Evaluation of Potential Volatile Allelopathic Plants from Bangladesh, with Sapindus mukorossi as a Candidate Species
Abstract
1. Introduction
2. Materials and Methods
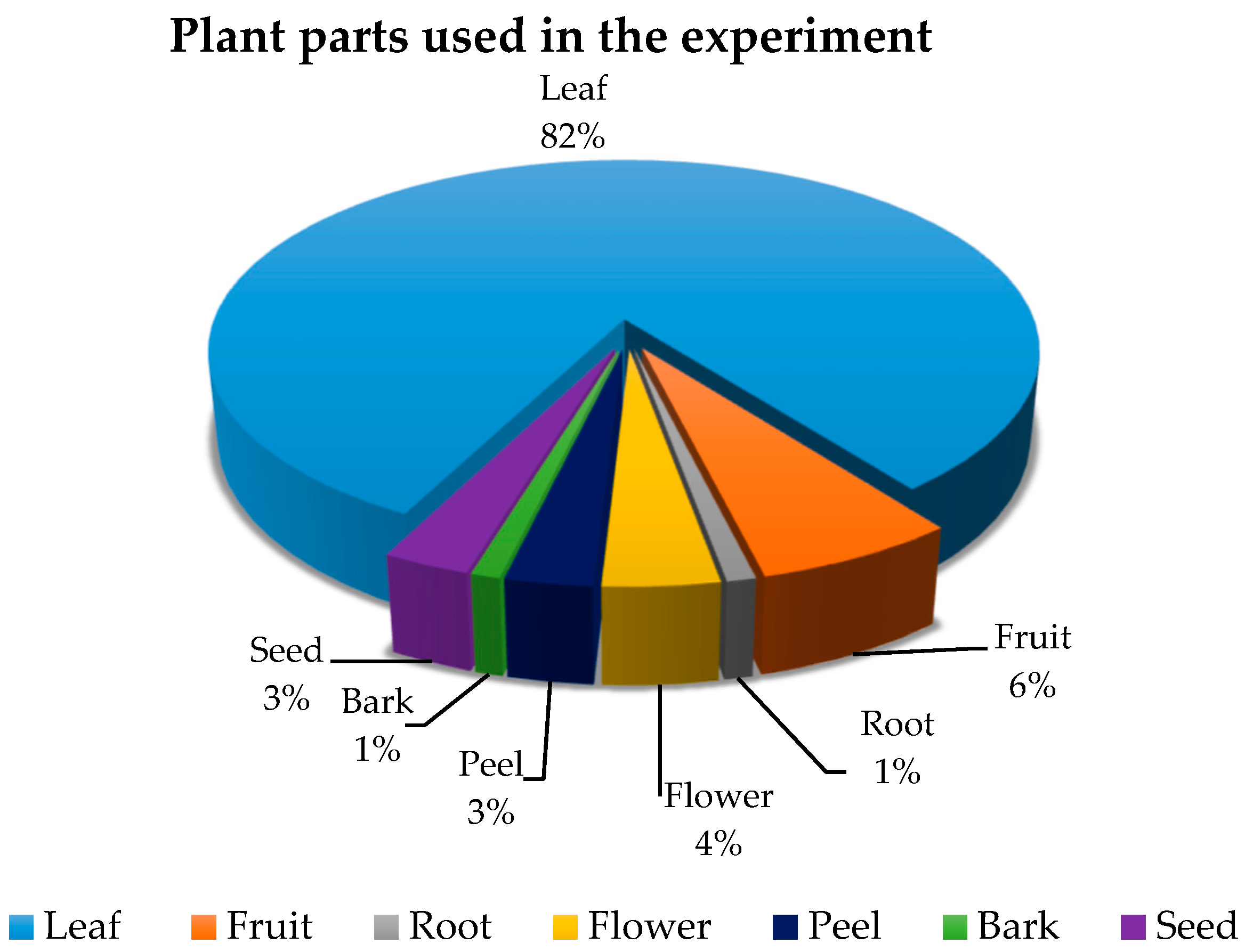
2.1. Plant Samples and Preparation
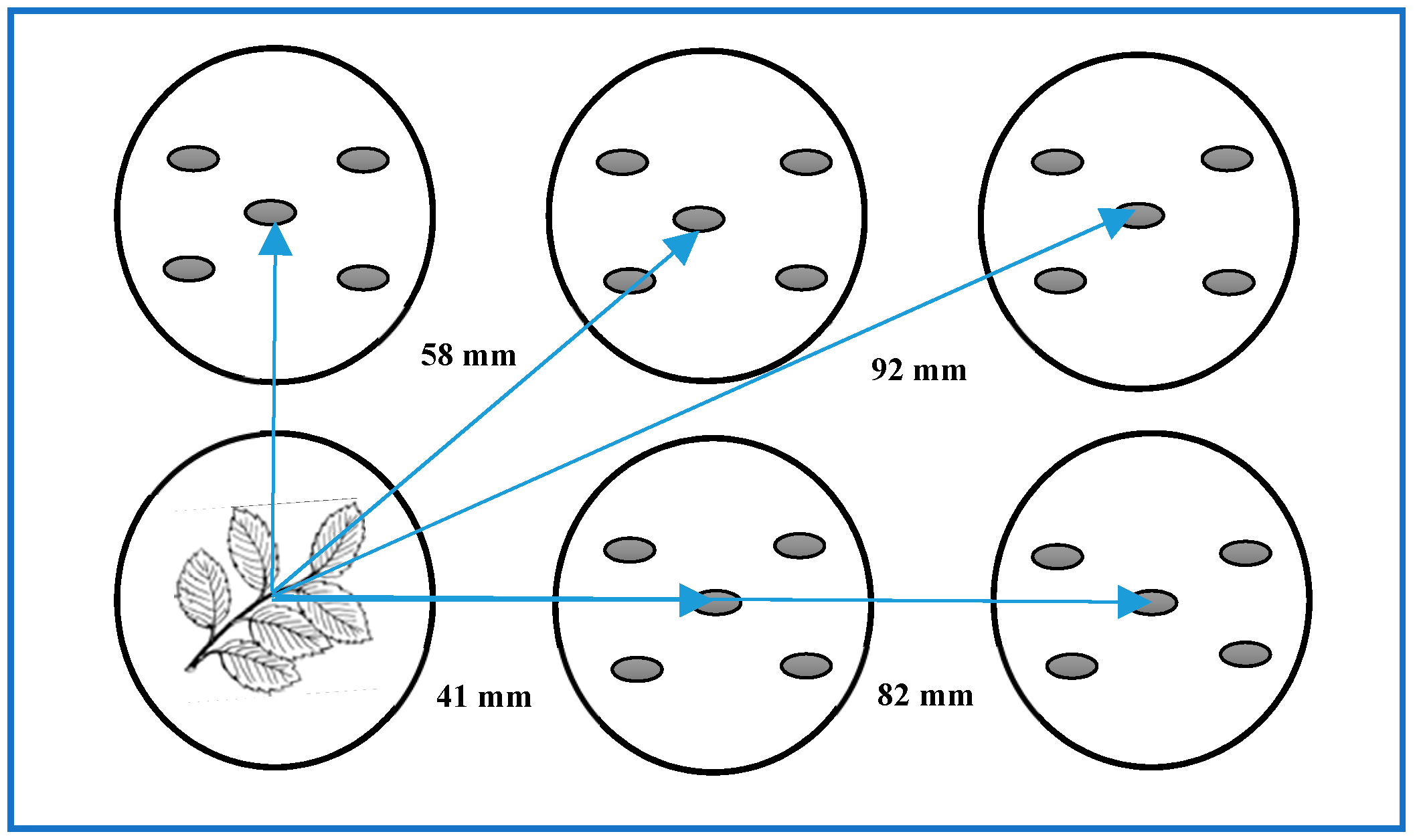
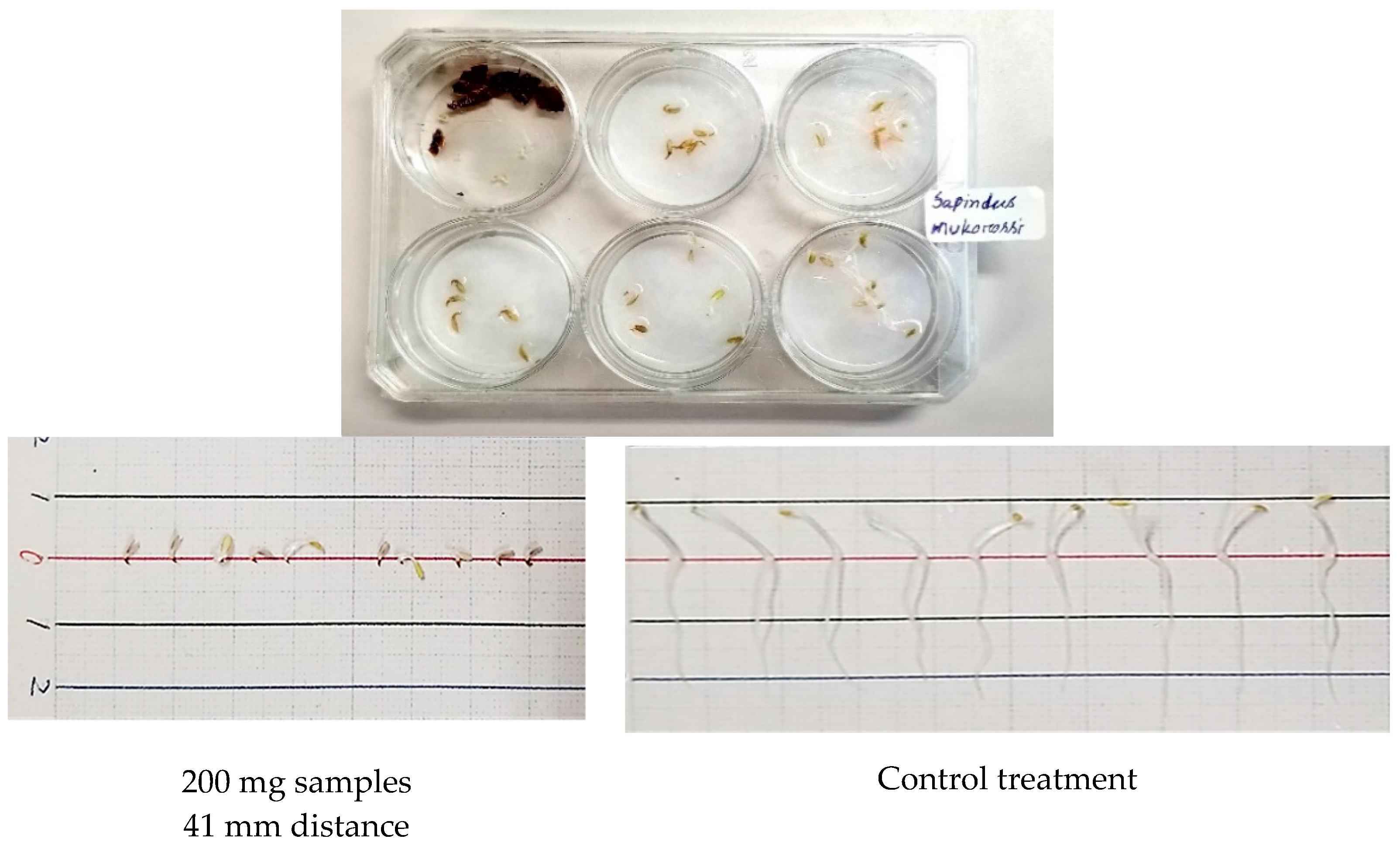
2.2. Dish Pack (DP) Method
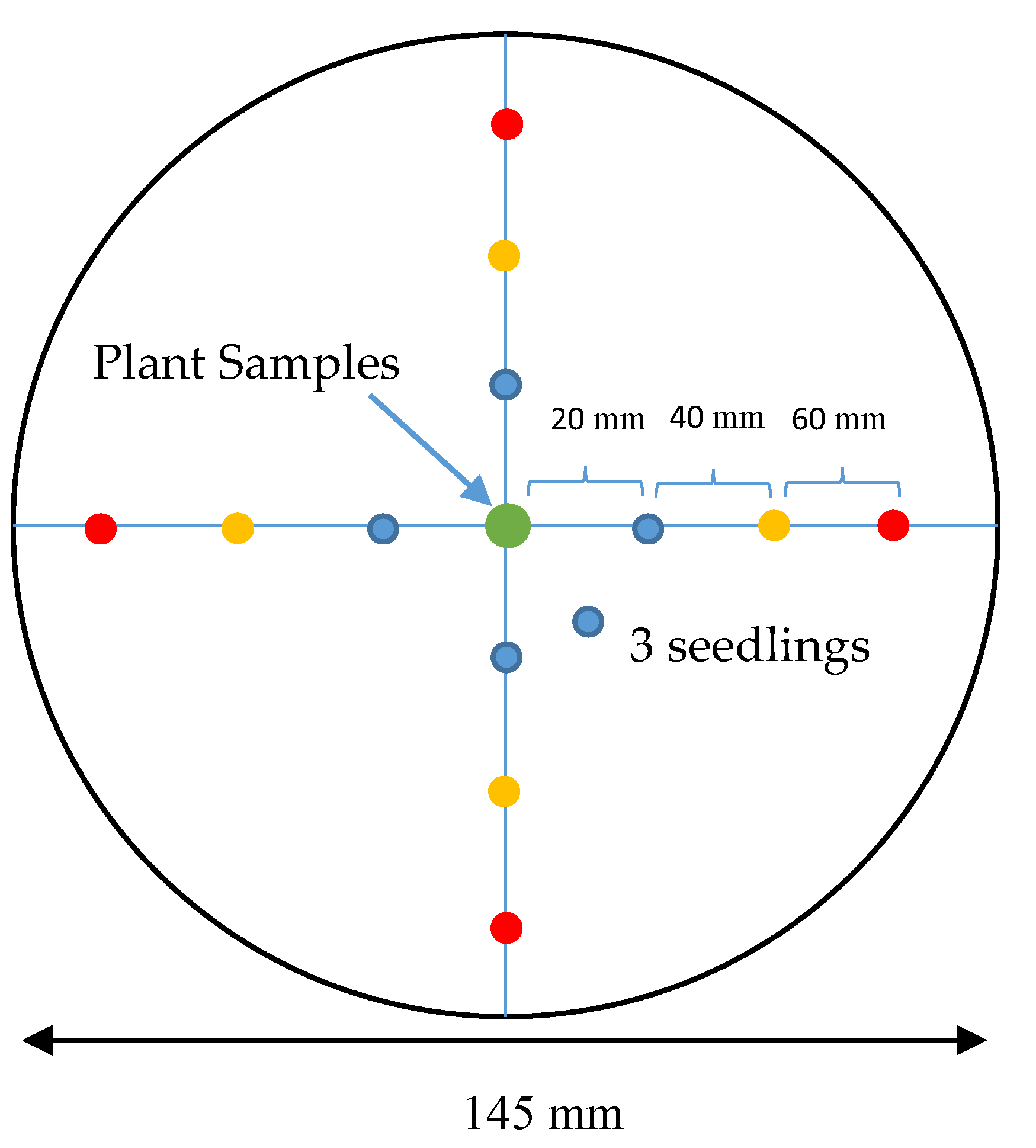
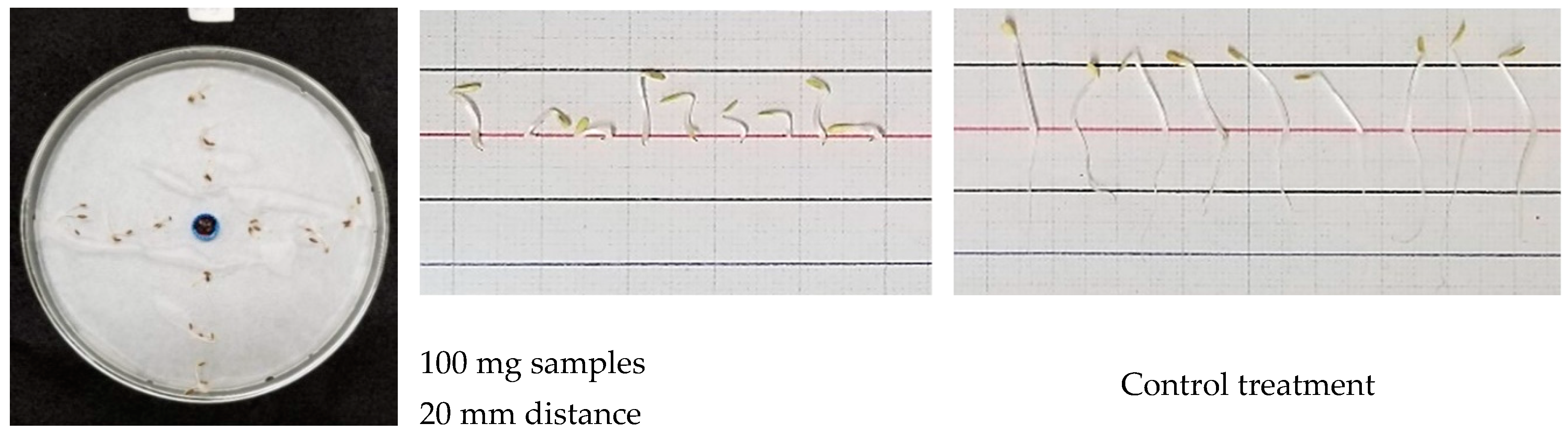
2.3. Single Petri Dish (SPD) Method
2.4. Comparison of Volatile Allelopathy of S. mukorossi by Dish Pack and Single Petri Dish Methods
2.5. Analytical Study
3. Results and Discussion
3.1. Volatile Allelopathic Effect of Evaluated Plant Species
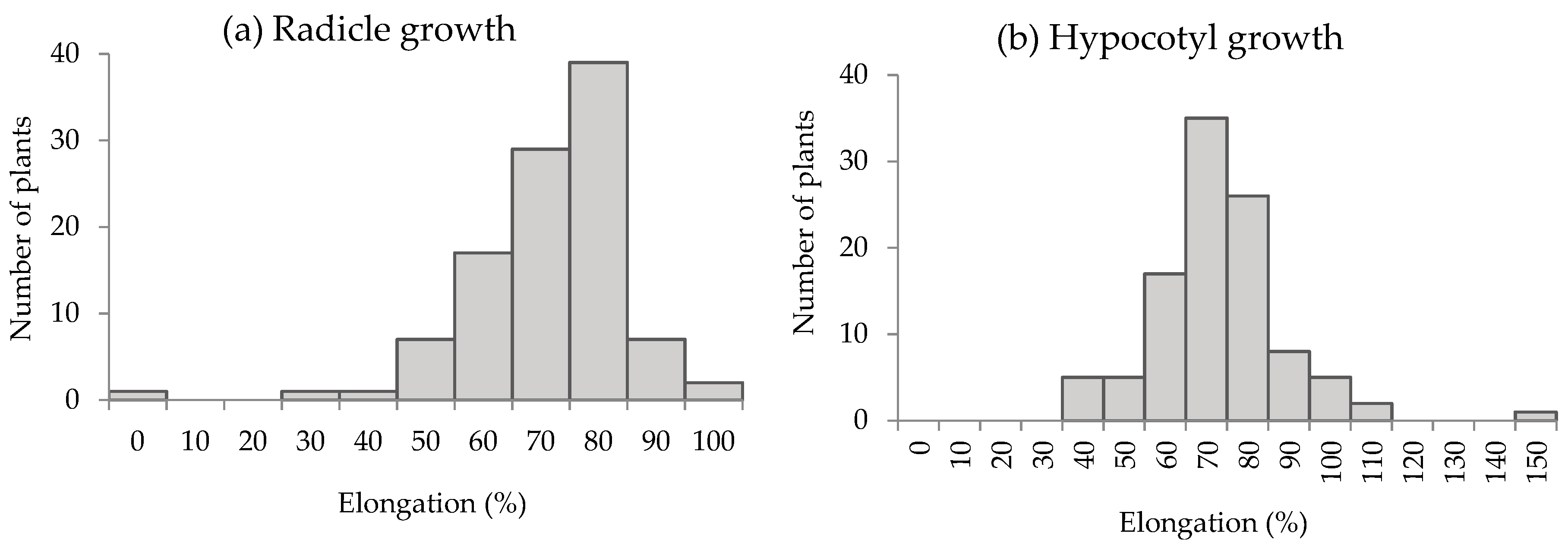
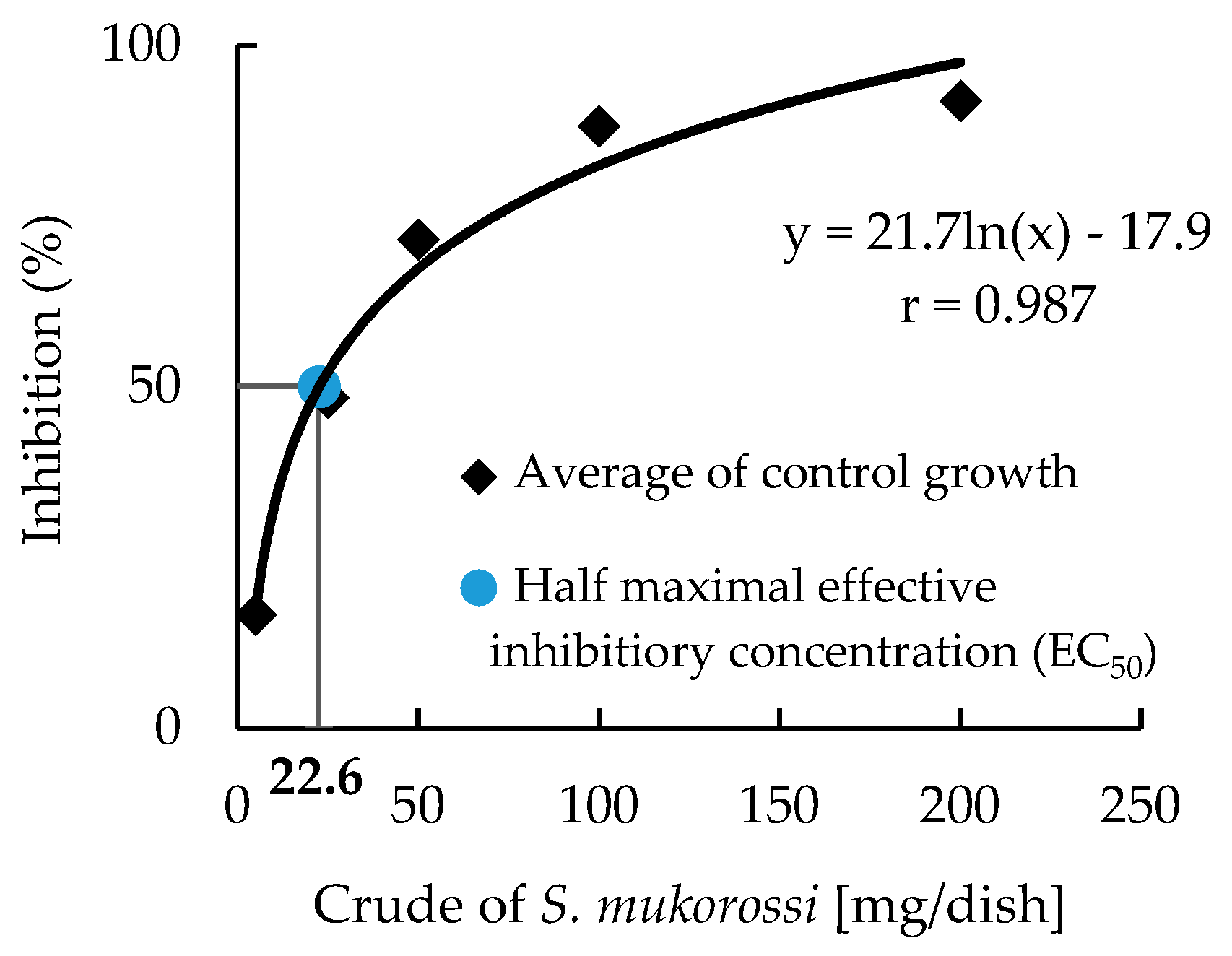
3.2. Growth Inhibitory Activity of S. mukorossi by DP Method
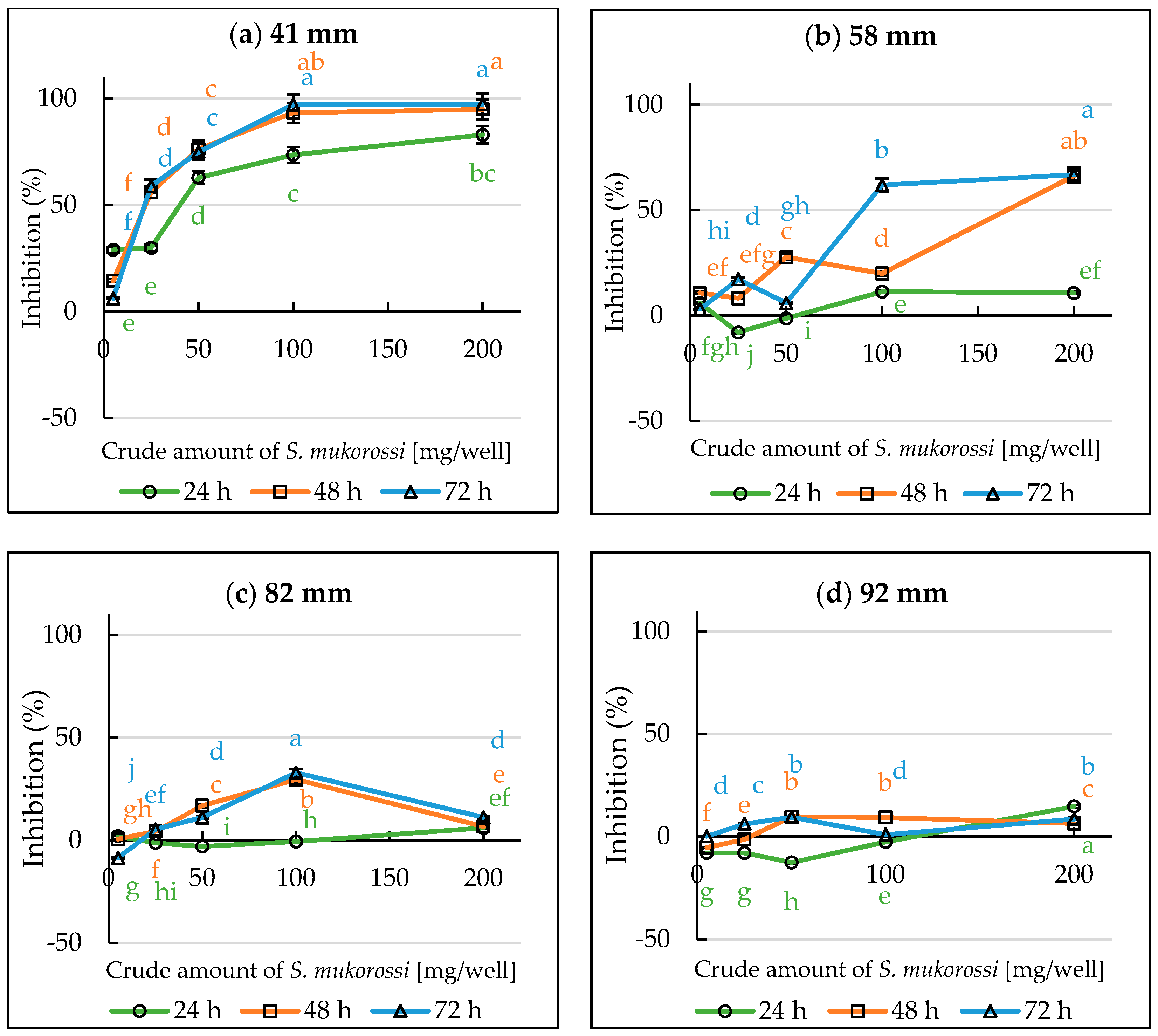
3.3. Inhibitory Effect of S. mukorossi in the Single Petri Dish (SPD) Method Evaluation
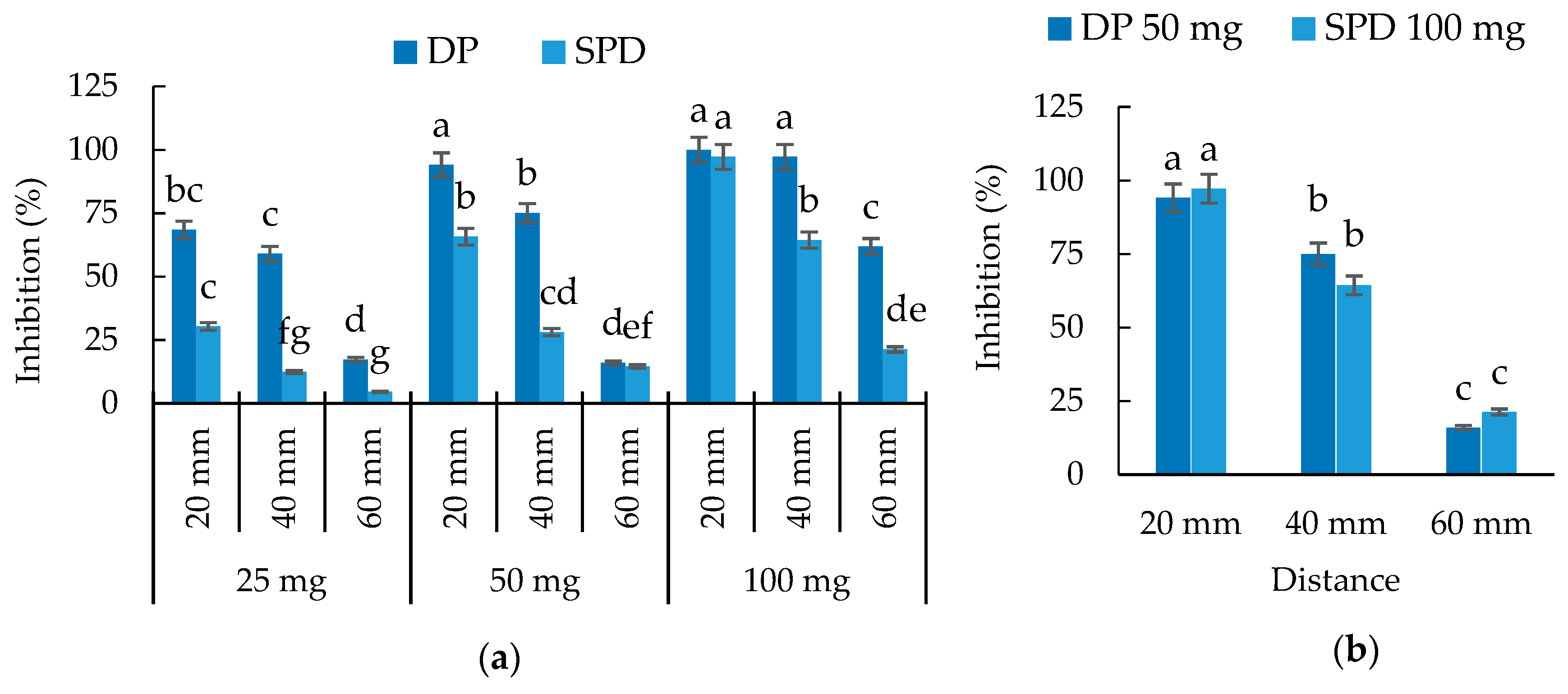
3.4. Evaluation of Volatile Allelopathy of S. mukorossi by Dish Pack Method and Single Petri Dish Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- 2014 Prototype Edition: Sustainable Development Knowledge Platform. Available online: https://sustainabledevelopment.un.org/globalsdreport/2014 (accessed on 15 July 2019).
- MoEF. Bangladesh Climate Change Strategy and Action Plan, 2008; Ministry of Environment and Forests, Government of the People’s Republic of Bangladesh: Dhaka, Bangladesh, 2008.
- Oerke, E. Crop losses to pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- Jabran, K.; Mahajan, G.; Sardana, V.; Chauhan, B.S. Allelopathy for weed control in agricultural systems. Crop Prot. 2015, 72, 57–65. [Google Scholar] [CrossRef]
- Breth, S.A. International Rice Research: 25 Years of Partnership; International Rice Research Inst.: Los Banos, Laguna, Philippines, 1985. [Google Scholar]
- Mamun, A.A. Weeds and Their Control: A Review of Weed Research in Bangladesh; Agricultural and Rural Development in Bangladesh; JSARD, Japan Intl Co-operation Agency: Dhaka. Bangladesh, 1990; Volume 19, pp. 45–72. [Google Scholar]
- Rajcan, I.; Swanton, C.J. Understanding maize–weed competition: Resource competition, light quality and the whole plant. Field Crops Res. 2001, 71, 139–150. [Google Scholar] [CrossRef]
- Heap, I. Global perspective of herbicide-resistant weeds. Pest. Manag. Sci. 2014, 70, 1306–1315. [Google Scholar] [CrossRef] [PubMed]
- Heap, I. The International Survey of Herbicide Resistant Weeds. Available online: http://weedscience.org (accessed on 5 August 2019).
- Weston, L.A.; Duke, S.O. Weed and crop allelopathy. Crit. Rev. Plant Sci. 2003, 22, 367–389. [Google Scholar] [CrossRef]
- Zeng, R.S.; Mallik, A.U.; Luo, S. Allelopathy in Sustainable Agriculture and Forestry; Springer: New York, NY, USA, 2008; Available online: www.springer.com/gp/book/9780387773360 (accessed on 22 January 2019).
- Fujii, Y. Allelopathy in the natural and agricultural ecosystems and isolation of potent allelochemicals from Velvet bean (Mucuna pruriens) and Hairy vetch (Vicia villosa). Biol. Sci. Space 2003, 17, 6–13. [Google Scholar] [CrossRef]
- International Allelopathy Society. International Allelopathy Society. June 2015. Available online: http://allelopathy-society.osupytheas.fr/about/ (accessed on 15 January 2019).
- Vyvyan, J.R. Allelochemicals as leads for new herbicides and agrochemicals. Tetrahedron 2002, 58, 1631–1646. [Google Scholar] [CrossRef]
- Barney, J.N.; Hay, A.G.; Weston, L.A. Isolation and characterization of allelopathic volatiles from mugwort (Artemisia vulgaris). J. Chem. Ecol. 2005, 31, 247–265. [Google Scholar] [CrossRef]
- Soltys, D.; Krasuska, U.; Bogatek, R.; Gniazdowsk, A. Allelochemicals as bioherbicides—Present and perspectives. In Herbicides—Current Research and Case Studies in Use; Price, A., Ed.; InTech: London, UK, 2013. [Google Scholar] [CrossRef]
- Fujii, Y.; Hiradate, S. Allelopathy: New Concepts & Methodology; Enfield, N.H., Ed.; Science Publishers Inc.: Hauppauge, NY, USA, 2007; Available online: https://bit.ly/2Y9DD23 (accessed on 18 January 2019).
- Singh, H.P.; Batish, R.D.; Kohli, R.K. Allelopathic interactions and allelochemicals: New possibilities for sustainable weed management. Crit. Rev. Plant Sci. 2003, 22, 239–311. [Google Scholar] [CrossRef]
- Kropff, M.J.; Walter, H. EWRS and the challenges for weed research at the start of a new millennium. Weed Res. (Oxf.) 2000, 40, 7–10. [Google Scholar] [CrossRef]
- Osbourn, A.E.; Lanzotti, V. Plant-Derived Natural Products: Synthesis, Function, and Application; Springer: Dordrecht, The Netherlands; New York, NY, USA, 2009; Available online: https://www.springer.com/gp/book/9780387854977 (accessed on 23 August 2019).
- El-Deek, M.H.; Hess, F.D. Inhibited Mitotic Entry is the Cause of Growth Inhibition by Cinmethylin. Weed Sci. 1986, 34, 684–688. [Google Scholar] [CrossRef]
- May, J.W.; Goss, J.R.; Moncorge, J.M.; Murphy, M.W. SD 95481 a versatile new herbicide with wide spectrum crop use. In Proceedings of the British Crop Protection Conference, Brighton, UK, 18–21 November 1985; Volume 12, pp. 265–270. [Google Scholar]
- Campe, R.; Hollenbach, E.; Kämmerer, L.; Hendriks, J.; Höffken, H.W.; Kraus, H.; Lerchl, J.; Mietzner, T.; Tresch, S.; Witschel, M.C.; et al. A new herbicidal site of action: Cinmethylin binds to acyl-ACP thioesterase and inhibits plant fatty acid biosynthesis. Pestic. Biochem. Phys. 2018, 148, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Duke, S.O.; Dayan, F.E.; Rimando, A.M.; Schrader, K.K.; Aliatra, G.; Oliva, A.; Romagni, J.G. Chemicals from nature for weed management. Weed Sci. 2002, 50, 138–151. [Google Scholar] [CrossRef]
- Harvey, A. Strategies for discovering drugs from previously unexplored natural products. Drug Discov. Today 2000, 5, 294–300. [Google Scholar] [CrossRef]
- Chu, C.; Mortimer, P.E.; Wang, H.; Wang, Y.; Liu, X.; Yu, S. Allelopathic effects of Eucalyptus on native and introduced tree species. For. Ecol. Manag. 2014, 323, 79–84. [Google Scholar] [CrossRef]
- Macías, F.A.; Lacret, R.; Varela, R.M.; Nogueiras, C.; Molinillo, J.M.G. Isolation and phytotoxicity of terpenes from Tectona grandis. J. Chem. Ecol. 2010, 36, 396–404. [Google Scholar] [CrossRef]
- Macías, F.A.; Marín, D.; Oliveros-Bastidas, A.; Varela, R.M.; Simonet, A.M.; Carrera, C.; Molinillo, J.M. Allelopathy as a new strategy for sustainable ecosystems development. Biol. Sci. Space 2003, 17, 18–23. [Google Scholar] [CrossRef]
- Fujii, Y.; Matsuyama, M.; Hiradate, S.; Shimozawa, H. Dish pack method: A new bioassay for volatile allelopathy. In Proceedings of the 4th World Congress on Allelopathy, “Establishing the Scientific Base”, Wagga, Australia, 21–26 August 2005; pp. 493–497. [Google Scholar]
- de Albuquerque, U.P.; de Medeiros, P.M.; de Almeida, A.L.; Monteiro, J.M.; Machado de Freitas Lins Neto, E.; Gomes de Melo, J.; dos Santos, J.P. Medicinal plants of the caatinga (semi-arid) vegetation of NE Brazil: A quantitative approach. J. Ethnopharmacol. 2007, 114, 325–354. [Google Scholar] [CrossRef]
- Begum, K.; Shammi, M.; Hasan, N.; Asaduzzaman, M.D.; Appiah, K.S.; Fujii, Y. Potential allelopathic candidates for land use and possible sustainable weed management in south asian ecosystem. Sustainability 2019, 11, 2649. [Google Scholar] [CrossRef]
- Dayan, F.E. Factors modulating the levels of the allelochemical sorgoleone in Sorghum bicolor. Planta 2006, 224, 339–346. [Google Scholar] [CrossRef]
- Appiah, K.; Li, Z.; Zeng, R.; Luo, S.; Oikawa, Y.; Fujii, Y. Determination of allelopathic potentials in plant species in Sino-Japanese floristic region by sandwich method and dish pack method. IJBAS 2015, 4, 381. [Google Scholar] [CrossRef]
- Reinhardt, C.F.; Bezuidenhout, S.R. Growth stage of Cyperus esculentus influences it’s allelopathic effect on ectomycorrhizal and higher plant species. J. Crop Prod. 2001, 4, 323–333. [Google Scholar] [CrossRef]
- Mardani, H.; Sekine, T.; Azizi, M.; Mishyna, M.; Fujii, Y. Identification of safranal as the main allelochemical from Saffron (Crocus sativus). Nat. Prod. Commun. 2015, 10, 775–777. [Google Scholar] [CrossRef] [PubMed]
- Turk, M.A.; Rahman, A.; Tawha, M. Inhibitory Effects of Aqueous Extracts of Black mustard on germination and growth of lentil. J. Agron. 2002, 1, 28–30. [Google Scholar] [CrossRef]
- Rao, M.S.; Asad, B.S.; Fazil, M.A.; Sudharshan, R.R.; Rasheed, S.A.; Pradeep, H.A.; Aboobacker, S.; Thayyil, A.H.; Riyaz, A.K.; Mansoor, M.; et al. Evaluation of protective effect of Sapindus mukorossi saponin fraction on CCl4-induced acute hepatotoxicity in rats. Clin. Exp. Gastroenterol. 2012, 5, 129–137. [Google Scholar] [CrossRef]
- Pelegrini, D.D.; Tsuzuki, J.K.; Amado, C.A.B.; Cortez, D.A.G.; Ferreira, I.C.P. Biological activity and isolated compounds in Sapindus saponaria L. and other plants of the genus Sapindus. Lat. Am. J. Pharm. 2008, 27, 922–927. [Google Scholar]
- Upadhyay, A.; Singh, D.K. Pharmacological effects of Sapindus mukorossi. Rev. Inst. Med. Trop. São Paulo 2012, 54, 273–280. [Google Scholar] [CrossRef]
- Kuo, Y.-H.; Huang, H.-C.; Yang Kuo, L.-M.; Hsu, Y.W.; Lee, K.H.; Chang, F.R.; Wu, Y.C. New dammarane-Type Saponins from the galls of Sapindus mukorossi. J. Agric. Food Chem. 2005, 53, 4722–4727. [Google Scholar] [CrossRef]
- Suhagia, B.N.; Rathod, I.S.; Sindhu, S. Sapindus mukorossi (Areetha): An overview. IJPSR 2011, 2, 1905–1913. [Google Scholar]
- Sharma, A.; Sati, S.C.; Sati, O.P.; Sati, M.D.; Kothiyal, S.K. Triterpenoid saponins from the pericarps of Sapindus mukorossi. J. Chem. 2013, 2013, 613190. [Google Scholar] [CrossRef]
- Sarethy, I.P.; Bhatia, N.; Maheshwari, N. Antibacterial activity of plant biosurfactant extract from Sapindus mukorossi and in silico evaluation of its bioactivity. Int. J. Pharm. Pharm. Sci. 2015, 7, 419–421. [Google Scholar]
- Heng, W.; Ling, Z.; Na, W.; Guo, Y.; Weng, Z.; Sun, Z.; Xu, D.; Xie, Y.; Yao, W. Analysis of the bioactive components of Sapindus saponins. Ind. Crops Prod. 2014, 61, 422–429. [Google Scholar] [CrossRef]
- Huang, H.-C.; Liao, S.-C.; Chang, F.-R.; Kuo, Y.-H.; Wu, Y.-C. Molluscicidal saponins from Sapindus mukorossi, Inhibitory Agents of Golden Apple Snails, Pomacea canaliculata. J. Agric. Food Chem. 2003, 51, 4916–4919. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Fu, L.; He, S.; Lu, X.; Wu, Y.; Ma, Z.; Zhang, X. Potent herbicidal activity of Sapindus mukorossi Gaertn. against Avena fatua L. and Amaranthus retroflexus L. Ind. Crops Prod. 2018, 122, 1–6. [Google Scholar] [CrossRef]
- Mishyna, M.; Laman, N.; Prokhorov, V.; Maninang, J.S.; Fujii, Y. Identification of octanal as plant growth inhibitory volatile compound released from Heracleum sosnowskyi fruit. Nat. Prod. Commun. 2015, 10, 771–774. [Google Scholar] [CrossRef]
- Appiah, K.S.; Mardani, H.K.; Omari, R.A.; Eziah, V.Y.; Ofosu-Anim, J.; Onwona-Agyeman, S.; Amoatey, C.A.; Kawada, K.; Katsura, K.; Oikawa, Y.; et al. Involvement of Carnosic Acid in the Phytotoxicity of Rosmarinus officinalis Leaves. Toxins 2018, 10, 498. [Google Scholar] [CrossRef]
- Nishihara, E.; Parvez, M.M.; Araya, H.; Kawashima, S.; Fujii, Y. L-3-(3,4-Dihydroxyphenyl)alanine (L-DOPA), an allelochemical exuded from velvetbean (Mucuna pruriens) roots. Plant Growth Regul. 2005, 45, 113–120. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Bachmann, K.A.; Bailer, A.J.; Bolger, P.M.; Borak, J.; Cai, L.; Cedergreen, N.; Cherian, M.G.; Chiueh, C.C.; Clarkson, T.W.; et al. Biological stress response terminology: Integrating the concepts of adaptive response and preconditioning stress within a hormetic dose–response framework. Toxicol. Appl. Pharmacol. 2007, 222, 122–128. [Google Scholar] [CrossRef]
- Chen, S.; Jeffrey, C. Gynostemma Blume. In Flora of China; Science Press: Beijing, China, 2011; Available online: http://www.efloras.org/florataxon.aspx?flora_id=2&taxon_id=114356 (accessed on 15 September 2019).
- Leuschner, C.; Backes, K.; Hertel, D.; Schipka, F.; Schmitt, U.; Terborg, O.; Runge, M.B. Drought responses at leaf, stem and fine root levels of competitive Fagus sylvatica L. and Quercus petraea (Matt.) Liebl. trees in dry and wet years. For. Ecol. Manag. 2001, 149, 33–46. [Google Scholar] [CrossRef]
- Khan, M.S.I.; Kato-Noguchi, H. Assessment of allelopathic potential of Couroupita guianensis Aubl. Plant Omics 2016, 9, 115–120. [Google Scholar] [CrossRef]
- Owen, C.S. Two dimensional diffusion theory: Cylindrical diffusion model applied to fluorescence quenching. J. Chem. Phys. 1975, 62, 3204–3207. [Google Scholar] [CrossRef]

| Dish Description | Dish Pack (DP) Method (Multiple Six-Wells Plastic Dish) | Single Petri Dish (SPD) Method (Single Glass Petri Dish) |
|---|---|---|
| Initial sample used | 200 mg | 100 mg |
| Sample placement | On a corner | At the centre |
| Diffusion of volatile compound | Might have equal diffusion | Might have equal diffusion to all directions |
| Volume | 120 mL | 240 mL |
| Size of dish | [36 mm (ϕ) × 18 mm (h)] × 6 wells | 145 mm (ϕ) × 20 mm (h) |
| Establishment | Already established [29]. | Developed in this study (resembles all sides of the DP method). |
| Interaction with light | Wrapped with aluminum foil. No interaction with light. | Wrapped with aluminum foil. No interaction with light. |
| Avoid contact with test plant | The test sample placed at the corner well of the six-well multiple dishes. Therefore, no contact with the test plant. | 10 mm small dish used at the centre of the 145 mm glass petri dish to place the sample. Therefore, no contact with the test plant. |
| Test seed | Five seeds in each well. | 4 directions, 3 locations, 3 seeds in each location. |
| Data collection | Three replications each. | Three replications each. |
| Family | Site Code | Botanical Name | Plant Part | Elongation Status | |||
|---|---|---|---|---|---|---|---|
| 41 mm Wells | |||||||
| R% | Score | H% | Score | ||||
| Sapindaceae | LM | Sapindus mukorossi | Fruit | 3.00 | ** ** ** | 59.5 | ** |
| Fabaceae | DNBG | Cassia nodosa | Leaf | 34.4 | ** ** ** | 76.7 | |
| Zingiberaceae | LM | Kaempferia galanga | Root | 43.4 | ** ** | 46.0 | ** * |
| Acanthaceae | CU | Justicia adhatoda | Leaf | 52.1 | ** * | 74.6 | |
| Piperaceae | DNBG | Piper longum | Fruit | 53.5 | ** * | 61.6 | * |
| Apiaceae | SAU | Cuminum cyminum | Seed | 56.1 | ** | 90.5 | |
| Apocynaceae | DNBG | Tabernaemontana dichotoma | Flower | 58.6 | ** | 44.6 | ** ** |
| Myristicaceae | DNBG | Myristica fragrance | Leaf | 59.6 | ** | 82.2 | |
| Asteraceae | SAU | Tagetes erecta | Flower | 59.7 | ** | 44.9 | ** ** |
| Commelinaceae | SAU | Commelina benghalensis | Leaf | 59.8 | ** | 77.5 | |
| Euphorbiaceae | BAU | Euphorbia neriifolia | Leaf | 60.3 | ** | 86.4 | |
| Lecythidaceae | SAU | Couroupita guianensis | Fruit | 61.4 | ** | 69.6 | |
| Anacardiaceae | LM | Mangifera indica | Seed | 62.0 | ** | 51.4 | ** * |
| Sapindaceae | JU | Lepisanthes rubiginosa | Leaf | 62.8 | * | 91.3 | |
| Combretaceae | DNBG | Terminalia chebula | Leaf | 63.1 | * | 45.8 | ** * |
| Asteraceae | SAU | Helianthus annuus | Leaf | 63.2 | * | 56.5 | ** |
| Lamiaceae | CU | Gmelina arborea | Bark | 63.7 | * | 81.0 | |
| Malvaceae | DNBG | Sida cordifolia | Leaf | 64.4 | * | 50.8 | ** * |
| Apiaceae | JU | Cordia dichotoma | Leaf | 65.9 | * | 45.6 | ** * |
| Rubiaceae | DNBG | Mitragyna parvifolia | Leaf | 65.9 | * | 80.8 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Begum, K.; Shammi, M.; Hasan, N.; Appiah, K.S.; Fujii, Y. Evaluation of Potential Volatile Allelopathic Plants from Bangladesh, with Sapindus mukorossi as a Candidate Species. Agronomy 2020, 10, 49. https://doi.org/10.3390/agronomy10010049
Begum K, Shammi M, Hasan N, Appiah KS, Fujii Y. Evaluation of Potential Volatile Allelopathic Plants from Bangladesh, with Sapindus mukorossi as a Candidate Species. Agronomy. 2020; 10(1):49. https://doi.org/10.3390/agronomy10010049
Chicago/Turabian StyleBegum, Kohinoor, Mashura Shammi, Nazmul Hasan, Kwame Sarpong Appiah, and Yoshiharu Fujii. 2020. "Evaluation of Potential Volatile Allelopathic Plants from Bangladesh, with Sapindus mukorossi as a Candidate Species" Agronomy 10, no. 1: 49. https://doi.org/10.3390/agronomy10010049
APA StyleBegum, K., Shammi, M., Hasan, N., Appiah, K. S., & Fujii, Y. (2020). Evaluation of Potential Volatile Allelopathic Plants from Bangladesh, with Sapindus mukorossi as a Candidate Species. Agronomy, 10(1), 49. https://doi.org/10.3390/agronomy10010049









