Effects of Elevated Temperature and CO2 Concentration on Seedling Growth of Ventenata dubia (Leers) Coss. and Bromus tectorum L.
Abstract
1. Introduction
2. Materials and Methods
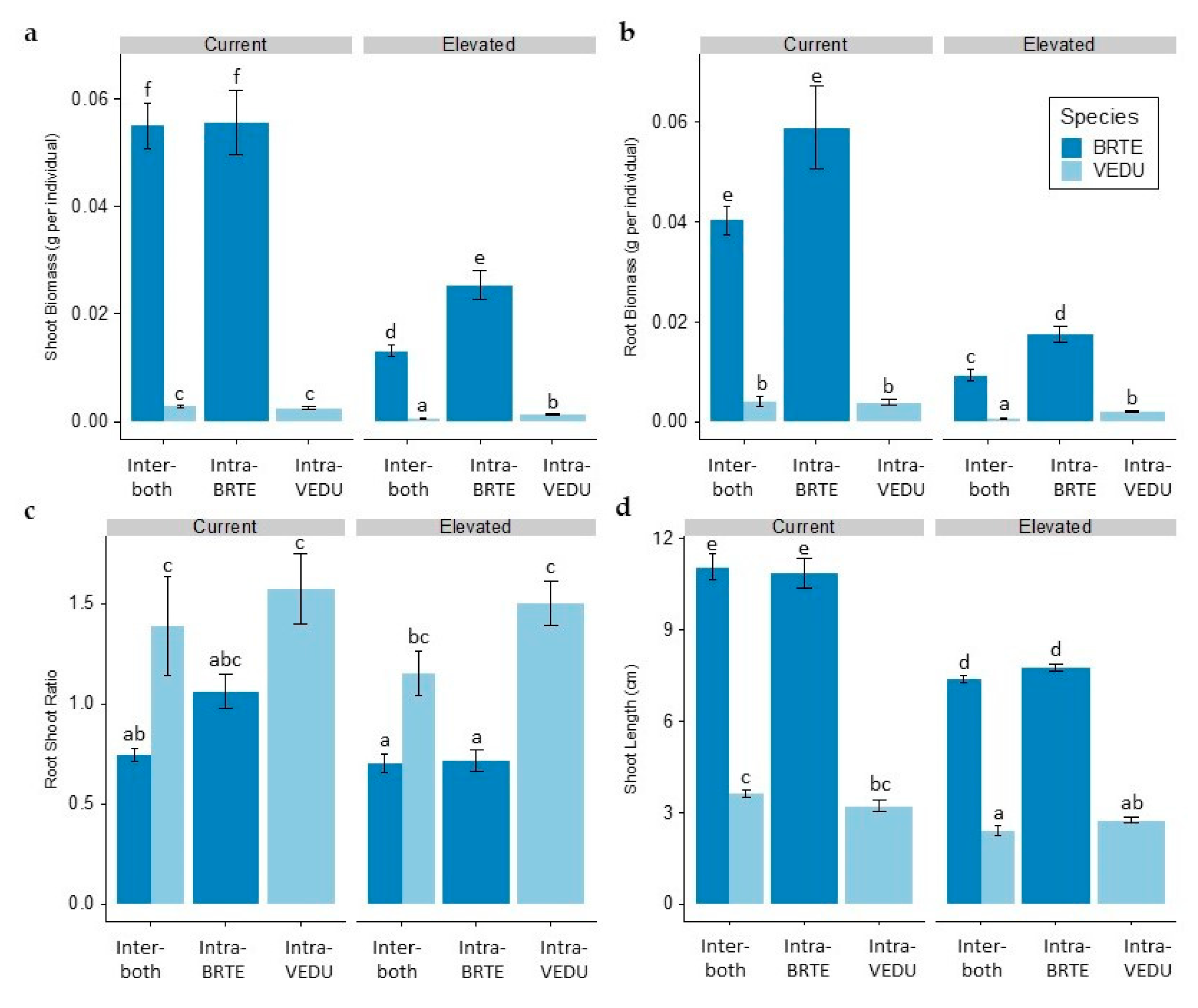
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Blumenthal, D.; Kray, J.; Ortmans, W.; Ziska, L.; Pendall, E. Cheatgrass is favored by warming but not CO2 enrichment in a semi-arid grassland. Glob. Chang. Biol. 2016, 22, 3026–3038. [Google Scholar] [CrossRef] [PubMed]
- Bradley, B.A.; Curtis, C.A.; Chambers, J.C. Bromus response to climate and projected changes with climate change. In Exotic Brome-Grasses in Arid and Semiarid Ecosystems of the Western US; Germino, M.J., Chambers, J.C., Brown, C.S., Eds.; Springer: New York, NY, USA, 2016; pp. 257–274. [Google Scholar] [CrossRef]
- Germino, M.; Chambers, J.C.; Brown, C.S. Exotic Brome-Grasses in Arid and Semiarid Ecosystems of the Western US; Springer: New York, NY, USA, 2016; 484p. [Google Scholar]
- Wallace, J.M.; Pavek, P.L.S.; Prather, T.S. Ecological characteristics of Ventenata dubia in the Intermountain Pacific Northwest. Invasive Plant Sci. Manag. 2015, 8, 57–71. [Google Scholar] [CrossRef]
- Harvey, A.; Mangold, J. Ventenata; Montana State University Extension: Bozeman, MT, USA, 2019; Publication MT201810AG. [Google Scholar]
- Bansal, S.; James, J.J.; Sheley, R.L. The effects of precipitation and soil type on three invasive annual grasses in the western United States. J. Arid Environ. 2014, 104, 38–42. [Google Scholar] [CrossRef]
- James, J.J. Effect of soil nitrogen stress on the relative growth rate of annual and perennial grasses in the Intermountain West. Plant Soil 2008, 310, 201–210. [Google Scholar] [CrossRef]
- James, J.J. Leaf nitrogen productivity as a mechanism driving the success of invasive annual grasses under low and high nitrogen supply. J. Arid Environ. 2008, 72, 1775–1784. [Google Scholar] [CrossRef]
- Whitlock, C.; Cross, W.; Maxwell, B.; Silverman, N.; Wade, A.A. 2017 Montana Climate Assessment; Montana State University and University of Montana: Bozeman, MT, USA; Montana Institute on Ecosystems: Missoula, MT, USA, 2017; 318p. [Google Scholar] [CrossRef]
- International Panel on Climate Change. Climate Change: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Core Writing Team, Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014. [Google Scholar] [CrossRef]
- Smith, S.D.; Strian, B.R.; Sharkey, T.D. Effects of CO2 enrichment on four Great Basin grasses. Funct. Ecol. 1987, 1, 139–143. [Google Scholar] [CrossRef]
- Smith, S.D.; Huxman, T.E.; Zitzer, S.F.; Charlet, T.N.; Housman, D.C.; Coleman, J.S.; Nowak, R.S. Elevated CO2 increases productivity and invasive species success in an arid ecosystem. Nature 2000, 408, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Blank, R.R.; White, R.H.; Ziska, L.H. Combustion properties of Bromus tectorum L.: Influence of ecotype and growth under four CO2 concentrations. Int. J. Wildland Fire 2006, 15, 227–236. [Google Scholar] [CrossRef]
- Ziska, L.H.; Reeves, J.B.; Blank, B. The impact of recent increases in atmospheric CO2 on biomass production and vegetative retention of cheatgrass (Bromus tectorum): Implications for fire disturbance. Glob. Chang. Biol. 2005, 11, 1325–1332. [Google Scholar] [CrossRef]
- Blank, R.R.; Morgan, T.; Ziska, L.; White, R.H. Effect of atmospheric CO2 levels on nutrients in cheatgrass tissue. Nat. Resour. Environ. Iss. 2011, 16, 3–8. [Google Scholar]
- Bansal, S.; Sheley, R.L. Annual grass invasion in sagebrush steppe: The relative importance of climate, soil properties and biotic interactions. Oecologia 2016, 181, 543–557. [Google Scholar] [CrossRef] [PubMed]
- Jabran, K.; Doğan, M.N. High carbon dioxide concentration and elevated temperature impact the growth of weeds but do not change the efficacy of glyphosate. Pest Manag. Sci. 2018, 74, 766–771. [Google Scholar] [CrossRef] [PubMed]
- Larson, C.D.; Lehnhoff, E.A.; Noffsinger, C.; Rew, L.J. Competition between cheatgrass and bluebunch wheatgrass is altered by temperature, resource availability, and atmospheric CO2 concentration. Oecologia 2018, 186, 855–868. [Google Scholar] [CrossRef]
- Zelikova, T.J.; Hufbauer, R.A.; Reed, S.C.; Wertin, T.; Fettig, C.; Belnap, J. Eco-evolutionary responses of Bromus tectorum to climate change: Implications for biological invasions. Ecol. Evol. 2013, 3, 1374–1387. [Google Scholar] [CrossRef] [PubMed]
- Compagnoni, A. Warming, soil moisture, and loss of snow increase Bromus tectorum’s population growth rate. Elem. Sci. Anth. 2014, 2, 000020. [Google Scholar] [CrossRef]
- Boyte, S.P.; Wylie, B.K.; Major, D.J. Cheatgrass percent cover change: Comparing recent estimates to climate change—Driven Predictions in the Northern Great Basin. Rangel. Ecol. Manag. 2016, 69, 265–279. [Google Scholar] [CrossRef]
- Larson, C.D.; Lehnhoff, E.A.; Rew, L.J. A warmer and drier climate in the northern sagebrush biome does not promote cheatgrass invasion or change its response to fire. Oecologia 2017, 185, 763–774. [Google Scholar] [CrossRef]
- Wolkovich, E.M.; Cleland, E.E. The phenology of plant invasions: A community ecology perspective. Front. Ecol. Environ. 2011, 9, 287–294. [Google Scholar] [CrossRef]
- Wolkovich, E.M.; Jonathan Davies, T.; Schaefer, H.; Cleland, E.E.; Cook, B.I.; Travers, S.E.; Davis, C.C. Temperature-dependent shifts in phenology contribute to the success of exotic species with climate change. Am. J. Bot. 2013, 100, 1407–1421. [Google Scholar] [CrossRef] [PubMed]
- Scheinost, P.; Stannard, M.; Prather, T. Plant Guide: Ventenata. United States Department of Agriculture, Natural Resources Conservation Service, 2008. Available online: https://plants.usda.gov/plantguide/pdf/pg_vedu.pdf (accessed on 13 August 2020).
- Jones, L.C.; Norton, N.; Prather, T.S. Indicators of ventenata (Ventenata dubia) invasion in sagebrush steppe rangelands. Invasive Plant Sci. Manag. 2018, 11, 1–9. [Google Scholar] [CrossRef]
- Bashkin, M.; Stohlgren, T.J.; Otsuki, Y.; Lee, M.; Evangelista, P.; Belnap, J. Soil characteristics and plant exotic species invasions in the Grand Staircase—Escalante National Monument, Utah, USA. Appl. Soil Ecol. 2003, 22, 67–77. [Google Scholar] [CrossRef]
- Averett, J.P.; McCune, B.; Parks, C.G.; Naylor, B.J.; DelCurto, T.; Mata-Gonzalez, R. Non-native plant invasion along elevation and canopy closure gradients in a middle rocky mountain ecosystem. PLoS ONE 2016, 11, e0147826. [Google Scholar] [CrossRef]
- Time and Date AS. Available online: https://www.timeanddate.com/weather/usa/bozeman/climate (accessed on 12 November 2019).
- de Wit, C.T. On competition. Versl. Landbouwkd. Onderz. 1960, 66, 1–82. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 1 September 2018).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis, 2nd ed.; Gentleman, R., Hornik, K., Parmigiani, G., Eds.; Springer: Houston, TX, USA, 2016. [Google Scholar] [CrossRef]
- Wickham, H.; Romain, F.; Henry, L.; Müller, K. A Grammar of Data Manipulation. Comprehensive R Archive Network (CRAN), 2018. Available online: https://cran.r-project.org/web/packages/dplyr/index.html (accessed on 18 August 2020). [CrossRef]
- Lenth, R.; Singmann, H.; Love, J.; Buerkner, P.; Herve, M. Estimated Marginal Means, Aka Least-Squares Means. Comprehensive R Archive Network (CRAN), 2018. Available online: https://cran.r-project.org/web/packages/emmeans/index.html (accessed on 25 October 2020). [CrossRef]
- Horn, K.J.; Bishop, T.B.B.; St. Clair, S.B. Precipitation timing and soil heterogeneity regulate the growth and seed production of the invasive grass red brome. Biol. Invasive 2017, 19, 1339–1350. [Google Scholar] [CrossRef]
- Rice, K.J.; Black, R.A.; Radamaker, G.; Evans, R.D. Photosynthesis, growth, and biomass allocation in habitat ecotypes of cheatgrass (Bromus tectorum). Func. Ecol. 1992, 6, 32–40. [Google Scholar] [CrossRef]
- Hulbert, L.C. Ecological studies of Bromus tectorum and other annual bromegrasses. Ecol. Monogr. 1955, 25, 181–213. [Google Scholar] [CrossRef]
- Shinozaki, K.; Kira, T. Intraspecific competition among higher plants. VII. Logistic theory of the C-D effect. J. Inst. Polytech. Osaka City Univ. D 1956, 7, 35–72. [Google Scholar]
- Yoda, K.; Kira, T.; Ogawa, H.; Hozumi, K. Self-thinning in over-crowded pure stands under cultivated and natural conditions. J. Biol. Osaka City Univ. 1963, 14, 107–129. [Google Scholar]
- Hopkins, W.G. Water relations of the whole plant. In Introduction to Plant Physiology; John Wiley & Sons, Inc.: New York, NY, USA, 1995; pp. 41–64. [Google Scholar]
- Abatzoglou, J.T.; Kkolden, C.A. Climate change and western US deserts: Potential for increased wildfire and invasive annual grasses. Rangel. Ecol. Manag. 2011, 64, 471–478. [Google Scholar] [CrossRef]
- Gurevitch, J.; Scheiner, S.M.; Fox, G.A. The Ecology of Plants, 2nd ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2006; 574p. [Google Scholar]
- Bazzaz, F.A. The response of natural ecosystems to the rising global CO2 levels. Ann. Rev. Ecol. Syst. 1990, 21, 167–196. [Google Scholar] [CrossRef]
- Jackson, R.B.; Sala, O.E.; Field, C.B.; Mooney, H.A. CO2 alters water use, carbon gain, and yield for the dominant species in a natural grassland. Oecologia 1994, 98, 257–262. [Google Scholar] [CrossRef] [PubMed]
| Time | Temperature (°C) | CO2 (ppm) |
|---|---|---|
| Current conditions 07:00–19:00 19:00–07:00 | 23 4 | 400 400 |
| Elevated conditions 07:00–19:00 19:00–07:00 | 29.6 10.6 | 800 800 |
| Shoot Biomass | Root Biomass | Root–Shoot Ratio | Shoot Length | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Source | Df | F | p | F | p | F | p | F | p |
| Species | 173 | 2064 | <0.01 | 769 | <0.01 | 50.2 | <0.01 | 1305 | <0.01 |
| Comp | 273 | 15.3 | <0.01 | 20.1 | <0.01 | 4.2 | 0.02 | 0.83 | 0.44 |
| Clim | 173 | 267 | <0.01 | 184 | <0.01 | 3.82 | 0.05 | 107 | <0.01 |
| Comp × Clim | 273 | 24 | <0.01 | 11.6 | <0.01 | 1.92 | 0.15 | 4.13 | 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harvey, A.J.; Rew, L.J.; Prather, T.S.; Mangold, J.M. Effects of Elevated Temperature and CO2 Concentration on Seedling Growth of Ventenata dubia (Leers) Coss. and Bromus tectorum L. Agronomy 2020, 10, 1718. https://doi.org/10.3390/agronomy10111718
Harvey AJ, Rew LJ, Prather TS, Mangold JM. Effects of Elevated Temperature and CO2 Concentration on Seedling Growth of Ventenata dubia (Leers) Coss. and Bromus tectorum L. Agronomy. 2020; 10(11):1718. https://doi.org/10.3390/agronomy10111718
Chicago/Turabian StyleHarvey, Audrey J., Lisa J. Rew, Tim S. Prather, and Jane M. Mangold. 2020. "Effects of Elevated Temperature and CO2 Concentration on Seedling Growth of Ventenata dubia (Leers) Coss. and Bromus tectorum L." Agronomy 10, no. 11: 1718. https://doi.org/10.3390/agronomy10111718
APA StyleHarvey, A. J., Rew, L. J., Prather, T. S., & Mangold, J. M. (2020). Effects of Elevated Temperature and CO2 Concentration on Seedling Growth of Ventenata dubia (Leers) Coss. and Bromus tectorum L. Agronomy, 10(11), 1718. https://doi.org/10.3390/agronomy10111718




