Stabilization of Inverse Miniemulsions by Silyl-Protected Homopolymers
Abstract
:1. Introduction
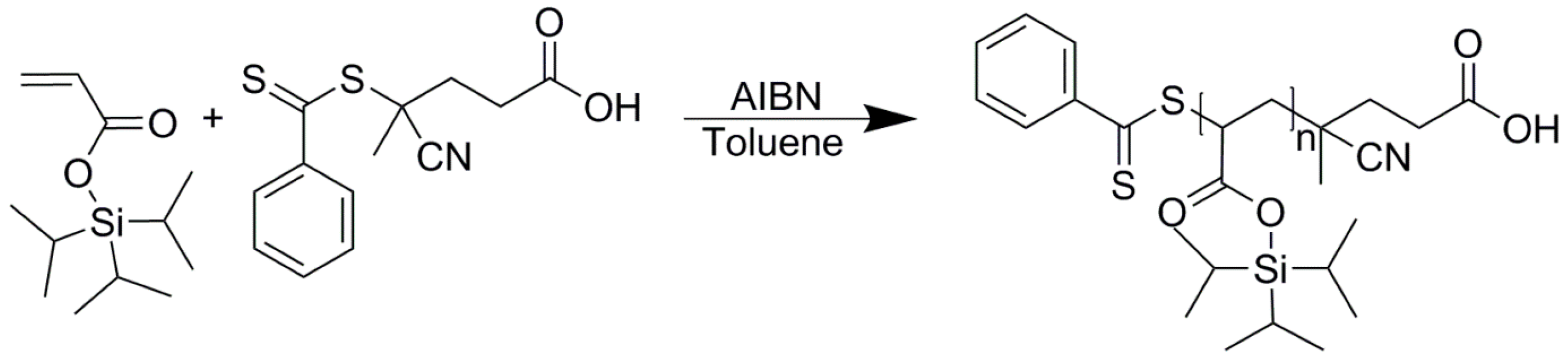
2. Materials and Methods
2.1. Materials
2.2. Analytical Tools
2.3. Analytical Tools
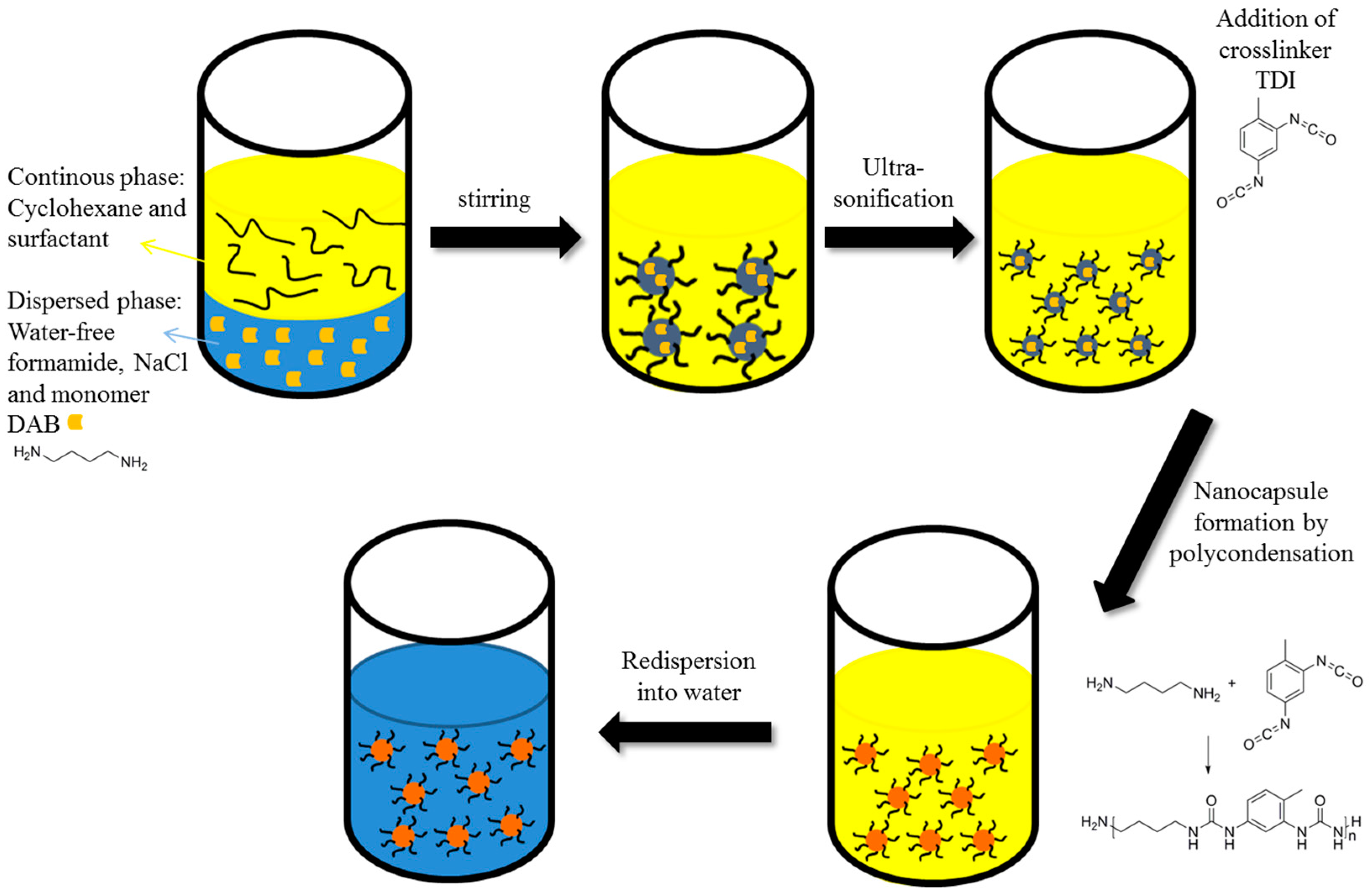
2.4. Preparation of the Inverse Miniemulsions
2.5. Deprotection of TIPSA in Solution
3. Results
4. Discussion
4.1. Stabilization of Inverse Miniemulsions by PTIPSA
4.2. Deprotection of the Triisopropylsilyl Groups
4.3. Transfer of the Nanocapsules in Water
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yan, M.; Du, J.J.; Gu, Z.; Liang, M.; Hu, Y.F.; Zhang, W.J.; Priceman, S.; Wu, L.L.; Zhou, Z.H.; Liu, Z.; et al. A novel intracellular protein delivery platform based on single-protein nanocapsules. Nat. Nanotechnol. 2010, 51, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.Y.; Liu, K.H.; Liu, D.M.; Chen, S.Y.; Chen, I.W. Temperature-sensitive nanocapsules for controlled drug release caused by magnetically triggered structural disruption. Adv. Funct. Mater. 2009, 19, 616–623. [Google Scholar] [CrossRef]
- Mora-Huertas, C.E.; Fessi, H.; Elaissari, A. Polymer-based nanocapsules for drug delivery. Int. J. Pharm. 2010, 385, 113–142. [Google Scholar] [CrossRef] [PubMed]
- Malzahn, K.; Ebert, S.; Schlegel, I.; Neudert, O.; Wagner, M.; Schütz, G.; Ide, A.; Roohi, F.; Münnenmann, K.; Crespy, D.; et al. Design and control of nanoconfinement to achieve magnetic resonance imaging contrast agents with high relaxivity. Adv. Health Mater. 2016, 5, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Caruso, F.; Spasova, M.; Saigueirino-Maceira, V.; Liz-Marzan, L.M. Multilayer assemblies of silica-encapsulated gold nanoparticles on decomposable colloid templates. Adv. Mater. 2001, 13, 1090–1094. [Google Scholar] [CrossRef]
- Caruso, F. Nanoengineering of particle surfaces. Adv. Mater. 2001, 13, 11–22. [Google Scholar] [CrossRef]
- Park, M.K.; Xia, C.J.; Advincula, R.C.; Schutz, P.; Caruso, F. Cross-linked, luminescent spherical colloidal and hollow-shell particles. Langmuir 2001, 17, 7670–7674. [Google Scholar] [CrossRef]
- Paiphansiri, U.; Tangboriboonrat, P.; Landfester, K. Polymeric nanocapsules containing an antiseptic agent obtained by controlled nanoprecipitation onto water-in-oil miniemulsion droplets. Macromol. Biosci. 2006, 6, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Staff, R.H.; Landfester, K.; Crespy, D. Recent advances in the emulsion solvent evaporation technique for the preparation of nanoparticles and nanocapsules. Adv. Polym. Sci. 2013, 262, 329–344. [Google Scholar]
- Alvarez-Roman, R.; Barre, G.; Guy, R.H.; Fessi, H. Biodegradable polymer nanocapsules containing a sunscreen agent: Preparation and photoprotection. Eur. J. Pharm. Biopharm. 2001, 52, 191–195. [Google Scholar] [CrossRef]
- Crespy, D.; Landfester, K. Preparation of nylon 6 nanoparticles and nanocapsules by two novel miniemulsion/solvent displacement hybrid techniques. Macromol. Chem. Physic. 2007, 208, 457–466. [Google Scholar] [CrossRef]
- Fickert, J.; Makowski, M.; Kappl, M.; Landfester, K.; Crespy, D. Efficient encapsulation of self-healing agents in polymer nanocontainers functionalized by orthogonal reactions. Macromolecules 2012, 45, 6324–6332. [Google Scholar] [CrossRef]
- Luo, Y.W.; Zhou, X.D. Nanoencapsulation of a hydrophobic compound by a miniemulsion polymerization process. J. Polym. Sci. Poly. Chem. 2004, 42, 2145–2154. [Google Scholar] [CrossRef]
- Bathfield, M.; Graillat, C.; Hamaide, T. Encapsulation of high biocompatible hydrophobe contents in nonionic nanoparticles by miniemulsion polymerization of vinyl acetate or styrene: Influence of the hydrophobe component on the polymerization. Macromol. Chem. Physic. 2005, 206, 2284–2291. [Google Scholar] [CrossRef]
- Crespy, D.; Stark, M.; Hoffmann-Richter, C.; Ziener, U.; Landfester, K. Polymeric nanoreactors for hydrophilic reagents synthesized by interfacial polycondensation on miniemulsion droplets. Macromolecules 2007, 40, 3122–3135. [Google Scholar] [CrossRef]
- Koh, H.D.; Lee, J.S. Polymeric nanocapsules containing nematic LCs as hydrophobes in miniemulsion polymerization. Macromol. Rapid Comm. 2007, 28, 315–321. [Google Scholar] [CrossRef]
- Crespy, D.; Musyanovych, A.; Landfester, K. Synthesis of polymer particles and nanocapsules stabilized with PEO/PPO containing polymerizable surfactants in miniemulsion. Colloid Polym. Sci. 2006, 284, 780–787. [Google Scholar] [CrossRef]
- Landfester, K. Synthesis of colloidal particles in miniemulsions. Annu. Rev. Mater. Res. 2006, 36, 231–279. [Google Scholar] [CrossRef]
- Li, W.W.; Yoon, J.A.; Matyjaszewski, K. Dual-reactive surfactant used for synthesis of functional nanocapsules in miniemulsion. J. Am. Chem. Soc. 2010, 132, 7823–7825. [Google Scholar] [CrossRef] [PubMed]
- Antonietti, M.; Landfester, K. Polyreactions in miniemulsions. Prog. Polym. Sci. 2002, 27, 689–757. [Google Scholar] [CrossRef]
- Medeiros, S.F.; Santos, A.M.; Fessi, H.; Elaissari, A. Synthesis of biocompatible and thermally sensitive poly(N-vinylcaprolactam) nanogels via inverse miniemulsion polymerization: Effect of the surfactant concentration. J. Polym. Sci. Poly. Chem. 2010, 48, 3932–3941. [Google Scholar] [CrossRef]
- Lin, C.L.; Chiu, W.Y.; Don, T.M. Superparamagnetic thermoresponsive composite latex via W/O miniemulsion polymerization. J. Appl. Polym. Sci. 2006, 100, 3987–3996. [Google Scholar] [CrossRef]
- Oh, J.K.; Perineau, F.; Matyjaszewski, K. Preparation of nanoparticles of well-controlled water-soluble homopolymers and block copolymers using an inverse miniemulsion ATRP. Macromolecules 2006, 39, 8003–8010. [Google Scholar] [CrossRef]
- Griffin, W.C. Classification of surface-active agents by HLB. J. Cosmet. Sci. 1949, 1, 311–326. [Google Scholar]
- Griffin, W.C. Calculation of HLB values of non-ionic surfactants. J. Cosmet. Sci. 1954, 5, 249–256. [Google Scholar]
- Kositza, M.J.; Bohne, C.; Alexandridis, P.; Hatton, T.A.; Holzwarth, J.F. Dynamics of micro- and macrophase separation of amphiphilic block-copolymers in aqueous solution. Macromolecules 1999, 32, 5539–5551. [Google Scholar] [CrossRef]
- Liu, H.Y.; Prevost, S.; Gradzielski, M. Solubilisation of oils of different polarity in aqueous solutions of pluronic trib lock copolymers. Z. Phys. Chem. 2012, 226, 675–694. [Google Scholar] [CrossRef]
- Rieger, J.; Passirani, C.; Benoit, J.P.; Van Butsele, K.; Jerome, R.; Jerome, C. Synthesis of amphiphilic copolymers of poly(ethylene oxide) and poly(epsilon-caprolactone) with different architectures, and their role in the preparation of stealthy nanoparticles. Adv. Funct. Mater. 2006, 16, 1506–1514. [Google Scholar] [CrossRef]
- Cajot, S.; Lecomte, P.; Jerome, C.; Riva, R. Novel functional degradable block copolymers for the building of reactive micelles. Polym. Chem. 2013, 4, 1025–1037. [Google Scholar] [CrossRef]
- Oh, J.K. Polylactide (PLA)-based amphiphilic block copolymers: synthesis, self-assembly, and biomedical applications. Soft Matter 2011, 7, 5096–5108. [Google Scholar] [CrossRef] [Green Version]
- Zhu, Z.X. Effects of amphiphilic diblock copolymer on drug nanoparticle formation and stability. Biomaterials 2013, 34, 10238–10248. [Google Scholar] [CrossRef] [PubMed]
- Kerwin, B.A. Polysorbates 20 and 80 used in the formulation of protein biotherapeutics: Structure and degradation pathways. J. Pharm. Sci. 2008, 97, 2924–2935. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Schlaad, H.; Smarsly, B.; Antonietti, M. Replication of lyotropic block copolymer mesophases into porous silica by nanocasting: Learning about finer details of polymer self-assembly. Langmuir 2003, 19, 4455–4459. [Google Scholar] [CrossRef]
- Klein, R.; Wurm, F. Aliphatic polyethers: Classical polymers for the 21st century. Macromol. Rapid Commun. 2015, 36, 1147–1165. [Google Scholar] [CrossRef] [PubMed]
- Chiefari, J.; Chong, Y.K.; Ercole, F.; Krstina, J.; Jeffery, J.; Le, T.P.T.; Mayadunne, R.T.A.; Meijs, G.F.; Moad, C.L.; Moad, G.; et al. Living free-radical polymerization by reversible addition-fragmentation chain transfer: The RAFT process. Macromolecules 1998, 31, 5559–5562. [Google Scholar] [CrossRef]
- Rizzardo, E.; Chiefari, J.; Chong, B.Y.K.; Ercole, F.; Krstina, J.; Jeffery, J.; Le, T.P.T.; Mayadunne, R.T.A.; Meijs, G.F.; Moad, C.L.; et al. Tailored polymers by free radical processes. Macromol. Symp. 1999, 143, 291–307. [Google Scholar] [CrossRef]
- Moad, G.; Rizzardo, E.; Thang, S.H. Living radical polymerization by the RAFT process. Aust. J. Chem. 2005, 58, 379–410. [Google Scholar] [CrossRef]
- Rizzardo, E.; Chiefari, J.; Mayadunne, R.T.A.; Moad, G.; Thang, S.H. Synthesis of defined polymers by reversible addition-fragmentation chain transfer: the RAFT process. In ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2000; Volume 768, pp. 278–296. [Google Scholar]
- Dorresteijn, R.; Ragg, R.; Rago, G.; Billecke, N.; Bonn, M.; Parekh, S.H.; Battagliarin, G.; Peneva, K.; Wagner, M.; Klapper, M.; et al. Biocompatible polylactide-block-polypeptide-block-polylactide nanocarrier. Biomacromolecules. 2013, 14, 1572–1577. [Google Scholar] [CrossRef] [PubMed]
- Geidel, C.; Schmachtel, S.; Riedinger, A.; Pfeiffer, C.; Mullen, K.; Klapper, M.; Parak, W.J. A general synthetic approach for obtaining cationic and anionic inorganic nanoparticles via encapsulation in amphiphilic copolymers. Small 2011, 7, 2929–2934. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Parowatkin, M.; Mailander, V.; Flechtner-Mors, M.; Graf, R.; Best, A.; Koynov, K.; Mohr, K.; Ziener, U.; Landfester, K.; et al. Nanocarrier for oral peptide delivery produced by polyelectrolyte complexation in nanoconfinement. Biomacromolecules 2015, 16, 2282–2287. [Google Scholar] [CrossRef] [PubMed]
- Brown, P.; Butts, C.P.; Eastoe, J. Stimuli-responsive surfactants. Soft Matter 2013, 9, 2365–2374. [Google Scholar] [CrossRef]
- Weaver, J.V.M.; Rannard, S.P.; Cooper, A.I. Polymer-mediated hierarchical and reversible emulsion droplet assembly. Angew. Chem. 2009, 121, 2165–2168. [Google Scholar] [CrossRef]
- Amalvy, J.; Unali, G.-F.; Li, Y.; Granger-Bevan, S.; Armes, S.; Binks, B.P.; Rodrigues, J.; Whitby, C.P. Synthesis of sterically stabilized polystyrene latex particles using cationic block copolymers and macromonomers and their application as stimulus-responsive particulate emulsifiers for oil-in-water emulsions. Langmuir 2004, 20, 4345–4354. [Google Scholar] [CrossRef] [PubMed]
- Dupin, D.; Armes, S.P.; Connan, C.; Reeve, P.; Baxter, S.M. How does the nature of the steric stabilizer affect the pickering emulsifier performance of lightly cross-linked, acid-swellable poly(2-vinylpyridine) latexes? Langmuir 2007, 23, 6903–6910. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Kim, T.H.; Choi, J.H.; Lee, J.Y.; Hah, S.S.; Yoo, H.O.; Hwang, S.S.; Ryu, K.N.; Kim, H.J.; Kim, J. Synthesis of a pH-sensitive PEO-based block copolymer and its application for the stabilization of iron oxide nanoparticles. Macromol. Chem. Physic 2010, 211, 1127–1136. [Google Scholar] [CrossRef]
- Lee, S.; Nam, J.H.; Kim, Y.J.; Cho, Y.J.; Kwon, N.H.; Lee, J.Y.; Kang, H.-J.; Kim, H.T.; Park, H.M.; Kim, S. Synthesis of PEO-based glucose-sensitive block copolymers and their application for preparation of superparamagnetic iron oxide nanoparticles. Macromol. Res. 2011, 19, 827–834. [Google Scholar] [CrossRef]
- Dorresteijn, R.; Billecke, N.; Parekh, S.H.; Klapper, M.; Mullen, K. Polarity reversal of nanoparticle surfaces by the use of light-sensitive polymeric emulsifiers. J. Polym. Sci. Pol. Chem. 2015, 53, 200–205. [Google Scholar] [CrossRef]
- Girifalco, L.A.; Good, R.J. A theory of the estimation of surface and interfacial energies. I. Derivation and application to interfacial tension. J. Phys. Chem. 1957, 61, 904–909. [Google Scholar] [CrossRef]
- Oyama, K.I.; Kondo, T.D. A novel and convenient chemoselective deprotection method for both silyl and acetyl groups on acidic hydroxyl groups such as phenol and carboxylic acid by using a nitrogen organic base, 1,1,3,3-tetramethylguanidine. Org. Lett. 2003, 5, 209–212. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.S.Y.; Su, F.Y. Solvent-modulated chemoselective deprotections of trialkylsilyl esters and chemoselective esterifications. Tetrahedron Lett. 2005, 46, 6305–6309. [Google Scholar] [CrossRef]
- Schelhaas, M.; Waldmann, H. Protecting group strategies in organic synthesis. Angew. Chem. Int. Edit. 1996, 35, 2056–2083. [Google Scholar] [CrossRef]
- Greene, T.W.; Wuts, P.G.M. Protective Groups in Organic Synthesis; Wiley and Sons: New York, NY, USA, 1999. [Google Scholar]
- Kocienski, P.J. Protecting Groups; George Thieme Verlag: New York, NY, USA, 1999. [Google Scholar]
- Heldmann, D.K.; Stohrer, J.; Zauner, R. 2-norbornyldimethylsilyl, ethers (NDMS): A new protecting group for alcohols and carboxylic acids. Synlett 2002, 11, 1919–1921. [Google Scholar] [CrossRef]
- Van Look, G.; Simchen, G.; Heberle, J. Silylating Agents; Fluka chemie AG: Buchs, Switzerland, 1995; Volume 2, pp. 1–45. [Google Scholar]
- Corey, E.J.; Rucker, C. Useful synthetic reagents derived from 1-triisopropylsilylpropyne and 1,3-bis-[triisopropylsilyl]propyne, direct, stereoselective synthesis of either Z-enynes or E-enynes. Tetrahedron Lett. 1982, 23, 719–722. [Google Scholar] [CrossRef]
- Corey, E.J.; Cho, H.; Rucker, C.; Hua, D.H. Studies with trialkylsilyltriflates - new syntheses and applications. Tetrahedron Lett. 1981, 22, 3455–3458. [Google Scholar] [CrossRef]
- Durand, A.; Marie, E. Macromolecular surfactants for miniemulsion polymerization. Adv. Colloid Interface 2009, 150, 90–105. [Google Scholar] [CrossRef] [PubMed]
- Behzadi, S.; Rosenauer, C.; Kappl, M.; Mohr, K.; Landfester, K.; Crespy, D. Osmotic pressure-dependent release profiles of payloads from nanocontainers by co-encapsulation of simple salts. Nanoscale 2016, 8, 12998–13005. [Google Scholar] [CrossRef] [PubMed]
- Staff, R.H.; Gallei, M.; Landfester, K.; Crespy, D. Hydrophobic nanocontainers for stimulus-selective release in aqueous environments. Macromolecules 2014, 47, 4876–4883. [Google Scholar] [CrossRef]
- Fickert, J.; Wohnhaas, C.; Turshatov, A.; Landfester, K. Copolymers structures tailored for the preparation of nanocapsules. Macromolecules 2013, 46, 573–579. [Google Scholar] [CrossRef]
- Nicolaou, K.C.; Ninkovic, S.; Sarabia, F.; Vourloumis, D.; He, Y.; Vallberg, H.; Finlay, M.R.V.; Yang, Z. Total syntheses of epothilones A and B via a macrolactonization-based strategy. J. Am. Chem. Soc. 1997, 119, 7974–7991. [Google Scholar] [CrossRef]
- Lalonde, M.; Chan, T.H. Use of organo-silicon reagents as protective groups in organic-synthesis. Synthesis-Stuttgart 1985, 9, 817–845. [Google Scholar] [CrossRef]

© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wald, S.; Wurm, F.R.; Landfester, K.; Crespy, D. Stabilization of Inverse Miniemulsions by Silyl-Protected Homopolymers. Polymers 2016, 8, 303. https://doi.org/10.3390/polym8080303
Wald S, Wurm FR, Landfester K, Crespy D. Stabilization of Inverse Miniemulsions by Silyl-Protected Homopolymers. Polymers. 2016; 8(8):303. https://doi.org/10.3390/polym8080303
Chicago/Turabian StyleWald, Sarah, Frederik R. Wurm, Katharina Landfester, and Daniel Crespy. 2016. "Stabilization of Inverse Miniemulsions by Silyl-Protected Homopolymers" Polymers 8, no. 8: 303. https://doi.org/10.3390/polym8080303
APA StyleWald, S., Wurm, F. R., Landfester, K., & Crespy, D. (2016). Stabilization of Inverse Miniemulsions by Silyl-Protected Homopolymers. Polymers, 8(8), 303. https://doi.org/10.3390/polym8080303





