Promising Composts as Growing Media for the Production of Baby Leaf Lettuce in a Floating System
Abstract
:1. Introduction
2. Materials and Methods
2.1. Compost Characterisation
2.2. Compost Microbial Community
2.3. In Vitro Suppressiveness against Pythium Irregulare
- MGI% = ((RGcontrol − RGcompost)/RGcontrol) × 100;
- RGcontrol = radial growth of pathogen in control plates;
- RGcompost = radial growth of pathogen in plates with compost.
2.4. Experimental Conditions
2.5. Germination
2.6. Plant Analysis at Harvesting
2.7. Statistical Analysis
3. Results
3.1. Compost Characterisation
3.2. Compost Microbial Community
3.3. Suppressiveness: Composts with Added Value
3.4. Compost as a Component of Growing Media in Floating Systems
3.4.1. Percentage of Germination and Yield
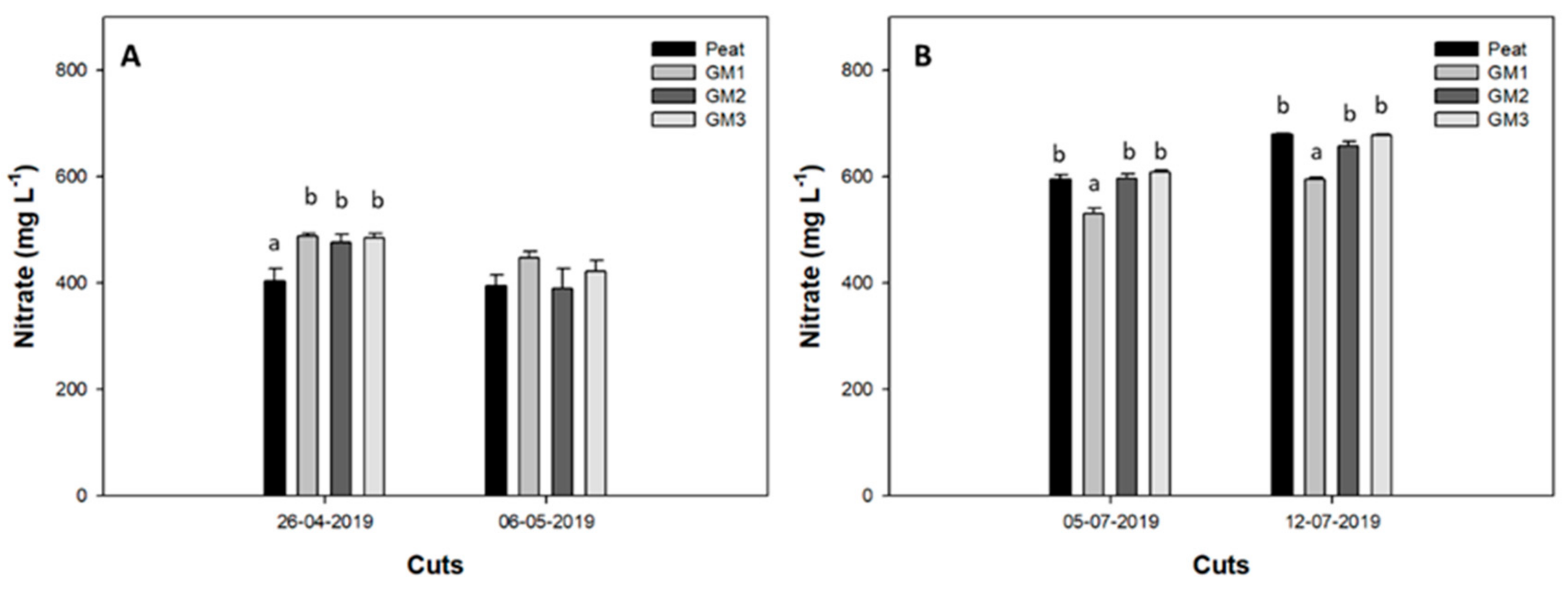
3.4.2. Nitrate Content in Leaves and in the Nutrient Solution
3.4.3. Total Phenolic and Antioxidant Capacity in Red Baby Leaf Lettuce Leaves and Roots
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nicola, S.; Egea-Gilabert, C.; Niñirola, D.; Conesa, E.; Pignata, G.; Fontana, E.; Fernández, J.A. Nitrogen and aeration levels of the nutrient solution in soilless cultivation systems as important growing conditions affecting inherent quality of baby leaf vegetables: A review. Acta Hortic. 2015, 1099, 167–177. [Google Scholar] [CrossRef]
- Mampholo, B.M.; Maboko, M.M.; Soundy, P.; Svakumar, D. Phytochemicals and overall quality of baby leaf lettuce (Lactuca sativa L.) varieties grown in closed hydroponic system. J. Food Qual. 2016, 39, 805–815. [Google Scholar] [CrossRef]
- Nicola, S.; Pignata, G.; Casale, M.; Lo Turco, P.E.; Gaino, W. Overview of a Lab-scale Pilot Plant for Studying Baby leaf Vegatables Grown in Soiless Culure. Hortic. J. 2016, 85, 97–104. [Google Scholar] [CrossRef]
- Cros, V.; Martínez-Sánchez, J.J.; Franco, J.A. Good yields of common purslane with a high fatty acid content can be obtained in a peat-based floating system. HortTechnology 2007, 17, 14–20. [Google Scholar] [CrossRef] [Green Version]
- Pennisi, G.; Sanyé-Mengual, E.; Orsini, F.; Crepaldi, A.; Nicola, S.; Ochoa, J.; Fernandez, J.A.; Gianquinto, G. Modelling environmental burdens of indoor-grown vegetables and herbs as affected by red and blue LED lighting. Sustainability 2019, 11, 4063. [Google Scholar] [CrossRef] [Green Version]
- Pane, C.; Spaccini, R.; Piccolo, A.; Scala, F.; Bonanomi, G. Compost amendments enhance peat suppressiveness to Pythium ultimum, Rhizoctonia solani and Sclerotinia minor. Biol. Control 2011, 56, 115–124. [Google Scholar] [CrossRef]
- Limpens, J.; Berendse, F.; Blodau, C.; Canadell, J.G.; Freeman, C.; Holden, J.; Roulet, N.; Rydin, H.; Schaepman-Strub, G. Peatlands and the carbon cycle: From local processes to global implications—A synthesis. Biogeosciences 2008, 5, 1475–1491. [Google Scholar] [CrossRef] [Green Version]
- Gruda, N.S. Increasing sustainability of growing media constituents and stand-alone substrates in soilless culture systems. Agronomy 2019, 9, 298. [Google Scholar] [CrossRef] [Green Version]
- Bonn, A.; Reed, M.S.; Evans, C.D.; Joosten, H.; Bain, C.; Farmer, J.; Emmer, I.; Couwenberg, J.; Moxey, A.; Artz, R.; et al. Investing in nature: Developing ecosystem service markets for peatland restoration. Ecosyst. Serv. 2014, 9, 54–65. [Google Scholar] [CrossRef] [Green Version]
- Gruda, N.; Bisbis, M.B.; Tanny, J. Impacts of protected vegetable cultivation on climate change and adaptation strategies for cleaner production—A review. J. Clean. Prod. 2019, 225, 324–339. [Google Scholar] [CrossRef]
- Blaya, J.; Lloret, E.; Ros, M.; Pascual, J.A. Identification of predictor parameters to determine agro-industrial compost suppressiveness against Fusarium oxysporum and Phytophthora capsici disease in muskmelon and pepper seedlings. J. Sci. Food Agric. 2015, 95, 1482–1490. [Google Scholar] [CrossRef] [PubMed]
- Giménez, A.; Fernández, J.A.; Pascual, J.A.; Ros, M.; López-Serrano, M.; Egea-Gilabert, C. An agroindustrial compost as alternative to peat for production of baby leaf red lettuce in a floating system. Sci. Hortic. 2019, 246, 907–915. [Google Scholar] [CrossRef]
- Razza, F.; D’Avino, L.; L’Abate, G.; Lazzeri, L. The role of compost in bio-waste management and circular economy. In Designing Sustainable Technologies, Products and Policies; Benetto, E., Gericke, K., Guiton, M., Eds.; Springer: Cham, Switzerland, 2018. [Google Scholar] [CrossRef] [Green Version]
- Prasad, M.; Maher, M.J. The use of composted green waste (cgw) as a growing medium component. Acta Hortic. 2001, 549, 107–114. [Google Scholar] [CrossRef]
- Raviv, M. Composts in growing media: What’s new and what’s next? Acta Hortic. 2013, 982, 39–52. [Google Scholar] [CrossRef]
- Tan, K.H. Humic Matter in Soil and the Environment, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar] [CrossRef]
- Zaccardelli, M.; Pane, C.; Scotti, R.; Maria, A.; Colturali, S.; Basilicata, U. Impiego di compost-tea come bioagrofarmaci e biostimolanti in orto- frutticoltura. Italus Horto 2012, 19, 17–28. [Google Scholar]
- Hadar, Y.; Papadopoulou, K.K. Suppressive composts: Microbial ecology links between abiotic environments and healthy plants. Annu. Rev. Phytopathol. 2012, 50, 133–153. [Google Scholar] [CrossRef]
- Ros, M.; Blaya, J.; Baldrian, P.; Bastida, F.; Richnow, H.; Jehmlich, N.; Pascual, J. In vitro elucidation of suppression effects of composts to soil-borne pathogen Phytophthora nicotianae on pepper plants using 16S amplicon sequencing and metaproteomics. Renew. Agric. Food Syst. 2020, 35, 206–214. [Google Scholar] [CrossRef]
- Bustamante, M.A.; Paredes, C.; Moral, R.; Agullo, E.; Perez-Murcia, M.D.; Abad, M. Compost from distillery wastes as peat subsitutes for transplant production. Resour. Conserv. Recycl. 2008, 52, 792–799. [Google Scholar] [CrossRef]
- McKenzie, J.R.; Young, P.N. Determination of ammonia-, nitrate-, and organic nitrogen in water and wastewater with an ammonia gas-sensing electrode. Analyst 1975, 100, 620–628. [Google Scholar] [CrossRef]
- Watanabe, F.S.; Olsen, S.R. Test of an ascorbic acid method for determining phosphorus in water and NaHCO3 extracts from soil. Soil Sci. Soc. Am. Proc. 1965, 29, 677–678. [Google Scholar] [CrossRef]
- Sánchez-Monedero, M.A.; Roig, A.; Martínez-Pardo, C.; Cegarra, J.; Paredes, C. A microanalysis method for determining total organic carbón in extracts of humic substances. Relationships between total organic carbón and oxidable carbón. Bioresour. Technol. 1996, 57, 291–295. [Google Scholar] [CrossRef]
- Garcia, C.; Hernandez, T.; Costa, F. Potential use of dehydrogenase activity as an index of microbial activity in degraded soil. Commun. Soil Plant 2008, 28, 123–134. [Google Scholar] [CrossRef]
- Taskin, B.; Gozen, A.G.; Duran, M. Selective quantification of viable Escherichia coli bacteria in biosolids by quantitative PCR with propidium monoazide modification. Appl. Environ. Microbiol. 2011, 77, 4329–4335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Huntley, J.; Fierer, N.; Owens, S.M.; Betley, J.; Fraser, L.; Bauer, M.; et al. Ultra-high-throughput microbial community analysis on the Illimina HiSeq and MiSeq platforms. ISME J. 2012, 6, 1621–1624. [Google Scholar] [CrossRef] [Green Version]
- Ihrmark, K.; Bödeker, I.T.M.; Cruz-Martinez, K.; Friberg, H.; Kubartova, A.; Schenck, J.; Strid, Y.; Stenlid, J.; Brandström-Durling, M.; Clemmensen, K.E.; et al. New primers to amplify the fungal ITS2 region–evaluation by454-sequencing of artificial and natural communities. FEMS Microbiol. Ecol. 2012, 82, 666–677. [Google Scholar] [CrossRef]
- Žifčáková, L.; Vetrovsky, T.; Howe, A.; Baldrian, P. Microbial activity in forest soil reflects the changes in ecosystem properties between summer and winter. Environ. Microbiol. 2016, 18, 288–301. [Google Scholar] [CrossRef]
- De Corato, U.; Viola, E.; Arcieri, G.; Valerio, V.; Zimbardi, F. Use of composted agro-energy co-products and agricultural residues against soil-borne pathogens in horticultural soil-less system. Sci. Hortic. 2016, 210, 166–179. [Google Scholar] [CrossRef]
- Niñirola, D.; Fernández, J.A.; Conesa, E.; Martínez, J.A.; Egea-Gilabert, C. Combined effects of growth cycle and different levels of aeration in nutrient solution on productivity, quality and shelf-life of watercress (Nasturtium officinale R. Br.) plants. HortScience 2014, 49, 567–573. [Google Scholar] [CrossRef]
- Egea-Gilabert, C.; Fernández, J.A.; Migliaro, D.; Martínez-Sánchez, J.J.; Vicente, M.J. Genetic variability in wild vs. cultivated Eruca vesicaria populations as assessed by morphological, agronomical and molecular analyses. Sci. Hortic. 2009, 121, 260–266. [Google Scholar] [CrossRef]
- Lara, L.J.; Egea-Gilabert, C.; Niñirola, D.; Conesa, E.; Fernández, J.A. Effect of aeration of the nutrient solution on the growth and quality of purslane (Portulaca oleracea). J. Hortic. Sci. Biotechnol. 2011, 86, 603–610. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Martínez-Hernández, G.B.; Gómez, P.A.; Pradas, I.; Artés, F.; Artés-Hernández, F. Moderate UV-C pretreatment as a quality enhancement tool in fresh-cut Bimi® broccoli. Postharvest Biol. Technol. 2011, 62, 327–337. [Google Scholar] [CrossRef]
- Klug, T.V.; Martínez-Hernández, G.B.; Collado, E.; Artés, F.; Artés-Hernández, F. Effect of microwave and high-pressure processing on quality of an innovative broccoli hummus. Food Bioprocess. Technol. 2018, 11, 1464–1477. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of free radical method to evaluate antioxidant activity. LWT—Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Benzie, L.F.; Strain, J.J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol. 1999, 299, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Cano, A.; Hernández-Ruíz, J.; García-Cánovas, F.; Acosta, M.; Arnao, M.B. An end-point method for estimation of the total antioxidant activity in plant material. Phytochem. Anal. 1998, 9, 196–202. [Google Scholar] [CrossRef]
- Větrovský, T.; Baldrian, P.; Morais, D. SEED 2: A user-friendly platform for amplicon high-throughput sequencing data analyses. Bioinformatics 2018, 34, 2292–2294. [Google Scholar] [CrossRef]
- Větrovský, T.; Baldrian, P. The variability of the 16S rRNA gene in bacterial genomes and its consequences for bacterial community analyses. PLoS ONE 2013, 8, e57923. [Google Scholar] [CrossRef] [Green Version]
- Aronesty, E. Comparison of sequencing utility programs. Open Bioinform. J. 2013, 7, 1–8. [Google Scholar] [CrossRef]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 1, 2460–2461. [Google Scholar] [CrossRef] [Green Version]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Kõljalg, U.; Nilsson, R.H.; Abarenkov, K.; Tedersoo, L.; Taylor, A.F.; Bahram, M.; Bates, S.T.; Bruns, T.D.; Bengtsson-Palme, J.; Callaghan, T.M.; et al. Towards a unified paradigm for sequence-based identification of fungi. Mol. Ecol. 2013, 22, 5271–5277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zaller, J.G. Vermicompost as a substitute for peat in potting media: Effects on germination, biomass allocation, yields and fruit quality of three tomato varieties. Sci. Hortic. 2007, 112, 191–199. [Google Scholar] [CrossRef]
- Clemmensen, A.W. Physical characteristics of Miscanthus composts compared to peat and wood fiber growth substrates. Compost Sci. Util. 2004, 12, 219–224. [Google Scholar] [CrossRef]
- Pascual, J.A.; Ceglie, F.; Tuzel, Y.; Koller, M.; Koren, A.; Hitchings, R.; Tittarelli, F. Organic substrate for transplant production in organic nurseries. A review. Agron. Sustain. Dev. 2018, 38, 35. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; McConnell, D.B.; Robinson, C.A.; Caldwell, R.D.; Huang, Y. Rooting foliage plant cuttings in compost formulated substrates. HorTechnology 2003, 13, 110–114. [Google Scholar] [CrossRef] [Green Version]
- Gruda, N.; Tucher, S.V.; Schnitzler, W.H. N-Immobilization of wood fiber substrates in the production of tomato transplants (Lycopersicon lycopersicum (L.) Karst. Ex Farw.). J. Appl. Bot. 2000, 74, 32–37. (In German) [Google Scholar]
- Gruda, N.; Schnitzler, W.H. Suitability of wood fiber substrates for production of vegetable transplants. I. Physical properties of wood fiber substrates. Sci. Hortic. 2004, 100, 309–322. [Google Scholar] [CrossRef]
- Gruda, N.; Schnitzler, W.H. Suitability of wood fiber substrates for production of vegeta-ble transplants. II. The effect of wood fiber substrates and their volume weights on the growth of tomato transplants. Sci. Hortic. 2004, 100, 333–340. [Google Scholar] [CrossRef]
- Bunt, A.C. Media and Mixes for Container-Grown Plants (Second Edition of Modern Potting Composts): A Manual on the Preparation and Use of Growing Media for Pot Plants; Academic Division of Unwin Hyman Ltd.: Broadwick Street, London, UK, 1988. [Google Scholar]
- Morales, A.B.; Bustamante, M.A.; Marhuenda-Egea, F.C.; Moral, R.; Ros, M.; Pascual, J.A. Agri-food sludge management using different co-composting strategies: Study of the added value of the composts obtained. J. Clean. Prod. 2016, 121, 186–197. [Google Scholar] [CrossRef]
- Lemaire, F.; Dartigues, A.; Rivière, L.M. Properties of substrate made with spent mushroom compost. Acta Hortic. 1985, 172, 13–30. [Google Scholar] [CrossRef]
- Singh, S.; Nain, L. Microorganisms in the conversion of agricultural wastes to compost. Proc. Indian Natl. Sci. Acad. 2014, 80, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Paradelo, R.; Basanta, R.; Barral, M.T. Water-holding capacity and plant growth in compost-based substrates modified with polyacrylamide, guar gum or bentonite. Sci. Hortic. 2019, 243, 344–349. [Google Scholar] [CrossRef]
- Trevisan, S.; Francioso, O.; Quaggiotti, S.; Nardi, S. Humic substances biological activity at the plant-soil interface: From environmental aspects to molecular factors. Plant Signal. Behav. 2010, 5, 635–643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rady, M.M.; Semida, W.M.; Hemida, K.A.; Abdelhamid, M.T. The effect of compost on growth and yield of Phaseolus vulgaris plants grown under saline soil. Int. J. Recycl. Organ. Waste Agric. 2016, 5, 311–321. [Google Scholar] [CrossRef] [Green Version]
- Castellano-Hinojosa, A.; Correa-Galeote, D.; Palau, J.; Bedmar, E.J. Isolation of N2-fixing rhizobacteria from Lolium perenne and evaluating their plant growth promoting traits. J. Basic Microbiol. 2016, 56, 85–91. [Google Scholar] [CrossRef]
- Kuan, K.B.; Othman, R.; Abdul Rahim, K.; Shamsuddin, Z.H. Plant growth-promoting rhizobacteria inoculation to enhance vegetative growth, nitrogen fixation and nitrogen remobilisation of maize under greenhouse conditions. PLoS ONE 2016, 11, e0152478. [Google Scholar] [CrossRef] [Green Version]
- Jasper, J.; Wagstaff, C.; Bell, L. Growth temperature influences postharvest glucosinolate concentrations and hydrolysis product formation in first and second cuts of rocket salad. Postharvest Biol. Technol. 2020, 163, 111157. [Google Scholar] [CrossRef]
- Awan, D.A.; Ahmad, F.; Imdad, M. Influence of row spacing and frequency of cuttings on spinach (Spinacia oleracea) production. J. Bioresour. Manag. 2016, 31, 9. [Google Scholar] [CrossRef]
- Suzuki, T.; Kamada, E.; Ishii, T. Effects of ratoon harvesting on the root systems of processing spinach. Plant Root 2019, 13, 23–28. [Google Scholar] [CrossRef] [Green Version]
- Conrath, U.; Beckers, G.J.M.; Flors, V.; García-Agustín, P.; Jakab, G.; Mauch, F.; Newman, M.A.; Pieterse, C.M.J.; Poinssot, B.; Pozo, M.J. Priming: Getting ready for battle. Mol. Plant Microbe Interact. 2006, 19, 1062–1071. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sang, M.K.; Kim, K.D. Biocontrol activity and primed systemic resistance by compost water extracts against anthracnoses of pepper and cucumber. Phytopathology 2011, 101, 732–740. [Google Scholar] [CrossRef] [PubMed]
- Hoitink, H.A.J.; Grebus, M.E. Status of biological control of plant diseases with composts. Compost Sci. Util. 1994, 2, 6–12. [Google Scholar] [CrossRef]
- Castaño, R.; Borrero, C.; Avilés, M. Organic matter fractions by SP-MAS 13C NMR and microbial communities involved in the suppression of Fusarium wilt in organic growth media. Biol. Control 2011, 58, 286–293. [Google Scholar] [CrossRef]
- Ting, A.S.Y.; Hermanto, A.; Peh, K.L. Indigenous actinomycetes from empty fruit bunch compost of oil palm: Evaluation on enzymatic and antagonistic properties. Biocatal. Agric. Biotechnol. 2014, 3, 310–315. [Google Scholar] [CrossRef]
- González-Sánchez, M.A.; Pérez-Jiménez, R.M.; Pliego, C.; Ramos, C.; De Vicente, A.; Cazorla, F.M. Biocontrol bacteria selected by a direct plant protection strategy against avocado white root rot show antagonism as a prevalent trait. J. Appl. Microbiol. 2009, 109, 65–78. [Google Scholar] [PubMed]
- Földes, T.; Bánhegyi, I.; Herpai, Z.; Varga, L.; Szigeti, J. Isolation of Bacillus strains from the rhizosphere of cereals and in vitro screening for antagonism against phytopathogenic, food-borne pathogenic and spoilage micro-organisms. J. Appl. Microbiol. 2000, 89, 840–846. [Google Scholar] [CrossRef] [Green Version]
- Tian, B.; Yang, J.; Zhang, K.Q. Bacteria used in the biological control of plant-parasitic nematodes: Populations, mechanisms of action, and future prospects. FEMS Microbiol. Ecol. 2007, 61, 197–213. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, H.H. Isolation and identification of biphenyl and polychlorinated biphenyl degrading microbes. Appl. Biochem. Biotechnol. 2017, 170, 391–398. [Google Scholar]
- Kirschbaum, M.U. The temperature dependence of soil organic matter decomposition, and the effect of global warming on soil organic C storage. Soil Biol. Biochem. 1995, 27, 753–760. [Google Scholar] [CrossRef]
- Burns, I.G.; Zhang, K.; Turner, M.K.; Edmondson, R. Iso-osmotic regulation of nitrate accumulation in lettuce. J. Plant Nutr. 2010, 34, 283–313. [Google Scholar] [CrossRef] [Green Version]
- Lakhdar, A.; Falleh, H.; Ouni, Y.; Oueslati, S.; Debez, A.; Ksouri, R.; Abdelly, C. Municipal solid waste compost application improves productivity, polyphenol content, and antioxidant capacity of Mesembryanthemum edule. J. Hazard. Mater. 2011, 191, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Santos, F.T.; Goufo, P.; Santos, C.; Botelho, D.; Fonseca, J.; Queirós, A.; Costa, M.S.S.M.; Trindade, H. Comparison of five agro-industrial waste-based composts as growing media for lettuce: Effect on yield, phenolic compounds and vitamin C. Food Chem. 2016, 209, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Marín, A.; Ferreres, F.; Barberá, G.G.; Gil, M.I. Weather variability influences color and phenolic content of pigmented baby leaf lettuces throughout the season. J. Agric. Food Chem. 2015, 63, 1673–1681. [Google Scholar] [CrossRef]
- Adebooye, O.C.; Schmitz-Eiberger, M.; Lankes, C.; Noga, G.J. Inhibitory effects of sub-optimal root zone temperature on leaf bioactive components, photosystem II (PS II) and minerals uptake in Trichosanthes cucumerina L. Cucurbitaceae. Acta Physiol. Plant. 2010, 32, 67–73. [Google Scholar] [CrossRef]
- Sakamoto, M.; Suzuki, T. Effect of root-zone temperature on growth and quality of hydroponically grown red leaf lettuce (Lactuca sativa L. cv. Red Wave). Am. J. Plant Sci. 2015, 10, 2350. [Google Scholar] [CrossRef] [Green Version]
- Yan, Q.-Y.; Duan, Z.-Q.; Mao, J.-D.; Li, X.; Dong, F. Low root zone temperature limits nutrient effects on cucumber seedling growth and induces adversity physiological response. J. Integr. Agric. 2013, 12, 1450–1460. [Google Scholar] [CrossRef]
- Chadirin, Y.; Hidaka, K.; Sago, Y.; Wajima, T.; Kitano, M. Aplication of temperature stress to root zone II. Effect of the high temperature pre-treatment on quality. Environ. Control Biol. 2011, 49, 157–164. [Google Scholar] [CrossRef]
- Costa, L.D.; Tomasi, N.; Gottardi, S.; Iacuzzo, F.; Cortella, G.; Manzocco, L.; Pinton, R.; Mimmo, T.; Cesco, S. The effect of growth medium temperature on corn salad [Valerianella locusta (L.) laterr] baby leaf yield and quality. HortScience 2011, 46, 1619–1625. [Google Scholar] [CrossRef]
- Salveit, M.E. Wound induced changes in phenolic metabolism and tissue browning are altered by heat shock. Postharvest Biol. Technol. 2000, 21, 61–69. [Google Scholar] [CrossRef]
- Kang, H.M.; Saltveit, M.E. Antioxidant capacity of lettuce leaf tissue increases after wounding. J. Agric. Food Chem. 2002, 26, 7536–7541. [Google Scholar] [CrossRef] [PubMed]
- Chrysargyris, A.; Antoniou, O.; Tzionis, A.; Prasad, M.; Tzortzakis, N. Alternative soilless media using olive-mill and paper waste for growing ornamental plants. Environ. Sci. Pollut. Res. 2018, 25, 35915–35927. [Google Scholar] [CrossRef] [PubMed]

| Composts. | C1 | C2 | C3 | |
|---|---|---|---|---|
| Feedstocks (% dry matter) | Vineyard wastes | 54 | 42 | 41 |
| Tomato wastes | 46 | 25 | 21 | |
| Leek wastes | - | - | 8 | |
| Olive mill cake | - | 33 | 30 | |
| Peat | C1 | C2 | C3 | ||
|---|---|---|---|---|---|
| Physical characteristics | |||||
| BD (g cm−3) | 0.38 ± 0.01 b | 0.19 ± 0.1 a | 0.20 ± 0.1 a | 0.20 ± 0.1 a | *** |
| TPS (Vol %) | 75.12 ± 0.10 | 88.4 ± 0.1 | 87.6 ± 0.2 | 87.9 ± 0.1 | n.s. |
| AC (Vol %) | 20.45 ± 1.23 a | 20.6 ± 0.4 a | 32.7 ± 0.3 b | 34.6 ± 0.7 b | *** |
| WHC (Vol %) | 547 ± 11.25 a | 678 ± 4.5 b | 548 ± 4.9 a | 533 ± 6.5 a | *** |
| Chemical characteristics | |||||
| pH | 5.6 ± 0.03 a | 8.41 ± 0.02 b | 8.59 ± 0.03 b | 8.84 ± 0.08 b | *** |
| EC (dS m−1) | 1.2 ± 0.01 a | 5.44 ± 0.01 d | 3.65 ± 0.04 b | 4.75 ± 0.01 c | *** |
| C/N | 49.6 ± 0.3 | 10.9 ± 0.1 | 13.2 ± 0.5 | 12.2 ± 0.3 | |
| TOC (g kg−1) | 466 ± 0.2 b | 378 ± 1 a | 433 ± 1 b | 404 ± 3 b | *** |
| HA (g kg−1) | 252.1 ± 9.0 c | 44.2 ± 2.0 a | 70.9 ± 1.0 b | 70.5 ± 0.3 b | *** |
| Total N (g kg−1) | 9.4 ± 0.3 a | 36.5 ± 0.5 b | 35.2 ± 0.6 b | 31.3 ± 0.4 b | *** |
| Organic N (g kg−1) | 9.3 ± 0.1 a | 35.1 ± 0.5 b | 34.1 ± 0.5 b | 29.8 ±0.5 b | *** |
| Nitric N (g kg−1) | 0.10 ± 0.03 a | 0.81 ± 0.07 b | 0.51 ± 0.09 b | 1.1 ± 0.09 b | *** |
| Ammonium N (g kg−1) | <0.01 a | 0.61 ± 0.02 b | 0.6 ± 0.03 b | 0.5 ± 0.02 b | *** |
| Total P (P2O5, g kg−1) | 4.5 ± 0.2 a | 21.4 ± 7.8 b | 12.9 ± 5.6 b | 15.1 ± 7.7 b | *** |
| Available P (P2O5, g kg−1) | 4.1 ± 0.1 a | 19.6 ± 0.7 b | 11.7 ± 0.8 b | 14.4 ± 0.6 b | *** |
| Total K (K2O, g kg−1) | 3.2 ± 0.3 a | 28.4 ± 1.8 b | 27.4 ± 3.8 b | 32.3 ± 2.8 b | *** |
| Available K (K2O, g kg−1) | 2.8 ± 0.3 a | 25.4 ± 3.7 b | 23.9 ± 2.8 b | 29.6 ± 3.0 b | *** |
| Ca (g kg−1) | 18.1 ± 3.0 a | 40.0 ± 3.0 b | 22.5 ± 2.2 a | 28.3 ± 0.41 ab | *** |
| Mg (g kg−1) | 1.8 ± 0.8 a | 6.4 ± 0.1 b | 4.0 ± 0.3 b | 4.4 ± 0.8 b | *** |
| B (mg kg−1) | 0.3 ± 0.1 | 0.6 ± 0.1 | 0.4 ± 0.1 | 0.4 ± 0.1 | n.s. |
| Cu (mg kg−1) | 5.5 ± 1.1 a | 21.6 ± 1.2 b | 19.3 ± 1.5 b | 20.1 ± 1.1 b | *** |
| Fe (g kg−1) | 1.2 ± 0.1 | 1.2 ± 0.1 | 0.9 ± 0.1 | 1.2 ± 0.1 | n.s. |
| Mn (mg kg−1) | 70.8 ± 5.7 a | 120.1 ± 2.7 b | 82.4 ± 5.7 ab | 98.6 ± 5.6 ab | *** |
| Mo (mg kg−1) | 1.5 ± 0.1 | 1.2 ± 0.1 | 0.7 ± 0.1 | 1.0 ± 0.3 | n.s. |
| Zn (mg kg−1) | 14.3 ± 1.1 a | 46.4 ± 1.3 b | 28.9 ± 1.8 b | 28.0 ± 1.9 b | *** |
| Biological characteristics | |||||
| Total fungi (log(10) CFUs g−1) | 4.88 ± 0.26 a | 4.51 ± 0.02 a | 5.40 ± 0.29 b | 5.18 ± 0.04 b | *** |
| Total Bacteria (log(10) CFUs g−1) | 7.23 ± 0.08 a | 8.60 ± 0.02 b | 9.19 ± 0.04 c | 9.41 ± 0.01 c | *** |
| DHA (µmol INTF g1− h−1) | 3.7 ± 0.1 a | 16.43 ± 0. 59 b | 20.49 ± 0.89 b | 17.35 ± 0.15 b | *** |
| Compost | C1 | C2 | C3 | ||
|---|---|---|---|---|---|
| Diversity Index | |||||
| Bacteria | Shannon (H) | 7.88 ± 0.14 | 7.80 ± 0.11 | 7.92 ± 0.04 | n.s. |
| Simpson (Ds) | 0.99 ± 0.01 | 0.98 ± 0.01 | 0.99 ± 0.01 | n.s. | |
| Fungi | Shannon (H) | 1.92 ± 0.07 a | 2.60 ± 0.10 b | 2.09 ± 0.14 a | *** |
| Simpson (Ds) | 0.49 ± 0.03 a | 0.73 ± 0.01 c | 0.60 ± 0.03 b | *** | |
| Spring | Summer | |||
|---|---|---|---|---|
| Yield (g Plant−1) | Nitrate Content (mg kg−1 FW) | Yield (g Plant−1) | Nitrate Content (mg kg−1 FW) | |
| Substrate (A) | ||||
| Peat | 1.95 ± 0.15 | 1618.6 ± 24.6 a | 1.28 ± 0.08 a | 1968.2 ± 39.4 a |
| GM1 | 1.91 ± 0.17 | 1806.7 ± 27.3 b | 1.55 ± 0.08 b | 1955.9 ± 58.6 a |
| GM2 | 1.94 ± 0.18 | 1848.6 ± 47.3 b | 1.31 ± 0.06 a | 2035.1 ± 32.0 b |
| GM3 | 2.14 ± 0.22 | 1833.6 ± 80.6 b | 1.40 ± 0.09 a, b | 2000.4 ± 22.2 a, b |
| Cut (B) | ||||
| 1st cut | 1.34 ± 0.04 a | 1654.5 ± 21.0 a | 1.26 ± 0.04 a | 1855.8 ± 18.4 a |
| 2nd cut | 2.63 ± 0.09 b | 1899.2 ± 40.3 b | 1.51 ± 0.07 b | 2124.0 ± 16.4 b |
| A × B | ||||
| Peat × 1st cut | 1.48 ± 0.05 | 1584.0 ± 46.7 a | 1.06 ± 0.04 | 1835.6 ± 34.7 b |
| Peat × 2nd cut | 2.42 ± 0.19 | 1653.3 ± 9.4 a, b | 1.49 ± 0.12 | 2100.7 ± 31.8 d, e |
| GM1 × 1st cut | 1.24 ± 0.07 | 1757.0 ± 20.2 b, c | 1.37 ± 0.03 | 1722.3 ± 25.0 a |
| GM1 × 2nd cut | 2.57 ± 0.09 | 1856.3 ± 46.2 c | 1.76 ± 0.13 | 2189.6 ± 18.5 f |
| GM2 × 1st cut | 1.27 ± 0.09 | 1699.8 ± 34.5 a, b | 1.32 ± 0.08 | 1906.8 ± 12.2 c |
| GM2 × 2nd cut | 2.61 ± 0.13 | 1997.5 ± 52.6 d | 1.35 ± 0.05 | 2163.4 ± 9.0 e, f |
| GM3 × 1st cut | 1.36 ± 0.10 | 1577.3 ± 31.9 a | 1.33 ± 0.07 | 1958.6 ± 5.5 c |
| GM3 × 2nd cut | 2.91 ± 0.24 | 2090.0 ± 99.7 d | 1.46 ± 0.16 | 2042.1 ± 40.4 d |
| Analysis of variance | ||||
| Substrate (A) | n.s. | *** | * | * |
| Cut (B) | *** | *** | ** | *** |
| A × B | n.s. | *** | n.s. | *** |
| Total Phenolic Content (mg GA 100 g−1 DW) | ||
|---|---|---|
| Spring | Summer | |
| Substrate (A) | ||
| Peat | 2467.8 ± 225.6 a | 2638.8 ± 177.5 a |
| GM1 | 2741.8 ± 226.9 a, b | 2932.7 ± 156.2 a, b |
| GM2 | 2838.3 ± 94.4 a, b | 3046.8 ± 216.5 b, c |
| GM3 | 2976.6 ± 192.2 b | 3268.4 ± 24.1 c |
| Cut (B) | ||
| 1st cut | 2461.0 ± 99.2 a | 2577.0 ± 68.0 a |
| 2nd cut | 3051.2 ± 119.7 b | 3366.4 ± 114.4 b |
| A × B | ||
| Peat × 1st cut | 2023.5 ± 133.4 a | 2304.9 ± 121.4 a |
| Peat × 2nd cut | 2912.2 ± 198.3 b, c | 2972.8 ± 176.4 b, c |
| GM1 × 1st cut | 2488.5 ± 144.5 a, b | 2624.2 ± 128.1 a, b |
| GM1 × 2nd cut | 2995.1 ± 415.1 b, c | 3241.1 ± 102.0 c, d |
| GM2 × 1st cut | 2641.7 ± 75.6 a, b, c | 2615.0 ± 72.5 a, b |
| GM2 × 2nd cut | 3035.0 ± 12.2 b, c | 3478.6 ± 206.8 d, e |
| GM3 × 1st cut | 2690.5 ± 182.8 b, c | 2763.9 ± 94.8 b |
| GM3 × 2nd cut | 3262.7 ± 263.5 c | 3772.9 ± 171.9 e |
| Analysis of variance | ||
| Substrate (A) | * | ** |
| Cut (B) | *** | *** |
| A × B | * | *** |
| Total Antioxidant Capacity (mg Trolox Equivalents 100 g−1 DW) | ||||
|---|---|---|---|---|
| Spring | Summer | |||
| ABTS | FRAP | ABTS | FRAP | |
| Substrate (A) | ||||
| Peat | 2552.4 ± 76.0 | 6444.5 ± 183.4 a | 2593.0 ± 114.6 a | 7223.6 ± 98.7 |
| GM1 | 2477.0 ± 93.9 | 6717.3 ± 196.9 a, b | 2654.5 ± 61.4 a | 7807.5 ± 258.6 |
| GM2 | 2755.9 ± 76.9 | 70004.7 ± 224.9 b, c | 2990.6 ± 226.9 b | 7518.7 ± 281.2 |
| GM3 | 2574.5 ± 133.3 | 7290.6 ± 197.3 c | 2906.4 ± 246.5 b | 7661.5 ± 392.2 |
| Cut (B) | ||||
| 1st cut | 2461.4 ± 49.4 a | 6557.0 ± 108.6 a | 2456.8 ± 49.9 a | 7158.3 ± 129.8 a |
| 2nd cut | 2717.5 ± 71.8 b | 7171.5 ± 161.2 b | 3115.4 ± 110.4 b | 7947.4 ± 183.5 b |
| A × B | ||||
| Peat × 1st cut | 2448.2 ± 103.9 a | 6221.2 ± 97.0 a | 2423.4 ± 133.9 a | 7224.9 ± 202.2 a, b |
| Peat × 2nd cut | 2656.5 ± 85.2 a, b, c | 6667.7 ± 330.1 a, b | 2762.7 ± 137.7 b | 7222.8 ± 88.6 a, b |
| GM1 × 1st cut | 2432.9 ± 107.9 a | 6449.6 ± 245.8 a, b | 2545.9 ± 82.5 a, b | 7361.2 ± 335.2 a, b |
| GM1 × 2nd cut | 2521.1 ± 247.0 a, b | 6985.0 ± 248.5 b, c | 2763.0 ± 15.7 b | 8253.7 ± 151.2 c |
| GM2 × 1st cut | 2635.12 ± 82.72 a, b, c | 6518.8 ± 102.4 a, b | 2493.3 ± 114.5 a, b | 6999.64 ± 36.10 a |
| GM2 × 2nd cut | 2872.74 ± 92.97 c | 7490.6 ± 79.2 c | 3484.8 ± 12.7 c | 8037.8 ± 353.1 b, c |
| GM3 × 1st cut | 2329.41 ± 6.17 a | 7038.5 ± 20.3 b, c | 2361.6 ± 84.1 a | 7047.2 ± 417.5 a |
| GM3 × 2nd cut | 2819.57 ± 169.57 b, c | 7542.7 ± 367.6 c | 3451.1 ± 10.8 c | 8275.8 ± 466.0 c |
| Analysis of variance | ||||
| Substrate (A) | n.s. | ** | ** | n.s. |
| Cut (B) | ** | ** | *** | ** |
| A × B | * | * | *** | * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Giménez, A.; Fernández, J.A.; Pascual, J.A.; Ros, M.; Saez-Tovar, J.; Martinez-Sabater, E.; Gruda, N.S.; Egea-Gilabert, C. Promising Composts as Growing Media for the Production of Baby Leaf Lettuce in a Floating System. Agronomy 2020, 10, 1540. https://doi.org/10.3390/agronomy10101540
Giménez A, Fernández JA, Pascual JA, Ros M, Saez-Tovar J, Martinez-Sabater E, Gruda NS, Egea-Gilabert C. Promising Composts as Growing Media for the Production of Baby Leaf Lettuce in a Floating System. Agronomy. 2020; 10(10):1540. https://doi.org/10.3390/agronomy10101540
Chicago/Turabian StyleGiménez, Almudena, Juan A. Fernández, José A. Pascual, Margarita Ros, José Saez-Tovar, Encarnación Martinez-Sabater, Nazim S. Gruda, and Catalina Egea-Gilabert. 2020. "Promising Composts as Growing Media for the Production of Baby Leaf Lettuce in a Floating System" Agronomy 10, no. 10: 1540. https://doi.org/10.3390/agronomy10101540
APA StyleGiménez, A., Fernández, J. A., Pascual, J. A., Ros, M., Saez-Tovar, J., Martinez-Sabater, E., Gruda, N. S., & Egea-Gilabert, C. (2020). Promising Composts as Growing Media for the Production of Baby Leaf Lettuce in a Floating System. Agronomy, 10(10), 1540. https://doi.org/10.3390/agronomy10101540








