Do Triticum aestivum L. and Triticum spelta L. Hybrids Constitute a Promising Source Material for Quality Breeding ofNew Wheat Varieties?
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Experiment
2.2. Biometric Measurements
2.3. Analysis of the Content of Macronutrients in Grain
2.4. Macro Element and Micro Element Analyses
2.4.1. Reagents and Standard Solutions
2.4.2. Digestion Procedure
2.4.3. ICP-SFMS Measurements
2.5. Statistical Analysis
3. Results
3.1. Yield Components
3.2. Macronutrient Analysis
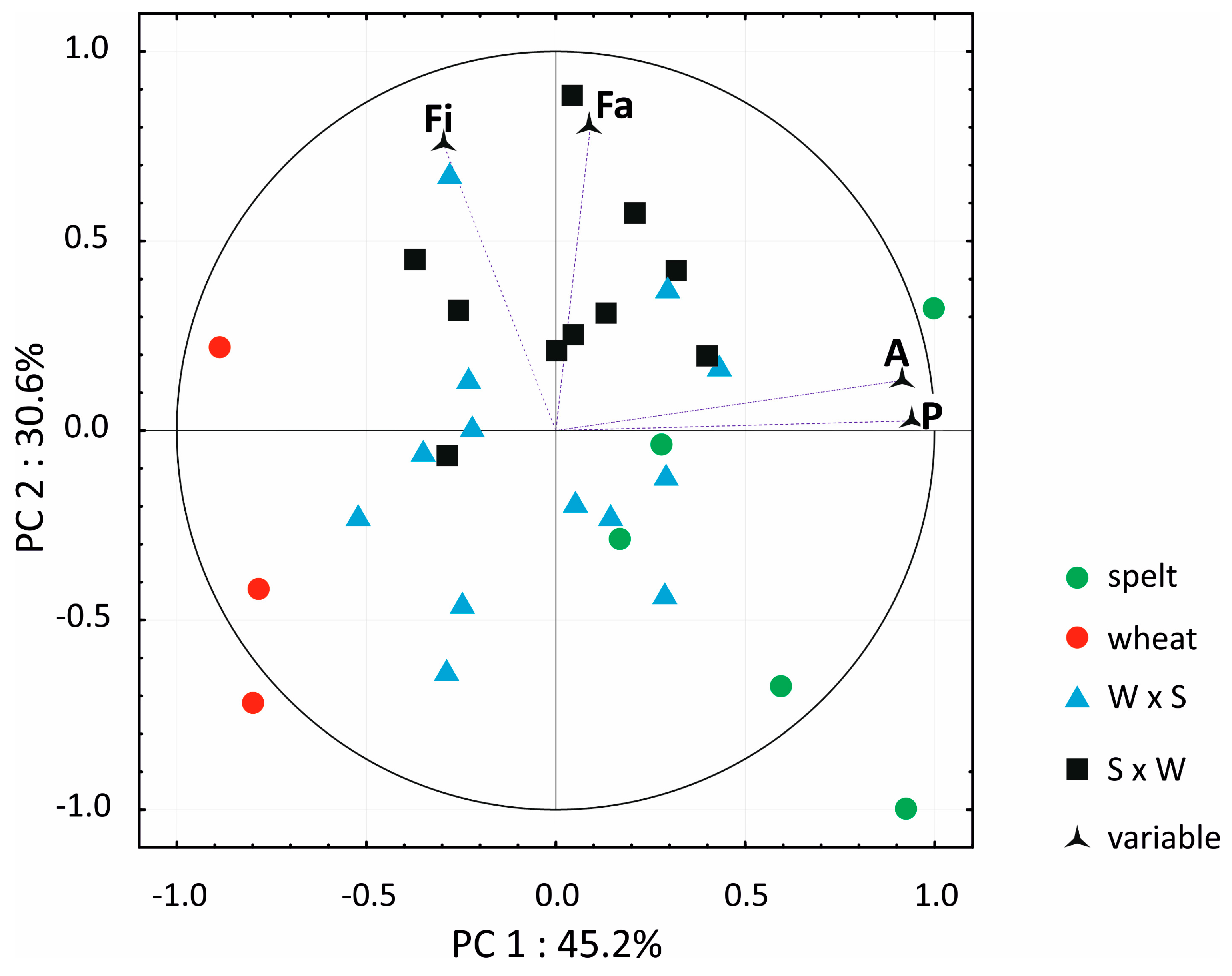
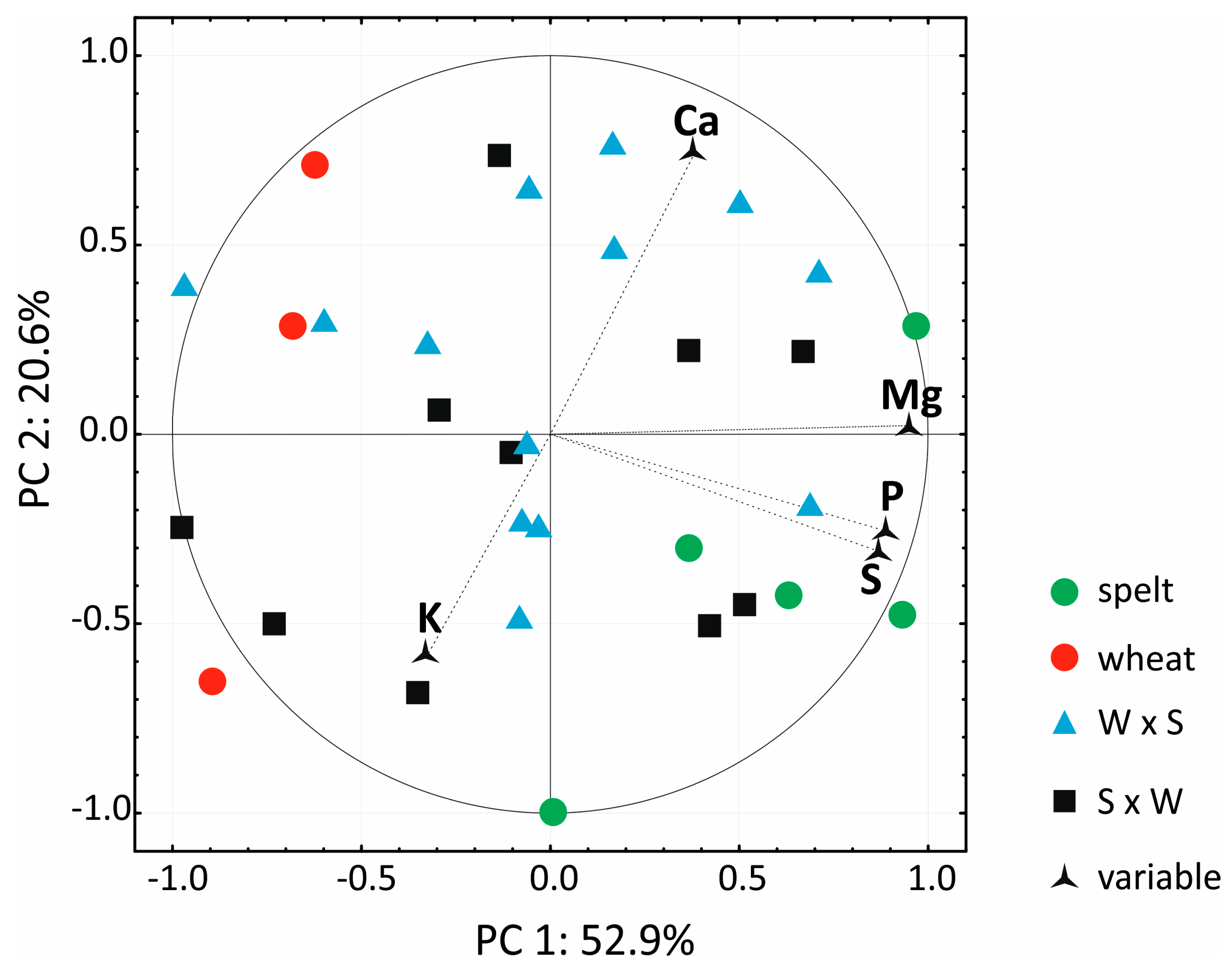
3.3. Macro Element Analysis
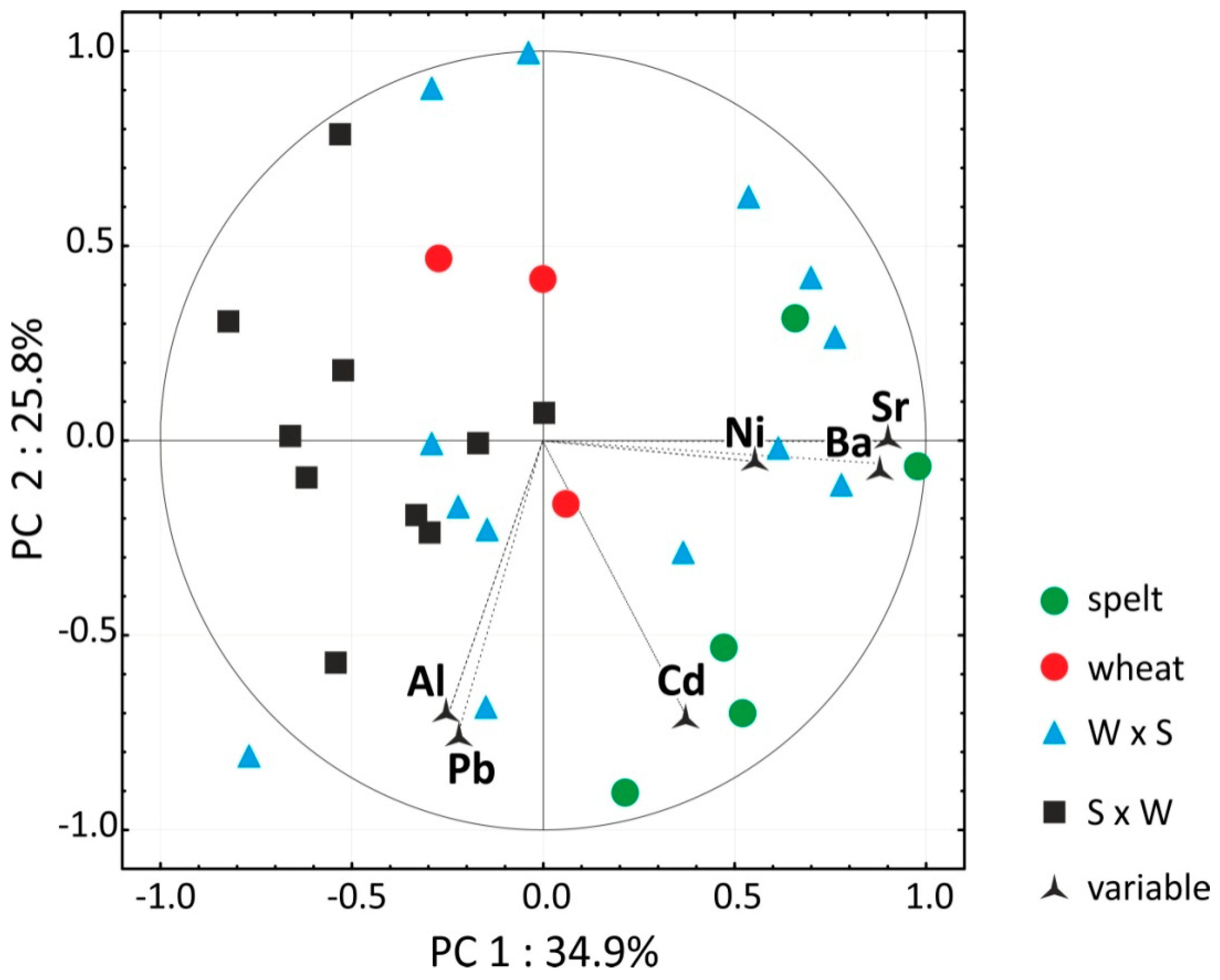
3.4. Micro Element Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- The Statistics Portal for Market Data, Market Research and Market Studies. Available online: https://www.statista.com/topics/1668/wheat/ (accessed on 2 November 2019).
- North America’s Grain Marketplace. Available online: https://farmlead.com/blog/graincents/april-8-durum-wheat-weekly-graincents-digest/ (accessed on 2 November 2019).
- Callejo, M.J.; Vargas-Kostiuk, M.E.; Rodríguez-Quijano, M. Selection, training and validation process of a sensory panel for bread analysis: Influence of cultivar on the quality of breads made from common wheat and spelt wheat. J. Cereal Sci. 2015, 61, 55–62. [Google Scholar] [CrossRef]
- Fan, M.S.; Zhao, F.J.; Fairweather-Tait, S.J.; Poulton, P.R.; Dunham, S.J.; McGrath, S.P. Evidence of decreasing mineral density in wheat grain over the last 160 years. J. Trace Elem. Med. Biol. 2008, 22, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.J.; Su, Y.H.; Dunham, S.J.; Rakszegi, M.; Bedo, Z.; McGrath, S.P.; Shewry, P.R. Variation in mineral micronutrient concentrations in grain of wheat lines of diverse origin. J. Cereal Sci. 2009, 49, 290–295. [Google Scholar] [CrossRef]
- Ward, J.L.; Poutanen, K.; Gebruers, K.; Piironen, V.; Lampi, A.-M.; Nyström, L.; Andersson, A.A.M.; Åman, P.; Boros, D.; Rakszegi, M.; et al. The HEALTHGRAIN cereal diversity screen: Concept, results, and prospects (Conference Paper). J. Agric. Food Chem. 2008, 21, 9699–9709. [Google Scholar] [CrossRef]
- Sharma, P.; Aggarwal, P.; Kaur, A. Biofortification: A new approach to eradicate hidden hunger. Food Rev. Int. 2016, 33, 1–21. [Google Scholar] [CrossRef]
- WHO. Guidelines on Food Fortification with Micronutrients; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Bouis, H.E.; Welch, R.M. Biofortification—A sustainable agricultural strategy for reducing micronutrient malnutrition in the global south. Crop Sci. 2010, 50, 20–30. [Google Scholar] [CrossRef]
- Piergiovanni, A.R.; Rizzi, R.; Pannacciulli, E.; Della Gatta, C. Mineral composition in hulled wheat grains: A comparison between emmer (Triticum dicoccon Schrank) and spelt (T. spelta L.) accessions. Int. J. Food Sci. Nutr. 1997, 4, 381–386. [Google Scholar] [CrossRef]
- Tolonen, M. Vitamins and Minerals in Health and Nutrition; Woodhead Publishing Limited: Cambridge, UK, 1990; pp. 148–151. [Google Scholar]
- Diaz, J.R.; de Las Cagigas, A.; Rodriguez, R. Micronutrient deficiencies in developing and affluent countries. Eur. J. Clin. Nutr. 2003, 57, 70–72. [Google Scholar] [CrossRef]
- Whatham, A.; Bartlett, H.; Eperjesi, F.; Blumenthal, C.; Allen, J.; Suttle, C.; Gaskin, K. Vitamin and mineral deficiencies in the developed world and their effect on the eye and vision. Ophthalmic Physiol. Opt. 2008, 28, 1–12. [Google Scholar] [CrossRef]
- Bhullar, N.K.; Gruissem, W. Nutritional enhancement of rice for human health: The contribution of biotechnology. Biotechnol. Adv. 2013, 31, 50–57. [Google Scholar] [CrossRef]
- Miller, D.D.; Welch, R.M. Food system strategies for preventing micronutrient malnutrition. ESA Work. Pap. 2013, 13, 1–34. [Google Scholar] [CrossRef]
- Quintaes, K.D.; Barberá, R.; Cilla, A. Iron bioavailability in iron-fortified cereal foods: The contribution of in vitro studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 2028–2041. [Google Scholar] [CrossRef] [PubMed]
- International Zinc Association. Available online: https://www.zinc.org/publications/ (accessed on 4 October 2019).
- Neeraja, C.N.; Kulkarni, K.S.; Madhu Babu, P.; Sanjeeva Rao, D.; Surekha, K.; Ravindra Babu, V. Transporter genes identified in landraces associated with high zinc in polished rice through panicle transcriptome for biofortification. PLoS ONE 2018, 13, e0192362. [Google Scholar]
- Velu, G.; Bhattacharjee, R.; Rai, K.N.; Sahrawat, K.L.; Longvah, T. A simple and rapid screening method for grain zinc content in pearl millet. J. SAT Agric. Res. 2018, 6, 1–4. [Google Scholar]
- Silva, C.A.; Abreu, A.F.B.; Ramalho, M.A.P. Genetic control of zinc and iron concentration in common bean seeds. Afr. J. Agric. Res. 2013, 8, 1001–1008. [Google Scholar]
- Pandey, A.; Khan, M.K.; Hakki, E.E.; Thomas, G.; Hamurcu, M.; Gezgin, S.; Gizlenci, O.; Akkaya, M.S. Assessment of genetic variability for grain nutrients from diverse regions potential for wheat improvement. Springer Plus 2016, 5, 1912. [Google Scholar] [CrossRef]
- Biel, W.; Stankowski, S.; Jaroszewska, A.; Pużyński, S.; Bośko, P. The influence of selected agronomic factors on the chemical composition of spelt wheat (Triticum aestivum ssp. spelta L.) grain. J. Integr. Agric. 2016, 15, 1763–1769. [Google Scholar] [CrossRef]
- Kohajdová, Z.; Karovicová, J. Nutritional value and baking applications of spelt wheat. Acta Sci. Pol. Technol. Aliment. 2008, 7, 5–14. [Google Scholar]
- Ruibal-Mendieta, N.L.; Delacroix, D.L.; Mignolet, J.M.P.; Marques, C.; Rozenberg, R.; Petitjean, G.; Habib-Jiwan, J.L.; Meurens, M.; Qeentin-Leclerco, J.; Delzenne, N.M.; et al. Spelt (Triticum aestivum ssp. spelta) as a source of bread making flours and bran naturally enriched in oleic acid and minerals but not phytic acid. J. Agric. Food Chem. 2005, 53, 2751–2759. [Google Scholar] [CrossRef]
- Suchowilska, E.; Wiwart, M.; Kandler, W.; Krska, R. A comparison of macro- and microelement concentrations in the wholegrain of four Triticum species. Plant Soil Environ. 2012, 58, 141–147. [Google Scholar] [CrossRef]
- Frakolaki, G.; Giannou, V.; Topakas, E.; Tzia, C. Chemical characterization and bread making potential of spelt versus wheat flour. J. Cereal Sci. 2018, 79, 50–56. [Google Scholar] [CrossRef]
- Dvorak, J.; Deal, K.R.; Luo, M.-C.; You, F.M.; von Borstel, K.; Dehghan, H. The Origin of Spelt and Free-Threshing Hexaploid Wheat. J. Hered. 2012, 103, 426–441. [Google Scholar] [CrossRef] [PubMed]
- Wiwart, M.; Suchowilska, E.; Lajszner, W.; Graban, Ł. Identification of hybrids of spelt and wheat and their parental forms using shape and color descriptors. Comput. Electron. Agric. 2012, 83, 68–76. [Google Scholar] [CrossRef]
- Srinivasa, J.; Arun, B.; Mishra, V.K.; Chand, R.; Sharma, D.; Bhardwaj, S.C.; Joshi, A.K. Accessing spelt gene pool to develop well-adapted Zinc-and Iron-Rich bread wheat. Crop Sci. 2014, 54, 2000–2010. [Google Scholar] [CrossRef]
- Cakmak, I. Enrichment of cereal grains with zinc: Agronomic or genetic biofortification? Plant Soil 2008, 302, 1–17. [Google Scholar] [CrossRef]
- Schmid, J.E.; Winzeler, M.; Winzeler, H. Analysis of disease resistance and quality characters of F1 hybrids of crosses between wheat (Triticum aestivum) and spelt (Triticum spelta). Euphytica 1994, 75, 105–110. [Google Scholar] [CrossRef]
- Gacek, E. Polish National List of Agricultural Varieties; Research Center for Cultivar Testing: Słupia Wielka, Poland, 2013. [Google Scholar]
- Witzenberger, A.H.; Hack, H.; van den Boom, T. Erläuterungen zum BBCH-Dezimal-Code für die Entwicklungsstadien des Getreides—mit Abbildungen. Gesunde Pflanz. 1989, 41, 384–388. [Google Scholar]
- Wiwart, M.; Suchowilska, E.; Kandler, W.; Sulyok, M.; Groenwald, P.; Krska, R. Can Polish wheat (Triticum polonicum L.) be an interesting gene source for breeding wheat cultivars with increased resistance to Fusarium head blight? Genet. Resour. Crop Evol. 2013, 60, 2359–2373. [Google Scholar] [CrossRef]
- Suchowilska, E.; Szafrańska, A.; Słowik, E.; Wiwart, M. Flour from Triticum polonicum L. as a potential ingredient in bread production. Cereal Chem. 2019, 96, 554–563. [Google Scholar] [CrossRef]
- Hoseney, R.C. Principles of Cereal Science and Technology; American Association of Cereal Chemists, Ed.; American Association of Cereal Chemists Inc.: StPaul, MN, USA, 1994; pp. 65–79, 87–93. [Google Scholar]
- Bieńkowska, T.; Suchowilska, E.; Kandler, W.; Krska, R.; Wiwart, M. Triticum polonicum L. as potential source material for the biofortification of wheat with essential micronutrients. Plant Genet. Resour. 2019, 17, 213–220. [Google Scholar] [CrossRef]
- TIBCO Software Inc. Statistica (Data Analysis Software System), version 13. 2017. Available online: http://statistica.io (accessed on 25 December 2019).
- European Commission. Commission Regulation (EC) No.1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in food stuffs. Off. J. Eur. Union 2006, 364, 5–24. [Google Scholar]
- Rogers, M.A.M.; Simon, D.G. Aluminium intake and risk of Alzheimer’s disease. Age Ageing 1999, 28, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Arzani, A.; Ashraf, M. Cultivated ancient wheats (Triticum spp.): A potential source of health-beneficial food products. Compr. Rev. Food Sci. Food Saf. 2017, 16, 477–488. [Google Scholar] [CrossRef]
- Gomez-Becerra, H.F.; Erdem, H.; Yazici, A.; Tutus, Y.; Torun, B.; Ozturk, L.; Cakmak, I. Grain concentrations of protein and mineral nutrients in a large collection of spelt wheat grown under different environments. J. Cereal Sci. 2010, 52, 342–349. [Google Scholar] [CrossRef]
- Miedaner, T.; Longin, C.F.H. Neglected Cereals: from Ancient Grains to Superfood; Agrimedia: Clenze, Germany, 2016; p. 157. [Google Scholar]
- Diordiieva, I.; Riabovol, L.; Riabovol, I.; Serzhyk, O.; Novak, A.; Kotsiuba, S. The characteristics of wheat collection samples created by Triticum aestivum L./Triticum spelta L.hybridization. Agron. Res. 2018, 16, 2005–2015. [Google Scholar]
- Wiwart, M.; Szafrańska, A.; Wachowska, U.; Suchowilska, E. Quality Parameters and Rheological Dough Properties of 15 Spelt (Triticum spelta L.) Varieties Cultivated Today. Cereal Chem. 2017, 94, 1037–1044. [Google Scholar]
- Rapp, M.; Beck, H.; Gütler, H.; Heilig, W.; Starck, N.; Römer, P.; Cuendet, C.; Kurz, H.; Uhlig, F.; Würschum, T.; et al. Spelt: Agronomy, Quality, and Flavor of Its Breads from 30 Varieties Tested across Multiple Environments. Crop Sci. 2017, 57, 1–9. [Google Scholar] [CrossRef]
- Graham, R.D.; Welch, R.M.; Saunders, D.A.; Ortiz-Monasterio, I.; Bouis, H.E.; Bonierbale, M.S.; deHaan, S.; Burgos, G.; Thiele, G.; Liria, R.; et al. Nutritious subsistence food systems. Adv. Agron. 2007, 92, 1–74. [Google Scholar]
- Lyons, G.; Stangoulis, J.; Graham, R. High-selenium wheat: Biofortification for better health. Nutr. Res. Rev. 2003, 16, 45–60. [Google Scholar] [CrossRef]
- Breuer, F.; Longin, F. Alte Weizenarten. Ein Comeback fast vergessener Kulturpflanzen. Backwarenaktuell 2011, 3, 9–13. [Google Scholar]
- Lachman, J.; Miholová, D.; Pivec, V.; Jírů, K.; Janovská, D. Content of phenolic antioxidants and selenium in grain of einkorn (Triticum monococcum), emmer (Triticum dicoccum) and spring wheat (Triticum aestivum) varieties. Plant Soil Environ. 2011, 57, 235–243. [Google Scholar] [CrossRef]
- Eurola, M.; Ekholm, P.; Ylinen, M.; Koivistoinen, P.; Varo, P. Effects of selenium fertilization on the selenium content of cereal grains, flour, and bread produced in Finland. Cereal Chem. 1990, 67, 334–337. [Google Scholar]
- Liu, M.; Zhao, Q.; Qi, F.; Stiller, J.; Tang, S.; Miao, J.; Vrána, J.; Holušová, K.; Liu, D.; Doležel, J.; et al. Sequence divergence between spelt and common wheat. Theor. Appl. Genet. 2018, 131, 1125–1132. [Google Scholar] [CrossRef] [PubMed]
- Zanetti, S.; Winzeler, M.; Keller, M.; Keller, B.; Messmer, M. Genetic analysis of pre-harvest sprouting resistance in a wheat x spelt cross. Crop Sci. 2000, 40, 1406–1417. [Google Scholar] [CrossRef]
- Sakai, Y.; Cao, L.; Funata, R.; Shiraishi, T.; Yoshikawa, K.; Maeno, K.; Miura, H.; Onishi, K. QTLs for agronomic traits detected in recombinant inbred lines derived from a bread wheat × spelt cross. Breed. Sci. 2018, 68, 587–595. [Google Scholar] [CrossRef]

| No | Parent | No | Hybrid | No | Hybrid | No | Hybrid |
|---|---|---|---|---|---|---|---|
| 1 | S10 | 1 | Torka × S10 | 9 | Kontesa × S14 | 17 | S13 × Torka |
| 2 | S11 | 2 | Torka × S11 | 10 | Zebra × S10 | 18 | S14 × Torka |
| 3 | S12 | 3 | Torka × S12 | 11 | Zebra × S11 | 19 | S10 × Kontesa |
| 4 | S13 | 4 | Torka × S14 | 12 | Zebra × S12 | 20 | S11 × Kontesa |
| 5 | S14 | 5 | Kontesa × S10 | 13 | Zebra × S14 | 21 | S12 × Kontesa |
| 6 | Torka | 6 | Kontesa × S11 | 14 | S10 × Torka | 22 | S13 × Kontesa |
| 7 | Kontesa | 7 | Kontesa × S12 | 15 | S11 × Torka | 23 | S14 × Kontesa |
| 8 | Zebra | 8 | Kontesa × S13 | 16 | S12 × Torka |
| SL (cm) | D | KNS | KWS (g) | OKW (mg) | Yield † (t ha−1) | |
|---|---|---|---|---|---|---|
| Spelt (n = 5) | 12.16 a ± 1.46 | 14.41 c ± 1.09 | 23.41 c ± 0.93 | 0.92 c ± 0.10 | 39.11 ± 3.79 | 4.48 a ± 0.57 |
| Wheat (n = 3) | 9.40 b ± 0.64 | 21.07 a ± 0.90 | 40.39 a ± 0.65 | 1.67 a ± 0.10 | 41.06 ± 2.57 | 5.98 a ± 0.44 |
| W × S (n = 13) | 9.92 b ± 0.78 | 18.02 b ± 1.02 | 29.85 b ± 2.00 | 1.24 b ± 0.06 | 41.74 ± 2.45 | 4.31 ab ± 0.59 |
| S × W (n = 10) | 10.21 b ± 1.88 | 18.11 b ± 2.25 | 29.83 b ± 2.60 | 1.25 b ± 0.14 | 41.81 ± 3.19 | 4.02 b ± 0.62 |
| Ash | Crude Fiber | Crude Protein | Crude Fat | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | |
| Spelt (n = 5) | 2.15 v | 1.87 v–y | 2.01 a | 2.23 y | 2.28 y | 2.25 b | 15.60 | 13.74 | 14.67 a | 2.37 xy | 2.12 y | 2.24 ab |
| Wheat (n = 3) | 1.88 v–y | 1.36 z | 1.62 b | 2.85 x | 2.35 xy | 2.60 a | 12.17 | 11.33 | 11.75 c | 2.07 y | 2.18 y | 2.12 b |
| W × S (n = 13) | 2.04 vx | 1.77 y | 1.90 a | 2.79 x | 2.31 y | 2.55 a | 13.72 | 12.23 | 12.97 b | 2.18 y | 2.27 y | 2.22 b |
| S × W (n = 10) | 1.98 vx | 1.87 v–y | 1.93 a | 2.80 x | 2.57 xy | 2.68 a | 14.30 | 12.07 | 13.19 b | 2.44 x | 2.32 y | 2.38 a |
| Mean (n = 31) | 2.02 A | 1.78 B | 2.71 A | 2.39 B | 14.06 A | 12.34 B | 2.28 | 2.25 | ||||
| F values for: | ||||||||||||
| Cereals (C) | F(3,54) = 69.37 | F(3,54) = 17.00 | F(3,54) = 34.69 | F(3,54) = 4.77 | ||||||||
| Years (Y) | F(1,54) = 14.11 | F(1,54) = 7.53 | F(1,54) = 14.23 | F(1,54) = 0.62 | ||||||||
| Interaction (CxY) | F(3,54) = 5.05 | F(3,54) = 3.61 | F(3,54) = 1.15 | F(3,54) = 3.01 | ||||||||
| K (103) | P (103) | S (103) | Mg (103) | Ca (102) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Year | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean |
| Spelt (n = 5) | 4.96 | 4.56 | 4.76 | 4.20 | 4.99 | 4.47 a | 1.65 | 1.91 | 1.71 a | 1.33 | 1.49 | 1.41 a | 3.49 | 3.44 | 3.47 |
| Wheat (n = 3) | 5.06 | 4.59 | 4.82 | 3.39 | 4.03 | 3.72 c | 1.32 | 1.51 | 1.39 c | 1.12 | 1.31 | 1.24 b | 3.38 | 3.45 | 3.42 |
| W × S (n = 13) | 5.04 | 4.53 | 4.78 | 3.78 | 4.57 | 4.15 b | 1.47 | 1.64 | 1.50 b | 1.28 | 1.40 | 1.34 ab | 3.70 | 3.81 | 3.76 |
| S × W (n = 10) | 4.94 | 4.70 | 4.82 | 3.89 | 4.57 | 4.16 b | 1.45 | 1.67 | 1.49 b | 1.29 | 1.40 | 1.35 ab | 3.69 | 3.24 | 3.47 |
| mean (n = 31) | 5.00 A | 4.60 B | 4.79 | 3.85 B | 4.59 A | 4.16 | 1.48 B | 1.68 A | 1.52 | 1.28 B | 1.41 A | 1.34 | 3.63 | 3.53 | 3.59 |
| F values for: | |||||||||||||||
| Cereals(C) | F(3,54) = 0.16 | F(3,54) = 5.97 | F(3,54) = 10.09 | F(3,54) = 2.95 | F(3,54) = 2.67 | ||||||||||
| Years (Y) | F(1,54) = 25.49 | F(1,54) = 45.63 | F(1,54) = 17.53 | F(1,54) = 15.29 | F(1,54) = 0.47 | ||||||||||
| Interaction (C × Y) | F(3,54) = 0.97 | F(3,54) = 0.72 | F(3,54) = 0.73 | F(3,54) = 0.11 | F(3,54) = 1.92 | ||||||||||
| SEW (n = 23) | 0.233 | 0.087 | 0.441 | 0.337 | 0.395 | ||||||||||
| SEK (n = 23) | 1.447 | 0.444 | 1.459 | 1.738 | 1.730 | ||||||||||
| Fe (101) | Zn (101) | Mn (101) | Cu | Na | ||||||||||||
| Year | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | |
| Spelt (n = 5) | 4.46 | 4.12 | 4.28 a | 3.24 x | 3.24 x | 3.24 a | 3.13 | 2.23 | 2.67 a | 3.47 | 4.93 | 4.20 a | 7.16 | 7.38 | 7.27 | |
| Wheat (n = 3) | 3.71 | 3.88 | 3.79 ab | 3.14 x | 2.28 v | 2.70 b | 2.76 | 2.05 | 2.41 ab | 2.35 | 4.28 | 3.31 bc | 7.53 | 7.51 | 7.52 | |
| W × S (n = 13) | 3.57 | 3.78 | 3.67 b | 3.05 xy | 2.84 v | 2.94 ab | 2.65 | 1.85 | 2.25 b | 2.94 | 4.60 | 3.77 ab | 7.95 | 7.71 | 7.83 | |
| S × W (n = 10) | 4.18 | 3.75 | 3.96 ab | 2.43 yv | 2.71 v | 2.56 b | 2.58 | 2.01 | 2.29 ab | 2.68 | 3.88 | 3.28 c | 7.44 | 8.13 | 7.79 | |
| mean (n = 31) | 3.92 | 3.83 | 2.89 | 2.81 | 2.72 A | 1.98 B | 2.88 A | 4.39 B | 7.62 | 7.77 | ||||||
| F values for: | Cereals (C) | F(3,54) = 3.05 | F(3,54) = 7.47 | F(3,54) = 2.85 | F(3,54 ) = 7.62 | F(3,54) = 1.08 | ||||||||||
| Years (Y) | F(1,54) = 0.36 | F(1,54) = 2.75 | F(1,54) = 38.07 | F(1,54) = 93.11 | F(1,54) = 0.372 | |||||||||||
| Int. (C × Y) | F(3,54) = 1.49 | F(3,54) = 3.58 | F(3,54) = 0.48 | F(3,54) = 1.02 | F(3,54) = 1.00 | |||||||||||
| SEW (n = 23) | 0.166 | 0.209 | −0.391 | −0.614 | 0.306 | |||||||||||
| SEK (n = 23) | 2.310 | 1.450 | −0.820 | 0.345 | 1.542 | |||||||||||
| Al | Sr | Rb | Ba | B (10−1) | ||||||||||||
| Year | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | |
| Spelt (n = 5) | 0.70 | 1.59 | 1.15 | 1.54 | 1.87 | 1.70 | 2.41 | 1.71 | 2.06 | 2.94 | 2.52 | 2.73 | 0.62 | 0.72 | 0.67 a | |
| Wheat (n = 3) | 0.89 | 1.38 | 1.14 | 1.21 | 1.56 | 1.39 | 4.34 | 1.25 | 2.79 | 1.51 | 1.83 | 1.67 | 0.53 | 0.65 | 0.59 ab | |
| W × S (n = 13) | 1.13 | 1.03 | 1.08 | 1.32 | 1.87 | 1.59 | 3.37 | 1.10 | 2.24 | 2.21 | 2.86 | 2.54 | 0.59 | 0.70 | 0.65 a | |
| S × W (n = 10) | 1.21 | 1.24 | 1.23 | 1.04 | 1.35 | 1.19 | 3.03 | 1.27 | 2.15 | 1.52 | 1.85 | 1.68 | 0.49 | 0.65 | 0.57 b | |
| mean (n = 31) | 1.06 | 1.22 | 1.25 B | 1.67 A | 3.20 A | 1.27 B | 2.04 | 2.38 | 0.56 B | 0.68 A | ||||||
| F values for: | Cereals (C) | F(3,54) = 0.52 | F(3,54) = 4.97 | F(3,54) = 0.57 | F(3,54) = 4.55 | F(3,54) = 4.74 | ||||||||||
| Years (Y) | F(1,54) = 3.49 | F(1,54) = 0.94 | F(1,54) = 31.13 | F(1,54) = 0.61 | F(1,54) = 26.44 | |||||||||||
| Int. (CxY) | F(3,54) = 2.53 | F(3,54) = 0.40 | F(3,54) = 1.59 | F(3,54) = 0.75 | F(3,54) = 0.42 | |||||||||||
| SEW (n = 23) | 0.155 | −0.349 | −0.184 | −0.102 | 0.270 | |||||||||||
| SEK (n = 23) | 1.449 | 0.167 | 1.521 | 0.642 | 1.642 | |||||||||||
| Mo (·10−1) | Ni (·10−1) | Cd (·10−2) | Se (·10−2) | Pb (·10−3) | ||||||||||||
| Year | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | 2014 | 2015 | Mean | |
| Spelt (n = 5) | 2.98 x–z | 4.08 x–z | 3.53 b | 2.41 x | 1.19 yz | 1.79 a | 7.99 | 5.46 | 6.73 | 2.70 | 1.70 | 2.20 a | 3.03 | 4.89 | 3.95 | |
| Wheat (n = 3) | 2.03 yz | 6.15 vx | 4.09 ab | 2.51 x | 0.67 z | 1.59 ab | 4.94 | 3.56 | 4.25 | 0.65 | 0.93 | 0.79 c | 1.72 | 3.31 | 2.51 | |
| W × S (n = 13) | 2.85 yz | 7.41 v | 5.13 a | 1.75 xy | 0.66 z | 1.20 bc | 4.29 | 4.53 | 4.41 | 1.27 | 0.97 | 1.12 bc | 2.59 | 2.10 | 2.34 | |
| S × W (n = 10) | 3.57 x–z | 4.81 xy | 4.19 ab | 1.13 yz | 0.73 z | 0.92 c | 3.53 | 4.55 | 4.04 | 1.85 | 0.95 | 1.40 b | 2.64 | 3.95 | 3.30 | |
| mean (n = 31) | 3.02 B | 5.91 A | 1.73 A | 0.81 B | 4.70 | 4.59 | 1.63 | 1.08 | 2.59 | 3.26 | ||||||
| F values for: | Cereals(C) | F(3,54) = 3.59 | F(3,54) = 8.77 | F(3,54) = 1.83 | F(3,54) = 4.09 | F(3,54) = 1.27 | ||||||||||
| Years (Y) | F(1,54) = 40.77 | F(1,54) = 65.68 | F(1,54) = 3.30 | F(1,54) = 3.03 | F(1,54) = 2.09 | |||||||||||
| Int. (C × Y) | F(3,54) = 6.72 | F(3,54) = 4.49 | F(3,54) = 2.43 | F(3,54) = 0.98 | F(3,54) = 0.84 | |||||||||||
| SEW (n = 23) | −0.344 | −0.552 | 0.280 | −0.542 | −2.305 | |||||||||||
| SEK (n = 23) | −0.314 | −1.243 | 1.870 | 0.722 | −6.627 | |||||||||||
| Macro Elements (103) | Micro Elements (101) | ||
|---|---|---|---|
| Desirable | Undesirable | ||
| Spelt (n = 5) | 15.8 a | 7.9 a | 5.8 a |
| Wheat (n = 3) | 14.6 b | 6.8 b | 4.4 b |
| W × S (n = 13) | 15.5 ab | 6.9 b | 5.2 ab |
| S × W (n = 10) | 15.3 ab | 6.9 b | 4.2 b |
| mean (n = 31) | 15.4 | 7.1 | 4.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suchowilska, E.; Wiwart, M.; Krska, R.; Kandler, W. Do Triticum aestivum L. and Triticum spelta L. Hybrids Constitute a Promising Source Material for Quality Breeding ofNew Wheat Varieties? Agronomy 2020, 10, 43. https://doi.org/10.3390/agronomy10010043
Suchowilska E, Wiwart M, Krska R, Kandler W. Do Triticum aestivum L. and Triticum spelta L. Hybrids Constitute a Promising Source Material for Quality Breeding ofNew Wheat Varieties? Agronomy. 2020; 10(1):43. https://doi.org/10.3390/agronomy10010043
Chicago/Turabian StyleSuchowilska, Elżbieta, Marian Wiwart, Rudolf Krska, and Wolfgang Kandler. 2020. "Do Triticum aestivum L. and Triticum spelta L. Hybrids Constitute a Promising Source Material for Quality Breeding ofNew Wheat Varieties?" Agronomy 10, no. 1: 43. https://doi.org/10.3390/agronomy10010043
APA StyleSuchowilska, E., Wiwart, M., Krska, R., & Kandler, W. (2020). Do Triticum aestivum L. and Triticum spelta L. Hybrids Constitute a Promising Source Material for Quality Breeding ofNew Wheat Varieties? Agronomy, 10(1), 43. https://doi.org/10.3390/agronomy10010043





