Sustainable Phenolic Fractions as Basis for Furfuryl Alcohol-Based Co-Polymers and Their Use as Wood Adhesives
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Formulation Preparation
2.3. Determination of Shelf- and Pot-Life
2.4. Gel/Curing Time
2.5. FT-IR Analysis
2.6. Water Solubility Test/Leaching Resistance
2.7. Solid Wood Gluing and Shearing Tests
2.8. Particleboard Production
2.9. Density and Density Profile
2.10. Mechanical Properties
3. Results
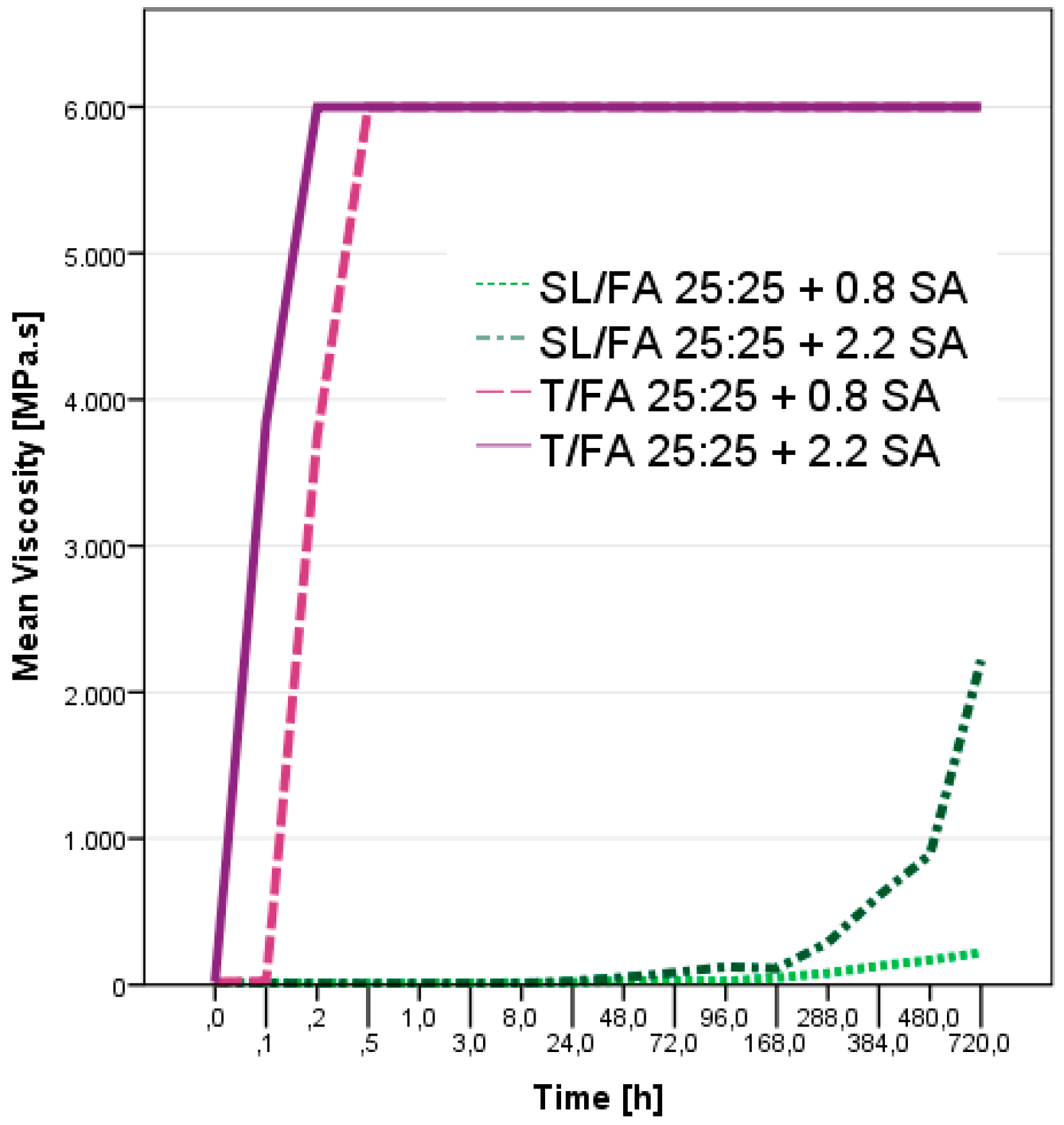
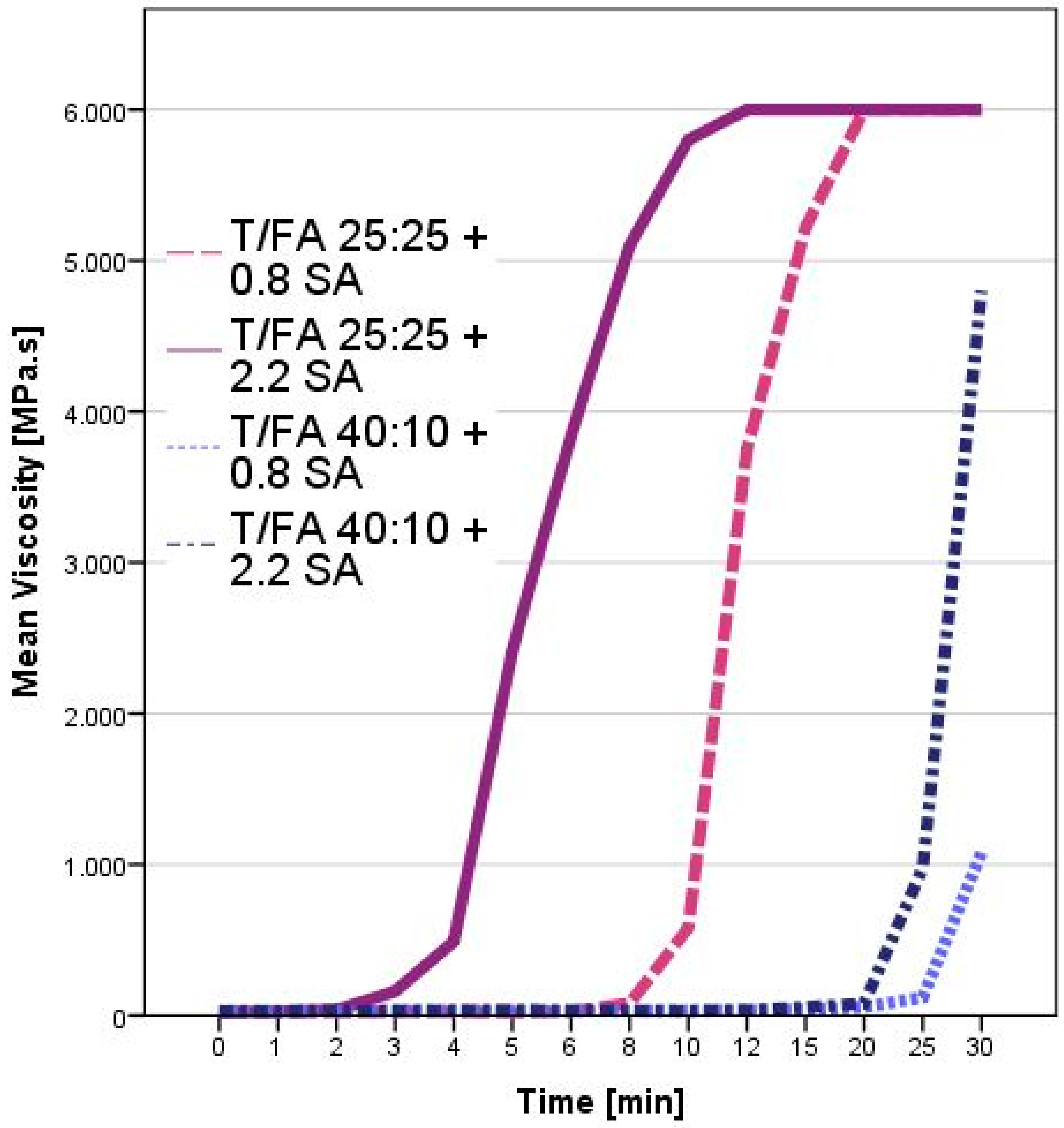
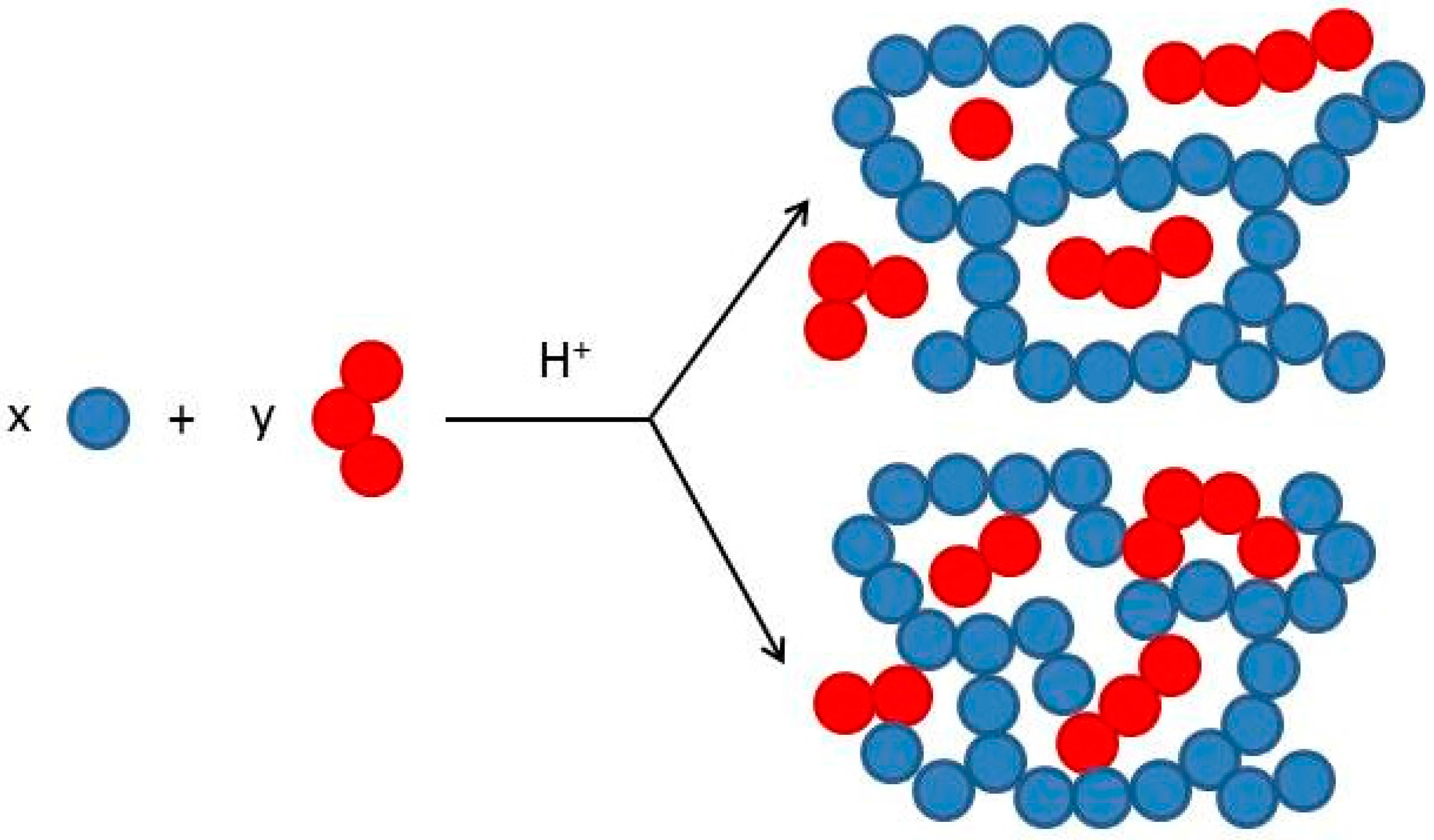
3.1. Curing Process Characterization
3.2. Adhesive Properties
3.2.1. Solid Wood Gluing: Shearing Tests
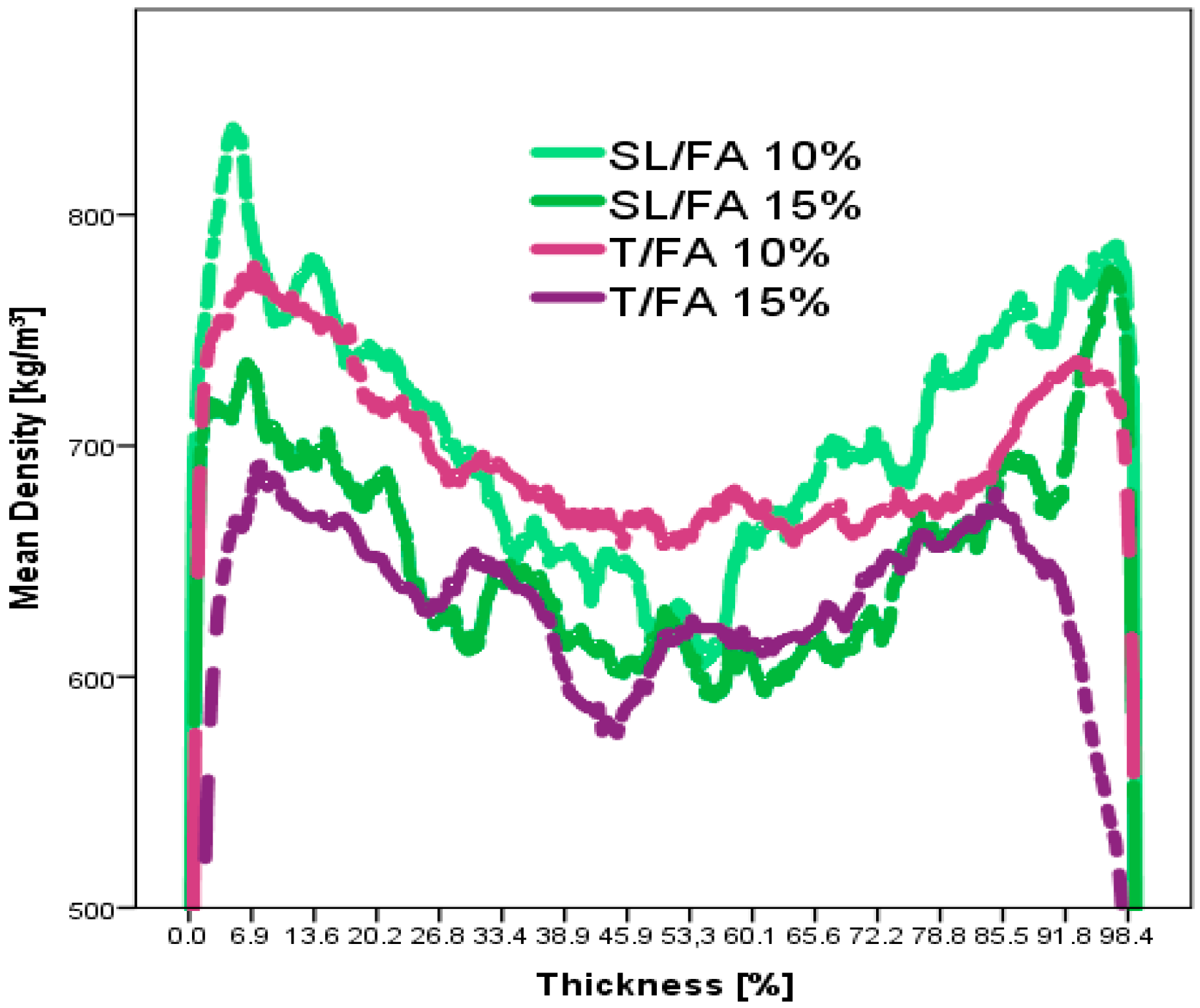
3.2.2. Particleboard Production
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gandini, A. Polymers from renewable resources: A challenge for the future of macromolecular materials. Macromolecules 2008, 41, 9491–9504. [Google Scholar] [CrossRef]
- Gandini, A. Furans as offspring of sugars and polysaccharides and progenitors of a family of remarkable polymers: A review of recent progress. Polym. Chem. 2010, 1, 245–251. [Google Scholar] [CrossRef]
- Deka, H.; Misra, M.; Mohanty, A. Renewable resource based “all green composites” from kenaf biofiber and poly (furfuryl alcohol) bioresin. Ind. Crop. Prod. 2013, 41, 94–101. [Google Scholar] [CrossRef]
- Gutiérrez, T.; Buszko, M.L.; Ingram, L.O.; Preston, J.F. Reduction of furfural to furfuryl alcohol by ethanologenic strains of bacteria and its effect on ethanol production from xylose. In Biotechnology for Fuels and Chemicals; Humana Press: Totowa, NJ, USA, 2002. [Google Scholar]
- Nagaraja, B.M.; Kumar, V.S.; Shasikala, V.; Padmasri, A.H.; Sreedhar, B.; Raju, B.D.; Rao, K.R. A highly efficient Cu/MgO catalyst for vapour phase hydrogenation of furfural to furfuryl alcohol. Catal. Commun. 2003, 4, 287–293. [Google Scholar] [CrossRef]
- Merlo, A.B.; Vetere, V.; Ruggera, J.F.; Casella, M.L. Bimetallic PtSn catalyst for the selective hydrogenation of furfural to furfuryl alcohol in liquid-phase. Catal. Commun. 2009, 10, 1665–1669. [Google Scholar] [CrossRef]
- Taherzadeh, M.J.; Gustafsson, L.; Niklasson, C.; Lidén, G. Conversion of furfural in aerobic and anaerobic batch fermentation of glucose by Saccharomyces cerevisiae. J. Biosci. Bioeng. 1999, 87, 169–174. [Google Scholar] [CrossRef]
- Vazquez, M.; Oliva, M.; Tellez-Luis, S.J.; Ramírez, J.A. Hydrolysis of sorghum straw using phosphoric acid: Evaluation of furfural production. Bioresour. Technol. 2007, 98, 3053–3060. [Google Scholar] [CrossRef] [PubMed]
- Weingarten, R.; Cho, J.; Conner, W.C., Jr.; Huber, G.W. Kinetics of furfural production by dehydration of xylose in a biphasic reactor with microwave heating. Green Chem. 2010, 12, 1423–1429. [Google Scholar] [CrossRef]
- De Jong, W.; Marcotullio, G. Overview of biorefineries based on co-production of furfural, existing concepts and novel developments. Int. J. Chem. React. Eng. 2010, 8. [Google Scholar] [CrossRef]
- Win, D.T. Furfural-gold from garbage. AU J. Technol. 2005, 8, 185–190. [Google Scholar]
- Choura, M.; Belgacem, N.M.; Gandini, A. Acid-catalyzed polycondensation of furfuryl alcohol: Mechanisms of chromophore formation and cross-linking. Macromolecules 1996, 29, 3839–3850. [Google Scholar] [CrossRef]
- Principe, M.; Ortiz, P.; Martínez, R. An NMR study of poly(furfuryl alcohol) prepared with p-toluenesulphonic acid. Polym. Int. 1999, 48, 637–641. [Google Scholar] [CrossRef]
- Guigo, N.; Mija, A.; Vincent, L.; Sbirrazzuoli, N. Chemorheological analysis and model-free kinetics of acid catalysed furfuryl alcohol polymerization. Phys. Chem. 2007, 9, 5359–5366. [Google Scholar] [CrossRef] [PubMed]
- Guigo, N.; Mija, A.; Vincent, L.; Sbirrazzuoli, N. Eco-friendly composite resins based on renewable biomass resources: Polyfurfuryl alcohol/lignin thermosets. Eur. Polym. J. 2010, 46, 1016–1023. [Google Scholar] [CrossRef]
- Tondi, G.; Link, M.; Oo, C.W.; Petutschnigg, A. A simple approach to distinguish classic and formaldehyde-free tannin based rigid foams by ATR FT-IR. J. Spectrosc. 2015, 2015, 902340. [Google Scholar] [CrossRef]
- Sricharoenchaikul, V.; Hicks, A.L.; Frederick, W.J. Carbon and char residue yields from rapid pyrolysis of kraft black liquor. Bioresour. Technol. 2001, 77, 131–138. [Google Scholar] [CrossRef]
- Zhao, L.; Tran, H.; Maki, K. Combustion behaviors of lignin-lean black liquor and lignin. Tappi J. 2015, 14, 451–458. [Google Scholar]
- Bacelo, H.A.; Santos, S.C.; Botelho, C.M. Tannin-based biosorbents for environmental applications—A review. Chem. Eng. J. 2016, 303, 575–587. [Google Scholar] [CrossRef]
- Barrado-Moreno, M.M.; Beltrán-Heredia, J.; Martín-Gallardo, J. Removal of Oocystis algae from freshwater by means of tannin-based coagulant. J. Appl. Phycol. 2016, 28, 1589–1595. [Google Scholar] [CrossRef]
- Hoareau, W.; Trindade, W.G.; Siegmund, B.; Castellan, A.; Frollini, E. Sugar cane bagasse and curaua lignins oxidized by chlorine dioxide and reacted with furfuryl alcohol: Characterization and stability. Polym. Degrad. Stabl. 2004, 86, 567–576. [Google Scholar] [CrossRef]
- Trindade, W.G.; Hoareau, W.; Megiatto, J.D.; Razera, I.A.T.; Castellan, A.; Frollini, E. Thermoset phenolic matrices reinforced with unmodified and surface-grafted furfuryl alcohol sugar cane bagasse and curaua fibers: Properties of fibers and composites. Biomacromolecules 2005, 6, 2485–2496. [Google Scholar] [CrossRef] [PubMed]
- Schneider, M.H.; Phillips, J.G. Furfuryl Alcohol and Lignin Adhesive Composition. U.S. Patent US6747076 B2, 8 June 2004. [Google Scholar]
- Trosa, A.; Pizzi, A. Industrial hardboard and other panels binder from waste lignocellulosic liquors/phenol-formaldehyde resins. Holz Roh Werkst. 1998, 56, 229–233. [Google Scholar] [CrossRef]
- Pin, J.M.; Guigo, N.; Mija, A.; Vincent, L.; Sbirrazzuoli, N.; van der Waal, J.C.; De Jong, E. Valorization of biorefinery side-stream products: Combination of humins with polyfurfuryl alcohol for composite elaboration. ACS Sustain. Chem. Eng. 2014, 2, 2182–2190. [Google Scholar] [CrossRef]
- Kraiwattanawong, K.; Mukai, S.R.; Tamon, H.; Lothongkum, A.W. Preparation of carbon cryogels from wattle tannin and furfural. Micropor. Mesopor. Mat. 2007, 98, 258–266. [Google Scholar] [CrossRef]
- Pizzi, A.; Orovan, E.; Cameron, F.A. The development of weather-and boil-proof phenol-resorcinol-furfural cold-setting adhesives. Holz Roh Werkst. 1984, 42, 467–472. [Google Scholar] [CrossRef]
- Pizzi, A.; Tondi, G.; Pasch, H.; Celzard, A. Matrix-assisted laser desorption/ionization time-of-flight structure determination of complex thermoset networks: Polyflavonoid tannin-furanic rigid foams. J. Appl. Polym. Sci. 2008, 110, 1451–1456. [Google Scholar] [CrossRef]
- Link, M.; Kolbitsch, C.; Tondi, G.; Ebner, M.; Wieland, S.; Petutschnigg, A. Formaldehyde-free tannin based foams and their use as lightweight panels. Bioresour. Resour. 2011, 6, 4218–4228. [Google Scholar]
- Tondi, G.; Link, M.; Kolbitsch, C.; Gavino, J.; Luckeneder, P.; Petutschnigg, A.; Herchl, R.; Van Doorslaer, C. Lignin-based foams: Production process and characterization. Bioresour. Resour. 2016, 11, 2972–2986. [Google Scholar] [CrossRef]
- Basso, M.C.; Pizzi, A.; Lacoste, C.; Delmotte, L.; Al-Marzouki, F.M.; Abdalla, S.; Celzard, A. MALDI-TOF and 13C NMR analysis of tannin–furanic–polyurethane foams adapted for industrial continuous lines application. Polymers 2014, 6, 2985–3004. [Google Scholar] [CrossRef]
- Tondi, G.; Zhao, W.; Pizzi, A.; Du, G.; Fierro, V.; Celzard, A. Tannin-based rigid foams: A survey of chemical and physical properties. Bioresour. Technol. 2009, 100, 5162–5169. [Google Scholar] [CrossRef] [PubMed]
- Celzard, A.; Fierro, V.; Amaral-Labat, G.; Pizzi, A. Torero, Flammability assessment of tannin-based cellular materials. J. Polym. Degrad. Stabl. 2011, 96, 477–482. [Google Scholar] [CrossRef]
- Tondi, G.; Link, M.; Kolbitsch, C.; Lesacher, R.; Petutschnigg, A. Pilot plant up-scaling of tannin foams. Ind. Crop. Prod. 2016, 79, 211–218. [Google Scholar] [CrossRef]
- Tondi, G.; Oo, C.W.; Pizzi, A.; Trosa, A.; Thevenon, M.F. Metal adsorption of tannin based rigid foams. Ind. Crop. Prod. 2009, 29, 336–340. [Google Scholar] [CrossRef]
- Sánchez-Martín, J.; Beltrán-Heredia, J.; Gibello-Pérez, P. Adsorbent biopolymers from tannin extracts for water treatment. Chem. Eng. J. 2011, 168, 1241–1247. [Google Scholar] [CrossRef]
- Tondi, G.; Pizzi, A.; Pasch, H.; Celzard, A. Structure degradation, conservation and rearrangement in the carbonisation of polyflavonoid tannin/furanic rigid foams–A MALDI-TOF investigation. Polym. Degrad. Stabl. 2008, 93, 968–975. [Google Scholar] [CrossRef]
- Reyer, A.; Tondi, G.; Berger, R.J.F.; Petutschnigg, A.; Musso, M. Raman spectroscopic investigation of tannin-furanic rigid foams. Vib. Spectrosc. 2016, 84, 58–66. [Google Scholar] [CrossRef]
- Čop, M.; Laborie, M.P.; Pizzi, A.; Sernek, M. Curing characterisation of spruce tannin-based foams using the advanced isoconversional method. Bioresour. Resour. 2014, 9, 4643–4655. [Google Scholar] [CrossRef]
- Foo, L.Y.; Hemingway, R.W. Condensed tannins: Reactions of model compounds with furfuryl alcohol and furfuraldehyde. J. Wood Chem. Technol. 1985, 5, 135–158. [Google Scholar] [CrossRef]
- Pizzi, A.; Mittal, K.L. (Eds.) Wood Adhesives; CRC Press: Leiden, The Netherlands, 2011.
- Thakur, V.K.; Kessler, M.R. Self-healing polymer nanocomposite materials: A review. Polymer 2015, 69, 369–383. [Google Scholar] [CrossRef]
- Dunky, M.; Niemz, P. Teil II Bindemittel Und Verleimung (Part 2: Adhesives and Adhesive Bonding). Holzwerkstoffe Und Leime—Technologie Und Einflussfaktoren (Wood Composites and Glues—Technology and Influencing Facors); Springer: Berlin, Germany, 2002. [Google Scholar]
- Egner, K. Untersuchung der faserschädigung bei der verwendung von säurehärtendem kunstharzleim. (Study of wood fiber damage by acid hardener synthetic resin glues). Holz Zbl 1952, 135, 1857–1858. [Google Scholar]
- Myers, G.E. Use of acid scavengers to improve durability of acid-catalyzed adhesive wood bonds. For. Prod. J. 1983, 33, 49–57. [Google Scholar]
- Pizzi, A.; Vosloo, R.; Cameron, F.A.; Orovan, E. Self-neutralizing acid-set PF wood adhesives. Holz Roh Werkst. 1986, 44, 229–234. [Google Scholar] [CrossRef]
- Adhesives for Load-Bearing Timber Structures–Test Methods–Part 1: Determination of Bond Strength in Longitudinal Tensile Shear Strength; British Standard: London, UK, 2004; DIN EN 302-1.
- Tondi, G.; Wieland, S.; Wimmer, T.; Schnabel, T.; Petutschnigg, A. Starch-sugar synergy in wood adhesion science: Basic studies and particleboard production. Eur. J. Wood Prod. 2012, 70, 271–278. [Google Scholar] [CrossRef]
- Wood-Based Panels; Determination of Modulus of Elasticity in Bending and of Bending Strength; British Standard: London, UK, 1993; DIN EN 310.
- Particleboards–Specifications; British Standard: London, UK, 2003; DIN EN 312.
- Tondi, G.; Petutschnigg, A. Middle infrared (ATR FT-MIR) characterization of industrial tannin extracts. Ind. Crop. Prod. 2015, 65, 422–428. [Google Scholar] [CrossRef]
- Schwanninger, M.; Rodrigues, J.C.; Pereira, H.; Hinterstoisser, B. Effects of short-time vibratory ball milling on the shape of FT-IR spectra of wood and cellulose. Vib. Spectrosc. 2004, 36, 23–40. [Google Scholar] [CrossRef]
- Nordstierna, L.; Lande, S.; Westin, M.; Karlsson, O.; Furo, I. Towards novel wood-based materials: Chemical bonds between lignin-like model molecules and poly (furfuryl alcohol) studied by NMR. Holzforschung 2008, 62, 709–713. [Google Scholar] [CrossRef]
- Lei, H.; Pizzi, A.; Du, G. Environmentally friendly mixed tannin/lignin wood resins. J. Appl. Polym. Sci. 2008, 107, 203–209. [Google Scholar] [CrossRef]
- Abdullah, U.H.B.; Pizzi, A. Tannin-furfuryl alcohol wood panel adhesives without formaldehyde. Eur. J. Wood Prod. 2013, 71, 131–132. [Google Scholar] [CrossRef]





| Spent liquor (SL) % | Furfuryl alcohol (FA) % | Sulfuric acid (SA) % | Water % |
| 45 | 5 | 0–0.8–1.5–2.2–2.8 | 47.2–47.8–48.5–49.2–50 |
| 40 | 10 | 0–0.8–1.5–2.2–2.8 | 47.2–47.8–48.5–49.2–50 |
| 35 | 15 | 0–0.8–1.5–2.2–2.8 | 47.2–47.8–48.5–49.2–50 |
| 30 | 20 | 0–0.8–1.5–2.2–2.8 | 47.2–47.8–48.5–49.2–50 |
| 25 | 25 | 0–0.8–1.5–2.2–2.8 | 47.2–47.8–48.5–49.2–50 |
| Tannin (T) % | Furfuryl alcohol (FA) % | Sulfuric acid (SA) % | Water % |
| 45 | 5 | 0–0.8–1.5–2.2 | 47.8–48.5–49.2–50 |
| 40 | 10 | 0–0.8–1.5–2.2 | 47.8–48.5–49.2–50 |
| 33 | 17 | 0–0.8–1.5–2.2 | 47.8–48.5–49.2–50 |
| 25 | 25 | 0–0.8–1.5–2.2 | 47.8–48.5–49.2–50 |
| Formulation | Amount of H2SO4 | ||
|---|---|---|---|
| 0.8% | 1.5% | 2.2% | |
| SL/FA 45:5 | 860 | 380 | 300 |
| SL/FA 40:10 | 520 | 460 | 290 |
| SL/FA 35:15 | 600 | 360 | 316 |
| SL/FA 30:20 | 122 | 60 | 46 |
| SL/FA 25:25 | 250 | 78 | 74 |
| Powder | Color of the leaching water | Insoluble fraction (%) | Estimated insoluble (%) | % of phenolic polymerized |
|---|---|---|---|---|
| Spent Liquor | Intense Brown | 2.2 (0.21) | (2.2) | - |
| Tannin | Intense Brown | 1.6 (0.35) | (1.6) | - |
| PFA | Transparent | 93.8 (3.93) | (93.8) | - |
| SL/FA 4:1 | Intense Brown | 47.9 (3.17) | 20.5 | 34.2 |
| SL/FA 1:1 | Brown | 76.1 (3.80) | 48.0 | 56.2 |
| T/FA 4:1 | Pale Brown | 88.7 (2.21) | 20.0 | 85.9 |
| T/FA 1:1 | Pale Brown | 92.1 (2.27) | 47.7 | 88.8 |
| Formulation | Temperature (°C) | Shearing strength (N/mm2) | Standard deviation | % of Broken wood |
|---|---|---|---|---|
| SL/FA 35:15 + 2.2 SA | 120 | 0 | 0 | 0 |
| SL/FA 35:15 + 2.2 SA | 150 | 9.4 | 1.37 | 11 |
| SL/FA 35:15 + 2.2 SA | 180 | 12.1 | 1.37 | 75 |
| SL/FA 30:20 + 2.2 SA | 120 | 8.2 | 3.31 | 0 |
| SL/FA 30:20 + 2.2 SA | 150 | 9.9 | 1.36 | 48 |
| SL/FA 30:20 + 2.2 SA | 180 | 14.1 | 1.33 | 97 |
| SL/FA 25:25 + 2.2 SA | 120 | 11.4 | 2.51 | 78 |
| SL/FA 25:25 + 2.2 SA | 150 | 12.6 | 1.59 | 63 |
| SL/FA 25:25 + 2.2 SA | 180 | 15.0 | 1.02 | 96 |
| SL/FA 25:25 + 2.2 SA * | 180 | 0 | 0 | 0 |
| T/FA 40:10 + 2.2 SA | 180 | 11.5 | 0.49 | 100 |
| T/FA 40:10 + 2.2 SA * | 180 | 1.9 | 0.50 | 60 |
| T/FA 25:25 + 0.8 SA | 180 | 0 | 0 | 0 |
| T/FA 25:25 + 2.2 SA | 120 | 0 | 0 | 0 |
| T/FA 25:25 + 2.2 SA | 180 | 12.7 | 1.29 | 100 |
| T/FA 25:25 + 2.2 SA * | 180 | 2.9 | 0.85 | 60 |
| Polyvinylacetate | - | 16.6 | 1.41 | 100 |
| Solid wood | - | 16.6 | 2.68 | 100 |
| Formulation of the glue (Dry) | |||||||
|---|---|---|---|---|---|---|---|
| Spent liquor (%) | Tannin (%) | FOH (%) | H2SO4 (g) | Amount of adhesive % | Density (kg/m³) | Internal bond (MPa) | Bending E-Mod (MPa) |
| 60 | - | 40 | 3.8 | 10 | 744.3 | 0.26 (0.06) | 6.46 (0.40) |
| 50 | 10 | 40 | 3.8 | 10 | 750.8 | 0.32 (0.03) | 7.56 (0.52) |
| 40 | 20 | 40 | 3.8 | 10 | 735.3 | 0.32 (0.06) | 6.47 (1.86) |
| - | 60 | 40 | 3.8 | 10 | 715.8 | 0.15 (0.05) | 9.71 (2.10) |
| 60 | - | 40 | 3.8 | 15 | 677.8 | 0.35 (0.14) | 11.35 (1.35) |
| 50 | 10 | 40 | 3.8 | 15 | 691.6 | 0.55 (0.16) | 10.81 (1.24) |
| 40 | 20 | 40 | 3.8 | 15 | 659.4 | 0.53 (0.16) | 9.49 (1.86) |
| - | 60 | 40 | 3.8 | 15 | 644.7 | 0.07 (0.03) | 6.41 (0.25) |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luckeneder, P.; Gavino, J.; Kuchernig, R.; Petutschnigg, A.; Tondi, G. Sustainable Phenolic Fractions as Basis for Furfuryl Alcohol-Based Co-Polymers and Their Use as Wood Adhesives. Polymers 2016, 8, 396. https://doi.org/10.3390/polym8110396
Luckeneder P, Gavino J, Kuchernig R, Petutschnigg A, Tondi G. Sustainable Phenolic Fractions as Basis for Furfuryl Alcohol-Based Co-Polymers and Their Use as Wood Adhesives. Polymers. 2016; 8(11):396. https://doi.org/10.3390/polym8110396
Chicago/Turabian StyleLuckeneder, Paul, Johannes Gavino, Robert Kuchernig, Alexander Petutschnigg, and Gianluca Tondi. 2016. "Sustainable Phenolic Fractions as Basis for Furfuryl Alcohol-Based Co-Polymers and Their Use as Wood Adhesives" Polymers 8, no. 11: 396. https://doi.org/10.3390/polym8110396
APA StyleLuckeneder, P., Gavino, J., Kuchernig, R., Petutschnigg, A., & Tondi, G. (2016). Sustainable Phenolic Fractions as Basis for Furfuryl Alcohol-Based Co-Polymers and Their Use as Wood Adhesives. Polymers, 8(11), 396. https://doi.org/10.3390/polym8110396