Bactericidal Effect of Lauric Acid-Loaded PCL-PEG-PCL Nano-Sized Micelles on Skin Commensal Propionibacterium acnes
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Synthesis of PCL-PEG-PCL Copolymers
2.3. Characterization of the PCL-PEG-PCL Copolymers
2.4. Preparation of Blank or LA-Loaded PCL-PEG-PCL Nano-Sized Micelles
2.5. Critical Micelle Concentration (CMC)
2.6. Loading Content of LA in the Micelles
2.7. Preparation of Bacteria for Antibacterial Testing
2.8. Minimum Inhibitory Concentration of the Micelles
2.9. The Minimum Bactericidal Concentration of Micelles
3. Results and Discussion
3.1. Characterization of PCL-PEG-PCL Copolymers (PCECs)
3.2. Characterization of PCL-PEG-PCL Micelles
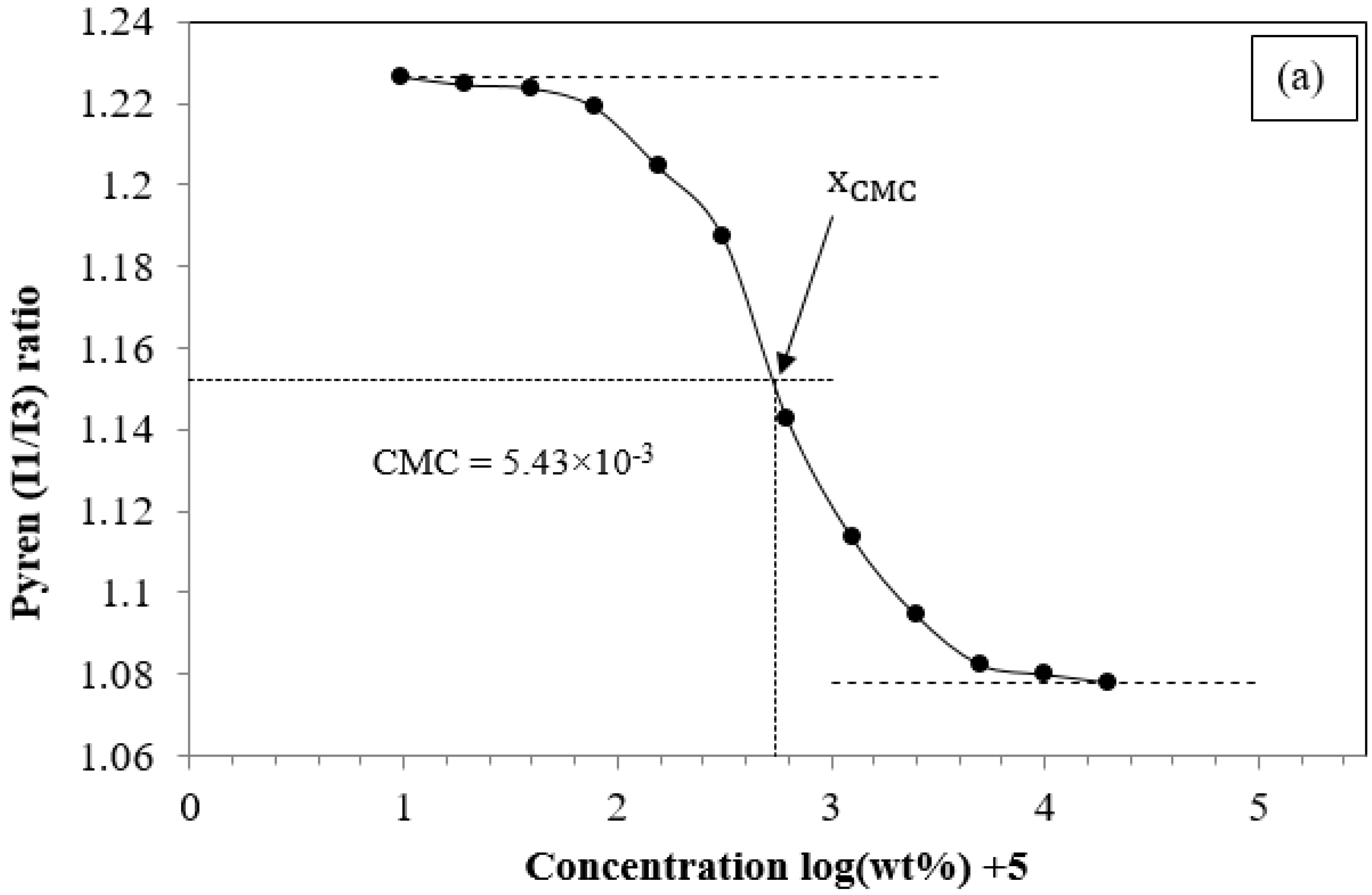
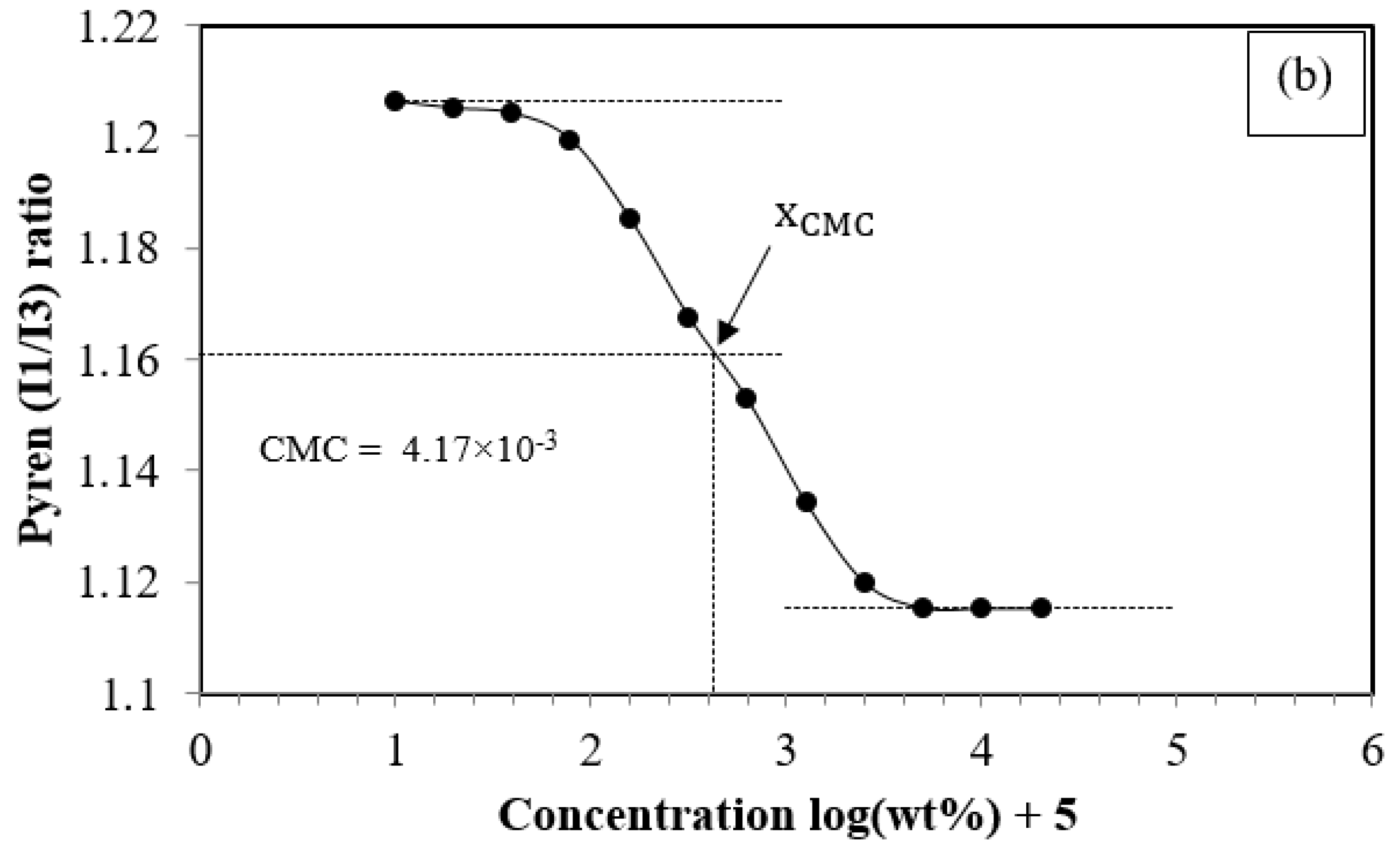
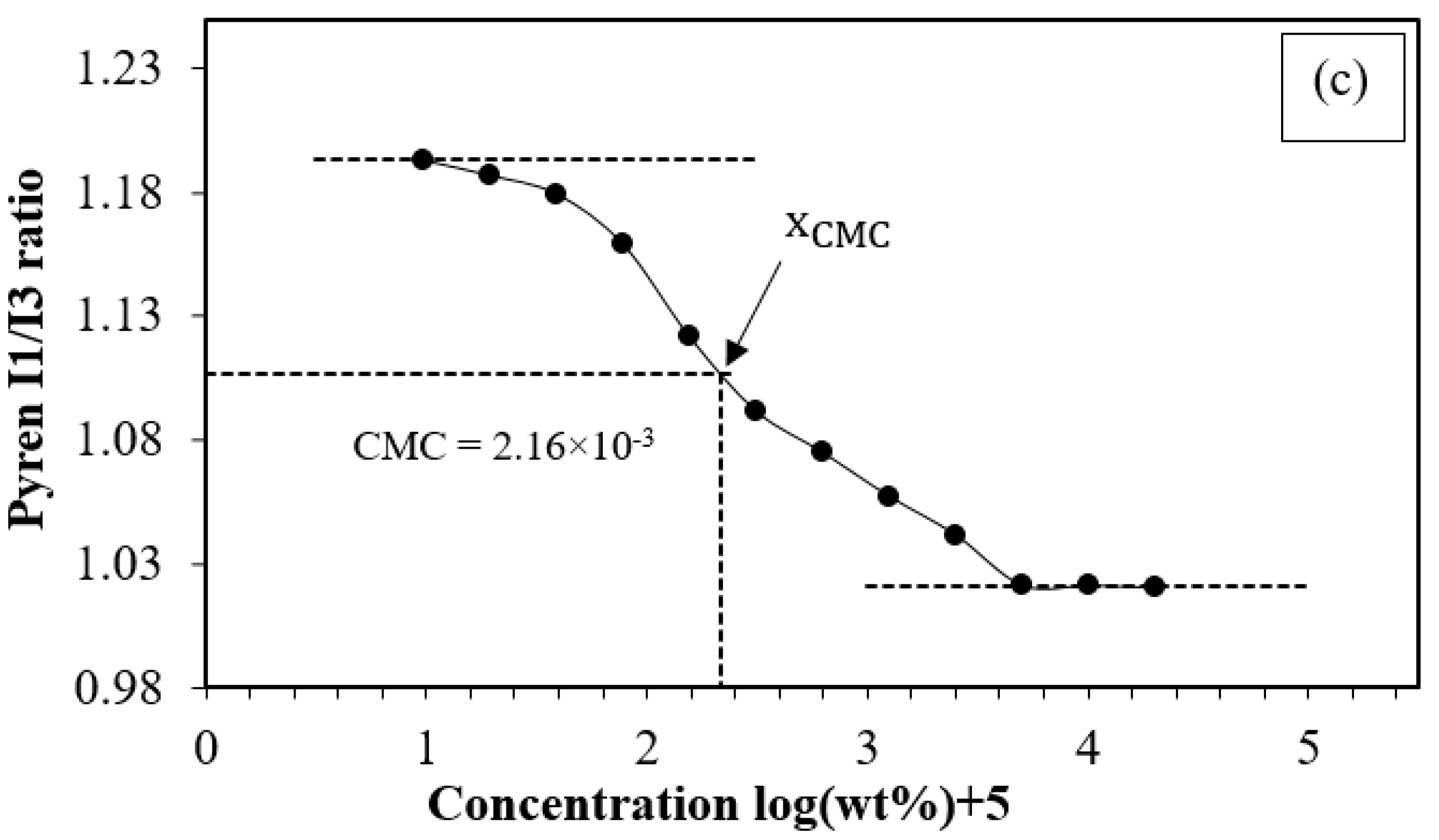
3.2.1. The Critical Micelle Concentration of PCL-PEG-PCL
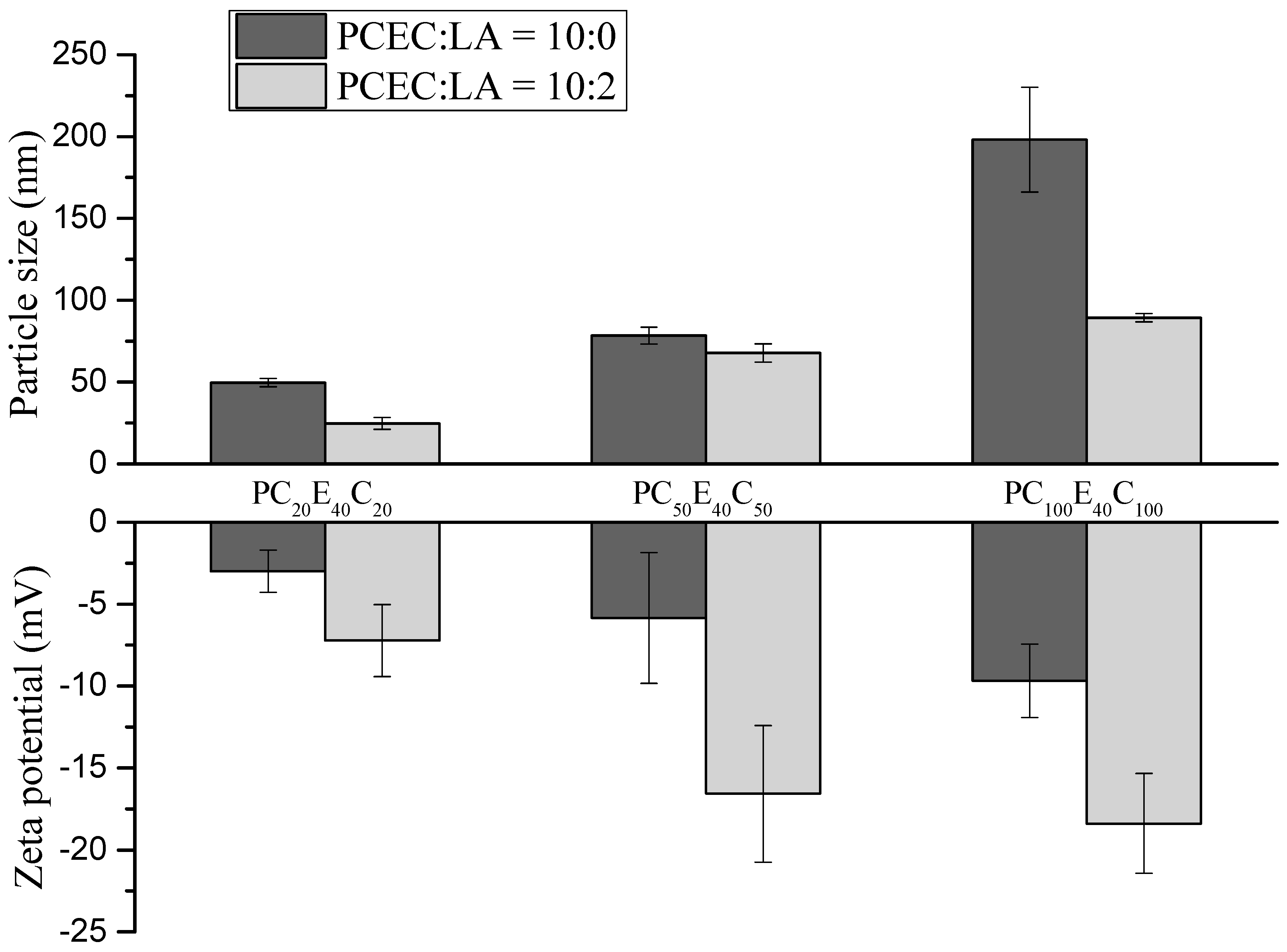
3.2.2. Particle Size and Surface Charges of the Micelles
3.2.3. Transmission Electron Microscopy of the Micelles
3.3. Drug Loading Content of LA in Micelle
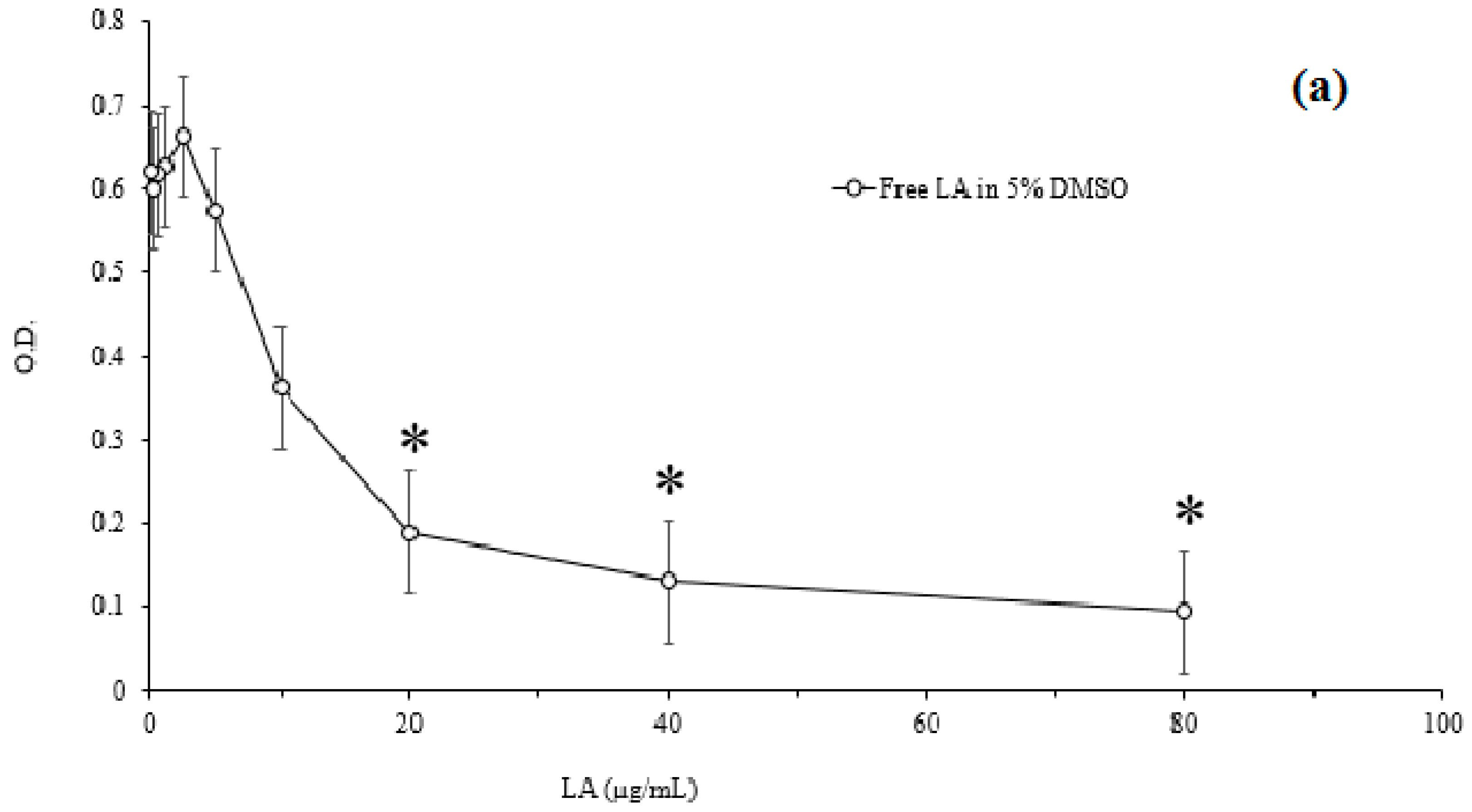
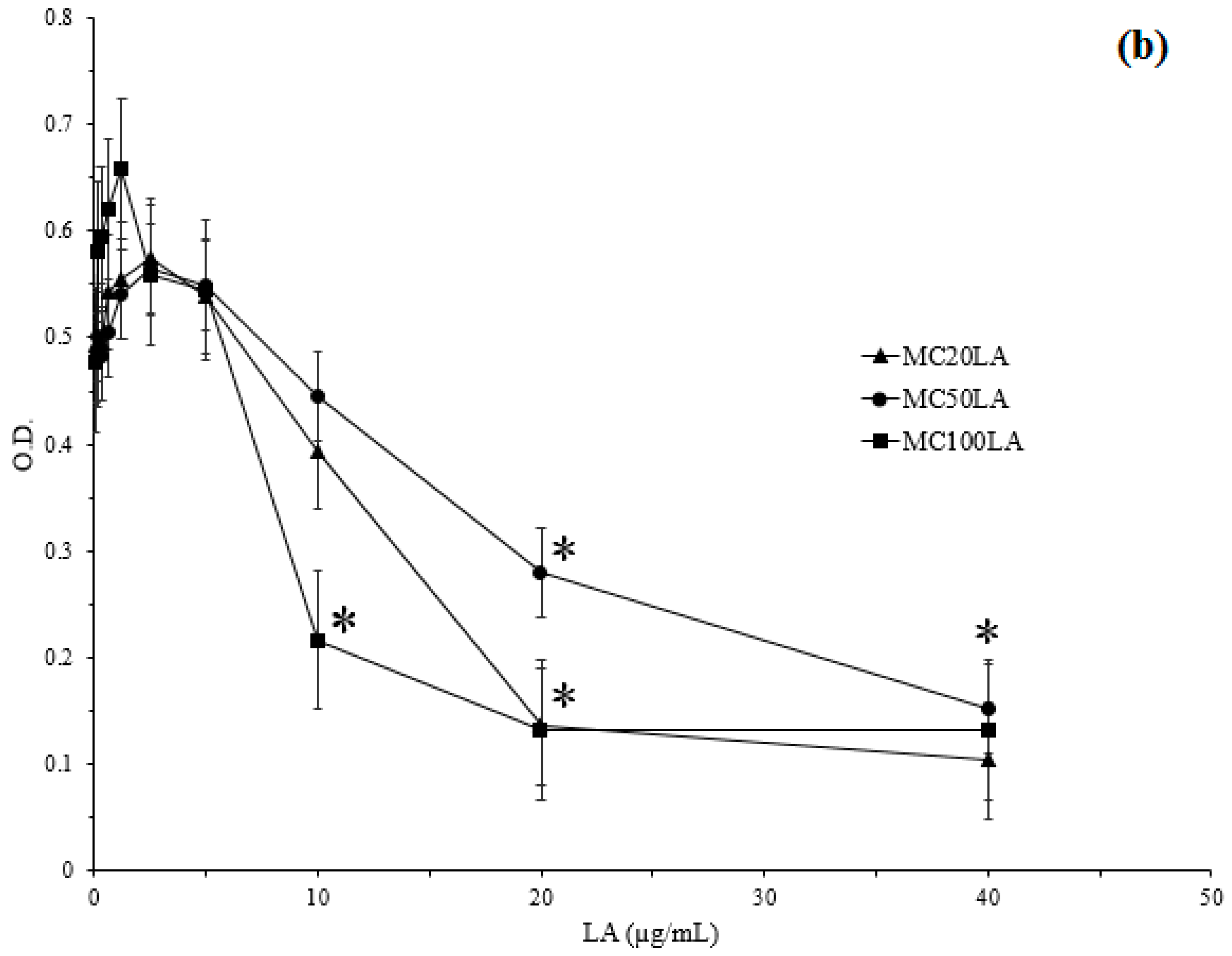
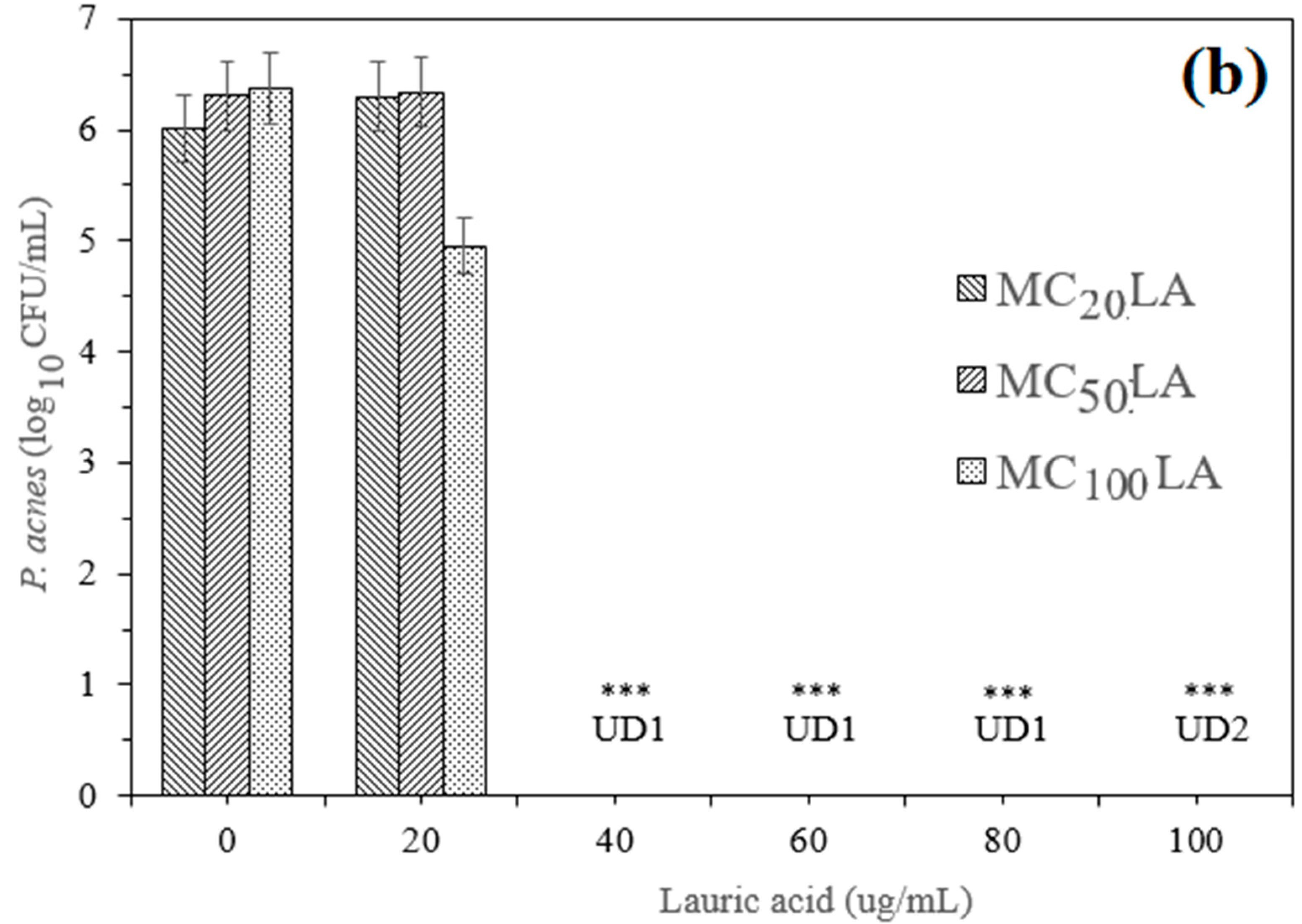
3.4. Minimum Inhibitory Concentration
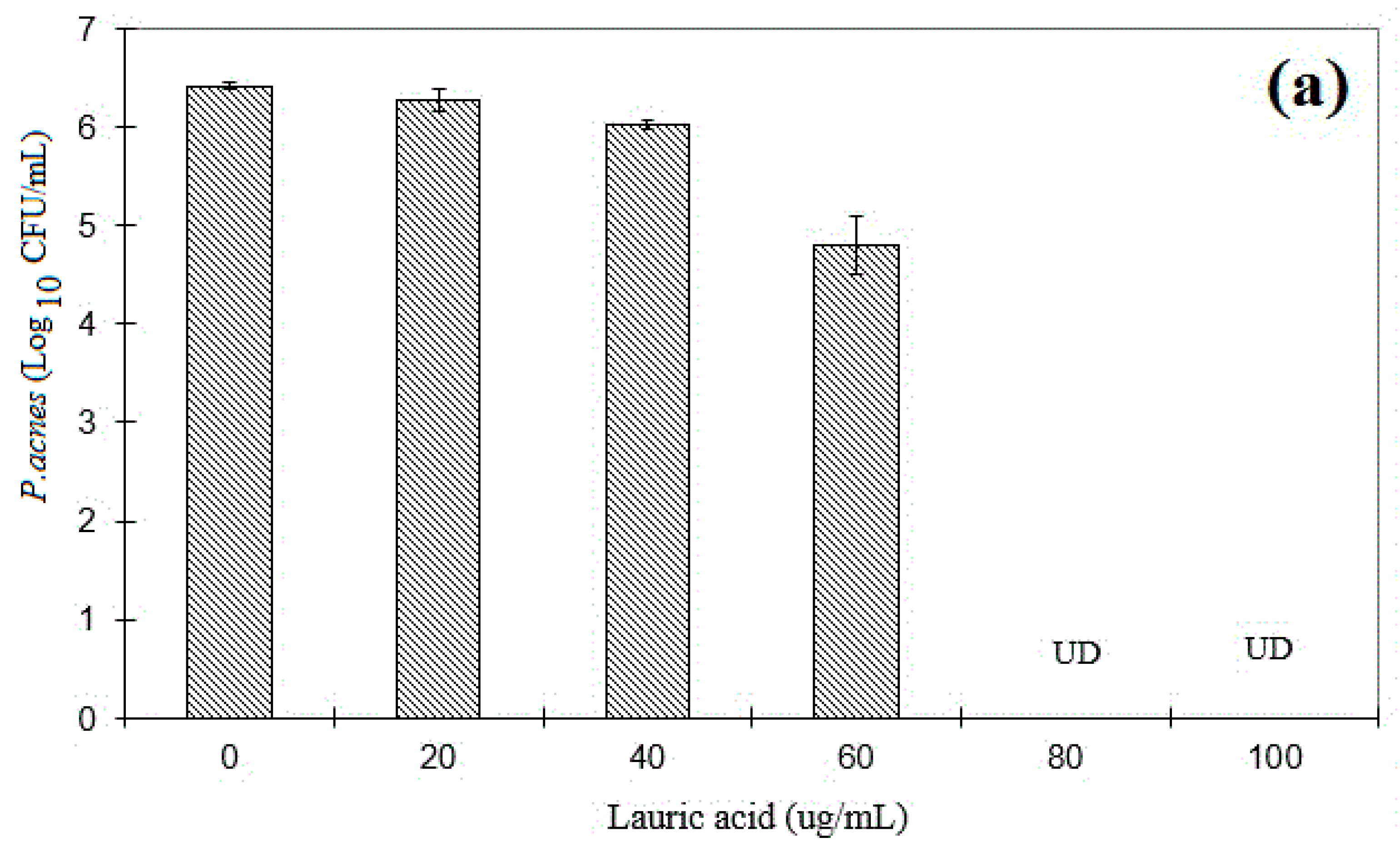
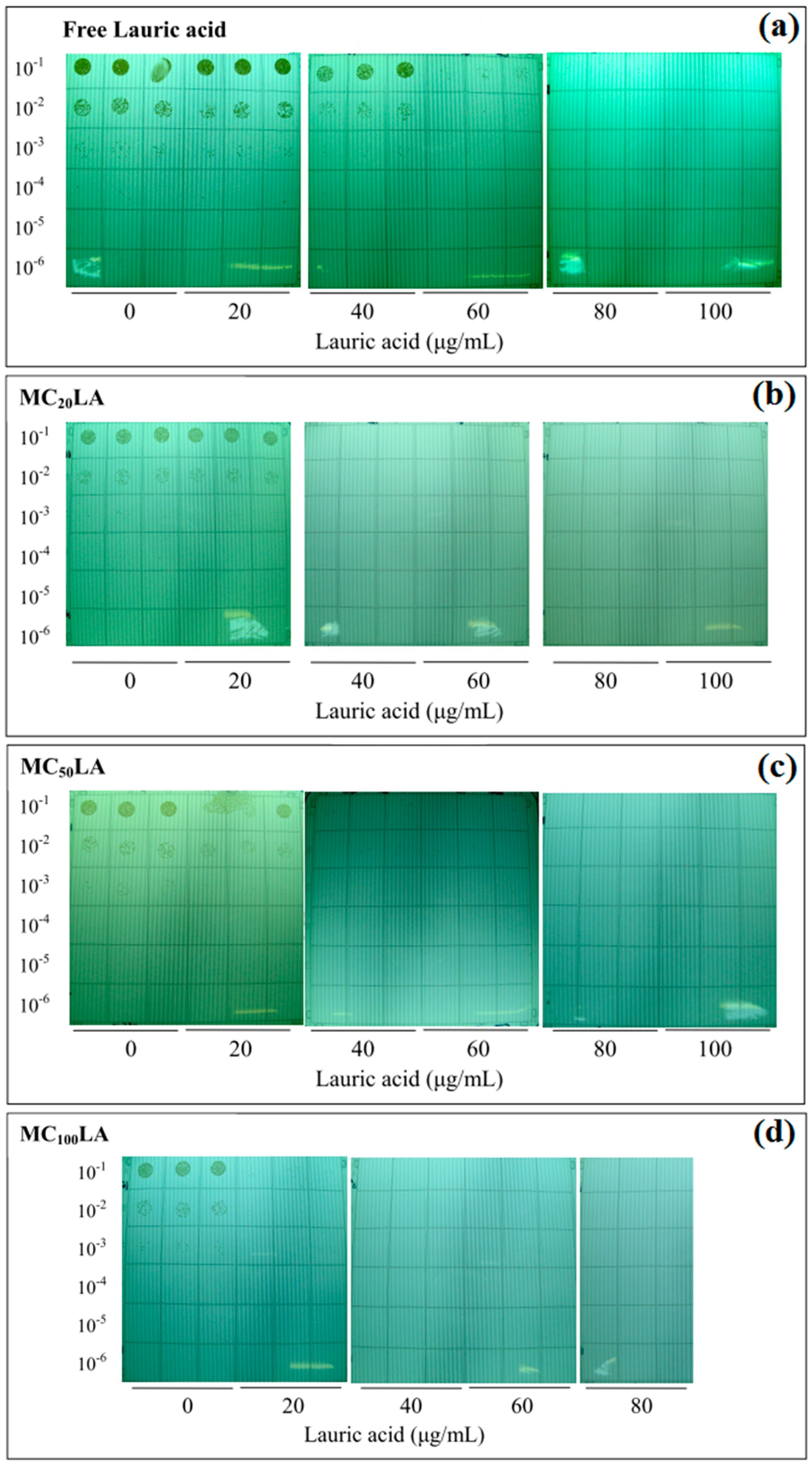
3.5. The Minimum Bactericidal Concentration
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dessinioti, C.; Katsambas, A.D. The role of Propionibacterium acnes in acne pathogenesis: Facts and controversies. Clin. Dermatol. 2010, 28, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Tsai, T.H.; Chuang, L.T.; Li, Y.Y.; Zouboulis, C.C.; Tsai, P.J. Anti-bacterial and anti-inflammatory properties of capric acid against Propionibacterium acnes: A comparative study with lauric acid. J. Dermatol. Sci. 2014, 73, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Pornpattananangkul, D.; Nakatsuji, T.; Chan, M.; Carson, D.; Huang, C.M.; Zhang, L. The antimicrobial activity of liposomal lauric acids against Propionibacterium acnes. Biomaterials 2009, 30, 6035–6040. [Google Scholar] [CrossRef] [PubMed]
- Zouboulis, C.C.; Eady, A.; Philpott, M.; Goldsmith, L.A.; Orfanos, C.; Cunliffe, W.C.; Rosenfield, R. What is the pathogenesis of acne? Exp. Dermatol. 2005, 14, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Bojar, R.A.; Holland, K.T. Acne and Propionibacterium acnes. Clin. Dermatol. 2004, 22, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Pornpattananangkul, D.; Fu, V.; Thamphiwatana, S.; Zhang, L.; Chen, M.; Vecchio, J.; Gao, W.; Huang, C.M.; Zhang, L. In vivo treatment of Propionibacterium acnes infection with liposomal lauric acids. Adv. Healthc. Mater. 2013, 2, 1322–1328. [Google Scholar] [CrossRef] [PubMed]
- Sabetta, J.R.; Rana, V.P.; Vadasdi, K.B.; Greene, R.T.; Cunningham, J.G.; Miller, S.R.; Sethi, P.M. Efficacy of topical benzoyl peroxide on the reduction of Propionibacterium acnes during shoulder surgery. J. Shoulder Elbow Surg. 2015, 24, 995–1004. [Google Scholar] [CrossRef] [PubMed]
- Anh, N.T.H.; Cuong, N.V.; Hoang, N.K. Formation of Biodegradable Copolymeric Nanoparticles for Anticancer Drug Delivery. In Proceedings of the Third International Conference on the Development of Biomedical Engineering in Vietnam, Ho Chi Minh City, Vietnam, 11–14 January 2010; Van Toi, V., Khoa, T., Eds.; Springer Berlin Heidelberg: Berlin, Germany, 2010; pp. 203–206. [Google Scholar]
- Cuong, N.V.; Li, Y.L.; Hsieh, M.F. In Vitro Biocompatibility of Folate-Decorated Star-Shaped Copolymeric Micelle for Targeted Drug Delivery. In Proceedings of the 5th Kuala Lumpur International Conference on Biomedical Engineering 2011, Kuala Lumpur, Malaysia, 20–23 June 2011; Osman, N., Abas, W., Wahab, A., Ting, H., Eds.; Springer Berlin Heidelberg: Berlin, Germany, 2011; pp. 84–87. [Google Scholar]
- Jiang, Z.; Hao, J.; You, Y.; Gu, Q.; Cao, W.; Deng, X. Biodegradable thermogelling hydrogel of P(CL-GL)-PEG-P(CL-GL) triblock copolymer: Degradation and drug release behavior. J. Pharm. Sci. 2009, 98, 2603–2610. [Google Scholar] [CrossRef] [PubMed]
- Khodaverdi, E.; Golmohammadian, A.; Mohajeri, S.A.; Zohuri, G.; Mirzazadeh Tekie, F.S.; Hadizadeh, F. Biodegradable in situ gel-forming controlled drug delivery system based on thermosensitive poly(epsilon-caprolactone)-poly(ethylene glycol)-poly(epsilon-caprolactone) hydrogel. ISRN Pharm. 2012, 2012, 9768–9779. [Google Scholar]
- Wei, X.W.; Gong, C.Y.; Gou, M.L.; Fu, S.Z.; Guo, Q.F.; Shi, S.; Luo, F.; Guo, G.; Qiu, L.Y.; Qian, Z.Y. Biodegradable poly(ɛ-caprolactone)–poly(ethylene glycol) copolymers as drug delivery system. Int. J. Pharm. 2009, 381, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, A.K.; Jana, U.; Manna, P.K.; Mohanta, G.P. Synthesis and evaluation of MePEG-PCL diblock copolymers: Surface properties and controlled release behavior. Prog. Biomater. 2015, 4, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Hallouard, F.; Briancon, S.; Anto, N.; Li, X.; Vandamme, T.; Fessi, H. Influence of diblock copolymer PCL-mPEG and of various iodinated oils on the formulation by the emulsion-solvent diffusion process of radiopaque polymeric nanoparticles. J. Pharm. Sci. 2013, 102, 4150–4158. [Google Scholar] [CrossRef] [PubMed]
- Gou, M.; Gong, C.; Zhang, J.; Wang, X.; Wang, X.; Gu, Y.; Guo, G.; Chen, L.; Luo, F.; Zhao, X.; et al. Polymeric matrix for drug delivery: Honokiol-loaded PCL-PEG-PCL nanoparticles in PEG-PCL-PEG thermosensitive hydrogel. J. Biomed. Mater. Res. A 2010, 93, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.C.; Alani, A.W.G.; Rao, D.A.; Rockich, N.C.; Kwon, G.S. Multi-drug loaded polymeric micelles for simultaneous delivery of poorly soluble anticancer drugs. J. Control. Release 2009, 140, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, J.; Carpena, P.; Molina-Bolı́var, J.A.; Carnero Ruiz, C. On the determination of the critical micelle concentration by the pyrene 1:3 ratio method. J. Colloid Interface Sci. 2003, 258, 116–122. [Google Scholar] [CrossRef]
- Rioux, V.C.D.; Bouriel, M.; Legrand, P. High performance liquid chromatography of fatty acids as naphthacyl derivatives. Analusis 1999, 27, 186–193. [Google Scholar] [CrossRef]
- Nakatsuji, T.; Kao, M.C.; Fang, J.Y.; Zouboulis, C.C.; Zhang, L.; Gallo, R.L.; Huang, C.M. Antimicrobial property of lauric acid against Propionibacterium acnes: Its therapeutic potential for inflammatory acne vulgaris. J. Investig. Dermatol. 2009, 129, 2480–2488. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Tudela, J.L.; Barchiesi, F.; Bille, J.; Chryssanthou, E.; Cuenca-Estrella, M.; Denning, D.; Donnelly, J.P.; Dupont, B.; Fegeler, W.; Moore, C.; et al. Method for the determination of minimum inhibitory concentration (MIC) by broth dilution of fermentative yeasts. Clin. Microbiol. Infect. 2003, 9, i–viii. [Google Scholar] [CrossRef]
- Holla, G.; Yeluri, R.; Munshi, A.K. Evaluation of minimum inhibitory and minimum bactericidal concentration of nano-silver base inorganic anti-microbial agent (Novaron(®)) against streptococcus mutans. Contemp. Clin. Dent. 2012, 3, 288–293. [Google Scholar] [PubMed]
- Gong, C.Y.; Shi, S.; Wu, L.; Gou, M.; Yin, Q.; Guo, Q.; Dong, P.; Zhang, F.; Luo, F.; Zhao, X.; et al. Biodegradable in situ gel-forming controlled drug delivery system based on thermosensitive PCL-PEG-PCL hydrogel: Part 1-Synthesis, characterization, and acute toxicity evaluation. J. Pharm. Sci. 2009, 98, 4684–4694. [Google Scholar] [CrossRef] [PubMed]
- Gong, C.Y.; Wu, Q.J.; Dong, P.W.; Shi, S.; Fu, S.Z.; Guo, G.; Hu, H.Z.; Zhao, X.; Wei, Y.Q.; Qian, Z.Y. Acute toxicity evaluation of biodegradable in situ gel-forming controlled drug delivery system based on thermosensitive PEG-PCL-PEG hydrogel. J. Biomed. Mater. Res. B 2009, 91, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Tamboli, V.; Mishra, G.P.; Mitra, A.K. Novel pentablock copolymer (PLA-PCL-PEG-PCL-PLA) based nanoparticles for controlled drug delivery: Effect of copolymer compositions on the crystallinity of copolymers and in vitro drug release profile from nanoparticles. Colloid Polym. Sci. 2013, 291, 1235–1245. [Google Scholar] [CrossRef] [PubMed]
- Tamboli, V.M. Pentablock Copolymers Based Controlled Release Formulations of Small and Macromolecules for Ophthalmic Applications. Ph.D. Thesis, School of Pharmacy and Department of Chemistry, University of Missouri-Kansas City, Kansas City, MI, USA, 2012. [Google Scholar]
- Liu, C.B.; Gong, C.Y.; Huang, M.J.; Wang, J.W.; Pan, Y.F.; Zhang, Y.D.; Li, G.Z.; Gou, M.L.; Wang, K.; Tu, M.J.; et al. Thermoreversible gel-sol behavior of biodegradable PCL-PEG-PCL triblock copolymer in aqueous solutions. J. Biomed. Mater. Res. B 2008, 84, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Piao, L.; Dai, Z.L.; Deng, M.X.; Chen, X.S.; Jing, X.B. Synthesis and characterization of PCL/PEG/PCL triblock copolymers by using calcium catalyst. Polymer 2003, 44, 2025–2031. [Google Scholar] [CrossRef]
- Zhang, X.; Jackson, J.K.; Burt, H.M. Determination of surfactant critical micelle concentration by a novel fluorescence depolarization technique. J. Biochem. Biophys. Methods 1996, 31, 145–150. [Google Scholar] [CrossRef]
- Hu, Y.; Jiang, X.; Ding, Y.; Zhang, L.; Yang, C.; Zhang, J.; Chen, J.; Yang, Y. Preparation and drug release behaviors of nimodipine-loaded poly(caprolactone)-poly(ethylene oxide)-polylactide amphiphilic copolymer nanoparticles. Biomaterials 2003, 24, 2395–2404. [Google Scholar] [CrossRef]
- Ahmad, Z.; Shah, A.; Siddiq, M.; Kraatz, H.-B. Polymeric micelles as drug delivery vehicles. RSC Adv. 2014, 4, 17028–17038. [Google Scholar] [CrossRef]
- Hoang, B.; Lee, H.; Reilly, R.M.; Allen, C. Noninvasive monitoring of the fate of 111In-labeled block copolymer micelles by high resolution and high sensitivity microSPECT/CT imaging. Mol. Pharm. 2009, 6, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Govender, T.; Stolnik, S.; Garnett, M.C.; Illum, L.; Davis, S.S. PLGA nanoparticles prepared by nanoprecipitation: Drug loading and release studies of a water soluble drug. J. Control Release 1999, 57, 171–185. [Google Scholar] [CrossRef]
- Hu, Y.; Jiang, X.; Ding, Y.; Zhang, L.; Yang, C.; Zhang, J.; Chen, J.; Yang, Y. Degradation behavior of poly(ε-caprolactone)-b-poly(ethylene glycol)-b-poly(ε-caprolactone) micelles in aqueous solution. Biomacromolecules 2004, 5, 1756–1762. [Google Scholar] [CrossRef] [PubMed]
- Kumari, A.; Singla, R.; Guliani, A.; Yadav, S.K. Nanoencapsulation for drug delivery. EXCLI J. 2014, 13, 265–286. [Google Scholar] [PubMed]
- Trivedi, R.; Kompella, U.B. Nanomicellar formulations for sustained drug delivery: Strategies and underlying principles. Nanomedicine 2010, 5, 485–505. [Google Scholar] [CrossRef] [PubMed]
- Allan, S.; Hoffman, J.Y.L. Handbook of Materials for Nanomedicine; Pan Stanford: Singapore, 2010; Volume 1, p. 868. [Google Scholar]
- Glavas, L.; Olsén, P.; Odelius, K.; Albertsson, A.-C. Achieving micelle control through core crystallinity. Biomacromolecules 2013, 14, 4150–4156. [Google Scholar] [CrossRef] [PubMed]
- Webster, G.F.; Rawlings, A.V. Acne and Its Therapy (Basic and Clinical Dermatology), 1st ed.; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Dannenfelser, R.-M.; Yalkowsky, S.H. QSAR in Environmental toxicology database of aqueous solubility for organic non-electrolytes. Sci. Total Environ. 1991, 109, 625–628. [Google Scholar] [CrossRef]
- Elton, L.S.; Lidiane, A.A.; Mariana, J.V.T.; Maria, I.Y.; Rodrigo, L.O.; Luis, M.F.; Maria, A.R.C.; Simone, G.S.; Gisele, A.C.G.; Ricardo, J.A.; et al. Solid lipid nanoparticles loaded with retinoic acid and lauric acid as an alternative for topical treatment of acne vulgaris. J. Nanosci. Nanotechnol. 2014, 14, 1–8. [Google Scholar]
- Bai, M.Y.; Hsueh, Y.W. Evaluation of silk fibroin protein/poly(vinyl alcohol) transparent membranes as prospective patch for acne care. J. Bioact. Compat. Polym. 2015, 30, 490–508. [Google Scholar] [CrossRef]
- Wang, Y.; Kuo, S.; Shu, M.; Yu, J.; Huang, S.; Dai, A.; Two, A.; Gallo, R.L.; Huang, C.M. Staphylococcus epidermidis in the human skin microbiome mediates fermentation to inhibit the growth of Propionibacterium acnes: Implications of probiotics in acne vulgaris. Appl. Microbiol. Biotechnol. 2014, 98, 411–424. [Google Scholar] [CrossRef] [PubMed]
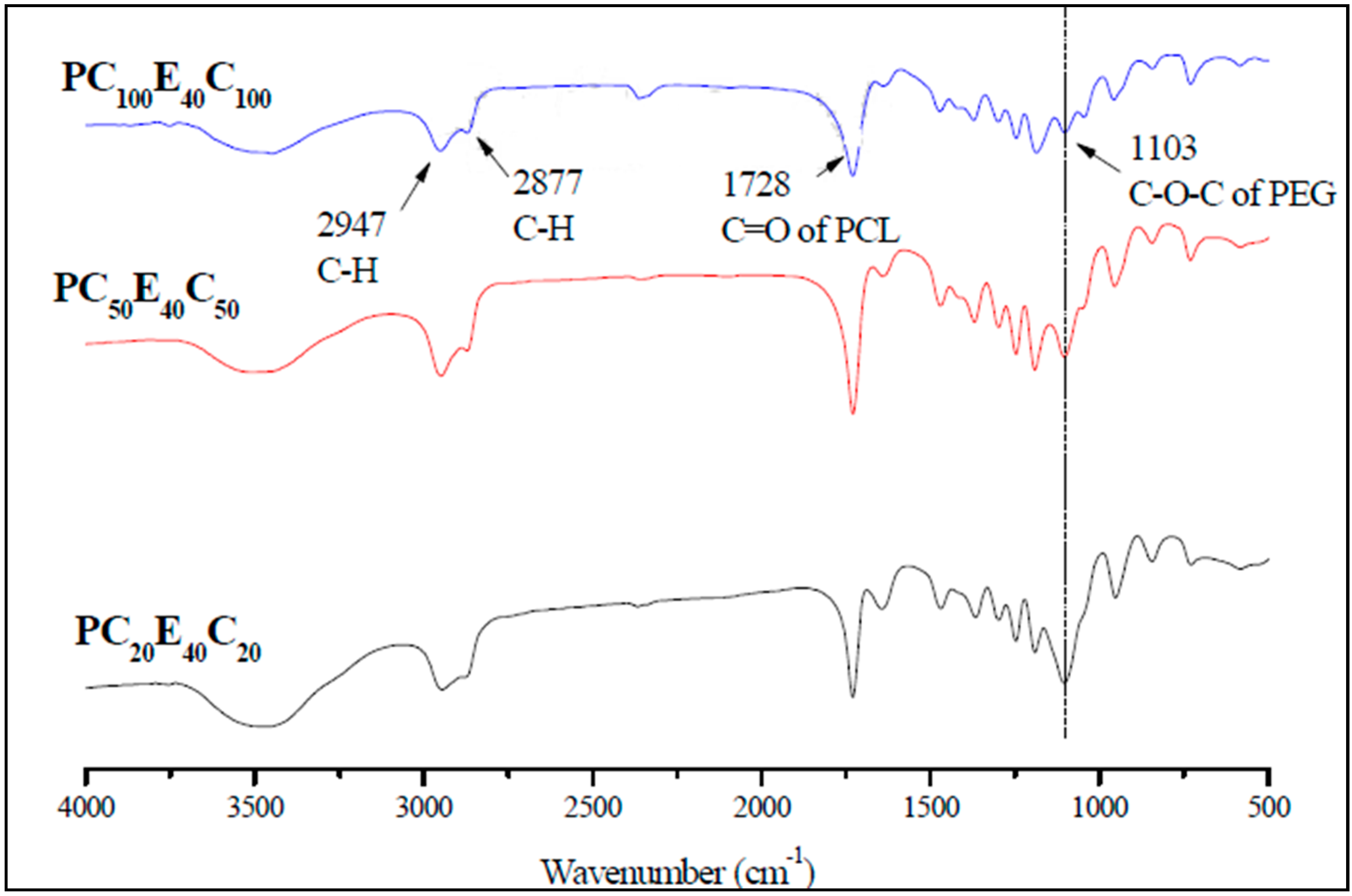



| PCEC copolymer | PCEC:LA (mg:mg) | Abbreviation |
|---|---|---|
| PC20E40C20 | 10:0 | MC20 |
| 10:2 | MC20LA | |
| PC50E40C50 | 10:0 | MC50 |
| 10:2 | MC50LA | |
| PC100E40C100 | 10:0 | MC100 |
| 10:2 | MC100LA |
| Polydispersity index | CMC (wt %) | |||||
|---|---|---|---|---|---|---|
| PC20E40C20 | 8000 | 8127 | 8320 | 9727 | 1.17 | 5.43 × 10−3 |
| PC50E40C50 | 14,000 | 13,698 | 10,831 | 13,999 | 1.29 | 4.17 × 10−3 |
| PC100E40C100 | 24,000 | 22,571 | 14,154 | 18,838 | 1.33 | 2.16 × 10−3 |
| Micelle | PCEC:LA (mg/mg) | Average particle size (nm) | PDI | Zeta potential (mV) |
|---|---|---|---|---|
| MC20 | 10:0 | 50 | 0.27 | −2.98 |
| MC20LA | 10:2 | 27 | 0.33 | −7.23 |
| MC50 | 10:0 | 78 | 0.24 | −5.90 |
| MC50LA | 10:2 | 68 | 0.21 | −16.57 |
| MC100 | 10:0 | 198 | 0.14 | −9.88 |
| MC100LA | 10:2 | 89 | 0.21 | −18.38 |
| Micelle | Concentration of LA in micelles a (µg/mL) | Concentration of micelles b (mg/mL) | Drug loading content c (%) |
|---|---|---|---|
| MC20LA | 172.24 ± 6.19 | 5.27 ± 0.06 | 3.27 ± 0.15 |
| MC50LA | 323.75 ± 15.33 | 3.43 ± 0.7 | 9.75 ± 2.35 |
| MC100LA | 105.30 ± 12.56 | 0.7 ± 0.1 | 15.42 ± 4.02 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, T.-Q.-M.; Hsieh, M.-F.; Chang, K.-L.; Pho, Q.-H.; Nguyen, V.-C.; Cheng, C.-Y.; Huang, C.-M. Bactericidal Effect of Lauric Acid-Loaded PCL-PEG-PCL Nano-Sized Micelles on Skin Commensal Propionibacterium acnes. Polymers 2016, 8, 321. https://doi.org/10.3390/polym8090321
Tran T-Q-M, Hsieh M-F, Chang K-L, Pho Q-H, Nguyen V-C, Cheng C-Y, Huang C-M. Bactericidal Effect of Lauric Acid-Loaded PCL-PEG-PCL Nano-Sized Micelles on Skin Commensal Propionibacterium acnes. Polymers. 2016; 8(9):321. https://doi.org/10.3390/polym8090321
Chicago/Turabian StyleTran, Thi-Quynh-Mai, Ming-Fa Hsieh, Keng-Lun Chang, Quoc-Hue Pho, Van-Cuong Nguyen, Ching-Yi Cheng, and Chun-Ming Huang. 2016. "Bactericidal Effect of Lauric Acid-Loaded PCL-PEG-PCL Nano-Sized Micelles on Skin Commensal Propionibacterium acnes" Polymers 8, no. 9: 321. https://doi.org/10.3390/polym8090321
APA StyleTran, T.-Q.-M., Hsieh, M.-F., Chang, K.-L., Pho, Q.-H., Nguyen, V.-C., Cheng, C.-Y., & Huang, C.-M. (2016). Bactericidal Effect of Lauric Acid-Loaded PCL-PEG-PCL Nano-Sized Micelles on Skin Commensal Propionibacterium acnes. Polymers, 8(9), 321. https://doi.org/10.3390/polym8090321







