Formose Reaction Controlled by a Copolymer of N,N-Dimethylacrylamide and 4-Vinylphenylboronic Acid
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Measurements
2.3. Preparation of Water Soluble Polymers
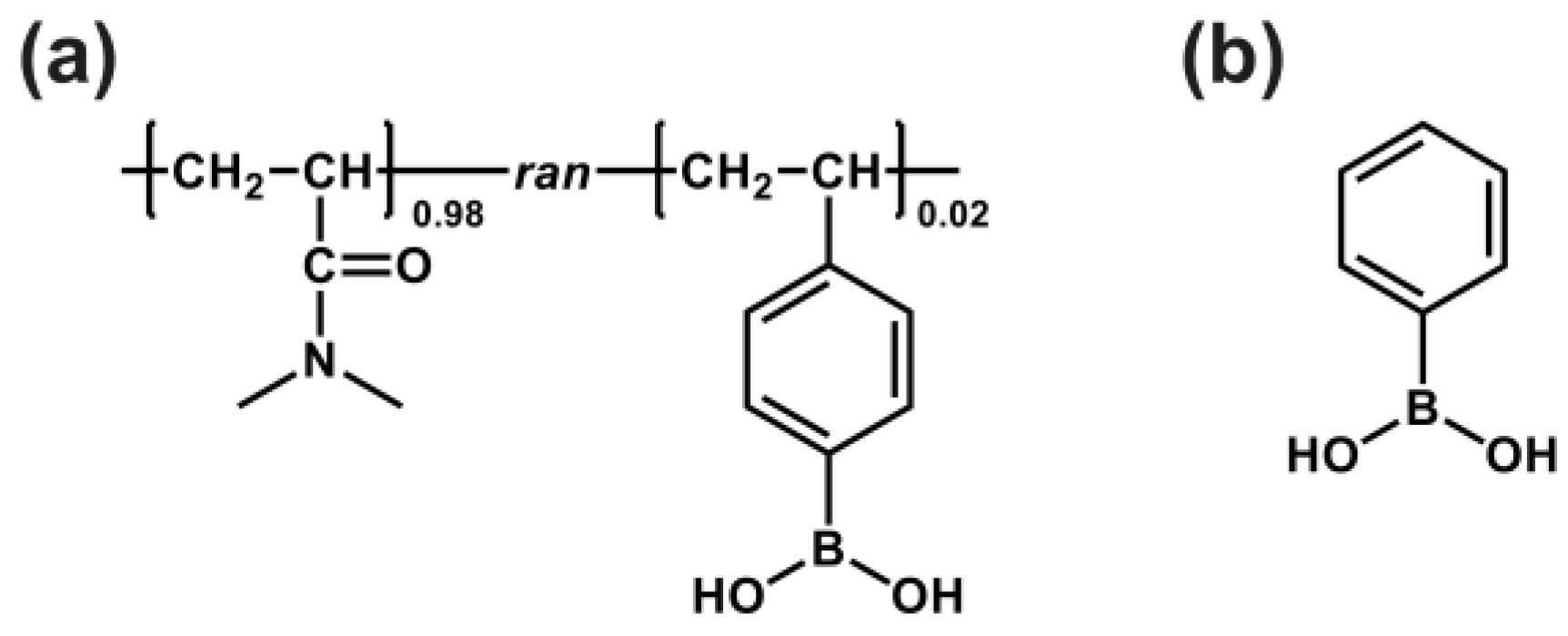
2.4. Preparation of N,N-Dimethylacrylamide/4-Vinylphenylboronic Acid Copolymer
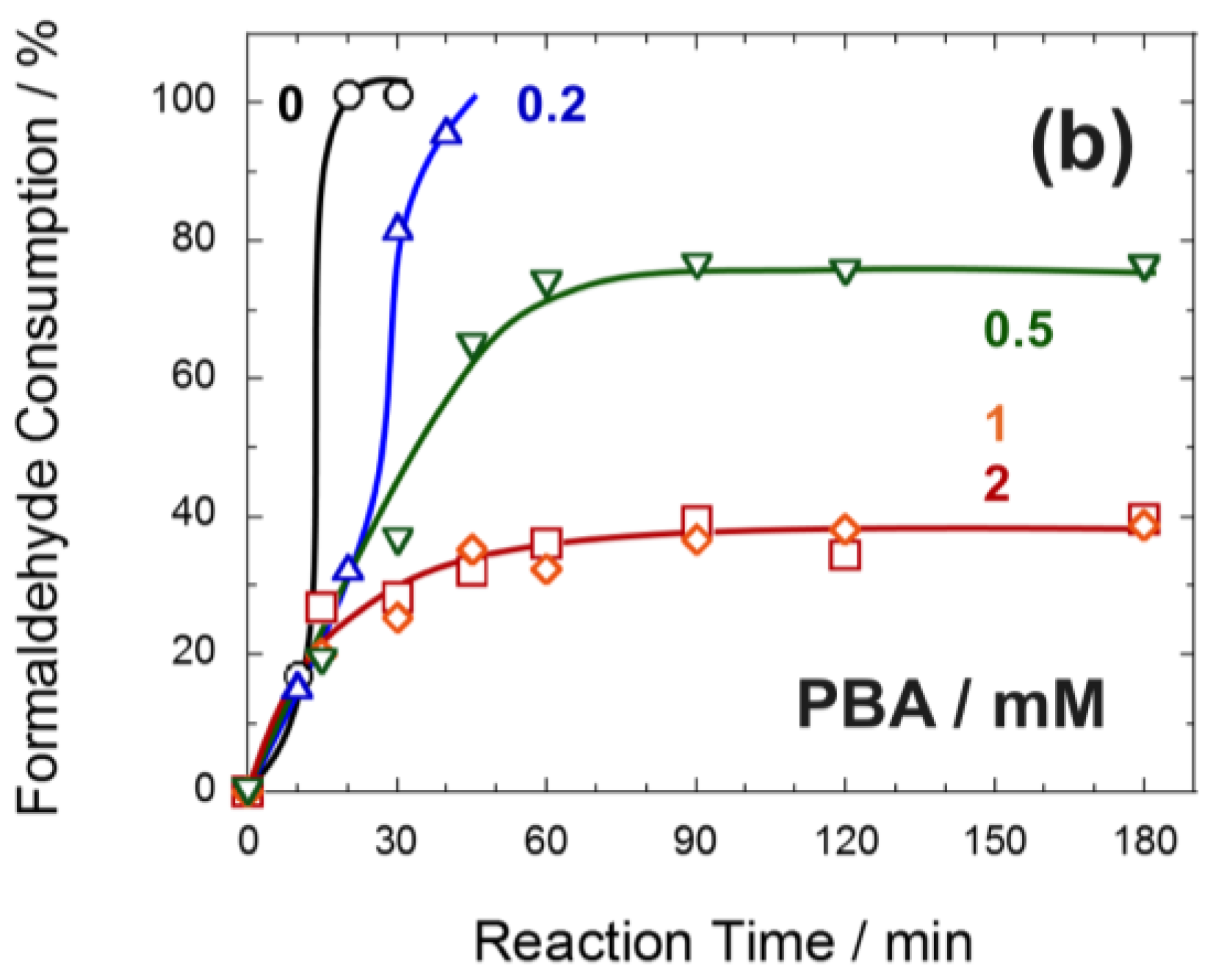
2.5. Formose Reaction in the Presence of Boronic Acid Compounds
3. Results
3.1. Effect of Water Soluble Polymers on the Formose Reaction
3.2. Preparation of N,N-Dimethylacrylamide/4-Vinylphenylboronic Acid Copolymer
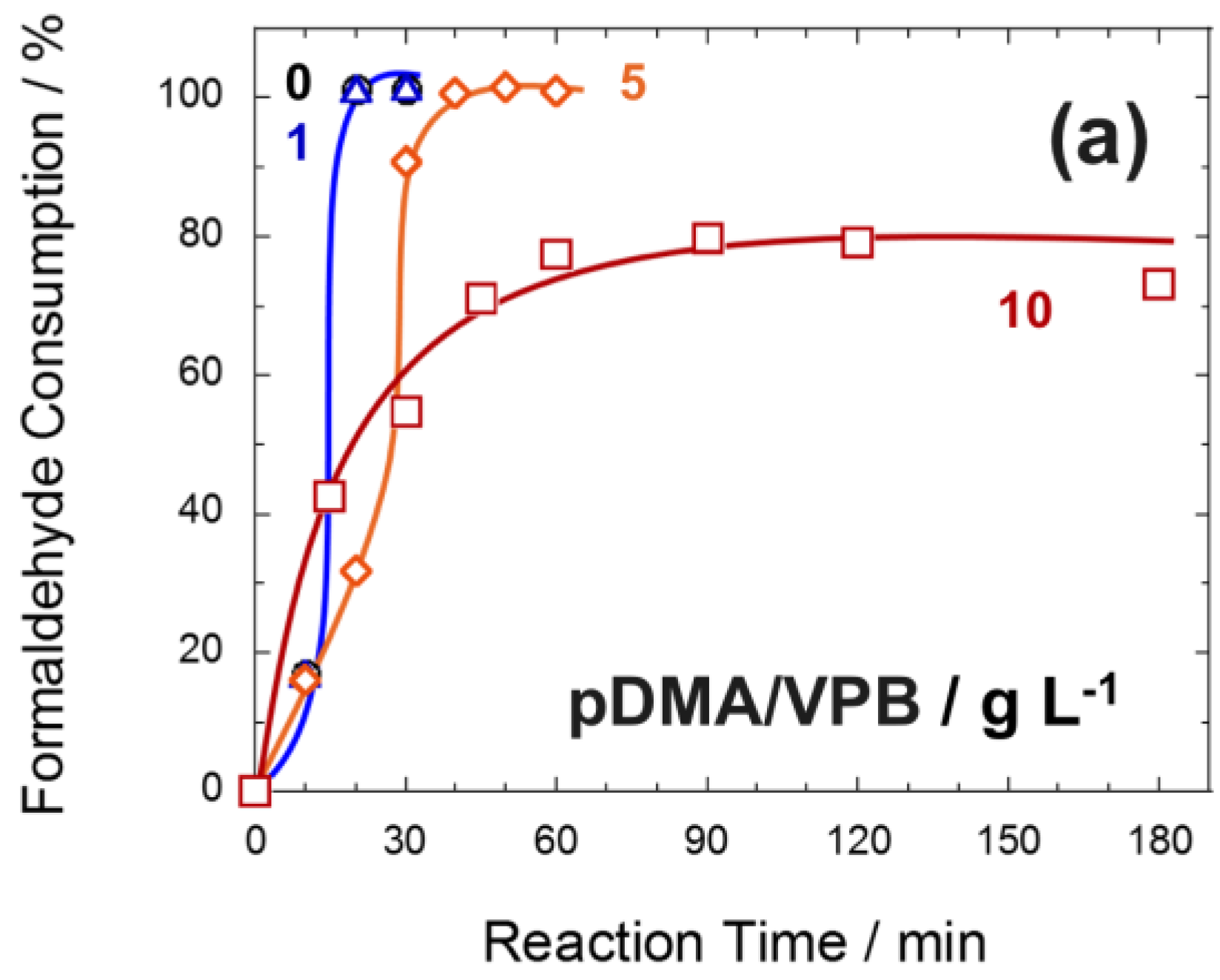
3.3. Effect of the N,N-Dimethylacrylamide/4-Vinylphenylboronic Acid Copolymer
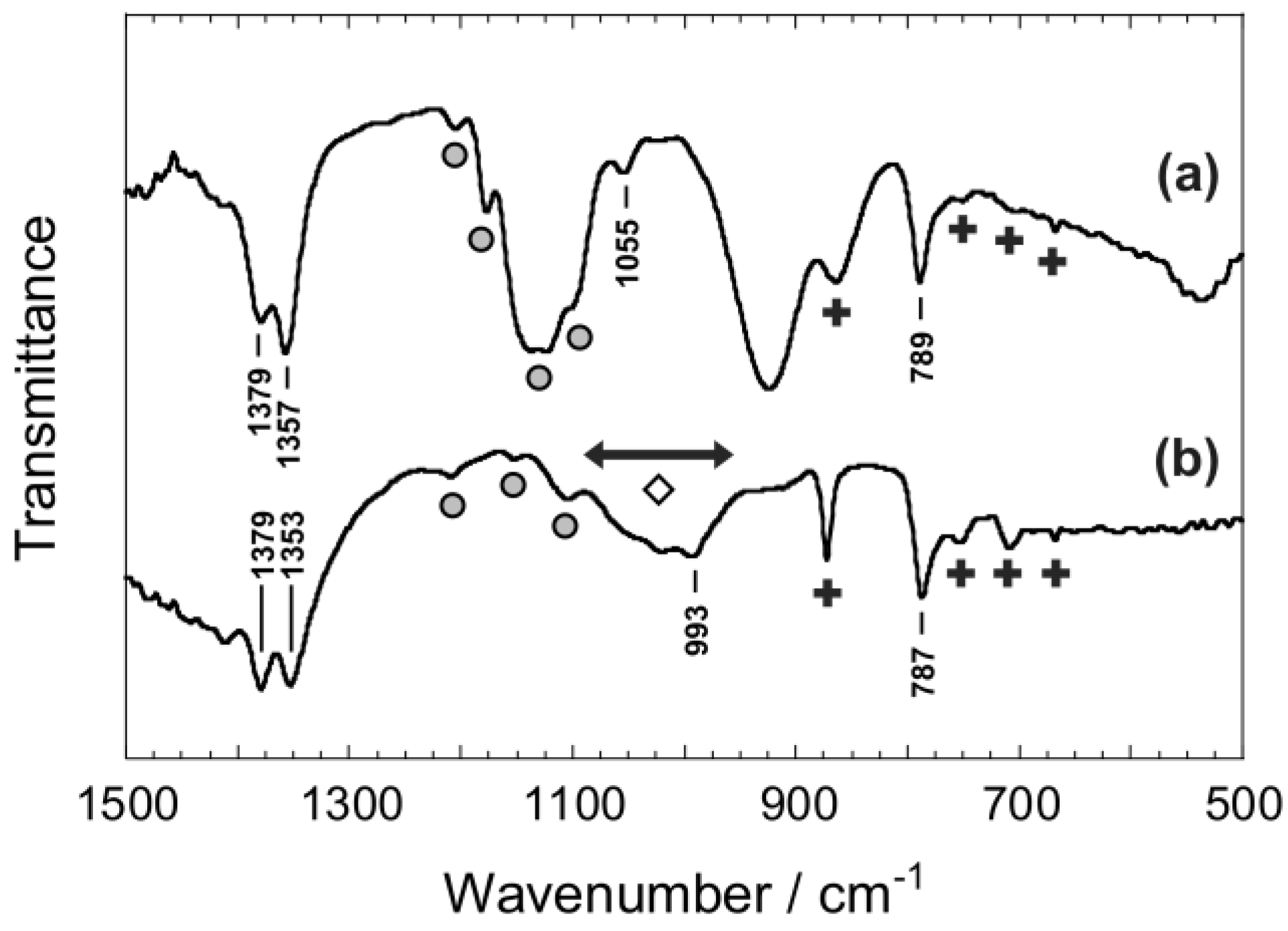
3.4. Confirmation of the Formation of Boronic Acid Esters in Formose Reaction Mixtures
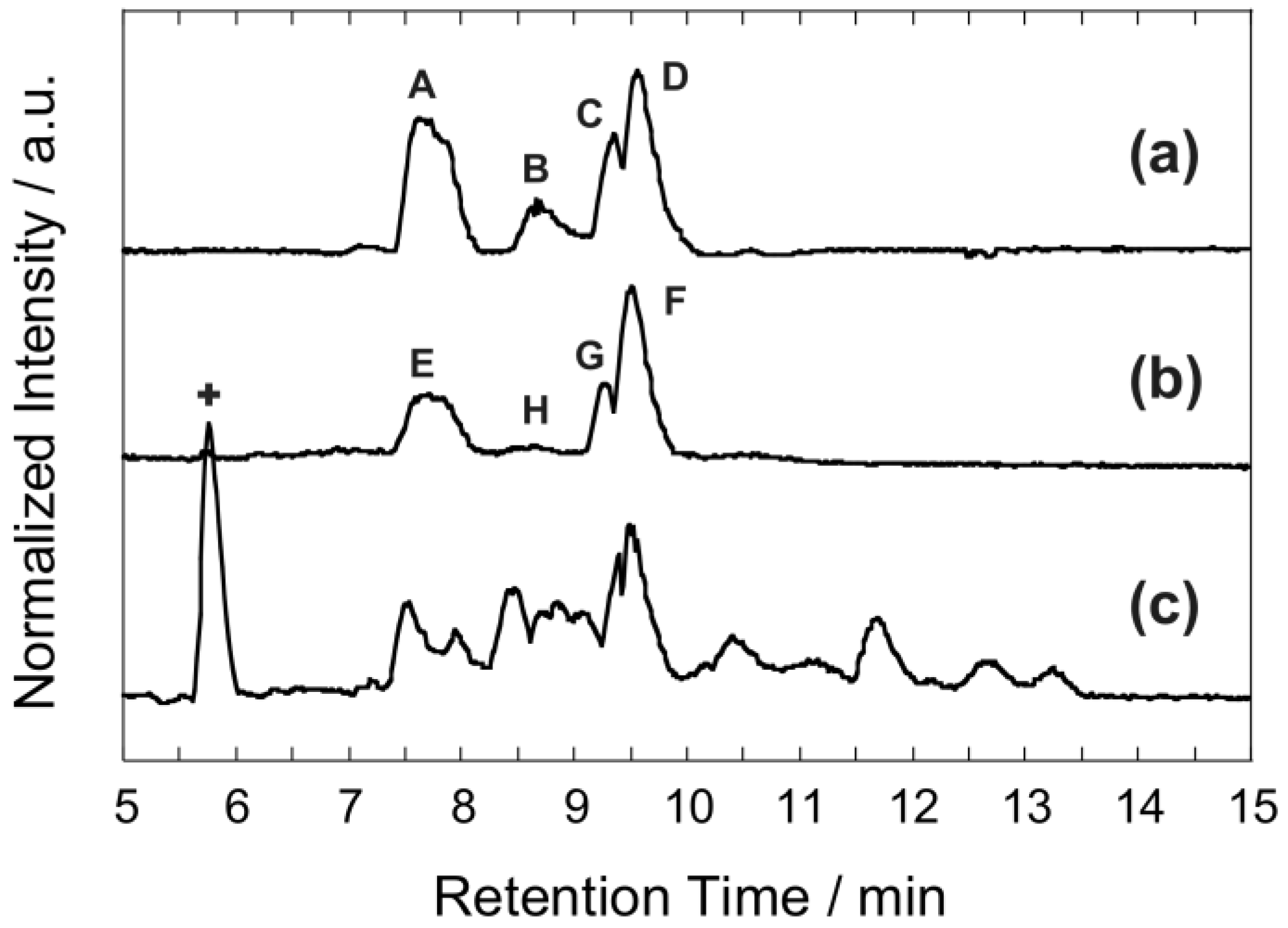
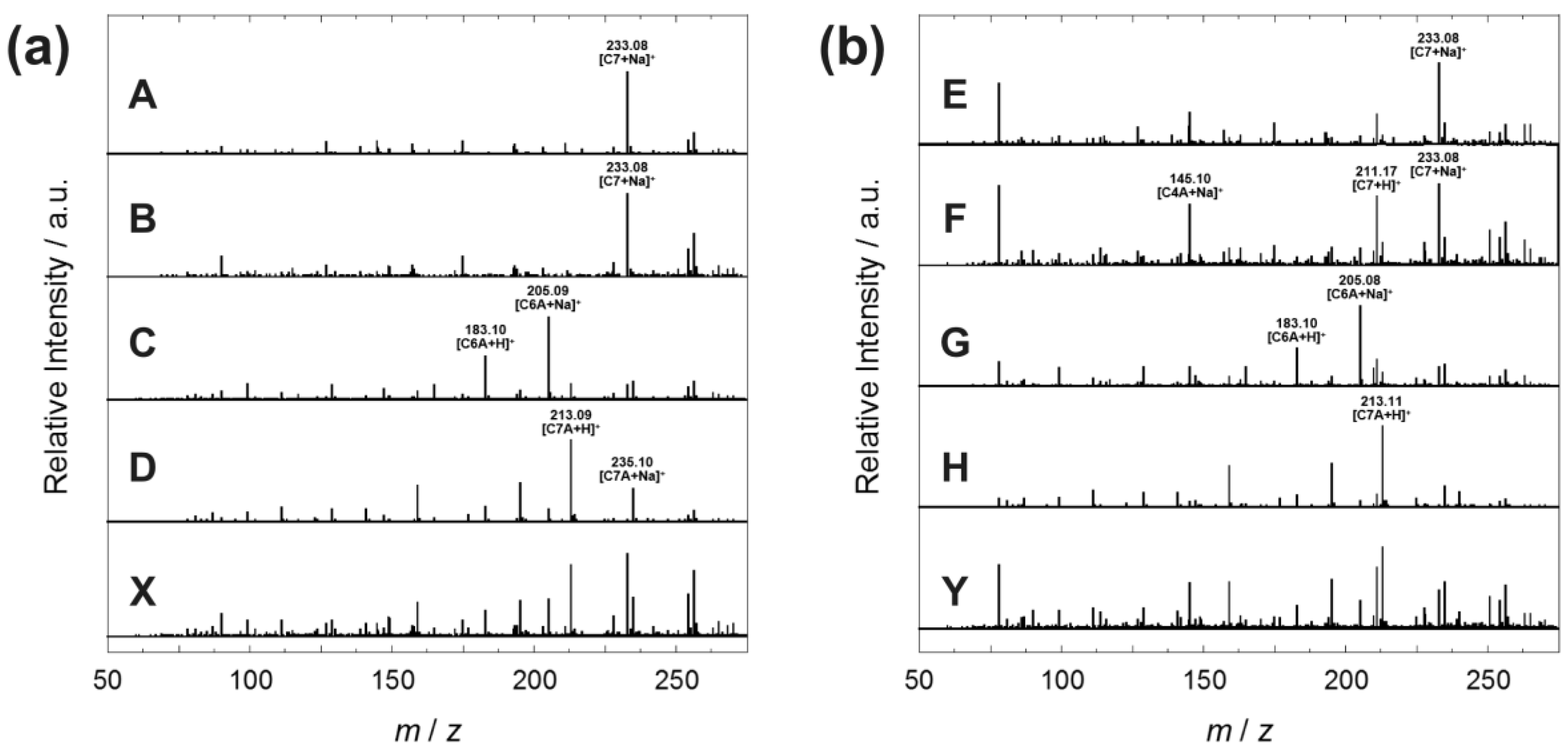
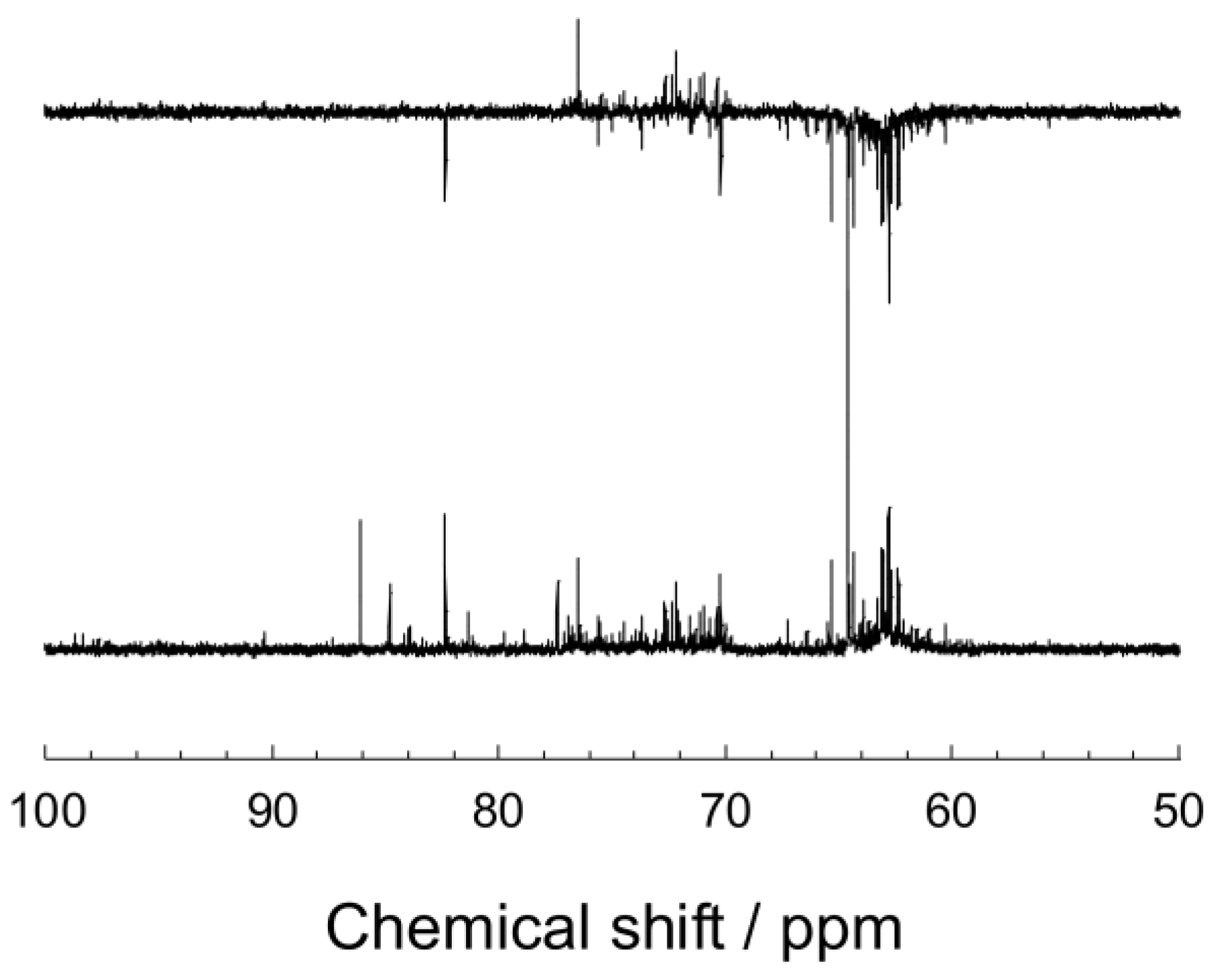
3.5. Characterization of Formose Reaction Products
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Voet, D.; Voet, J.G. Biochemistry, 4th ed.; Wiley & Sons: New York, NY, USA, 2010. [Google Scholar]
- Alberts, B.; Johnson, A.; Lewis, J.; Morgan, D.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell, 6th ed.; Garland Publishing Inc.: New York, NY, USA, 2014. [Google Scholar]
- Ager, D.J.; East, M.B. The synthesis of carbohydrate derivatives from acyclic precursors. Tetrahedron 1993, 49, 5683–5765. [Google Scholar] [CrossRef]
- Bertozzi, C.R.; Kiessling, L.L. Chemical glycobiology. Science 2001, 291, 2357. [Google Scholar] [CrossRef] [PubMed]
- Kinnaert, C.; Daugaard, M.; Nami, F.; Clausen, M.H. Chemical synthesis of oligosaccharides related to the cell walls of plants and algae. Chem. Rev. 2017, 117, 11337–11405. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Wang, S.; Tang, W. De novo synthesis of mono- and oligosaccharides via dihydropyran intermediates. Chem. Asian J. 2017, 12, 1027–1042. [Google Scholar] [CrossRef] [PubMed]
- Northrup, A.B.; MacMillan, D.W.C. Two-step synthesis of carbohydrates by selective aldol reactions. Science 2004, 305, 1752–1755. [Google Scholar] [CrossRef] [PubMed]
- Northrup, A.B.; Mangion, I.K.; Hettche, F.; MacMillan, D.W.C. Enantioselective organo-catalytic direct aldol reactions of α-oxy-aldehydes: Step one in a two-step synthesis of carbohydrates. Angew. Chem. Int. Ed. 2004, 43, 2152–2154. [Google Scholar] [CrossRef] [PubMed]
- Butlerow, A. Bildung einer zuckerartigen substanz durch synthese. Ann. Chem. 1861, 120, 295–298. [Google Scholar] [CrossRef]
- Breslow, R. Mechanism of the formose reaction. Tetrahedron Lett. 1959, 1, 22–26. [Google Scholar] [CrossRef]
- Mizuno, T.; Weiss, A.H. Synthesis and utilization of formose sugars. Adv. Carbohydr. Chem. Biochem. 1974, 29, 173–227. [Google Scholar] [CrossRef]
- Weiss, A.H.; Socha, R.F.; Likholobov, V.A.; Sakharov, M.M. Formose sugars from formaldehyde. Appl. Cat. 1981, 1, 237–246. [Google Scholar] [CrossRef]
- Delidovich, I.V.; Simonov, A.N.; Taran, O.P.; Parmon, V.N. Catalytic formation of monosaccharides: From the formose reaction towards selective synthesis. ChemSusChem 2014, 7, 1833–1846. [Google Scholar] [CrossRef] [PubMed]
- Kofoed, J.; Reymond, J.-L.; Darbre, T. Prebiotic carbohydrate synthesis: Zinc-proline catalyzes direct aqueous aldol reactions of α-hydroxy aldehydes and ketones. Org. Biomol. Chem. 2005, 3, 1850–1855. [Google Scholar] [CrossRef] [PubMed]
- Jalbout, A.F.; Abrell, L.; Adamowicz, L.; Polt, R.; Apponi, A.J.; Ziurys, L.M. Sugar synthesis from a gas-phase formose reaction. Astrobiology 2007, 7, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Jalbout, A.F. Prebiotic synthesis of simple sugars by an interstellar formose reaction. Orig. Life Evol. Biosph. 2008, 38, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Breslow, R.; Cheng, Z.-L. L-Amino acids catalyze the formation of an excess of D-glyceraldehyde, and thus of other D sugars, under credible prebiotic conditions. Proc. Natl. Acad. Sci. USA 2010, 107, 5723–5725. [Google Scholar] [CrossRef] [PubMed]
- Mauksch, M.; Wei, S.; Freund, M.; Zamfir, A.; Tsogoeva, S.B. Spontaneous mirror symmetry breaking in the aldol reaction and its potential relevance in prebiotic chemistry. Orig. Life Evol. Biosph. 2010, 40, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Ricardo, A.; Illangkoon, H.I.; Kim, M.J.; Carrigan, M.A.; Frye, F.; Benner, S.A. Synthesis of carbohydrates in mineral-guided prebiotic cycles. J. Am. Chem. Soc. 2011, 133, 9457–9468. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, T.; Shigemasa, Y.; Sakazawa, C. Potentiometric analysis of the formose reaction. Chem. Lett. 1974, 713–714. [Google Scholar] [CrossRef]
- Shigemasa, Y.; Shimao, M.; Sakazawa, C.; Matsuura, T. Potentiometric analysis and fundamental technique. Bull. Chem. Soc. Jpn. 1975, 48, 2099–2102. [Google Scholar] [CrossRef]
- Shigemasa, Y.; Saimoto, H. Formose. Synthesis of saccharides from formaldehyde. Trends Glycosci. Glycotechnol. 1990, 2, 119–123. [Google Scholar] [CrossRef]
- Saimoto, H.; Sugiyama, N.; Sashiwa, H.; Shigemasa, Y. Formation of C2 and C3 sugars in the formose reaction. Chem. Express 1993, 8, 737–740. [Google Scholar]
- Shigemasa, Y.; Nagae, O.; Sakazawa, C.; Nakashima, R.; Matsuura, T. Formose reactions. 5. A selective formose reaction. J. Am. Chem. Soc. 1978, 100, 1309–1310. [Google Scholar] [CrossRef]
- Shigemasa, Y.; Kawahara, M.; Sakazawa, C.; Nakashima, R.; Matsuura, T. Formose reactions. IX. Selective formation of branched sugar alcohols in a modified formose reaction and factors affecting the selectivity. J. Cat. 1980, 62, 107–116. [Google Scholar] [CrossRef]
- Matsumoto, T.; Inoue, S. Selective formation of triose from formaldehyde catalyzed by ethylbenzothiazolium bromide. J. Chem. Soc. Chem. Commun. 1983, 171–172. [Google Scholar] [CrossRef]
- Matsumoto, T.; Yamamoto, H.; Inoue, S. Selective formation of triose from formaldehyde catalyzed by thiazolium salt. J. Am. Chem. Soc. 1984, 106, 4829–4832. [Google Scholar] [CrossRef]
- Ricardo, A.; Carrigan, M.A.; Olcott, A.N.; Benner, S.A. Borate minerals stabilize ribose. Science 2004, 303, 196. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.B.; Gurusamy-Thangavelu, S.A.; Ma, K. The silicate-mediated formose reaction: Bottom-up synthesis of sugar silicates. Science 2010, 327, 984–986. [Google Scholar] [CrossRef] [PubMed]
- Hashidzume, A.; Fujimoto, T.; Masaoka, M.; Sanada, Y.; Sato, T. Preparation and catalytic activity of porous alumina by the sol-gel process in the presence of saccharides. Kobunshi Ronbunshu 2010, 67, 312–317. [Google Scholar] [CrossRef]
- Masaoka, M.; Michitaka, T.; Hashidzume, A. Formose reaction accelerated in aerosol-OT reverse micelles. Beilstein J. Org. Chem. 2016, 12, 2663–2667. [Google Scholar] [CrossRef] [PubMed]
- Imai, T.; Michitaka, T.; Hashidzume, A. Formose reaction controlled by boronic acid compounds. Beilstein J. Org. Chem. 2016, 12, 2668–2672. [Google Scholar] [CrossRef] [PubMed]
- James, T.D.; Sandanayake, K.R.A.S.; Shinkai, S. Saccharide sensing with molecular receptors based on boronic acid. Angew. Chem. Int. Ed. Engl. 1996, 35, 1910–1922. [Google Scholar] [CrossRef]
- James, T.D.; Phillips, M.D.; Shinkai, S. Boronic Acids in Saccharide Recognition; The Royal Society of Chemistry: Cambridge, UK, 2006. [Google Scholar]
- Kubo, Y.; Nishiyabu, R.; James, T.D. Hierarchical supramolecules and organization using boronic acid building blocks. Chem. Commun. 2015, 51, 2005–2020. [Google Scholar] [CrossRef] [PubMed]
- Zhai, W.; Sun, X.; James, T.D.; Fossey, J.S. Boronic acid-based carbohydrate sensing. Chem. Asian J. 2015, 10, 1836–1848. [Google Scholar] [CrossRef] [PubMed]
- Brooks, W.L.A.; Sumerlin, B.S. Synthesis and applications of boronic acid-containing polymers: From materials to medicine. Chem. Rev. 2016, 116, 1375–1397. [Google Scholar] [CrossRef] [PubMed]
- Maruo, Y.Y.; Nakamura, J.; Uchiyama, M. Development of formaldehyde sensing element using porous glass impregnated with β-diketone. Talanta 2008, 74, 1141–1147. [Google Scholar] [CrossRef] [PubMed]
- Faniran, J.A.; Shurvell, H.F. Infrared spectra of phenylboronic acid (normal and deuterated) and diphenyl phenylboronate. Can. J. Chem. 1968, 46, 2089–2095. [Google Scholar] [CrossRef]
- Smith, M.K.; Northrop, B.H. Vibrational properties of boroxine anhydride and boronate ester materials: Model systems for the diagnostic characterization of covalent organic frameworks. Chem. Mater. 2014, 26, 3781–3795. [Google Scholar] [CrossRef]
- As can be seen in Figure S2 in Supplementary Materials, the theoretical calculation indicates the absorption band due to the B–O–H in the quaternized boronic acid ester is observed at ca. 974 cm−1.
- Shigemasa, Y.; Akagi, S.; Nakashima, R.; Saito, S. Formose reactions. Part 10. A selective synthesis of 2,4-di-C-(hydroxymethyl)-3-pentulose in the formose reaction. Carbohydr. Res. 1980, 80, C1–C3. [Google Scholar] [CrossRef]
- Hall, D.G. Boronic Acids: Preparation, Application in Organic Synthesis and Medicine; Wiley-VCH: Weinheim, Germany, 2005. [Google Scholar]
- Wulff, G.; Schauhoff, S. Enzyme-analog-built polymers. 27. Racemic resolution of free sugars with macroporous polymers prepared by molecular imprinting. Selectivity dependence on the arrangement of functional groups versus spatial requirements. J. Org. Chem. 1991, 56, 395–400. [Google Scholar] [CrossRef]
- Wulff, G. Enzyme-like catalysis by molecularly imprinted polymers. Chem. Rev. 2002, 102, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Hashidzume, A.; Zimmerman, S.C. Switching the selectivity of a polyglycerol dendrimer monomolecularly imprinted with D-(–)-fructose. Tetrahedron Lett. 2009, 50, 2204–2207. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michitaka, T.; Imai, T.; Hashidzume, A. Formose Reaction Controlled by a Copolymer of N,N-Dimethylacrylamide and 4-Vinylphenylboronic Acid. Polymers 2017, 9, 549. https://doi.org/10.3390/polym9110549
Michitaka T, Imai T, Hashidzume A. Formose Reaction Controlled by a Copolymer of N,N-Dimethylacrylamide and 4-Vinylphenylboronic Acid. Polymers. 2017; 9(11):549. https://doi.org/10.3390/polym9110549
Chicago/Turabian StyleMichitaka, Tomohiro, Toru Imai, and Akihito Hashidzume. 2017. "Formose Reaction Controlled by a Copolymer of N,N-Dimethylacrylamide and 4-Vinylphenylboronic Acid" Polymers 9, no. 11: 549. https://doi.org/10.3390/polym9110549
APA StyleMichitaka, T., Imai, T., & Hashidzume, A. (2017). Formose Reaction Controlled by a Copolymer of N,N-Dimethylacrylamide and 4-Vinylphenylboronic Acid. Polymers, 9(11), 549. https://doi.org/10.3390/polym9110549




