tree growth and mineral nutrient status. Acta Sci. Pol. Hortorum Cultus 2015, 14, 17–37. [Google Scholar]
- Li, M.; Wang, W.H.; Liu, R.J. Influences of arbuscular mycorrhizal fungi and Fusarium oxysporumf.sp. niveum on lipid peroxidation and membrane permeability in watermelon roots. Acta Phytopathol. Sin. 2003, 33, 229–232. [Google Scholar]
- Chen, S.; Jin, W.; Liu, A.; Zhang, S.; Liu, D.; Wang, F.; Lin, X.; He, C. Arbuscular mycorrhizal fungi (AMF) increase growth and secondary metabolism in cucumber subjected to low temperature stress. Sci. Hortic. 2013, 160, 222–229. [Google Scholar] [CrossRef]
- Tian, L.; Li, Y.; Wu, Q.S. Exogenous carbon magnifies mycorrhizal effects on growth behaviour and sucrose metabolism in trifoliate orange. Not. Bot. Horti Agrobot. 2018, 46, 365–370. [Google Scholar] [CrossRef]
- Lopez-Raez, J.A.; Verhage, A.; Fernandez, I.; Garcia, J.M.; Azcon-Aguilar, C.; Flors, V. Hormonal and transcriptional profiles highlight common and differential host responses to arbuscular mycorrhizal fungi and the regulation of the oxylipin pathway. J. Exp. Bot. 2010, 61, 2589–2601. [Google Scholar] [CrossRef]

| Gene Name | Gene Description | ID in GDR Database | Primer Sequence (5′→3′) |
|---|---|---|---|
| TEF2 | translation elongation factor 2 | JQ732180 | F: AGCAAGCACCCAACAAGCATA |
| R: CCAACCAAACTCTTCAGCCAAT | |||
| PpPAL1 | phenylalanine ammonia-lyase 1 | ppa002099m | F: ACCTCCCACAGAAGAACAAAG |
| R: CAAATCTTATGCCAGAGTAGCC | |||
| Pp4CL3 | 4-coumarate-CoA ligase 3 | ppa022401m | F:GCCGCAGGGAAAGGAGTT |
| R: GGTTGTAGCCAAGGGAGCA | |||
| PpCHI | chitinase | ppa008859m | F:TGCTGCTGCTCGGACTTT |
| R: TATTGGGCGGATGGTGTA | |||
| PpAOC3 | allene oxide cyclase | ppa010397m | F:ACTCGGTGACCTTGTTCCA |
| R: GCCCAATCAGTGTCCTCGTAA | |||
| PpAOC4 | allene oxide cyclase | ppa012079m | F:CGTATCTGGCTGTGACTGGT |
| R: GAAGTTGGAGATTGTGGCTTGA | |||
| PpLOX1 | linoleate 9S lipoxygenase 1 | ppa001293m | F:CCCAACCGCCCAACTATAAG |
| R: AGGAGTGTCTCTCTGCCCCA | |||
| PpLOX5 | lipoxygenase | ppa001207m | F:CGACGAGGTCCACAGTGATAC |
| R: GTTAGGGAGGAAGCCAGCATA | |||
| PpOPR2 | 12-oxophytodienoate reductase 2 | ppa007490m | F: GCCGAGAATGAGGACAGT |
| R: AGAACCAACGACCAAAGG |
| Treatments | Mycorrhizal Colonization (%) | Total Plant Biomass (g FW/Plant) | Root Morphology | |||
|---|---|---|---|---|---|---|
| Total Length (cm) | Surface Area (cm2) | Projected Area (cm2) | Volume (cm3) | |||
| NR − AMF | 0.0 ± 0.0c | 7.37 ± 0.40b | 532 ± 33b | 69.1 ± 5.8b | 21.3 ± 1.6b | 0.72 ± 0.04b |
| NR + AMF | 52.0 ± 4.4a | 9.73 ± 0.46a | 616 ± 16a | 84.1 ± 1.1a | 26.8 ± 0.4a | 0.91 ± 0.06a |
| R−AMF | 0.0 ± 0.0c | 5.86 ± 0.24c | 435 ± 43c | 64.2 ± 3.5b | 21.2 ± 1.6b | 0.72 ± 0.04b |
| R + AMF | 29.8 ± 2.6b | 7.25 ± 0.56b | 484 ± 35bc | 66.6 ± 5.8b | 20.4 ± 1.5b | 0.76 ± 0.10ab |
| Treatments | SA (pmol/g FW) | JA (pmol/g FW) | PAL Activity (U/g FW) | Chitinase Activity (U/g FW) | Total Soluble Phenol (μg/g FW) | Lignin (mg/g FW) |
|---|---|---|---|---|---|---|
| NR − AMF | 59.65 ± 3.69b | 60.95 ± 5.20c | 6.31 ± 0.29b | 11.99 ± 0.13b | 111.3 ± 2.3a | 43.2 ± 6.8a |
| NR + AMF | 71.81 ± 6.36a | 98.30 ± 2.10a | 7.31 ± 0.52a | 13.21 ± 0.33a | 100.4 ± 2.7b | 32.5 ± 5.0bc |
| R − AMF | 54.97 ± 3.27b | 72.23 ± 3.67b | 1.91 ± 0.43c | 8.74 ± 0.53c | 94.4 ± 4.0c | 26.2 ± 2.2c |
| R + AMF | 67.75 ± 1.46a | 94.02 ± 4.49a | 7.24 ± 0.36a | 13.41 ± 0.67a | 103.4 ± 2.4b | 37.9 ± 5.9ab |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, W.-Q.; Lü, L.-H.; Srivastava, A.K.; Wu, Q.-S.; Kuča, K. Effects of Mycorrhizae on Physiological Responses and Relevant Gene Expression of Peach Affected by Replant Disease. Agronomy 2020, 10, 186. https://doi.org/10.3390/agronomy10020186
Gao W-Q, Lü L-H, Srivastava AK, Wu Q-S, Kuča K. Effects of Mycorrhizae on Physiological Responses and Relevant Gene Expression of Peach Affected by Replant Disease. Agronomy. 2020; 10(2):186. https://doi.org/10.3390/agronomy10020186
Chicago/Turabian StyleGao, Wei-Qin, Li-Hui Lü, A. K. Srivastava, Qiang-Sheng Wu, and Kamil Kuča. 2020. "Effects of Mycorrhizae on Physiological Responses and Relevant Gene Expression of Peach Affected by Replant Disease" Agronomy 10, no. 2: 186. https://doi.org/10.3390/agronomy10020186
APA StyleGao, W.-Q., Lü, L.-H., Srivastava, A. K., Wu, Q.-S., & Kuča, K. (2020). Effects of Mycorrhizae on Physiological Responses and Relevant Gene Expression of Peach Affected by Replant Disease. Agronomy, 10(2), 186. https://doi.org/10.3390/agronomy10020186








