Flame Retardancy of PA6 Using a Guanidine Sulfamate/Melamine Polyphosphate Mixture
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Preparation of Samples
| Materials | PA6 wt% | GAS wt% | MPP wt% |
|---|---|---|---|
| PA6 | 100 | 0 | 0 |
| PA6/MPP 5% | 95 | 0 | 5 |
| PA6/GAS 1%/MPP 4% | 95 | 1 | 4 |
| PA6/GAS 2.5%/MPP 2.5% | 95 | 2.5 | 2.5 |
| PA6/GAS 4%/MPP 1% | 95 | 4 | 1 |
| PA6/GAS 5% | 95 | 5 | 0 |
| Zone | 1 * | 2 | 3 | 4 | 5 ** | 6 | 7 | 8 | 9 | 10 |
|---|---|---|---|---|---|---|---|---|---|---|
| Temperature (°C) | 300 | 280 | 260 | 260 | 240 | 240 | 235 | 230 | 230 | 210 |
2.3. Fire Testing Methods
2.4. Thermogravimetric Analysis
2.5. Analysis of the Decomposition Gases
2.6. Analysis of the Condensed Phase
3. Results and Discussion
3.1. Preliminary Investigation of the MPP/GAS Combination
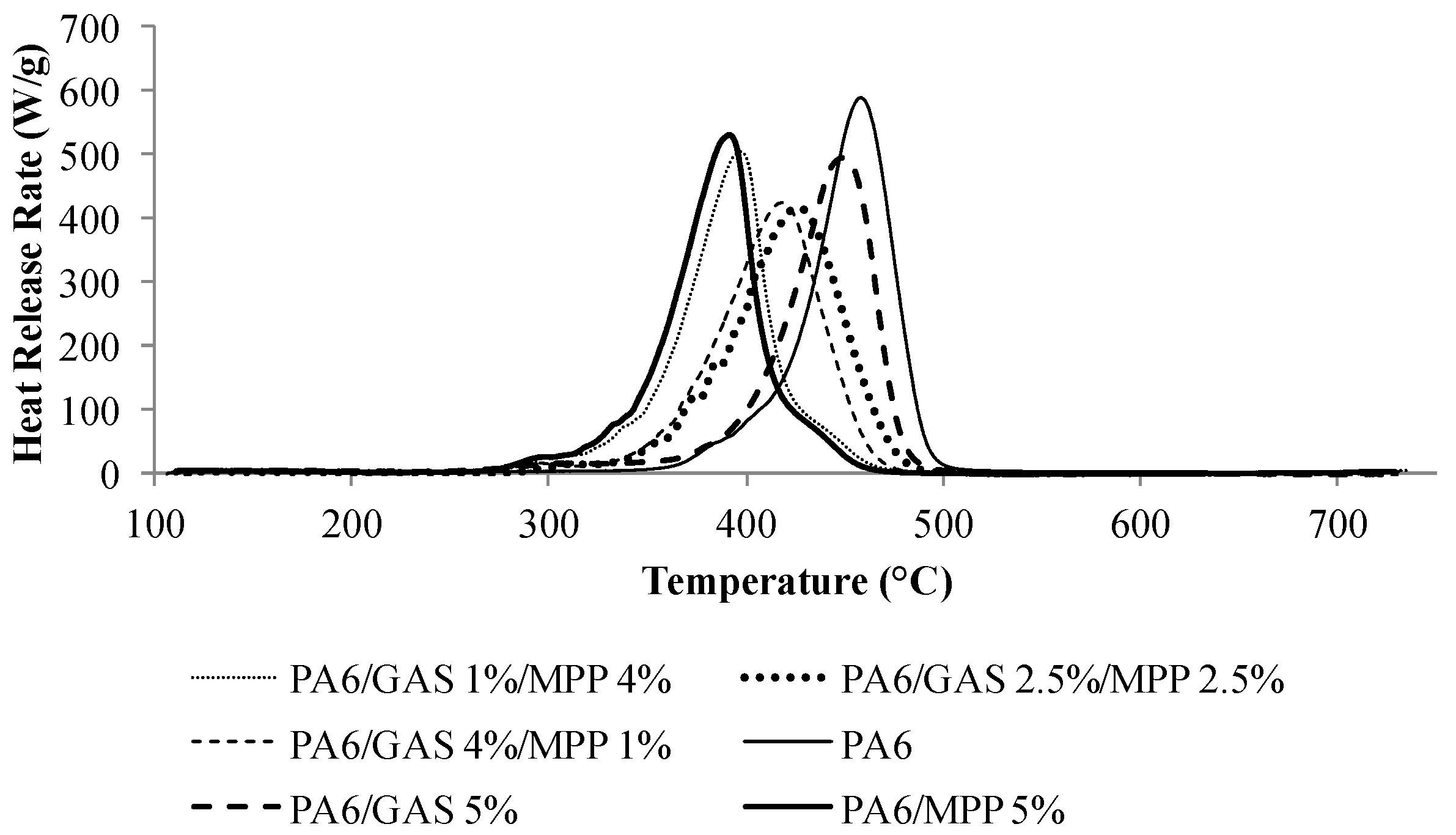
| Formulation | pHRR (W/g) | ΔpHRR/PA6 (%) | TpHRR (°C) | THR (kJ/g) | ΔTHR/PA6 (%) |
|---|---|---|---|---|---|
| PA6 | 588 | – | 456 | 30.0 | – |
| PA6/MPP 5% | 530 | −10 | 391 | 29.7 | −1.0 |
| PA6/GAS 1%/MPP 4% | 498 | −15 | 395 | 28.1 | −6.3 |
| PA6/GAS 2.5%/MPP 2.5% | 411 | −30 | 428 | 28.7 | −4.3 |
| PA6/GAS 4%/MPP 1% | 429 | −27 | 420 | 28.1 | −6.3 |
| PA6/GAS 5% | 500 | −15 | 449 | 27.4 | −8.7 |
3.2. Fire Retardant Properties of the PA6/GAS 2.5%/MPP 2.5%
3.2.1. Cone Calorimetry
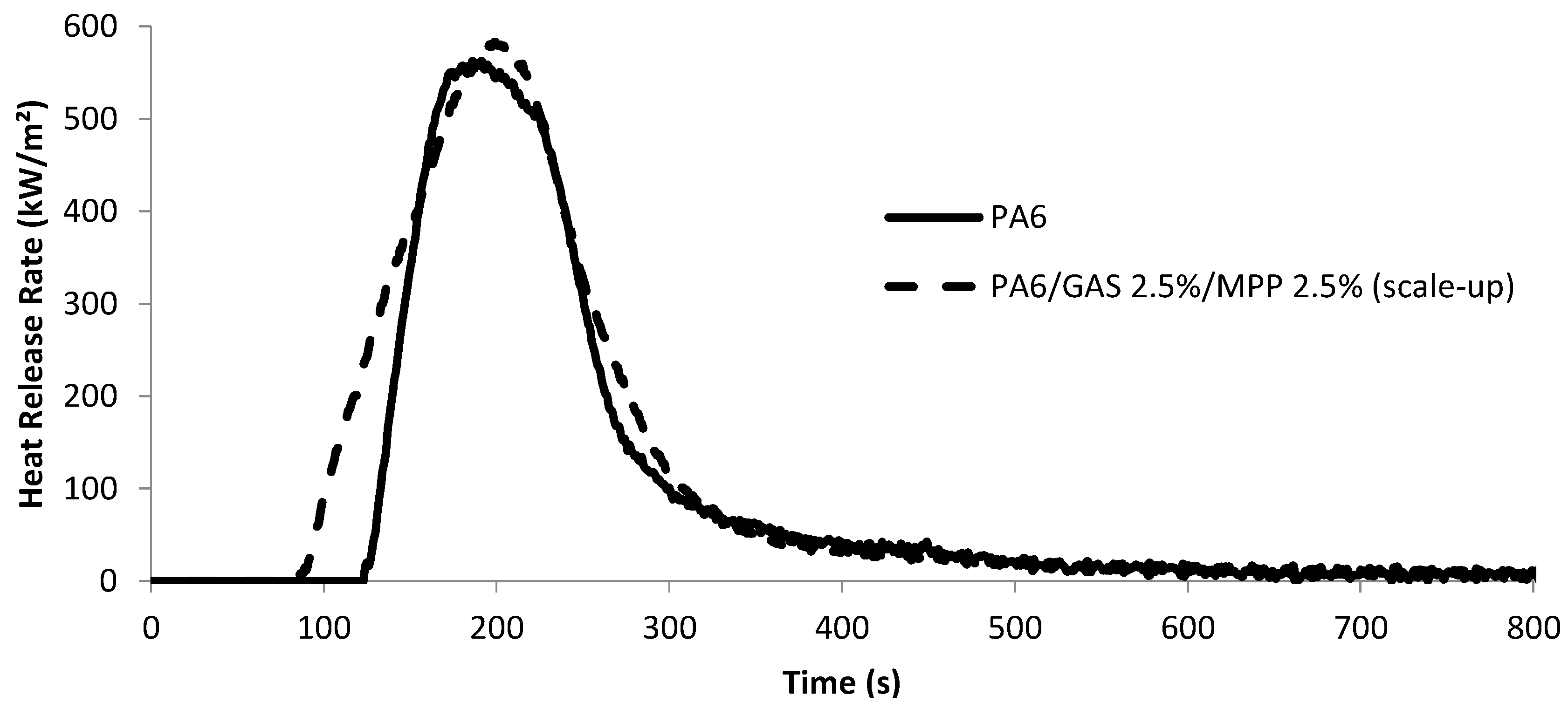
| Property | PA6 | PA6/GAS 2.5%/MPP 2.5% | Δ/PA6 (%) |
|---|---|---|---|
| peak HRR (kW/m2) | 603 ± 13 | 582 ± 20 | −4 |
| time peak HRR (s) | 218 ± 16 | 213 ± 9 | −2 |
| TTI (s) | 124 ± 13 | 87 ± 2 | −30 |
| Total heat release (MJ/m2) | 73 ± 2 | 79 ± 1 | +8 |

3.2.2. UL 94 and LOI

| Formulation | t1/t2 * (s) | Dripping ** | Cotton ignition ** | rating |
|---|---|---|---|---|
| PA6 | 3.6/2.4 | Y | Y | V2 |
| PA6/GAS 2.5%/MPP 2.5% | 1.6/1.9 | Y | N | V0 |
3.3. Mechanism of Degradation of PA6/GAS/MPP
3.3.1. TGA-FTIR
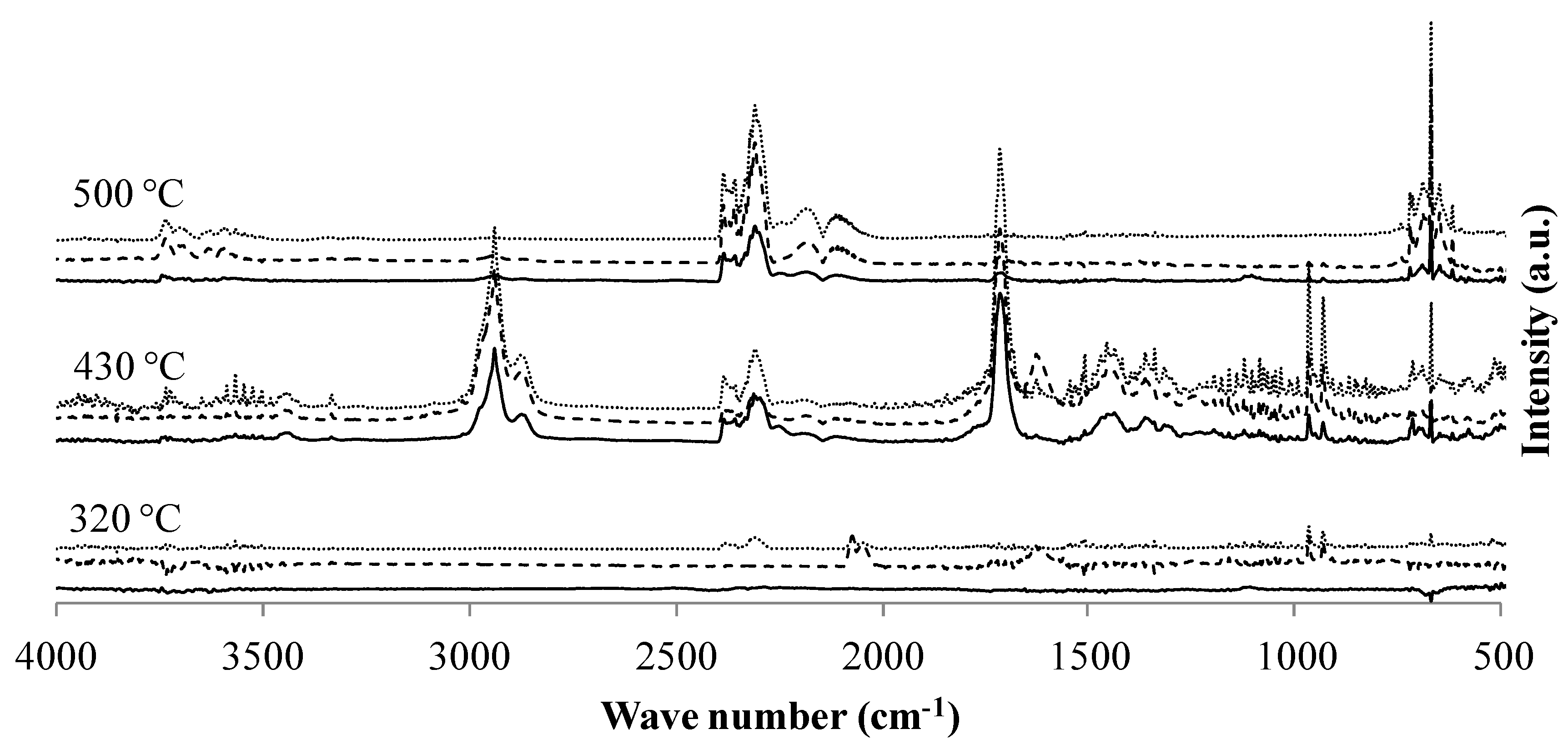
| Functional Group or Component | Wave Number (cm−1) | Vibration Type | Additional Ref. |
|---|---|---|---|
| CO2 | 669 | δ | [22] |
| 2354 | νas | ||
| NH3 | 930 | δ δ ν | [22,26] |
| 965 | |||
| 1626 | |||
| 3332 | |||
| CH2 | 2873 | δ | [22] |
| 2938 | δ | ||
| N−CH2 | 1440 | δ | [22] |
| Amide I (prim. *, sec. **) | 1713 | ν | [22,23] |
| Amide II (sec. **) | 1508 | δ, ν | [22,23] |
| Amide III (prim. *) | 1340 | δ, ν | [22,23] |
| −C≡N | 2250 | ν | [22,25] |
| H2O | 1400–1700 | δ | [22] |
| 3400–3700 | νas |
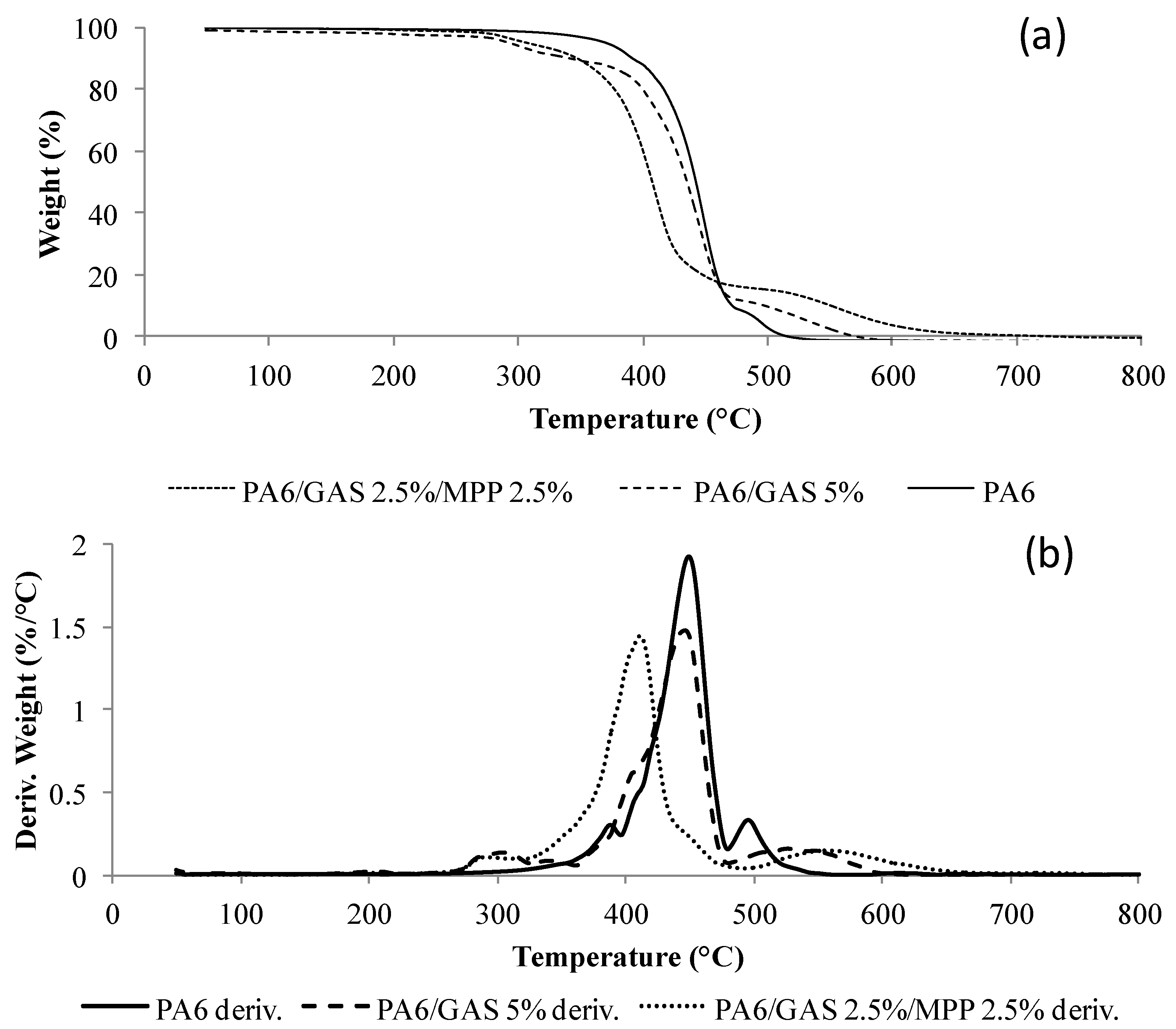
3.4. Analysis of the Solid Phase
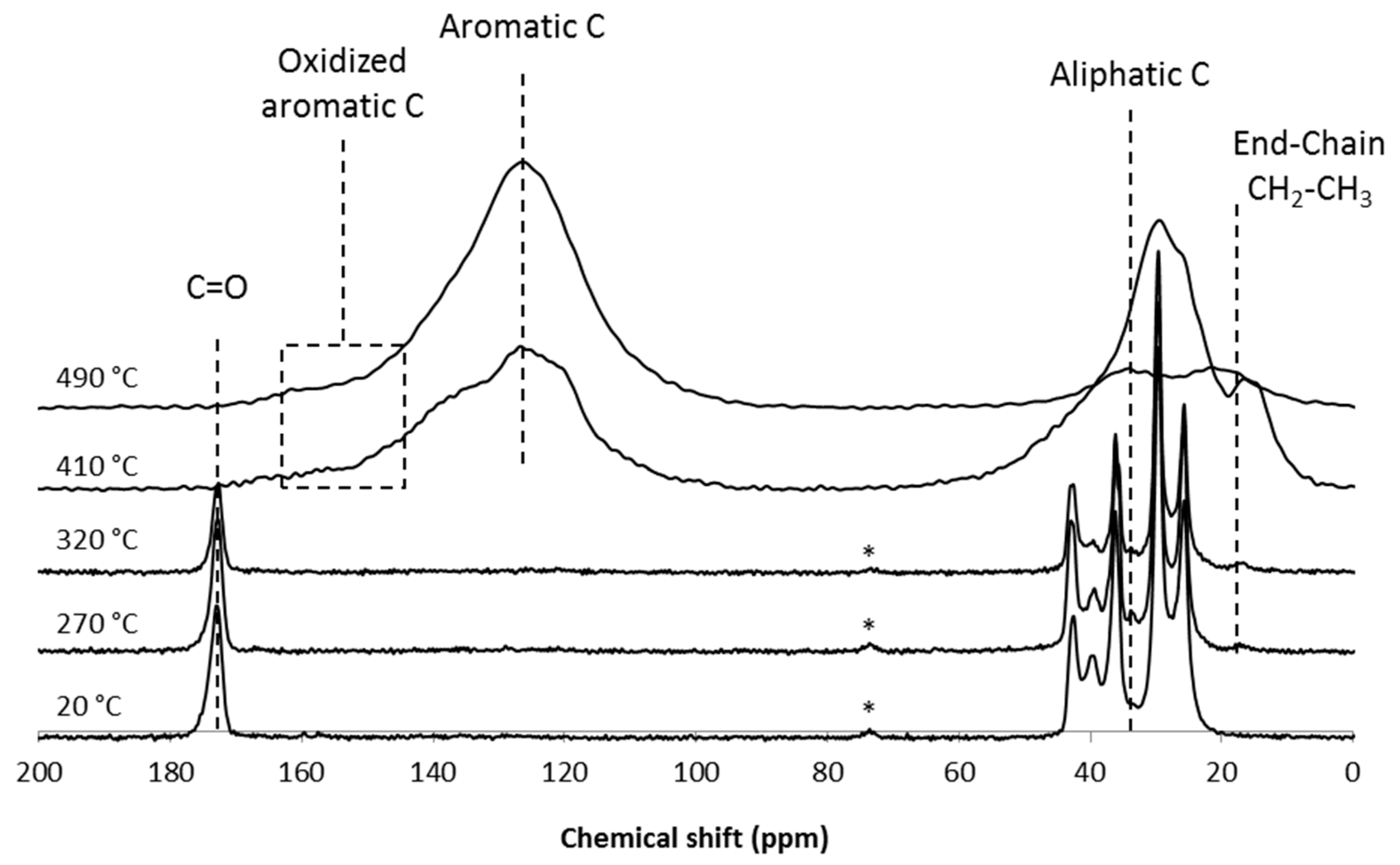
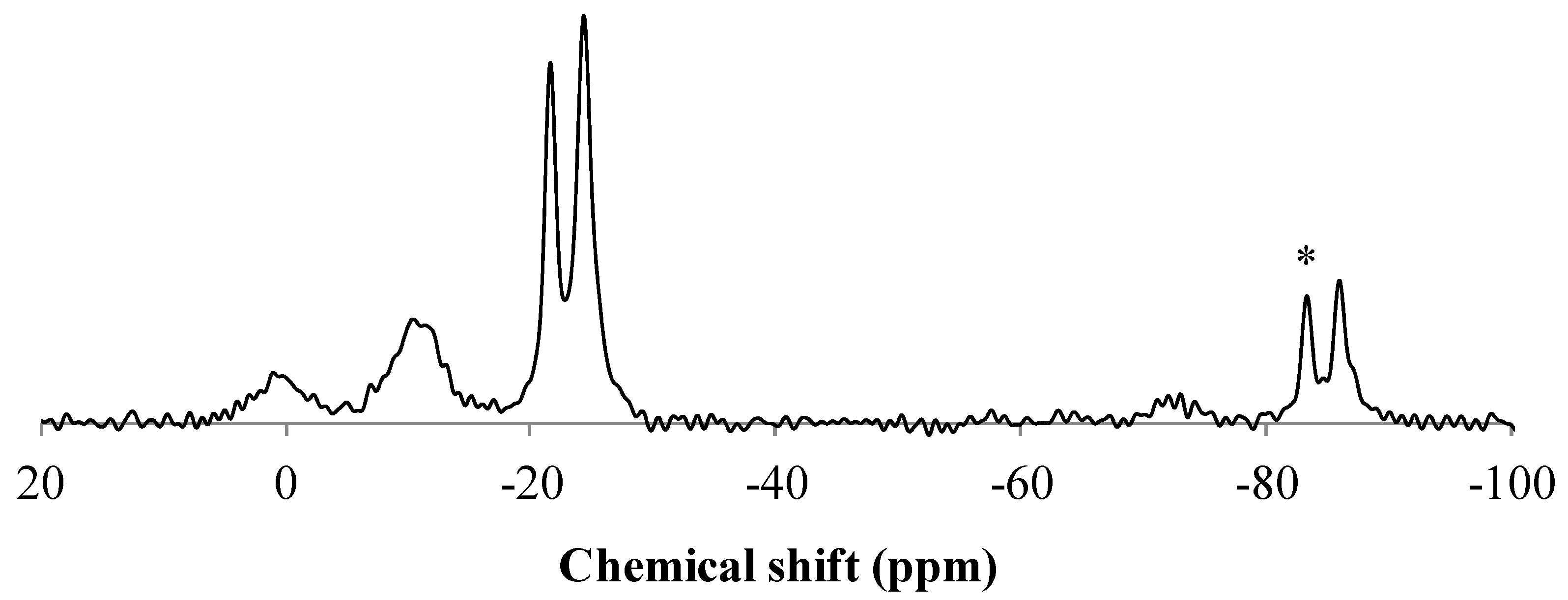

4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lewin, M.; Brozek, J.; Martens, M.M. The system polyamide/sulfamate/dipentaerythritol: Flame retardancy and chemical reactions. Polym. Adv. Technol. 2002, 13, 1091–1102. [Google Scholar] [CrossRef]
- Dahiya, J.B.; Kandola, B.K.; Sitpalan, A.; Horrocks, A.R. Effects of nanoparticles on the flame retardancy of the ammonium sulphamate-dipentaerythritol flame-retardant system in polyamide 6. Polym. Adv. Technol. 2013, 24, 398–406. [Google Scholar] [CrossRef]
- Coquelle, M.; Duquesne, S.; Casetta, M.; Sun, J.; Zhang, S.; Bourbigot, S. Investigation of the decomposition pathway of polyamide 6/ammonium sulfamate fibers. Polym. Degrad. Stab. 2014, 106, 150–157. [Google Scholar] [CrossRef]
- Sasaki, Y.; Suzuki, Y.; Fujimoto, T.; Miki, K. Jpn. patent 53133257. Chem. Abstr. 1978, 90, 105016w. [Google Scholar]
- Yang, H.H. Polyamide fibers. In Handbook of Fiber Chemistry, 3rd ed.; Lewin, M., Ed.; CRC Press: Boca Raton, FL, USA, 2006; Volume 8. [Google Scholar]
- Levchik, S.V.; Costa, L.; Camino, G. Effect of the fire-retardant, ammonium polyphosphate, on the thermal decomposition of aliphatic polyamides: Part II—Polyamide 6. Polym. Degrad. Stab. 1992, 36, 229–237. [Google Scholar] [CrossRef]
- Levchik, S.V.; Weil, E.D. Combustion and fire retardancy of aliphatic nylons. Polym. Int. 2000, 49, 1033–1073. [Google Scholar] [CrossRef]
- Tests for Flammability of Plastic Materials for Parts in Devices and Appliances; UL 94; Underwriters Laboratories Inc. (UL): Northbook, IL, USA, 2001.
- Standard Test Method for Determining Flammability Characteristics of Plastics and Other Solid Materials Using Microscale Combustion Calorimetry; ASTM D7309-07a; ASTM International: West Conshohocken, PA, USA, 2007.
- International Organization for Standardization. Plastics—Simple Heat Release Test Using a Conical Radiant Heater and a Thermopile Detector; ISO 13927:2001; International Organization for Standardization: Geneva, Switzerland, 2001. [Google Scholar]
- International Organization for Standardization. Plastics—Determination of Burning Behaviour by Oxygen Index—Part 2: Ambient-Temperature Test; ISO 4589–2:1996; International Organization for Standardization: Geneva, Switzerland, 1996. [Google Scholar]
- Samyn, F.; Bourbigot, S.; Jama, C.; Bellayer, S. Fire retardancy of polymer clay nanocomposites: Is there an influence of the nanomorphology? Polym. Degrad. Stab. 2008, 93, 2019–2024. [Google Scholar] [CrossRef]
- Schartel, B.; Pawlowski, K.H.; Lyon, R.E. Pyrolysis combustion flow calorimeter: A tool to assess flame retarded PC/ABS materials? Thermochim. Acta 2007, 462, 1–14. [Google Scholar] [CrossRef]
- Morgan, A.B. Cone calorimeter and pyrolysis combustion flow calorimeter testing of polyurethane foams—A call for collaboration to establish a predictive model. In Proceedings of the 23rd Annual Conference on Recent Advances in Flame Retardancy of Polymeric Materials 2012, Stamford, CT, USA, 21–23 May 2012; pp. 254–266.
- Lewin, M.; Zhang, J.; Pearce, E.; Gilman, J. Flammability of polyamide 6 using the sulfamate system and organo-layered silicate. Polym. Adv. Technol. 2007, 18, 737–745. [Google Scholar] [CrossRef]
- Schartel, B.; Pötschke, P.; Knoll, U.; Abdel-Goad, M. Fire behaviour of polyamide 6/multiwall carbon nanotube nanocomposites. Eur. Polym. J. 2005, 41, 1061–1070. [Google Scholar] [CrossRef]
- Samyn, F. Compréhension des procédés d'ignifugation du polyamide 6; Université Lille I: Lille, Nord, France, 2007. (In French) [Google Scholar]
- Levchik, S.V.; Balabanovich, A.I.; Levchik, G.F.; Costa, L. Effect of melamine and its salts on combustion and thermal decomposition of polyamide 6. Fire Mater. 1997, 21, 75–83. [Google Scholar] [CrossRef]
- Kandola, B.K.; Price, D.; Milnes, G.J.; da Silva, A. Development of a novel experimental technique for quantitative study of melt dripping of themoplastic polymers. Polym. Degrad. Stab. 2013, 98, 52–63. [Google Scholar] [CrossRef]
- Dupretz, R.; Fontaine, G.; Duquesne, S.; Bourbigot, S. Understanding of phenomena through instrumentation of UL-94 test. In Proceedings of Journée des Jeunes Chercheurs de l’UGéPE, Mons, Belgium, 23 October 2014.
- Levchik, S.V.; Weil, E.D.; Lewin, M. Thermal decomposition of aliphatic nylons. Polym. Int. 1999, 48, 532–557. [Google Scholar] [CrossRef]
- Stein, S.E. Infrared Spectra. In NIST Chemistry WebBook; NIST Standard Reference Database No. 69; Linstrom, P.J., Mallard, W.G., Eds.; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2001. [Google Scholar]
- Seefeldt, H.; Duemichen, E.; Braun, U. Flame retardancy of glass fiber reinforced high temperature polyamide by use of aluminum diethylphosphinate: Thermal and thermo-oxidative effects. Polym. Int. 2013, 62, 1608–1616. [Google Scholar]
- Pretsch, E.; Bühlmann, P.; Badertscher, M. IR Spectroscopy. In Structure Determination of Organic Compounds: Tables of Spectral Data, 4th ed.; Springer: Berlin, Germany, 2009. [Google Scholar]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies: Tables and Charts, 3rd ed.; Wiley: Hoboken, NJ, USA, 2004. [Google Scholar]
- Fuller, M.P.; Griffiths, P.R. Infrared microsampling by diffuse reflectance fourier transform spectrometry. Appl. Spectrosc. 1980, 34, 533–539. [Google Scholar] [CrossRef]
- Pramoda, K.P.; Liu, T.; Liu, Z.; He, C.; Sue, H.J. Thermal degradation behavior of polyamide 6/clay nanocomposites. Polym. Degrad. Stab. 2003, 81, 47–56. [Google Scholar] [CrossRef]
- Davis, R.D.; Gilman, J.W.; VanderHart, D.L. Processing degradation of polyamide 6/montmorillonite clay nanocomposites and clay organic modifier. Polym. Degrad. Stab. 2003, 79, 111–121. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, Q. Reaction of melamine phosphate with pentaerythritol and its products for flame retardation of polypropylene. Polym. Adv. Technol. 2007, 18, 587–600. [Google Scholar] [CrossRef]
- Bourbigot, S.; Bras, M.L.; Delobel, R. Carbonization mechanisms resulting from intumescence association with the ammonium polyphosphate-pentaerythritol fire retardant system. Carbon 1993, 31, 1219–1230. [Google Scholar] [CrossRef]
- Duquesne, S.; Futterer, T. Intumescent Systems. In The Non-halogenated Flame Retardant Handbook; Morgan, A.B., Wilkie, C.A., Eds.; Wiley: Hoboken, NJ, USA, 2014; pp. 293–346. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coquelle, M.; Duquesne, S.; Casetta, M.; Sun, J.; Gu, X.; Zhang, S.; Bourbigot, S. Flame Retardancy of PA6 Using a Guanidine Sulfamate/Melamine Polyphosphate Mixture. Polymers 2015, 7, 316-332. https://doi.org/10.3390/polym7020316
Coquelle M, Duquesne S, Casetta M, Sun J, Gu X, Zhang S, Bourbigot S. Flame Retardancy of PA6 Using a Guanidine Sulfamate/Melamine Polyphosphate Mixture. Polymers. 2015; 7(2):316-332. https://doi.org/10.3390/polym7020316
Chicago/Turabian StyleCoquelle, Mathieu, Sophie Duquesne, Mathilde Casetta, Jun Sun, Xiaoyu Gu, Sheng Zhang, and Serge Bourbigot. 2015. "Flame Retardancy of PA6 Using a Guanidine Sulfamate/Melamine Polyphosphate Mixture" Polymers 7, no. 2: 316-332. https://doi.org/10.3390/polym7020316
APA StyleCoquelle, M., Duquesne, S., Casetta, M., Sun, J., Gu, X., Zhang, S., & Bourbigot, S. (2015). Flame Retardancy of PA6 Using a Guanidine Sulfamate/Melamine Polyphosphate Mixture. Polymers, 7(2), 316-332. https://doi.org/10.3390/polym7020316