Enhancement in Mechanical and Shape Memory Properties for Liquid Crystalline Polyurethane Strengthened by Graphene Oxide
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Procedure of Producing GO/LCPU Composites
2.2.1. Preparation of LCPU
2.2.2. Preparation of GO/LCPU Composite Membrane
2.3. Characterization
3. Results and Discussion
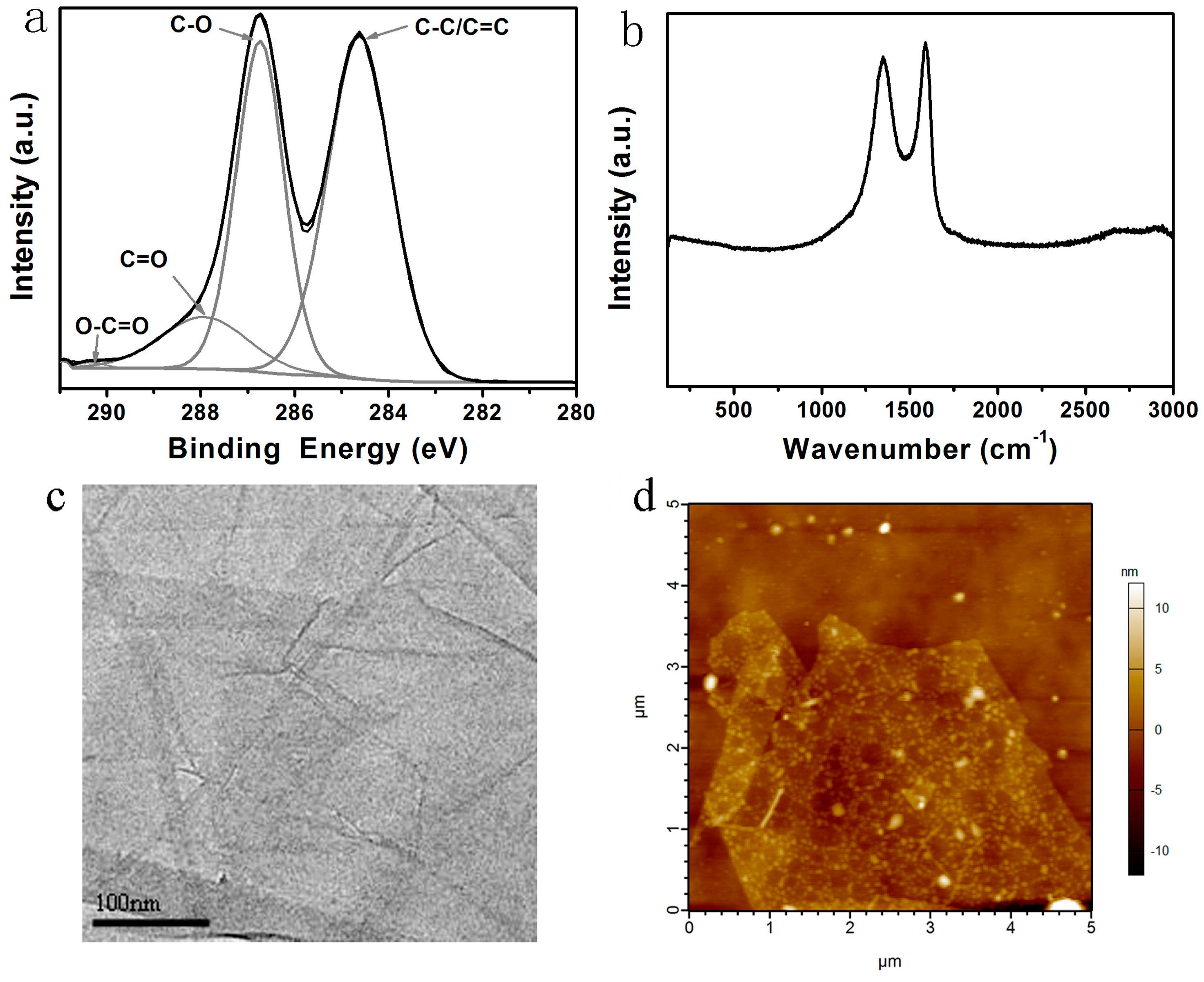
3.1. Characterization of GO
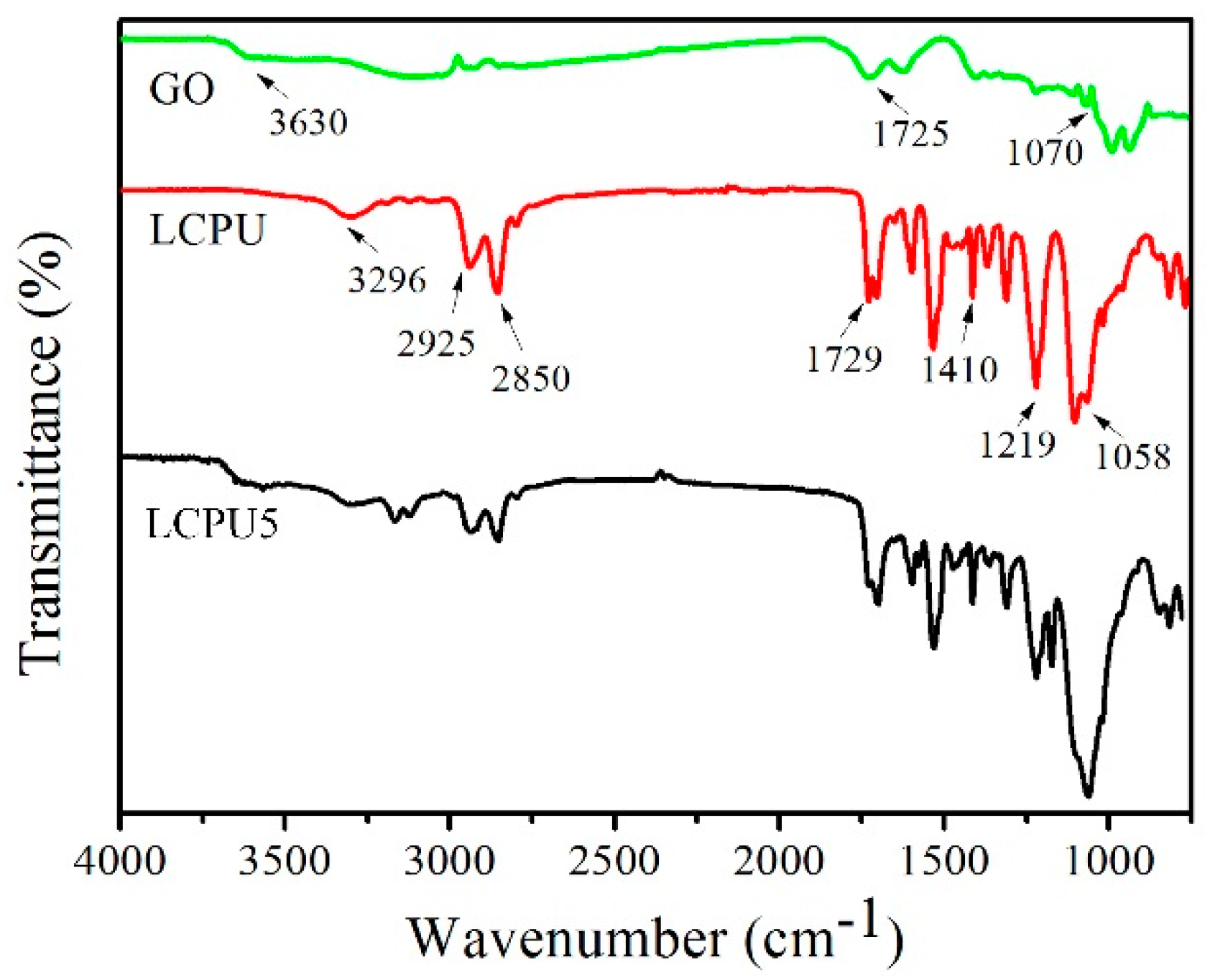
3.2. FT-IR
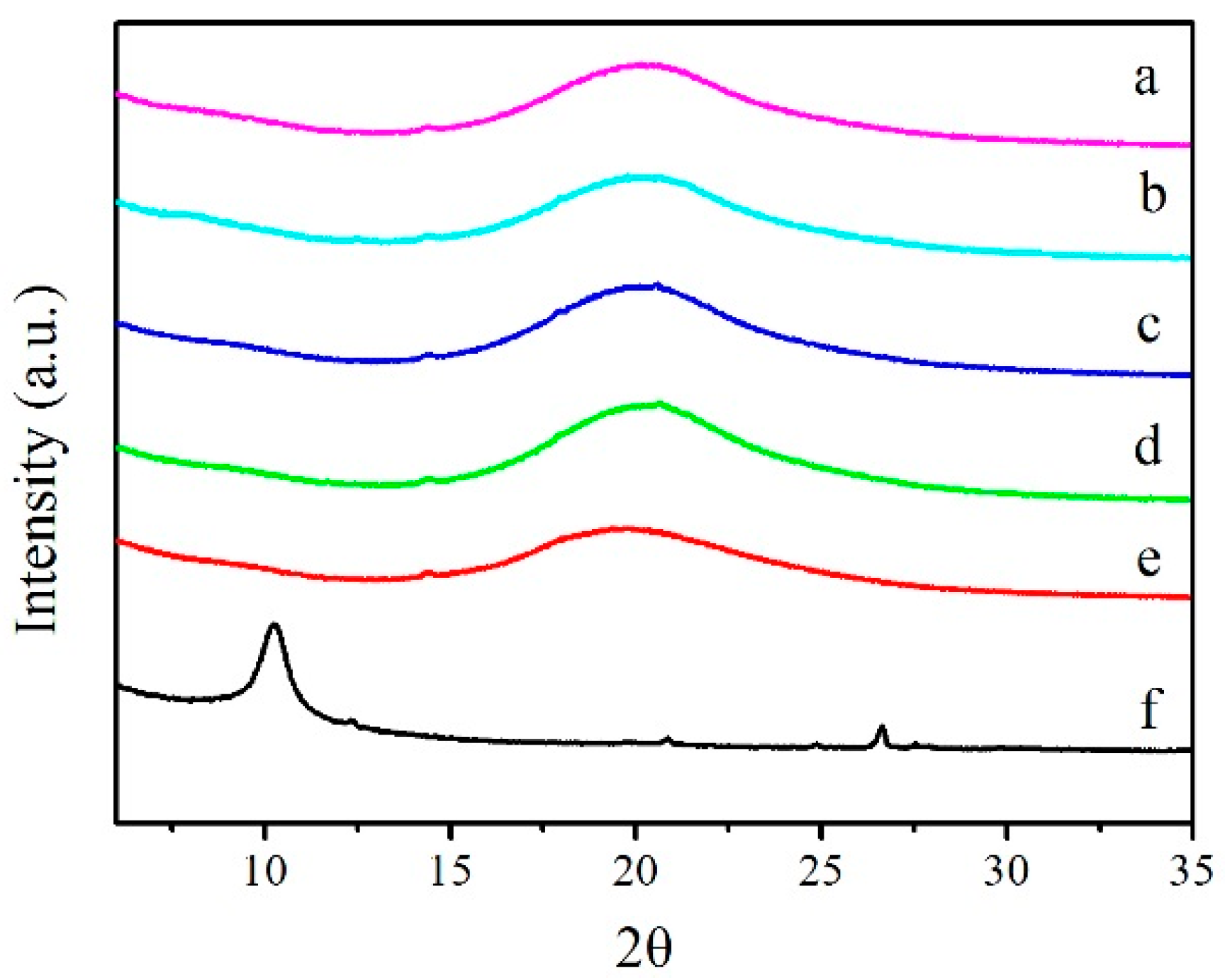
3.3. XRD
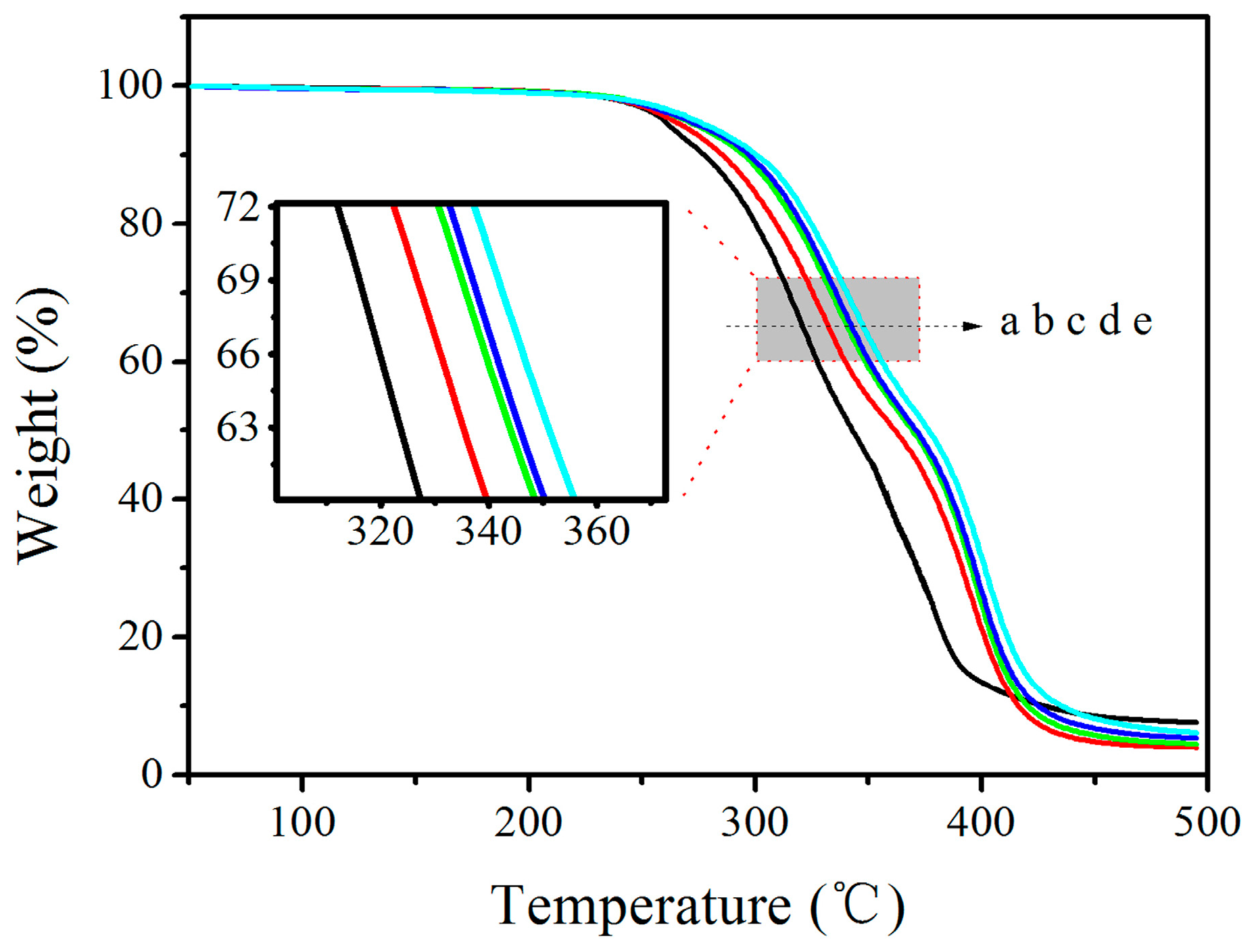
3.4. TGA
3.5. DSC
3.6. POM
3.7. SEM
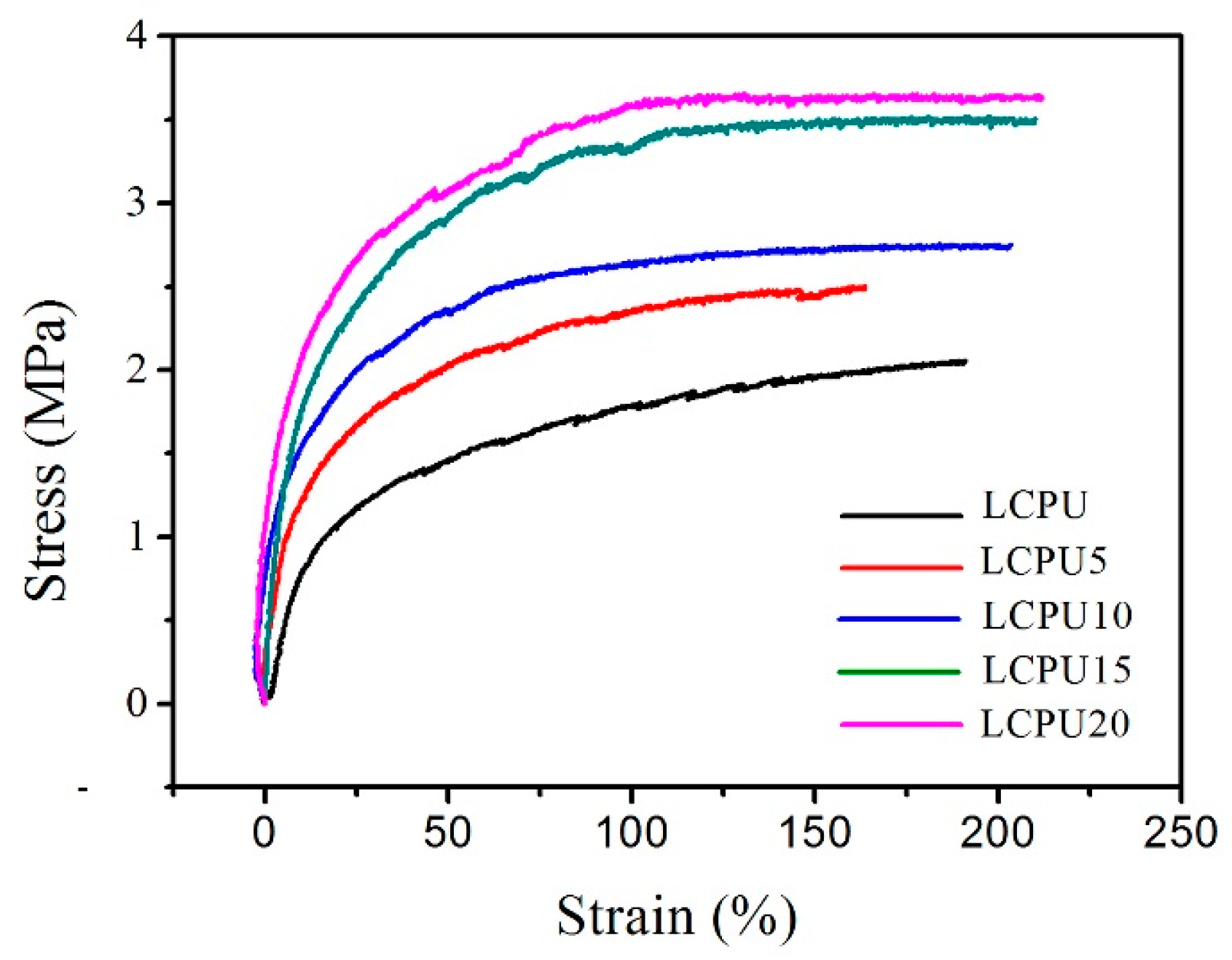
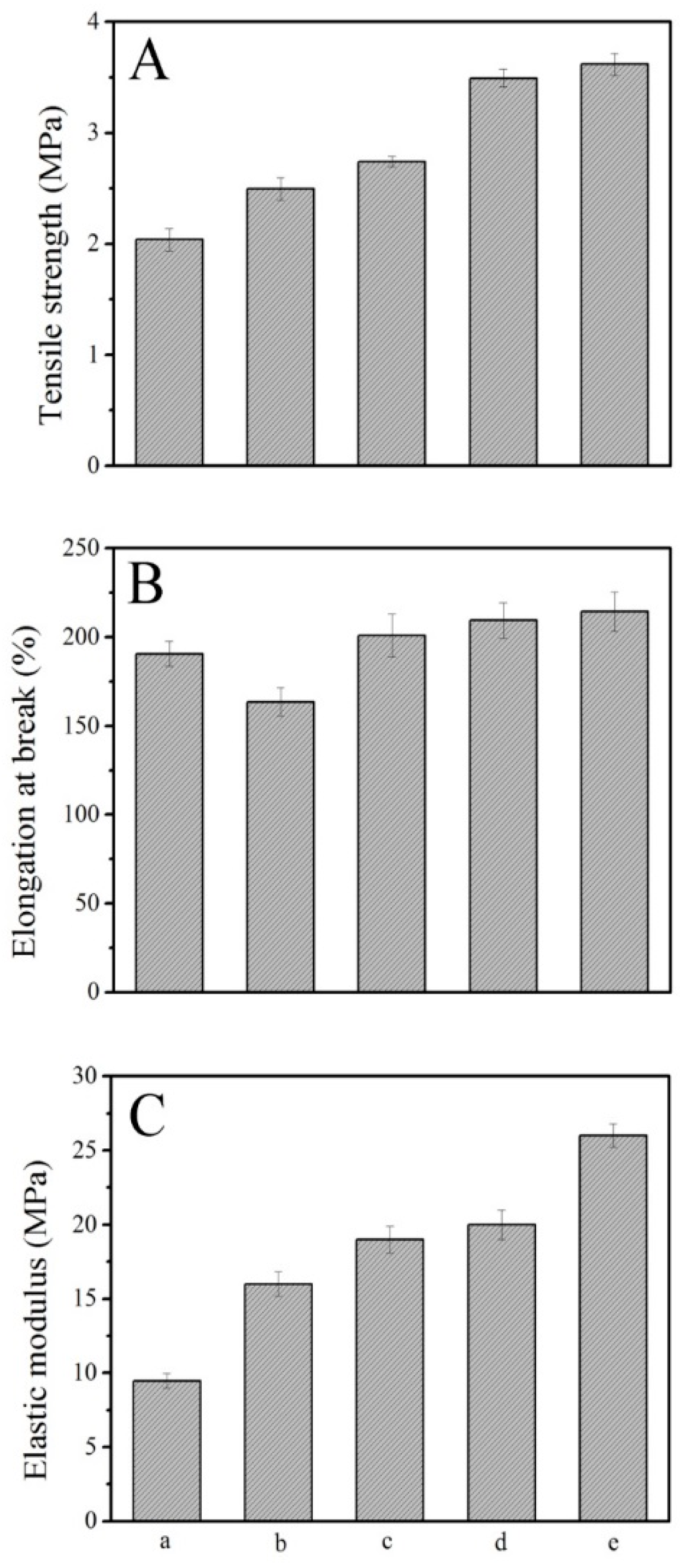
3.8. Mechanical Properties
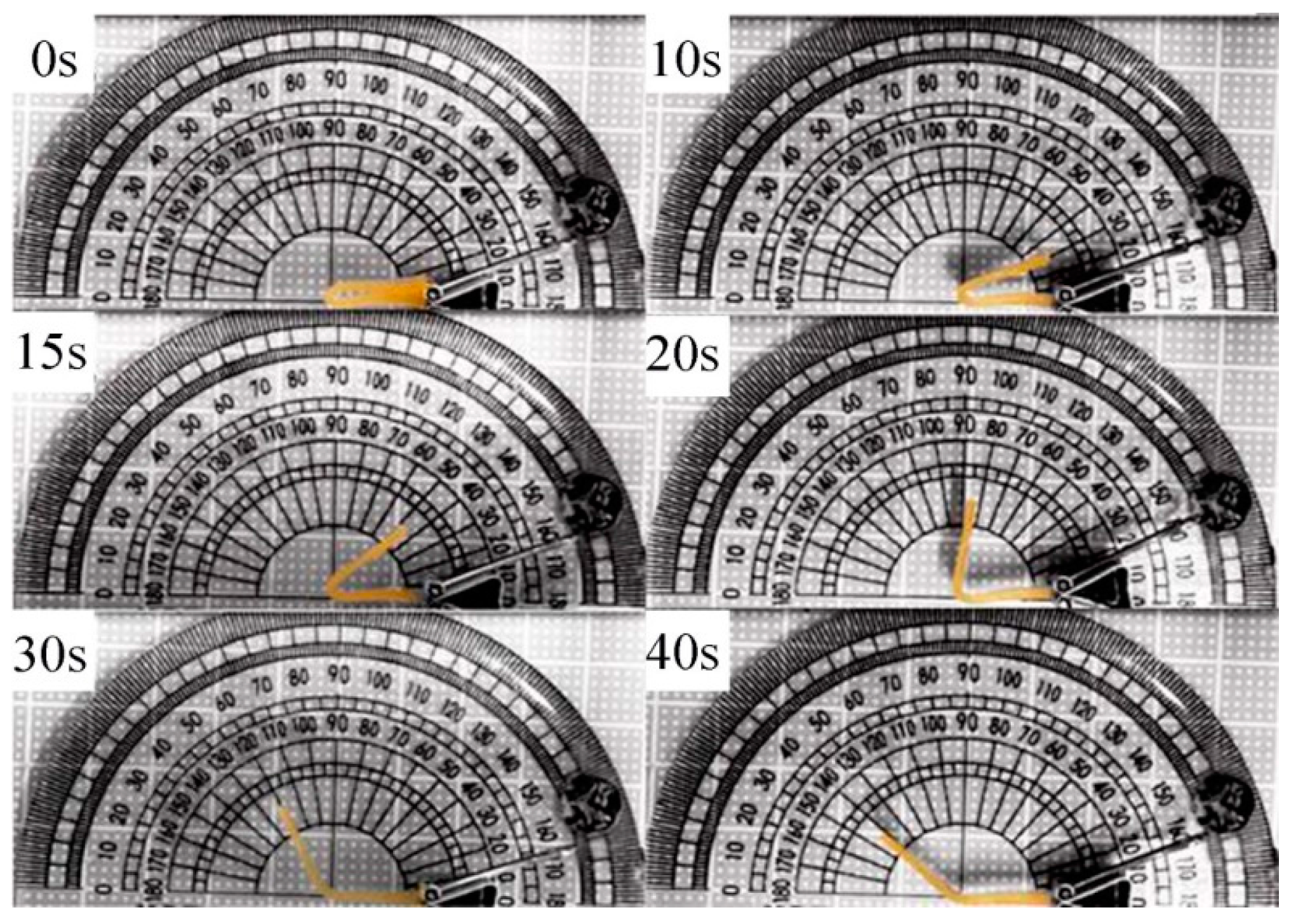
3.9. Shape Memory Property
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Warwar Damouny, C.; Silverstein, M.S. Hydrogel-filled, semi-crystalline, nanoparticle-crosslinked, porous polymers from emulsion templating: Structure, properties, and shape memory. Polymer 2016, 82, 262–273. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, J.; Su, X.; Han, M. Shape memory polybenzoxazines based on polyetheramine. React. Funct. Polym. 2016, 102, 62–69. [Google Scholar] [CrossRef]
- Kim, J.T.; Kim, B.K.; Kim, E.Y.; Park, H.C.; Jeong, H.M. Synthesis and shape memory performance of polyurethane/graphene nanocomposites. React. Funct. Polym. 2014, 74, 16–21. [Google Scholar] [CrossRef]
- Arnebold, A.; Hartwig, A. Fast switchable, epoxy based shape-memory polymers with high strength and toughness. Polymer 2016, 83, 40–49. [Google Scholar] [CrossRef]
- Chevigny, C.; Foucat, L.; Rolland-Sabaté, A.; Buléon, A.; Lourdin, D. Shape-memory effect in amorphous potato starch: The influence of local orders and paracrystallinity. Carbohydr. Polym. 2016, 146, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Qi, H.J.; Xie, T. Recent progress in shape memory polymer: New behavior, enabling materials, and mechanistic understanding. Prog. Polym. Sci. 2015, 49–50, 79–120. [Google Scholar] [CrossRef]
- Gao, W.; Xing, L.; Zhang, Q.; Chen, K.; Liu, P.; Chen, L.; Yang, N.; Zhang, X.; Wang, K.; Wei, Y. Nanocomposite hydrogel incorporated with polymerizable liquid crystal surfactant: Shape transition from layered to honeycomb pore structure and thermo/swelling behavior. React. Funct. Polym. 2016, 102, 53–61. [Google Scholar] [CrossRef]
- Burke, K.A.; Rousseau, I.A.; Mather, P.T. Reversible actuation in main-chain liquid crystalline elastomers with varying crosslink densities. Polymer 2014, 55, 5897–5907. [Google Scholar] [CrossRef]
- Hiraoka, K.; Tagawa, N.; Baba, K. Shape-memory effect controlled by the crosslinking topology in uniaxially-deformed smectic C* elastomers. Macromol. Chem. Phys. 2008, 209, 298–307. [Google Scholar] [CrossRef]
- Shayan, S.; Joselito, M.R.; Peter, C.I.; Gordon, G.W. A facile approach to spinning multifunctional conductive elastomer fibres with nanocarbon fillers. Smart Mater. Struct. 2016, 25, 035015. [Google Scholar]
- Khan, U.; Young, K.; O’Neill, A.; Coleman, J.N. High strength composite fibres from polyester filled with nanotubes and graphene. J. Mater. Chem. 2012, 22, 12907–12914. [Google Scholar] [CrossRef]
- Kang, S.-H.; Fang, T.-H.; Hong, Z.-H. Electrical and mechanical properties of graphene oxide on flexible substrate. J. Phys. Chem. Solids 2013, 74, 1783–1793. [Google Scholar] [CrossRef]
- Seyedin, M.Z.; Razal, J.M.; Innis, P.C.; Jalili, R.; Wallace, G.G. Achieving outstanding mechanical performance in reinforced elastomeric composite fibers using large sheets of graphene oxide. Adv. Funct. Mater. 2015, 25, 94–104. [Google Scholar] [CrossRef]
- Seyedin, S.; Razal, J.M.; Innis, P.C.; Jalili, R.; Wallace, G.G. Compositional effects of large graphene oxide sheets on the spinnability and properties of polyurethane composite fibers. Adv. Mater. Interfaces 2016, 3. [Google Scholar] [CrossRef]
- Pant, H.R.; Pokharel, P.; Joshi, M.K.; Adhikari, S.; Kim, H.J.; Park, C.H.; Kim, C.S. Processing and characterization of electrospun graphene oxide/polyurethane composite nanofibers for stent coating. Chem. Eng. J. 2015, 270, 336–342. [Google Scholar] [CrossRef]
- González, Z.; Botas, C.; Álvarez, P.; Roldán, S.; Blanco, C.; Santamaría, R.; Granda, M.; Menéndez, R. Thermally reduced graphite oxide as positive electrode in Vanadium Redox Flow Batteries. Carbon 2012, 50, 828–834. [Google Scholar] [CrossRef]
- Kim, J.; Jeon, J.H.; Kim, H.J.; Lim, H. Durable and water-floatable ionic polymer actuator with hydrophobic and asymmetrically laser-scribed reduced graphene oxide paper electrodes. ACS Nano 2014, 8, 2986–2997. [Google Scholar] [CrossRef] [PubMed]
- Barrioni, B.R.; de Carvalho, S.M.; Oréfice, R.L.; de Oliveira, A.A.R.; Pereira, M.D.M. Synthesis and characterization of biodegradable polyurethane films based on HDI with hydrolyzable crosslinked bonds and a homogeneous structure for biomedical applications. Mater. Sci. Eng. C 2015, 52, 22–30. [Google Scholar] [CrossRef] [PubMed]
- Cakić, S.M.; Ristić, I.S.; Marinović-Cincović, M.; Špírková, M. The effects of the structure and molecular weight of the macrodiol on the properties polyurethane anionic adhesives. Int. J. Adhes. Adhes. 2015, 41, 132–139. [Google Scholar] [CrossRef]
- Li, X.; Deng, H.; Li, Z.; Xiu, H.; Qi, X.; Zhang, Q.; Wang, K.; Chen, F.; Fu, Q. Graphene/thermoplastic polyurethane nanocomposites: Surface modification of graphene through oxidation, polyvinyl pyrrolidone coating and reduction. Compos. Part A 2015, 68, 264–275. [Google Scholar] [CrossRef]
- Han, W.; Tu, M.; Zeng, R.; Zhao, J.; Zhou, C. Preparation, characterization and cytocompatibility of polyurethane/cellulose based liquid crystal composite membranes. Carbohydr. Polym. 2012, 90, 1353–1361. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Shu, X.; Xie, H.L.; Zhang, H.L. Synthesis and liquid crystalline behavior of 2,5-disubstituted styrene-based random copolymers: Effect of difference in length of the rigid core on the mesomorphic behavior of mesogen-jacketed liquid crystalline polymers. Polymer 2013, 54, 3556–3565. [Google Scholar] [CrossRef]
- Pokharel, P.; Lee, D.S. High performance polyurethane nanocomposite films prepared from a masterbatch of graphene oxide in polyether polyol. Chem. Eng. J. 2014, 253, 356–365. [Google Scholar] [CrossRef]
- Potts, J.R.; Dreyer, D.R.; Bielawski, C.W.; Ruoff, R.S. Graphene-based polymer nanocomposites. Polymer 2011, 52, 5–25. [Google Scholar] [CrossRef]
- Quaresimin, M.; Schulte, K.; Zappalorto, M.; Chandrasekaran, S. Toughening mechanisms in polymer nanocomposites: From experiments to modelling. Compos. Sci. Technol. 2016, 123, 187–204. [Google Scholar] [CrossRef]
- Nadiv, R.; Shtein, M.; Buzaglo, M.; Peretz-Damari, S.; Kovalchuk, A.; Wang, T.; Tour, J.M.; Regev, O. Graphene nanoribbon–polymer composites: The critical role of edge functionalization. Carbon 2016, 99, 444–450. [Google Scholar] [CrossRef]
- Bian, J.; Lin, H.L.; He, F.X.; Wei, X.W.; Chang, I.T.; Sancaktar, E. Fabrication of microwave exfoliated graphite oxide reinforced thermoplastic polyurethane nanocomposites: Effects of filler on morphology, mechanical, thermal and conductive properties. Compos. Part A 2013, 47, 72–82. [Google Scholar] [CrossRef]
- Ramezanzadeh, B.; Ghasemi, E.; Mahdavian, M.; Changizi, E.; Mohamadzadeh Moghadam, M.H. Characterization of covalently-grafted polyisocyanate chains onto graphene oxide for polyurethane composites with improved mechanical properties. Chem. Eng. J. 2015, 281, 869–883. [Google Scholar] [CrossRef]
- Kalaitzidou, K.; Fukushima, H.; Askeland, P.; Drzal, L.T. The nucleating effect of exfoliated graphite nanoplatelets and their influence on the crystal structure and electrical conductivity of polypropylene composites. J. Mater. Sci. 2008, 43, 2895–2907. [Google Scholar] [CrossRef]
- Jing, Q.; Liu, W.; Pan, Y.; Silberschmidt, V.V.; Li, L.; Dong, Z. Chemical functionalization of graphene oxide for improving mechanical and thermal properties of polyurethane composites. Mater. Des. 2015, 85, 808–814. [Google Scholar] [CrossRef]
- Gu, S.; Yan, B.; Liu, L.; Ren, J. Carbon nanotube–polyurethane shape memory nanocomposites with low trigger temperature. Eur. Polym. J. 2013, 49, 3867–3877. [Google Scholar] [CrossRef]
- Raja, M.; Ryu, S.H.; Shanmugharaj, A.M. Influence of surface modified multiwalled carbon nanotubes on the mechanical and electroactive shape memory properties of polyurethane (PU)/poly(vinylidene diflouride) (PVDF) composites. Colloids Surf. A 2014, 450, 59–66. [Google Scholar] [CrossRef]
- Lian, H.; Li, S.; Liu, K.; Xu, L.; Wang, K.; Guo, W. Study on modified graphene /butyl rubber nanocomposites—Part I: Preparation and Characterization. Polym. Eng. Sci. 2011, 51, 2254–2260. [Google Scholar] [CrossRef]
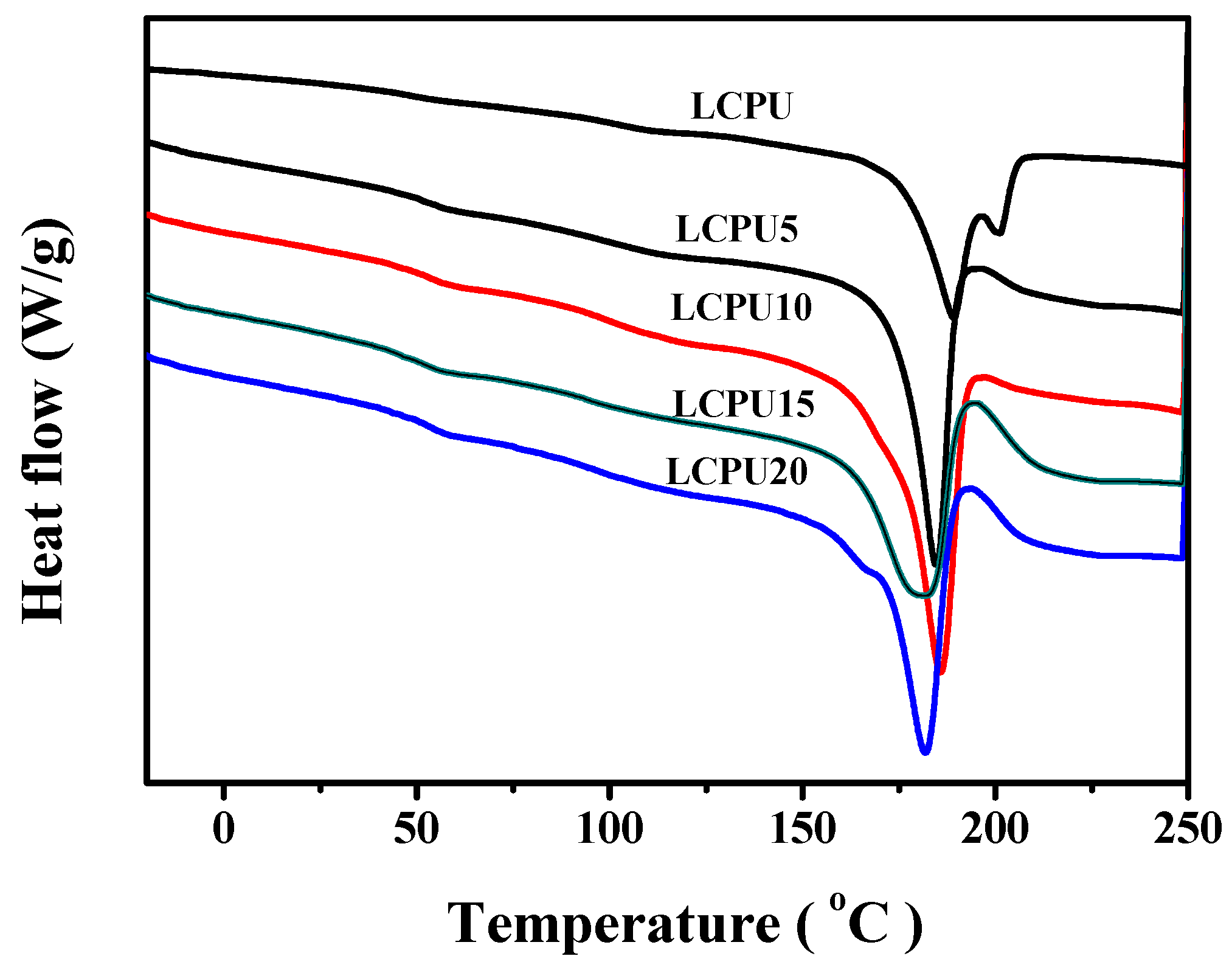


| Sample | Tg (°C) | Tm (°C) | Ti (°C) |
|---|---|---|---|
| LCPU | 40.8 ± 0.6 | 189.3 ± 0.2 | 200.6 ± 0.1 |
| LCPU5 | 41.9 ± 0.7 | 188.0 ± 0.2 | - |
| LCPU10 | 42.7 ± 0.5 | 185.7 ± 0.3 | - |
| LCPU15 | 42.1 ± 0.6 | 181.9 ± 0.2 | - |
| LCPU20 | 42.8 ± 0.5 | 181.8 ± 0.3 | - |
| Cycles | LCPU | LCPU5 | LCPU10 | LCPU15 | LCPU20 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Rf | Rr | Rf | Rr | Rf | Rr | Rf | Rr | Rf | Rr | |
| 1 | 98 | 99 | 96 | 100 | 95 | 99 | 92 | 99 | 92 | 100 |
| 2 | 98 | 99 | 95 | 99 | 95 | 98 | 93 | 98 | 93 | 99 |
| 3 | 97 | 98 | 95 | 96 | 95 | 97 | 93 | 98 | 93 | 99 |
| 4 | 98 | 97 | 96 | 96 | 94 | 97 | 93 | 98 | 93 | 98 |
| Cycles | LCPU (°/s) | LCPU5 (°/s) | LCPU10 (°/s) | LCPU15 (°/s) | LCPU20 (°/s) |
|---|---|---|---|---|---|
| 1 | 2.6 | 5.6 | 6.3 | 8.6 | 9.2 |
| 2 | 2.6 | 4.5 | 5.9 | 8.1 | 8.9 |
| 3 | 2.4 | 4.9 | 5.5 | 6.8 | 7.8 |
| 4 | 2.3 | 4.6 | 5.3 | 6.0 | 7.1 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Lian, H.; Hu, Y.; Chang, W.; Cui, X.; Liu, Y. Enhancement in Mechanical and Shape Memory Properties for Liquid Crystalline Polyurethane Strengthened by Graphene Oxide. Polymers 2016, 8, 236. https://doi.org/10.3390/polym8070236
Li Y, Lian H, Hu Y, Chang W, Cui X, Liu Y. Enhancement in Mechanical and Shape Memory Properties for Liquid Crystalline Polyurethane Strengthened by Graphene Oxide. Polymers. 2016; 8(7):236. https://doi.org/10.3390/polym8070236
Chicago/Turabian StyleLi, Yueting, Huiqin Lian, Yanou Hu, Wei Chang, Xiuguo Cui, and Yang Liu. 2016. "Enhancement in Mechanical and Shape Memory Properties for Liquid Crystalline Polyurethane Strengthened by Graphene Oxide" Polymers 8, no. 7: 236. https://doi.org/10.3390/polym8070236
APA StyleLi, Y., Lian, H., Hu, Y., Chang, W., Cui, X., & Liu, Y. (2016). Enhancement in Mechanical and Shape Memory Properties for Liquid Crystalline Polyurethane Strengthened by Graphene Oxide. Polymers, 8(7), 236. https://doi.org/10.3390/polym8070236







