Growing Medium Type Affects Organic Fertilizer Mineralization and CNPS Microbial Enzyme Activities
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design and Treatments
| Growing Media | Fertilizers | |||||
|---|---|---|---|---|---|---|
| Peat | Coir | Composted Bark | F1 | F2 | Horn | |
| Feedstock nature and origin | Sphagnum dark peat, H5 [42], Ireland | Coconut mesocarp (pith), Sri Lanka | Pinus bark, France | Plant based | Plant based | Animal based |
| Form and size | Fine fractions below 5 mm | Dried granular (<2 mm) | Dried meal (<5 mm) | |||
| Physical properties | ||||||
| Total porosity 1 (%) | 96 ± 0.06 | 96 ± 0.07 | 92 ± 0.05 | - | - | - |
| WHC 2 (%) | 75 ± 1 | 84 ± 2 | 55 ± 1 | - | - | - |
| AFP 3 (%) | 21 ± 1 | 12 ± 2 | 37 ± 0.6 | - | - | - |
| EAW 4 (%) | 33 ± 1 | 45 ± 2 | 17 ± 0.3 | - | - | - |
| Bulk density 5 (g cm−3) | 0.07 ± 0.002 | 0.06 ± 0.0 | 0.18 ± 0.001 | - | - | - |
| Chemical properties | ||||||
| pH 6 (water) | 4 ± 0.06 | 6.5 ± 0.0 | 6.1 ± 0.02 | - | - | - |
| EC 7 (µS cm−1) | 35 ± 0.3 | 75 ± 1.7 | 113 ± 2 | - | - | - |
| CEC 8 (cmol+ kg−1) | 48 | 55 | 50 | - | - | - |
| OM 9 (%) | 98 ± 0.2 | 92 ± 0.3 | 88 ± 0.5 | 78 ± 0.2 | 75 ± 0.1 | 83 ± 0.4 |
| Elemental composition 10,11 (g kg−1 dry mass) | ||||||
| C | 516 ± 5 | 453 ± 13 | 450 ± 0.6 | 401 ± 0.2 | 404 ± 9 | 436 ± 20 |
| N | 9.9 ± 0.1 | 5.8 ± 0.1 | 5.8 ± 0.3 | 72 ± 1 | 32 ± 0.5 | 136.3 ± 5 |
| P | 0.19 | 0.3 | 0.74 | 16 | 5.6 | 26 |
| S | 2.52 | 0.94 | 0.78 | 20 | 20 | 18 |
| C:N ratio | 52 | 78 | 78 | 5.5 | 13 | 3.2 |
| C:P ratio | 2716 | 1512 | 608 | 25 | 72 | 17 |
| C:S ratio | 205 | 482 | 577 | 20 | 20 | 24 |
| N:P ratio | 52 | 19 | 7.8 | 4.4 | 5.6 | 5.3 |
| N:S ratio | 3.9 | 6.2 | 7.4 | 3.6 | 1.5 | 7.4 |
| P:S ratio | 0.08 | 0.32 | 0.95 | 0.82 | 0.27 | 1.4 |
2.2. Measurement during Incubation
2.2.1. Chemical Analyses
2.2.2. Enzyme Activity Assays
2.2.3. Statistical Analyses
3. Results
3.1. Growing Media and Fertilizer Effects on pH and Electrical Conductivity
3.2. Effect of Growing Media and Fertilizer Types on Nutrient Dynamics
3.3. Effect of Growing Media and Fertilizer Types on Enzyme Activities
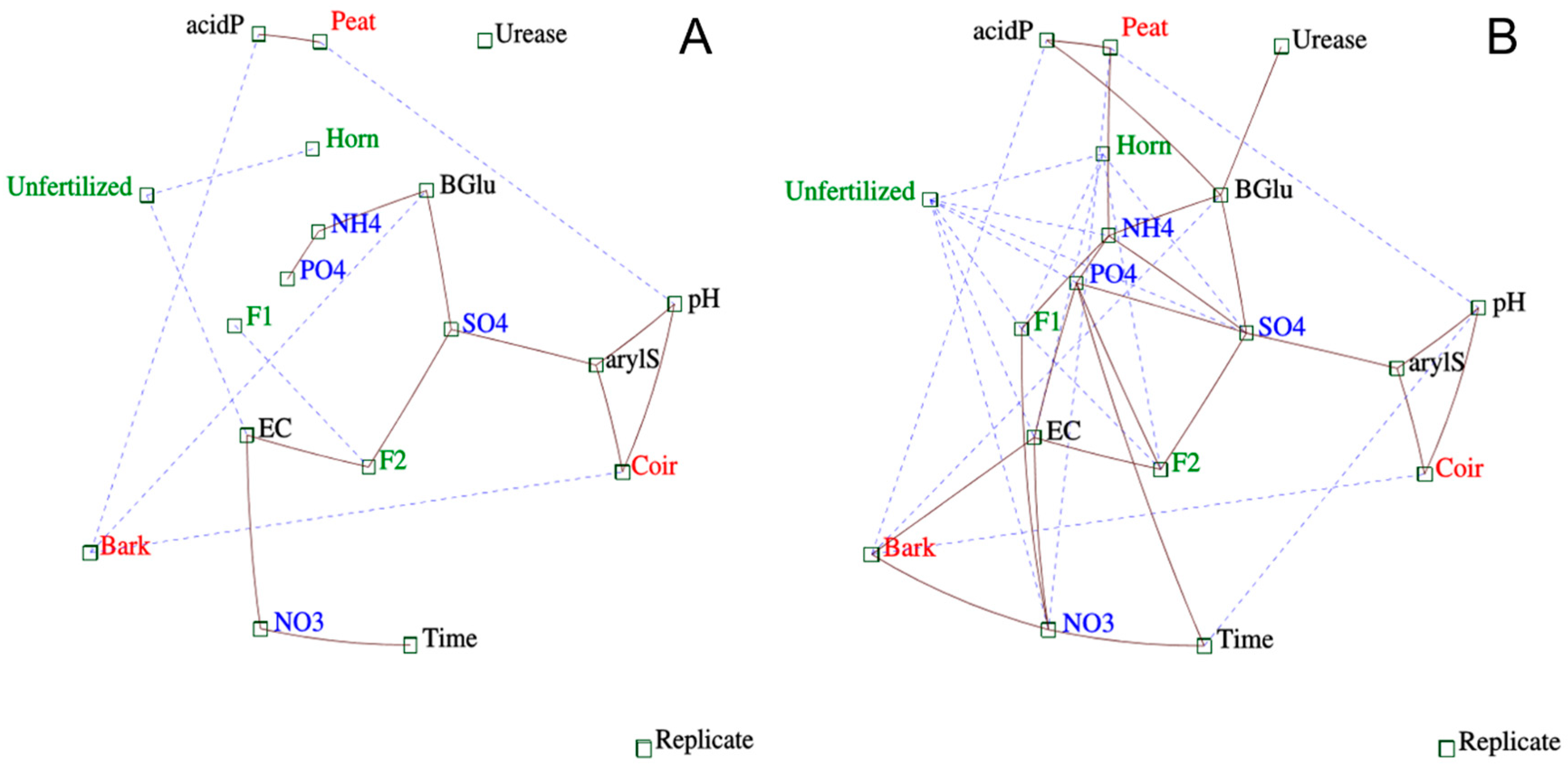
3.4. Multivariate and Correlation Analyses between Nutrient Releases and Enzyme Activities
4. Discussion
4.1. Nutrient Availability from Fertilizer Mineralization Is Driven by the GM Type
4.2. The Role of Fertilizer’s Biochemical Composition on Nutrient Availability
4.3. Enzyme Activities Are Driven by the GM Type
4.4. Remarkable Relationships between Enzyme Activities and Nutrient Availabilities in Each Growing Media
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carlile, W.R.; Raviv, M.; Prasad, M. Organic Soilless Media Components. In Soilless Culture, 2nd ed.; Raviv, M., Lieth, J.H., Bar-Tal, A., Eds.; Elsevier: Boston, MA, USA, 2019; pp. 303–378. ISBN 978-0-444-63696-6. [Google Scholar]
- Zulfiqar, F.; Allaire, S.E.; Akram, N.A.; Méndez, A.; Younis, A.; Peerzada, A.M.; Shaukat, N.; Wright, S.R. Challenges in organic component selection and biochar as an opportunity in potting substrates: A review. J. Plant Nutr. 2019, 42, 1386–1401. [Google Scholar] [CrossRef]
- Bohlin, C.; Holmberg, P. Peat: Dominating growing medium in swedish horticulture. In Proceedings of the International Symposium on Growing Media and Hydroponics, Alnarp, Sweden, 8 September 2001; Acta Horticulturae. International Society for Horticultural Science (ISHS): Leuven, Belgium, 2004; Volume 644, pp. 177–181. [Google Scholar]
- Gruda, N.S. Increasing Sustainability of Growing Media Constituents and Stand-Alone Substrates in Soilless Culture Systems. Agronomy 2019, 9, 298. [Google Scholar] [CrossRef]
- Carlile, B.; Coules, A. Towards sustainability in growing media. In Proceedings of the International Symposium on Growing Media, Composting and Substrate Analysis, Barcelona, Spain, 21 October 2011; Acta Horticulturae. International Society for Horticultural Science (ISHS): Leuven, Belgium, 2013; Volume 1013, pp. 341–349. [Google Scholar]
- Blok, C. Growing media for food and quality of life in the period 2020–2050. In Acta Horticulturae; International Society for Horticultural Science (ISHS): Leuven, Belgium, 2020. (In press)
- Clarke, D. The wise use of peat in horticulture. In Proceedings of the the International Symposium on Growing Media, Angers, France, 4 September 2005; Acta Horticulturae. International Society for Horticultural Science (ISHS): Leuven, Belgium, 2008; Volume 779, pp. 161–164. [Google Scholar]
- Barrett, G.E.; Alexander, P.D.; Robinson, J.S.; Bragg, N.C. Achieving environmentally sustainable growing media for soilless plant cultivation systems—A review. Sci. Hortic. 2016, 212, 220–234. [Google Scholar] [CrossRef]
- Burnett, S.E.; Mattson, N.S.; Williams, K.A. Substrates and fertilizers for organic container production of herbs, vegetables, and herbaceous ornamental plants grown in greenhouses in the United States. Sci. Hortic. 2016, 208, 111–119. [Google Scholar] [CrossRef]
- Treadwell, D.D.; Hochmuth, G.J.; Hochmuth, R.C.; Simonne, E.H.; Davis, L.L.; Laughlin, W.L.; Li, Y.; Olczyk, T.; Sprenkel, R.K.; Osborne, L.S. Nutrient management in organic greenhouse herb production: Where are we now? Horttechnology 2007, 17, 461–466. [Google Scholar] [CrossRef]
- Carlile, W.R.; Wilson, D.P. Microbial activity in growing media—A brief review. In Proceedings of the II Symposium on Horticultural Substrates and their Analysis, XXIII IHC, Guernsey–Firenze, UK–Italy, 10 September 1990; Acta Horticulturae. International Society for Horticultural Science (ISHS): Leuven, Belgium, 1991; Volume 294, pp. 197–206. [Google Scholar]
- Caldwell, B.A. Enzyme activities as a component of soil biodiversity: A review. Pedobiologia 2005, 49, 637–644. [Google Scholar] [CrossRef]
- Prosser, J.I.; Nicol, G.W. Archaeal and bacterial ammonia-oxidisers in soil: The quest for niche specialisation and differentiation. Trends Microbiol. 2012, 20, 523–531. [Google Scholar] [CrossRef]
- Prosser, J.I. Autotrophic Nitrification in Bacteria. In Advances in Microbial Physiology; Rose, A.H., Tempest, D.W., Eds.; Elsevier: Boston, MA, USA, 1990; Volume 30, pp. 125–181. [Google Scholar]
- Norton, J.; Ouyang, Y. Controls and Adaptive Management of Nitrification in Agricultural Soils. Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef]
- Hoitink, H.; Boehm, M. Biocontrol within the context of soil microbial communities: A substrate-dependent phenomenon. Annu. Rev. Phytopathol. 1999, 37, 427–446. [Google Scholar] [CrossRef]
- Montagne, V.; Capiaux, H.; Cannavo, P.; Charpentier, S.; Renaud, S.; Liatard, E.; Grosbellet, C.; Lebeau, T. Protective effect of organic substrates against soil-borne pathogens in soilless cucumber crops. Sci. Hortic. 2016, 206, 62–70. [Google Scholar] [CrossRef]
- Fascella, G.; Mammano, M.M.; D’Angiolillo, F.; Pannico, A.; Rouphael, Y. Coniferous wood biochar as substrate component of two containerized Lavender species: Effects on morpho-physiological traits and nutrients partitioning. Sci. Hortic. 2020, 267, 109356. [Google Scholar] [CrossRef]
- Lévesque, V.; Rochette, P.; Ziadi, N.; Dorais, M.; Antoun, H. Mitigation of CO2, CH4 and N2O from a fertigated horticultural growing medium amended with biochars and a compost. Appl. Soil Ecol. 2018, 126, 129–139. [Google Scholar] [CrossRef]
- Domeño, I.; Irigoyen, I.; Muro, J. Comparison of traditional and improved methods for estimating the stability of organic growing media. Sci. Hortic. 2011, 130, 335–340. [Google Scholar] [CrossRef]
- Aendekerk, T.G.L. Decomposition of peat substrates in relation to physical properties and growth of chamaecyparis. In Proceedings of the International Symposium Growing Media and Plant Nutrition in Horticulture, Freising, Germany, 2 September 1996; Acta Horticulturae. International Society for Horticultural Science (ISHS): Leuven, Belgium, 1997; Volume 450, pp. 191–198. [Google Scholar]
- Prasad, M. Physical, chemical and biological properties of coir dust. In Proceedings of the International Symposium Growing Media and Plant Nutrition in Horticulture, Freising, Germany, 2 September 1996; Acta Horticulturae. International Society for Horticultural Science (ISHS): Leuven, Belgium, 1997; Volume 450, pp. 21–30. [Google Scholar]
- Naasz, R.; Michel, J.C.; Charpentier, S. Microbial respiration and its consequences on oxygen availability in peat substrate. In Proceedings of the International Symposium on Growing Media, Angers, France, 4 September 2005; Acta Horticulturae 779. International Society for Horticultural Science (ISHS): Leuven, Belgium, 2008; Volume 779, pp. 91–96. [Google Scholar]
- Verhagen, J.B.G.M. Stability of growing media from a physical, chemical and biological perspective. In Proceedings of the International Symposium on Growing Media, Nottingham, UK, 2–8 September 2007; Acta Horticulturae. International Society for Horticultural Science (ISHS): Leuven, Belgium, 2009; Volume 819, pp. 135–142. [Google Scholar]
- Montagne, V.; Charpentier, S.; Cannavo, P.; Capiaux, H.; Grosbellet, C.; Lebeau, T. Structure and activity of spontaneous fungal communities in organic substrates used for soilless crops. Sci. Hortic. 2015, 192, 148–157. [Google Scholar] [CrossRef]
- Carlile, W.R.; Dickinson, K. Dehydrogenase as an indicator of microbial activity in growing media. In Proceedings of the International Symposium on Growing Media and Hydroponics, Alnarp, Sweden, 8 September 2001; Acta Horticulturae. International Society for Horticultural Science (ISHS): Leuven, Belgium, 2004; Volume 644, pp. 517–523. [Google Scholar]
- Handreck, K.A. Rapid assessment of the rate of nitrogen immobilisation in organic components of potting media: I. Method development. Commun. Soil Sci. Plant Anal. 1992, 23, 201–215. [Google Scholar] [CrossRef]
- Handreck, K.A. Rapid assessment of the rate of nitrogen immobilisation in organic components of potting media: II. Nitrogen drawdown index and plant growth. Commun. Soil Sci. Plant Anal. 1992, 23, 217–230. [Google Scholar] [CrossRef]
- Handreck, K.A. Phosphorus immobilization in wood waste-based potting media. Commun. Soil Sci. Plant Anal. 1996, 27, 2295–2314. [Google Scholar] [CrossRef]
- Plante, A.F.; Stone, M.M.; McGill, W.B. The Metabolic Physiology of Soil Microorganisms. In Soil Microbiology, Ecology and Biochemistry, 4th ed.; Paul, E.A., Ed.; Elsevier: Boston, MA, USA, 2015; pp. 245–272. ISBN 978-0-12-415955-6. [Google Scholar]
- Sinsabaugh, R.L.; Lauber, C.L.; Weintraub, M.N.; Ahmed, B.; Allison, S.D.; Crenshaw, C.; Contosta, A.R.; Cusack, D.; Frey, S.; Gallo, M.E.; et al. Stoichiometry of soil enzyme activity at global scale. Ecol. Lett. 2008, 11, 1252–1264. [Google Scholar] [CrossRef]
- Mooshammer, M.; Wanek, W.; Zechmeister-Boltenstern, S.; Richter, A. Stoichiometric imbalances between terrestrial decomposer communities and their resources: Mechanisms and implications of microbial adaptations to their resources. Front. Microbiol. 2014, 5. [Google Scholar] [CrossRef]
- Burns, R.G.; DeForest, J.L.; Marxsen, J.; Sinsabaugh, R.L.; Stromberger, M.E.; Wallenstein, M.D.; Weintraub, M.N.; Zoppini, A. Soil enzymes in a changing environment: Current knowledge and future directions. Soil Biol. Biochem. 2013, 58, 216–234. [Google Scholar] [CrossRef]
- Grunert, O.; Robles-Aguilar, A.A.; Hernandez-Sanabria, E.; Schrey, S.D.; Reheul, D.; Labeke, M.-C.V.; Vlaeminck, S.E.; Vandekerckhove, T.G.L.; Mysara, M.; Monsieurs, P.; et al. Tomato plants rather than fertilizers drive microbial community structure in horticultural growing media. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Nannipieri, P.; Giagnoni, L.; Renella, G.; Puglisi, E.; Ceccanti, B.; Masciandaro, G.; Fornasier, F.; Moscatelli, M.C.; Marinari, S. Soil enzymology: Classical and molecular approaches. Biol. Fertil. Soils 2012, 48, 743–762. [Google Scholar] [CrossRef]
- Grunert, O.; Reheul, D.; Van Labeke, M.; Perneel, M.; Hernandez-Sanabria, E.; Vlaeminck, S.E.; Boon, N. Growing media constituents determine the microbial nitrogen conversions in organic growing media for horticulture. Microb. Biotechnol. 2016, 9, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Aldén, L.; Demoling, F.; Bååth, E. Rapid Method of Determining Factors Limiting Bacterial Growth in Soil. Appl. Environ. Microbiol. 2001, 67, 1830–1838. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.B.; Spurway, M.I.; Stewart, D.P.C. A review of factors influencing organic matter decomposition and nitrogen immobilisation in container media. In The International Plant Propagators’ Society Combined Proceedings; The International Plant Propagators’ Society: Carlisle, PA, USA, 1999; Volume 48, pp. 66–71. [Google Scholar]
- Van Gerrewey, T.; Ameloot, N.; Navarrete, O.; Vandecruys, M.; Perneel, M.; Boon, N.; Geelen, D. Microbial activity in peat-reduced plant growing media: Identifying influential growing medium constituents and physicochemical properties using fractional factorial design of experiments. J. Clean. Prod. 2020, 256, 120323. [Google Scholar] [CrossRef]
- Thuriès, L.; Larré-Larrouy, M.-C.; Pansu, M. Evaluation of three incubation designs for mineralization kinetics of organic materials in soil. Commun. Soil Sci. Plant Anal. 2000, 31, 289–304. [Google Scholar] [CrossRef]
- Voroney, R.P.; Heck, R.J. The Soil Habitat. In Soil Microbiology, Ecology, and Biochemistry, 4th ed.; Paul, E.A., Ed.; Elsevier: Boston, MA, USA, 2015; pp. 14–40. ISBN 978-0-12-415955-6. [Google Scholar]
- Von Post, L. Sveriges Geologiska Undersöknings torvinventering och några av dess hittills vunna resultat. Svenska mosskulturföreningens tidskrift 1922, 36, 1–27. [Google Scholar]
- Bunt, A.C. Physical properties of mixtures of peats and minerals of different particle size and bulk density for potting substrates. In Proceedings of the International Symposium on Substrates in Horticulture other than Soils In Situ, Barcelona, Spain, 28 August 1983; Acta Horticulturae. International Society for Horticultural Science (ISHS): Leuven, Belgium, 1984; Volume 150, pp. 143–154. [Google Scholar]
- Caron, J.; Parent, L.E.; Elrick, D.E.; Michel, J.C.; Naasz, R. Physical properties of organic soils and growing media: Water and air storage and flow dynamics. In Soil Sampling and Methods of Analysis, 2nd ed.; Carter, M.R., Gregorich, E.G., Eds.; CRC Press: Boca Raton, FL, USA, 2008; pp. 885–912. ISBN 978-0-8493-3586-0. [Google Scholar]
- AFNOR Amendements du Sol et Supports de Culture—Détermination des Propriétés Physiques—Masse Volumique Apparente Sèche, Volume D’air, Volume d’eau, Valeur de Rétraction et Porosité Totale. NF EN 13041. U44-307. 2000. Available online: https://www.boutique.afnor.org/normes (accessed on 10 March 2018).
- AFNOR Amendements Organiques et Supports de Culture—Préparation des Echantillons Pour les Essais Physiques et Chimiques, Détermination de la Teneur en Matière Sèche, du Taux D’humidité et de la Masse Volumique Compactée en Laboratoire. NF EN 13040. U44-306. 2000. Available online: https://www.boutique.afnor.org/normes (accessed on 10 March 2018).
- AFNOR Amendements du Sol et Supports de Culture—Détermination du pH. NF EN 13037. U44-308. 2000. Available online: https://www.boutique.afnor.org/normes (accessed on 10 March 2018).
- AFNOR Amendements du Sol et Supports de Culture—Détermination de la Conductivité Electrique. NF EN 13038. U44-309. 2000. Available online: https://www.boutique.afnor.org/normes (accessed on 10 March 2018).
- AFNOR Qualité du Sol—Determination de la Capacité D’échange Cationique (CEC) Effective et des Cations Echangeables à L’aide D’une Solution de Trichlorure de Cobaltihexammine. NF EN ISO 23470. X31-440. 2011. Available online: https://www.boutique.afnor.org/normes (accessed on 10 March 2018).
- AFNOR Amendements du Sol et Supports de Culture—Détermination de la Teneur en Matieres Organiques. NF EN 13039. U44-304. 2000. Available online: https://www.boutique.afnor.org/normes (accessed on 10 March 2018).
- AFNOR Qualité du Sol—Détermination de la Teneur Totale en Azote par Combustion Sèche. NF ISO 13878. X31-418. 1998. Available online: https://www.boutique.afnor.org/normes (accessed on 10 March 2018).
- AFNOR Qualité de L’eau—Dosage D’éléments Choisis par Spectroscopie D’émission Optique avec Plasma Induit par Haute Fréquence (ICP-OES). NF EN ISO 11885. T90-136. 2009. Available online: https://www.boutique.afnor.org/normes (accessed on 10 March 2018).
- Keeney, D.R.; Nelson, D.W. Nitrogen—Inorganic Forms. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties, 1st ed.; Page, A.L., Ed.; Agronomy Monographs; Soil Science Society of America, Inc.: Madison, WI, USA, 1982; pp. 643–698. ISBN 0-89118-072-9. [Google Scholar]
- Mulvaney, R.L. Nitrogen—Inorganic Forms. In Methods of Soil Analysis: Part 3, Chemical Methods; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnson, C.T., Sumner, M.E., Eds.; SSSA Book Series No. 5; American Society of Agronomy, Inc.: Madison, WI, USA, 1996; pp. 1123–1184. ISBN 978-0-89118-825-4. [Google Scholar]
- Murphy, J.; Riley, J.P. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Tabatabai, M.A. A Rapid Method for Determination of Sulfate in Water Samples. Environ. Lett. 1974, 7, 237–243. [Google Scholar] [CrossRef]
- Sörbo, B. Sulfate: Turbidimetric and nephelometric methods. In Methods in Enzymology; Sulfur and Sulfur Amino Acids; Elsevier: Boston, MA, USA, 1987; Volume 143, pp. 3–6. [Google Scholar]
- Eivazi, F.; Tabatabai, M.A. Glucosidases and galactosidases in soils. Soil Biol. Biochem. 1988, 20, 601–606. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol. Biochem. 1969, 1, 301–307. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Arylsulfatase Activity of Soils. Soil Sci. Soc. Am. J. 1970, 34, 225–229. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Assay of urease activity in soils. Soil Biol. Biochem. 1972, 4, 479–487. [Google Scholar] [CrossRef]
- TIBCO Software Inc. Statistica (Logiciel D’analyse des Données), 13 ed.; 2017; Available online: http://statistica.io (accessed on 10 July 2019).
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. Available online: https://ggplot2.tidyverse.org (accessed on 10 July 2019).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 10 July 2019).
- Anderson, M.J.; Gorley, R.N.; Clarke, K.R. PERMANOVA+ for PRIMER. Guide to Software and Statistical Methods; Primer-E Plymouth: Plymouth, UK, 2008. [Google Scholar]
- Legendre, P.; Anderson, M.J. Distance-Based Redundancy Analysis: Testing Multispecies Responses in Multifactorial Ecological Experiments. Ecol. Monogr. 1999, 69, 1–24. [Google Scholar] [CrossRef]
- Lesty, M. Analyse Informatique de Données Géochimiques sur les Echanges Air-Mer. Thèse de doctorat (PhD. Thesis), Université de Paris VII, Paris, France, 1981. [Google Scholar]
- Lesty, M. Une nouvelle approche dans le choix des régresseurs de la régression multiple en presence d’intéractions et de colinéarités. Modulad 1999, 22, 41–77. [Google Scholar]
- Engrand, C.; Lespagnol, J.; Martin, P.; Thirkell, L.; Thomas, R. Multi-correlation analyses of TOF-SIMS spectra for mineralogical studies. Appl. Surf. Sci. 2004, 231–232, 883–887. [Google Scholar] [CrossRef]
- Lesty, C.; Pleau-Varet, J.; Kujas, M. Geometric Method and Generalized Linear Models. Two Opposite Multiparametric Approaches Illustrated on a Sample of Pituitary Adenomas. J. Appl. Stat. 2004, 31, 191–213. [Google Scholar] [CrossRef]
- Agehara, S.; Warncke, D.D. Soil Moisture and Temperature Effects on Nitrogen Release from Organic Nitrogen Sources. Soil Sci. Soc. Am. J. 2005, 69, 1844–1855. [Google Scholar] [CrossRef]
- Handreck, K.A.; Black, N.D. Growing Media for Ornamental Plants and Turf; Revised Edition; 1989; New South Wales University Press: Kensington, Australia, 1984; ISBN 0 86840 177 3. [Google Scholar]
- Comerford, N.B. Soil Factors Affecting Nutrient Bioavailability. In Nutrient Acquisition by Plants: An Ecological Perspective; BassiriRad, H., Ed.; Ecological Studies; Springer: Berlin/Heidelberg, Germany, 2005; pp. 1–14. ISBN 978-3-540-27675-3. [Google Scholar]
- Caron, J.; Rivière, L.M.; Parent, L.E.; Ilnicki, P. Quality of peat substrates for plants grown in containers. In Organic Soils and Peat Materials for Sustainable Agriculture, 1st ed.; Parent, L.E., Ilnicki, P., Eds.; CRC Press: Boca Raton, FL, USA, 2003; pp. 67–92. ISBN 0-8493-1458-5. [Google Scholar]
- Enwezor, W.O. The mineralization of nitrogen and phosphorus in organic materials of varying C:N and C:P ratios. Plant Soil 1976, 44, 237–240. [Google Scholar] [CrossRef]
- Nannipieri, P.; Paul, E.A. The chemical and functional characterization of soil N and its biotic components. Soil Biol. Biochem. 2009, 41, 2357–2369. [Google Scholar] [CrossRef]
- Bunt, A.C. Some physical and chemical characteristics of loamless pot-plant substrates and their relation to plant growth. In Proceedings of the I Symposium on Artificial Media in Horticulture, Gent, Belgium, 10 September 1973; Acta Horticulturae 37. International Society for Horticultural Science (ISHS): Leuven, Belgium, 1974; pp. 1954–1965. [Google Scholar]
- Wang, B.; Yang, W.; McKittrick, J.; Meyers, M.A. Keratin: Structure, mechanical properties, occurrence in biological organisms, and efforts at bioinspiration. Prog. Mater. Sci. 2016, 76, 229–318. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, W.; Hayashi, C.; Gatesy, J.; McKittrick, J. Microstructure and mechanical properties of different keratinous horns. J. R. Soc. Interface 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Durand, J.H. Les Sols Irrigables: Etude Pedologique; Agence de Coopération Culturelle et Technique, Presses Universitaires de France: Paris, France, 1983; 339p, ISBN 2-85319-110-9. [Google Scholar]
- Cleveland, C.C.; Liptzin, D. C:N:P stoichiometry in soil: Is there a “Redfield ratio” for the microbial biomass? Biogeochemistry 2007, 85, 235–252. [Google Scholar] [CrossRef]
- Xu, X.; Thornton, P.E.; Post, W.M. A global analysis of soil microbial biomass carbon, nitrogen and phosphorus in terrestrial ecosystems. Glob. Ecol. Biogeogr. 2013, 22, 737–749. [Google Scholar] [CrossRef]
- Allison, S.D.; Weintraub, M.N.; Gartner, T.B.; Waldrop, M.P. Evolutionary-Economic Principles as Regulators of Soil Enzyme Production and Ecosystem Function. In Soil Enzymology; Shukla, G., Varma, A., Eds.; Soil Biology; Springer: Berlin/Heidelberg, Germany, 2011; pp. 229–243. ISBN 978-3-642-14225-3. [Google Scholar]
- Lashermes, G.; Gainvors-Claisse, A.; Recous, S.; Bertrand, I. Enzymatic Strategies and Carbon Use Efficiency of a Litter-Decomposing Fungus Grown on Maize Leaves, Stems, and Roots. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef]
- Finn, D.; Kopittke, P.M.; Dennis, P.G.; Dalal, R.C. Microbial energy and matter transformation in agricultural soils. Soil Biol. Biochem. 2017, 111, 176–192. [Google Scholar] [CrossRef]
- Allison, S.D.; Vitousek, P.M. Responses of extracellular enzymes to simple and complex nutrient inputs. Soil Biol. Biochem. 2005, 37, 937–944. [Google Scholar] [CrossRef]
- Jian, S.; Li, J.; Chen, J.; Wang, G.; Mayes, M.A.; Dzantor, K.E.; Hui, D.; Luo, Y. Soil extracellular enzyme activities, soil carbon and nitrogen storage under nitrogen fertilization: A meta-analysis. Soil Biol. Biochem. 2016, 101, 32–43. [Google Scholar] [CrossRef]
- Guerra, P.A.M.; Salas Sanjúan, M.D.C.; López, M.J. Evaluation of physicochemical properties and enzymatic activity of organic substrates during four crop cycles in soilless containers. Food Sci. Nutr. 2018, 6, 2066–2078. [Google Scholar] [CrossRef]
- McCarty, G.W.; Shogren, D.R.; Bremner, J.M. Regulation of urease production in soil by microbial assimilation of nitrogen. Biol. Fertil. Soils 1992, 12, 261–264. [Google Scholar] [CrossRef]
- Mondini, C.; Cayuela, M.L.; Sinicco, T.; Sánchez-Monedero, M.A.; Bertolone, E.; Bardi, L. Soil application of meat and bone meal. Short-term effects on mineralization dynamics and soil biochemical and microbiological properties. Soil Biol. Biochem. 2008, 40, 462–474. [Google Scholar] [CrossRef]
- Margalef, O.; Sardans, J.; Fernández-Martínez, M.; Molowny-Horas, R.; Janssens, I.A.; Ciais, P.; Goll, D.; Richter, A.; Obersteiner, M.; Asensio, D.; et al. Global patterns of phosphatase activity in natural soils. Sci. Rep. 2017, 7, 1337. [Google Scholar] [CrossRef] [PubMed]
- Heuck, C.; Weig, A.; Spohn, M. Soil microbial biomass C:N:P stoichiometry and microbial use of organic phosphorus. Soil Biol. Biochem. 2015, 85, 119–129. [Google Scholar] [CrossRef]
- Chen, J.; Seven, J.; Zilla, T.; Dippold, M.A.; Blagodatskaya, E.; Kuzyakov, Y. Microbial C:N:P stoichiometry and turnover depend on nutrients availability in soil: A 14C, 15N and 33P triple labelling study. Soil Biol. Biochem. 2019, 131, 206–216. [Google Scholar] [CrossRef]
- Bünemann, E.K.; Oberson, A.; Liebisch, F.; Keller, F.; Annaheim, K.E.; Huguenin-Elie, O.; Frossard, E. Rapid microbial phosphorus immobilization dominates gross phosphorus fluxes in a grassland soil with low inorganic phosphorus availability. Soil Biol. Biochem. 2012, 51, 84–95. [Google Scholar] [CrossRef]
- Bünemann, E.K. Assessment of gross and net mineralization rates of soil organic phosphorus—A review. Soil Biol. Biochem. 2015, 89, 82–98. [Google Scholar] [CrossRef]
- Vong, P.-C.; Dedourge, O.; Lasserre-Joulin, F.; Guckert, A. Immobilized-S, microbial biomass-S and soil arylsulfatase activity in the rhizosphere soil of rape and barley as affected by labile substrate C and N additions. Soil Biol. Biochem. 2003, 35, 1651–1661. [Google Scholar] [CrossRef]
- Schmalenberger, A.; Fox, A. Bacterial Mobilization of Nutrients from Biochar-Amended Soils. In Advances in Applied Microbiology; Sariaslani, S., Gadd, G.M., Eds.; Elsevier: Boston, MA, USA, 2016; Volume 94, pp. 109–159. [Google Scholar]
- Acosta-Martínez, V.; Tabatabai, M.A. Enzyme activities in a limed agricultural soil. Biol. Fertil. Soils 2000, 31, 85–91. [Google Scholar] [CrossRef]
- Ekenler, M.; Tabatabai, M.A. Responses of phosphatases and arylsulfatase in soils to liming and tillage systems. J. Plant Nutr. Soil Sci. 2003, 166, 281–290. [Google Scholar] [CrossRef]
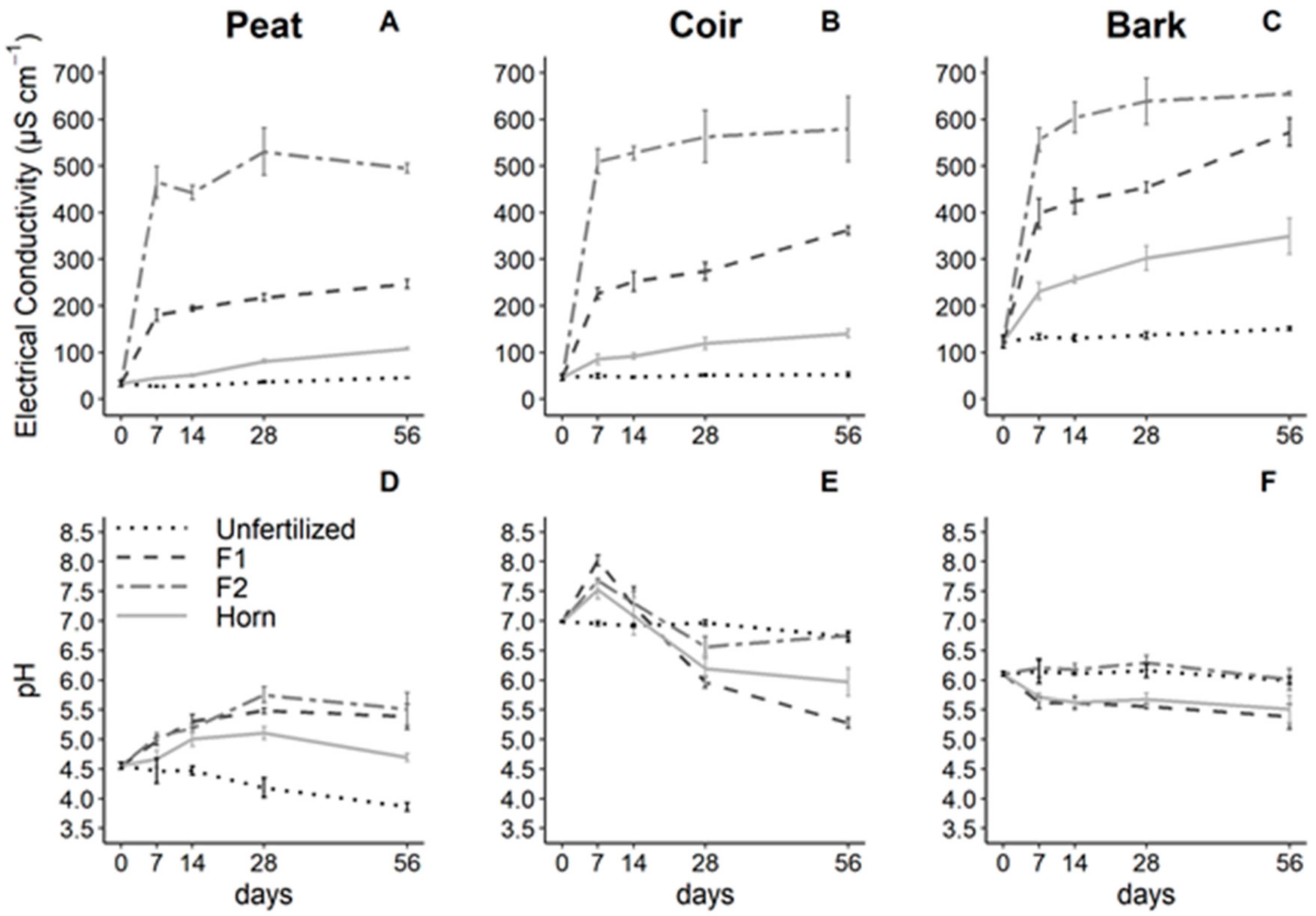
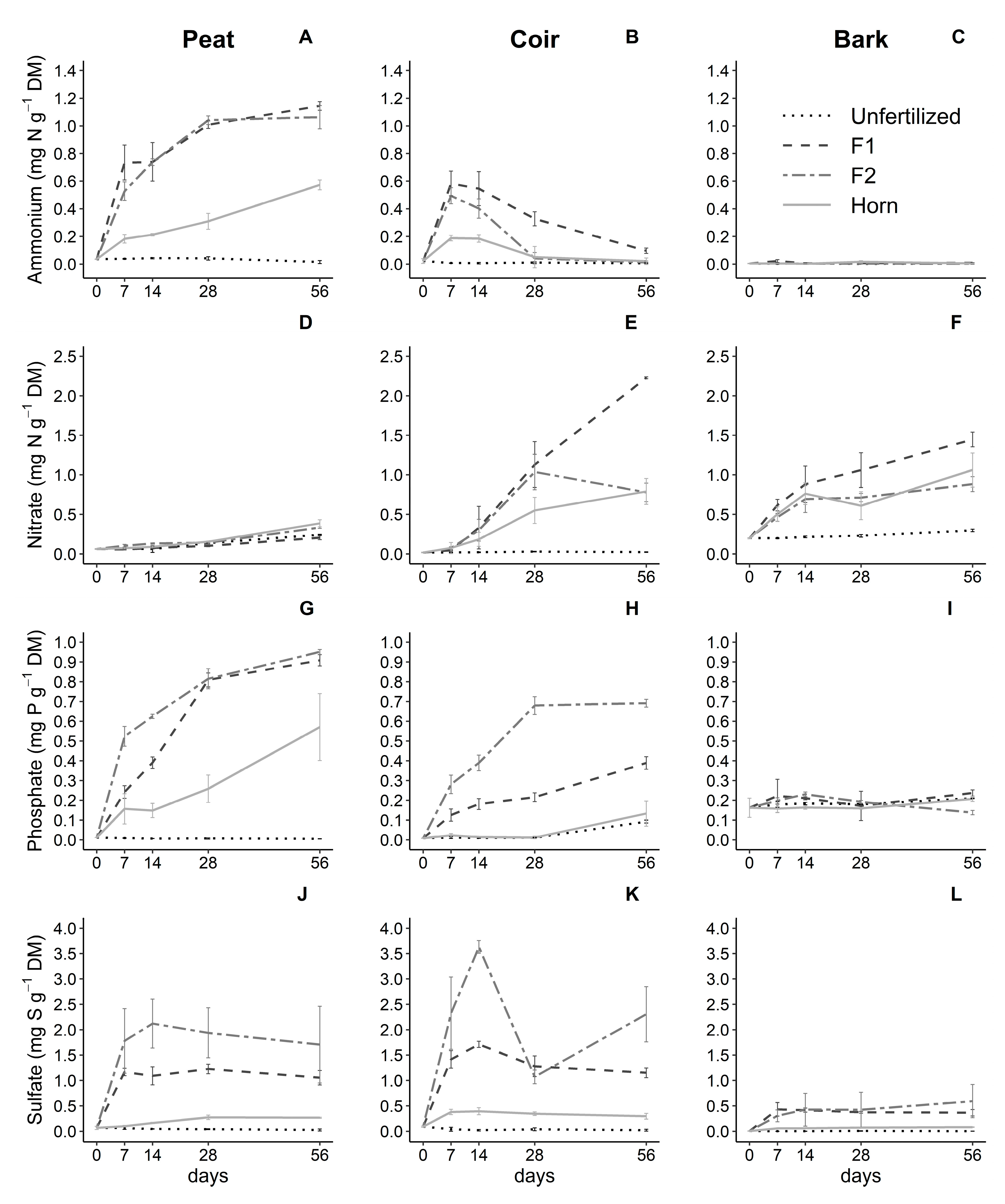
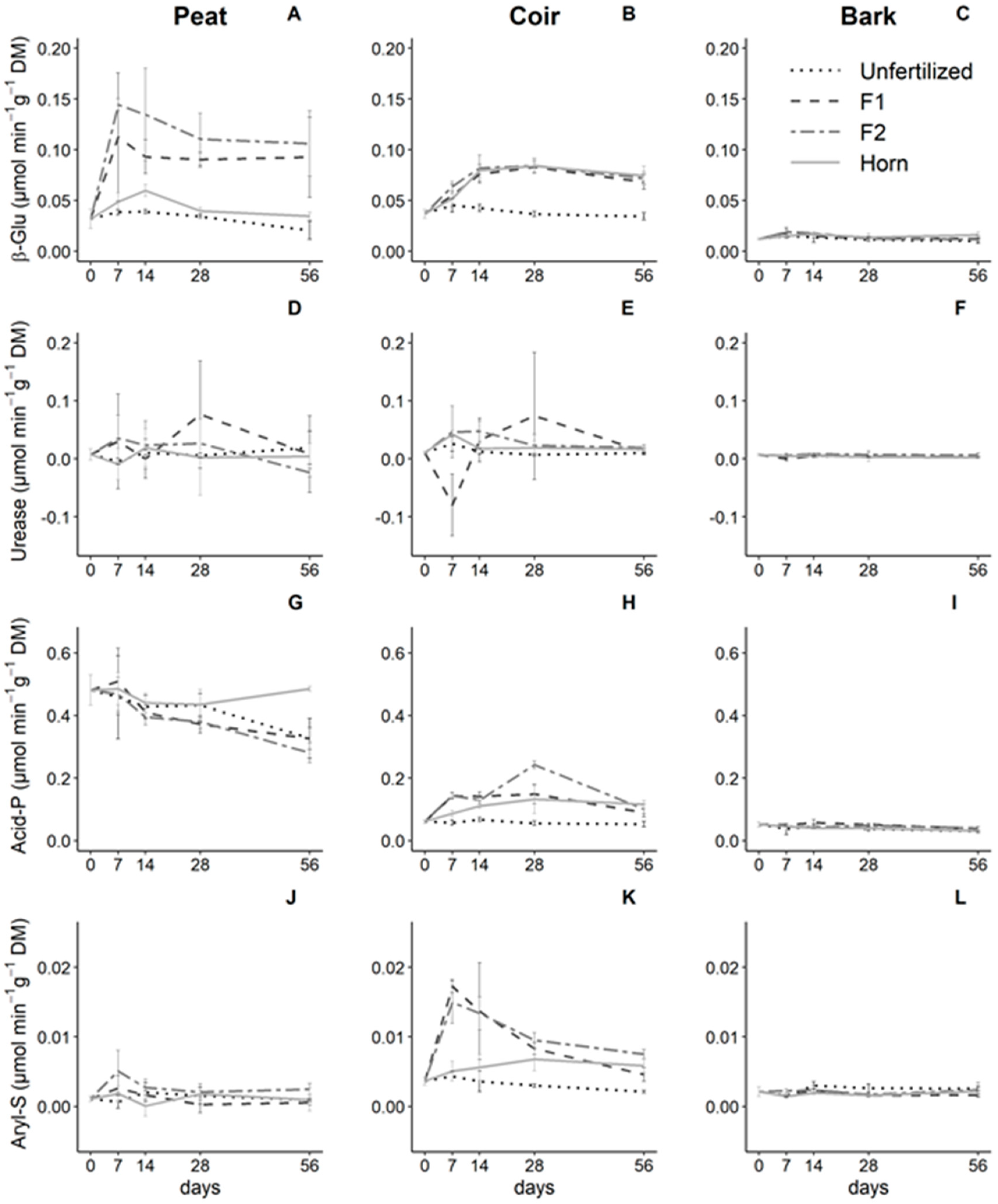
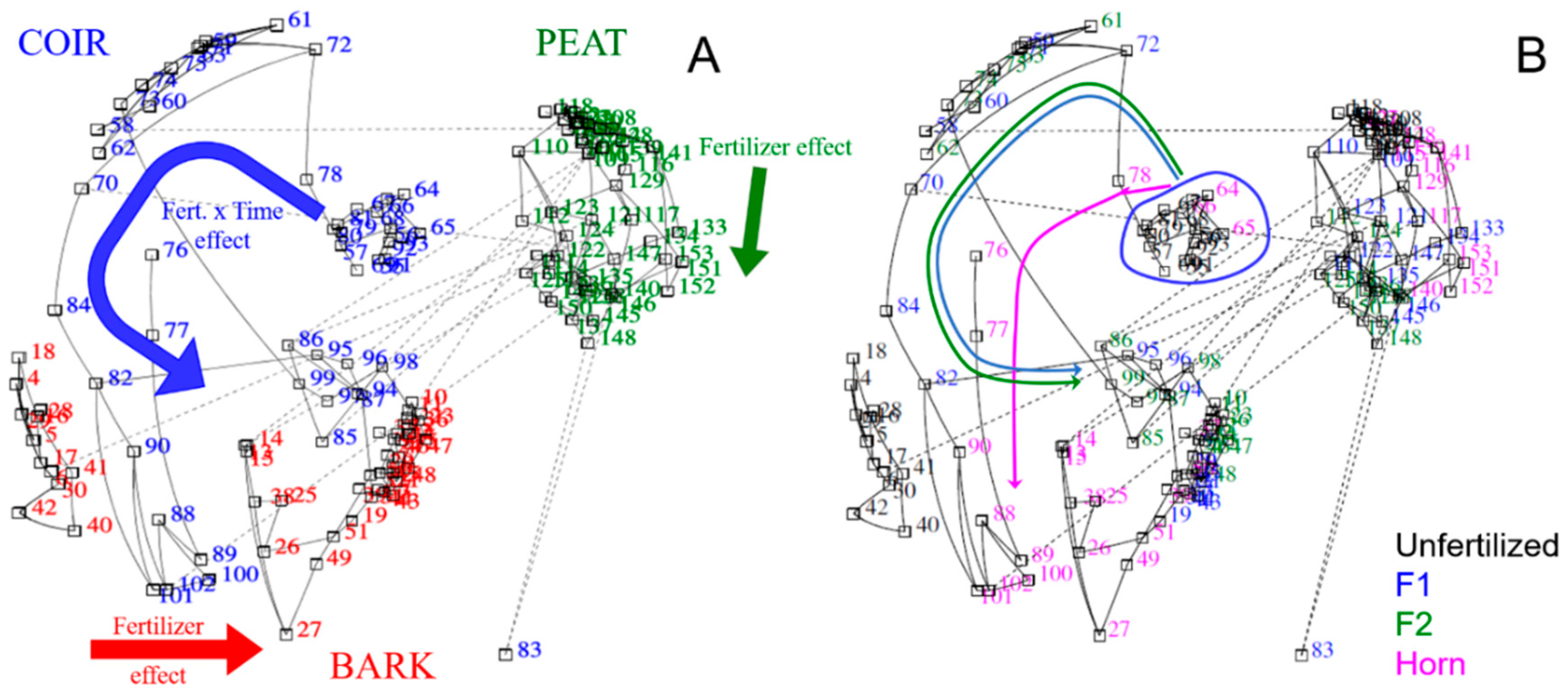
| Factors | pH | EC | NH4+-N | NO3−-N | PO43−-P | SO42−-S | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Df | F | p | F | p | F | p | F | p | F | p | F | p | |
| GM | 2 | 1946.6 | *** | 773.1 | *** | 1606.8 | *** | 273.1 | *** | 415.4 | *** | 140.7 | *** |
| Fert. | 3 | 82.8 | *** | 3065.5 | *** | 647.4 | *** | 155.7 | *** | 710.9 | *** | 305.2 | *** |
| Time | 3 | 79.9 | *** | 70.8 | *** | 0.5 | ns | 160.3 | *** | 176.8 | *** | 6.9 | *** |
| GM × Fert. | 6 | 71.3 | *** | 35.1 | *** | 263.4 | *** | 44.0 | *** | 213.8 | *** | 39.5 | *** |
| GM × Time | 6 | 78.3 | *** | 3.7 | ** | 119.7 | *** | 31.0 | *** | 54.9 | *** | 6.9 | *** |
| Fert. × Time | 9 | 7.9 | *** | 8.7 | *** | 2.4 | * | 24.4 | *** | 22.1 | *** | 5.5 | *** |
| GM × Fert. × Time | 18 | 17.9 | *** | 1.2 | ns | 20.9 | *** | 13.6 | *** | 16.1 | *** | 4.1 | *** |
| Error | 96 | - | - | - | - | - | - | - | - | - | - | - | - |
| Factors | β-Glu | Urease | Acid-P | Aryl-S | |||||
|---|---|---|---|---|---|---|---|---|---|
| Df | F | p | F | p | F | p | F | p | |
| GM | 2 | 222.7 | *** | 2.8 | ns | 1653.7 | *** | 248.7 | *** |
| Fert. | 3 | 56.9 | *** | 0.7 | ns | 7.0 | *** | 34.8 | *** |
| Time | 3 | 2.6 | ns | 1.4 | ns | 15.0 | *** | 13.5 | *** |
| GM × Fert. | 6 | 27.8 | *** | 1.0 | ns | 11.6 | *** | 25.9 | *** |
| GM × Time | 6 | 3.7 | ** | 0.5 | ns | 9.4 | *** | 9.1 | *** |
| Fert. × Time | 9 | 0.6 | ns | 2.2 | * | 3.3 | ** | 5.4 | *** |
| GM × Fert. × Time | 18 | 0.6 | ns | 1.6 | ns | 2.0 | * | 2.7 | *** |
| Error | 96 | - | - | - | - | - | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paillat, L.; Cannavo, P.; Barraud, F.; Huché-Thélier, L.; Guénon, R. Growing Medium Type Affects Organic Fertilizer Mineralization and CNPS Microbial Enzyme Activities. Agronomy 2020, 10, 1955. https://doi.org/10.3390/agronomy10121955
Paillat L, Cannavo P, Barraud F, Huché-Thélier L, Guénon R. Growing Medium Type Affects Organic Fertilizer Mineralization and CNPS Microbial Enzyme Activities. Agronomy. 2020; 10(12):1955. https://doi.org/10.3390/agronomy10121955
Chicago/Turabian StylePaillat, Louise, Patrice Cannavo, Fabrice Barraud, Lydie Huché-Thélier, and René Guénon. 2020. "Growing Medium Type Affects Organic Fertilizer Mineralization and CNPS Microbial Enzyme Activities" Agronomy 10, no. 12: 1955. https://doi.org/10.3390/agronomy10121955
APA StylePaillat, L., Cannavo, P., Barraud, F., Huché-Thélier, L., & Guénon, R. (2020). Growing Medium Type Affects Organic Fertilizer Mineralization and CNPS Microbial Enzyme Activities. Agronomy, 10(12), 1955. https://doi.org/10.3390/agronomy10121955





