Soil Type and Zinc Doses in Agronomic Biofortification of Lettuce Genotypes
Abstract
1. Introduction
2. Materials and Methods
2.1. Location and Experimental Design
2.2. Installation and Conduction of the Experiment
2.3. Growth and Biofortification with Zn
2.4. Statistical Analysis
3. Results
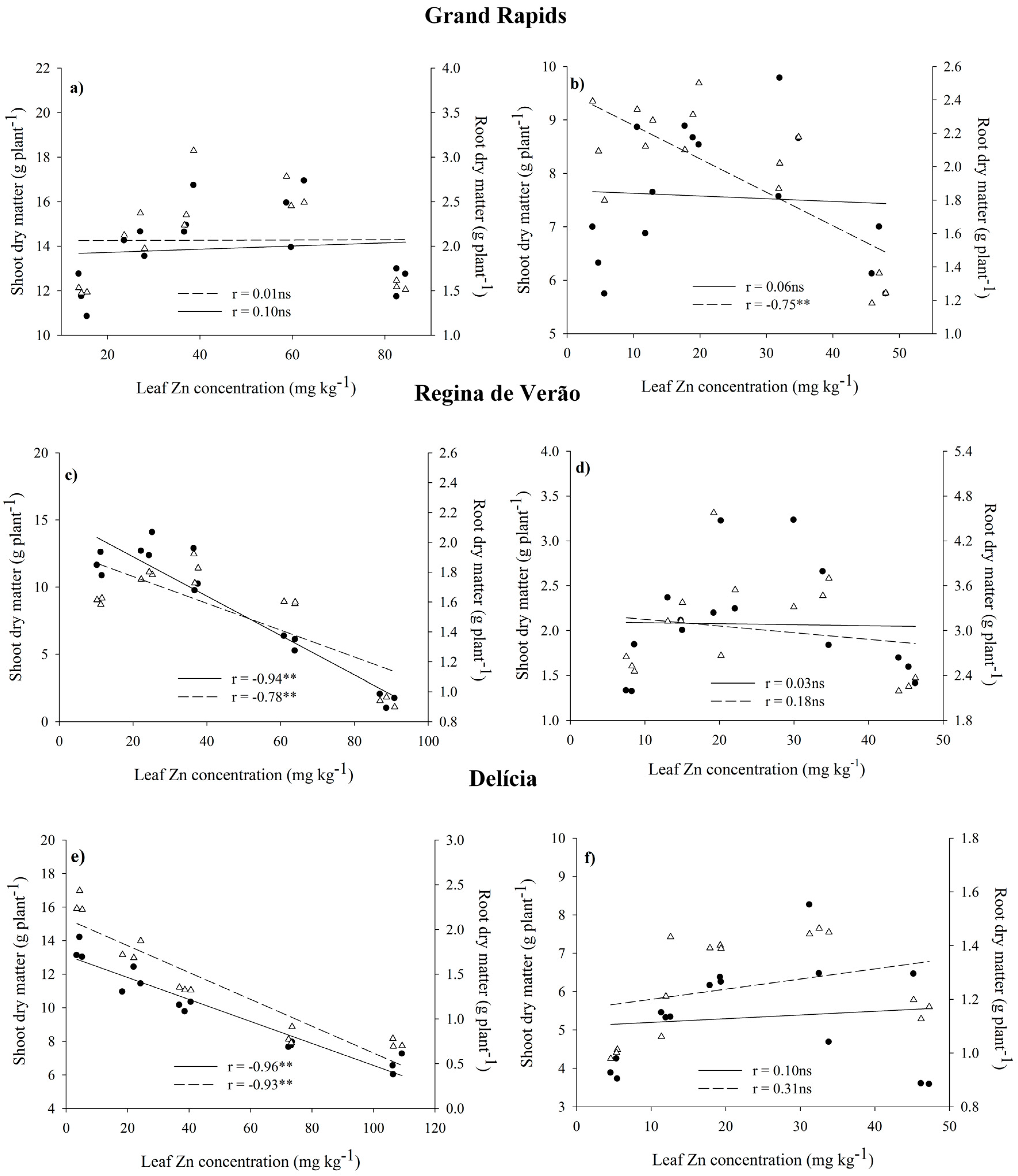
3.1. Growth of Lettuce Genotypes
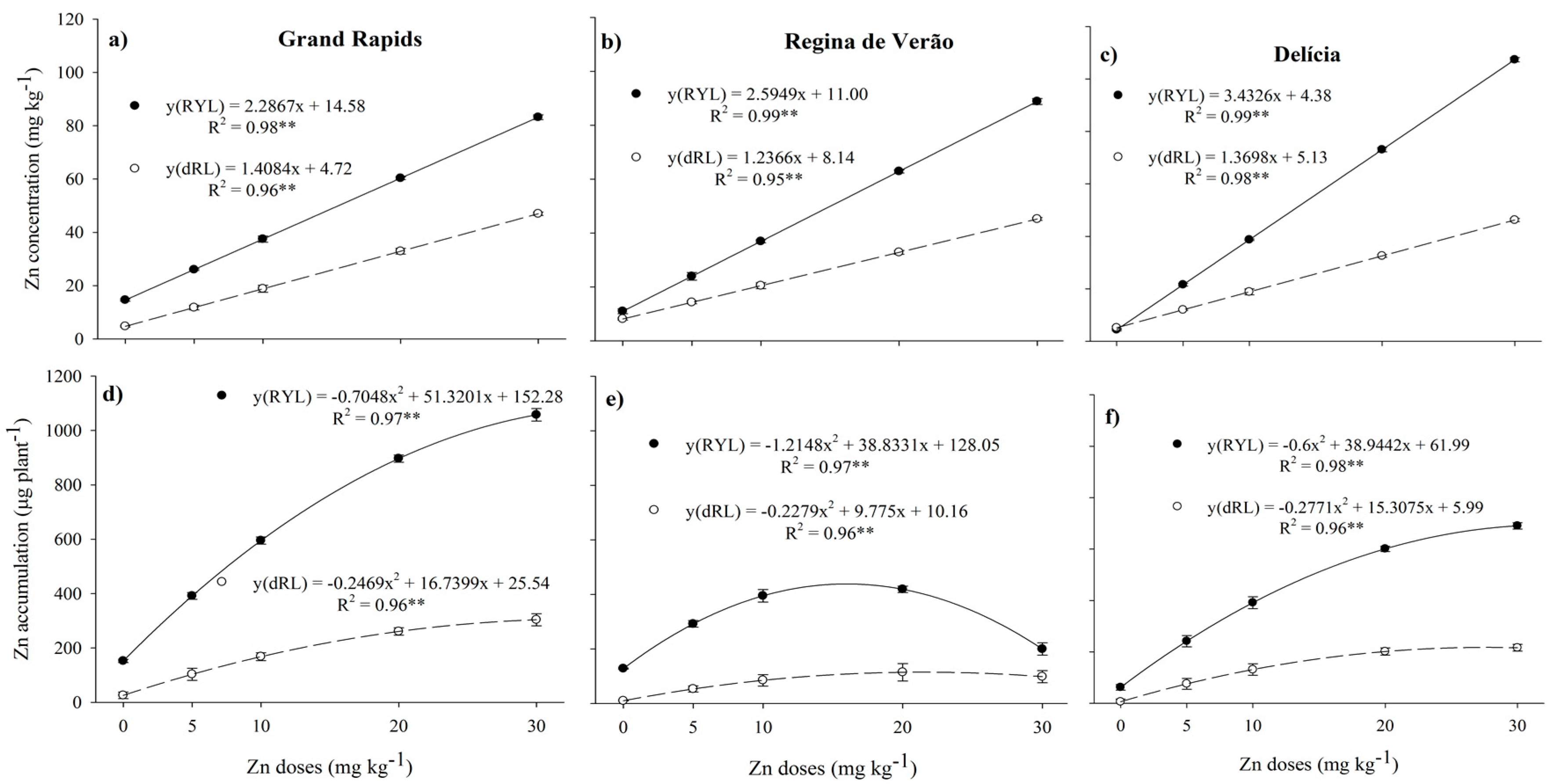
3.2. Leaf Zn Concentration and Accumulation-Biofortification
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Worldmeters. Available online: http://www.worldometers.info/world-population/#growthrate (accessed on 27 September 2019).
- United Nations. The Impact of Population Momentum on Future Population Growth; United Nations: New York, NY, USA, 2017. [Google Scholar]
- Sustainable Development Solutions Network. Solutions for Sustainable Agriculture and Food Systems: Technical Report for the Post-2015 Development Agenda; Sustainable Development Solutions Network: New York, NY, USA, 2015. [Google Scholar]
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food Security: The challenge of feeding 9 billion people. Science 2010, 327, 812–818. [Google Scholar] [CrossRef] [PubMed]
- FAO; WFP; IFAD. Economic growth is necessary but not sufficient to accelerate reduction of hunger and malnutrition. In The State of Food Insecurity in the World 2012; FAO: Rome, Italy, 2012. [Google Scholar]
- Beal, T.; Massiot, E.; Joanne, E.; Arsenault, J.E.; Matthew, R.; Smith, M.R.; Hijmans, R.J. Global trends in dietary micronutrient supplies and estimated prevalence of inadequate intakes. PLoS ONE 2017, 12, e0175554. [Google Scholar] [CrossRef] [PubMed]
- Wessells, K.R.; Brown, K.H. Estimating the global prevalence of zinc deficiency: Results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 2012, 7, e50568. [Google Scholar] [CrossRef] [PubMed]
- Hefferon, K. Biotechnological approaches for generating zinc-enriched crops to combat malnutrition. Nutrients 2019, 11, 253. [Google Scholar] [CrossRef] [PubMed]
- Lopes, A.S.; Guilherme, L.R.G. A career perspective on soil management in the Cerrado region of Brazil. Adv. Agron. 2016, 137, 1–72. [Google Scholar]
- Corguinha, A.P.B.; Carvalho, C.A.; de Souza, G.A.; de Carvalho, T.S.; Vieira, E.A.; Fialho, J.F.; Guilherme, L.R.G. Potential of cassava clones enriched with β-carotene and lycopene for zinc biofortification under different soil Zn conditions. J. Sci. Food Agric. 2019, 99, 666–674. [Google Scholar] [CrossRef] [PubMed]
- Tittonell, P.; Giller, K.E. When yield gaps are poverty traps: The paradigm of ecological intensification in african smallholder agriculture. Field Crop. Res. 2013, 143, 76–90. [Google Scholar] [CrossRef]
- Manzeke, M.G.; Mtambanengwe, F.; Nezomba, H.; Watts, M.J.; Broadley, M.R.; Mapfumo, P. Zinc fertilization increases productivity and grain nutritional quality of cowpea (Vigna unguiculata [L.] Walp.) under integrated soil fertility management. Field Crop. Res. 2017, 213, 231–244. [Google Scholar] [CrossRef]
- Barrameda-Medina, Y.; Blasco, B.; Lentini, M.; Esposito, S.; Baenas, N.; Moreno, D.A.; Ruiz, J.M. Zinc biofortification improves phytochemicals and amino-acidic profile in Brassica oleracea cv. Bronco. Plant Sci. 2017, 258, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Bouis, H.E.; Saltzman, A. Improving nutrition through biofortification: A review of evidence from HarvestPlus, 2003 through 2016. Glob. Food Secur. 2017, 12, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Poblaciones, M.J.; Rengel, Z. Soil and foliar zinc biofortification in field pea (Pisum sativum L.): Grain accumulation and bioavailability in raw and cooked grains. Food Chem. 2016, 212, 427–433. [Google Scholar] [CrossRef] [PubMed]
- White, P.J.; Pongrac, P.; Sneddon, C.C.; Thompson, J.A.; Wright, G. Limits to the biofortification of leafy brassicas with zinc. Agriculture 2018, 8, 32. [Google Scholar] [CrossRef]
- White, P.J.; Broadley, M.R. Physiological limits to zinc biofortification of edible crops. Front. Plant Sci. 2011, 2, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Maynard, D.N.; Hochmuth, G.J. Knott’s Handbook for Vegetable Growers, 5th ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2007; pp. 179–181. [Google Scholar]
- Padash, A.; Shahabivand, S.; Behtash, F.; Aghaee, A. A practicable method for zinc enrichment in lettuce leaves by the endophyte fungus Piriformospora indica under increasing zinc supply. Sci. Hortic. 2016, 213, 367–372. [Google Scholar] [CrossRef]
- Faostat. Available online: http://www.fao.org/faostat/es/#data/QC (accessed on 29 December 2019).
- Souza, R.F.; Faquin, V.; Fernandes, L.A.; Avila, F.W. Nutrição fosfatada e rendimento do feijoeiro sob influência da calagem e adubação orgânica. Ciênc. Agrotec. 2006, 30, 656–664. [Google Scholar] [CrossRef]
- Barbosa, J.C.; Maldonado Júnior, W. Experimentação Agronômica e Agroestat—Sistema Para Análises Estatísticas de Ensaios Agronômicos, 1st ed.; Gráfica Multipress Ltda: Jaboticabal, Brazil, 2015. [Google Scholar]
- Pongrac, P.; McNicol, J.W.; Lilly, A.; Thompson, J.A.; Wright, G.; Hillier, S.; White, P.J. Mineral element composition of cabbage as affected by soil type and phosphorus and zinc fertilization. Plant Soil 2019, 434, 151–165. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E.; Moller, I.M.; Murphy, A. Fisiologia e Desenvolvimento Vegetal, 6th ed.; Artmed: Porto Alegre, Brazil, 2017; pp. 119–167. [Google Scholar]
- Rafaey, Y.; Jansen, B.; Parsons, J.R.; Voogt, P.; Bagnis, S.; Markus, A.; El-Shater, A.-H.; El-Haddad, A.-A.; Kalbitz, K. Effects of clay minerals, hydroxides, and timing of dissolved organic matter addition on the competitive sorption of copper, nickel, and zinc: A column experiment. J. Environ. Manag. 2017, 187, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Broadley, M.; Brown, P.; Cakmak, I.; Rengel, Z.; Zhao, F. Function of nutrients: Micronutrients. In Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Marschner, P., Ed.; Academic Press: London, UK, 2012; pp. 191–248. [Google Scholar]

| Soil Attribute | Soil | Soil Attribute | Soil | ||
|---|---|---|---|---|---|
| RYL | dRL | RYL | dRL | ||
| pH (H2O) | 5.0 | 4.9 | CEC-T (cmolc kg−1) | 4.3 | 9.1 |
| Al3+ (cmolc kg−1) | 0.7 | 1.3 | V (%) | 16.3 | 3.3 |
| Ca2+ (cmolc kg−1) | 0.4 | 0.1 | Sand (g kg−1) | 730.0 | 240.0 |
| Mg2+ (cmolc kg−1) | 0.2 | 0.1 | Silt (g kg−1) | 50.0 | 110.0 |
| K+ (mg kg−1) | 41.0 | 37.0 | Clay (g kg−1) | 220.0 | 650.0 |
| H++Al3+ (cmolc kg−1) | 3.6 | 8.8 | Ds (kg dm−3) | 1.2 | 1.0 |
| P (mg kg−1) | 0.8 | 0.8 | SiO2 (g kg−1) | 95.1 | 130 |
| P rem (mg kg−1) | 26.5 | 8.5 | Al2O3 (g kg−1) | 97.4 | 319 |
| S (mg kg−1) | 6.2 | 7.1 | Fe2O3 (g kg−1) | 36.2 | 172 |
| B (mg kg−1) | 0.7 | 0.4 | TiO2 (g kg−1) | 6.2 | 22.0 |
| Cu2+ (mg kg−1) | 0.9 | 1.7 | P2O5 (g kg−1) | 0.0 | 0.9 |
| Fe2+ (mg kg−1) | 64.7 | 93.4 | Fed (g kg−1) | 10.8 | 102 |
| Mn2+ (mg kg−1) | 5.9 | 3.3 | Feo (g kg−1) | 0.1 | 9.2 |
| Zn2+ (mg kg−1) | 0.6 | 0.8 | Ct (g kg−1) | 752 | 293 |
| OM (g kg−1) | 24.0 | 48.0 | Gibbsite (g kg−1) | 63.0 | 359 |
| BS (cmolc kg−1) | 0.7 | 0.3 | Ki | 1.0 | 0.4 |
| CEC-t (cmolc kg−1) | 1.4 | 1.6 | Kr | 0.7 | 0.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Almeida, H.J.; Vergara Carmona, V.M.; Ferreira Inocêncio, M.; Furtini Neto, A.E.; Cecílio Filho, A.B.; Mauad, M. Soil Type and Zinc Doses in Agronomic Biofortification of Lettuce Genotypes. Agronomy 2020, 10, 124. https://doi.org/10.3390/agronomy10010124
de Almeida HJ, Vergara Carmona VM, Ferreira Inocêncio M, Furtini Neto AE, Cecílio Filho AB, Mauad M. Soil Type and Zinc Doses in Agronomic Biofortification of Lettuce Genotypes. Agronomy. 2020; 10(1):124. https://doi.org/10.3390/agronomy10010124
Chicago/Turabian Stylede Almeida, Hilário Júnior, Victor Manuel Vergara Carmona, Maykom Ferreira Inocêncio, Antônio Eduardo Furtini Neto, Arthur Bernardes Cecílio Filho, and Munir Mauad. 2020. "Soil Type and Zinc Doses in Agronomic Biofortification of Lettuce Genotypes" Agronomy 10, no. 1: 124. https://doi.org/10.3390/agronomy10010124
APA Stylede Almeida, H. J., Vergara Carmona, V. M., Ferreira Inocêncio, M., Furtini Neto, A. E., Cecílio Filho, A. B., & Mauad, M. (2020). Soil Type and Zinc Doses in Agronomic Biofortification of Lettuce Genotypes. Agronomy, 10(1), 124. https://doi.org/10.3390/agronomy10010124





